3.1. Morphology and Structure of the Films
From the cross-section of the pure tungsten oxide WO
3|comp thin films prepared from the precursor solution without additives (
Figure S1c), a full coverage of the substrate by WO
3 with a film thickness of around 100 nm can be observed, thinner than the reported 250 nm in [
14]. This could be caused by differences in the details of experimental conditions during spin-coating. The samples reveal a smooth and compact film structure with less than 2 nm RMS surface roughness (WO
3|comp) on the crystalline FTO substrate as also observed in [
14]. While the addition of PEG 400 or PEG 600 in the volume ratio of 3:10 or 5:10 into the precursor solution led to inhomogeneous films, the addition of PEG in the volume ratio of up to 1.5:10 provided films of WO
3|µp400 and WO
3|µp600 with a smooth coverage of the substrate (
Figure 1a and
Figure S1b) with RMS surface roughness similar to the WO
3|comp films (
Figure S1a). The cross-section of these films indicates a grainy film structure and film thicknesses of about 130–150 nm (
Figure 1d and
Figure S1d). Such a grainy film structure has been also observed for WO
3 thin films prepared via spin-coating from a peroxotungstic acid precursor solution with PEG 20,000 as structure-directing agent and annealed at 300 °C [
30]. Using PIB
50-
b-PEO
45 as structure-directing agent results in around 200–230 nm thick WO
3|mp films (
Figure 1e) with isolated spherical mesopores of around 10–20 nm diameter (
Figure 1b,e). These pore sizes are comparable to the ones obtained for mesoporous titanium oxide using a similar block copolymer [
35,
38]. Films prepared with PIB
50-
b-PEO
45 as additive were light-brownish in color, indicating some remaining carbon within the films. The combination of PEG 600 and PIB
50-
b-PEO
45 as structure-directing agents led to 230–250 nm thick WO
3|µmp films with interconnected mesopores (
Figure 1c,f).
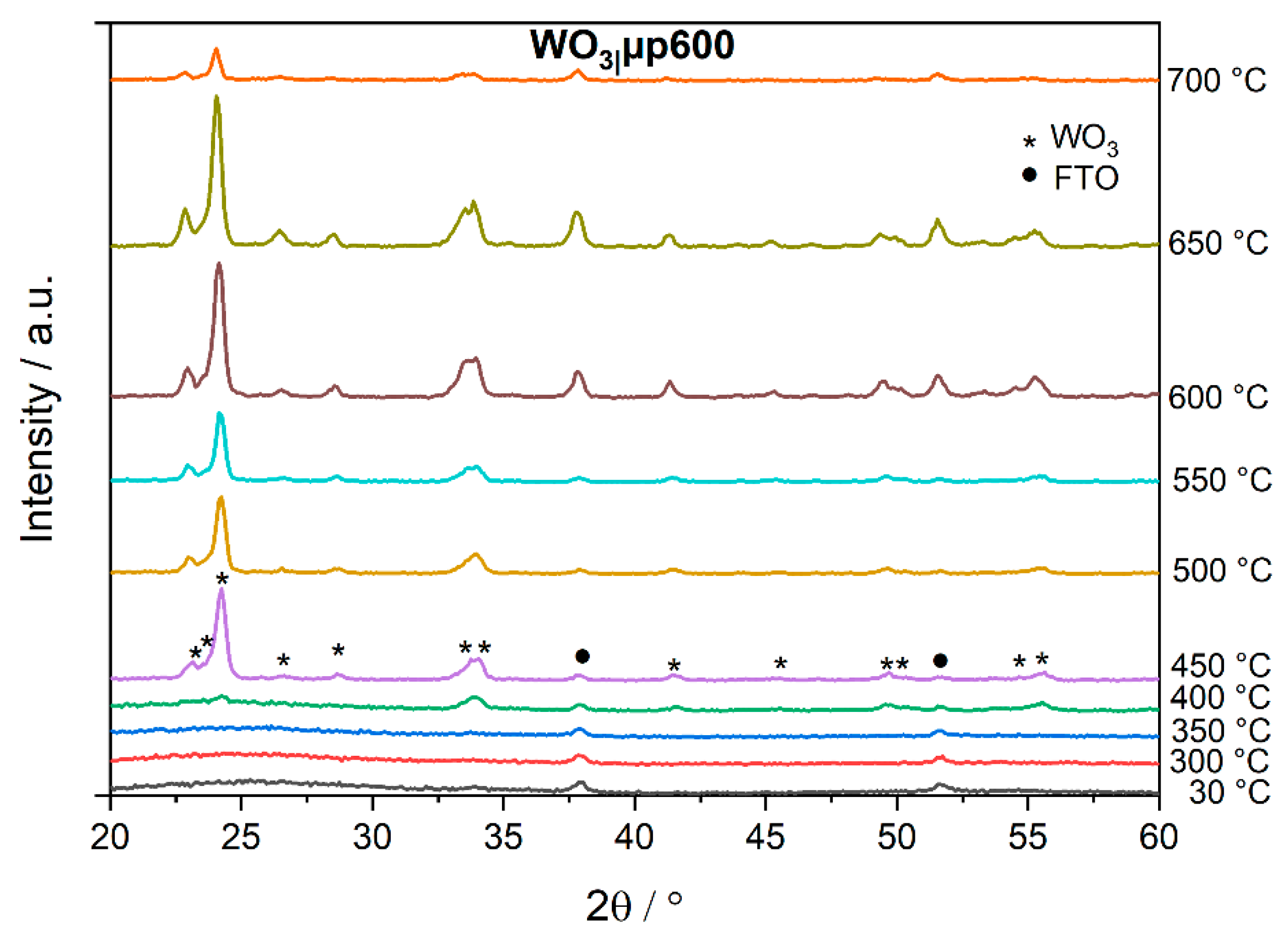
The amorphous character of the films which were annealed at temperatures
T ≤ 300 °C is revealed by GIXRD at room temperature (30 °C) as depicted in
Figure 2 for a WO
3|µp600 film. No reflexes between 20–60° are observable which would be characteristic of crystalline WO
3. All other reflexes correspond to FTO as confirmed by a GIXRD measurement of a bare FTO glass substrate. Annealing the film during the GIXRD analysis showed the transition of an amorphous film into a crystalline film. At 400 °C, reflexes assigned to triclinic WO
3 (JCPDS card 83-0947) start to appear and become more obvious from 450 °C on. For WO
3|comp as well as for the WO
3|µp400, WO
3|mp and WO
3|µmp films, similar series were obtained, indicating the presence of triclinic WO
3 only after annealing the samples at 450 °C (
Figure S2). The crystallite size of the films annealed at 450 °C was estimated from the reflexes at 2Θ around 23° and 42° using the Scherrer equation [
41,
42,
43]. An average crystallite size of around 21 nm was obtained which is well in line with the crystallite size reported in [
44] for WO
3 films prepared by dip-coating and annealed at 500 °C. Zhao et al. [
14] and Wu et al. [
30] have also obtained triclinic WO
3 from peroxotungstic acid solutions. At 600 °C and 650 °C, the relative intensity of the substrate signals increased, presumably caused by thermal expansion of the sample leading to a higher contribution from the substrate. At 700 °C, the glass of the substrate started to melt leading to a decrease of the intensity of all reflexes.
The amorphous character of the WO
3 thin films annealed at temperatures
T ≤ 300 °C was confirmed by Raman spectroscopy. In the Raman spectra (
Figure S3) measured for the films prepared without and with PEG at
T ≤ 300 °C a broad peak at 770 cm
−1 was observed caused by vibrations of W
6+-O bonds [
12,
45] while a relatively sharp peak at 950 cm
−1 reflects the stretching of W
6+=O bonds characteristic for amorphous WO
3 [
12,
45,
46]. For the WO
3|µp600 thin film, another broad peak at 220 cm
−1 was noticed which might correspond to the bridging of O-W
4+-O bonds and indicates the presence of W
4+ ions and oxygen vacancies within the films [
12,
45].
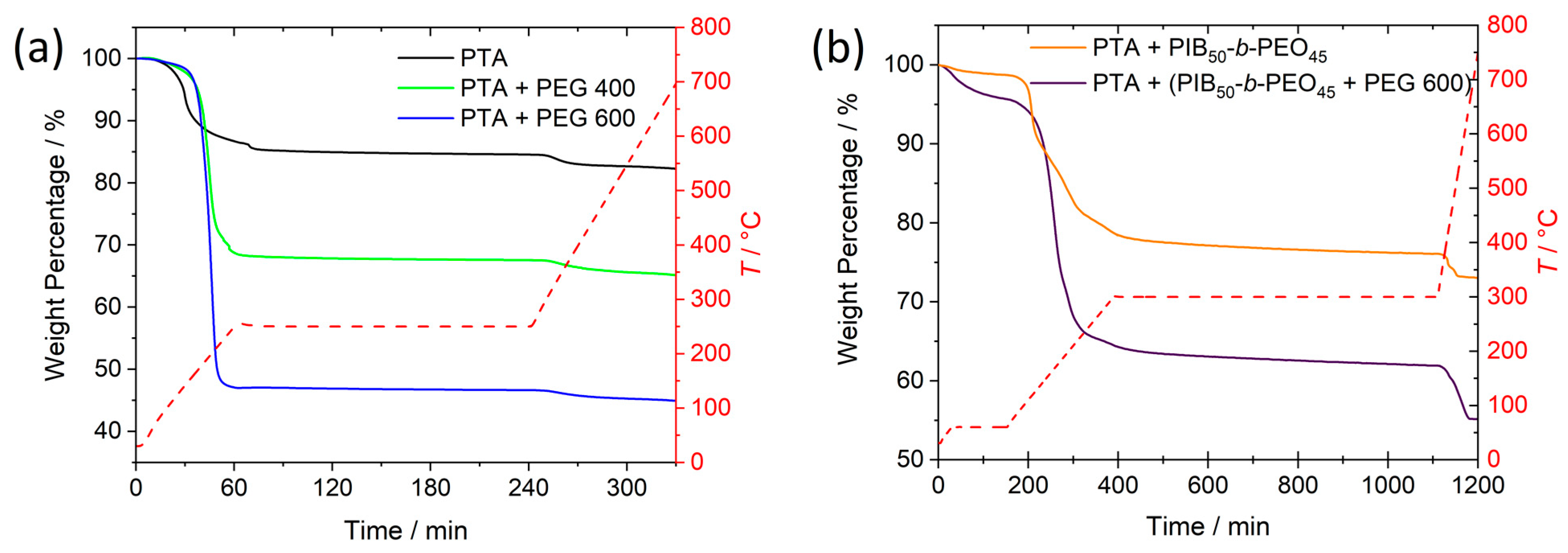
Thermogravimetry (TG) of the dried precursor, resembling films following spin-coating, was performed to monitor the decomposition of the polymers during heat treatment and to detect possible phase transformations of WO
3. In
Figure 3a, the TG curves for the dried peroxotungstic acid precursor solutions with and without PEG are shown. Starting from room temperature up to a temperature of 100 °C, a small loss in mass of around 1% followed by a larger loss from 100 °C to 200 °C was observed caused by the desorption of water and organic solvents as well as the transformation of peroxotungstic acid to WO
3 [
14,
27,
29]. For the precursors with PEG, mainly its decomposition contributed to this mass loss [
27]. As expected, the drop in mass from room temperature to 250 °C is larger for the precursor containing PEG 600 since a larger amount of polymer is present. Within the three hours of treatment at 250 °C, the mass of the precursor without additives decayed by 2% whereas the precursors containing PEG 400 or PEG 600 reduced their mass by only 1% or 0.4%. All three precursors quickly stabilized indicating that keeping them for about 1 h at 250 °C is sufficient for the preparation of amorphous WO
3 thin films. The subsequent small loss of mass (2%) upon further heating to 700 °C can be assigned to the phase transition of amorphous to crystalline WO
3 [
14,
27,
29], which was independently proven by XRD (
Figure 2 and
Figure S2). The TG curves for the precursors containing the block copolymer PIB
50-
b-PEO
45 or PIB
50-
b-PEO
45 in combination with PEG 600 (
Figure 3b) follow the same trend upon heating to 300 °C. At this temperature, however, a mass loss of about 3% was observed over 12 h, indicating continued decomposition of the polymer. Upon subsequent heating to 800 °C, a significant mass loss of another 3% (WO
3|mp) or 7% (WO
3|µmp) was observed consisting of both, crystallization of WO
3 [
27] and completion of polymer decomposition in the case of WO
3|µmp. For WO
3|mp, however, the mass loss of the precursor is similar to the one of the precursor without any additives confirming that residuals of the polymer were still present in the films. Since amorphous WO
3 thin films were aimed at, a temperature not higher than 300 °C (
Figure 2 and
Figure S2) had to be chosen for the heat treatment of the films and it cannot be excluded that the WO
3|mp thin films might still contain some polymer fragments. Comparing the initial loss of mass for the different precursors reveal that the precursor without any additives and the one containing PIB
50-
b-PEO
45 have lost about the same amount of mass (15–18%) up to a temperature of 250 °C while the precursors with PEG 600 have lost more than half of their original mass. The reason for the similar decrease in mass for the pure precursor and the one with PIB
50-
b-PEO
45 can be explained by the small polymer content and its only partial decomposition. While for the precursors with PIB
50-
b-PEO
45, around 37 mg of PIB
50-
b-PEO
45 was added in 1 mL precursor solution, the precursor solution with PEG 600 in a volume ratio of 1.5:10 contained 165.5 mg PEG 600 in 1 mL precursor solution. The observed larger initial loss of mass is explained by almost complete decomposition of PEG at 250 °C.
3.2. Elemental Composition, Density, Surface Area and Porosity
To verify that WO
3 and not any other related compound was obtained when using the precursor solutions containing different polymers as additives, the O:W ratio of the films was determined by EDX (
Table 2, (O:W)
EDX). Since the tungsten oxide films are thinner than 300 nm and the penetration depth of EDX into the sample is in the micrometer range [
47] emission from the substrate has to be accounted for. The spectra revealed the presence of tin in addition to tungsten and oxygen as well as some carbon contamination. Assuming a ratio of O:Sn = 2 for the FTO substrate, meaningful values of (O:W)
EDX in the films can still be calculated. At first glance, (O:W)
EDX was found to be around 3 for all the films (
Table 2) which can be considered a confirmation that WO
3 was obtained from the different precursor solutions. However, (O:W)
EDX decreases for the films prepared with PEG indicating the presence of a higher concentration of oxygen vacancies. It has been reported before that PEG can inhibit the crystallization of the films and might lead to formation of oxygen-deficient WO
3 [
48]. For the WO
3|µmp films, (O:W)
EDX deviates in a larger range compared to the other samples. Thus, the presence of mesopores interconnected by micropores (
Figure 1c,f) might lead to locally differing transmission of X-rays emitted from Sn in FTO within a given sample area and, therefore, to larger variations in (O:W)
EDX. Further, for the WO
3|mp films, the amount of carbon detected in the films was higher directly indicating that polymer fragments were still present in the films.
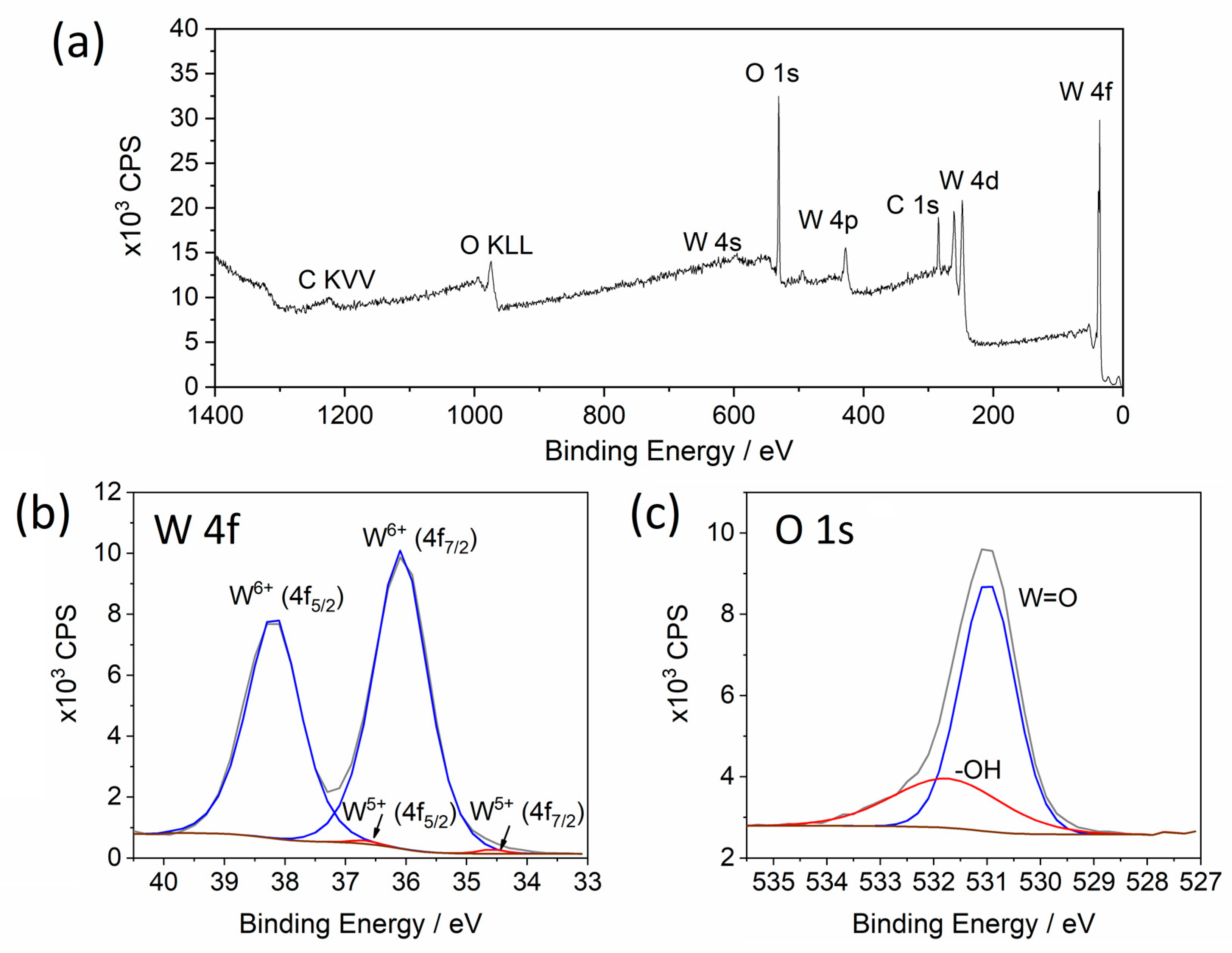
X-ray photoelectron spectroscopy (XPS) was performed to complement the elemental analysis by EDX and, in particular, to obtain direct insight into the W oxidation state in pristine films. The overview spectra, which are quite similar for all the films (
Figure 4a and
Figures S4a–S7a), only show the expected elements such as tungsten and oxygen as well as some carbon impurities, to similar extent in all samples. The W 4f spectra (
Figure 4b and
Figures S4b–S7b) exhibit a doublet at binding energies around 38 eV and 36 eV, which corresponds to W
6+ 4
f5/2 and W
6+ 4
f7/2 states [
14,
24,
49,
50]. The small shoulders at lower binding energies of about 36.4 eV and 34.2 eV point out the presence of W
5+ 4
f5/2 and W
5+ 4
f7/2 states assigned to defects in the film surface [
24,
49]. The positions of these peaks are in good accordance with the ones reported for WO
3 thin films prepared via sol–gel processes [
14,
24] or sputter deposition [
50]. The O 1s spectra (
Figure 4c and
Figures S4c–S7c) show a main peak around 530.8 eV which can be assigned to the W=O bonds [
24]. The shoulder with less intensity at higher binding energy of about 532.0 eV can be assigned to W–OH groups [
24,
51].
From the peak areas of the O 1s spectra and the W 4f spectra, the O:W ratio at the surface (
Table 3, (O:W)
XPS) was determined [
52]. The values of (O:W)
XPS are higher than the (O:W)
EDX values (
Table 2). The reason for this lies in the surface sensitivity of XPS as opposed to EDX [
47] leading to a higher contribution of hydroxyl groups at the surface and therefore to higher values for (O:W)
XPS. However, the trend of a decreasing (O:W)
EDX with PEG as precursor additive was confirmed by (O:W)
XPS (
Table 3). In the films prepared with any of the additives but in particular for WO
3|µp400 and WO
3|µp600, a higher concentration of W
5+ was seen when compared to WO
3|comp (
Table 4). Hence, as reported in [
48], PEG can lead to porous films with more defects and, in particular, more oxygen vacancies, represented by the existence of W
5+ states.
The C 1s signal seems to be dominated by adventitious carbon and no conclusion can be drawn about any residual polymer fragments since even for the WO
3|comp film prepared without any additives a larger amount of carbon was detected than for the WO
3|mp film (
Table 3), for which residual carbon was apparent in the color and in EDX. Therefore, the amount of carbon detected by XPS cannot give any evidence for polymer fragments remaining in the films.
During the stepwise etching with Ar
+, the amount of W
6+ states for all the films decreased while the amount of W
5+ states increased and subsequently even W
4+, and tungsten in even lower ionization states (W
x+) and metallic W
0 states arose indicating the stepwise reduction of WO
3 under Ar
+-bombardment even up to metallic tungsten as shown in
Figure S8. These observations are well in line with the results obtained for Ar
+ bombardment of WO
3 films prepared via thermal evaporation [
49].
Film characteristics derived from wet chemical analysis and profilometry compared to those independently obtained from the analysis of ellipsometry are summarized in
Table 5.
ρ of the WO
3|comp films was found similar to that reported for bulk WO
3 (
= 7.2 g cm
−3 [
53]). By addition of PEG or PIB
50-
b-PEO
45 as structure-directing agents,
ρ of the prepared films decreased, according to the strategy. The porosity
Pchem derived from microwave plasma atomic emission spectroscopy of the films dissolved in NH
3 solution increased according to the decreasing
ρ of the films (
Table 5). The porosity was independently calculated from the ellipsometry data, yielding
PE, which directly confirmed the range of porosity of the samples, but with a trend to slightly lower values than
Pchem (
Table 5). For the ellipsometric analysis, the films were prepared on smooth silicon wafers as opposed to rough FTO substrates. Such difference and the fundamentally different method of analysis can well explain the small differences between
d and
dE and the small systematic difference between
Pchem and
PE. Both methods, however, independently yield the same trend of increasing porosity when using the polymers as structure-directing agents with
P(WO
3|µp400) <
P(WO
3|µp600) <<
P(WO
3|mp) <<
P(WO
3|µmp). The combination of PIB
50-
b-PEO
45 and PEG 600 as additives clearly provided the highest porosity as already revealed by SEM (
Figure 1c).
The specific surface area
SBET of the as-prepared films was analyzed from the BET plots obtained from Kr physisorption (77 K) shown in
Figure S9. The WO
3|mp sample was annealed at 550 °C for 1 h before the BET analysis to remove any residual polymer and obtain a measure for the intrinsic surface area of WO
3, albeit transformation to crystalline WO
3 under such conditions. For the WO
3|comp films prepared without additives, no significant
SBET could be determined, as expected for a compact thin film. For the WO
3|µp600 films, a significant surface area of around 3 m
2/g was obtained, comparable to the surface area reported for WO
3 powder (~2 m
2/g) [
54]. Such surface area is compatible with the presence of micropores between grains of the films (
Figure 1d). The values of
SBET of the WO
3|mp films and WO
3|µmp films turned out in the same range as reported for WO
3 nanoflakes (~10 m
2/g [
55]) and mesoporous WO
3 (~32 m
2/g [
31]).
SBET ≅ 10 m
2/g determined for the WO
3|mp films thereby confirmed the presence of mesopores within the films but the considerably higher
SBET ≅ 20 m
2/g of the WO
3|µmp films shows the significantly increased accessibility of the mesopores by the simultaneous presence of micropores serving as interconnecting channels. These
SBET values appear quite small, compared to the surface area and pore volume usually measured for templated metal oxides. Note that the BET surface area is referred to the mass and that WO
3 has a quite high density (ca. 4.2–7.2 g cm
−3), thus resulting in lower
SBET values compared to a material with identical porosity, but smaller skeleton density, e.g., SiO
2 (ρ ≅ 2.2 g·cm
−3 [
53]). For instance, a value of
SBET ≅ 20 m
2/g (WO
3|µmp), assuming an identical pore space, translates into a value of
SBET ≅ 65 m
2/g for a corresponding SiO
2 material, which is in accordance with a network of connected spherical mesopores of ca. 20 nm in diameter.
3.3. Redox Characteristics and Intercalation of Li+
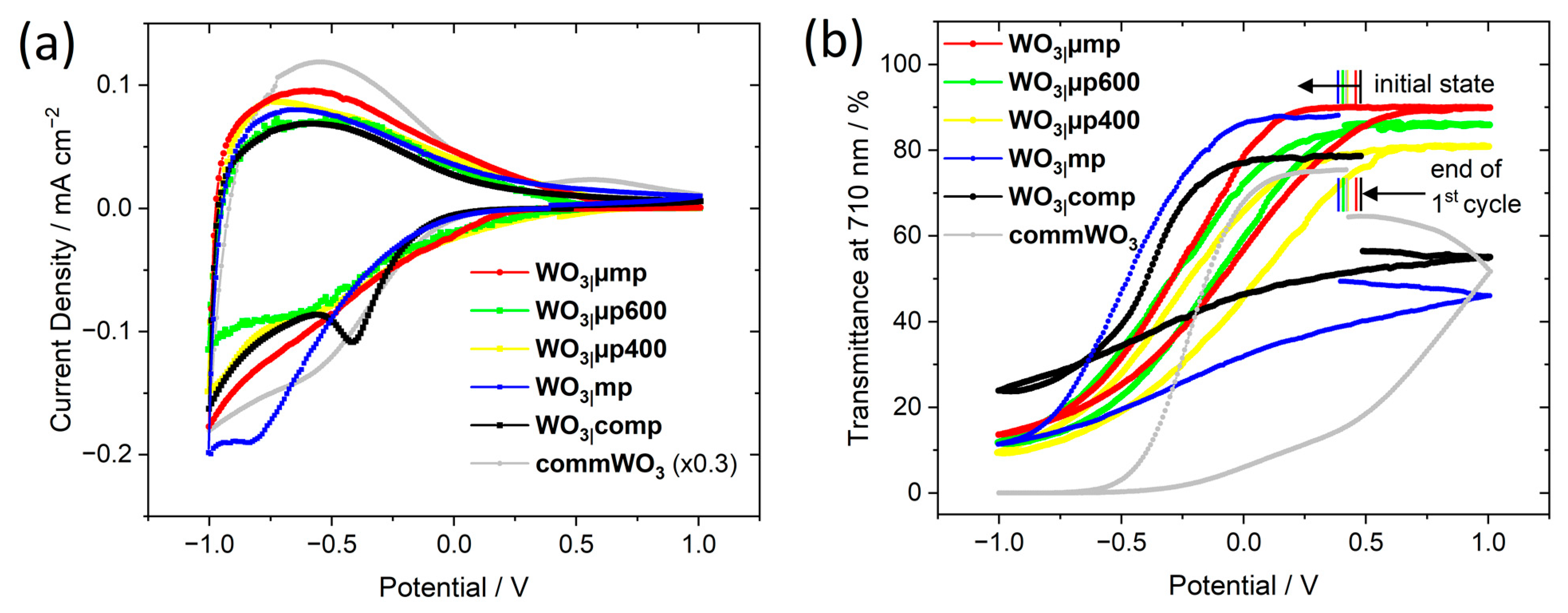
In order to analyze the electrochromic reduction reaction and the diffusion of Li
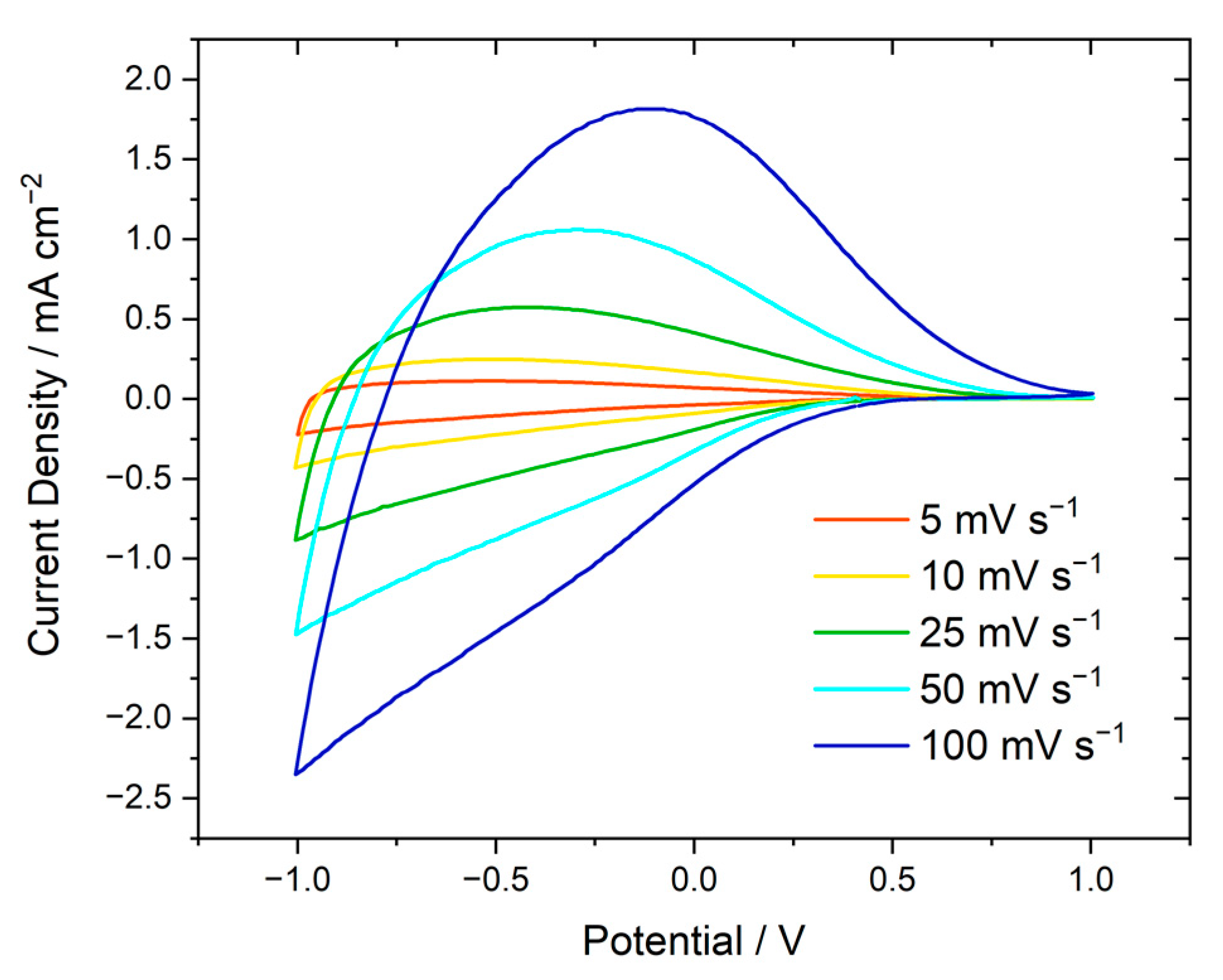
+ ions in the films, cyclic voltammetry (CV) was performed at different scan rates as shown for a WO
3|µmp film in
Figure 5 and for all other types of films in
Figure S10. Following a CV curve with a just slightly different shape during a first conditioning cycle (
Figure 6a), reproducible CV curves were obtained at each scan rate representing a reversible intercalation and deintercalation of Li
+ upon reduction and reoxidation as expected for amorphous WO
3 films [
7].
The reversibility
K of the bleaching and coloration process can be determined from the ratio
qout/
qin of the extracted and inserted charge densities [
56]. During the first conditioning cycle (
Table 6) and especially for the WO
3|comp and WO
3|mp films, the reversibility is rather low indicating that not all inserted ions can be deintercalated but are trapped within the films. Slow or irreversible movement in compact WO
3 can be expected. For the porous WO
3|mp films, however, this originally came unexpected and points at partial pore filling by residual polymer or polymer fragments, as also concluded from a brownish color after annealing during film preparation and relevant mass losses at higher temperature during TG analysis. For the WO
3|µp400, WO
3|µp600 and WO
3|µmp films, the reversibility is close to 100% confirming that only a few ions are trapped within the films [
39,
57]. Hence, diffusion in the microporous network and in the absence of residual polymer fragments assures complete extraction of Li
+ even during the first bleaching process. However, from the second cycle on, all the films showed a high reversibility which means that less ions were trapped inside the films and that the sites where ions might be trapped were already occupied during the first coloration. In this respect, the films are similar to the commercially sputtered commWO
3 film.
The anodic peak current densities
jp,
a and the cathodic current densities at negative scan reversal
jr,
c (
Figure 5 and
Figure S10) were plotted against the square root of the scan rate
v1/2 following the Randles Ševčík equation at room temperature
where
n is the number of electrons transferred in the reaction (1 in the present case [
14]) and
c is the concentration of Li
+ in the bulk of the electrolyte [
14,
56] to determine two approximations of the effective diffusion coefficient of Li
+ and electrons in the films,
D =
Da,CV in the anodic branch and
D =
Dc,CV in the cathodic branch. Diffusion of Li
+ in the electrolyte can be considered much faster than that in the films, and it has not to be considered separately. The average values of the effective diffusion coefficients determined for a given type of sample are shown in
Table 7. The values of the effective diffusion coefficient are in the range of 10
−11–10
−9 cm
2·s
−1, similar to the values reported for sol–gel derived [
14,
29,
58] or sputter-deposited [
59] WO
3 thin films. Slightly larger effective diffusion coefficients were found for WO
3|µp400 and WO
3|µp600 than for WO
3|comp. Obviously, the ions can diffuse more easily through the film network along the microporous pathways available within the films prepared with PEG 400 or PEG 600. For WO
3|mp, on the other hand, an effective diffusion coefficient even smaller than for WO
3|comp was obtained. Despite the mesopores detected in SEM, the remaining polymer fragments of PIB
50-
b-PEO
45 within the films seem to significantly attenuate the diffusion of Li
+. An effective diffusion coefficient about 6 times higher than that for WO
3|comp, WO
3|µp400 or WO
3|µp600 was observed in WO
3|µmp using a combination of both, PEG and PIB
50-
b-PEO
45 as structure-directing agents. This combination provided films with similarly fast diffusion of Li
+ as obtained for commWO
3 (
Table 7).
3.4. Electrochromic Switching Characteristics
To investigate the switching processes of the films, their optical transmittance
Tλ was measured
in operando during CV analysis. As expected, the reduction of WO
3 and the intercalation of Li
+ led to a decrease of
Tλ (
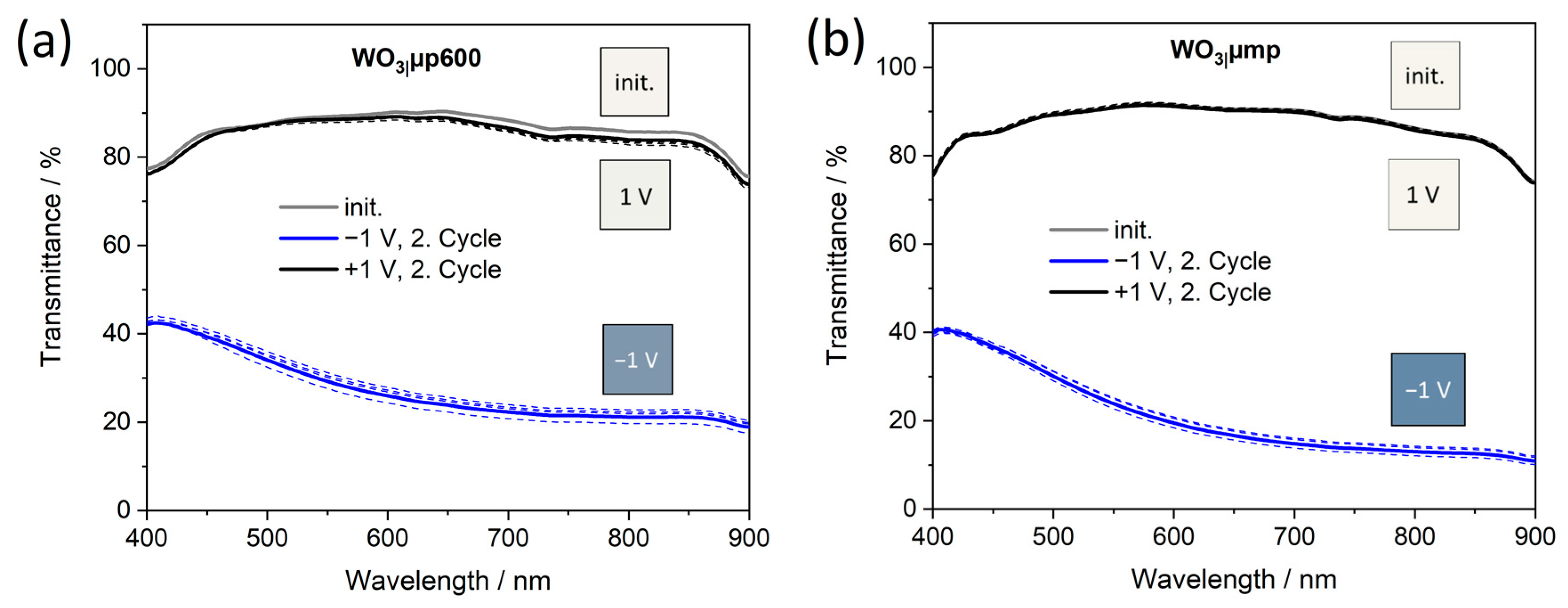
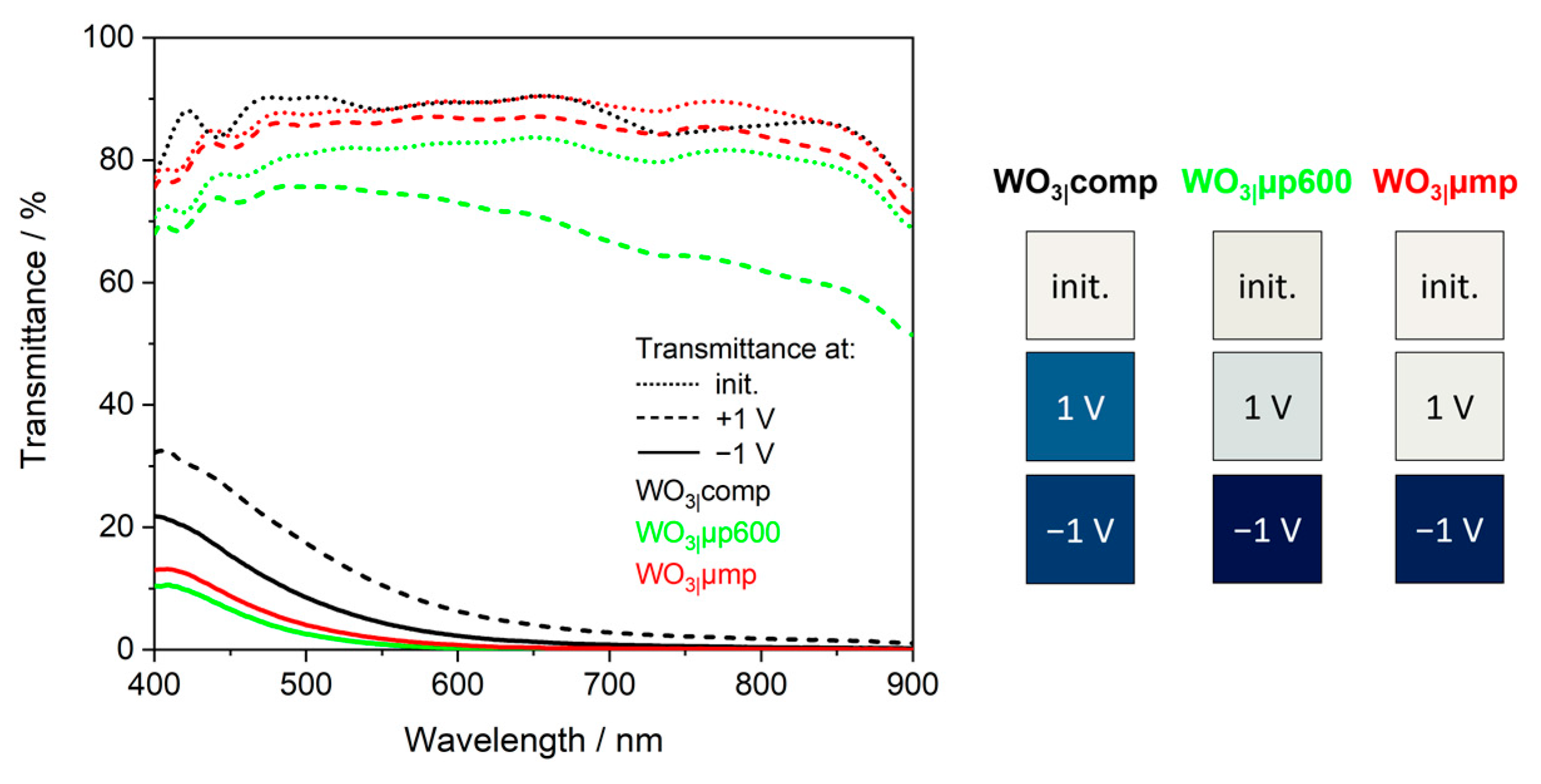
Figure 6b) and deeply blue-colored films as shown by the transmittance spectra at a potential of −1.0 V (
Figure 7 and
Figure S11) and the color impressions, which were calculated as reported in [
60].
By reoxidation of WO
3 and deintercalation of the ions,
Tλ reversibly increased [
1,
6]. For the WO
3|comp and the WO
3|mp films (
Figure S11b,c),
Tλ in the bleached state was found lower compared to the initial state indicating partial irreversibility and the presence of trapped ions [
39,
57] within the films. This is in accordance with their slightly lower reversibility
K1 (
Table 6). The WO
3|µp400, WO
3|µp600 and WO
3|µmp films (
Figure 7 and
Figure S11a) could be fully bleached reaching transmittance characteristics close to the initial state. Facile deintercalation rather than trapping of ions as already revealed by the high reversibility (
Table 6) is thereby confirmed.
Comparing the change in transmittance at a wavelength of 710 nm upon reduction and reoxidation of the films (
Table 8) reveals that for WO
3|µp400, WO
3|µp600 and WO
3|µmp films the transmittance modulation ∆
T710nm is larger than for the WO
3|comp or WO
3|mp films. These observations correlate well with the values of the effective diffusion coefficient (
Table 7). Residual polymer fragments in the WO
3|mp films might possibly hinder the diffusion of the ions within the mesoporous film structure leading to a change in transmittance comparable to compact films. Facile diffusion of ions supported by micropores present in the films prepared in the presence of PEG allows a high transmittance modulation, which has been also observed earlier for WO
3 prepared by dip-coating from similar precursor solutions [
27]. The combination of PEG 600 with PIB
50-
b-PEO
45 led to films reaching even higher values for the transmittance modulation compared to the films prepared with PEG only (
Table 8) indicating facilitated diffusion of ions through the film network composed of interconnected mesopores and pore clusters already revealed by SEM (
Figure 1f). For the WO
3|comp and WO
3|mp films, the transmittance started to change during the intercalation step at a more negative potential of around −0.25 V compared to films of WO
3|µp400, WO
3|µp600 and WO
3|µmp (
Figure 6b). Such a delayed change in transmittance indicates a slower initial intercalation of the ions within the WO
3|comp and WO
3|mp films. For both films, the CV curves at the first cycle (
Figure 6a) also stay at smaller current and show a small peak at around −0.4 V and −0.7 V, respectively. Such characteristics are expected for a hindered reaction, typically observed for crystalline WO
3 films [
33] but observed here for the amorphous WO
3|comp and WO
3|mp films, indicative of an initial barrier for ion intercalation in line with the compact film structure or the presence of remaining polymer fragments. Nevertheless, from −0.25 V to −1.0 V the slope of the transmittance is comparable for all the films. During the bleaching process, however, a significantly steeper slope of the transmittance–potential curves can be observed for films of WO
3|µp400, WO
3|µp600 and WO
3|µmp suggesting a significantly more facile deintercalation of Li
+ through the porous film network.
Another important parameter for the comparison of switching characteristics of the electrochromic films is provided by the coloration efficiency
CE. It is defined as the change in optical density ∆
OD with the intercalated charge density
q (see Equation (2)) [
2,
7].
CE710nm of the films at 710 nm, a wavelength for which the change was most significant, was determined from a linear fit in a plot of ∆
OD = log
Tbleach − log
Tcol over
q, the charge inserted during the switching process.
CE710nm for the WO
3|µp400, WO
3|µp600 and WO
3|µmp films was found considerably higher than for WO
3|comp or WO
3|mp (
Table 8). Less charge is needed to allow a high transmittance modulation for the films with interconnected micropores compared to the compact or just mesoporous films. Such enhancement of
CE by using appropriate structure-directing agents is in good accordance with earlier reports [
27,
31]. The highest obtained values for ∆
T710nm and
CE710nm for the porous films were reached for WO
3|µmp, similar or even better than those measured for commWO
3, the commercial benchmark sample, speaking in favor of the present films with a combination of mesopores interconnected by micropores. Interestingly, the
CE values reported here are significantly higher than those previously reported for WO
3 films with ordered spherical mesopores caused by a similar block copolymer, and also similar thickness [
34]. The observed higher
CE in the present study is indicative of differences in the pore space: the mesoporous films reported in Sallard et al. [
34] were prepared without additional PEG and, thus, without the improved connectivity between the spherical mesopores in WO
3|µmp. Hence, this comparison clearly supports the need for connecting micropores to maximize the electrochromic response.
CV measurements were also performed for WO
3|comp and WO
3|µp600 following their annealing at temperatures higher than 250 °C (
Table S1). For the films calcined at 350 °C,
Da,CV was found in the same order of magnitude as for WO
3|comp calcined at 250 °C (
Table 7) and also the values of ∆
T710nm and ∆
CE710nm lie in the range of the upper limit of the obtained values for the films annealed at 250 °C (
Table 8). These observations confirm facile diffusion of Li
+ in amorphous WO
3 that by GIXRD was observed to persist up to 350 °C (
Figure 2 and
Figure S2). After calcination at
T ≥ 450 °C, the values of ∆
T710nm and
CE710nm decreased and were found close to the lower limit of the values for the films calcined at 250 °C and a drastic change can be clearly seen for the effective diffusion coefficients, which are up to two orders of magnitude smaller than
Da,CV determined after annealing at 250 °C. Slow diffusion in crystalline films formed after calcination at
T ≥ 400 °C (GIXRD,
Figure 2 and
Figure S2) confirmed the disadvantage of a crystalline film structure for electrochromic performance as observed earlier [
14,
61,
62]. However, the direct comparison of WO
3|comp films fabricated without additives with WO
3|µp600 prepared with PEG 600 as additive (
Table S1) shows a benefit of a microporous starting structure even for a crystalline film resulting in a more facile diffusion of ions.
As shown in
Figure 1, the WO
3|µp400, WO
3|µp600 and WO
3|µmp films were thicker than the films prepared without additives. Therefore, to check if the observed improvement of the transmittance modulation over WO
3|comp was provided by such increased thickness of the films and to directly compare the performance to commWO
3, thicker films of WO
3|comp, WO
3|µp600 and WO
3|µmp with similar thickness were prepared by subsequent preparation of several films onto each other. The thicker films (
Figure 8) changed into an almost as deep blue as commWO
3 at, however, even slightly improved transparency in the bleached state (
Figure S11d). Despite deep initial coloration, a reversible ∆
T710nm (
Figure 8) of just 0.35% was measured for a 646 nm thick WO
3|comp film allowing a reversible change between 0.76% and 0.41% only, indicating heavily trapped ions after the initial coloration step. In contrast, for a 702 nm thick WO
3|µp600 film impressive ∆
T710nm = 65.9% and for a 620 nm thick WO
3|µmp film ∆
T710nm = 84.8% were achieved, values even slightly larger than those obtained for the 230–250 nm thick WO
3|µmp films (
Table 8). It is thereby directly confirmed that the observed improvements of the switching characteristics are a direct consequence of a fast intercalation and deintercalation of Li
+ ions in the films facilitated by their porous morphology.
3.5. Long-Term Stability of Switching Characteristics
Response times
tcol and
tbleach (
Table 9) corresponding to the time needed to color or bleach, respectively, the films to 90% of ∆
T710nm [
24,
28] were calculated from the transmittance curves (
Figure 9,
Figure 10,
Figures S12 and S13).
tcol shorter than 6 s were consistently achieved for all films speaking in favor of a generally fast intercalation. However, rather long
tbleach > 40 s was determined for WO
3|comp, WO
3|mp and commWO
3 whereas the WO
3|µp400, WO
3|µp600 and WO
3|µmp were bleached within
tbleach < 25 s with short response times of just 2–3 s obtained for some samples. This tendency is again in conformity with the study of Sallard et al. [
34], where response times of
tcol ca. 20–30 s and
tbleach ca. 70 s were observed for samples similar to WO
3|mp. Despite higher ∆
T710nm for the commercial WO
3 film (
Figure S12d), the short bleaching times of the films with micropores confirmed a facile switching of these films (
Figure 9,
Figure 10,
Figures S12 and S13). The trend of
tbleach >
tcol for all films fits to the slower transmittance change during positive scans in CV (
Figure 6b) and
Dc,CV >
Da,CV (
Table 7). While the intercalation of Li
+ into all presently studied films is fast, their extraction is slow for compact films (WO
3|comp and commWO
3) and for WO
3|mp with remaining polymer fragments. By using PEG 400, PEG 600 or both, PIB
50-
b-PEO
45 and PEG 600 as additives, micropores were created that provide pathways for fast deintercalation of the ions.
A tendency of smaller
CE710nm was revealed in CA compared to CV (
Table 8). The direct fast application of −1.0 V leads to higher current densities and chances of local overcharging at the beginning of the coloration process as opposed to a slow, well-defined change of the potential during CV. Overcharging or undesired side reactions can result in lower
CE710nm. Comparing the different kinds of films, however,
CE710nm follows the same trend as observed before during CV. From CV,
CE710nm even higher for WO
3|µp400, WO
3|µp600 and WO
3|µmp compared to commWO
3 were reached as opposed to similar
CE710nm for WO
3|comp, WO
3|mp and commWO
3.
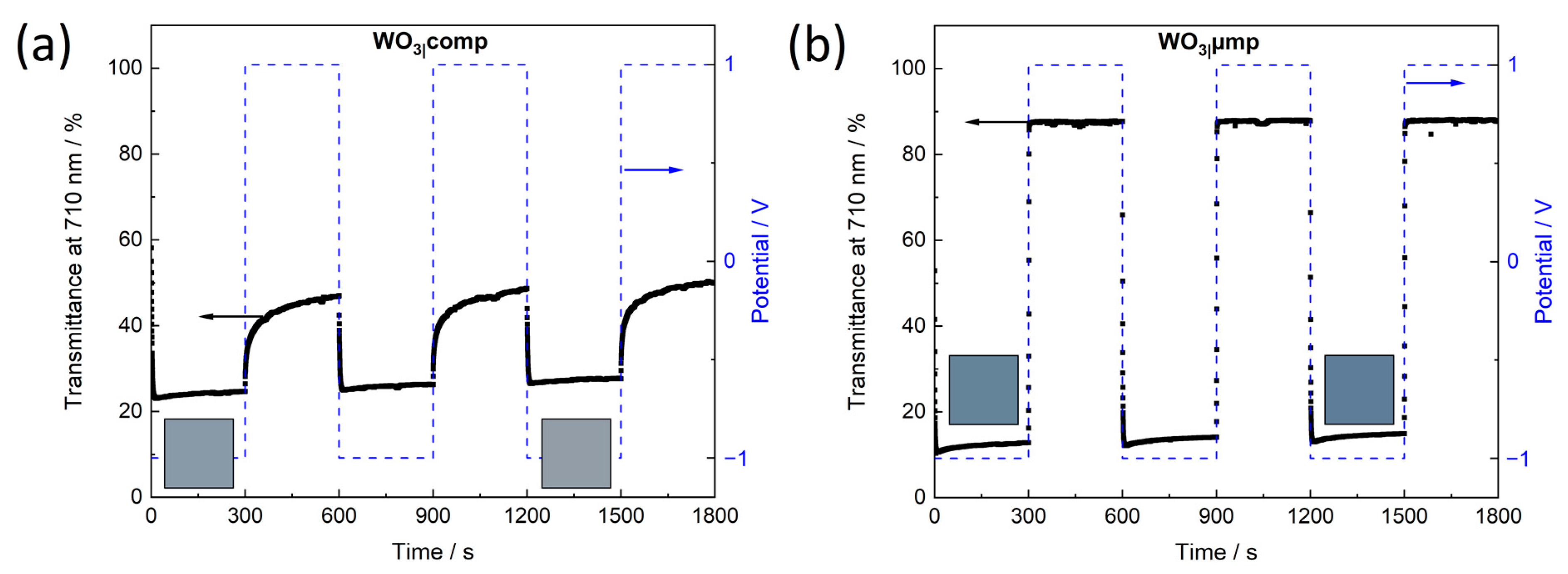
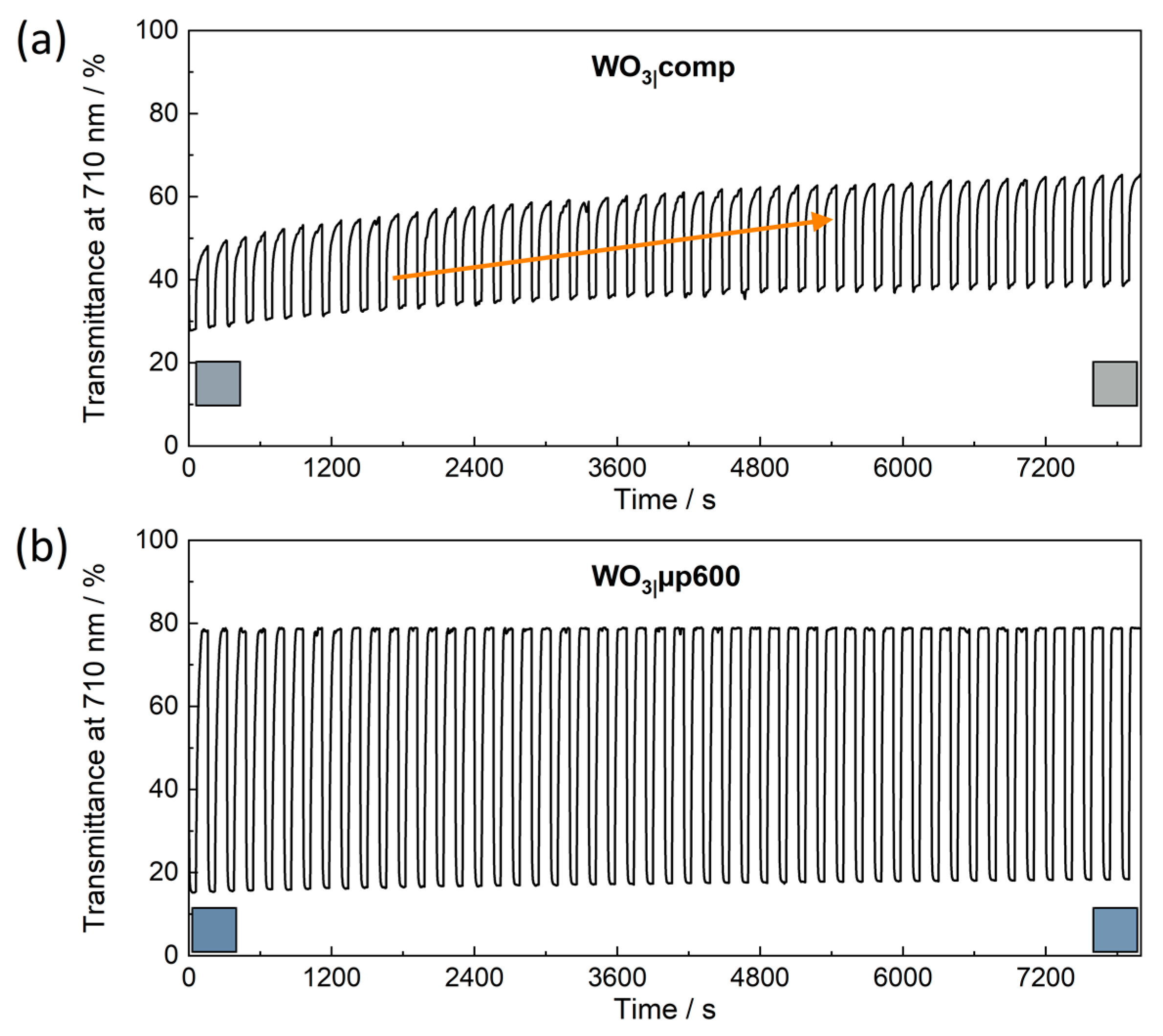
Reversibility of the switching process deserves further attention. Films of WO
3|comp showed a monotonous upward shift of
Tλ when the films were switched back and forth between −1.0 V and 1.0 V for 300 s each over three cycles (
Figure 9a). This trend continued when the films were studied over 50 more cycles (
Figure 10a). The compact film obviously contained a considerable concentration of trapped ions [
57]. It has been found earlier [
63,
64] that the concentration of intercalated ions
x in Li
xWO
3 in the films, clearly influenced the equilibria of W
6+ ⇌ W
5+ and W
5+ ⇌ W
4+ leading to the observed changes in the optical absorption of WO
3. It was observed that up to
x = 0.3–0.5, the transmittance first decreased and then shifted back to higher transmittance for larger
x whereby after exceeding
x ≈ 0.7 the films became optically irreversible showing a light-brownish color. The range of
x < 0.3–0.7 in the present experiments was chosen to provide a good compromise between intense coloration and high reversibility. However, in
Figure S14a it can be seen that after the first intercalation of WO
3|comp for 300 s at −1.0 V (1. Cycle) ions were trapped within the films leading to a bluish film even after reversal to 1.0 V (
Figure S14c). The color impressions of the film after 53 cycles indicate that the concentration of trapped ions must have increased subsequently until the films became light-brownish with
Tλ for both −1.0 V and 1.0 V shifted to higher values (
Figure S14a,c). For WO
3|mp, despite mesoporous morphology, the remaining polymer in the film also led to trapped ions and, thus, a similar trend towards increasing
Tλ (
Figure S13a) as obtained for WO
3|comp. Hence, long coloration times are disadvantageous for such films and lead to the observed irreversibilities. For the WO
3|µp400, WO
3|µp600 and WO
3|µmp films, however,
Tλ in the bleached as well as in the colored states remained widely constant at 1.0 V and −1.0 V, respectively (
Figure 10b,
Figures S13b,c and S14b,c). These films point out the possibility to endure multiple reversible switching at a high stability of the films. The porous network in these films without significant amounts of residual polymer fragments enables a facile intercalation and deintercalation of the Li
+ ions allowing the observed reversible switching of these films.
The reversibility dependent on the applied negative potential is compared for WO
3|comp and WO
3|µp600 in
Figure S15. It could be observed that independent of the negative potential, WO
3|µp600 could be fully bleached again at 1.0 V reaching a transmittance equal to the one in the initial state. For WO
3|comp, however, the transmittance was lower than in the initial state already after prior polarization at −0.25 V only, indicating trapped ions already for weakly reduced WO
3|comp. For both films it can be observed that the spectral shape also changed with increasing negative potential. The transmittance at longer wavelengths decreased less than expected when compared to the change at short wavelengths to a degree that around 600–900 nm the transmittance even increased from −0.75 V to −1.0 V, most clearly seen for WO
3|comp (
Figure S15a). This trend can be explained by a Li
+ concentration increased to
x ≈ 0.6 at −1.0 V, a value higher than the limit of
x ≥ 0.5, for which such spectral changes were already reported [
63,
64].
3.6. Chemical Analysis of Reduced Films
The spectroelectrochemical analysis has shown that PEG 600 in combination with PIB
50-
b-PEO
45 as structure-directing agents led to WO
3 films with optimum electrochromic switching characteristics (
Table 8,
Figure 7b,
Figure 9b and
Figure S13c). For films prepared with just PEG 600 or PEG 400 as additive, switching characteristics almost as good (
Table 8,
Figure 7a,
Figure 10b,
Figures S11a, S12a,b and S13b) were achieved. From a technological point of view, application of PEG only provides other advantages over using both, PEG and PIB
50-
b-PEO
45. The preparation of the films is not only saving the additional demanding precursor but also saves time and energy since a heat treatment of the films is only needed at 250 °C for one hour as opposed to 300 °C for 12 h. Therefore, and in order to allow further optimization of such films in the future, details of the intercalation reactions and of the charge transport were studied for WO
3|µp400 and WO
3|µp600 and compared to WO
3|comp.
The presence of reduced states of WO
3 was directly proven by XPS analysis (
Figure S16 vs.
Figure 4 and
Figures S4–S7) after applying −1.0 V for 300 s in the electrochemical cell and subsequent transfer to vacuum. All films showed an increase in the concentration of W
5+ from around 0.3–2.8% in the pristine state to 16–38% in the reduced state (
Figure S8b,
Table 4), comparable to the values of 30–35% reported for colored WO
3 films prepared by a cathodic arc plasma [
65]. A trend of higher concentrations of W
5+ (at least at the surface) in WO
3|µp400 and WO
3|µp600 and to lower concentrations in WO
3|mp and WO
3|µmp, also when compared to WO
3|comp was observed. Efficient reduction of microporous WO
3 was thereby confirmed. Despite high coloration of WO
3|µmp and low coloration of WO
3|mp, both these mesoporous films showed a similarly low concentration of W
5+ and degree of reduction
x, in line with a high
CE710nm observed for WO
3|µmp, but rather low
CE710nm for WO
3|mp (
Table 8). Ar
+-sputtering of the reduced films with intercalated Li
+ allowed to discuss the depth profile of the redox reactions. The concentration of W
5+ for the reduced films decreased and then saturated with proceeding etching time, i.e., deeper in the films (
Figure S8). This is consistent with a reduction reaction proceeding from the surface towards the inner volume of the films as would be expected for a movement of e
− still faster also in porous WO
3 than Li
+, which can be expected to be fast within the pores but slow within solid, albeit porous WO
3, as confirmed by increased but still quite moderate
DCV (
Table 7). The subsequent reduction to W
4+, W
x+ and W
0 occurred faster than in the pristine films, reasonable in view of a start already in a reduced state of WO
3 (
Figure S16).
To directly prove the impact of the microporous network and to complement this analysis, the distribution of Li
+ in the reduced films was also analyzed by ToF-SIMS (
Figure 11 and
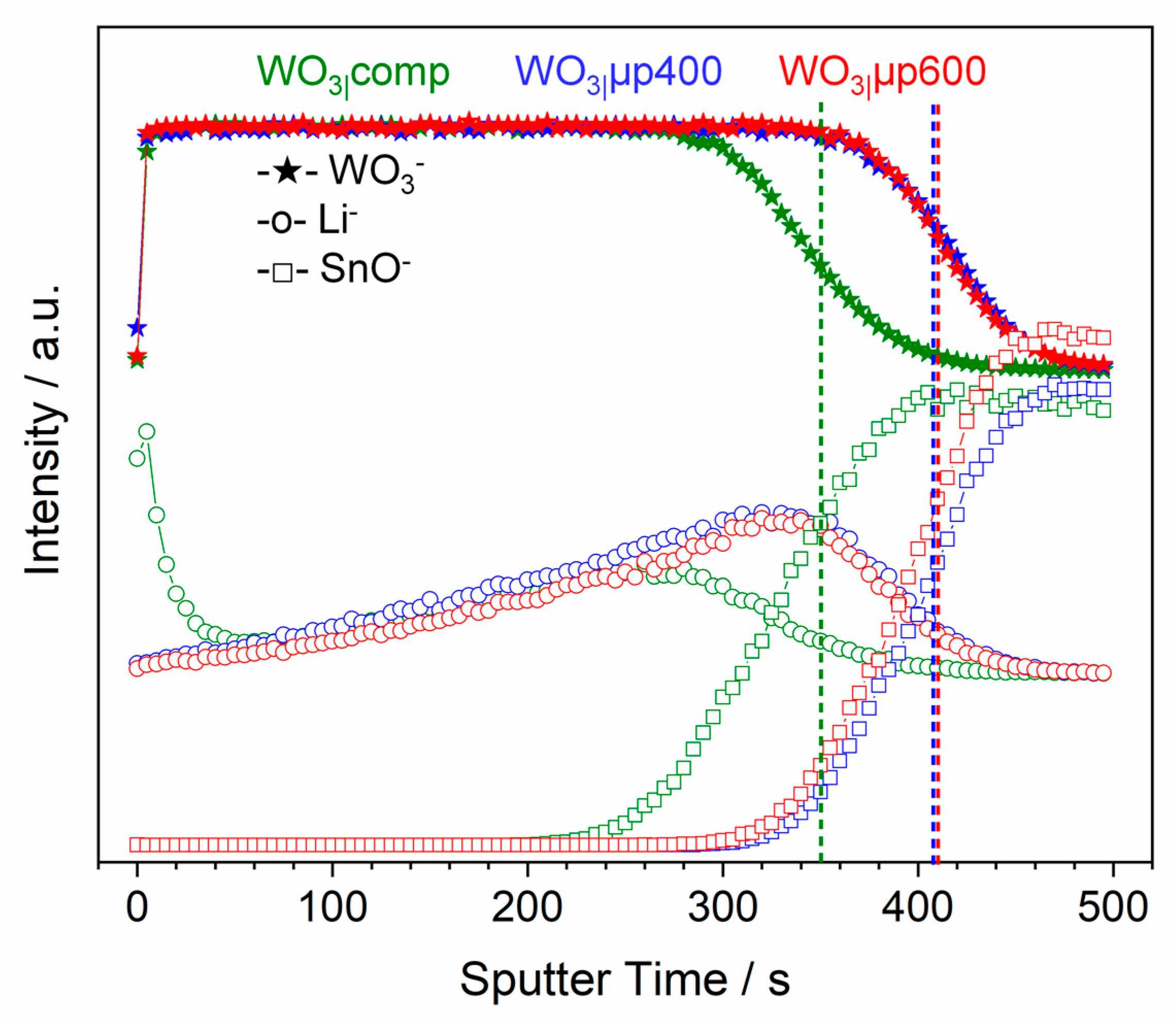
Figure S17). The WO
3− and the SnO
− signals (
Figure 11) are used for correlating the sputter times and the etched thickness of the films. For the WO
3|µp400 and WO
3|µp600 samples, the decrease in intensity of the WO
3− signal and the increase in intensity of the SnO
− signal occurred at longer sputter times compared to the WO
3|comp films, well in line with a higher film thickness for WO
3|µp400 and WO
3|µp600 (
Table 5). A rather constant sputter rate of about 0.3 nm/s was obtained for all films. All depth profiles (
Figure S17) confirm that an intercalation for around 60 s is sufficient to completely charge the WO
3 film since the depth profiles of the films intercalated for 60 s or 300 s are quite similar, as also revealed by transmittance spectra measured for the films after 60 s and 300 s of intercalation (
Figure S18). For WO
3|comp films, an accumulation of Li
+ at the film surface up to a depth (sputter time) of around 20 nm (60 s) can be clearly observed (
Figure 11 and
Figure S17a) after reduction for
t ≥ 60 s, confirming a compact film structure and, thus, a low accessibility of the inner volume of the film for Li
+. In strong contrast, reduced WO
3|µp400 and WO
3|µp600 did not show any Li
+ accumulation at the surface, speaking in favor of a more homogeneous reduction of the films enabled by a good accessibility of the inner parts of the films for Li
+. An increase of the Li
− signal at increased depths and for increased intercalation times observed for all films (
Figure S17) points at intercalation of Li
+ also in the inner parts of the films, but, since unrealistically high Li
− concentrations were detected close to the interface with FTO, seems to be superimposed by artefacts of the measurements caused by changing ionization probabilities (matrix effects) and by different sputter probabilities of the elements [
66,
67], which hinders a more detailed analysis of the depth profiles.
3.7. Charge Transport within Microporous vs. Compact Tungsten Oxide
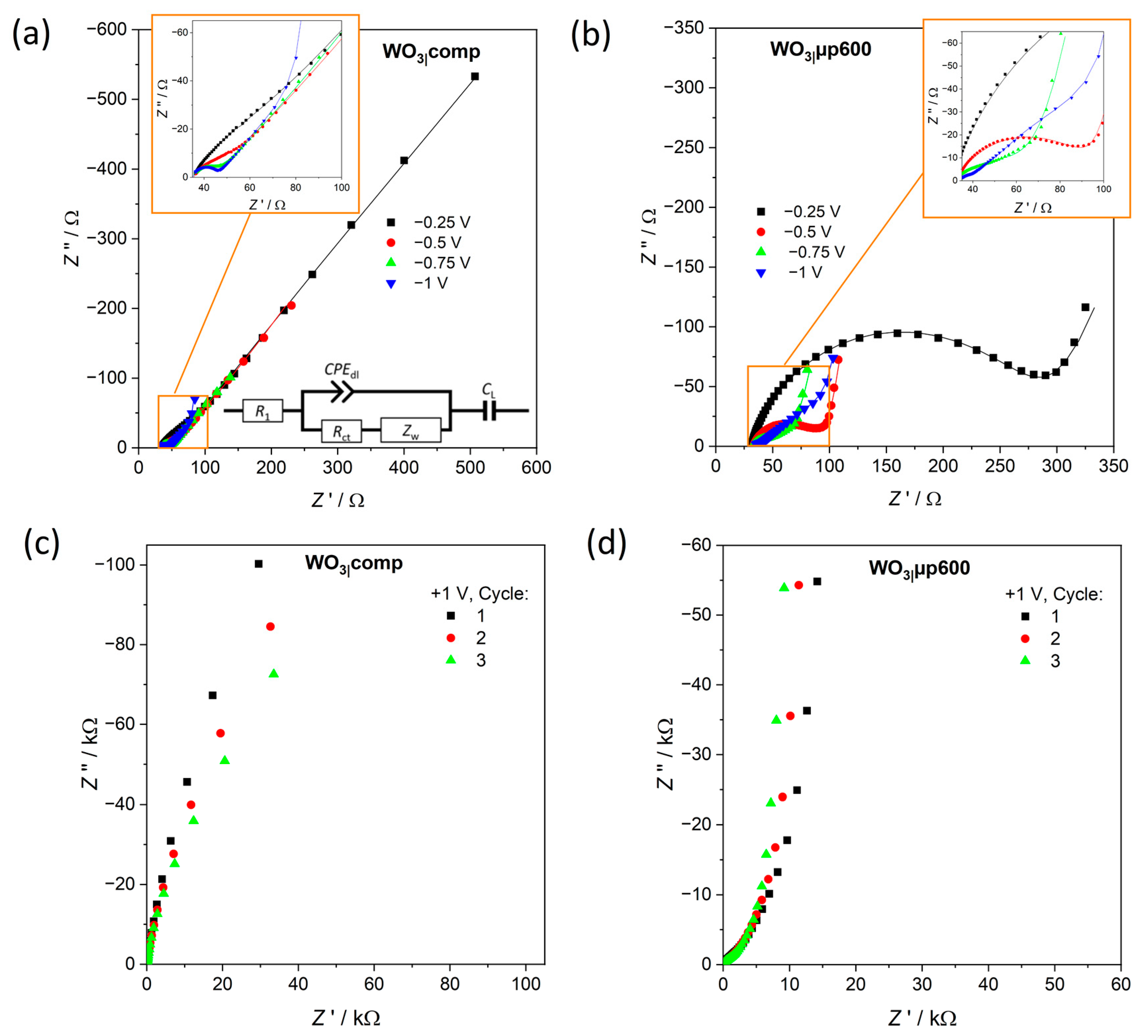
The direct influence of the microporous network on electrode kinetics of the electrochromic reaction was independently analyzed by impedance spectroscopy in different redox states of WO
3|comp and WO
3|µp600 films. The resulting Nyquist plots (
Figure 12) can be fitted well with the equivalent circuit shown in
Figure 12a. This circuit is typically used for the description of intercalation processes in electrochromic films [
68,
69].
R1 corresponds to a series resistance caused by the electrolyte and the substrate [
69,
70,
71,
72].
Rct is assigned to the resistance of the charge transfer from the WO
3 surface to the electrolyte and describes the intercalation of the ions into the WO
3 film as well as the interfacial redox processes [
68,
69,
71,
72,
73]. The electrochemical double layer consisting of the electrons at the electrode surface and ions on the electrolyte side can be modelled by a constant phase element or a capacitance [
68,
69,
71,
72,
73]. A more accurate fit of the present data was obtained by using a constant phase element
CPEdl. A semi-infinite type Warburg impedance
Zw represents the diffusion of Li
+ ions in the WO
3 films [
68,
69,
71,
73] but had to be replaced by a finite-length-type Warburg element
Zw,s [
72,
73] for the impedance spectra measured at a potential of −1.0 V, which led to more reliable fits of the data.
CL refers to a limiting capacitance caused by the finite length of the WO
3 film and considers a diffusion limited by charge accumulation in the film at low frequencies [
68,
69,
74].
As expected, the series resistance
R1 in the high frequency range of the Nyquist plot (
Figure 12) was found quite constant around 30–35 Ω for compact or microporous WO
3 and at different applied potentials. Similarly,
QCPE and α representing the double layer capacitance and
CL representing the limiting capacitance of WO
3 showed no significant changes either with potential or among the films speaking in favor of a rather constant arrangement of ions and electrons at the electrode surface and of a rather constant maximum charge uptake. The resistance
Rct, however, for WO
3|comp decreased from about 51 Ω at −0.25 V to 9 Ω at −1.0 V, a trend directly seen by a decreasing radius of the semicircle in the moderate frequency range (
Figure 12a). At negative potentials, a driving force is established to insert Li
+ into the films and the barrier to enter is decreased. The higher value of
Rct at −0.25 V compared to the value at −1.0 V points out that a higher barrier has still to be overcome to insert Li
+ at −0.25 V, possibly caused by slow transport of ions and/or electrons within the WO
3|comp structure. For WO
3|µp600,
Rct of about 248 Ω at −0.25 V was found considerably higher than for WO
3|comp. Since ion transport should not be hindered in a microporous compared to a compact material, the reason should be sought in an attenuated electron transport. The porous morphology and a thicker film can both lead to a larger number of grain boundaries posing additional barriers for electron transport. However,
Rct of the microporous film decreased to around 38 Ω at a potential of −0.75 V and then to about 11 Ω at −1.0 V. Hence, the values of
Rct at −1.0 V are quite similar for both types of films indicating a similar facile electron transport within the films at this potential and allowing fast intercalation of Li
+. For both types of films, the impedance values at 1.0 V (
Figure 12c,d) are much higher than at −1.0 V representing a high barrier for intercalation of the ions in the bleached state as already observed in earlier reports [
75,
76].
The Warburg coefficient
Aw obtained from fits of the impedance spectra allows to obtain an independent measure of Li
+ diffusion by the ion diffusion coefficient
DI in the films with the active surface area
A, the molar gas constant
R and the temperature
T [
77]:
The WO
3|comp film (
Table S2) provided generally smaller
DI compared to the WO
3|µp600 film, which is well in line with the tendency observed from the effective diffusion coefficients obtained from the CV measurements. However,
DI in the range of 10
−13–10
−11 cm
2 s
−1 were found around two orders of magnitude smaller than
Dc,CV and
Da,CV. This is a consequence of the fact that
DCV are effective diffusion coefficients describing the combined diffusion of electrons and ions while
DI is characteristic for the diffusion of ions only. Since CV was performed across the whole range of potentials and since the fits of the impedance spectra at −1.0 V needed a dedicated Warburg element, values at intermediate potentials (−0.25 V to −0.75 V) are used for comparison purposes. Throughout this range,
DI was found significantly larger for WO
3|µp600 than for WO
3|comp, well in line with larger
DCV for WO
3|µp600, confirming faster ion transport in WO
3|µp600. The difference in
DCV, however, was found smaller than that in
DI presumably caused by slower electron transport in WO
3|µp600 which also is considered by the effective diffusion coefficient
DCV.