Propolis as a Potential Novel Histological Tissue Fixative: A Preliminary Analysis
Abstract
1. Introduction
2. Materials and Methods
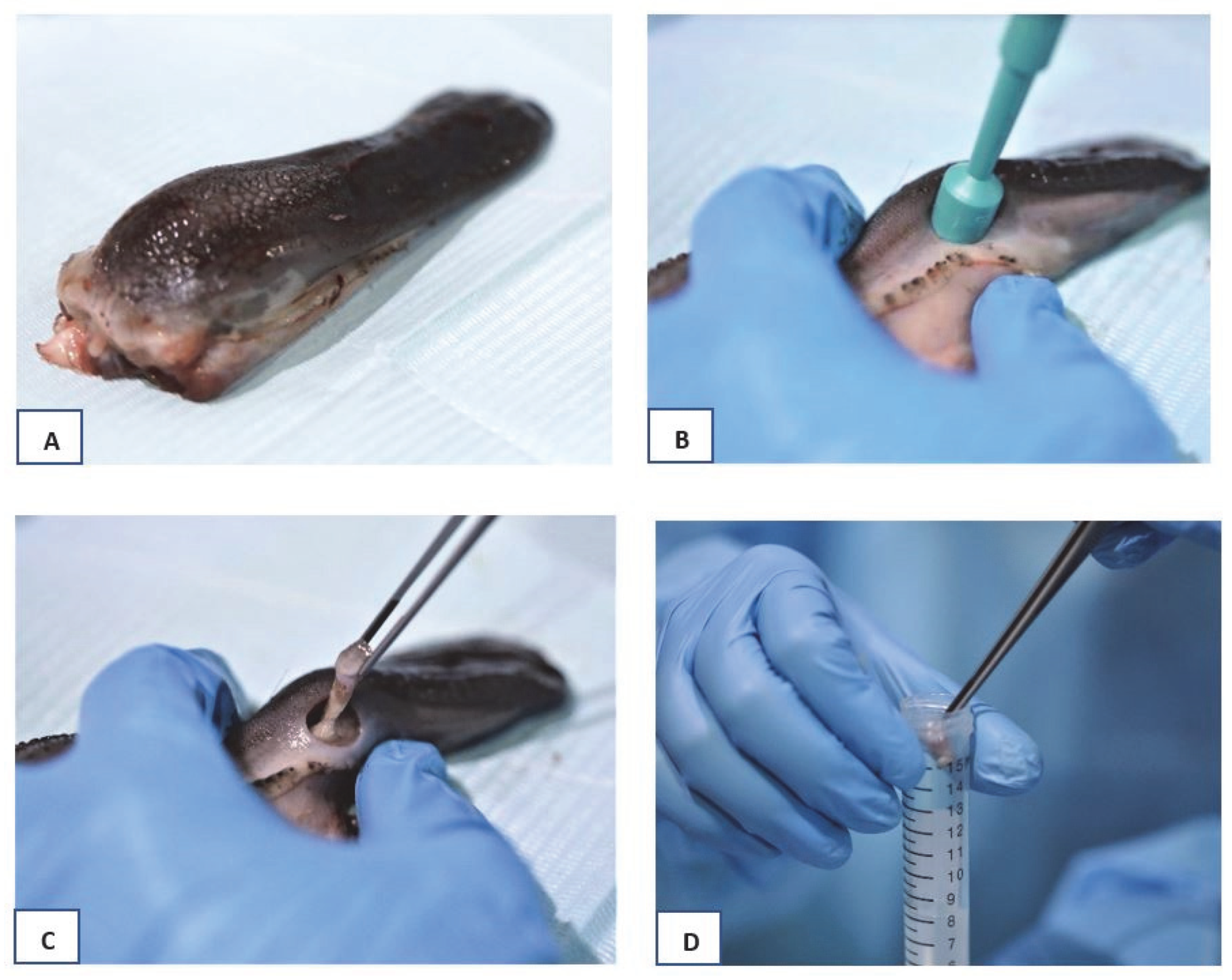
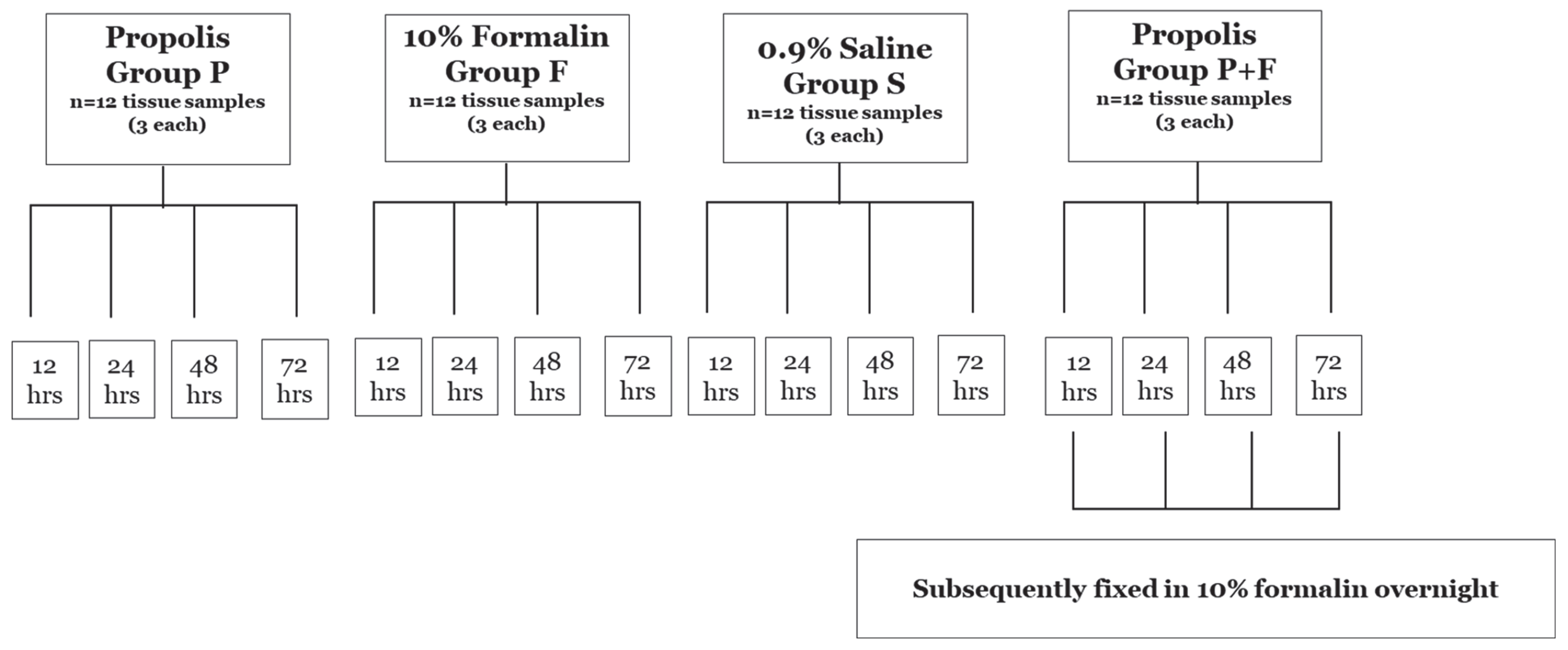
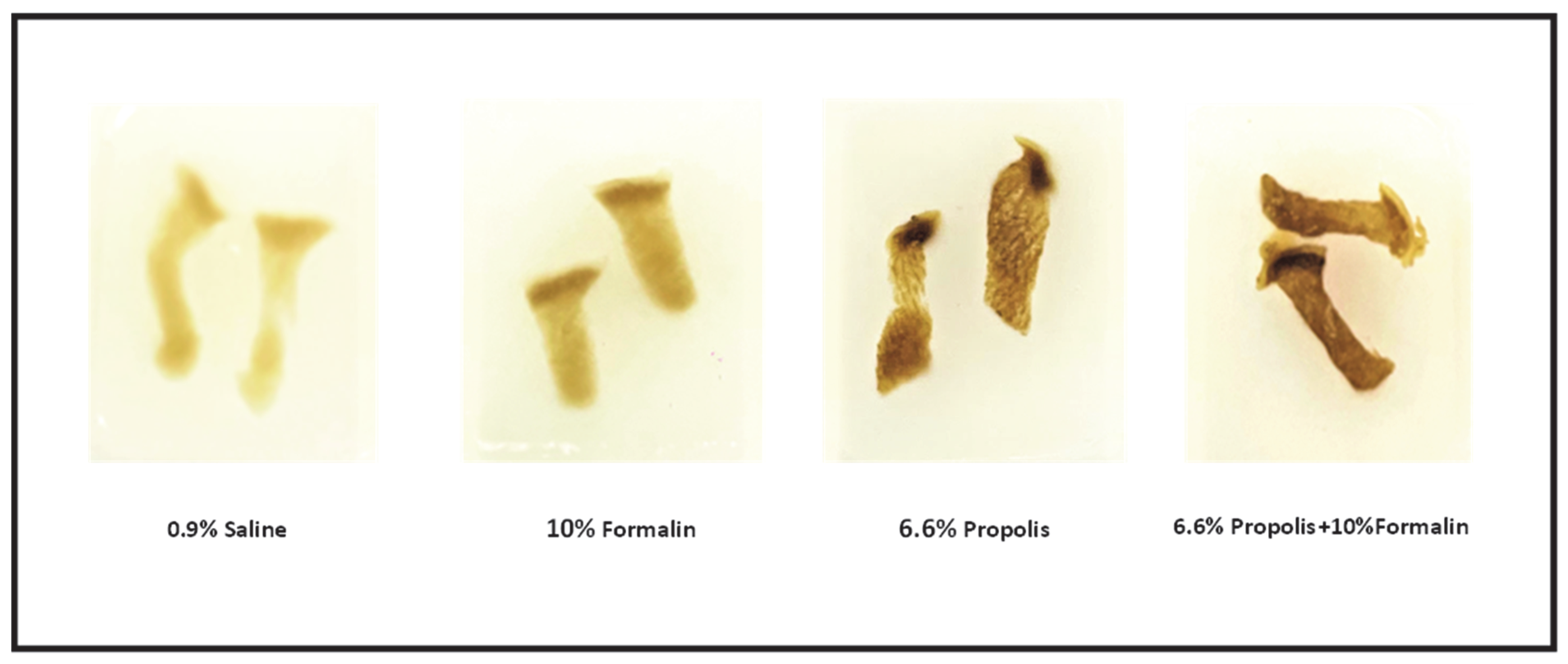
2.1. Tissue Samples Preparation
2.2. Preparation of Histological Slides
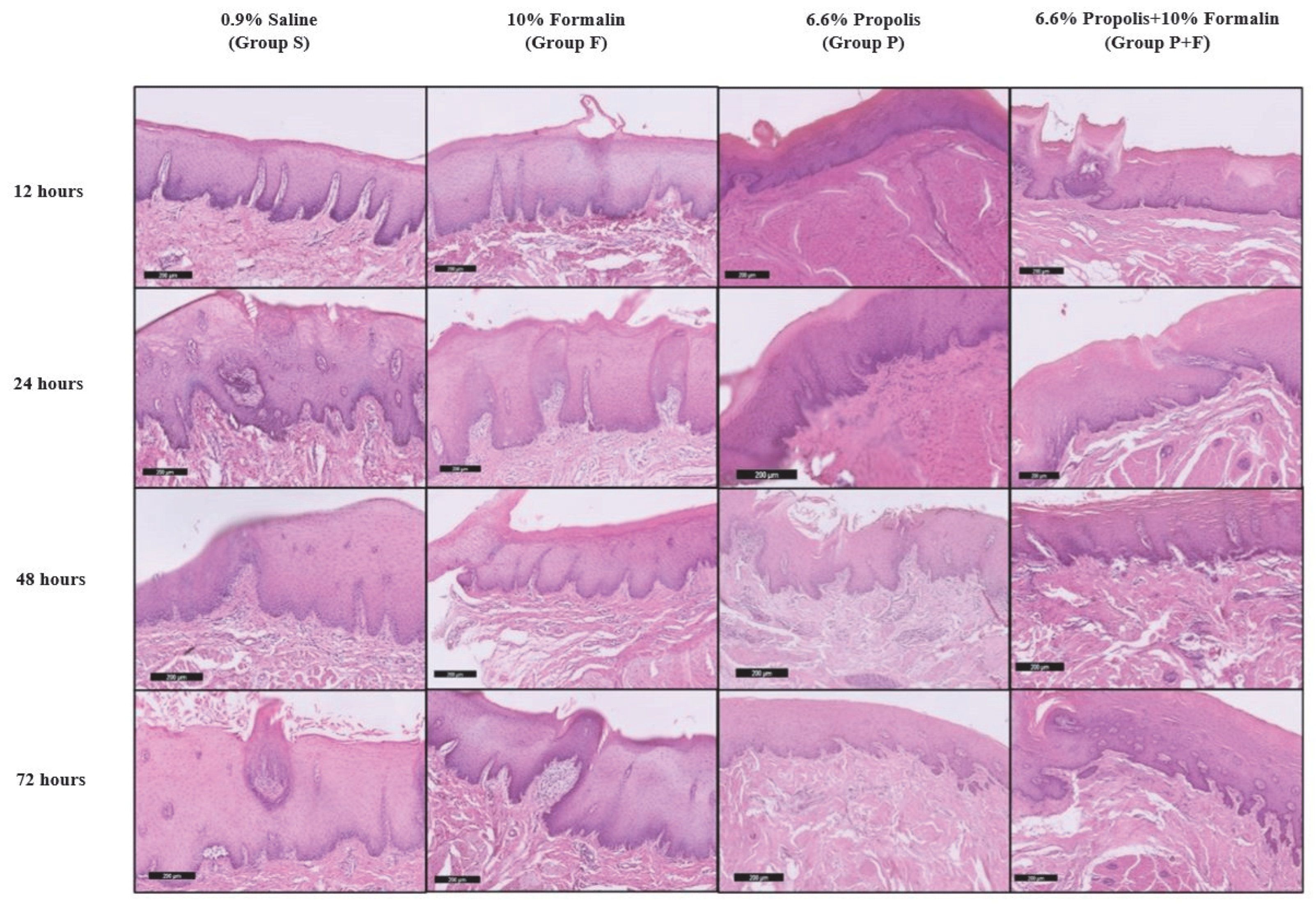
2.3. Histological Assessment
2.4. Statistical Analysis
3. Results
3.1. Examiners’ Agreement
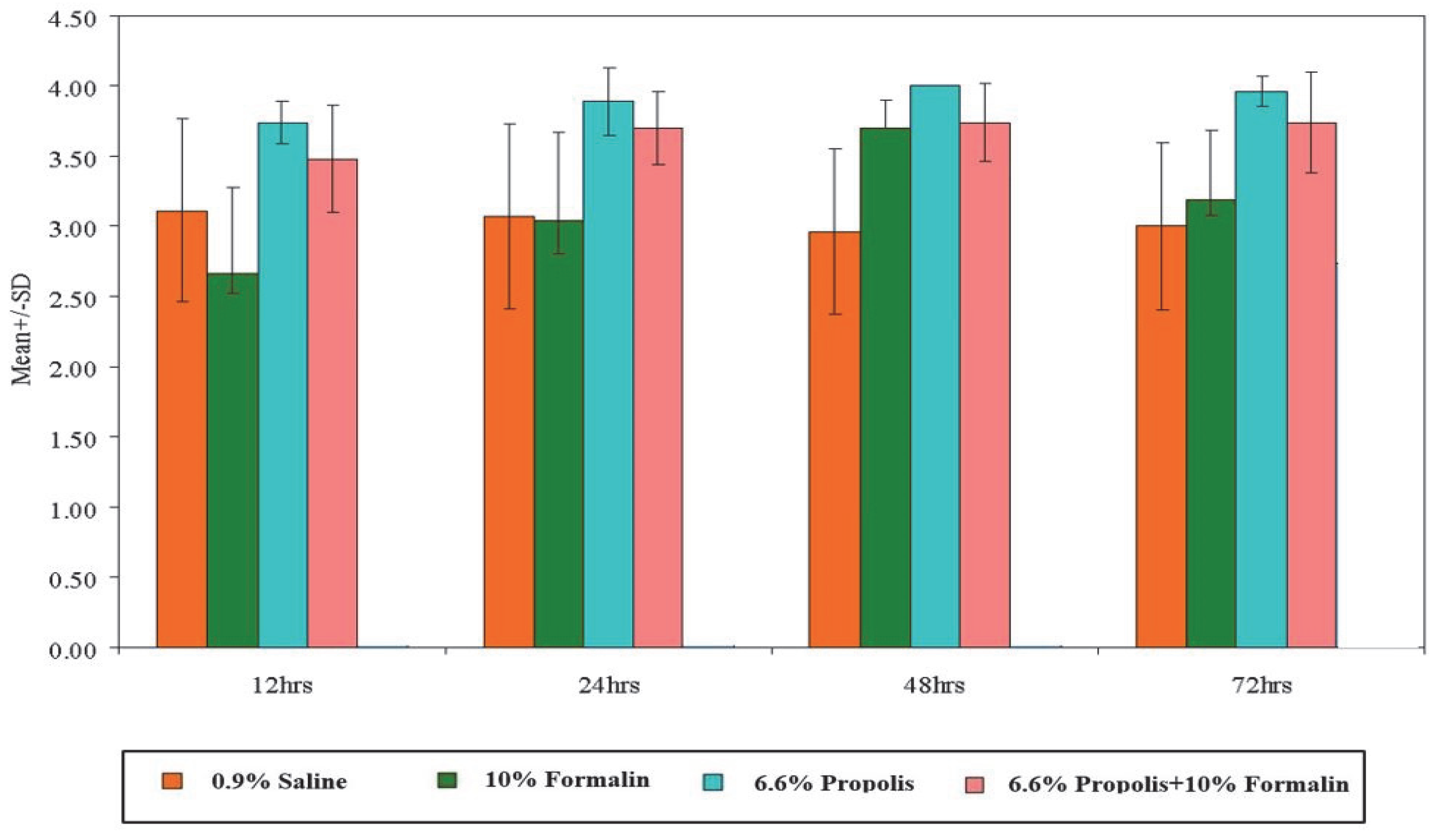
3.2. Comparison between Different Groups at Different Time Points
3.2.1. Twelve Hours
3.2.2. Twenty-Four Hours
3.2.3. Forty-Eight Hours
3.2.4. Seventy-Two Hours
3.3. Changes in the Mean Score at Different Points Compared with 12 h
3.3.1. Changes from 12 to 24 h
Between-Group Comparison
Within-Group Comparison
3.3.2. Changes from 12 to 48 h
Between-Group Comparison
Within-Group Comparison
3.3.3. Changes from 12 to 72 h
Between-Group Comparison
Within-Group Comparison
3.4. Comparison of Fixation Ability of Different Solutions throughout Various Microscopic Structures
4. Discussion
4.1. Study Limitations
4.2. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bancroft, J.D.; Gamble, M. Theory and Practice of Histological Techniques; Churchill Livingstone: Edinburgh, UK, 1996; p. 766. [Google Scholar]
- Bhat, A.H.; Hussein, S. Fixation and different types of fixatives: Their role and functions: A review. Int. J. Clin. Diagn. Pathol. 2021, 4, 113–119. [Google Scholar] [CrossRef]
- Thavarajah, R.; Mudimbaimannar, V.K.; Elizabeth, J.; Rao, U.K.; Ranganathan, K. Chemical and physical basics of routine formaldehyde fixation. J. Oral. Maxillofac. Pathol. 2012, 16, 400–405. [Google Scholar] [CrossRef] [PubMed]
- Grizzle, W.E. Special symposium: Fixation and tissue processing models. Biotechnol. Histochem. 2009, 84, 185–193. [Google Scholar] [CrossRef] [PubMed]
- Fox, C.H.; Johnson, F.B.; Whiting, J.; Roller, P.P. Formaldehyde fixation. J. Histochem. Cytochem. 1985, 33, 845–853. [Google Scholar] [CrossRef] [PubMed]
- Swenberg, J.A.; Moeller, B.C.; Lu, K.; Rager, J.E.; Fry, R.C.; Starr, T.B. Formaldehyde carcinogenicity research: 30 years and counting for mode of action, epidemiology, and cancer risk assessment. Toxicol. Pathol. 2013, 41, 181–189. [Google Scholar] [CrossRef]
- Piatek-Koziej, K.; Holda, J.; Koziej, M.; Tyrak, K.; Jasinska, K.A.; Bonczar, A.; Walocha, J.A.; Holda, M.K. Fixative properties of honey solutions as a formaldehyde substitute in cardiac tissue preservation. Folia Med. Cracov. 2019, 59, 101–114. [Google Scholar]
- Ramamoorthy, A.; Ravi, S.; Jeddy, N.; Thangavelu, R.; Janardhanan, S. Natural Alternatives for Chemicals Used in Histopathology Lab- A Literature Review. J. Clin. Diagn. Res. 2016, 10, EE01–EE04. [Google Scholar] [CrossRef]
- Swenberg, J.A.; Kerns, W.D.; Mitchell, R.I.; Gralla, E.J.; Pavkov, K.L. Induction of squamous cell carcinomas of the rat nasal cavity by inhalation exposure to formaldehyde vapor. Cancer Res. 1980, 40, 3398–3402. [Google Scholar]
- Hauptmann, M.; Stewart, P.A.; Lubin, J.H.; Beane Freeman, L.E.; Hornung, R.W.; Herrick, R.F.; Hoover, R.N.; Fraumeni, J.F., Jr.; Blair, A.; Hayes, R.B. Mortality from lymphohematopoietic malignancies and brain cancer among embalmers exposed to formaldehyde. J. Natl. Cancer Inst. 2009, 101, 1696–1708. [Google Scholar] [CrossRef]
- Kolayli, S.; Palabiyik, I.; Atik, D.; Keskin, M.; Bozdeveci, A.; Karaoglu, S. Comparison of Antibacterial and Antifungal Effects of Different Varieties of Honey and Propolis Samples. Acta Aliment. 2020, 49, 8. [Google Scholar] [CrossRef]
- Castaldo, S.; Capasso, F. Propolis, an old remedy used in modern medicine. Fitoterapia 2002, 73 (Suppl. S1), S1–S6. [Google Scholar] [CrossRef]
- Ahmed, R.; Tanvir, E.M.; Hossen, S.; Afroz, R.; Ahmmed, I.; Rumpa, N.-E.; Paul, S.; Gan, S.H.; Sulaiman, S.A.; Khalil, I. Antioxidant Properties and Cardioprotective Mechanism of Malaysian Propolis in Rats. Evid.-Based Complement. Altern. Med. 2017, 2017, 5370545. [Google Scholar] [CrossRef]
- Martinotti, S.; Ranzato, E. Propolis: A new frontier for wound healing? Burn. Trauma 2015, 3, 9. [Google Scholar] [CrossRef] [PubMed]
- Kubiliene, L.; Laugaliene, V.; Pavilonis, A.; Maruska, A.; Majiene, D.; Barcauskaite, K.; Kubilius, R.; Kasparaviciene, G.; Savickas, A. Alternative preparation of propolis extracts: Comparison of their composition and biological activities. BMC Complement. Altern. Med. 2015, 15, 156. [Google Scholar] [CrossRef] [PubMed]
- Bankova, V.S.; De Castro, S.L.; Marcucci, M.C. Propolis: Recent advances in chemistry and plant origin. Apidologie 2000, 31, 3–15. [Google Scholar] [CrossRef]
- Ahangari, Z.; Naseri, M.; Vatandoost, F. Propolis: Chemical Composition and Its Applications in Endodontics. Iran. Endod. J. 2018, 13, 285–292. [Google Scholar] [CrossRef] [PubMed]
- Farooqui, T. Beneficial effects of propolis on human health and neurological diseases. Front. Biosci. 2012, 4, 779–793. [Google Scholar] [CrossRef]
- Mavri, A.; Abramovič, H.; Polak, T.; Bertoncelj, J.; Jamnik, P.; Možina, S.S.; Jeršek, B. Chemical Properties and Antioxidant and Antimicrobial Activities of Slovenian Propolis. Chem. Biodivers. 2012, 9, 1545–1558. [Google Scholar] [CrossRef]
- Gjertsen, A.W.; Stothz, K.A.; Neiva, K.G.; Pileggi, R. Effect of propolis on proliferation and apoptosis of periodontal ligament fibroblasts. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endodontol. 2011, 112, 843–848. [Google Scholar] [CrossRef]
- Godkar, P.B.; Godkar, P.D. Textbook of Medical Laboratory Technology, 2nd ed.; Bhalani Publication House: Mumbai, India, 1996. [Google Scholar]
- Kasetty, S.; Dwivedi, D.; Ragavendra, T.R.; Kallianpur, S.; Gupta, S.; Prabhakar, N. Comparison of efficacy of local anesthetic solution, distilled water and normal saline as emergency fixatives. J. Oral Maxillofac. Pathol. 2018, 22, 283. [Google Scholar] [CrossRef]
- Landis, J.R.; Koch, G.G. The measurement of observer agreement for categorical data. Biometrics 1977, 33, 159–174. [Google Scholar] [CrossRef] [PubMed]
- Easton-Calabria, A.; Demary, K.C.; Oner, N.J. Beyond Pollination: Honey Bees (Apis mellifera) as Zootherapy Keystone Species. Front. Ecol. Evol. 2019, 6, 161. [Google Scholar] [CrossRef]
- Sabarinath, B.; Sivapathasundharam, B.; Sathyakumar, M. Fixative properties of honey in comparison with formalin. J. Histotechnol. 2014, 37, 5. [Google Scholar] [CrossRef]
- Patil, S.; Premalatha, B.; Rao, R.S.; Ganavi, B. Revelation in the field of tissue preservation—A preliminary study on natural formalin substitutes. J. Int. Oral Health 2013, 5, 31–38. [Google Scholar] [PubMed]
- Patil, S.; Rao, R.S.; Ganavi, B.S.; Majumdar, B. Natural sweeteners as fixatives in histopathology: A longitudinal study. J. Nat. Sci. Biol. Med. 2015, 6, 67–70. [Google Scholar] [CrossRef]
- Al-Maaini, R.; Bryant, P. The Effectiveness of Honey as a Substitute for Formalin in the Histological Fixation of Tissue. J. Histotechnol. 2013, 29, 173–176. [Google Scholar] [CrossRef]
- Lalwani, V.; Surekha, R.; Vanishree, M.; Koneru, A.; Hunasgi, S.; Ravikumar, S. Honey as an alternative fixative for oral tissue: An evaluation of processed and unprocessed honey. J. Oral. Maxillofac. Pathol. 2015, 19, 342–347. [Google Scholar] [CrossRef]
- Kuriachan, D.; Suresh, R.; Janardhanan, M.; Savithri, V.; Aravind, T.; Thampy, L. Analysis of Fixative Properties of Three Eco-friendly Substances: A Comparison with Formalin. Oral Maxillofac. Pathol. J. 2017, 8, 79–84. [Google Scholar]
- Bachevski, D.; Damevska, K.; Simeonovski, V.; Dimova, M. Back to the basics: Propolis and COVID-19. Dermatol. Ther. 2020, 33, 3–5. [Google Scholar] [CrossRef]


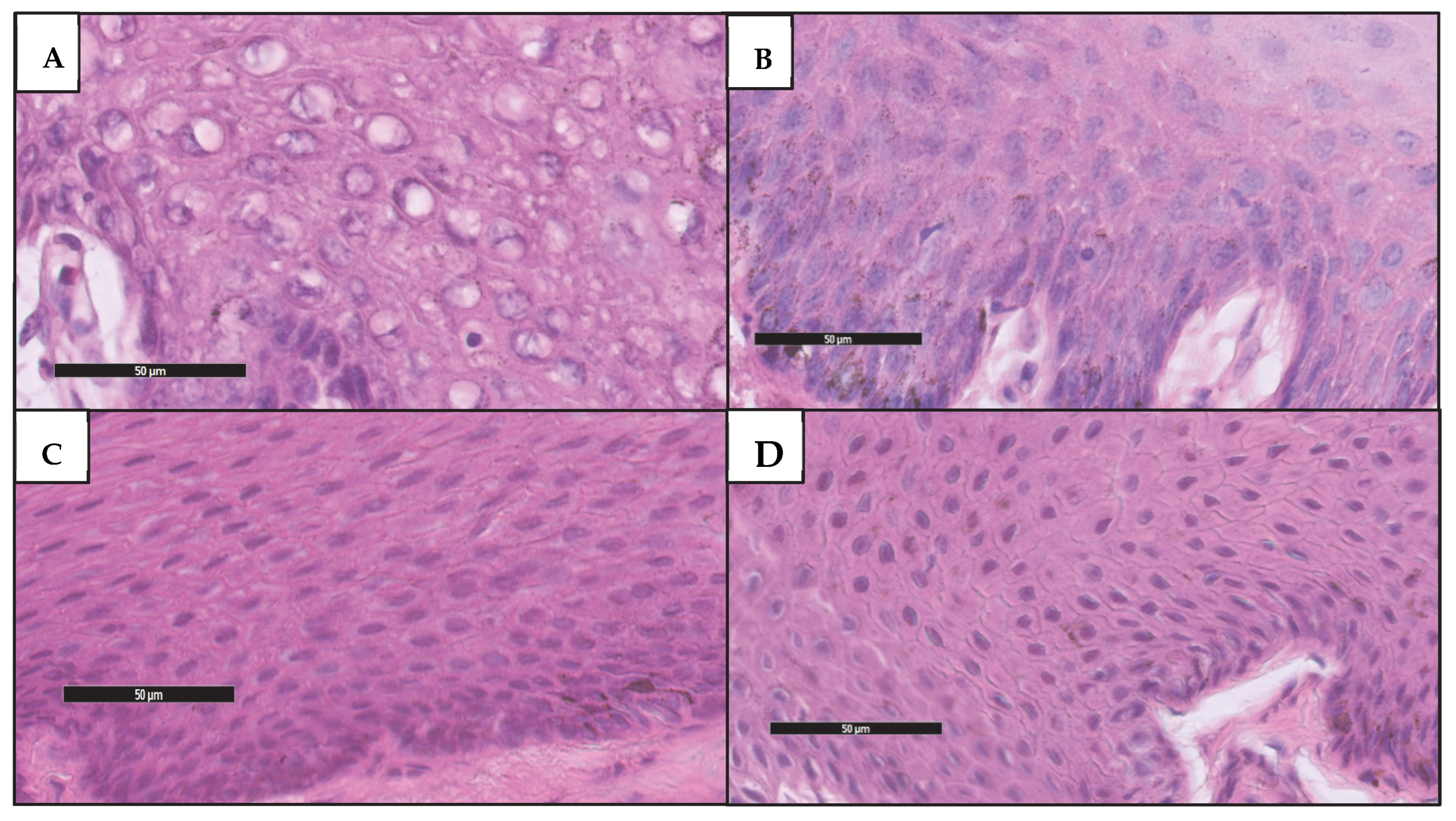
| Score | Histological Criteria |
|---|---|
| 4 | No apparent change |
| 3 | Minor changes that do not affect the diagnosis |
| 2 | Minor changes that may affect the diagnosis |
| 1 | Major changes that affect the diagnosis |
| Fixative Agents | Agreement | Kappa | Strength of Agreement |
|---|---|---|---|
| 0.9% Saline | 83.33% | 0.47 | Moderate |
| 10% Formalin | 77.78% | 0.55 | Moderate |
| 6.6% Propolis | 88.89% | 0.61 | Substantial |
| 6.6% Propolis + 10% Formalin | 77.78% | 0.57 | Moderate |
| Solution | 12 h | 24 h | 48 h | 72 h | ||||
|---|---|---|---|---|---|---|---|---|
| Mean | SD § | Mean | SD | Mean | SD | Mean | SD | |
| 0.9% Saline | 3.11 | 0.65 | 3.07 | 0.66 | 2.96 | 0.59 | 3.00 | 0.60 |
| 10% Formalin | 2.67 | 0.60 | 3.04 | 0.63 | 3.70 | 0.20 | 3.19 | 0.50 |
| 6.6% Propolis | 3.74 | 0.15 | 3.89 | 0.24 | 4.00 | 0.00 | 3.96 | 0.11 |
| 6.6% Propolis + 10% Formalin | 3.48 | 0.38 | 3.70 | 0.26 | 3.74 | 0.28 | 3.74 | 0.36 |
| Pairwise comparisons by Tukey’s multiple post hoc procedures | ||||||||
| 6.6% Saline vs. 10% Formalin | p = 0.3702 | p = 0.9999 | p = 0.0006 * | p = 0.9037 | ||||
| 0.9% Saline vs. 6.6% Propolis | p = 0.0905 | p = 0.0129 * | p = 0.0001 * | p = 0.0006 * | ||||
| 0.9% Saline vs. 6.6% Propolis + 10% Formalin | p = 0.5518 | p = 0.0852 | p = 0.0004 * | p = 0.0096 * | ||||
| 10% Formalin vs. 6.6% Propolis | p = 0.0008 * | p = 0.0086 * | p = 0.3814 | p = 0.0059 * | ||||
| 10% Formalin vs. 6.6% Propolis + 10% Formalin | p = 0.0142 * | p = 0.0602 | p = 0.9994 | p = 0.0837 | ||||
| 6.6% Propolis vs. 6.6% Propolis + 10% Formalin | p = 0.8213 | p = 0.9374 | p = 0.5153 | p = 0.8293 | ||||
| Solution | Changes from 12 h to | |||||
|---|---|---|---|---|---|---|
| 24 h | 48 h | 72 h | ||||
| Mean | SD § | Mean | SD | Mean | SD | |
| 0.9% Saline | 0.04 | 0.45 | 0.15 | 0.38 | 0.11 | 0.53 |
| 10% Formalin | −0.37 | 0.39 | −1.04 | 0.51 | −0.52 | 0.34 |
| 6.6% Propolis | −0.15 | 0.24 | −0.26 | 0.15 | −0.22 | 0.17 |
| 6.6% Propolis + 10% Formalin | −0.22 | 0.24 | −0.26 | 0.28 | −0.26 | 0.22 |
| Pairwise comparisons by Tukey’s multiple post hoc procedures | ||||||
| 0.9% Saline vs. 10% Formalin | p = 0.1569 | p = 0.0001 * | p = 0.0152 * | |||
| 0.9% Saline vs. 6.6% Propolis | p = 0.8262 | p = 0.2024 | p = 0.4077 | |||
| 0.9% Saline vs. 6.6%Propolis + 10% Formalin | p = 0.5797 | p = 0.2024 | p = 0.3033 | |||
| 10% Formalin vs. 6.6% Propolis | p = 0.7104 | p = 0.0014 * | p = 0.5254 | |||
| 10% Formalin vs. 6.6% Propolis + 10% Formalin | p = 0.9142 | p = 0.0014 * | p = 0.6482 | |||
| 6.6% Propolis vs. 6.6% Propolis + 10%Formalin | p = 0.9931 | p = 1.0000 | p = 0.9997 | |||
| Solution | Change (time) | Mean Diff. ‡ | SD Diff. † | Changes (%) | p-Value |
|---|---|---|---|---|---|
| 0.9% Saline | 12 to 24 h | 0.04 | 0.45 | 1.19 | 0.8131 |
| 12 to 48 h | 0.15 | 0.38 | 4.76 | 0.2721 | |
| 12 to 72 h | 0.11 | 0.53 | 3.57 | 0.5447 | |
| 10% Formalin | 12 to 24 h | −0.37 | 0.39 | −13.89 | 0.0212 * |
| 12 to 48 h | −1.04 | 0.51 | −38.89 | 0.0003 * | |
| 12 to 72 h | −0.52 | 0.34 | −19.44 | 0.0017 * | |
| 6.6% Propolis | 12 to 24 h | −0.15 | 0.24 | −3.96 | 0.1038 |
| 12 to 48 h | −0.26 | 0.15 | −6.93 | 0.0007 * | |
| 12 to 72 h | −0.22 | 0.17 | −5.94 | 0.0039 * | |
| 6.6% Propolis + 10% Formalin | 12 to 24 h | −0.22 | 0.24 | −6.38 | 0.0222 * |
| 12 to 48 h | −0.26 | 0.28 | −7.45 | 0.0232 * | |
| 12 to 72 h | −0.26 | 0.22 | −7.45 | 0.0081 * |
| (a) | ||||||
|---|---|---|---|---|---|---|
| Solution | Epithelium | |||||
| Superficial 1/3rd | Middle 1/3rd | Lower 1/3rd | ||||
| Mean | SD § | Mean | SD | Mean | SD | |
| 0.9% Saline | 2.58 | 0.44 | 2.83 | 0.30 | 3.42 | 0.10 |
| 10% Formalin | 3.12 | 0.39 | 3.21 | 0.29 | 3.41 | 0.22 |
| 6.6% Propolis | 3.37 | 0.16 | 3.21 | 0.21 | 3.41 | 0.22 |
| 6.6% Propolis + 10% Formalin | 3.16 | 0.36 | 3.21 | 0.16 | 3.50 | 0.36 |
| Pairwise comparisons by Tukey’s multiple post hoc procedures | ||||||
| 0.9% Saline vs. 10% Formalin | p = 0.2342 | p = 0.4502 | p = 1.0000 | |||
| 0.9% Saline vs. 6.6% Propolis | p = 0.0388 * | p = 0.4502 | p = 1.0000 | |||
| 0.9% Saline vs. 6.6% Propolis + 10% Formalin | p = 0.1779 | p = 0.4502 | p = 0.9859 | |||
| 10% Formalin vs. 6.6% Propolis | p = 0.8374 | p = 1.0000 | p = 1.0000 | |||
| 10% Formalin vs. 6.6% Propolis + 10% Formalin | p = 0.9998 | p = 1.0000 | p = 0.9842 | |||
| 10% Propolis vs. 6.6% Propolis + 10% Formalin | p = 0.9080 | p = 1.0000 | p = 0.9842 | |||
| (b) | ||||||
| Solution | Basement Membrane | Nerve Bundles | Fibrous Tissue | |||
| Mean | SD | Mean | SD | Mean | SD | |
| 0.9% Saline | 3.62 | 0.16 | 3.66 | 0.13 | 2.83 | 0.20 |
| 10% Formalin | 3.70 | 0.09 | 3.66 | 0.24 | 2.92 | 0.59 |
| 6.6% Propolis | 3.58 | 0.21 | 3.71 | 0.29 | 3.25 | 0.10 |
| 6.6% Propolis + 10% Formalin | 3.87 | 0.09 | 3.54 | 0.21 | 3.16 | 0.24 |
| 0.9% Saline + 10% Formalin | 3.54 | 0.44 | 3.46 | 0.44 | 2.79 | 0.48 |
| Pairwise comparisons by Tukey’s multiple post hoc procedures | ||||||
| 0.9% Saline vs. 10% Formalin | p = 0.9883 | p = 1.0000 | p = 0.9973 | |||
| 0.9% Saline vs. 6.6% Propolis | p = 0.9990 | p = 0.9995 | p = 0.5225 | |||
| 0.9% Saline vs. 6.6% Propolis + 10% Formalin | p = 0.5814 | p = 0.9697 | p = 0.7081 | |||
| 10% Formalin vs. 6.6% Propolis | p = 0.9456 | p = 0.9995 | p = 0.7136 | |||
| 10% Formalin vs. 6.6% Propolis + 10% Formalin | p = 0.8442 | p = 0.9697 | p = 0.8724 | |||
| 6.6% Propolis vs. 6.6% Propolis + 10% Formalin | p = 0.4367 | p = 0.9157 | p = 0.9976 | |||
| (c) | ||||||
| Solution | Blood Vessels | Skeletal Muscles | Adipose Tissue | |||
| Mean | SD | Mean | SD | Mean | SD | |
| 0.9% Saline | 3.71 | 0.29 | 3.04 | 0.29 | 3.29 | 0.25 |
| 10% Formalin | 3.79 | 0.16 | 2.75 | 0.40 | 3.29 | 0.34 |
| 6.6% Propolis | 3.87 | 0.09 | 3.16 | 0.24 | 3.08 | 0.35 |
| 6.6% Propolis + 10% Formalin | 3.66 | 0.13 | 2.96 | 0.21 | 2.87 | 0.16 |
| 0.9% Saline + 10% Formalin | 3.50 | 0.19 | 2.71 | 0.37 | 2.58 | 0.57 |
| Pairwise comparisons by Tukey’s multiple post hoc procedures | ||||||
| 0.9% Saline vs. 10% Formalin | p = 0.9673 | p = 0.6760 | p = 1.0000 | |||
| 0.9% Saline vs. 6.6% Propolis | p = 0.7040 | p = 0.9769 | p = 0.9195 | |||
| 0.9% Saline vs. 6.6% Propolis + 10% Formalin | p = 0.9973 | p = 0.9952 | p = 0.4988 | |||
| 10% Formalin vs. 6.6% Propolis | p = 0.9636 | p = 0.3547 | p = 0.9226 | |||
| 10% Formalin vs. 6.6% Propolis + 10% Formalin | p = 0.8691 | p = 0.8711 | p = 0.5044 | |||
| 6.6% Propolis vs. 6.6% Propolis + 10% Formalin | p = 0.5132 | p = 0.8711 | p = 0.9226 | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bugshan, A.S.; AlJanobi, H.A.; AlMunif, R.A.; AlShubaili, R.R.; AlHarbi, N.M.; Khusheim, S.A.; AlShuyukh, M.H.; Khan, A.M. Propolis as a Potential Novel Histological Tissue Fixative: A Preliminary Analysis. Appl. Sci. 2022, 12, 9842. https://doi.org/10.3390/app12199842
Bugshan AS, AlJanobi HA, AlMunif RA, AlShubaili RR, AlHarbi NM, Khusheim SA, AlShuyukh MH, Khan AM. Propolis as a Potential Novel Histological Tissue Fixative: A Preliminary Analysis. Applied Sciences. 2022; 12(19):9842. https://doi.org/10.3390/app12199842
Chicago/Turabian StyleBugshan, Amr S., Hawra A. AlJanobi, Raghad A. AlMunif, Rand R. AlShubaili, Nawal M. AlHarbi, Sarah A. Khusheim, Muneer H. AlShuyukh, and Asim M. Khan. 2022. "Propolis as a Potential Novel Histological Tissue Fixative: A Preliminary Analysis" Applied Sciences 12, no. 19: 9842. https://doi.org/10.3390/app12199842
APA StyleBugshan, A. S., AlJanobi, H. A., AlMunif, R. A., AlShubaili, R. R., AlHarbi, N. M., Khusheim, S. A., AlShuyukh, M. H., & Khan, A. M. (2022). Propolis as a Potential Novel Histological Tissue Fixative: A Preliminary Analysis. Applied Sciences, 12(19), 9842. https://doi.org/10.3390/app12199842






