Beta vulgaris subsp. maritima: A Valuable Food with High Added Health Benefits
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material
2.2. Plant Extraction
2.3. HPLC-DAD Analysis
2.4. Determination of Total Phenolic Content
2.5. Determination of Total Flavonoid Content
2.6. Antioxidant Activities
2.6.1. ABTS+ Radical Scavenging Assay
2.6.2. DPPH Radical Scavenging Assay
2.6.3. Ferric Reducing Antioxidant Power Assay (FRAP)
2.6.4. Metal Chelating Activity
2.7. Antiglycation Activities
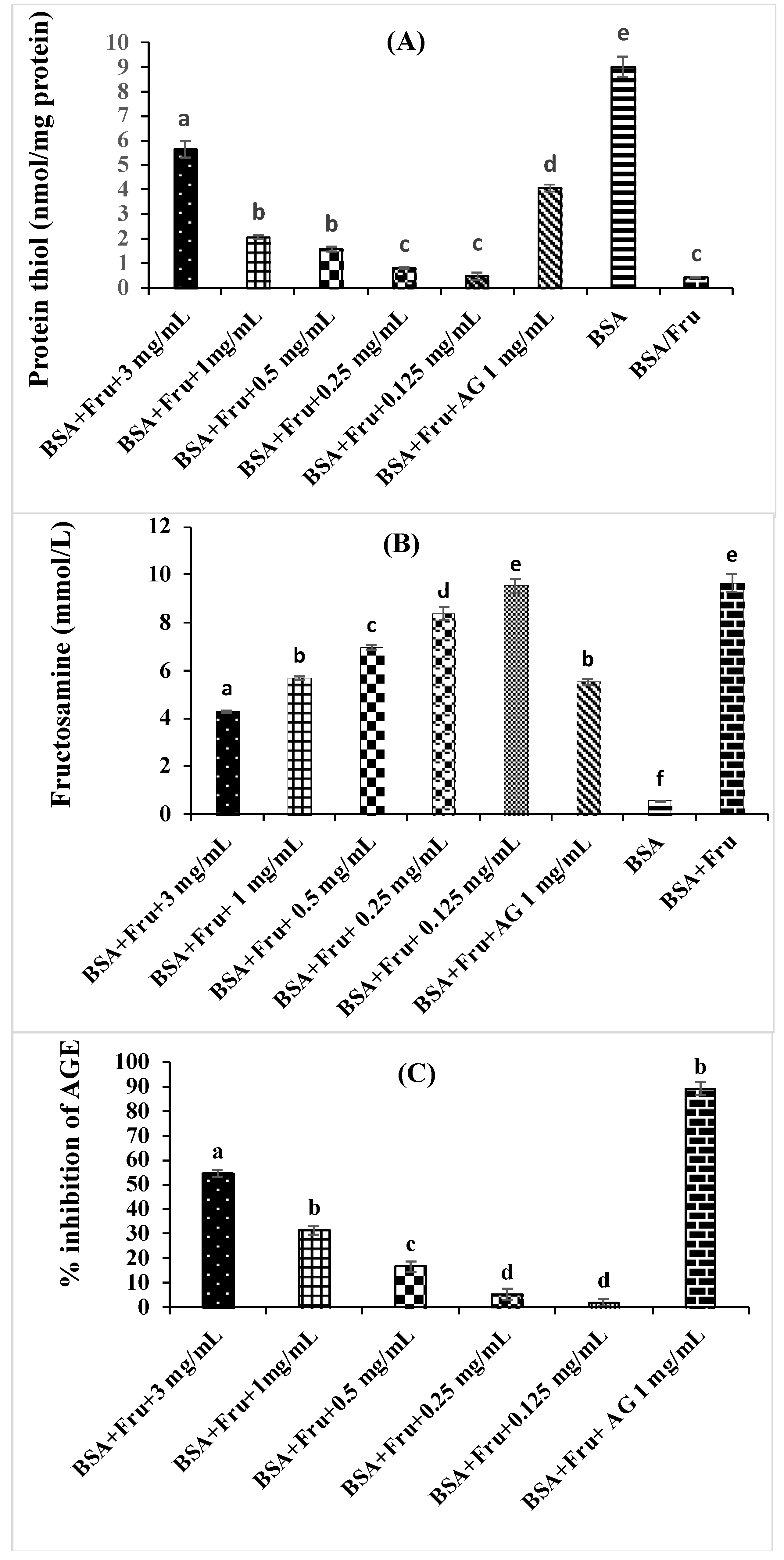
2.7.1. Determination of Fructosamine
2.7.2. Determination of Protein Thiol Group
2.8. Antidiabetic Enzymatic Activities
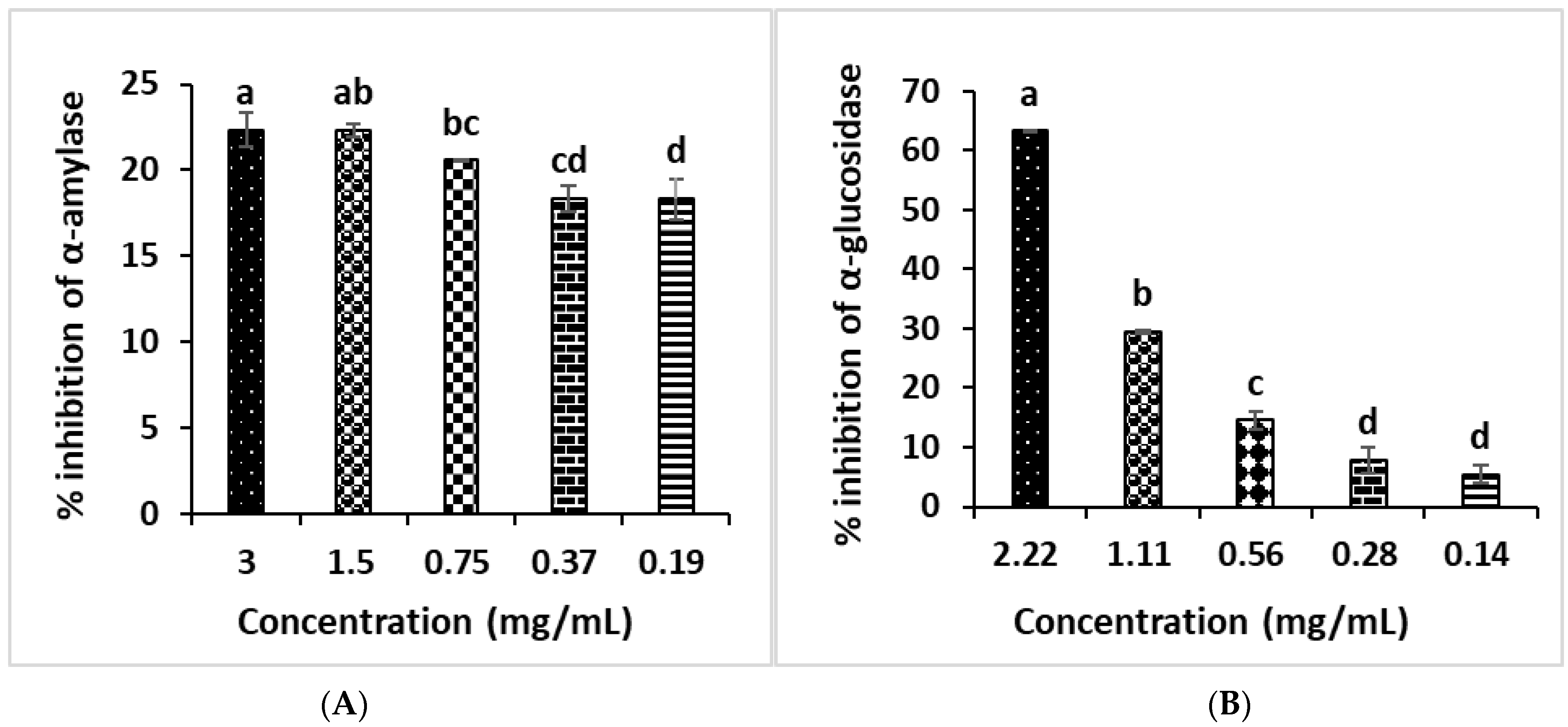
2.8.1. α-Amylase Inhibition Effect
2.8.2. α-Glucosidase Inhibitory Assay
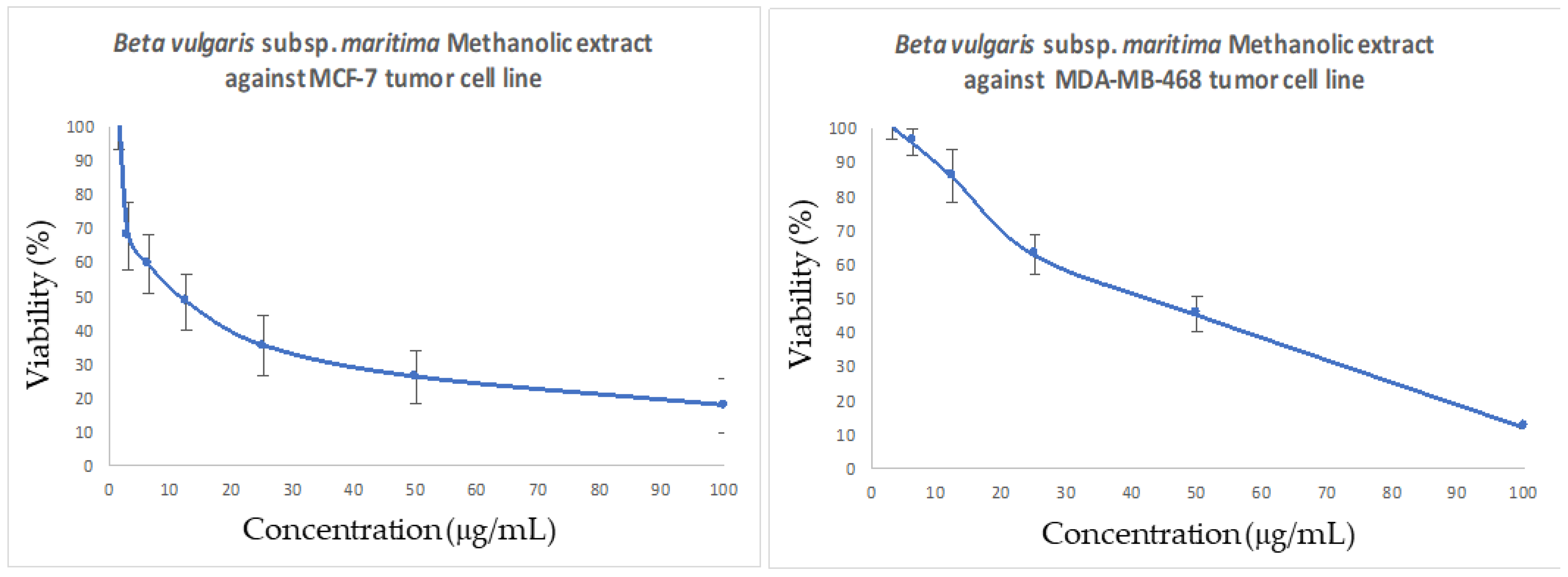
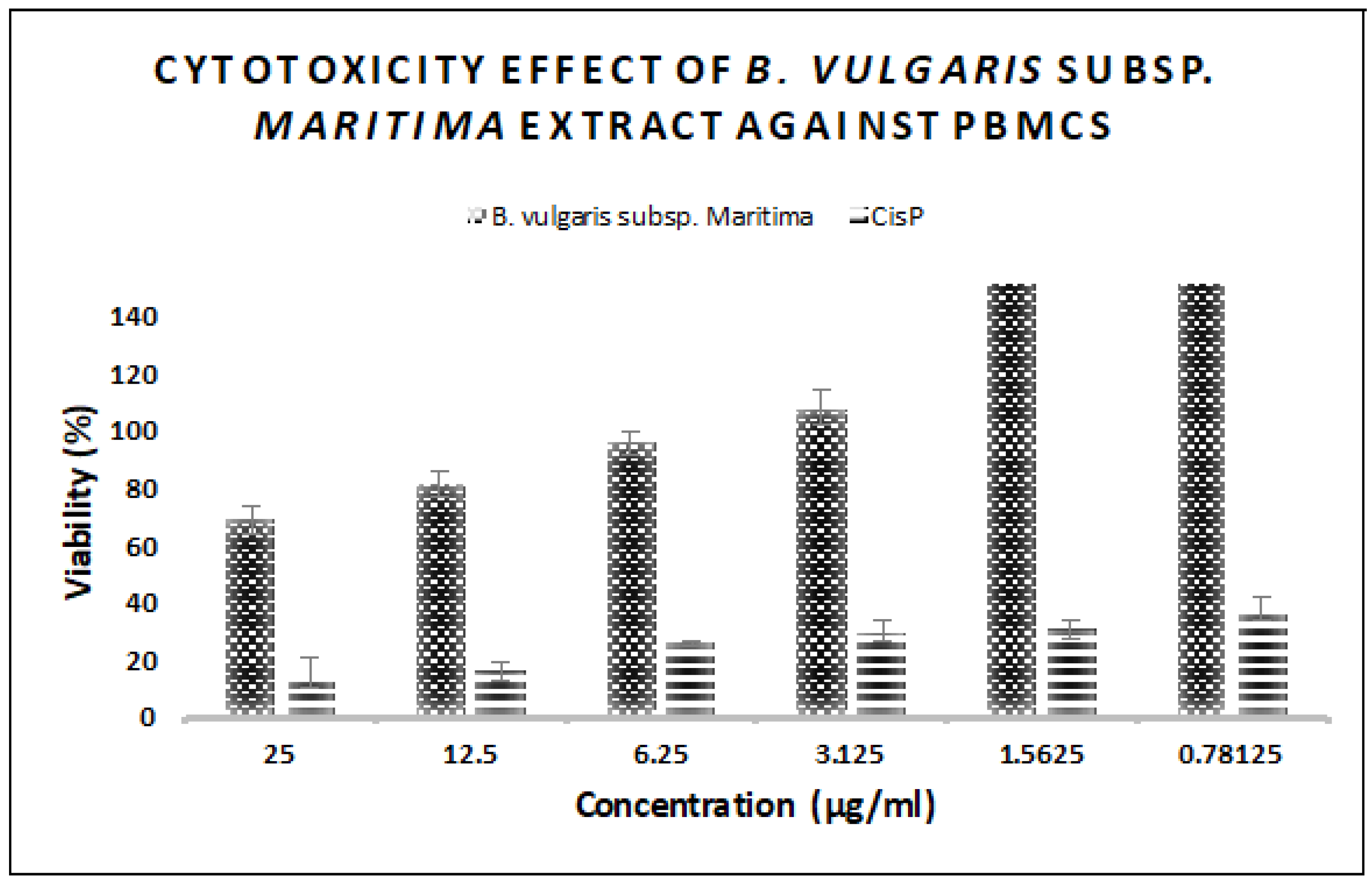
2.9. In Vitro Cytotoxic Activity
2.9.1. Cell Culture
2.9.2. MTT Assay
2.10. Antibacterial Activity
2.10.1. Microorganisms and Growth Conditions
2.10.2. Agar Disc Diffusion Method and Broth Microdilution Method
2.11. Statistical Analysis
3. Results and Discussion
3.1. Total Polyphenols’ and Flavonoid Contents
3.2. Identification and Quantification of Phenolic Compounds by HPLC
3.3. Antioxidant Capacity
3.4. Inhibition of Beta vulgaris subsp. maritima Leaves on Non-Enzymatic Glycation Process
3.5. In Vitro Study of Anti-Diabetic Activities of B. vulgaris subsp. maritima Leaves’ Extract
3.6. In Vitro Antitumor Activity of B. vulgaris subsp. maritima Leaves against Breast Cancer
3.7. Antibacterial Activity of B. vulgaris subsp. maritima Leaves’ Extract
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Saeedian, M.-E.; Amini, M. 2-[2-Methyl-5-phenyl-1-(3, 4, 5-trimethoxyphenyl)-1H-pyrrol-3-yl]-2-oxo-N-(pyridin-4-yl) acetamide. Molbank 2018, 2018, 1002. [Google Scholar] [CrossRef]
- Bouchmaa, N.; Ben Mrid, R.; Boukharsa, Y.; Nhiri, M.; Ait Mouse, H.; Taoufik, J.; Zyad, A. Cytotoxicity of new pyridazin-3 (2H)-one derivatives orchestrating oxidative stress in human triple-negative breast cancer (MDA-MB-468). Arch. Pharm. 2018, 351, 1800128. [Google Scholar] [CrossRef] [PubMed]
- Bouchmaa, N.; Tilaoui, M.; Boukharsa, Y.; Jaâfari, A.; Mouse, H.A.; Oukerrou, M.A.; Taoufik, J.; Ansar, M.; Zyad, A. In vitro antitumor activity of newly synthesized pyridazin-3 (2H)-one derivatives via apoptosis induction. Pharm. Chem. J. 2018, 51, 893–901. [Google Scholar] [CrossRef]
- Zhu, A.-G.; Jiang, Z.-S.; Gong, P.-Y.; Zhang, D.-M.; Zou, Z.-W.; Zhang, Q.; Ma, H.-M.; Guo, Z.-g.; Zhao, J.-y.; Dong, J.-j.; et al. Efficacy of low-protein diet for diabetic nephropathy: A systematic review of randomized controlled trials. Lipids Health Dis. 2018, 17, 141. [Google Scholar] [CrossRef] [PubMed]
- Ben Mrid, R.; Bouargalne, Y.; El Omari, R.; Nhiri, M. New insights into the therapeutic effects of phenolic acids from sorghum seeds. J. Rep. Pharm. Sci. 2019, 8, 91. [Google Scholar]
- Li, Y.; Li, S.; Meng, X.; Gan, R.Y.; Zhang, J.J.; Li, H.B. Dietary natural products for prevention and treatment of breast cancer. Nutrients 2017, 9, 728. [Google Scholar] [CrossRef] [PubMed]
- Oukerrou, M.A.; Tilaoui, M.; Mouse, H.A.; Bouchmaa, N.; Zyad, A. Differential cytotoxic activity of essential oil of Lippia citriodora from different regions in Morocco. Chem. Biodivers. 2017, 14, e1600497. [Google Scholar] [CrossRef]
- Ben Mrid, R.; Bouchmaa, N.; Bouargalne, Y.; Ramdan, B.; Karrouchi, K.; Kabach, I.; Nhiri, M. Phytochemical characterization, antioxidant and in vitro cytotoxic activity evaluation of Juniperus oxycedrus Subsp. oxycedrus needles and berries. Molecules 2019, 24, 502. [Google Scholar] [CrossRef]
- Kazimierczak, R.; Hallmann, E.; Lipowski, J.; Drela, N.; Kowalik, A.; Püssa, T.; Matt, D.; Luik, A.; Gozdowski, D.; Rembiałkowska, E. Beetroot (Beta vulgaris L.) and naturally fermented beetroot juices from organic and conventional production: Metabolomics, antioxidant levels and anticancer activity. J. Sci. Food Agric. 2014, 94, 2618–2629. [Google Scholar] [CrossRef]
- Nade, V.S.; Kawale, L.A.; Zambre, S.S.; Kapure, A.B. Neuroprotective potential of Beta vulgaris L. in Parkinson’s disease. Ind. J. Pharmacol. 2015, 47, 403. [Google Scholar] [CrossRef]
- Oztay, F.; Sacan, O.; Kayalar, O.; Bolkent, S.; Ipci, Y.; Kabasakal, L.; Sener, G.; Yanardag, R. Chard (Beta vulgaris var. cicla) extract improved hyperglycemia-induced oxidative stress and surfactant-associated protein alterations in rat lungs. Pharm. Biol. 2015, 53, 1639–1646. [Google Scholar] [CrossRef] [PubMed]
- Citores, L.; Iglesias, R.; Gay, C.; Ferreras, J.M. Antifungal activity of the ribosome-inactivating protein BE 27 from sugar beet (Beta vulgaris L.) against the green mould Penicillium digitatum. Mol. Plant Pathol. 2016, 17, 261–271. [Google Scholar] [CrossRef] [PubMed]
- Duke, J.A. Handbook of Energy Crops; Purdue University, Center for New Crops & Plants Products: West Lafayette, IN, USA, 1983. [Google Scholar]
- Asraoui, F.; Kounnoun, A.; El Cadi, H.; Cacciola, F.; Oulad El Majdoub, Y.; Alibrando, F.; Mandolfino, F.; Dugo, P.; Mondello, L.; Louajri, A. Phytochemical Investigation and Antioxidany Activity of Globularia alypum L. Molecules 2021, 26, 759. [Google Scholar] [CrossRef] [PubMed]
- Kabach, I.; Mrid, R.; Bouchmaa, N.; Bouargalne, Y.; Nhiri, A. Phytochemical screening, antioxidant and cytotoxic activities of M. vulgare. Int. J. Pharm. Res. 2019, 11, 338–345. [Google Scholar]
- Oyaizu, M. Studies on products of browning reaction antioxidative activities of products of browning reaction prepared from glucosamine. Jpn. Nutr. Diet. 1986, 44, 307–315. [Google Scholar] [CrossRef]
- Dinis, T.C.; Madeira, V.M.; Almeida, L.M. Action of phenolic derivatives (acetaminophen, salicylate, and 5-aminosalicylate) as inhibitors of membrane lipid peroxidation and as peroxyl radical scavengers. Arch. Biochem. Biophys. 1994, 315, 161–169. [Google Scholar] [CrossRef]
- Adisakwattana, S.; Sompong, W.; Meeprom, A.; Ngamukote, S.; Yibchok-Anun, S. Cinnamic acid and its derivatives inhibit fructose-mediated protein glycation. Int. J. Mol. Sci. 2012, 13, 1778–1789. [Google Scholar] [CrossRef]
- Zhang, L.; Meng, Z.; Jiang, Z.; Liu, Z.; Hou, L.; Cai, G.; Ou, H.; Huang, S.; Song, Q.; Fang, J.; et al. Indicators of glucose dysregulation and the relationship with iron overload in Chinese children with beta thalassemia major. Pediatric Diabetes 2021. [Google Scholar] [CrossRef]
- Dai, J.; Dong, A.; Xiong, G.; Liu, Y.; Hossain, M.S.; Liu, S.; Gao, N.; Li, S.; Wang, J.; Qiu, D. Production of highly active extracellular amylase and cellulase from Bacillus subtulis ZIM3 and a recombinant strain with a potential application in tobacco fermentation. Front. Microbiol. 2020, 11, 1539. [Google Scholar] [CrossRef]
- Bouchmaa, N.; Ben Mrid, R.B.; Boukharsa, Y.; Bouargalne, Y.; Nhiri, M.; Idir, A.; Taoufik, J.; Ansar, M.; Zyad, A. Reactive Oxygen Species-Mediated Apoptosis and Cytotoxicity of Newly Synthesized Pyridazin-3-Ones In P815 (Murin Mastocytoma) Cell Line. Drug Res. 2019, 69, 528–536. [Google Scholar] [CrossRef]
- Benali, T.; Chtibi, H.; Bouyahya, A.; Khabbach, A.; Hammani, K. Detection of antioxidant and antimicrobial activities in phenol components and essential oils of cistus ladaniferus and mentha suaveolens extracts. Biomed. Pharmacol. J. 2020, 13, 603–612. [Google Scholar] [CrossRef]
- Gulluce, M.; Sahin, M.; Sokmen, M.; Ozer, H.; Daferera, D.; Sokmen, A.; Polissiou, M.; Adiguzel, A.; Ozkan, H. Antimicrobial and antioxidant properties of the essential oils and methanol extract of Mentha longifolia L. ssp longifolia. Food Chem. 2007, 103, 1449–1456. [Google Scholar] [CrossRef]
- Shahidi, F.; Chandrasekara, A. Millet grain phenolics and their role in disease risk reduction and health promotion: A review. J. Funct. Food. 2013, 5, 570–581. [Google Scholar] [CrossRef]
- Biondo, P.B.F.; Boeing, J.S.; Barizão, É.O.; Souza, N.E.D.; Matsushita, M.; Oliveira, C.C.D.; Boroski, M.; Visentainer, J.V. Evaluation of beetroot (Beta vulgaris L.) leaves during its developmental stages: A chemical composition study. Food Sci. Technol. 2014, 34, 94–101. [Google Scholar] [CrossRef]
- Hajihosseini, S.; Setorki, M. The antioxidant activity of Beta vulgaris leaf extract in improving scopolamine-induced spatial memory disorders in rats. Avic. J. Phytomed. 2017, 7, 417. [Google Scholar]
- Gennari, L.; Felletti, M.; Blasa, M.; Angelino, D.; Celeghini, C.; Corallini, A.; Ninfali, P. Total extract of Beta vulgaris var. cicla seeds versus its purified phenolic components: Antioxidant activities and antiproliferative effects against colon cancer cells. Phytochem. Anal. 2011, 22, 272–279. [Google Scholar] [CrossRef]
- Kujala, T.S.; Vienola, M.S.; Klika, K.D.; Loponen, J.M.; Pihlaja, K. Betalain and phenolic compositions of four beetroot (Beta vulgaris) cultivars. Eur. Food Res. Technol. 2002, 214, 505–510. [Google Scholar] [CrossRef]
- Georgiev, V.G.; Weber, J.; Kneschke, E.M.; Denev, P.N.; Bley, T.; Pavlov, A.I. Antioxidant activity and phenolic content of betalain extracts from intact plants and hairy root cultures of the red beetroot Beta vulgaris cv. Detroit dark red. Plant Food. Hum. Nutr. 2010, 65, 105–111. [Google Scholar] [CrossRef]
- Barros, L.; Morales, P.; Carvalho, A.M.; Ferreira, I.C. Antioxidant potential of wild plant foods. In Mediterranean Wild Edible Plants; Springer: New York, NY, USA, 2016; pp. 209–232. [Google Scholar]
- Cheng, D.; Li, W.; Wang, L.; Li, T.; Poiani, G.; Wassef, A.; Hudilikar, R.; Ondar, P.; Brunetti, L.; Kong, A.-N. Pharmacokinetics, Pharmacodynamics, and PKPF Modeling of Curcumin in Regulating Antioxidant and Epigenetic Gene Expression I Healthy Human Volunteers. Mol. Pharm. 2019, 16, 1881–1889. [Google Scholar] [CrossRef]
- Xu, X.; Guo, S.; Hao, X.; Ma, H.; Bai, Y.; Huang, Y. Improving antioxidant and antiproliferative activities of colla corii asini hydrolysates using ginkgo biloba extracts. Food Sci. Nutr. 2018, 6, 765–772. [Google Scholar] [CrossRef]
- Zardi-Bergaoui, A.; Ben Nejma, A.; Harzallah-Skhiri, F.; Flamini, G.; Ascrizzi, R.; Ben Jannet, H. Chemical composition and biological studies of the essential oil from aerial parts of Beta vulgaris subsp. maritima (L.) arcang. growing in Tunisia. Chem. Biodivers. 2017, 14, e1700234. [Google Scholar] [CrossRef] [PubMed]
- Mzoughi, Z.; Chahdoura, H.; Chakroun, Y.; Cámara, M.; Fernández-Ruiz, V.; Morales, P.; Mosbah, H.; Flamini, G.; Snoussi, M.; Majdoub, H. Wild edible Swiss chard leaves (Beta vulgaris L. var. cicla): Nutritional, phytochemical composition and biological activities. Food Res. Int. 2019, 119, 612–621. [Google Scholar] [CrossRef]
- Aktumsek, A.; Zengin, G.; Guler, G.O.; Cakmak, Y.S.; Duran, A. Antioxidant potentials and anticholinesterase activities of methanolic and aqueous extracts of three endemic Centaurea L. species. Food Chem. Toxicol. 2013, 55, 290–296. [Google Scholar] [CrossRef] [PubMed]
- Valifard, M.; Mohsenzadeh, S.; Kholdebarin, B.; Rowshan, V. Effects of salt stress on volatile compounds, total phenolic content and antioxidant activities of Salvia mirzayanii. S. Afr. J. Bot. 2014, 93, 92–97. [Google Scholar] [CrossRef]
- Rowan, S.; Bejarano, E.; Taylor, A. Mechanistic targeting of advanced glycation end-products in age-related diseases. Biochim. Biophys. Acta 2018, 1864, 3631–3643. [Google Scholar] [CrossRef]
- Gill, V.; Kumar, V.; Singh, K.; Kumar, A.; Kim, J.J. Advanced glycation end products (AGEs) may be a striking link between modern diet and health. Biomolecules 2019, 9, 888. [Google Scholar] [CrossRef]
- Sarmah, S.; Roy, A.S. A review on prevention of glycation of proteins: Potential therapeutic substances to mitigate the severity of diabetes complications. Int. J. Biol. Macromol. 2021, 195, 565–588. [Google Scholar] [CrossRef]
- Wu, C.H.; Huang, S.M.; Lin, J.A.; Yen, G.C. Inhibition of advanced glycation endproduct formation by foodstuffs. Food Funct. 2011, 2, 224–234. [Google Scholar] [CrossRef]
- Grzegorczyk-Karolak, I.; Gołąb, K.; Gburek, J.; Wysokińska, H.; Matkowski, A. Inhibition of advanced glycation end-product formation and antioxidant activity by extracts and polyphenols from Scutellaria alpina L. and S. altissima L. Molecules 2016, 21, 739. [Google Scholar] [CrossRef]
- Fernández-Fernández, A.M.; Iriondo-DeHond, A.; Dellacassa, E.; Medrano-Fernandez, A.; del Castillo, M.D. Assessment of antioxidant, antidiabetic, antiobesity, and anti-inflammatory properties of a Tannat winemaking by-product. Eur. Food Res. Technol. 2019, 245, 1539–1551. [Google Scholar] [CrossRef]
- Lordan, S.; Smyth, T.J.; Soler-Vila, A.; Stanton, C.; Ross, R.P. The a-Amylase and a-Glucosidase Inhibitory Effects of Irish Seaweed Extracts. Food Chem. 2013, 141, 2170–2176. [Google Scholar] [CrossRef]
- Ozsoy-Sacan, O.; Karabulut-Bulan, O.; Bolkent, S.; Yanardag, R.; Ozgey, Y. Effects of chard (Beta vulgaris L. var cicla) on the liver of the diabetic rats: A morphological and biochemical study. Biosci. Biotechnol. Biochem. 2004, 68, 1640–1648. [Google Scholar] [CrossRef] [PubMed]
- Song, J.; Kwon, O.; Chen, S.; Daruwala, R.; Eck, P.; Park, J.B.; Levine, M. Flavonoid inhibition of sodium-dependent vitamin C transporter 1 (SVCT1) and glucose transporter isoform 2 (GLUT2), intestinal transporters for vitamin C and glucose. J. Biol. Chem. 2002, 277, 15252–15260. [Google Scholar] [CrossRef] [PubMed]
- Tundis, R.; Loizzo, M.R.; Statti, G.A.; Menichini, F. Inhibitory effects on the digestive enzyme α-amylase of three Salsola species (Chenopodiaceae) in vitro. Int. J. Pharm. Sci. 2007, 62, 473–475. [Google Scholar]
- Li, H.; Song, F.; Xing, J.; Tsao, R.; Liu, Z.; Liu, S. Screening and structural characterization of α-glucosidase inhibitors from hawthorn leaf flavonoids extract by ultrafiltration LC-DAD-MS n and SORI-CID FTICR MS. J. Am. Soc. Mass Spectrom. 2009, 20, 1496–1503. [Google Scholar] [CrossRef] [PubMed]
- Gennari, A.; Stockler, M.; Puntoni, M.; Sormani, M.; Nanni, O.; Amadori, D.; Wilcken, N.; D’Amico, M.; DeCensi, A.; Bruzzi, P. Duration of chemotherapy for metastatic breast cancer: A systematic review and meta-analysis of randomized clinical trials. J. Clin. Oncol. 2011, 29, 2144–2149. [Google Scholar] [CrossRef]
- Kongtawelert, P.; Wudtiwai, B.; Shwe, T.H.; Pothacharoen, P.; Phitak, T. Inhibitory effect of Hesperidin on the expression of programmed death ligand (PD-L1) in breast cancer. Molecules 2020, 25, 252. [Google Scholar] [CrossRef]
- Al-Mutairi, A.; Rahman, A.; Rao, M.S. Low doses of thymoquinone and ferulic acid in combination effectively inhibit proliferation of cultured MDA-MB 231 breast adenocarcinoma cells. Nutr. Canc. 2021, 73, 282–289. [Google Scholar] [CrossRef]
- Mekki, L.; Mansour, H.; Eldean, E.G.; Nasser, A.A. Evaluation of anti-genotoxicity of the ethanolic plant extract of Beta vulgaris Maritima using Allium cepa root assay. Egypt J. Exp. Biol. 2015, 11, 147–153. [Google Scholar]
- Anand, U.; Jacobo-Herrera, N.; Altemimi, A.; Lakhssassi, N. A comprehensive review on medicinal plants as antimicrobial therapeutics: Potential avenues of biocompatible drug discovery. Metabolites 2019, 9, 258. [Google Scholar] [CrossRef]
- Othman, L.; Sleiman, A.; Abdel-Massih, R.M. Antimicrobial activity of polyphenols and alkaloids in middle eastern plants. Front. Microbiol. 2019, 10, 911. [Google Scholar] [CrossRef] [PubMed]
- Jubair, N.; Rajagopal, M.; Chinnappan, S.; Abdullah, N.B.; Fatima, A. Review on the antibacterial mechanism of plant-derived compounds against multidrug-resistant bacteria (MDR). Evid.-Based Complement. Alternat. Med. 2021, 2021, 3663315. [Google Scholar] [CrossRef] [PubMed]
- Mishra, A.; Sharma, A.K.; Kumar, S.; Saxena, A.K.; Pandey, A.K. Bauhinia variegata leaf extracts exhibit considerable antibacterial, antioxidant, and anticancer activities. Biomed. Res. Int. 2013, 2013, 915436. [Google Scholar] [CrossRef] [PubMed]
| Extract Yield (%) | Polyphenols (mg GAE/g dw) | Flavonoids (mg QE/g dw) | |
|---|---|---|---|
| Aqueous extract | 21.71 | 12.377 ± 3.324 a | 26.023 ± 3.565 a |
| Methanolic extract | 18.82 | 37.117 ± 6.336 b | 38.785 ± 1.148 b |
| Phenolic Compounds mg/100 g dw | Aqueous Extract | Methanolic Extract |
|---|---|---|
| Hydroxycinnamic acids | ||
| Caffeic acid | 0.61 | ND |
| P-coum acid | 0.49 | 0.49 |
| Hydroxybenzoic acids | ||
| Salicylic acid | 758.52 | ND |
| Syringic acid | 2.16 | ND |
| Gallic acid | ND | ND |
| p-hydroxybenzoic acid | 0.58 | ND |
| Flavonoids | ||
| Hesperidin | 189.07 | 68.82 |
| Naringinin | 5.28 | 11.12 |
| Rutin | 47.86 | ND |
| Terpernes | ||
| Thymoquinone | 0.57 | 16.48 |
| Limonene | ND | ND |
| Antioxidants’ Properties (IC50 Values; mg/mL) | Reducing Power (mg AAE/g dw) | |||
|---|---|---|---|---|
| DPPH Scavenging Activity | ABTS | Metal Chelating Activity | ||
| Aqueous extract | 1.789 ± 0.062 a | 1.551 ± 0.094 a | 0.484 ± 0.007 a | 98.498 ± 4.198 a |
| Methanolic extract | 0.919 ± 0.044 b | 1.218 ± 0.044 a | 0.739 ± 0.057 b | 300.057 ±10.824 b |
| IC50 (mg/mL) | ||
|---|---|---|
| α-Amylase | α-Glucosidase | |
| Beta vulgaris subsp. maritima leaves | - | 1.792 ± 0.005 a |
| Acarbose | 0.046 ± 0.001 b | 0.329 ± 0.041 b |
| Sample Tested | IC50 of Cytotoxic Activity against Tumor Cells | % of Viability in PBMCs | ||||
|---|---|---|---|---|---|---|
| MCF-7 | MDA-MB-468 | PBMCs | Concentration (µg/mL) | |||
| 12.5 | 3.125 | 0.78125 | ||||
| B. v. subsp. Maritima | 11.57 ± 5.09 a | 42.50 ± 7.27 a | >50 a | 81.15 ±5.91 a | 108.02 ±1.49 a | 224.9 ±6.46 a |
| CisP | 0.20 ± 0.0 c | 2.20 ± 0.40 c | 0.27 b | 16.08 ± 3.39 b | 30.08 ± 3.58 b | 37.96 ± 3.44 b |
| Bacterial Strains | Inhibition Zone Diameter (mm) | |
|---|---|---|
| B. vulgaris subsp. maritima | Gentamicine | |
| Proteus mirabilis | 9.0 ± 0.0 | 18.7± 0.6 |
| Bacillus subtilis | 10.7 ± 0.6 | 19.7 ± 0.6 |
| Staphylococcus aureus | n.e | 15 ± 1 |
| Pseudomonas aeruginosa | n.e | n.e |
| Escherichia coli | n.e | 16 ± 1 |
| Species | Proteus mirabilis | Bacillus subtilis | ||
|---|---|---|---|---|
| MIC (mg/mL) | MBC (mg/mL) | MIC(mg/mL) | MBC(mg/mL) | |
| B. vulgaris subsp. maritima | ND | ND | 25 ± 0 | >25 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bouchmaa, N.; Mrid, R.B.; Kabach, I.; Zouaoui, Z.; Karrouchi, K.; Chtibi, H.; Zyad, A.; Cacciola, F.; Nhiri, M. Beta vulgaris subsp. maritima: A Valuable Food with High Added Health Benefits. Appl. Sci. 2022, 12, 1866. https://doi.org/10.3390/app12041866
Bouchmaa N, Mrid RB, Kabach I, Zouaoui Z, Karrouchi K, Chtibi H, Zyad A, Cacciola F, Nhiri M. Beta vulgaris subsp. maritima: A Valuable Food with High Added Health Benefits. Applied Sciences. 2022; 12(4):1866. https://doi.org/10.3390/app12041866
Chicago/Turabian StyleBouchmaa, Najat, Reda Ben Mrid, Imad Kabach, Zakia Zouaoui, Khalid Karrouchi, Houda Chtibi, Abdelmajid Zyad, Francesco Cacciola, and Mohamed Nhiri. 2022. "Beta vulgaris subsp. maritima: A Valuable Food with High Added Health Benefits" Applied Sciences 12, no. 4: 1866. https://doi.org/10.3390/app12041866
APA StyleBouchmaa, N., Mrid, R. B., Kabach, I., Zouaoui, Z., Karrouchi, K., Chtibi, H., Zyad, A., Cacciola, F., & Nhiri, M. (2022). Beta vulgaris subsp. maritima: A Valuable Food with High Added Health Benefits. Applied Sciences, 12(4), 1866. https://doi.org/10.3390/app12041866







