Influence of the Polymer and Solvent Variables on the Nanoencapsulation of the Flavonoid Quercetin: Preliminary Study Based on Eudragit® Polymers
Abstract
Featured Application
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethical Statement
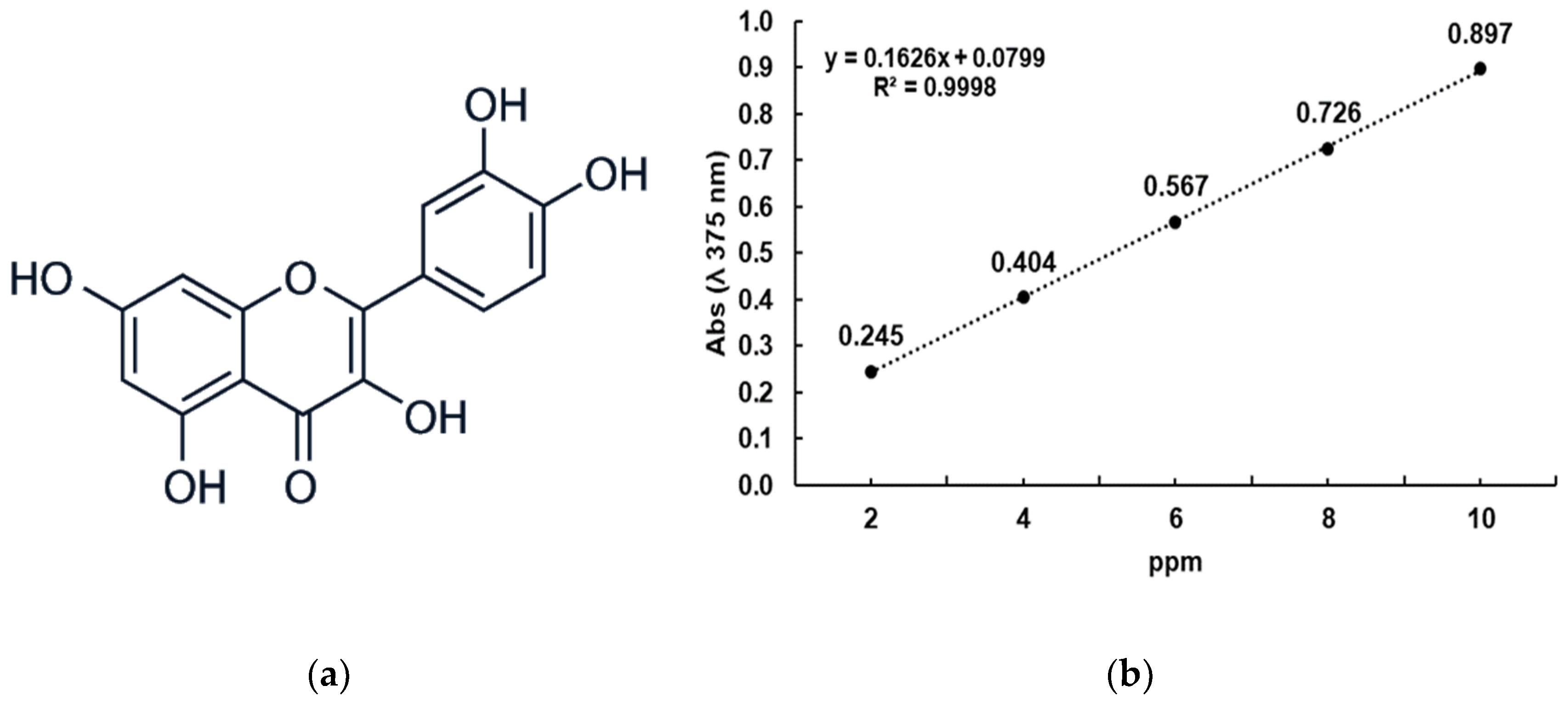
2.2. Chemicals and Reagents
2.3. Polymeric Nanoparticles Formulation
2.3.1. Eudragit® PNP Synthesis
2.3.2. Quercetin Encapsulation into PNPs
2.4. Encapsulation Efficiency
2.5. Size and Morphology of Polymer Nanoparticles
2.6. Hemolytic Activity Test
2.7. Statistical Analysis
3. Results
3.1. Polymeric Nanoparticle Preparation
3.2. Qr-Loaded PNPs
3.3. Morphology of Polymer Nanoparticles
3.4. Determination of the Hemolytic Activity of the PNPs
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Chan, H.-K. Nanodrug particles and nanoformulations for drug delivery. Adv. Drug Deliv. Rev. 2011, 63, 405. [Google Scholar] [CrossRef]
- Pacheco-Ordaz, A.; Sánchez-García, E.; Quintanilla-Licea, R.; Bazaldúa-Rodríguez, A.F.; Pérez-Hernández, R.A.; Hernández-García, M.E.; Báez-González, J.G.; Castro-Ríos, R.; Elizondo-Luévano, J.H.; Chávez-Montes, A. Amoebicidal and trichomonicidal capacity of polymeric nanoparticles loaded with extracts of the plants Curcuma longa (Zingiberaceae) and Berberis vulgaris (Berberidaceae). Rev. Biol. Trop. 2022, 70, 319–331. [Google Scholar] [CrossRef]
- Lee, B.K.; Yun, Y.; Park, K. PLA micro- and nano-particles. Adv. Drug Deliv. Rev. 2016, 107, 176–191. [Google Scholar] [CrossRef]
- Heya, M.S.; Cordero-Díaz, A.; Galindo-Rodríguez, S.A.; Verde-Star, M.J.; Sánchez-García, E.; Villarreal-Villarreal, J.P.; Guillén-Meléndez, G.A. Overcoming tumor and mucosal barriers through active-loaded nanocarriers: Nanoparticles and exosomes. Appl. Nanosci. 2023, 13, 4485–4495. [Google Scholar] [CrossRef]
- Asimuddin, M.; Shaik, M.R.; Adil, S.F.; Siddiqui, M.R.H.; Alwarthan, A.; Jamil, K.; Khan, M. Azadirachta indica based biosynthesis of silver nanoparticles and evaluation of their antibacterial and cytotoxic effects. J. King Saud Univ.-Sci. 2020, 32, 648–656. [Google Scholar] [CrossRef]
- Cetin, M.; Atila, A.; Kadioglu, Y. Formulation and In vitro Characterization of Eudragit® L100 and Eudragit® L100-PLGA Nanoparticles Containing Diclofenac Sodium. AAPS PharmSciTech 2010, 11, 1250–1256. [Google Scholar] [CrossRef]
- Kriplani, P.; Guarve, K. Eudragit, a Nifty Polymer for Anticancer Preparations: A Patent Review. Recent Pat. Anticancer Drug Discov. 2022, 17, 92–101. [Google Scholar] [CrossRef]
- Patra, C.N.; Priya, R.; Swain, S.; Kumar Jena, G.; Panigrahi, K.C.; Ghose, D. Pharmaceutical significance of Eudragit: A review. Future J. Pharm. Sci. 2017, 3, 33–45. [Google Scholar] [CrossRef]
- Chong-Cerda, R.; Levin, L.; Castro-Ríos, R.; Hernández-Luna, C.E.; González-Horta, A.; Gutiérrez-Soto, G.; Chávez-Montes, A. Nanoencapsulated Laccases Obtained by Double-Emulsion Technique. Effects on Enzyme Activity pH-Dependence and Stability. Catalysts 2020, 10, 1085. [Google Scholar] [CrossRef]
- Caddeo, C.; Gabriele, M.; Fernàndez-Busquets, X.; Valenti, D.; Fadda, A.M.; Pucci, L.; Manconi, M. Antioxidant activity of quercetin in Eudragit-coated liposomes for intestinal delivery. Int. J. Pharm. 2019, 565, 64–69. [Google Scholar] [CrossRef]
- D’Andrea, G. Quercetin: A flavonol with multifaceted therapeutic applications? Fitoterapia 2015, 106, 256–271. [Google Scholar] [CrossRef]
- Rodríguez-Garza, N.E.; Quintanilla-Licea, R.; Romo-Sáenz, C.I.; Elizondo-Luevano, J.H.; Tamez-Guerra, P.; Rodríguez-Padilla, C.; Gomez-Flores, R. In Vitro Biological Activity and Lymphoma Cell Growth Inhibition by Selected Mexican Medicinal Plants. Life 2023, 13, 958. [Google Scholar] [CrossRef]
- Cárdenas Garza, G.R.; Elizondo Luévano, J.H.; Bazaldúa Rodríguez, A.F.; Chávez Montes, A.; Pérez Hernández, R.A.; Martínez Delgado, A.J.; López Villarreal, S.M.; Rodríguez Rodríguez, J.; Sánchez Casas, R.M.; Castillo Velázquez, U.; et al. Benefits of Cardamom (Elettaria cardamomum (L.) Maton) and Turmeric (Curcuma longa L.) Extracts for Their Applications as Natural Anti-Inflammatory Adjuvants. Plants 2021, 10, 1908. [Google Scholar] [CrossRef]
- Elizondo-Luévano, J.H.; Castro-Ríos, R.; Sánchez-García, E.; Hernández-García, M.E.; Vargas-Villarreal, J.; Rodríguez-Luis, O.E.; Chávez-Montes, A. In Vitro Study of Antiamoebic Activity of Methanol Extracts of Argemone mexicana on Trophozoites of Entamoeba histolytica HM1-IMSS. Can. J. Infect. Dis. Med. Microbiol. 2018, 2018, 7453787. [Google Scholar] [CrossRef]
- Elizondo-Luévano, J.H.; Hernández-García, M.E.; Pérez-Narváez, O.A.; Castro-Ríos, R.; Chávez-Montes, A. Berberina, curcumina y quercetina como potenciales agentes con capacidad antiparasitaria. Rev. Biol. Trop. 2020, 68, 1241–1249. [Google Scholar] [CrossRef]
- Tienda-Vázquez, M.A.; Melchor-Martínez, E.M.; Elizondo-Luévano, J.H.; Parra-Saldívar, R.; Lara-Ortiz, J.S.; Luna-Sosa, B.; Scheckhuber, C.Q. Antidiabetic Plants for the Treatment of Type 2 Diabetes Mellitus and Associated Bacterial Infections. Processes 2023, 11, 1299. [Google Scholar] [CrossRef]
- Ji, Y.; Lemberg, M.; Prudic, A.; Paus, R.; Sadowski, G. Modeling and analysis of dissolution of paracetamol/Eudragit® formulations. Chem. Eng. Res. Des. 2017, 121, 22–31. [Google Scholar] [CrossRef]
- Sester, C.; Ofridam, F.; Lebaz, N.; Gagnière, E.; Mangin, D.; Elaissari, A. pH-Sensitive methacrylic acid–methyl methacrylate copolymer Eudragit L100 and dimethylaminoethyl methacrylate, butyl methacrylate, and methyl methacrylate tri-copolymer Eudragit E100. Polym. Adv. Technol. 2020, 31, 440–450. [Google Scholar] [CrossRef]
- Ofridam, F.; Lebaz, N.; Gagnière, É.; Mangin, D.; Elaissari, A. Effect of secondary polymer on self-precipitation of pH-sensitive polymethylmethacrylate derivatives Eudragit E100 and Eudragit L100. Polym. Adv. Technol. 2020, 31, 1270–1279. [Google Scholar] [CrossRef]
- Rodríguez-Luis, O.; Verde-Star, J.; González-Horta, A.; Báez-González, G.; Castro-Ríos, R.; Sánchez-García, E.; Chávez-Montes, A. Preparation of polymer nanoparticles loaded with Syzygium aromaticum essential oil: An oral potential application. Bol. Latinoam. Caribe Plantas Med. Aromát. 2020, 19, 65–76. [Google Scholar]
- Elizondo-Luevano, J.H.; Verde-Star, J.; González-Horta, A.; Castro-Ríos, R.; Hernández-García, M.E.; Chávez-Montes, A. In Vitro Effect of Methanolic Extract of Argemone mexicana against Trichomonas vaginalis. Korean J. Parasitol. 2020, 58, 135–145. [Google Scholar] [CrossRef] [PubMed]
- Elizondo-Luévano, J.H.; Gomez-Flores, R.; Verde-Star, M.J.; Tamez-Guerra, P.; Romo-Sáenz, C.I.; Chávez-Montes, A.; Rodríguez-Garza, N.E.; Quintanilla-Licea, R. In Vitro Cytotoxic Activity of Methanol Extracts of Selected Medicinal Plants Traditionally Used in Mexico against Human Hepatocellular Carcinoma. Plants 2022, 11, 2862. [Google Scholar] [CrossRef] [PubMed]
- Fessi, H.; Puisieux, F.; Devissaguet, J.P.; Ammoury, N.; Benita, S. Nanocapsule formation by interfacial polymer deposition following solvent displacement. Int. J. Pharm. 1989, 55, R1–R4. [Google Scholar] [CrossRef]
- Urbán-Morlán, Z. Preparation of Ethyl Cellulose Nanoparticles by Solvent-Displacement Using the Conventional Method and a Recirculation System. J. Mex. Chem. Soc. 2017, 59, 173–180. [Google Scholar] [CrossRef]
- Bilati, U.; Allémann, E.; Doelker, E. Development of a nanoprecipitation method intended for the entrapment of hydrophilic drugs into nanoparticles. Eur. J. Pharm. Sci. 2005, 24, 67–75. [Google Scholar] [CrossRef]
- D’Addio, S.M.; Prud’homme, R.K. Controlling drug nanoparticle formation by rapid precipitation. Adv. Drug Deliv. Rev. 2011, 63, 417–426. [Google Scholar] [CrossRef]
- Wang, Y.; Li, P.; Tran, T.T.D.; Zhang, J.; Kong, L. Manufacturing techniques and surface engineering of polymer based nanoparticles for targeted drug delivery to cancer. Nanomaterials 2016, 6, 26. [Google Scholar] [CrossRef]
- Das, S.; Suresh, P.K.; Desmukh, R. Design of Eudragit RL 100 nanoparticles by nanoprecipitation method for ocular drug delivery. Nanomed. Nanotechnol. Biol. Med. 2010, 6, 318–323. [Google Scholar] [CrossRef]
- Mersmann, A. Crystallization and precipitation. Chem. Eng. Process. Process Intensif. 1999, 38, 345–353. [Google Scholar] [CrossRef]
- Sinha, B.; Müller, R.H.; Möschwitzer, J.P. Bottom-up approaches for preparing drug nanocrystals: Formulations and factors affecting particle size. Int. J. Pharm. 2013, 453, 126–141. [Google Scholar] [CrossRef]
- Murnane, D.; Marriott, C.; Martin, G.P. Developing an environmentally benign process for the production of microparticles: Amphiphilic crystallization. Eur. J. Pharm. Biopharm. 2008, 69, 72–82. [Google Scholar] [CrossRef]
- Shrimal, P.; Jadeja, G.; Patel, S. A review on novel methodologies for drug nanoparticle preparation: Microfluidic approach. Chem. Eng. Res. Des. 2020, 153, 728–756. [Google Scholar] [CrossRef]
- Danaei, M.; Dehghankhold, M.; Ataei, S.; Hasanzadeh Davarani, F.; Javanmard, R.; Dokhani, A.; Khorasani, S.; Mozafari, M. Impact of Particle Size and Polydispersity Index on the Clinical Applications of Lipidic Nanocarrier Systems. Pharmaceutics 2018, 10, 57. [Google Scholar] [CrossRef] [PubMed]
- Horn, D.; Rieger, J. Organic Nanoparticles in the Aqueous Phase-Theory, Experiment, and Use. Angew. Chem. Int. Ed. Engl. 2001, 40, 4330–4361. [Google Scholar] [CrossRef] [PubMed]
- Dillen, K.; Vandervoort, J.; Van den Mooter, G.; Ludwig, A. Evaluation of ciprofloxacin-loaded Eudragit® RS100 or RL100/PLGA nanoparticles. Int. J. Pharm. 2006, 314, 72–82. [Google Scholar] [CrossRef]
- Beck-Broichsitter, M.; Rytting, E.; Lebhardt, T.; Wang, X.; Kissel, T. Preparation of nanoparticles by solvent displacement for drug delivery: A shift in the “ouzo region” upon drug loading. Eur. J. Pharm. Sci. 2010, 41, 244–253. [Google Scholar] [CrossRef]
- Elizondo-Luévano, J.H.; Pérez-Narváez, O.A.; Sánchez-García, E.; Castro-Ríos, R.; Hernández-García, M.E.; Chávez-Montes, A. In-Vitro Effect of Kalanchoe daigremontiana and Its Main Component, Quercetin against Entamoeba histolytica and Trichomonas vaginalis. Iran. J. Parasitol. 2021, 16, 394–401. [Google Scholar] [CrossRef]
- Firuzi, O.; Lacanna, A.; Petrucci, R.; Marrosu, G.; Saso, L. Evaluation of the antioxidant activity of flavonoids by “ferric reducing antioxidant power” assay and cyclic voltammetry. Biochim. Biophys. Acta-Gen. Subj. 2005, 1721, 174–184. [Google Scholar] [CrossRef]
- Gašić, U.; Ćirić, I.; Pejčić, T.; Radenković, D.; Djordjević, V.; Radulović, S.; Tešić, Ž. Polyphenols as Possible Agents for Pancreatic Diseases. Antioxidants 2020, 9, 547. [Google Scholar] [CrossRef]
- Zheng, Y.; Haworth, I.S.; Zuo, Z.; Chow, M.S.S.; Chow, A.H.L. Physicochemical and Structural Characterization of Quercetin-β-Cyclodextrin Complexes. J. Pharm. Sci. 2005, 94, 1079–1089. [Google Scholar] [CrossRef]
- Zhang, Y.; Yang, Y.; Tang, K.; Hu, X.; Zou, G. Physicochemical characterization and antioxidant activity of quercetin-loaded chitosan nanoparticles. J. Appl. Polym. Sci. 2008, 107, 891–897. [Google Scholar] [CrossRef]
- Patel, A.R.; Heussen, P.C.M.; Hazekamp, J.; Drost, E.; Velikov, K.P. Quercetin loaded biopolymeric colloidal particles prepared by simultaneous precipitation of quercetin with hydrophobic protein in aqueous medium. Food Chem. 2012, 133, 423–429. [Google Scholar] [CrossRef] [PubMed]
- Budhian, A.; Siegel, S.J.; Winey, K.I. Haloperidol-loaded PLGA nanoparticles: Systematic study of particle size and drug content. Int. J. Pharm. 2007, 336, 367–375. [Google Scholar] [CrossRef]
- Rathod, L.V.; Kapadia, R.; Sawant, K.K. A novel nanoparticles impregnated ocular insert for enhanced bioavailability to posterior segment of eye: In vitro, in vivo and stability studies. Mater. Sci. Eng. C 2017, 71, 529–540. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Zhang, M.; Tian, Y.; Liu, R.; Wang, Y.; Guo, F.; Gong, Y.; Yan, M. Development, Characterization, and Investigation of In Vivo Targeted Delivery Efficacy of Luteolin-Loaded, Eudragit S100-Coated mPEG-PLGA Nanoparticles. AAPS PharmSciTech 2022, 23, 100. [Google Scholar] [CrossRef]
- Jat, R.; Jain, S.; Singh, S.K.; Gupta, R. Formulation and In vitro—In vivo Evaluation of Quercetin Loaded Eudragit S100 Microspheres. Asian J. Pharm. 2018, 12, 31–37. [Google Scholar]
- Vauthier, C.; Bouchemal, K. Methods for the Preparation and Manufacture of Polymeric Nanoparticles. Pharm. Res. 2009, 26, 1025–1058. [Google Scholar] [CrossRef]
- Date, A.A.; Jain, A.; Khachane, P.; Nagarsenker, M.S. Organic solvent-free approach to single step fabrication of Eudragit nanoparticles using Labrasol. Pharmazie 2010, 65, 733–736. [Google Scholar] [CrossRef]
- Wang, C.; Cui, B.; Guo, L.; Wang, A.; Zhao, X.; Wang, Y.; Sun, C.; Zeng, Z.; Zhi, H.; Chen, H.; et al. Fabrication and Evaluation of Lambda-Cyhalothrin Nanosuspension by One-Step Melt Emulsification Technique. Nanomaterials 2019, 9, 145. [Google Scholar] [CrossRef]
- Elizondo-Luévano, J.H.; Rodríguez-Garza, N.E.; Bazaldúa-Rodríguez, A.F.; Romo-Sáenz, C.I.; Tamez-Guerra, P.; Verde-Star, M.J.; Gomez-Flores, R.; Quintanilla-Licea, R. Cytotoxic, Anti-Hemolytic, and Antioxidant Activities of Ruta chalepensis L. (Rutaceae) Extract, Fractions, and Isolated Compounds. Plants 2023, 12, 2203. [Google Scholar] [CrossRef]
- De La Cruz-Jiménez, L.; Hernández-Torres, M.A.; Monroy-García, I.N.; Rivas-Morales, C.; Verde-Star, M.J.; Gonzalez-Villasana, V.; Viveros-Valdez, E. Biological Activities of Seven Medicinal Plants Used in Chiapas, Mexico. Plants 2022, 11, 1790. [Google Scholar] [CrossRef] [PubMed]
- Mombeshora, M.; Mukanganyama, S. Antibacterial activities, proposed mode of action and cytotoxicity of leaf extracts from Triumfetta welwitschii against Pseudomonas aeruginosa. BMC Complement. Altern. Med. 2019, 19, 315. [Google Scholar] [CrossRef] [PubMed]
- Kalegaril, M.; Miguel, M.D.; Dias, J.D.F.G.; Lordelloz, A.L.L.; de Lima, C.P.; Miyazaki, C.M.S.; Zanin, S.M.W.; Verdam, M.C.D.S.; Miguel, O.G. Phytochemical constituents and preliminary toxicity evaluation of leaves from Rourea induta Planch. (Connaraceae). Braz. J. Pharm. Sci. 2011, 47, 635–642. [Google Scholar] [CrossRef]




| Constant Variable | Combinations |
|---|---|
| AP 12 mL | OP volumes: 5, 8, 10, 12, 15, 18, and 20 mL |
| + | |
| PA 50 mg | |
| OP 12 mL | AP volumes: 5, 8, 10, 12, 15, 18, and 20 mL |
| + | |
| PA 50 mg | |
| AP 12 mL | PA volumes: 5, 10, 25, 50, 75, and 100 mg |
| + | |
| OP 12 mL |
| Constant Variables | Combinations |
|---|---|
| AP 12 mL | PVA content in AP: 0.0%, 0.5%, 1.0%, 2.0%, 4.0%, and 8.0% w/v |
| + | |
| OP 12 mL | |
| + | |
| PA 50 mg | |
| + | |
| Qr 5 mg |
| Polymers | % PVA | S (nm) | PDI | % EE |
|---|---|---|---|---|
| EPO | 0.0 | 150.7 ± 3.7d | 0.15 ± 0.01 d | 26.2 ± 0.8 a |
| 0.5 | 106.2 ± 1.4 c | 0.14 ± 0.01 d | 66.6 ± 2.0 b | |
| 1.0 | 104.5 ± 2.2 c | 0.12 ± 0.02 c | 73.6 ± 2.4 b | |
| 2.0 | 80.7 ± 0.8 b | 0.07 ± 0.00 a | 83.4 ± 2.0 c | |
| 4.0 | 73.9 ± 2.5 b | 0.09 ± 0.01 b | 91.1 ± 3.1 cd | |
| 8.0 | 43.6 ± 1.9 a | 0.10 ± 0.01 c | 98.6 ± 1.8 d | |
| ANOVA p | 0.001 | <0.001 | <0.001 | |
| E100 | 0.0 | 201.2 ± 4.3 d | 0.3 ± 0.02 e | 8.5 ± 2.1 a |
| 0.5 | 94.5 ± 1.7 c | 0.13 ± 0.01 d | 81.1 ± 1.2 b | |
| 1.0 | 81.6 ± 3.0 b | 0.09 ± 0.00 c | 81.6 ± 3.3 b | |
| 2.0 | 76.3 ± 3.3 b | 0.08 ± 0.01 bc | 87.2 ± 1.7 b | |
| 4.0 | 38.9 ± 2.0 a | 0.07 ± 0.00 b | 91.5 ± 1.8 bc | |
| 8.0 | 26.1 ± 1.6 a | 0.04 ± 0.00 a | 98.7 ± 3.0 c | |
| ANOVA p | <0.001 | <0.001 | <0.001 | |
| L100 | 0.0 | 186.5 ± 1.7 d | 0.23 ± 0.01 e | 23.9 ± 1.7 a |
| 0.5 | 101.5 ± 1.6 c | 0.19 ± 0.02 d | 48.7 ± 2.1 b | |
| 1.0 | 78.9 ± 2.1 b | 0.16 ± 0.02 c | 52.9 ± 2.7 b | |
| 2.0 | 78.1 ± 2.8 b | 0.16 ± 0.01 c | 53.0 ± 1.9 b | |
| 4.0 | 72.3 ± 3.5 b | 0.12 ± 0.02 b | 76.1 ± 1.6 c | |
| 8.0 | 52.3 ± 2.7 a | 0.08 ± 0.01 a | 78.5 ± 3.4 c | |
| ANOVA p | <0.001 | 0.001 | <0.001 | |
| L100-55 | 0.0 | 222.8 ± 3.4 d | 0.39 ± 0.02 c | 11.2 ± 2.1 a |
| 0.5 | 192.5 ± 1.7 c | 0.15 ± 0.01 b | 82.4 ± 2.4 b | |
| 1.0 | 174.6 ± 5.4 b | 0.15 ± 0.02 b | 87.1 ± 1.6 bc | |
| 2.0 | 168.2 ± 3.2 b | 0.15 ± 0.02 b | 88.9 ± 1.9 bc | |
| 4.0 | 53.1 ± 2.6 a | 0.13 ± 0.00 a | 89.1 ± 3.1 bc | |
| 8.0 | 51.6 ± 1.1 a | 0.13 ± 0.01 a | 96.0 ± 2.7 c | |
| ANOVA p | <0.001 | <0.001 | <0.001 |
| μg/mL | Polymers | Qr | ANOVA p⟊ | |||
|---|---|---|---|---|---|---|
| EPO | E100 | L100 | L100-55 | |||
| 100 | 0.01 ± 0.002 a,1 | 0.01 ± 0.00 a,1 | 0.03 ± 0.01 a,2 | 0.01 ± 0.00 a,1 | 0.01 ± 0.00 a,1 | <0.05 |
| 200 | 0.02 ± 0.002 ab,2 | 0.01 ± 0.00 a,1 | 0.04 ± 0.01 ab,3 | 0.02 ± 0.01 b,2 | 0.01 ± 0.00 a,1 | <0.05 |
| 400 | 0.02 ± 0.001 ab,1 | 0.02 ± 0.00 a,1 | 0.04 ± 0.01 ab,2 | 0.02 ± 0.01 b,1 | 0.11 ± 0.04 b,3 | <0.01 |
| 600 | 0.02 ± 0.002 ab,1 | 0.02 ± 0.00 a,1 | 0.04 ± 0.01 ab,2 | 0.02 ± 0.00 b,1 | 0.20 ± 0.04 c,3 | <0.001 |
| 800 | 0.02 ± 0.004 ab,1 | 0.02 ± 0.00 a,1 | 0.05 ± 0.01 b,2 | 0.02 ± 0.01 b,1 | 0.45 ± 0.12 d,3 | <0.001 |
| 1000 | 0.03 ± 0.003 b,1 | 0.02 ± 0.01 ab,1 | 0.05 ± 0.01 b,2 | 0.02 ± 0.01 b,1 | 0.68 ± 0.10 e,3 | <0.001 |
| ANOVA p | <0.001 | <0.05 | <0.001 | <0.01 | <0.001 | - |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Elizondo-Luevano, J.H.; Castro-Ríos, R.; Parra-Saldívar, R.; Larqué-García, H.; Garza-Tapia, M.; Melchor-Martínez, E.M.; Chávez-Montes, A. Influence of the Polymer and Solvent Variables on the Nanoencapsulation of the Flavonoid Quercetin: Preliminary Study Based on Eudragit® Polymers. Appl. Sci. 2023, 13, 7816. https://doi.org/10.3390/app13137816
Elizondo-Luevano JH, Castro-Ríos R, Parra-Saldívar R, Larqué-García H, Garza-Tapia M, Melchor-Martínez EM, Chávez-Montes A. Influence of the Polymer and Solvent Variables on the Nanoencapsulation of the Flavonoid Quercetin: Preliminary Study Based on Eudragit® Polymers. Applied Sciences. 2023; 13(13):7816. https://doi.org/10.3390/app13137816
Chicago/Turabian StyleElizondo-Luevano, Joel H., Rocío Castro-Ríos, Roberto Parra-Saldívar, Horacio Larqué-García, Marsela Garza-Tapia, Elda M. Melchor-Martínez, and Abelardo Chávez-Montes. 2023. "Influence of the Polymer and Solvent Variables on the Nanoencapsulation of the Flavonoid Quercetin: Preliminary Study Based on Eudragit® Polymers" Applied Sciences 13, no. 13: 7816. https://doi.org/10.3390/app13137816
APA StyleElizondo-Luevano, J. H., Castro-Ríos, R., Parra-Saldívar, R., Larqué-García, H., Garza-Tapia, M., Melchor-Martínez, E. M., & Chávez-Montes, A. (2023). Influence of the Polymer and Solvent Variables on the Nanoencapsulation of the Flavonoid Quercetin: Preliminary Study Based on Eudragit® Polymers. Applied Sciences, 13(13), 7816. https://doi.org/10.3390/app13137816








