Cocoa Bean Shell as Promising Feedstock for the Production of Poly(3-hydroxybutyrate) (PHB)
Abstract
1. Introduction
2. Materials and Methods
2.1. Cocoa Bean Shell
2.2. Hydrothermal Hydrolysis
- -
- Non-centrifuged medium: the content of the autoclaved bottles was adjusted to pH 6–7 with 5 M NaOH (VWR) and then, it was placed in a flask for subsequent inoculation.
- -
- Centrifuged medium: solids were removed from the hydrolysate mixture with a sieve and the liquid phase was centrifuged for 10 min at 10,000 rpm and, after that, the pH of the supernatant was adjusted to 6–7 with 5 M NaOH (VWR). Finally, the broth was placed in a fermentation flask for inoculation.
2.3. Fermentation Process
2.4. Morphological Analysis
2.5. Analytical Methods
2.5.1. Determination of Dry Extract
2.5.2. Determination of Total Carbohydrates: Phenol–Sulfuric Acid Method
2.5.3. Determination of Reducing Sugars: Dinitrosalicylic Acid (DNS) Method
2.5.4. Determination of PHB
2.5.5. Determination of Fermentation Inhibitors
2.5.6. Elemental Analysis
2.5.7. Determination of Total Suspended Solids
3. Results and Discussion
3.1. Characterization of Hydrolysed Broths and CBS Solids
3.2. Fermentation with B. firmus
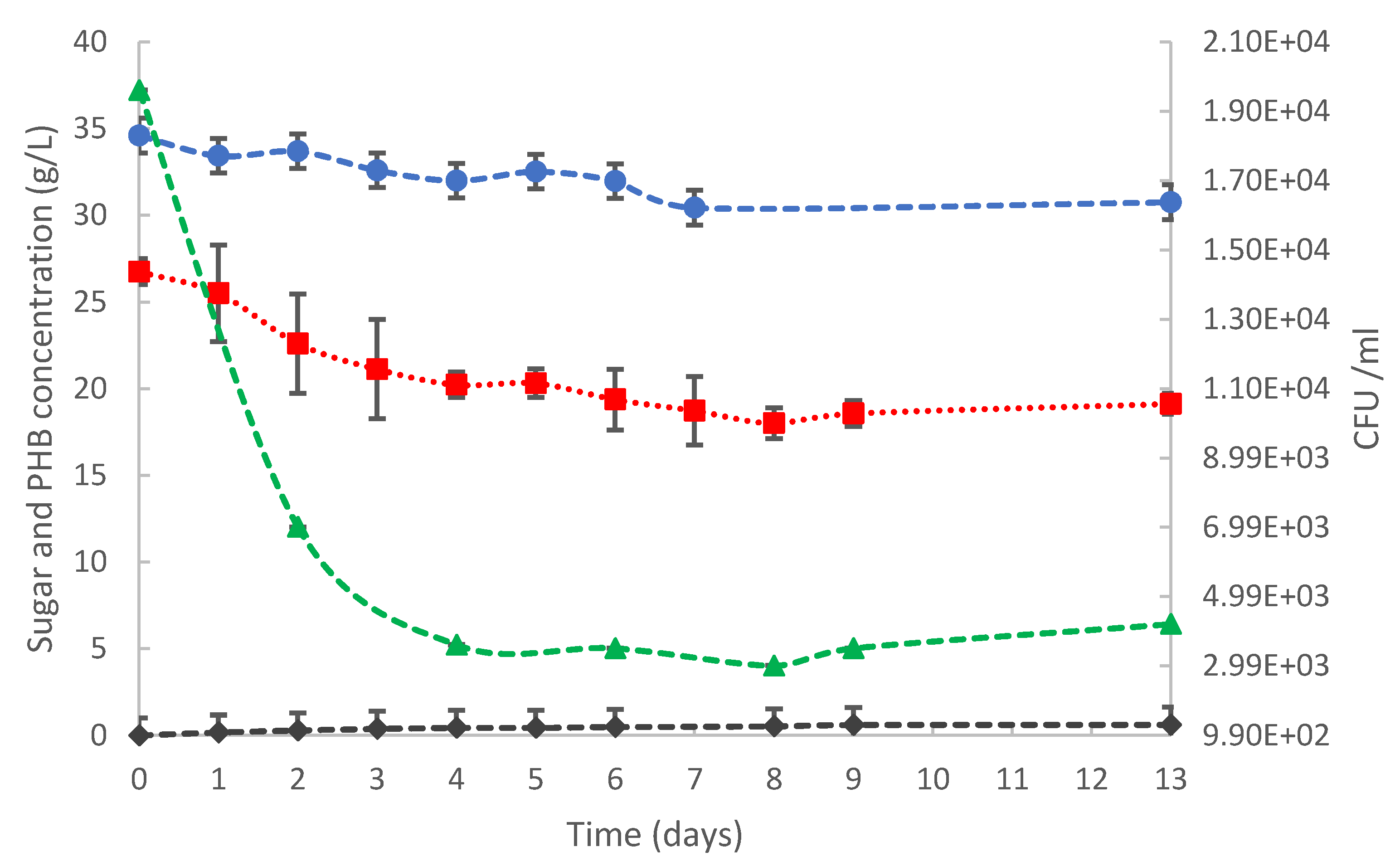
3.2.1. Centrifuged Medium
3.2.2. Non-Centrifuged Medium
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Pavlović, N.; Jokić, S.; Jakovljević, M.; Blažić, M.; Molnar, M. Green Extraction Methods for Active Compounds from Food Waste—Cocoa Bean Shell. Foods 2020, 9, 140. [Google Scholar] [CrossRef]
- Mariatti, F.; Gunjević, V.; Boffa, L.; Cravotto, G. Process Intensification Technologies for the Recovery of Valuable Compounds from Cocoa By-Products. Innov. Food Sci. Emerg. Technol. 2021, 68, 102601. [Google Scholar] [CrossRef]
- Sánchez, M.; Laca, A.; Laca, A.; Díaz, M. Towards Food Circular Economy: Hydrothermal Treatment of Mixed Vegetable and Fruit Wastes to Obtain Fermentable Sugars and Bioactive Compounds. Environ. Sci. Pollut. Res. 2022. [Google Scholar] [CrossRef] [PubMed]
- Rajput, S.; Singh, S.P. Connecting Circular Economy and Industry 4.0. Int. J. Inf. Manag. 2019, 49, 98–113. [Google Scholar] [CrossRef]
- Acosta, N.; De Vrieze, J.; Sandoval, V.; Sinche, D.; Wierinck, I.; Rabaey, K. Cocoa Residues as Viable Biomass for Renewable Energy Production through Anaerobic Digestion. Bioresour. Technol. 2018, 265, 568–572. [Google Scholar] [CrossRef] [PubMed]
- Rojo-poveda, O.; Barbosa-pereira, L.; Zeppa, G.; Stévigny, C. Cocoa Bean Shell—A By-Product with Nutritional Properties and Biofunctional Potential. Nutrients 2020, 12, 1123. [Google Scholar] [CrossRef]
- Ho, V.T.T.; Zhao, J.; Fleet, G. Yeasts Are Essential for Cocoa Bean Fermentation. Int. J. Food Microbiol. 2014, 174, 72–87. [Google Scholar] [CrossRef]
- Vásquez, Z.S.; de Carvalho Neto, D.P.; Pereira, G.V.M.; Vandenberghe, L.P.S.; de Oliveira, P.Z.; Tiburcio, P.B.; Rogez, H.L.G.; Góes Neto, A.; Soccol, C.R. Biotechnological Approaches for Cocoa Waste Management: A Review. Waste Manag. 2019, 90, 72–83. [Google Scholar] [CrossRef]
- Okiyama, D.C.G.; Navarro, S.L.B.; Rodrigues, C.E.C. Cocoa Shell and Its Compounds: Applications in the Food Industry. Trends Food Sci. Technol. 2017, 63, 103–112. [Google Scholar] [CrossRef]
- Briassoulis, D.; Tserotas, P.; Athanasoulia, I. Alternative Optimization Routes for Improving the Performance of Poly (3-Hydroxybutyrate) (PHB) Based Plastics. J. Clean. Prod. 2021, 318, 128555. [Google Scholar] [CrossRef]
- Sabbagh, F.; Muhamad, I.I. Production of Poly-Hydroxyalkanoate as Secondary Metabolite with Main Focus on Sustainable Energy. Renew. Sustain. Energy Rev. 2017, 72, 95–104. [Google Scholar] [CrossRef]
- Luef, K.P.; Stelzer, F.; Wiesbrock, F. Poly(Hydroxy Alkanoate)s in Medical Applications. Chem. Biochem. Eng. Q. 2015, 29, 287–297. [Google Scholar] [CrossRef] [PubMed]
- Murthy, G.S.; Kumar, D.; Strauss, S.; Dalton, D.; Vionocur, J. Feasibility Analysis of Poly-β-Hydroxybutyrate (PHB) Extraction from Hybrid Poplar Leaves. Am. Soc. Agric. Biol. Eng. 2010, 6, 4461–4471. [Google Scholar]
- Balakrishna Pillai, A.; Jaya Kumar, A.; Kumarapillai, H. Enhanced Production of Poly(3-Hydroxybutyrate) in Recombinant Escherichia Coli and EDTA–Microwave-Assisted Cell Lysis for Polymer Recovery. AMB Express 2018, 8, 142. [Google Scholar] [CrossRef] [PubMed]
- Sindhu, R.; Silviya, N.; Binod, P.; Pandey, A. Pentose-Rich Hydrolysate from Acid Pretreated Rice Straw as a Carbon Source for the Production of Poly-3-Hydroxybutyrate. Biochem. Eng. J. 2013, 78, 67–72. [Google Scholar] [CrossRef]
- Salehizadeh, H.; Van Loosdrecht, M.C.M. Production of Polyhydroxyalkanoates by Mixed Culture: Recent Trends and Biotechnological Importance. Biotechnol. Adv. 2004, 22, 261–279. [Google Scholar] [CrossRef]
- Mohanrasu, K.; Premnath, N.; Siva Prakash, G.; Sudhakar, M.; Boobalan, T.; Arun, A. Exploring Multi Potential Uses of Marine Bacteria; an Integrated Approach for PHB Production, PAHs and Polyethylene Biodegradation. J. Photochem. Photobiol. B Biol. 2018, 185, 55–65. [Google Scholar] [CrossRef]
- Poltronieri, P.; Mezzolla, V.; D’Urso, O.F. PHB Production in Biofermentors Assisted through Biosensor Applications. Proceedings 2016, 1, 4. [Google Scholar] [CrossRef]
- Dhingra, H.K.; Priya, K. Physiological and Molecular Identification of Polyhydroxybutyrates (PHB) Producing Micro-Organisms Isolated from Root Nodules of Leguminous Plants. Afr. J. Microbiol. Res. 2013, 7, 3961–3967. [Google Scholar] [CrossRef]
- Mohapatra, S.; Maity, S.; Dash, H.R.; Das, S.; Pattnaik, S.; Rath, C.C.; Samantaray, D. Bacillus and Biopolymer: Prospects and Challenges. Biochem. Biophys. Rep. 2017, 12, 206–213. [Google Scholar] [CrossRef]
- Dubois, M.; Gilles, K.; Hamilton, J.K.; Rebers, P.A.; Smith, F. A Colorimetric Method for the Determination of Sugars. Anal. Chem. 1956, 28, 350–356. [Google Scholar] [CrossRef]
- Miller, G.L. Use of Dinitrosalicylic Acid Reagent for Determination of Reducing Sugar. Anal. Chem. 1959, 31, 426–428. [Google Scholar] [CrossRef]
- Díaz, A.I.; Laca, A.; Laca, A.; Díaz, M. Treatment of Supermarket Vegetable Wastes to Be Used as Alternative Substrates in Bioprocesses. Waste Manag. 2017, 67, 59–66. [Google Scholar] [CrossRef] [PubMed]
- Law, J.H.; Slepecky, R.A. Ralph a. Slepecky. J. Bacteriol. Res. 1960, 82, 546–558. [Google Scholar]
- Pola, L.; Fernández-García, L.; Collado, S.; Oulego, P.; Díaz, M. Macronutrient Solubilisation during Hydrothermal Treatment of Sewage Sludge. J. Water Process. Eng. 2021, 43, 102270. [Google Scholar] [CrossRef]
- Sukruansuwan, V.; Napathorn, S.C. Use of Agro-Industrial Residue from the Canned Pineapple Industry for Polyhydroxybutyrate Production by Cupriavidus Necator Strain A-04. Biotechnol. Biofuels 2018, 11, 202. [Google Scholar] [CrossRef]
- Yustinah; Hidayat, N.; Alamsyah, R.; Roslan, A.M.; Hermansyah, H.; Gozan, M. Production of Polyhydroxybutyrate from Oil Palm Empty Fruit Bunch (OPEFB) Hydrolysates by Bacillus Cereus Suaeda B-001. Biocatal. Agric. Biotechnol. 2019, 18, 101019. [Google Scholar] [CrossRef]
- Naranjo, J.M.; Posada, J.A.; Higuita, J.C.; Cardona, C.A. Valorization of Glycerol through the Production of Biopolymers: The PHB Case Using Bacillus Megaterium. Bioresour. Technol. 2013, 133, 38–44. [Google Scholar] [CrossRef]
- Hamdy, S.M.; Danial, A.W.; Gad El-Rab, S.M.F.; Shoreit, A.A.M.; Hesham, A.E.L. Production and Optimization of Bioplastic (Polyhydroxybutyrate) from Bacillus Cereus Strain SH-02 Using Response Surface Methodology. BMC Microbiol. 2022, 22, 183. [Google Scholar] [CrossRef]
- Trakunjae, C.; Boondaeng, A.; Apiwatanapiwat, W.; Kosugi, A.; Arai, T.; Sudesh, K.; Vaithanomsat, P. Enhanced Polyhydroxybutyrate (PHB) Production by Newly Isolated Rare Actinomycetes Rhodococcus Sp. Strain BSRT1-1 Using Response Surface Methodology. Sci. Rep. 2021, 11, 1896. [Google Scholar] [CrossRef]
- Oliveira-Filho, E.R.; Silva, J.G.P.; de Macedo, M.A.; Taciro, M.K.; Gomez, J.G.C.; Silva, L.F. Investigating Nutrient Limitation Role on Improvement of Growth and Poly(3-Hydroxybutyrate) Accumulation by Burkholderia Sacchari LMG 19450 From Xylose as the Sole Carbon Source. Front. Bioeng. Biotechnol. 2020, 7, 416. [Google Scholar] [CrossRef] [PubMed]
- Sánchez, M.; Laca, A.; Laca, A.; Díaz, M. Value-added Products from Fruit and Vegetable Wastes: A Review. CLEAN Soil Air Water 2021, 49, 2000376. [Google Scholar] [CrossRef]
- van der Maas, L.; Driessen, J.L.S.P.; Mussatto, S.I. Effects of Inhibitory Compounds Present in Lignocellulosic Biomass Hydrolysates on the Growth of Bacillus Subtilis. Energies 2021, 14, 8419. [Google Scholar] [CrossRef]
- Abdel-Rahman, M.A.; Hassan, S.E.D.; Fouda, A.; Radwan, A.A.; Barghoth, M.G.; Desouky, S.G. Evaluating the Effect of Lignocellulose-Derived Microbial Inhibitors on the Growth and Lactic Acid Production by Bacillus Coagulans Azu-10. Fermentation 2021, 7, 17. [Google Scholar] [CrossRef]
- Martínez-Herrera, R.E.; Alemán-Huerta, M.E.; Almaguer-Cantú, V.; Rosas-Flores, W.; Martínez-Gómez, V.J.; Quintero-Zapata, I.; Rivera, G.; Rutiaga-Quiñones, O.M. Efficient Recovery of Thermostable Polyhydroxybutyrate (PHB) by a Rapid and Solvent-Free Extraction Protocol Assisted by Ultrasound. Int. J. Biol. Macromol. 2020, 164, 771–782. [Google Scholar] [CrossRef]
- Kovalcik, A.; Pernicova, I.; Obruca, S.; Szotkowski, M.; Enev, V.; Kalina, M.; Marova, I. Grape Winery Waste as a Promising Feedstock for the Production of Polyhydroxyalkanoates and Other Value-Added Products. Food Bioprod. Process. 2020, 124, 1–10. [Google Scholar] [CrossRef]
- Mahboubi, A.; Elyasi, S.; Doyen, W.; De Wever, H.; Taherzadeh, M.J. Concentration-Driven Reverse Membrane Bioreactor for the Fermentation of Highly Inhibitory Lignocellulosic Hydrolysate. Process. Biochem. 2020, 92, 409–416. [Google Scholar] [CrossRef]
- Muñoz-Páez, K.M.; Alvarado-Michi, E.L.; Buitrón, G.; Valdez-Vazquez, I. Distinct Effects of Furfural, Hydroxymethylfurfural and Its Mixtures on Dark Fermentation Hydrogen Production and Microbial Structure of a Mixed Culture. Int. J. Hydrogen Energy 2019, 44, 2289–2297. [Google Scholar] [CrossRef]
- Anli Dino, A.; Brindha, R.; Jayamuthunagai, J.; Bharathiraja, B. Biodegradation of Aniline from Textile Industry Waste Using Salt Tolerant Bacillus Firmus BA01. Eng. Agric. Environ. Food 2019, 12, 360–366. [Google Scholar] [CrossRef]
- Sarmiento-Vásquez, Z.; Vanderberghe, L.P.D.S.; Karp, S.G.; Soccol, C.R. Production of Polyhydroxyalkanoates through Soybean Hull and Waste Glycerol Valorization: Subsequent Alkaline Pretreatment and Enzymatic Hydrolysis. Fermentation 2022, 8, 433. [Google Scholar] [CrossRef]
- Khiyami, M.A.; Al-fadual, S.M.; Bahklia, A.H. Polyhydroxyalkanoates Production via Bacillus (PCS) Biofilm and Date Palm Syrup. J. Med. Plants Res. 2011, 5, 3312–3320. [Google Scholar]
- Manikandan, N.A.; Pakshirajan, K.; Pugazhenthi, G. A Closed-Loop Biorefinery Approach for Polyhydroxybutyrate (PHB) Production Using Sugars from Carob Pods as the Sole Raw Material and Downstream Processing Using the Co-Product Lignin. Bioresour. Technol. 2020, 307, 123247. [Google Scholar] [CrossRef]
- Valdez-Calderón, A.; Barraza-Salas, M.; Quezada-Cruz, M.; Islas-Ponce, M.A.; Angeles-Padilla, A.F.; Carrillo-Ibarra, S.; Rodriguez, M.; Rojas-Avelizapa, N.G.; Garrido-Hernández, A.; Rivas-Castillo, A.M. Production of Polyhydroxybutyrate (PHB) by a Novel Klebsiella Pneumoniae Strain Using Low-Cost Media from Fruit Peel Residues. Biomass Convers. Biorefinery 2022, 12, 4925–4938. [Google Scholar] [CrossRef]
- Saratale, R.G.; Saratale, G.D.; Cho, S.K.; Kim, D.S.; Ghodake, G.S.; Kadam, A.; Kumar, G.; Bharagava, R.N.; Banu, R.; Shin, H.S. Pretreatment of Kenaf (Hibiscus cannabinus L.) Biomass Feedstock for Polyhydroxybutyrate (PHB) Production and Characterization. Bioresour. Technol. 2019, 282, 75–80. [Google Scholar] [CrossRef] [PubMed]
- Florian, K.; Michael, M.; Back, W.; Kurz, T.; Krottenthaler, M. Effect of Hot Trub and Particle Addition on Fermentation Performance of Saccharomyces Cerevisiae. Enzym. Microb. Technol. 2007, 41, 711–720. [Google Scholar] [CrossRef]
- Bathgate, G.N. The Influence of Malt and Wort Processing on Spirit Character: The Lost Styles of Scotch Malt Whisky. J. Inst. Brew. 2019, 125, 200–213. [Google Scholar] [CrossRef]
- Liu, S.; Huang, J.; Zhang, C.; Wang, L.; Fan, C.; Zhong, C. Probing the Growth and Mechanical Properties of Bacillus Subtilis Biofilms through Genetic Mutation Strategies. Synth. Syst. Biotechnol. 2022, 7, 965–971. [Google Scholar] [CrossRef] [PubMed]
- Rath, S.; Palit, K.; Das, S. Variable PH and Subsequent Change in PCO2 Modulates the Biofilm Formation, Synthesis of Extracellular Polymeric Substances, and Survivability of a Marine Bacterium Bacillus Stercoris GST-03. Environ. Res. 2022, 214, 114128. [Google Scholar] [CrossRef] [PubMed]
- Fioresi, F.; Vieillard, J.; Bargougui, R.; Bouazizi, N.; Nkuigue, P.; Djoufac, E.; Brun, N.; Mofaddel, N.; Le, F. Chemical Modification of the Cocoa Shell Surface Using Diazonium Salts. J. Colloid Interface Sci. 2017, 494, 92–97. [Google Scholar] [CrossRef]
- Al-yousef, H.A.; Alotaibi, B.M.; Aouaini, F.; Bonilla-petriciolet, A. Adsorption of Ibuprofen on Cocoa Shell Biomass-Based Adsorbents: Interpretation of the Adsorption Equilibrium via Statistical Physics Theory. J. Mol. Liq. 2021, 331, 115697. [Google Scholar] [CrossRef]
- Balentić, J.P.; Ačkar, Đ.; Jokić, S.; Jozinović, A.; Babić, J.; Miličević, B.; Šubarić, D.; Pavlović, N. Cocoa Shell: A By-Product with Great Potential for Wide Application. Molecules 2018, 23, 1404. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Arellano, G.; Barajas-Fernández, J.; García-Alamilla, R.; Lagunes-Gálvez, L.; Lara-Rivera, A.H.; García-Alamilla, P. Evaluation of Cocoa Beans Shell Powder as a Bioadsorbent of Congo Red Dye Aqueous Solutions. Materials 2021, 14, 2763. [Google Scholar] [CrossRef] [PubMed]



| Centrifuged Medium | Non-Centrifuged Medium | |
|---|---|---|
| Soluble total sugars | 34.5 ± 0.05 | 35.9 ± 0.6 |
| Soluble reducing sugars | 26.8 ± 0.02 | 31.5 ± 0.7 |
| Soluble total nitrogen | 2.21 | 2.14 |
| Soluble total phosphorus | 0.45 | 0.64 |
| pH | 6.8 | 6.8 |
| Total suspended solids | ~0 | 152 ± 0.03 |
| Broth | k1 (Days−1) | k2 (Days−1) | k3 (Days−1) |
|---|---|---|---|
| Non-centrifuged | 0.072 | 1.223 | 0.4213 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sánchez, M.; Laca, A.; Laca, A.; Díaz, M. Cocoa Bean Shell as Promising Feedstock for the Production of Poly(3-hydroxybutyrate) (PHB). Appl. Sci. 2023, 13, 975. https://doi.org/10.3390/app13020975
Sánchez M, Laca A, Laca A, Díaz M. Cocoa Bean Shell as Promising Feedstock for the Production of Poly(3-hydroxybutyrate) (PHB). Applied Sciences. 2023; 13(2):975. https://doi.org/10.3390/app13020975
Chicago/Turabian StyleSánchez, Marta, Amanda Laca, Adriana Laca, and Mario Díaz. 2023. "Cocoa Bean Shell as Promising Feedstock for the Production of Poly(3-hydroxybutyrate) (PHB)" Applied Sciences 13, no. 2: 975. https://doi.org/10.3390/app13020975
APA StyleSánchez, M., Laca, A., Laca, A., & Díaz, M. (2023). Cocoa Bean Shell as Promising Feedstock for the Production of Poly(3-hydroxybutyrate) (PHB). Applied Sciences, 13(2), 975. https://doi.org/10.3390/app13020975







