Toxicity of Different Chemical Components in Sun Cream Filters and Their Impact on Human Health: A Review
Abstract
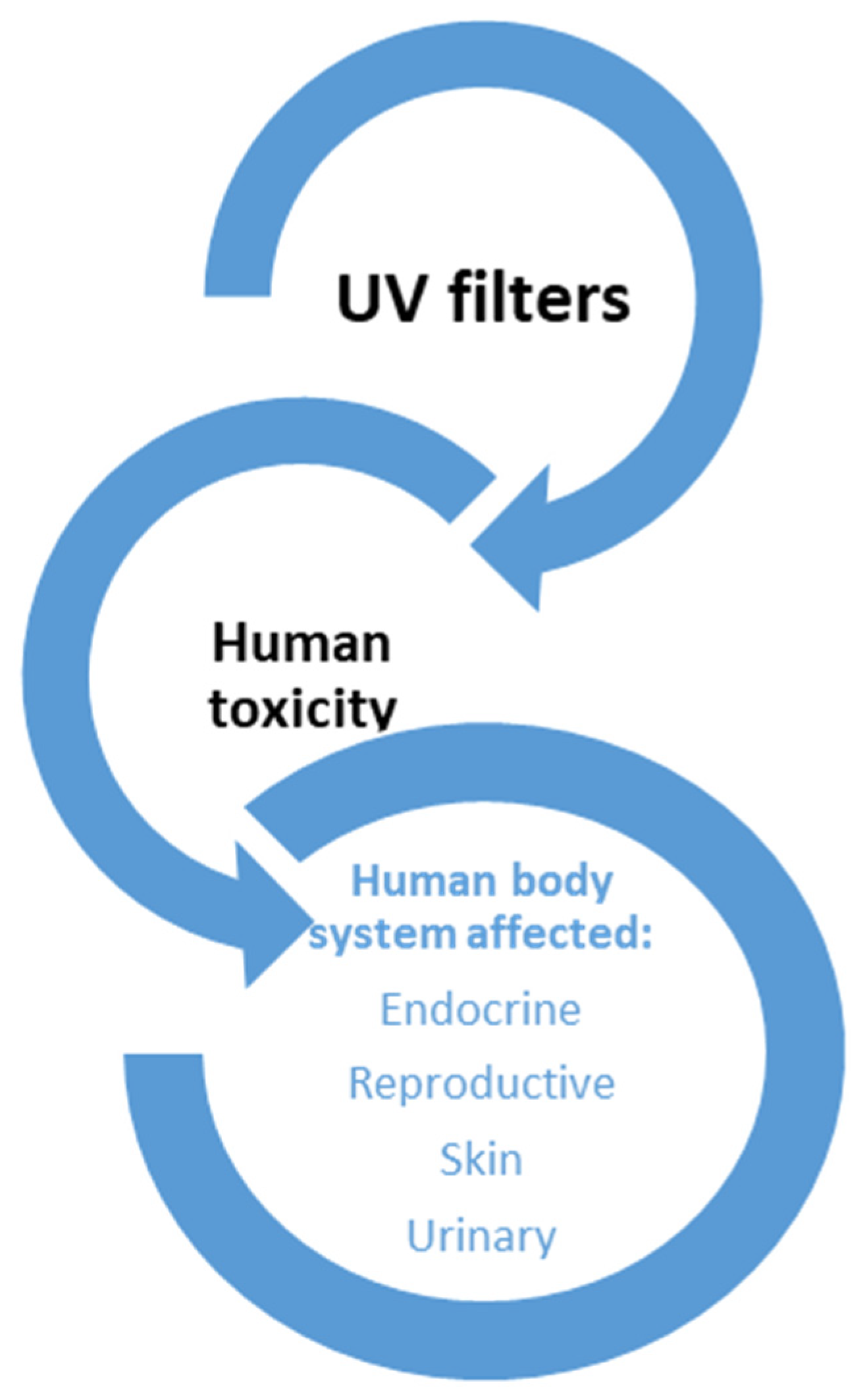
:1. Introduction
2. Materials and Methods
3. Results
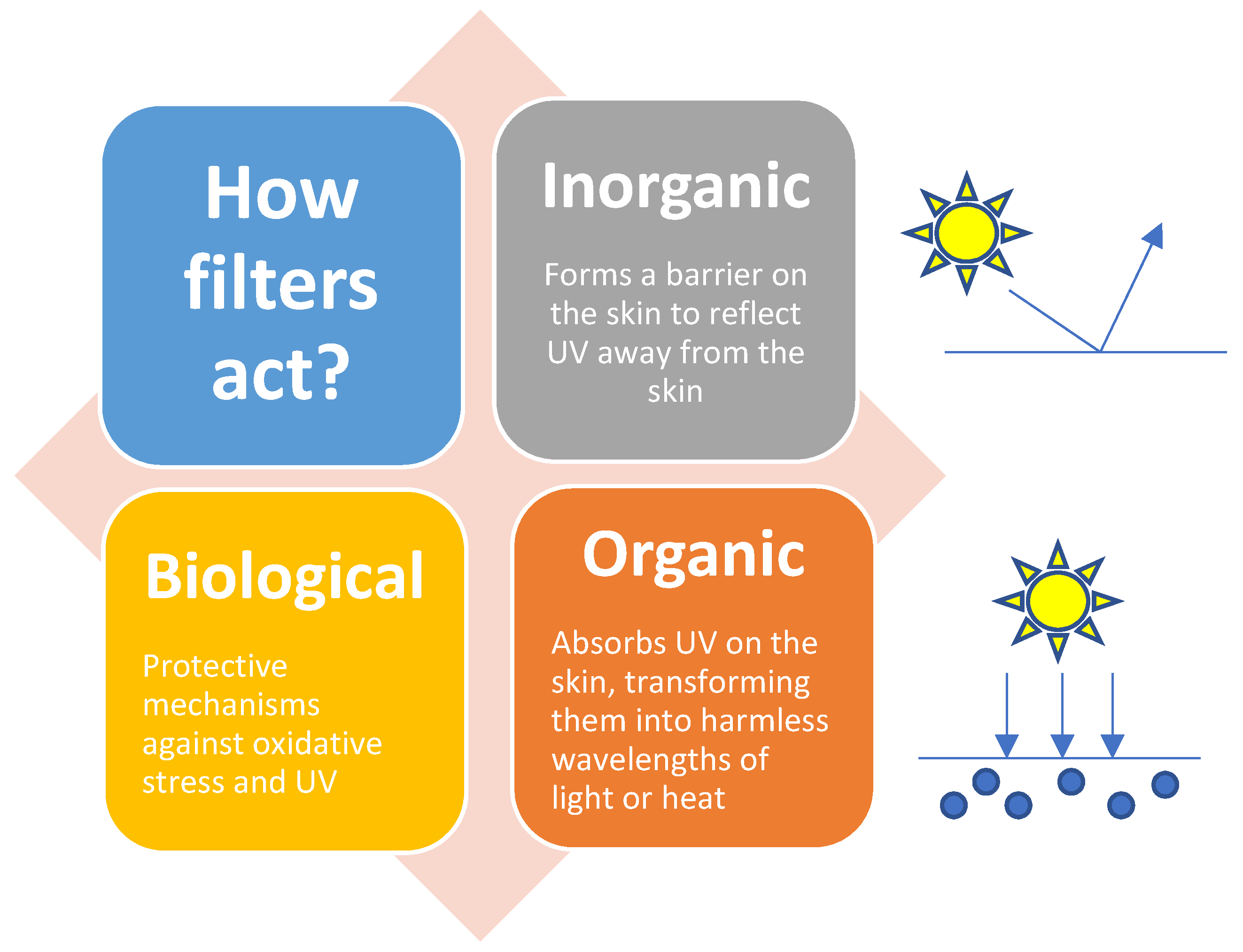
3.1. Organic Filters
3.1.1. UVB Filters
- PABA and derivates:
- -
- Octyldimethyl PABA or Padimate O: EDC (estrogenic), liberator of free radicals, allergen reactions, ecotoxixin and environmental pollutant. Always avoid (EWG sec 5 fair)
- -
- Ethylhexyl triazone (octyl triazone): Not authorized in the USA. Increases the effectiveness of other filters. Free radical released in sunlight. Very little data. Prudence (EWG 1 None)
- Cinnamates:
- -
- Octinoxate (Octyl Methoxycinnamate, OMC, Ethylhexyl Methoxycinnamate, EHMC): confirmed EDC, persistent and bioaccumulative, is absorbed through the skin and can increase the absorption of other substances. It appears in breast milk. Ecotoxic. One of the most problematic filters today. Banned in Denmark in creams for children. It pollutes rivers and affects the populations of fish and other aquatic animals. It appears in tap water and even some bottled water. Always avoid (EWG 6 limited)
- -
- Cinoxato (2-Ethoxyethyl-P-Methoxycinnamate): In disuse. There is little data on its safety. Avoid as a precaution (EWG 3 limited)
- Salicylates: They are weaker; they increase the power of other UVB radiation absorbers. Better safety profile.
- -
- Octisalate (ethylhexyl salicylate, 2-Ethylhexyl 2-Hydroxybenzoate): It is quite safe, it is frequently used in association with Avobenzone to extend the duration of protection. Ecotoxin. Today, it is an acceptable chemical filter (EWG sec 3)
- -
- Homosalate (3,3,5-Trimethyl-Salicylate Cyclohexanol; 3,3,5-Trimethylcyclohexyl 2-Hydroxybenzoate): Highly polluting, weak endocrine disruptor, decomposes with light into oxidizing substances that are harmful to the skin. Increases the absorption of other substances. Avoid (EWG 4 limited)
- Octocrylene or octocrilene (CAS n. 6197-30-4) (2-ethylhexyl 2-cyano-3,3-diphenyl-2-propenoate; 2-ethylhexyl 2-cyano-3,3-diphenylacrylate or the 2-ethylhexyl ester of 2-cyano-3,3-diphenyl acrylic acid): It is an organic ultraviolet (UV) filter with an aromatic structure, which absorbs mainly UVB radiation and short UVA wavelengths [64]. It is used in sunscreens with other UV filters to provide an adequate sun protection factor (SPF) due to its UV radiation absorption properties [65]. According to regulation, octocrylene is authorized as a UV filter in cosmetic formulations at a maximum concentration of 10.0% in acid form in Europe (Annex VI/10). Octocrylene seems to have endocrine disrupting activity and may cause allergies and/or (photo)allergies. It is included in the list elaborated in 2019 by the European Commission that collected 14 ingredients with potential disrupting properties used in cosmetic products [66].
- Ensulizole: (2-Pheny 1H-benzimidazole 5-Sulfonic acid, PBSA): In contact with the sun, it produces free radicals that can damage DNA and potentially cause skin cancer. Avoid if possible (EWG 3 limited)
- 4-MBC (4-Methylbenzylidene Camphor) (1,7,7-Trimethyl-3- [(4-Methylphenyl) Methylene] Bicyclo [2.2.1] Heptan-2-On)): EDC, persistent and bioaccumulative. Not authorized in the US for sun creams. Possible thyroid toxicity. Important environmental contaminant, affects aquatic populations. Always avoid. (EWG 7 fair)
3.1.2. UVA Filters
- Benzophenones: benzophenone-3, (2-hydroxy-4-methoxyphenyl) phenyl- methanone; 2-benzoyl-5-methoxyphenol; 2-hydroxy-4-methoxybenzophenone; (2-hydroxy-4-methoxyphenyl) phenylmethanone; 4-methoxy-2-hydroxybenzophenone; advastab 45; ai3-23644; anuvex; b3; benzophenone. Widely used, although less and less since it is one of the worst chemical filters that exists, especially BP-3 or Oxybenzone, which has multiple endocrine disrupting effects, is bioaccumulative, persistent in the environment, is associated with photoallergic reactions, is absorbed in significant amounts through the skin, passes into breast milk. There is evidence of neurotoxicity and ecotoxicity affecting many aquatic species [15,68]. With the current data, it should be prohibited. The CDC found it in the bodies of 97% of Americans tested in one study. Always avoid. (EGW 8 fair)
- Anthranilates: They are weak UVB absorbers and mainly UVA absorbers, but less effective than benzophenones.
- Avobenzone (Butyl Methoxydibenzoylmethane, Parsol 1798) Today one of the safest chemical filters. Allergies rare. It degrades easily in sunlight, losing its effectiveness, although it can be stabilized with other safe substances such as Octisalate. Ecotoxin. The most recommended by the EWG. (EWG 2 limited)
- Mexoryl SX: Tetraphthalidine sulfonic acid dialcamphor: Broad UVA absorber with efficacy similar to avobenzone. It is also considered safe. (EWG sec 2 limited)
3.1.3. Broad Spectrum Filters (UVA and UVB)
- Tinosorb M (Methylene bis-benzotriazolyl tetramethylbutylphenol) is not absorbed, is photostable, and there are few studies on it. Possible environmental contaminant. Not allowed in the USA. Acceptable filter but caution due to lack of studies. (EWG 1 limited).
- Tinosorb S (Bis-Ethylhexyloxyphenol methoxyphenyl triazine: Photostable and non-estrogenic. Few studies on toxicity. Acceptable but caution due to lack of studies. (EWG 0 limited). Not allowed in the USA.
- Iscotrizinol (Uvasorb HEB, diethylhexyl butamido triazone) No data, acceptable/caution. (EWG 2 limited).
3.2. Inorganic Filters (Minerals)
3.3. Biological Sunscreens
- Lignin: this compound acts as UV blocking agent, with antioxidant properties due to its ability to capture free radicals [83].
- Silymarin: native to the Silybum marianum, itis known for its antioxidant properties. Silymarin and its flavonolignans are useful agents that may protect the skin against the adverse impacts of solar radiation [84].
- Plants: recently, many other plant-derived extracts have been used as UV blocking agents:
- -
- Sphaeranthus indicus (SI) Linn (Asteraceae): rich in phenols, flavonoids, and mushroom tyrosinase [87].
- -
- Elaeagnus angustifolia (E. angustifolia): leaf extracts from this plant have been used to develop a topical sunscreen formulation [88].
- -
- Moringa oleifera: their extracts are rich in polyphenols such as quercetin, rutin, chlorogenic acid, ellagic acid, and ferulic acid that can be used in sunscreens [89].
- -
- Helianthus annuus: its seed oil belongs to the linoleic acid and oleic acid category of oils [90]. The alkyl polyglucoside (APG) emulsifier exhibits good emulsifying properties with a good SPF.
- -
- Cistus incanus L. and Cistus ladanifer L.: its components in their extracts have abundant polyphenolic that are beneficial sources of sunscreen and preserve the skin from UVR-mediated oxidative damage [91].
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Anastas, P.T.; Warner, J.C. Green Chemistry: Theory and Practice; Oxford University Press: Oxford, UK, 1998; p. 148. [Google Scholar]
- Bopp, S.K.; Kienzler, A.; Richarz, A.N.; van der Linden, S.C.; Paini, A.; Parissis, N.; Worth, A.P. Regulatory Assessment and Risk Management of Chemical Mixtures: Challenges and Ways Forward. Crit. Rev. Toxicol. 2019, 49, 174–189. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Berardesca, E.; Zuberbier, T.; Sanchez Viera, M.; Marinovich, M. Review of the safety of octocrylene used as an ultraviolet filter in cosmetics. European Academy of Dermatology and Venereology. J. Eur. Acad. Dermatol. Venereol. 2019, 33 (Suppl. S7), 25–33. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vom Saal, F.S.; Woodruff, T.J.; Soto, A.M.; Skakkebaek, N.E.; Gore, A.C.; Doan, L.L.; Brown, T.R.; Zoeller, R.T. Endocrine-Disrupting Chemicals and Public Health Protection: A Statement of Principles from The Endocrine Society. Endocrinology 2012, 153, 4097–4110. [Google Scholar] [CrossRef]
- WHO. State of the Science of Endocrine Disrupting Chemicals 2012; WHO Press: Geneva, Switzerland, 2013. [Google Scholar]
- Bokobza, E.; Hinault, C.; Tiroille, V.; Clavel, S.; Bost, F.; Chevalier, N. The Adipose Tissue at the Crosstalk Between EDCs and Cancer Development. Front. Endocrinol. 2021, 12, 691658. [Google Scholar] [CrossRef]
- Komarowska, M.D.; Grubczak, K.; Czerniecki, J.; Hermanowicz, A.; Hermanowicz, J.M.; Debek, W.; Matuszczak, E. Identification of the Bisphenol A (BPA) and the Two Analogues BPS and BPF in Cryptorchidism. Front. Endocrinol. 2021, 12, 694669. [Google Scholar] [CrossRef]
- Rodprasert, W.; Toppari, J.; Virtanen, H.E. Endocrine Disrupting Chemicals and Reproductive Health in Boys and Men. Front. Endocrinol. 2021, 12, 706532. [Google Scholar] [CrossRef]
- Zhang, Y.; Lu, Y.; Ma, H.; Xu, Q.; Wu, X. Combined Exposure to Multiple Endocrine Disruptors and Uterine Leiomyomata and Endometriosis in US Women. Front. Endocrinol. 2021, 12, 726876. [Google Scholar] [CrossRef]
- Delfosse, V.; Maire Al Balaguer, P.; Bourguet, W. A Structural Perspective on Nuclear Receptors as Targets of Environmental Compounds. Acta Pharmacol. Sin. 2014, 36, 88–101. [Google Scholar] [CrossRef] [Green Version]
- Fransway, A.F.; Fransway, P.J.; Belsito, D.V.; Yiannias, J.A. Paraben Toxicology. Dermatitis 2019, 30, 32–45. [Google Scholar] [CrossRef]
- Kockler, J.; Oelgemöller, M.; Robertson, S.; Glass, B.D. Photostability of sunscreens. J. Photochem. Photobiol. C Photochem. Rev. 2012, 13, 91–110. [Google Scholar] [CrossRef]
- Valle-Sistac, J.; Molins-Delgado, D.; Díaz, M.; Ibáñez, L.; Barceló, D.; Silvia Díaz-Cruz, M. Determination of parabens and benzophenone-type UV filters in human placenta: First description of the existence of benzyl paraben and benzophenone-4. Environ. Int. 2016, 88, 243–249. [Google Scholar] [CrossRef] [PubMed]
- Rehfeld, A.; Egeberg, D.L.; Almstrup, K.; Petersen, J.H.; Dissing, S.; Skakkebæk, N.E. EDC IMPACT: Chemical UV filters can affect human sperm function in a progesterone-like manner. Endocr. Connect. 2018, 7, 16–25. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- López-Hera, D. Medicina de Familia en la Red. Cremas Para el Sol Seguras: Como Elegir Fotoprotectores Solares No Tóxicos y Respetuosos Con El Medio Ambiente. Available online: https://www.drlopezheras.com/2014/07/cremas-para-el-sol-seguras-como-elegir.html (accessed on 1 September 2022).
- Skotarczak, K.; Osmola-Mańkowska, A.; Lodyga, M.; Polańska, A.; Mazur, M.; Adamski, Z. Photoprotection: Facts and controversies. Eur. Rev. Med. Pharmacol. Sci. 2015, 19, 98–112. [Google Scholar] [PubMed]
- Narbutt, J. Does the use of protective creams with UV filters inhibit the synthesis of vitamin D?—For and against. Prz. Pediatry 2009, 41, 75–81. [Google Scholar]
- Yeager, D.G.; Lim, H.W. What’s new in photoprotection: A review of new concepts and controversies. Dermatol. Clin. 2019, 37, 149–157. [Google Scholar] [CrossRef]
- Wang, S.Q.; Balagula, Y.; Osterwalder, U. Photoprotection: A review of the current and future technologies. Dermatol. Ther. 2010, 23, 31–47. [Google Scholar] [CrossRef]
- De Gruijl, F.R.; van Kranen, H.J.; Mullenders, L.H. UV-induced DNA damage, repair, mutations and oncogenic pathways in skin cancer. J. Photochem. Photobiol. B 2001, 63, 19–27. [Google Scholar] [CrossRef]
- Liu, F.C.; Grimsrud, T.K.; Veierød, M.B.; Robsahm, T.E.; Ghiasvand, R.; Babigumira, R.; Shala, N.K.; Stenehjem, J.S. Ultraviolet radiation and risk of cutaneous melanoma and squamous cell carcinoma in males and females in the Norwegian Offshore Petroleum Workers cohort. Am. J. Ind. Med. 2021, 64, 496–510. [Google Scholar] [CrossRef]
- Duro Mota, E.; Campillos Páez, M.T.; Causín Serrano, S. El sol y los filtros solares. Medifam 2003, 13, 39–45. [Google Scholar] [CrossRef] [Green Version]
- Cole, C.; Shyr, T.; Ou-Yang, H. Metal Oxide Sunscreens Protect Skin by Absorption, Not by Reflection or Scattering. Photodermatol. Photoimmunol. Photomed. 2016, 32, 5–10. [Google Scholar] [CrossRef] [Green Version]
- Kollias, N. The absorption properties of “physical” sunscreens. Arch. Dermatol. 1999, 135, 209–210. [Google Scholar] [CrossRef] [PubMed]
- Garrote, A.; Bonet, R. Fotoprotección. Factores de protección y filtros solares. Offarm 2008, 27, 63–73. [Google Scholar]
- Food and Drug Administration (US). CFR—Code of Federal Regulations Title 21; 2017, FDA Approved UV Filters for Sunscreens. Available online: https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfcfr (accessed on 15 April 2022).
- FDA Advances New Proposed Regulation to Make Sure That Sunscreens are Safe and Effective. Federal Register 84FR6204, 2019-03019. 2019. Available online: https://www.fda.gov/news-events/press-announcements/fda-advances-new-proposed-regulation-make-sure-sunscreens-are-safe-and-effective (accessed on 18 May 2022).
- Danovaro, R.; Bongiorni, L.; Corinaldesi, C.; Giovannelli, D.; Damiani, E.; Astolfi, P.; Greci, L.; Pusceddu, A. Sunscreens cause coral bleaching by promoting viral infections. Environ. Health Perspect. 2008, 116, 441–447. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Watkins, Y.S.D.; Sallach, J.B. Investigating the exposure and impact of chemical UV filters on coral reef ecosystems: Review and research gap prioritization. Integr. Environ. Assess Manag. 2021, 17, 967–981. [Google Scholar] [CrossRef] [PubMed]
- National Institute of Health 2020. Endocrine Disruptors. Available online: https://www.niehs.nih.gov/health/topics/agents/endocrine/index.cfm (accessed on 13 June 2022).
- Environmental Protection Agency (US). Nanomaterial Case Studies: Nanoscale Titanium Dioxide in Water Treatment and in Topical Sunscreen (Final). EPA/600/R-09/057F. 2010. Available online: https://cfpub.epa.gov/ncea/risk/recordisplay.cfm?deid=230972 (accessed on 13 June 2022).
- Environmental Protection Agency (US). Endocrine Disruptor Screening Program. 2010. Available online: https://www.epa.gov/endocrine-disruption (accessed on 14 July 2022).
- OECD. Revised Guidance Document 150 on Standardised Test Guidelines for Evaluating Chemicals for Endocrine Disruption; OECD Publishing: Paris, France, 2018. [Google Scholar]
- DeLeo, V. Sunscreen. In Bolognia J. Dermatology; Elsevier: London, UK, 2012; pp. 2197–2204. [Google Scholar]
- Garnacho Saucedo, G.M.; Salido Vallejo, R.; Moreno Giménez, J.C. Effects of solar radiation and an update on photoprotection. An. Pediatr. 2020, 92, 377. [Google Scholar] [CrossRef]
- Maier, T.; Korting, H.C. Sunscreens—Which and what for? Skin Pharm. Physiol. 2005, 18, 253–262. [Google Scholar] [CrossRef] [Green Version]
- Heneweer, M.; Muusse, M.; van den Berg, M.; Sanderson, J.T. Additive estrogenic effects of mixtures of frequently used UV filters on pS2-gene transcription in MCF-7 cells. Toxicol. Appl. Pharmacol. 2005, 208, 170–177. [Google Scholar] [CrossRef]
- Schlumpf, M.; Cotton, B.; Conscience, M.; Hailer, V.; Steinmann, B.; Lichtensteiger, W. In vitro and in vivo estrogenicity of UV screens. Environ. Health Perspect. 2001, 109, 239–244. [Google Scholar] [CrossRef]
- Coronado, M.; De Haro, H.; Deng, X.; Rempel, M.A.; Lavado, R. Estrogenic activity and reproductive effects of the UV-filter oxybenzone (2-hydroxy-4-methoxyphenyl-methanone) in fish. Aquat. Toxicol. 2008, 90, 182–187. [Google Scholar] [CrossRef]
- Krause, M.; Klit, A.; Blomberg Jensen, M.; Søeborg, T.; Frederiksen, H.; Schlumpf, M.; Lichtensteiger, W.; Skakkebaek, N.E.; Drzewiecki, K.T. Sunscreens: Are they beneficial for health? An overview of endocrine disrupting properties of UV-Filters. Int. J. Androl. 2012, 35, 424–436. [Google Scholar] [CrossRef]
- Broniowska, Z.; Slusarczyk, J.; Starek-Swiechowicz, B.; Trojan, E.; Pomierny, B.; Krzyzanowska, W.; Basta-Kaim, A.; Budziszewska, B. The effect of dermal benzophenone-2 administration on immune system activity, hypothalamic-pituitary-thyroid axis activity and hematological parameters in male Wistar rats. Toxicology 2018, 1, 1–8. [Google Scholar] [CrossRef]
- Krzyzanowska, W.; Pomierny, B.; Starek-Swiechowicz, B.; Broniowska, Ż.; Strach, B.; Budziszewska, B. The effects of benzophenone-3 on apoptosis and the expression of sex hormone receptors in the frontal cortex and hippocampus of rats. Toxicol. Lett. 2018, 296, 63–72. [Google Scholar] [CrossRef] [PubMed]
- Schreurs, R.; Lanser, P.; Seinen, W.; van der Burg, B. Estrogenic activity of UV filters determined by an in vitro reporter gene assay and an in vivo transgenic zebrafish assay. Arch. Toxicol. 2002, 76, 257–261. [Google Scholar] [CrossRef] [PubMed]
- Akhiyat, S.; Olasz-Harken, E.B. Update on human safety and the environmental impact of physical and chemical sunscreen filters: What do we know about the effects of these commonly used and important molecules? Pract. Dermatol. 2019, 48–51. Available online: https://practicaldermatology.com/articles/201 (accessed on 31 October 2022).
- Klimova, Z.; Hojerova, J.; Beránkova, M. Skin absorption and human exposure estimation of three widely discussed UV filters in sunscreens–In vitro study mimicking real-life consumer habits. Food Chem. Toxicol. 2015, 83, 237–250. [Google Scholar] [CrossRef]
- Joensen, U.N.; Jorgensen, N.; Thyssen, J.P.; Petersen, J.H.; Szecsi, P.B.; Stender, S.; Andersson, A.M.; Skakkebaek, N.E.; Frederiksen, H. Exposure to phenols, parabens and UV filters: Associations with loss-of-function mutations in the filaggrin gene in men from the general population. Environ. Int. 2017, 105, 105–111. [Google Scholar] [CrossRef]
- Schlumpf, M.; Durrer, S.; Faass, O.; Ehnes, C.; Fuetsch, M.; Gaille, C.; Henseler, M.; Hofkamp, L.; Maerkel, K.; Reolon, S.; et al. Developmental toxicity of UV filters and environmental exposure: A review. Int. J. Androl. 2008, 31, 144–151. [Google Scholar] [CrossRef]
- Kim, S.; Choi, K. Occurrences, toxicities, and ecological risks of benzophenone-3, a common component of organic sunscreen products: A mini-review. Environ. Int. 2014, 70, 143–157. [Google Scholar] [CrossRef]
- Olson, E. The rub on sunscreen. New York Times, 19 June 2006. [Google Scholar]
- DiNardo, J.C.; Downs, C.A. Dermatological and environmental toxicological impact of the sunscreen ingredient oxybenzone/benzophenone-3. J. Cosmet. Dermatol. 2018, 17, 15–19. [Google Scholar] [CrossRef]
- Huo, W.; Cai, P.; Chen, M.; Li, H.; Tang, J.; Xu, C.; Zhu, D.; Tang, W.; Xia, Y. The relationship between prenatal exposure to BP-3 and Hirschsprung’s disease. Chemosphere 2016, 144, 1091–1097. [Google Scholar] [CrossRef]
- DiNardo, J.C.; Downs, C.A. Can oxybenzone cause Hirschsprung’s disease? Reprod. Toxicol. 2019, 86, 98–100. [Google Scholar] [CrossRef] [PubMed]
- Alamer, M.; Darbre, P.D. Effects of exposure to six chemical ultraviolet filters commonly used in personal care products on motility of MCF-7 and MDA-MB-231 human breast cancer cells in vitro. J. Appl. Toxicol. 2018, 38, 148–159. [Google Scholar] [CrossRef] [PubMed]
- Pollack, A.Z.; Buck Louis, G.M.; Chen, Z.; Sun, L.; Trabert, B.; Guo, Y.; Kannan, K. Bisphenol A, benzophenone-type ultraviolet filters, and phthalates in relation to uterine leiomyoma. Environ. Res. 2015, 137, 101–107. [Google Scholar] [CrossRef] [PubMed]
- Phiboonchaiyanan, P.P.; Busaranon, K.; Ninsontia, C.; Chanvorachote, P. Benzophenone-3 increases metastasis potential in lung cancer cells via epithelial to mesenchymal transition. Cell Biol. Toxicol. 2017, 33, 251–261. [Google Scholar] [CrossRef]
- Wang, W.Q.; Duan, H.X.; Pei, Z.T.; Xu, R.R.; Qin, Z.T.; Zhu, G.C.; Sun, L.W. Evaluation by the Ames assay of the mutagenicity of UV filters using benzophenone and benzophenone-1. Int. J. Environ. Res. Public Health 2018, 15, 1907. [Google Scholar] [CrossRef] [Green Version]
- Lim, H.W.; Thomas, L.; Rigel, D.S. Photoprotection. In Photoaging; Rigel, D.S., Weiss, R.A., Lim, H.W., Eds.; Marcel Dekker: New York, NY, USA, 2004; pp. 73–88. [Google Scholar]
- Schauder, S.; Ippen, H. Contact and photocontact sensitivity to sunscreens. Review of a 15-year experience and of the literature. Contact Dermat. 1997, 37, 221–232. [Google Scholar] [CrossRef]
- Heurung, A.R.; Raju, S.I.; Warshaw, E.M. Adverse reactions to sunscreen agents: Epidemiology, responsible irritants and allergens, clinical characteristics, and management. Dermatitis 2014, 25, 289–326. [Google Scholar] [CrossRef]
- Carrascosa, J.M. El futuro se hace presente en fotoprotección solar. Rev. Piel 2011, 26, 311–314. [Google Scholar] [CrossRef]
- Sánchez Saldaña, L.; Lanchipa Yokota, P.; Pancorbo Mendoza, J.; Regis Roggero, A.; Saenz Anduaga, E.M. Fotoprotectores tópicos. Rev. Peru. Dermatol. 2002, 12, 156–163. [Google Scholar]
- EWG’s Low Danger: 1–2. 2018. Available online: https://www.ewg.org/sunscreen/report/executive-summary/#.WxcjvVMvxmB (accessed on 26 September 2021).
- Cosmetics Regulation (EC) No. 1223/2009. Available online: https://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=OJ:L:2009:342:0059:0209:es:PDF (accessed on 26 September 2021).
- Manová, E.; von Goetz, N.; Hungerbühler, K. Ultraviolet filter contact and photocontact allergy: Consumer exposure and risk assessment for octocrylene from personal care products and sunscreens. Br. J. Dermatol. 2014, 171, 1368–1374. [Google Scholar] [CrossRef]
- Bury, D.; Belov, V.N.; Qi, Y.; Hayen, H.; Volmer, D.A.; Brüning, T.; Koch, H.M. Determination of urinary metabolites of the emerging UV filter octocrylene by online-SPE-LC-MS/MS. Anal. Chem. 2018, 90, 944–951. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Available online: https://ec.europa.eu/growth/content/call-data-ingredients-potential-endocrine-disrupting-properties-used-cosmetic-products_en (accessed on 26 January 2021).
- Elder, D.P.; Snodin, D.J. Drug substances presented as sulfonic acid salts: Overview of utility, safety and regulation. J. Pharm. Pharmacol. 2009, 61, 269–278. [Google Scholar] [CrossRef] [PubMed]
- Fivenson, D.; Sabzevari, N.; Qiblawi, S.; Blitz, J.; Norton, B.B.; Norton, S.A. Sunscreens: UV filters to protect us: Part 2-Increasing awareness of UV filters and their potential toxicities to us and our environment. Int. J. Womens Dermatol. 2020, 7, 45–69, PMCID:PMC7838327. [Google Scholar] [CrossRef] [PubMed]
- Scheuer, E.; Warshaw, E. Sunscreen allergy: A review of epidemiology, clinical characteristics, and responsible allergens. Dermatitis 2006, 17, 3–11. [Google Scholar] [CrossRef]
- Osmond, M.J.; McCall, M.J. Zinc oxide nanoparticles in modern sunscreens: An analysis of potential exposure and hazard. Nanotoxicology 2010, 4, 15–41. [Google Scholar] [CrossRef]
- Hanigan, D.; Truong, L.; Schoepf, J.; Nosaka, T.; Mulchandani, A.; Tanguay, R.L.; Westerhoff, P. Trade-offs in ecosystem impacts from nanomaterial versus organic chemical ultraviolet filters in sunscreens. Water Res. 2018, 139, 281–290. [Google Scholar] [CrossRef] [PubMed]
- Smijs, T.G.; Pavel, S. Titanium dioxide and zinc oxide nanoparticles in sunscreens: Focus on their safety and effectiveness. Nanotechnol. Sci. Appl. 2011, 4, 95–112. [Google Scholar] [CrossRef] [Green Version]
- Schilling, K.; Bradford, B.; Castelli, D.; Dufour, E.; Nash, J.F.; Pape, W.; Schulte, S.; Tooley, I.; van den Bosch, J.; Schellauf, F. Human safety review of “nano” titanium dioxide and zinc oxide. Photochem. Photobiol. Sci. 2010, 9, 495–509. [Google Scholar] [CrossRef]
- Senzui, M.; Tamura, T.; Miura, K.; Ikarashi, Y.; Watanabe, Y.; Fujii, M. Study on penetration of titanium dioxide (TiO2) nanoparticles into intact and damaged skin in vitro. J. Toxicol. Sci. 2010, 35, 107–113. [Google Scholar] [CrossRef] [Green Version]
- Dussert, A.S.; Gooris, E.; Hemmerle, J. Characterization of the mineral content of a physical sunscreen emulsion and its distribution onto human stratum corneum. Int. J. Cosmet. Sci. 1997, 19, 119–129. [Google Scholar] [CrossRef]
- Zvyagin, A.V.; Zhao, X.; Gierden, A.; Sanchez, W.; Ross, J.A.; Roberts, M.S. Imaging of zinc oxide nanoparticle penetration in human skin in vitro and in vivo. J. Biomed. Opt. 2008, 13, 064031. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Scientific Committee on Consumer Safety (SCCS). Opinion on Zinc Oxide (Nano Form). 2012. Available online: http://ec.europa.eu/health/scientific_committ (accessed on 24 February 2021).
- Yabe, S.; Sato, T. Cerium oxide for sunscreen cosmetics. J. Solid State Chem. 2003, 171, 7–11. [Google Scholar] [CrossRef]
- Seixas, V.C.; Serra, O.A. Stability of sunscreens containing CePO4: Proposal for a new inorganic UV filter. Molecules 2014, 19, 9907–9925. [Google Scholar] [CrossRef] [Green Version]
- He, H.; Li, A.; Li, S.; Tang, J.; Li, L.; Xiong, L. Natural components in sunscreens: Topical formulations with sun protection factor (SPF). Biomed Pharmacother. 2021, 134, 111161. [Google Scholar] [CrossRef] [PubMed]
- Matsui, M.S.; Hsia, A.; Miller, J.D.; Hanneman, K.; Scull, H.; Cooper, K.D.; Baron, E. Non-sunscreen photoprotection: Antioxidants add value to a sunscreen. J. Investig. Dermatol. Symp. Proc. 2009, 14, 56–59. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Landaeta, K.V.; Piombo, M.E. Estrés oxidativo, carcinogénesis cutánea por radiación solar y quimioproteccion con polifenoles. Piel 2012, 27, 446–452. [Google Scholar] [CrossRef]
- Li, S.X.; Li, M.F.; Bian, J.; Wu, X.F.; Peng, F.; Ma, M.G. Preparation of organic acid lignin submicrometer particle as a natural broad-spectrum photo-protection agent. Int. J. Biol. Macromol. 2019, 132, 836–843. [Google Scholar] [CrossRef]
- Netto, M.G.; Jose, J. Development, characterization, and evaluation of sunscreen cream contain-ing solid lipid nanoparticles of silymarin. J. Cosmet. Dermatol. 2018, 17, 1073–1083. [Google Scholar] [CrossRef]
- Álvarez-Gómez, F.; Korbee, N.; Casas-Arrojo, V.; Abdala-Díaz, R.T.; Figueroa, F.L. UV photoprotection, cytotoxicity and immunology capacity of red algae extracts. Molecules 2019, 24, 341. [Google Scholar] [CrossRef] [Green Version]
- Garcia-Pichel, F.; Wingard, C.E.; Castenholz, R.W. Evidence regarding the UV sunscreen role of a mycosporine-like compound in the cyanobacterium Gloeocapsa sp. Appl. Environ. Microbiol. 1993, 59, 170–176. [Google Scholar] [CrossRef] [Green Version]
- Ahmad, H.I.; Khan, H.M.S.; Akhtar, N. Development of topical drug delivery system with Sphaeranthus indicus flower extract and its investigation on skin as a cosmeceutical product. J. Cosmet. Dermatol. 2020, 19, 985–994. [Google Scholar] [CrossRef] [PubMed]
- Ahmady, A.; Amini, M.H.; Zhakfar, A.M.; Babak, G.; Sediqi, M.N. Sun protective potential and physical stability of herbal sunscreen de-veloped from afghan medicinal plants. Turk. J. Pharm. Sci. 2020, 17, 285–292. [Google Scholar] [CrossRef] [PubMed]
- Baldisserotto, A.; Buso, P.; Radice, M.; Dissette, V.; Lampronti, I.; Gambari, R.; Manfredini, S.; Vertuani, S. Moringa oleifera leaf extracts as multifunctional ingredients for “natural and organic” sunscreens and photoprotective preparations. Molecules 2018, 23, 664. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Banerjee, K.; Thiagarajan, N.; Thiagarajan, P. Formulation and characterization of a Heli annuus-alkyl polyglucoside emulsion cream for topical applications. J. Cosmet. Dermatol. 2019, 18, 628–637. [Google Scholar] [CrossRef] [PubMed]
- Gaweł-Bęben, K.; Kukula-Koch, W.; Hoian, U.; Czop, M.; Strzępek-Gomółka, M.; Antosiewicz, B. Characterization of Cistus × incanus L. and Cistus ladanifer L. ex-tracts as potential multifunctional antioxidant ingredients for skin protecting cosmetics. Antioxidants 2020, 9, 202. [Google Scholar] [CrossRef] [Green Version]
- Hiller, J.; Klotz, K.; Meyer, S.; Uter, W.; Hof, K.; Greiner, A.; Göen, T.; Drexler, H. Systemic availability of lipophilic organic UV filters through dermal sunscreen exposure. Environ. Int. 2019, 132, 105068. [Google Scholar] [CrossRef]
- Witorsch, R.J.; Thomas, J.A. Personal care products and endocrine disruption: A critical review of the literature. Crit. Rev. Toxicol. 2010, 40, 515563. [Google Scholar] [CrossRef]
- Rehfeld, A.; Dissing, S.; Skakkebæk, N.E. Chemical UV filters mimic the effect of progesterone on Ca2+ signaling in human sperm cells. Endocrinology 2016, 157, 4297–4308. [Google Scholar] [CrossRef] [Green Version]
- Ponzo, O.J.; Silvia, C. Evidence of reproductive disruption associated with neuroendocrine changes induced by UV-B filters, phtalates and nonylphenol during sexual maturation in rats of both gender. Toxicology 2013, 311, 41–51. [Google Scholar] [CrossRef]
- Axelstad, M.; Boberg, J.; Hougaard, K.S.; Christiansen, S.; Jacobsen, P.R.; Mandrup, K.R.; Nellemann, C.; Lund, S.P.; Hass, U. Effects of pre- and postnatal exposure to the UV-filter Octyl Methoxycinnamate (OMC) on the reproductive, auditory and neurological development of rat offspring. Toxicol. Appl. Pharmacol. 2011, 250, 278–290. [Google Scholar] [CrossRef]
- Ozáez, I.; Martínez-Guitarte, J.L.; Morcillo, G. Effects of in vivo exposure to UV filters (4-MBC, OMC, BP-3, 4-HB, OC, OD-PABA) on endocrine signaling genes in the insect Chironomus riparius. Sci. Total Environ. 2013, 456, 120–126. [Google Scholar] [CrossRef] [PubMed]
- Li, A.J.; Law, J.C.F.; Chow, C.H.; Huang, Y.; Li, K.; Leung, K.S.Y. Joint Effects of Multiple UV Filters on Zebrafish Embryo Development. Environ. Sci. Technol. 2018, 52, 9460–9467. [Google Scholar] [CrossRef] [PubMed]
- Li, V.W.T.; Tsui, M.P.M.; Chen, X.; Hui, M.N.Y.; Jin, L.; Lam, R.H.W.; Yu, R.M.K.; Murphy, M.B.; Cheng, J.; Lam, P.K.S.; et al. Effects of 4-methylbenzylidene camphor (4-MBC) on neuronal and muscular development in zebrafish (Danio rerio) embryos. Environ. Sci. Pollut. Res. 2016, 23, 8275–8285. [Google Scholar] [CrossRef]
- Balázs, A.; Krifaton, C.; Orosz, I.; Szoboszlay, S.; Kovács, R.; Csenki, Z.; Urbányi, B.; Kriszt, B. Hormonal activity, cytotoxicity and developmental toxicity of UV filters. Ecotoxicol. Environ. Saf. 2016, 131, 45–53. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.H.; Hsieh, C.Y.; Ko, F.C.; Cheng, J.O. Effect of the UV-filter benzophenone-3 on intra-colonial social behaviors of the false clown anemonefish (Amphiprion ocellaris). Sci. Total Environ. 2018, 644, 1625–1629. [Google Scholar] [CrossRef]
- Carvalhais, A.; Pereira, B.; Sabato, M.; Seixas, R.; Dolbeth, M.; Marques, A.; Guilherme, S.; Pereira, P.; Pacheco, M.; Mieiro, C. Mild effects of sunscreen agents on a marine flatfish: Oxidative stress, energetic profiles, neurotoxicity and behaviour in response to titanium dioxide nanoparticles and oxybenzone. Int. J. Mol. Sci. 2021, 22, 1567. [Google Scholar] [CrossRef]
- Bahia, M.F. Proteção Solar—Actualização, 1st ed.; Universidade do Porto: Porto, Portugal, 2003. [Google Scholar]
- Bonda, C.A.; Lott, D. Sunscreen photostability. In Principles and Practice of Photoprotection; Springer: Berlin/Heidelberg, Germany, 2016; pp. 247–273. [Google Scholar]
- Tarras-Wahlberg, N.; Stenhagen, G.; Larko, O.; Rosen, A.; Wennberg, A.M.; Wennerstrom, O. Changes in ultraviolet absorption of sunscreens after ultraviolet irradiation. J. Investig. Dermatol. 1999, 113, 547–553. [Google Scholar] [CrossRef]
- Stiefel, C.; Schwack, W. Reactions of cosmetic UV filters with skin proteins: Model studies of esters with primary amines. Trends Photochem. Photobiol. 2013, 15, 105–116. [Google Scholar]
| UVB Filters/Risks | UVA Filters/Risks | Broad Spectrum Filters (UVA and UVB)/Risks |
|---|---|---|
| PABA and derivates - Octyldimethyl PABA or Padimate O: EDC (estrogenic), liberator of free radicals, allergic reactions. Always avoid. - Ethylhexyl triazone (octyl triazone): Free radical liters with sunlight. Prudence. | Benzophenones (BP-3 or Oxybenzone): EDC effects, bioaccumulative, photoallergic reactions, absorbed through the skin, passing into breast milk. Neurotoxicity. Always avoid | Tinosorb M (Methylene bis-benzotriazolyl tetramethylbutylphenol): Possible environmental contaminant. Acceptable/caution. |
| Cinnamates - Octinoxate (Octyl Methoxycinnamate, OMC, Ethylhexyl Methoxycinnamate, EHMC): confirmed EDC, persistent and bioaccumulative, absorbed through the skin, appears in breast milk. Always avoid. - Cinoxato (2-Ethoxyethyl-P-Methoxycinnamate): In disuse. Little data on its safety. Avoid as a precaution. | Anthranilates: less effective than benzophenones. | Tinosorb S (Bis-Ethylhexyloxyphenol methoxyphenyl triazine): non-estrogenic. Acceptable/caution. |
| Salicylates: Better security profile. - Octisalate (ethylhexyl salicylate, 2-Ethylhexyl 2-Hydroxybenzoate): Acceptable chemical filter. - Homosalate (3,3,5-Trimethyl-Salicylate Cyclohexanol; 3,3,5-Trimethylcyclohexyl 2-Hydroxybenzoate): Highly Polluting, weak EDC, decomposes with light into oxidizing substances harmful skin. Increases the absorption of other substances. Avoid. | Avobenzone (Butyl Methoxydibenzoylmethane): Rare allergies. The most recommended by the EWG. | Iscotrizinol (Uvasorb HEB, diethylhexyl butamido triazone): No data, Acceptable/caution. |
| Octocrylene: EDC activity, allergies and/or (photo)allergies. Avoid. | Mexoryl SX (Tetraphthalidine sulfonic acid dialcamphor): also considered safe. | |
| Ensulizole (2-Pheny 1H-benzimidazole 5-Sulfonic acid, PBSA): produces free radicals (DNA damage and potentially skin cancer. Avoid. | ||
| 4-Methylbenzylidene Camphor: EDC, persistent and bioaccumulative. Possible thyroid toxicity. Always avoid. |
| Inorganic Filters | Organic Filters | Biological Filters |
|---|---|---|
| Titamiun dioxide | PABA and derivates | Lignin |
| Zinc Oxide | Cinnamates | Silimarine |
| Salicylates | Marine antioxidants | |
| Octocrylene | Plant-derived extracts | |
| Ensulizole | ||
| 4-Methylbenzylidene Camphor | ||
| Benzophenones | ||
| Anthranilates | ||
| Avobenzone | ||
| Mexoryl SX |
| Compounds in Organic/Inorganic Filters | Toxicity Range (1–10) |
|---|---|
| Oxybenzone | High Danger: 7–10 |
| Ethylhexyl methoxycinnamate | High Danger: 7–10 |
| Homosalate | High Danger: 7–9 |
| Octisalate | High Danger: 7–9 |
| Octocrylene | Medium Danger: 3–6 |
| Titanium dioxide | Low Danger: 1–2 |
| Zinc oxide | Low Danger: 1–2 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Santander Ballestín, S.; Luesma Bartolomé, M.J. Toxicity of Different Chemical Components in Sun Cream Filters and Their Impact on Human Health: A Review. Appl. Sci. 2023, 13, 712. https://doi.org/10.3390/app13020712
Santander Ballestín S, Luesma Bartolomé MJ. Toxicity of Different Chemical Components in Sun Cream Filters and Their Impact on Human Health: A Review. Applied Sciences. 2023; 13(2):712. https://doi.org/10.3390/app13020712
Chicago/Turabian StyleSantander Ballestín, Sonia, and María José Luesma Bartolomé. 2023. "Toxicity of Different Chemical Components in Sun Cream Filters and Their Impact on Human Health: A Review" Applied Sciences 13, no. 2: 712. https://doi.org/10.3390/app13020712
APA StyleSantander Ballestín, S., & Luesma Bartolomé, M. J. (2023). Toxicity of Different Chemical Components in Sun Cream Filters and Their Impact on Human Health: A Review. Applied Sciences, 13(2), 712. https://doi.org/10.3390/app13020712







