Abstract
Currently, there is an increase in the use of preparations from collagen-containing raw materials in the production of meat products. The high functional and technological properties of such preparations make it possible to significantly improve the rheological properties of food products, as well as organoleptic indicators, and enrich meat products with dietary fibers. The aim of this study was to study the effect of an emulsion of 5% protein hydrolysate and 1% purslane powder on herodietic boiled sausages for the elderly. The results showed that the experimental samples of boiled sausages contained 15.44% protein, 6.6% fat, 4.2% carbohydrates and 71.1% moisture; this shows that boiled sausage with the addition of protein hydrolysate contains optimal chemical composition for dietary nutrition, a certain moisture index confirms the excellent consistency of boiled sausage. The change in the peroxide number was studied for 7 days. The results showed that the peroxide number did not change significantly from 2.0 meq/kg to 4.6 meq/kg. Thus, the addition of purslane with antioxidant properties helps to slow down the oxidation of fats and thereby increases the shelf life of boiled herodietic sausage. The results of the study of color characteristics showed stable color formation, which was influenced by an emulsion of collagen and purslane. It was found that products using emulsions from collagen-containing raw materials are characterized by a higher concentration of tyrosine due to the action of proteolytic enzymes (pepsin and trypsin)—from 728.1 mcg/mL (during the first three hours of hydrolysis) to 392.5 mcg/mL (during 6 h of hydrolysis)—which indicates a higher degree of protein digestibility these products. Thus, the addition of an emulsion of 5% protein hydrolysate and 1% purslane powder can be recommended for the production of boiled sausages with improved nutritional and taste properties.
1. Introduction
The increase in the world’s population leads to an increase in the consumption of food, including meat. Meat production volumes are increasing by 4–5% annually. At the same time, only 50–54% of each carcass reaches the end consumer in the form of meat [1,2]. The rest goes into the category of industrial waste or secondary products [3,4]. Waste disposal involves environmental risks and additional costs, so the processing of by-products into value-added products is more acceptable [5,6].
Boiled sausages, also known as frankfurters [7] or hot dogs [8], are popular processed meat products enjoyed worldwide [9]. They are characterized by their smooth texture [10], savory flavor [11] and convenience [12]. Boiled sausages are typically made from a combination of finely ground meat [13], fat [14], spices [15] and additives [16], which are mixed and stuffed into casings before undergoing cooking processes such as boiling or steaming [17].
The popularity of boiled sausages can be attributed to several factors, including their versatility, affordability and widespread availability [18,19,20]. They are commonly consumed as a quick snack, part of a meal or as an ingredient in various dishes [21,22,23]. In recent years, there has been increasing interest in improving the nutritional profile of boiled sausages to meet consumer demands for healthier options [24].
Boiled sausages are a popular food product that is widely consumed in many countries around the world [25,26,27]. These sausages are distinguished by their delicate texture, unique taste and many variations in the recipe [28,29,30]. They are a source of protein, vitamins and minerals, which makes them a popular choice to meet the nutritional needs of people of different age groups, including the elderly [31,32,33,34].
Elderly people have special nutritional needs associated with changes in the body, age restrictions and loss of appetite [35]. In this context, the development of boiled sausages using protein hydrolysate from recycled raw materials represents a significant potential for improving the nutritional value and popularity of these products among the elderly [36,37,38].
Studies have been conducted to optimize the development process of low-sodium chicken cutlets with improved quality characteristics, which can also be applied to the development of boiled sausages using protein hydrolysate [39].
The optimization of pork meat sausage with a healthier fat content and improved sensory characteristics can also be applied to the development of cooked sausages using protein hydrolysate [40].
Various approaches to the formation of a healthier lipid profile of functional meat-based products, including boiled sausages, have been considered, including the replacement of meat fats with non-meat fats to improve the nutritional composition of these products [41].
This study investigates the effect of natural antioxidants on the quality and shelf-life stability of frankfurters formulated with olive oil. It highlights the importance of using natural ingredients to enhance the sensory and nutritional attributes of boiled sausages [42].
The potential use of plasma protein hydrolysates as functional ingredients for the preparation of sausages is considered to investigate the effect of the inclusion of these hydrolysates on the physico-chemical and sensory properties of boiled sausages [43].
Secondary products of the meat industry can become a rich source of protein and protein hydrolysates for use in the food industry, including in the production of sausages. Due to their chemical composition and functional properties, animal proteins are an alternative to proteins isolated from soy and can be used in the production of meat products as a meat substitute, which achieves the following: increasing nutritional and biological value; improving structural, mechanical and organoleptic properties; enhancing the taste of meat; and reducing the cost of meat products [44]. Consumers today are guided not only by the taste qualities of the product but also by those aspects that are related to health and environmental friendliness. The combination of these factors leads manufacturers to search for new formulations and a way to reduce waste [45].
For normal processes of digestion and absorption in the gastrointestinal tract, the composition of the chyme must necessarily include dietary fibers involved in many metabolic processes [46]. According to this hypothesis, the physiological role of dietary fibers is determined primarily by the physicochemical properties of the components; they consist of cellulose, pectin, acidic polysaccharides, collagen and its fractions, as well as other so-called ballast substances [47]. The ability to gel and change the volume depending on environmental conditions determines their effect on intestinal motility and chyme evacuation processes. The presence on the surface of the fibers of functional groups capable of interacting with dissolved substances ensures the participation of dietary fibers in the processes regulating the distribution of substances inside the intestinal cavity and their transport into the internal environment. The participation of fibers in the adsorption of substances from the liquid phase of the chyme changes the composition of this phase and, consequently, the kinetics of transport of these substances into the body. Carboxyl groups, which are part of dietary fiber molecules, participate as a buffer in the pH change process. They are also functional main groups that ensure the adsorption of ions, especially multicharged cations forming [48,49,50].
The appearance of products containing dietary fibers, in particular collagen and its fractions, must fully comply with the historically established consumer habits [51]. Nevertheless, using methods of creating environmentally friendly full-fledged food products and knowing the biological functions of collagen as a representative of dietary fiber and, at the same time, dietary protein, it is possible, obviously, to choose such options for dietary diets containing collagen that will not reduce and, in some cases, may even increase the effectiveness of their use [52]. Collagen and its fractions occupy a special place in the preparation of human diets. This is due to the fact that, on the one hand, they play the role of full-fledged dietary fibers, and on the other hand, they are sources of dietary protein [53]. Another aspect of this study is the creation of multifunctional meat products that have the property of reducing the oxidative properties of meat fats and, therefore, prolonging shelf life in the absence of standard synthetic preservatives. As previously established in the report of the study of the functional properties of Portulaca oleracea L., it is a source of potassium, followed by magnesium and calcium, and has the potential to be used as a vegetable source of omega-3 fatty acids. It is a very good source of alpha-linolenic acid and linoleic acid. It contains a sufficient amount of carotenoids and B vitamins. Also, in view of the presence of flavonoids, the established antioxidant capacity of iron and sufficiently good antioxidant activity, it is assumed that its addition to meat products will serve as a source of enrichment and will be able to affect the storage capacity of products.
The purpose of this study is to improve nutritional value, technological and organoleptic characteristics and increase shelf life by adding collagen hydrolysate and purslane.
2. Materials and Methods
2.1. Materials
The objects of the study are boiled beef sausages using emulsions from collagen-containing raw materials (sausages beef + 5% hydrolysate + 1% purslane). Protein hydrolysate was obtained by fermented hydrolysis of beef, horse, and lamb legs with a put joint.
To carry out effective hydrolysis of protein substrates, the enzyme concentration was chosen: 5% by the enzyme BLT 7 [54]. The hydrolysis time is 24 h at a temperature of 45 °C.
The drying of the hydrolysate was carried out using a Spray Dryer NSP-1500 (NANBEI LTD, Zhengzhou, China) shown in Figure 1.

Figure 1.
Drying mode of protein hydrolysate in Spray Dryer NSP-1500.
Boiled sausages with the addition of protein hydrolysate are produced according to the standard technology of boiled sausages. Purslane powder (in the amount of 1% by weight of minced meat) and protein hydrolysate shown in Figure 2 (in the amount of 5% by weight of minced meat) were added during minced meat cutting.

Figure 2.
Protein hydrolysate from by-products.
The heat treatment was carried out to a temperature inside the sausage of 72 °C.
The samples were developed at the pilot production at the Saken Seifullin Kazakh Agrotechnical Research University.
2.2. Determination of Methyl Esters of Fatty Acids
Methyl esters of fatty acids were analyzed on an Agilent 7890 gas chromatograph manufactured by Agilent Technologies (Santa Clara, CA, USA) with a flame ionization detector and a capillary column HP-Innowax 60 m × 0.32 mm × 0.5 mkm in nitrogen current. Temperature gradient from 100 to 260 °C at a rate of 10 °C/min. Injection of 1 µL, mixing of the gas flow 1:100, detector temperature within 250…300 °C, respectively. We used a standard mixture of comparison of methyl esters of fatty acids Supelco No. 47885U with automatic calculation of data on the content of C6…C24 fatty acids. Calculation of the quantitative content of fatty acids was carried out using the method of internal normalization.
Statistical processing of the results (standard methodologies) was carried out in accordance with the metrological characteristics stated in the methods; in the absence of such, the principles set out in accordance with paragraph 5.5 of RMG 76-2014 were applied (the critical significance level of the null statistical hypothesis (p) was assumed to be 0.05) [55].
2.3. Determination of Amino Acid Composition
The amino acid composition of proteins was determined by ion-column chromatography using a T339 analyzer (Mikrotehna, Prague, Czech Republic) [56].
2.4. Protein Digestibility
The digestibility of proteins of the studied products by digestive enzymes “in vitro” was determined by the Pokrovsky–Yertanov method.
For samples of boiled sausage products, digestion was simulated according to the Pokrovsky and Yertanov protocol, which is presented below. In particular, a control sample was simulated, with the addition of enzymes but without the addition of samples.
A total of 25 mL of freshly prepared pepsin solution was added to 0.5 g of the crushed sample (concentration of 1 mg/mL: 25 mL of 0.02 N hydrochloric acid solution (pH = 1.2) was mixed with 25 mg of crystalline pepsin), thoroughly mixed and heated at 37 °C, and then kept at this temperature for 3 h.
The remains of the samples after digestion with pepsin (25 mL) were neutralized by stirring 0.65 mL of 2 N sodium hydroxide, after which 25 mL of 0.02 N sodium bicarbonate solution (pH 8.2) and 25 mg of crystalline trypsin (the final concentration of the enzyme in solution 0.5 mg/mL) was added with further incubation at 37 °C for 3 h.
After the digestion was completed, the samples were immediately frozen at −40 °C for several hours. To further measure the protein concentration, the samples were defrosted and separated in a centrifuge for 20 min at 14,000 rpm and a supernatant was taken.
The protein concentration was measured using the Lowry method in three repeats in each sample. The concentration of the control sample was also measured, which was subtracted from the sausage indicators in order to exclude the influence of enzymes on the protein values in the samples [57].
2.5. Determination of Color Characteristics
The color characteristics of the samples were determined using a Konica Minolta CM-2300d spectrophotometer calibrated using standard black-and-white calibration plates. The color values were expressed as L (lightness), a (redness) and b (yellowness).
To determine the color resistance to light, the criterion for assessing color stability (Y) was used. Color stability was calculated using the following formula:
where
L1 and L2 are the values of the light index before and after exposure to light;
a1 and a2—the values of the redness index before and after exposure to light;
b1 and b2—the values of the yellowness index before and after exposure to light.
When determining the color resistance to light, the sample was placed under an artificial light source (an incandescent, fluorescent lamp with a power of at least 40 watts). After 1 h after the start of the experiment, the change in color characteristics was instrumentally determined [58].
The studies were carried out in a five-fold repetition. Data processing was carried out using the Microsoft Excel 16.76 in combination with XLSTAT program.
2.6. Determination of the Peroxide Number
The method is based on the reaction of interaction of primary products of fat oxidation (peroxides and hydroperoxides) with potassium iodide in an acidic medium, followed by titration with a solution of sodium thiosulfate and quantitative determination of the released iodine.
2.7. Analysis of the Molecular Mass Distribution of Protein Fractions of a Sample by One-Dimensional Electrophoresis
A total of 100 mg of the sample was taken, and 500 mL of a lysing solution was added (4.5 M urea, 2.5% β-mercaptoethanol, 1% triton X-100, 1% ampholines with a pH of 3–10). The resulting homogenate was clarified by centrifugation at 14,000 rpm for 20 min. After that, the supernatant was separated and added to it in a 1:1 ratio of protein buffer. To prepare this, 1 mL of sodium dodecyl sulfate (SDS) 10%, 250 µL of concentrated β-mercaptoethanol, 625 µL of Tris-HCl 0.5 M, and 1.5 g of urea were added to Eppendorf-type tubes; bromophenol blue was added until they were dark colored and brought to a volume of 5 mL with water; and then the samples were heated in a boiling water bath for 5 min.
Visualization and analysis of images. The staining of Coomassie G250 proteins was carried out in a solution of the following composition: 10% acetic acid, 25% isopropanol and 0.05% Coomassie G-250. A total of 10% acetic acid was used to remove the unbound dye. For computer densitometry, one-dimensional electrophoregrams that were in a wet state were used. Their full digital images were obtained using a Bio-5000 Plus scanner (Serva, Heidelberg, Germany) in 600 dpi 2D-RGB mode. The resulting digital images were edited in a graphic editor [59].
3. Results and Discussion
3.1. The Results of the Study of the Chemical Composition of Herodietic Sausages
The evaluation of the chemical composition of experimental samples of boiled meat products did not reveal any abnormal deviations, and all indicators were in the generally accepted contents of this type of product (Table 1).

Table 1.
Physico-chemical indicators of herodietic boiled sausage products with the addition of purslane and collagen raw materials.
Attention is only drawn to the high content of carbohydrates and low content of fat. Thus, with the generally accepted technology of production of this type of product, the fat content ranges from 10–20% absolute units, and carbohydrates less than 2–3%. Perhaps this is due to the addition of a carbohydrate additive, purslane, and the use of less fatty raw materials for the production.
The study of the dynamics of changes in the composition of protein fractions based on the results of comparative studies of the ratio of sarcoplasmic proteins, based on the extraction of sarcoplasmic proteins from muscle tissue with a buffer solution of low ionic strength and obtaining fractions of water-soluble, salt-soluble and alkali-soluble proteins, followed by the determination of their number by the Kjeldahl method, are presented in Table 1. The largest number of fractions in the experimental sample were isolated alkali-soluble proteins, the least salt-soluble, which does not contradict the literature data.
The strength properties of muscle tissue depend on the type, fatness and variety of meat and change during processing. The tensile strength of muscle tissue is (10–20) × 105 Pa, collagen fibers (2000–6500) × 105 and elastin fibers (1000–2000) × 105. The tension of the cut of raw meat lies within (1.3–1.9) × 105; after cooking, it increases to (2.7–4.7) × 105. The results of the experiment to determine the ultimate shear stress and shear stress of cooked sausages are presented in Table 1. It was determined that boiled sausage products using emulsions from collagen-containing raw materials are characterized by a rather delicate consistency, which is obviously due to lower values of BCC and VUS in comparison with products without the introduction of collagen protein.
3.2. Results of the Study of Fatty Acid Composition, Oxidation of Lipids and Proteins
Table 2 shows the main content of the fatty acid composition of the studied samples.

Table 2.
Fatty acid composition of herodietic sausages.
As a result of the conducted studies, out of 36 fatty acids, only 14 indicators were isolated in significant concentrations.
Currently, synthetic antioxidants are widely used to slow down the oxidation of lipids and proteins in the meat industry. However, consumers are concerned about these synthetic antioxidants because of their potential toxicological effects. Accordingly, natural antioxidants can be used because of their health safety compared to synthetic antioxidants. The aim of this study was to evaluate the protective effects of Portulaca extract (Portulaca oleracea L.) against the oxidation of lipids and proteins during refrigerated storage for 7 days.
The dynamics of oxidative spoilage and cooked products are presented in Table 3.

Table 3.
Dynamics of fat oxidation and protein oxidation (background) in herodietic sausages during storage.
When evaluating the results obtained, it can be concluded that the accumulation of the peroxide number is proceeding at a low rate; perhaps this can be attributed to the introduction of 1% purslane into the component composition, which slows down, among other things, oxidative processes in both proteins and fats. Purslane extract has good antioxidant activity because it contains polyphenols, organic acids with a small chain and saccharides. Purslane, as a potential natural antioxidant, has excellent properties, including good stability, non-toxicity and medicinal value, compared to synthetic antioxidants; in addition, it is a popular low-calorie product and has therapeutic efficacy. Therefore, the use of purslane in sausage products seems relevant and appropriate.
3.3. Results of Color Stability
When studying the stability before and after exposure to light on the indicators of cooked sausages, the following values were obtained for the main characteristics of color: L (lightness), a (redness) and b (yellowness). The data are presented in Table 4.

Table 4.
Color characteristics of herodietic sausages.
Thus, when determining the color stability of meat products to the effects of temperature conditions (heating to a temperature of 70–72 °C), the influence of added chemical agents (according to the generally accepted formulation: chlorides, phosphates, etc.) and to determine the color stability during storage, color characteristics were studied, and color stability was established. The obtained values for lightness and redness decreased slightly in the region of 2% and 8%, respectively. At the same time, yellowness, on the contrary, when exposed to light, had higher values (by 20%) than before its exposure. This indicates a fairly stable color formation in the product, which may be associated with the introduction of purslane, which has good antioxidant properties that apparently prevent the oxidation of protein pigments.
3.4. Fractional Composition of Proteins of Herodietic Sausage
As a result of the study of the fractional composition of the protein, the following values were obtained. The data are presented in Figure 3.
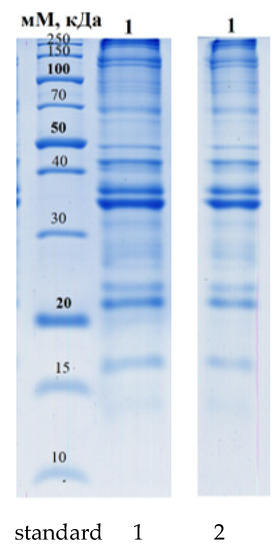
Figure 3.
One-dimensional electrophoregram of the studied samples. Symbols: St—Standard of molecular weights: 250, 150, 100, 70, 50, 40, 30, 20, 15, 10 kDa (from top to bottom); 1—stabbing 6 µL; 2—stabbing 4 µL (sample).
The isolated protein fractions are mainly related to beef. Additionally, proteins belonging to the purslane family were identified in this sample (Table 5).

Table 5.
Identified proteins related to Portulaca.
As a result, 158 protein fractions were isolated using computer densitometry; the main proteins belonged to the species Bos taurus (Bovine), which is confirmed by the composition of the sample. Three proteins (enzymes) related to plant genes were also isolated:
- -
- Large-chain ribulose bisphosphate carboxylase;
- -
- 4,5-DOPA dioxygenase extradiol;
- -
- Maturase K.
Ribulose bisphosphate carboxylase (RBFCO) is the main enzyme of plant leaves [60]. The use of RBFKO protein for medicinal and nutritional purposes still attracts little attention, although it has long been noted that RBFKO protein is close to milk casein in amino acid composition. Especially attractive from this point of view seems to be the possibility of enriching protein with essential amino acids using genetic engineering techniques.
4,5-DOPA dioxygenase extradiol: the kinetic properties of the enzyme are characterized by the formation of betalamic acid (where they act as anthocyanins in other plants), a structural, chromophoric and bioactive unit of the plant pigment betalains [61].
Dioxygenases are non-heme iron-containing enzymes that catalyze the incorporation of both molecular oxygen atoms into catechol derivatives, which leads to ring cleavage [62]. 4,5-DODA catalyzes the oxidation of DOPA, which opens the ring, with the formation of an intermediate product, 4,5-secodopa, which undergoes spontaneous cyclization to form betalamic acid.
In addition to their physiological role as plant pigments, betalains are molecules with strong antiradical activity. It has been demonstrated that tumor formation in the skin and liver is inhibited at very low concentrations of pigments in the diet of mice [63]. In addition, it has also been reported that the administration of betalaine is a protective factor against the effects of gamma radiation in these animals [59]. In humans, the concentrations of betalains in the oral plasma are sufficient to promote their inclusion in LDL and erythrocytes, which are then protected from oxidative damage and hemolysis [64].
Maturase K (matK) is a plastid gene of plants. The protein it encodes is an organelle intron maturase, a protein that splices group II introns. It is necessary for the connection of group II introns in vivo [65]. This protein preserves only the well-conserved X domain and the remnants of the reverse transcriptase domain.
Universal matK primers can be used to encode the DNA of angiosperms [65].
3.5. Results of the Study of Amino Acid Composition
One of the main indicators of food quality is the biological value, reflecting the degree of compliance with the amino acid balance of the body’s needs, necessary for the course of physiological processes in the body (Table 6).

Table 6.
Amino acid composition of herodietic sausages.
A significant increase in the content of essential amino acids such as lysine and valine and interchangeable amino acids such as arginine, glycine and serine in the test samples confirms that the protein hydrolysate introduced into the prototype is rich and well-balanced in amino acid composition.
3.6. Determination of the Digestibility of Herodietic Sausages by Food Enzymes
In vitro digestive models play a key role in studying the functional characteristics of protein compounds that enter the human body and undergo enzymatic hydrolysis in the gastrointestinal tract. The bioconversion in the reproduction of the Pokrovsky–Yertanov protocol is limited at the stage of trypsinolysis but allows the achievement of the last phase of the destruction of protein molecules into individual amino acids [56]. This method has a high potential for evaluating the biological effects and mechanisms of action of peptides and amino acids obtained as a result of the enzymatic hydrolysis of proteins.
When studying the digestibility of boiled sausage products, the following values were obtained (presented in Table 7).

Table 7.
Results of the evaluation of the digestibility of boiled sausages.
A comparative analysis of protein digestibility of experimental and control samples of boiled sausage products was carried out. It was found that products using emulsions from collagen-containing raw materials are characterized by a higher concentration of tyrosine due to the action of proteolytic enzymes (pepsin and trypsin)—from 728.1 mcg/mL (during the first three hours of hydrolysis) to 392.5 mcg/mL (during 6 h of hydrolysis)—which indicates a higher degree of protein digestibility these products. From the data obtained, it can be concluded that the use of a collagen complex of hydrolyzed proteins in a prototype of cooked meat products and the concentration of tyrosine increased the availability of macromolecules to the action of digestive enzymes, thereby improving the digestibility of the product.
4. Conclusions
In conclusion, the physicochemical parameters of experimental samples of meat products were studied, and the effectiveness of the use of protein hydrolysate and vegetable additives of purslane powder was proved. The addition of protein hydrolysate in an amount of 5% improves the consistency and nutritional value of cooked sausages. The use of purslane powder in an amount of 1% has an antioxidant effect, which increases the shelf life of cooked sausage, thereby reducing the amount of sodium nitrite in the sausage. The data obtained allow us to recommend these boiled sausages for the diet of the elderly.
Author Contributions
Conceptualization, K.M. and T.T.; methodology, G.T.; validation, G.Z. (Galia Zamaratskaia); formal analysis, K.M. and R.M.; investigation, G.T. and A.M. (Ainur Matibayeva); resources, T.T.; data curation, A.M. (Almas Mukhametov); writing—original draft preparation, K.M.; writing—review and editing, A.M. (Ainur Matibayeva), G.Z. (Gulmira Zhakupova) and D.K.; visualization, R.M.; supervision, K.M. All authors have read and agreed to the published version of the manuscript.
Funding
This research is funded by the Ministry of Agriculture of the Republic of Kazakhstan (BR10764998).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
All data generated or analyzed during this study are included in this published article.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Geiker, N.R.W.; Bertram, H.C.; Mejborn, H.; Dragsted, L.O.; Kristensen, L.; Carrascal, J.R.; Bügel, S.; Astrup, A. Meat and Human Health—Current Knowledge and Research Gaps. Foods 2021, 10, 1556. [Google Scholar] [CrossRef]
- Tieland, M.; Borgonjen-Van den Berg, K.J.; van Loon, L.J.C.; de Groot, L.C.P.G.M. Dietary Protein Intake in Community-Dwelling, Frail, and Institutionalized Elderly People: Scope for Improvement. Eur. J. Nutr. 2011, 51, 173–179. [Google Scholar] [CrossRef]
- Kowalski, Z.; Krupa-Żuczek, K. A Model of the Meat Waste Management. Pol. J. Chem. Technol. 2007, 9, 91–97. [Google Scholar] [CrossRef]
- Toldrá, F.; Mora, L.; Reig, M. New Insights into Meat By-Product Utilization. Meat Sci. 2016, 120, 54–59. [Google Scholar] [CrossRef]
- Adewale, P.; Dumont, M.-J.; Ngadi, M. Enzyme-Catalyzed Synthesis and Kinetics of Ultrasonic Assisted Methanolysis of Waste Lard for Biodiesel Production. Chem. Eng. J. 2016, 284, 158–165. [Google Scholar] [CrossRef]
- Wancura, J.H.C.; Brondani, M.; dos Santos, M.S.N.; Oro, C.E.D.; Wancura, G.C.; Tres, M.V.; Oliveira, J.V. Demystifying the Enzymatic Biodiesel: How Lipases Are Contributing to Its Technological Advances. Renew. Energy 2023, 216, 119085. [Google Scholar] [CrossRef]
- Tokysheva, G.; Tultabayeva, T.; Mukhtarkhanova, R.; Zhakupova, G.; Gorbulya, V.; Kakimov, M.; Makangali, K. The Study of Physicochemical and Technological Properties of Boiled Sausage Recommended for the Older Adults. Potravin. Slovak J. Food Sci. 2023, 17, 16–29. [Google Scholar] [CrossRef]
- de Lima, A.L.; Guerra, C.A.; Costa, L.M.; de Oliveira, V.S.; Lemos Junior, W.J.F.; Luchese, R.H.; Guerra, A.F. A Natural Technology for Vacuum-Packaged Cooked Sausage Preservation with Potentially Postbiotic-Containing Preservative. Fermentation 2022, 8, 106. [Google Scholar] [CrossRef]
- Pasichnyi, V.; Bozhko, N.; Tischenko, V.; Kotliar, Y. Development of cooked smoked sausage on the basis of muskovy duck meat. Food Sci. Technol. 2019, 12, 102–109. [Google Scholar] [CrossRef]
- Rigdon, M.; Stelzleni, A.M.; McKee, R.W.; Pringle, T.D.; Bowker, B.; Zhuang, H.; Thippareddi, H. Texture and Quality of Chicken Sausage Formulated with Woody Breast Meat. Poult. Sci. 2021, 100, 100915. [Google Scholar] [CrossRef]
- Chen, H.; Wang, H.; Qi, J.; Wang, M.; Xu, X.; Zhou, G. Chicken Breast Quality—Normal, Pale, Soft and Exudative (PSE) and Woody—Influences the Functional Properties of Meat Batters. Int. J. Food Sci. Technol. 2017, 53, 654–664. [Google Scholar] [CrossRef]
- Cross, H.R.; Carpenter, Z.L.; Smith, G.C. Effects of intramuscular collagen and elastin on bovine muscle tenderness. J. Food Sci. 1973, 38, 998–1003. [Google Scholar] [CrossRef]
- Bogard, A.K.; Fuller, C.C.; Radke, V.; Selman, C.A.; Smith, K.E. Ground Beef Handling and Cooking Practices in Restaurants in Eight States. J. Food Prot. 2013, 76, 2132–2140. [Google Scholar] [CrossRef] [PubMed]
- Valsta, L.M.; Tapanainen, H.; Männistö, S. Meat Fats in Nutrition. Meat Sci. 2005, 70, 525–530. [Google Scholar] [CrossRef] [PubMed]
- Lu, F.; Kuhnle, G.K.; Cheng, Q. The Effect of Common Spices and Meat Type on the Formation of Heterocyclic Amines and Polycyclic Aromatic Hydrocarbons in Deep-Fried Meatballs. Food Control 2018, 92, 399–411. [Google Scholar] [CrossRef]
- Surendran Nair, M.; Nair, D.V.T.; Kollanoor Johny, A.; Venkitanarayanan, K. Use of Food Preservatives and Additives in Meat and Their Detection Techniques. In Meat Quality Analysis; Academic Press: Cambridge, MA, USA, 2020; pp. 187–213. [Google Scholar] [CrossRef]
- Villalobos-Delgado, L.H.; Núñez-González, F.A.; Alarcon-Rojo, A.D.; Silva-Avila, N.J. Quality of Cooked Sausages with Added Beef or Pork Heart Surimi. J. Food Process. Preserv. 2020, 44, e14939. [Google Scholar] [CrossRef]
- Strashynskiy, I.; Fursik, O.; Pasichniy, V.; Marynin, A.; Goncharov, G. The study of properties of minces in boiled sausages with functional food composition use. Eureka Life Sci. 2016, 6, 31–36. [Google Scholar] [CrossRef][Green Version]
- Strashynskiy, I.; Fursik, O.; Pasichniy, V.; Marynin, A.; Goncharov, G. Influence of Functional Food Composition on the Properties of Meat Mince Systems. East.-Eur. J. Enterp. Technol. 2016, 6, 53–58. [Google Scholar] [CrossRef][Green Version]
- Rudy, M.; Kucharyk, S.; Duma-Kocan, P.; Stanisławczyk, R.; Gil, M. Unconventional Methods of Preserving Meat Products and Their Impact on Health and the Environment. Sustainability 2020, 12, 5948. [Google Scholar] [CrossRef]
- Morimoto, J.M.; Marchioni, D.M.L.; Cesar, C.L.G.; Fisberg, R.M. Statistical Innovations Improve Prevalence Estimates of Nutrient Risk Populations: Applications in São Paulo, Brazil. J. Acad. Nutr. Diet. 2012, 112, 1614–1618. [Google Scholar] [CrossRef]
- Pasichnyi, V.; Ukrainets, A.; Khrapachov, O.; Marynin, A.; Svyatnenko, R.; Moroz, O. Research into Efficiency of Pasterization of Boiled Sausage Products in Order to Improve Their Storage Term. East.-Eur. J. Enterp. Technol. 2018, 6, 21–28. [Google Scholar] [CrossRef]
- Akpan, I.P. Trends in Sausage Production. Afr. J. Food Sci. Technol. 2017, 8, 81–84. [Google Scholar] [CrossRef]
- Naik, H.R.; Amin, T. Processing and Preservation Of Meat and Meat Products. In Food Processing and Preservation; CRC Press: Boca Raton, FL, USA, 2021; pp. 159–170. [Google Scholar] [CrossRef]
- Danyliv, M.M.; Vasilenko, O.A.; Ozherelyeva, O.N.; Shestakova, Y.A. Improvement of Sausage Production Technology. IOP Conf. Ser. Earth Environ. Sci. 2019, 341, 012131. [Google Scholar] [CrossRef]
- Danyliv, M.M.; Vasilenko, O.A.; Ozherelyeva, O.N.; Stanislavskaya, E.B. Developing a Technology for Ground Meat Low-Fat Semi-Products. IOP Conf. Ser. Earth Environ. Sci. 2021, 624, 012165. [Google Scholar] [CrossRef]
- Kurćubić, V.; Vukašinović, M.; Mašković, P.; Petrović, M. Examination of the certain chemical characteristics of different types of boiled sausages produced in Serbia. J. Cent. Eur. Agric. 2012, 13, 643–653. [Google Scholar] [CrossRef]
- Sinegubov, J.K. Study of Consumer Requirements for the Quality of Boiled Sausage Products; Russian State Agrarian University-Moscow State Agricultural Academy named after K. A. Timiryazev: Moscow, Russia, 2021. [Google Scholar] [CrossRef]
- Ivanova, L.A.; Charushina, E.B.; Khodareva, E.V. Of Discrepancy Prediction Algorithm in the Model of Improve the Quality of Serial Processes of Engineering Production. J. Phys. Conf. Ser. 2019, 1399, 033012. [Google Scholar] [CrossRef]
- Fursik, O.; Strashynskiy, I.; Pasichny, V.; Kochubei-Lytvynenko, O. Quality assessment of proteins in cooked sausages with food compositions. Food Sci. Technol. 2018, 12, 80–88. [Google Scholar] [CrossRef]
- Güemes-Vera, N.; Zamora-Natera, J.F.; Soto, S.S. Frankfurter Sausage Texture Is Affected by Using Isolate, Concentrate and Flour of Lupinus Albus and Pork Skin Proteins. Food Res. 2018, 2, 234–239. [Google Scholar] [CrossRef]
- Cardona-Hincapié, J.A.; Restrepo-Molina, D.A.; López-Vargas, J.H. Effect of a Total Substitution of Vegetable Protein and Phosphates on Shrinkage by Cooking and Purging in Chopped York Ham. Revista Facultad Nacional de Agronomía Medellín 2020, 73, 9333–9340. [Google Scholar] [CrossRef]
- Fursik, O.; Strashynskyi, I.; Pasichnyi, V.; Svyatnenko, R. Biological Efficiently of Cooked Sausages Protein. Sci. Messenger LNU Vet. Med. Biotechnol. 2019, 21, 48–53. [Google Scholar] [CrossRef]
- Houmy, N.; Melhaoui, R.; Belhaj, K.; Richel, A.; Sindic, M.; Hano, C.; Kodad, S.; Mihamou, A.; Addi, M.; Abid, M.; et al. Chemical Characterization of Almond Meal as a Co-Product of the Mechanical Extraction of Almond Oil. E3S Web Conf. 2020, 183, 04004. [Google Scholar] [CrossRef]
- Kaur, D.; Rasane, P.; Singh, J.; Kaur, S.; Kumar, V.; Mahato, D.K.; Dey, A.; Dhawan, K.; Kumar, S. Nutritional Interventions for Elderly and Considerations for the Development of Geriatric Foods. Curr. Aging Sci. 2019, 12, 15–27. [Google Scholar] [CrossRef] [PubMed]
- Al-Atif, H. Collagen Supplements for Aging and Wrinkles: A Paradigm Shift in the Field of Dermatology and Cosmetics. Dermatol. Pract. Concept. 2022, 12, e2022018. [Google Scholar] [CrossRef] [PubMed]
- Tobin, D.J. Introduction to Skin Aging. J. Tissue Viability 2017, 26, 37–46. [Google Scholar] [CrossRef] [PubMed]
- Barbosa, M.C.; Grosso, R.A.; Fader, C.M. Hallmarks of Aging: An Autophagic Perspective. Front. Endocrinol. 2019, 9, 790. [Google Scholar] [CrossRef]
- Zhang, L.; Zhang, S.; Song, H.; Li, B. Ingestion of Collagen Hydrolysates Alleviates Skin Chronological Aging in an Aged Mouse Model by Increasing Collagen Synthesis. Food Funct. 2020, 11, 5573–5580. [Google Scholar] [CrossRef]
- Exposito, J.-Y.; Valcourt, U.; Cluzel, C.; Lethias, C. The Fibrillar Collagen Family. Int. J. Mol. Sci. 2010, 11, 407–426. [Google Scholar] [CrossRef]
- Domingo, C.J.A.; Sartagoda, K.J.; Catandijan, N.J.C.; Yasin, N.K. Impact of Vegetable Fat on the Sensory and Physicochemical Quality Characteristics of Chevon Sausage. Appl. Food Res. 2023, 3, 100265. [Google Scholar] [CrossRef]
- Guntarti, A.; Ahda, M.; Kusbandari, A. Determining Fatty Acids and Halal Authentication of Sausage. Food Res. 2019, 4, 495–499. [Google Scholar] [CrossRef]
- Choi, S.-M.; Choi, J.-H.; Choi, Y.-S.; Kim, H.-W.; Kim, H.-Y.; Lee, M.-A.; Hwang, K.-E.; Lee, J.-W.; Kim, C.-J. Effects of Kimchi Powder on the Quality Properties of Emulsion Type Sausage Made with Irradiated Pork Meat. Korean J. Food Sci. Anim. Resour. 2011, 31, 649–657. [Google Scholar] [CrossRef]
- Álvarez García, C.; Marcet Manrique, I. Meat Proteins as a Potential Source of Bioactive Ingredients for Food and Pharmaceutical Use. In Novel Proteins for Food, Pharmaceuticals and Agriculture; Wiley: Hoboken, NJ, USA, 2018; pp. 29–49. [Google Scholar] [CrossRef]
- Mora, L.; Gallego, M.; Toldrá, F. ACEI-Inhibitory Peptides Naturally Generated in Meat and Meat Products and Their Health Relevance. Nutrients 2018, 10, 1259. [Google Scholar] [CrossRef]
- Su, M.; Chen, D.; Zhou, J.; Shen, Q. Effects of Different Dietary Carbohydrate Sources on the Meat Quality and Flavor Substances of Xiangxi Yellow Cattle. Animals 2022, 12, 1136. [Google Scholar] [CrossRef] [PubMed]
- Yadav, S.; Malik, A.; Pathera, A.; Islam, R.U.; Sharma, D. Development of Dietary Fibre Enriched Chicken Sausages by Incorporating Corn Bran, Dried Apple Pomace and Dried Tomato Pomace. Nutr. Food Sci. 2016, 46, 16–29. [Google Scholar] [CrossRef]
- Yadav, S.; Pathera, A.K.; Islam, R.U.; Malik, A.K.; Sharma, D.P. Effect of Wheat Bran and Dried Carrot Pomace Addition on Quality Characteristics of Chicken Sausage. Asian-Australas. J. Anim. Sci. 2018, 31, 729–737. [Google Scholar] [CrossRef] [PubMed]
- Parkash, J.; Yadav, S.; Sharma, D.P.; Pathera, A.K.; Raut, S. Development of Dietary Fibre Enriched Chevon Rolls by Incorporating Corn Bran and Dried Apple Pomace. J. Anim. Res. 2016, 6, 603. [Google Scholar] [CrossRef]
- Pathera, A.K.; Riar, C.S.; Yadav, S.; Sharma, D.P. Effect of Dietary Fiber Enrichment and Different Cooking Methods on Quality of Chicken Nuggets. Korean J. Food Sci. Anim. Resour. 2017, 37, 410–417. [Google Scholar] [CrossRef]
- Pereira, A.G.T.; Ramos, E.M.; Teixeira, J.T.; Cardoso, G.P.; Ramos, A.L.S.; Fontes, P.R. Effects of the Addition of Mechanically Deboned Poultry Meat and Collagen Fibers on Quality Characteristics of Frankfurter-Type Sausages. Meat Sci. 2011, 89, 519–525. [Google Scholar] [CrossRef]
- Kamani, M.H.; Meera, M.S.; Bhaskar, N.; Modi, V.K. Partial and Total Replacement of Meat by Plant-Based Proteins in Chicken Sausage: Evaluation of Mechanical, Physico-Chemical and Sensory Characteristics. J. Food Sci. Technol. 2019, 56, 2660–2669. [Google Scholar] [CrossRef]
- Ahmad, S.; Rizawi, J.A.; Srivastava, P.K. Effect of Soy Protein Isolate Incorporation on Quality Characteristics and Shelf-Life of Buffalo Meat Emulsion Sausage. J. Food Sci. Technol. 2010, 47, 290–294. [Google Scholar] [CrossRef]
- Aktayeva, S.; Baltin, K.; Kiribayeva, A.; Akishev, Z.; Silayev, D.; Ramankulov, Y.; Khassenov, B. Isolation of Bacillus Sp. A5.3 Strain with Keratinolytic Activity. Biology 2022, 11, 244. [Google Scholar] [CrossRef]
- Ichihara, K.; Fukubayashi, Y. Preparation of Fatty Acid Methyl Esters for Gas-Liquid Chromatography. J. Lipid Res. 2010, 51, 635–640. [Google Scholar] [CrossRef] [PubMed]
- Vasilevskaya, E.R.; Aruzina, M.A.; Akhremko, A.G. In Vitro Bioconversion: Comparison of Multi-Enzyme Systems INFOGEST 2.0 and Pokrovkiy-Ertanov Protocol. Vsyo O Myase 2022, 2022, 44–49. [Google Scholar] [CrossRef]
- Skaljac, S.; Jokanovic, M.; Tomovic, V.; Ivic, M.; Sojic, B.; Ikonic, P.; Peulic, T. Colour Characteristics of Vacuum Packed Fermented Sausage during Storage. IOP Conf. Ser. Earth Environ. Sci. 2019, 333, 012101. [Google Scholar] [CrossRef]
- Lu, X.; Wang, Y.; Zhang, Z. Radioprotective Activity of Betalains from Red Beets in Mice Exposed to Gamma Irradiation. Eur. J. Pharmacol. 2009, 615, 223–227. [Google Scholar] [CrossRef] [PubMed]
- Drachuk, U.; Simonova, I.; Halukh, B.; Basarab, I.; Romashko, I. The study of lentil flour as a raw material for production of semi-smoked sausages. East.-Eur. J. Enterp. Technol. 2018, 11, 44–50. [Google Scholar]
- Mate, C.J.; von Caemmerer, S.; Evans, J.R.; Hudson, G.S.; Andrews, T.J. The Relationship between CO2-Assimilation Rate, Rubisco Carbamylation and Rubisco Activase Content in Activase-Deficient Transgenic Tobacco Suggests a Simple Model of Activase Action. Planta 1996, 198, 604–613. [Google Scholar] [CrossRef]
- Gandía-Herrero, F.; García-Carmona, F. Characterization of Recombinant Beta Vulgaris 4,5-DOPA-Extradiol-Dioxygenase Active in the Biosynthesis of Betalains. Planta 2012, 236, 91–100. [Google Scholar] [CrossRef]
- Lipscomb, J.D. Mechanism of Extradiol Aromatic Ring-Cleaving Dioxygenases. Curr. Opin. Struct. Biol. 2008, 18, 644–649. [Google Scholar] [CrossRef]
- Kapadia, G.J.; Azuine, M.A.; Sridhar, R.; Okuda, Y.; Tsuruta, A.; Ichiishi, E.; Mukainake, T.; Takasaki, M.; Konoshima, T.; Nishino, H.; et al. Chemoprevention of DMBA-Induced UV-B Promoted, NOR-1-Induced TPA Promoted Skin Carcinogenesis, and DEN-Induced Phenobarbital Promoted Liver Tumors in Mice by Extract of Beetroot. Pharmacol. Res. 2003, 47, 141–148. [Google Scholar] [CrossRef]
- Tesoriere, L.; Butera, D.; D’Arpa, D.; Di Gaudio, F.; Allegra, M.; Gentile, C.; Livrea, M.A. Increased Resistance to Oxidation of Betalain-Enriched Human Low Density Lipoproteins. Free Radic. Res. 2003, 37, 689–696. [Google Scholar] [CrossRef]
- Yu, J.; Xue, J.-H.; Zhou, S.-L. New Universal matK Primers for DNA Barcoding Angiosperms. J. Syst. Evol. 2011, 49, 176–181. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).