Dynamic Multi-Task Graph Isomorphism Network for Classification of Alzheimer’s Disease
Abstract
1. Introduction
2. Related Work
2.1. Deep Learning-Based Alzheimer’s Disease Classification Methods
2.1.1. CNN-Based Methods
2.1.2. GNN-Based Methods
2.1.3. Other Deep Learning Methods
2.2. Application of Multi-Task Learning in AD Classification
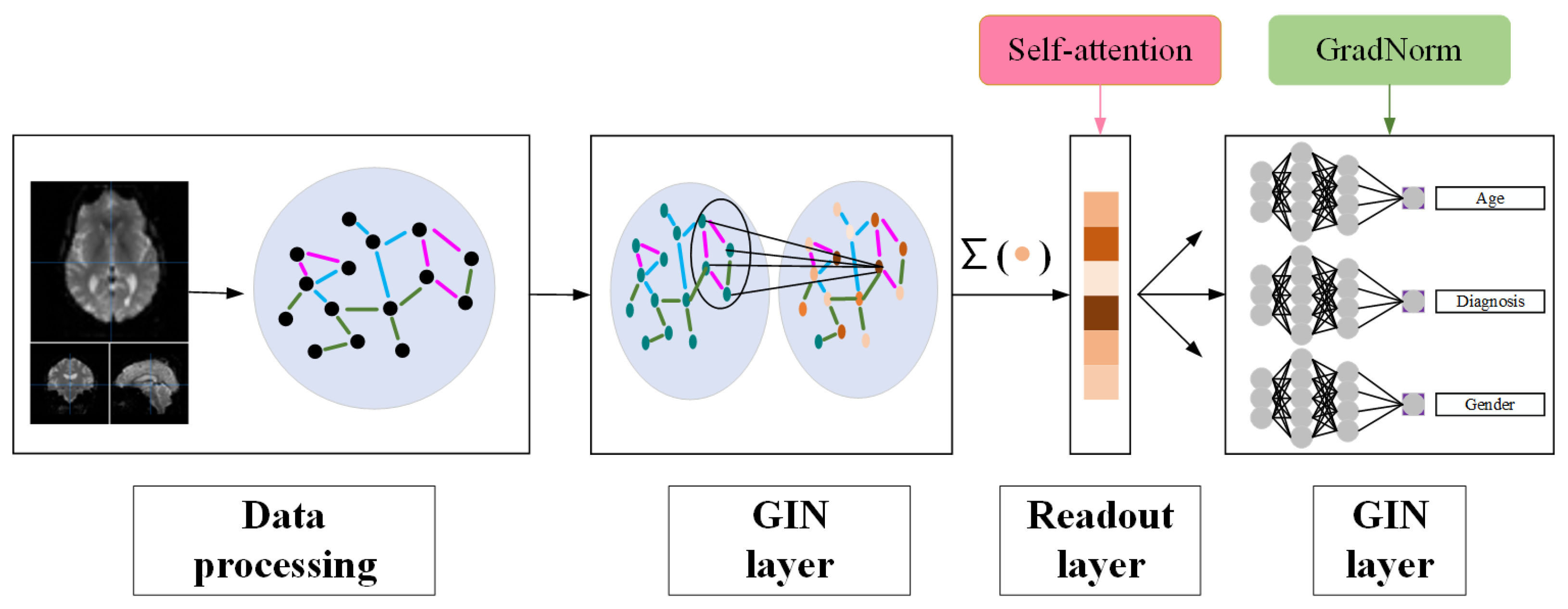
3. Methods
3.1. Data Preprocessing
3.2. GIN Architecture
3.3. Multi-Task Integration
4. Experiments
4.1. Benchmark Dataset
4.2. Experimental Settings
4.2.1. Baseline
4.2.2. Evaluation Metrics
4.2.3. Implementation Details
4.3. Model Comparison
4.4. Ablation Study
4.4.1. Effectiveness of the Attention Mechanism
4.4.2. Effectiveness of Multi-Task Learning
4.4.3. Effectiveness of the GradNorm Algorithm
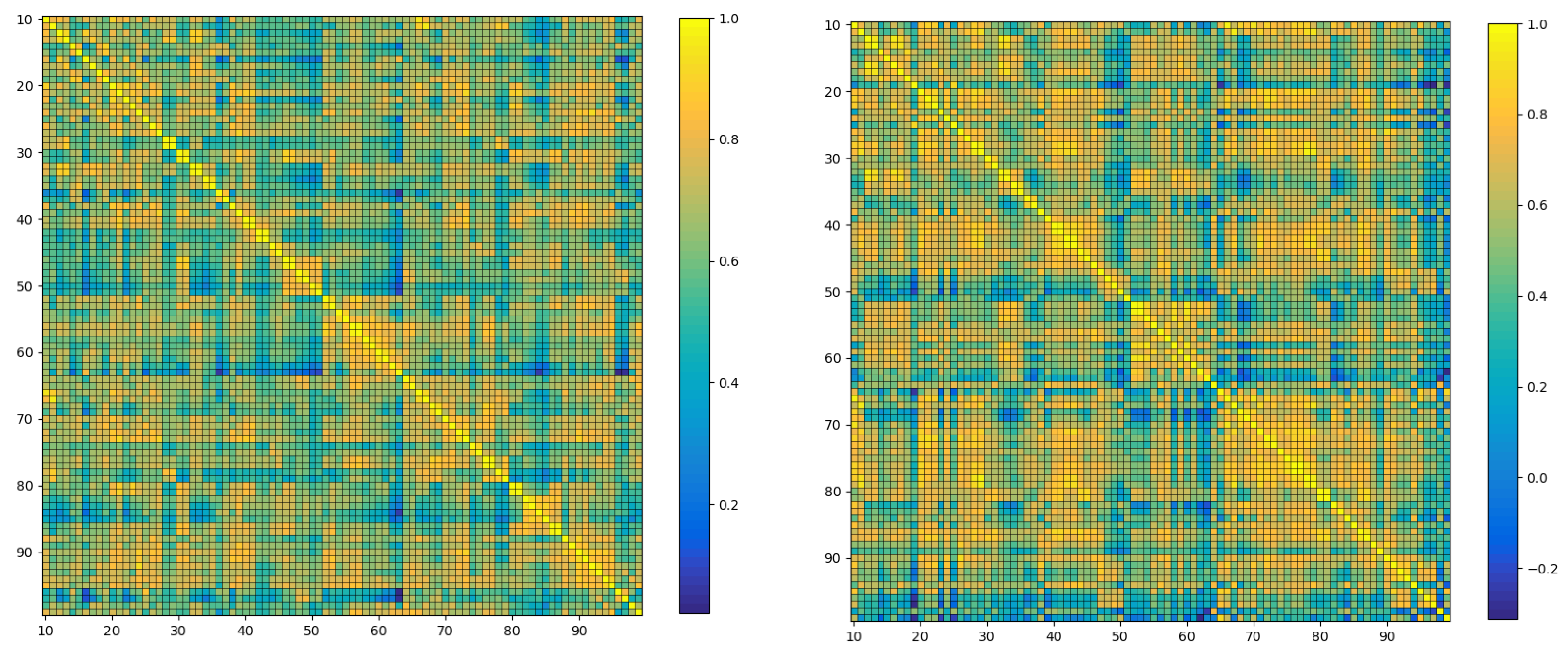
4.5. Correlation Analysis
4.6. Model Interpretability
5. Discussion and Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Smith, M.A. Alzheimer disease. Int. Rev. Neurobiol. 1998, 42, 1–54. [Google Scholar] [PubMed]
- Cummings, J.L.; Cole, G. Alzheimer disease. JAMA 2002, 287, 2335–2338. [Google Scholar] [CrossRef] [PubMed]
- Nichols, E.; Szoeke, C.E.; Vollset, S.E.; Abbasi, N.; Abd-Allah, F.; Abdela, J.; Aichour, M.T.E.; Akinyemi, R.O.; Alahdab, F.; Asgedom, S.W.; et al. Global, regional, and national burden of Alzheimer’s disease and other dementias, 1990–2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol. 2019, 18, 88–106. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. The World Health Report 2000. Available online: https://www.who.int/publications/i/item/924156198X (accessed on 20 June 2023).
- Huang, Y.; Mucke, L. Alzheimer mechanisms and therapeutic strategies. Cell 2012, 148, 1204–1222. [Google Scholar] [CrossRef] [PubMed]
- Calisto, F.M.G.F.; de Matos Fernandes, J.G.; Morais, M.; Santiago, C.; Abrantes, J.M.V.; Nunes, N.J.; Nascimento, J.C. Assertiveness-based Agent Communication for a Personalized Medicine on Medical Imaging Diagnosis. In Proceedings of the 2023 CHI Conference on Human Factors in Computing Systems, Hamburg, Germany, 23–28 April 2023; pp. 1–20. [Google Scholar]
- Sarraf, S.; Tofighi, G. Deep learning-based pipeline to recognize Alzheimer’s disease using fMRI data. In Proceedings of the Future Technologies Conference, San Francisco, CA, USA, 6–7 December 2016; pp. 816–820. [Google Scholar]
- Matthews, P.M.; Jezzard, P. Functional magnetic resonance imaging. J. Neurol. Neurosurg. Psychiatry 2004, 75, 6–12. [Google Scholar]
- Tuovinen, T.; Kananen, J.; Rajna, Z.; Lieslehto, J.; Korhonen, V.; Rytty, R.; Mattila, H.; Huotari, N.; Raitamaa, L.; Helakari, H.; et al. The variability of functional MRI brain signal increases in Alzheimer’s disease at cardiorespiratory frequencies. Sci. Rep. 2020, 10, 21559. [Google Scholar] [CrossRef]
- Xu, K.; Hu, W.; Leskovec, J.; Jegelka, S. How powerful are graph neural networks? arXiv 2018, arXiv:1810.00826. [Google Scholar]
- Kim, B.H.; Ye, J.C. Understanding graph isomorphism network for rs-fMRI functional connectivity analysis. Front. Neurosci. 2020, 14, 630. [Google Scholar] [CrossRef]
- Caruana, R. Multitask Learning; Springer: Berlin/Heidelberg, Germany, 1998. [Google Scholar]
- Braak, H.; Braak, E. Frequency of stages of Alzheimer-related lesions in different age categories. Neurobiol. Aging 1997, 18, 351–357. [Google Scholar] [CrossRef]
- Podcasy, J.L.; Epperson, C.N. Considering sex and gender in Alzheimer disease and other dementias. Dialogues Clin. Neurosci. 2022, 18, 437–446. [Google Scholar] [CrossRef]
- Xing, X.; Li, Q.; Yuan, M.; Wei, H.; Xue, Z.; Wang, T.; Shi, F.; Shen, D. DS-GCNs: Connectome classification using dynamic spectral graph convolution networks with assistant task training. Cereb. Cortex 2021, 31, 1259–1269. [Google Scholar] [CrossRef] [PubMed]
- Kazemi, Y.; Houghten, S.K. A deep learning pipeline to classify different stages of Alzheimer’s disease from fMRI data. In Proceedings of the IEEE Conference on Computational Intelligence in Bioinformatics and Computational Biology, Saint Louis, MO, USA, 30 May–2 June 2018; pp. 1–8. [Google Scholar]
- Janghel, R.; Rathore, Y. Deep convolution neural network based system for early diagnosis of Alzheimer’s disease. IRBM 2021, 42, 258–267. [Google Scholar] [CrossRef]
- Ghafoori, S.; Shalbaf, A. Predicting conversion from MCI to AD by integration of rs-fMRI and clinical information using 3D-convolutional neural network. Int. J. Comput. Assist. Radiol. Surg. 2022, 17, 1245–1255. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Zhou, Y.; Dvornek, N.C.; Zhang, M.; Zhuang, J.; Ventola, P.; Duncan, J.S. Pooling Regularized Graph Neural Network for fMRI Biomarker Analysis. Lecture Notes in Computer Science. In Proceedings of the Medical Image Computing and Computer Assisted Intervention, Lima, Peru, 4–8 October 2020; Volume 12267, pp. 625–635. [Google Scholar]
- Yang, C.; Wang, P.; Tan, J.; Liu, Q.; Li, X. Autism spectrum disorder diagnosis using graph attention network based on spatial-constrained sparse functional brain networks. Comput. Biol. Med. 2021, 139, 104963. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Song, X.; Qiu, Y.; Zhao, C.; Lei, B. Structure and Feature Based Graph U-Net for Early Alzheimer’s Disease Prediction. In Proceedings of the 11th International Workshop on Multimodal Learning for Clinical Decision Support, Strasbourg, France, 1 October 2021; Volume 13050, pp. 93–104. [Google Scholar]
- Song, X.; Zhou, F.; Frangi, A.F.; Cao, J.; Xiao, X.; Lei, Y.; Wang, T.; Lei, B. Multi-center and multi-channel pooling GCN for early AD diagnosis based on dual-modality fused brain network. IEEE Trans. Med. Imaging 2022, 42, 354–367. [Google Scholar] [CrossRef] [PubMed]
- Lin, K.; Jie, B.; Dong, P.; Ding, X.; Bian, W.; Liu, M. Extracting Sequential Features from Dynamic Connectivity Network with rs-fMRI Data for AD Classification. In Proceedings of the Machine Learning in Medical Imaging—12th International Workshop, Strasbourg, France, 27 September 2021; Volume 12966, pp. 664–673. [Google Scholar]
- Alorf, A.; Khan, M.U.G. Multi-label classification of Alzheimer’s disease stages from resting-state fMRI-based correlation connectivity data and deep learning. Comput. Biol. Med. 2022, 151, 106240. [Google Scholar] [CrossRef]
- Xia, Z.; Zhou, T.; Mamoon, S.; Lu, J. Recognition of dementia biomarkers with deep finer-DBN. IEEE Trans. Neural Syst. Rehabil. Eng. 2021, 29, 1926–1935. [Google Scholar] [CrossRef]
- Shao, W.; Peng, Y.; Zu, C.; Wang, M.; Zhang, D.; The Alzheimer’s Disease Neuroimaging Initiative. Hypergraph based multi-task feature selection for multimodal classification of Alzheimer’s disease. Comput. Med. Imaging Graph. 2020, 80, 101663. [Google Scholar] [CrossRef]
- Zeng, N.; Li, H.; Peng, Y. A new deep belief network-based multi-task learning for diagnosis of Alzheimer’s disease. Neural Comput. Appl. 2021, 35, 11599–11610. [Google Scholar] [CrossRef]
- Friston, K.J.; Williams, S.; Howard, R.; Frackowiak, R.S.; Turner, R. Movement-related effects in fMRI time-series. Magn. Reson. Med. 1996, 35, 346–355. [Google Scholar] [CrossRef]
- Jenkinson, M.; Bannister, P.; Brady, M.; Smith, S. Improved optimization for the robust and accurate linear registration and motion correction of brain images. Neuroimage 2002, 17, 825–841. [Google Scholar] [CrossRef] [PubMed]
- Tzourio-Mazoyer, N.; Landeau, B.; Papathanassiou, D.; Crivello, F.; Etard, O.; Delcroix, N.; Mazoyer, B.; Joliot, M. Automated anatomical labeling of activations in SPM using a macroscopic anatomical parcellation of the MNI MRI single-subject brain. Neuroimage 2002, 15, 273–289. [Google Scholar] [CrossRef] [PubMed]
- Rubinov, M.; Sporns, O. Complex network measures of brain connectivity: Uses and interpretations. Neuroimage 2010, 52, 1059–1069. [Google Scholar] [CrossRef]
- Petersen, R.C.; Aisen, P.S.; Beckett, L.A.; Donohue, M.C.; Gamst, A.C.; Harvey, D.J.; Jack, C.R.; Jagust, W.J.; Shaw, L.M.; Toga, A.W.; et al. Alzheimer’s disease neuroimaging initiative (ADNI): Clinical characterization. Neurology 2010, 74, 201–209. [Google Scholar] [CrossRef] [PubMed]
- Efron, B. Bootstrap Methods: Another Look at the Jackknife; Springer: Berlin/Heidelberg, Germany, 1992. [Google Scholar]
- Vaswani, A.; Shazeer, N.; Parmar, N.; Uszkoreit, J.; Jones, L.; Gomez, A.N.; Kaiser, L.; Polosukhin, I. Attention is All you Need. In Proceedings of the Advances in Neural Information Processing Systems 30: Annual Conference on Neural Information Processing Systems, Long Beach, CA, USA, 4–9 December 2017; pp. 5998–6008. [Google Scholar]
- He, K.; Gan, C.; Li, Z.; Rekik, I.; Yin, Z.; Ji, W.; Gao, Y.; Wang, Q.; Zhang, J.; Shen, D. Transformers in medical image analysis: A review. Intell. Med. 2023, 3, 59–78. [Google Scholar] [CrossRef]
- Lin, Z.; Feng, M.; Santos, C.N.d.; Yu, M.; Xiang, B.; Zhou, B.; Bengio, Y. A structured self-attentive sentence embedding. arXiv 2017, arXiv:1703.03130. [Google Scholar]
- Nair, V.; Hinton, G.E. Rectified Linear Units Improve Restricted Boltzmann Machines. In Proceedings of the 27th International Conference on Machine Learning, Haifa, Israel, 21–24 June 2010; pp. 807–814. [Google Scholar]
- Zhang, Y.; Yang, Q. A survey on multi-task learning. IEEE Trans. Knowl. Data Eng. 2021, 34, 5586–5609. [Google Scholar] [CrossRef]
- Ruder, S. An overview of multi-task learning in deep neural networks. arXiv 2017, arXiv:1706.05098. [Google Scholar]
- Nebel, R.A.; Aggarwal, N.T.; Barnes, L.L.; Gallagher, A.; Goldstein, J.M.; Kantarci, K.; Mallampalli, M.P.; Mormino, E.C.; Scott, L.; Yu, W.H.; et al. Understanding the impact of sex and gender in Alzheimer’s disease: A call to action. Alzheimer’s Dement. 2018, 14, 1171–1183. [Google Scholar] [CrossRef]
- Chen, Z.; Badrinarayanan, V.; Lee, C.; Rabinovich, A. GradNorm: Gradient Normalization for Adaptive Loss Balancing in Deep Multitask Networks. In Proceedings of the 35th International Conference on Machine Learning, Stockholm, Sweden, 10–15 July 2018; Volume 80, pp. 793–802. [Google Scholar]
- Weiner, M.W.; Veitch, D.P.; Aisen, P.S.; Beckett, L.A.; Cairns, N.J.; Cedarbaum, J.; Donohue, M.C.; Green, R.C.; Harvey, D.; Jack, C.R., Jr.; et al. Impact of the Alzheimer’s disease neuroimaging initiative, 2004 to 2014. Alzheimer’s Dement. 2015, 11, 865–884. [Google Scholar] [CrossRef]
- Jack, C.R., Jr.; Bernstein, M.A.; Fox, N.C.; Thompson, P.; Alexander, G.; Harvey, D.; Borowski, B.; Britson, P.J.; Whitwell, J.L.; Ward, C.; et al. The Alzheimer’s disease neuroimaging initiative (ADNI): MRI methods. J. Magn. Reson. Imaging Off. J. Int. Soc. Magn. Reson. Med. 2008, 27, 685–691. [Google Scholar] [CrossRef]
- Kaiser, A.; Haller, S.; Schmitz, S.; Nitsch, C. On sex/gender related similarities and differences in fMRI language research. Brain Res. Rev. 2009, 61, 49–59. [Google Scholar] [CrossRef]
- Cortes, C.; Vapnik, V. Support-vector networks. Mach. Learn. 1995, 20, 273–297. [Google Scholar] [CrossRef]
- Altman, N.S. An introduction to kernel and nearest-neighbor nonparametric regression. Am. Stat. 1992, 46, 175–185. [Google Scholar]
- Al Fahoum, A.; Ghobon, T.A. Performance predictions of Sci-Fi films via machine learning. Appl. Sci. 2023, 13, 4312. [Google Scholar] [CrossRef]
- LeCun, Y.; Bengio, Y.; Hinton, G. Deep learning. Nature 2015, 521, 436–444. [Google Scholar] [CrossRef] [PubMed]
- Kipf, T.N.; Welling, M. Semi-supervised classification with graph convolutional networks. arXiv 2016, arXiv:1609.02907. [Google Scholar]
- Velickovic, P.; Cucurull, G.; Casanova, A.; Romero, A.; Liò, P.; Bengio, Y. Graph Attention Networks. In Proceedings of the 6th International Conference on Learning Representations, Vancouver, BC, Canada, 30 April–3 May 2018. [Google Scholar]
- Xu, B.; Shen, H.; Cao, Q.; Qiu, Y.; Cheng, X. Graph wavelet neural network. arXiv 2019, arXiv:1904.07785. [Google Scholar]
- Du, J.; Zhang, S.; Wu, G.; Moura, J.M.F.; Kar, S. Topology adaptive graph convolutional networks. arXiv 2017, arXiv:1710.10370. [Google Scholar]
- Hanley, J.A.; McNeil, B.J. The meaning and use of the area under a receiver operating characteristic (ROC) curve. Radiology 1982, 143, 29–36. [Google Scholar] [CrossRef]
- Kohavi, R. A Study of Cross-Validation and Bootstrap for Accuracy Estimation and Model Selection. In Proceedings of the 14th International Joint Conference on Artificial Intelligence, Montréal, QC, Canada, 20–25 August 1995; pp. 1137–1145. [Google Scholar]
- Kingma, D.P.; Ba, J. Adam: A method for stochastic optimization. arXiv 2014, arXiv:1412.6980. [Google Scholar]
- Lundberg, S.M.; Lee, S.I. A unified approach to interpreting model predictions. Adv. Neural Inf. Process. Syst. 2017, 30, 4768–4777. [Google Scholar]
- Al Fahoum, A.S.; Abu Al-Haija, A.O.; Alshraideh, H.A. Identification of coronary artery diseases using photoplethysmography signals and practical feature selection process. Bioengineering 2023, 10, 249. [Google Scholar] [CrossRef] [PubMed]



| Dataset | Subgroup | Number | Gender (M/F) | Age (Mean ± Std.) |
|---|---|---|---|---|
| ADNI | AD | 118 | 62/65 | 74.69 ± 7.44 |
| CN | 185 | 84/101 | 74.79 ± 5.97 |
| Hyper-Parameters | Accuracy | Specificity | Recall | F1 Score | |
|---|---|---|---|---|---|
| batch_size = 32 | lr = 0.0001 | 85.11 | 94.15 | 73.30 | 81.03 |
| batch_size = 32 | lr = 0.001 | 81.06 | 96.09 | 61.60 | 73.92 |
| batch_size = 32 | lr = 0.01 | 72.74 | 97.24 | 40.00 | 55.97 |
| batch_size = 64 | lr = 0.0001 | 76.65 | 87.69 | 61.23 | 68.64 |
| batch_size = 64 | lr = 0.001 | 90.44 | 95.97 | 83.22 | 89.14 |
| batch_size = 64 | lr = 0.01 | 81.80 | 92.30 | 62.33 | 74.41 |
| batch_size = 128 | lr = 0.0001 | 78.86 | 90.25 | 63.98 | 72.42 |
| batch_size = 128 | lr = 0.001 | 86.58 | 92.85 | 77.47 | 82.49 |
| batch_size = 128 | lr = 0.01 | 76.83 | 97.46 | 48.47 | 63.79 |
| Model | Accuracy | Specificity | Recall | F1 Score |
|---|---|---|---|---|
| SVM | 83.12 | 82.91 | 83.36 | 79.36 |
| KNN | 70.24 | 72.62 | 67.69 | 64.65 |
| WKNN | 72.58 | 74.31 | 70.43 | 67.80 |
| CNN | 83.24 | 83.54 | 78.21 | 82.19 |
| GCN | 88.93 | 95.94 | 79.82 | 86.25 |
| GAT | 85.66 | 95.42 | 73.10 | 81.69 |
| GWNN | 85.29 | 96.95 | 67.59 | 78.49 |
| TAGCN | 86.02 | 96.12 | 72.64 | 81.73 |
| DMT–GIN | 90.44 | 95.97 | 83.22 | 89.14 |
| Methods | Accuracy | Specificity | Recall | F1 Score |
|---|---|---|---|---|
| (No. 1) Backbone | 85.76 | 93.85 | 78.45 | 84.91 |
| (No. 2) Backbone + AT | 86.34 | 94.04 | 75.67 | 86.04 |
| (No. 3) Backbone + AT + MT | 88.72 | 95.86 | 81.29 | 85.89 |
| (No. 4) Backbone + MT + GN | 89.88 | 95.76 | 82.15 | 88.21 |
| (No. 5) Backbone + AT + MT + GN | 90.44 | 95.97 | 83.22 | 89.14 |
| D | B | C | E | C | P | |
|---|---|---|---|---|---|---|
| D | 1.00 | 0.75 | −0.56 | 0.67 | −0.75 | 0.82 |
| B | 0.75 | 1.00 | 0.45 | −0.52 | 0.63 | −0.72 |
| C | −0.56 | 0.45 | 1.00 | 0.71 | −0.59 | 0.49 |
| E | 0.67 | −0.52 | 0.71 | 1.00 | 0.84 | −0.76 |
| C | −0.75 | 0.63 | −0.59 | 0.84 | 1.00 | 0.87 |
| P | 0.82 | −0.72 | 0.49 | −0.76 | 0.87 | 1.00 |
| Rank | Brain Region | SHAP Value | Model |
|---|---|---|---|
| 1 | Precentral gyrus | 0.234 | DMT-GIN |
| 2 | Middle frontal gyrus | 0.201 | DMT-GIN |
| 3 | Inferior frontal gyrus, opercular part | 0.176 | DMT-GIN |
| 4 | Inferior frontal gyrus, triangular part | 0.152 | DMT-GIN |
| 5 | Supplementary motor area | 0.138 | DMT-GIN |
| 1 | Fusiform gyrus | 0.231 | GCN |
| 2 | Superior parietal lobule | 0.203 | GCN |
| 3 | Precentral gyrus | 0.191 | GCN |
| 4 | Middle temporal gyrus | 0.165 | GCN |
| 5 | Superior frontal gyrus | 0.143 | GCN |
| 1 | Fusiform gyrus | 0.240 | GIN |
| 2 | Precentral gyrus | 0.210 | GIN |
| 3 | Superior parietal lobule | 0.200 | GIN |
| 4 | Middle frontal gyrus | 0.173 | GIN |
| 5 | Supplementary motor area | 0.140 | GIN |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, Z.; Lin, Z.; Li, S.; Wang, Y.; Zhong, W.; Wang, X.; Xin, J. Dynamic Multi-Task Graph Isomorphism Network for Classification of Alzheimer’s Disease. Appl. Sci. 2023, 13, 8433. https://doi.org/10.3390/app13148433
Wang Z, Lin Z, Li S, Wang Y, Zhong W, Wang X, Xin J. Dynamic Multi-Task Graph Isomorphism Network for Classification of Alzheimer’s Disease. Applied Sciences. 2023; 13(14):8433. https://doi.org/10.3390/app13148433
Chicago/Turabian StyleWang, Zhiqiong, Zican Lin, Shuo Li, Yibo Wang, Weiying Zhong, Xinlei Wang, and Junchang Xin. 2023. "Dynamic Multi-Task Graph Isomorphism Network for Classification of Alzheimer’s Disease" Applied Sciences 13, no. 14: 8433. https://doi.org/10.3390/app13148433
APA StyleWang, Z., Lin, Z., Li, S., Wang, Y., Zhong, W., Wang, X., & Xin, J. (2023). Dynamic Multi-Task Graph Isomorphism Network for Classification of Alzheimer’s Disease. Applied Sciences, 13(14), 8433. https://doi.org/10.3390/app13148433







