Sensing Hydrogen Seeps in the Subsurface for Natural Hydrogen Exploration
Abstract
:1. Introduction
2. Materials and Methods
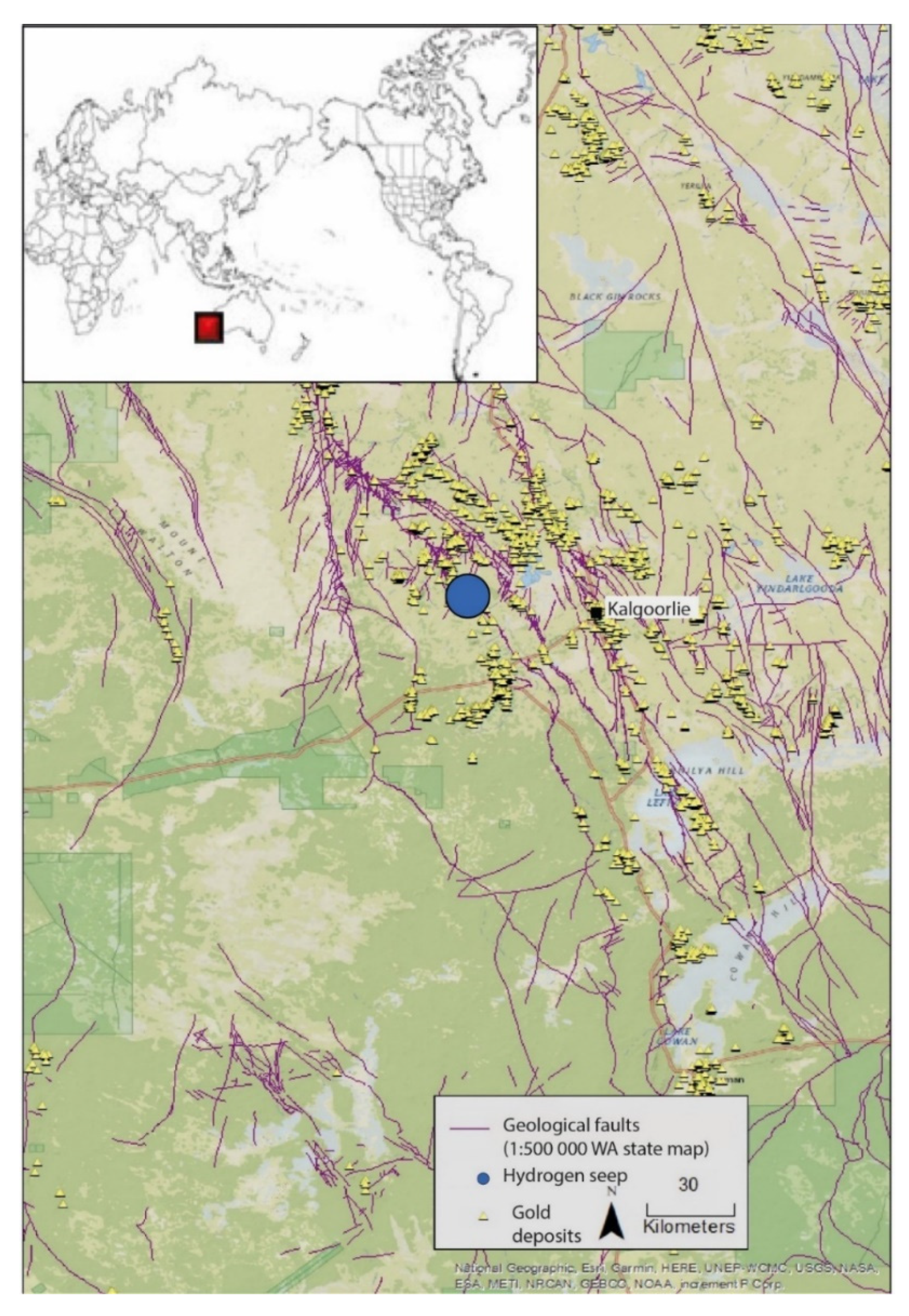
2.1. Sampling Site and Geology
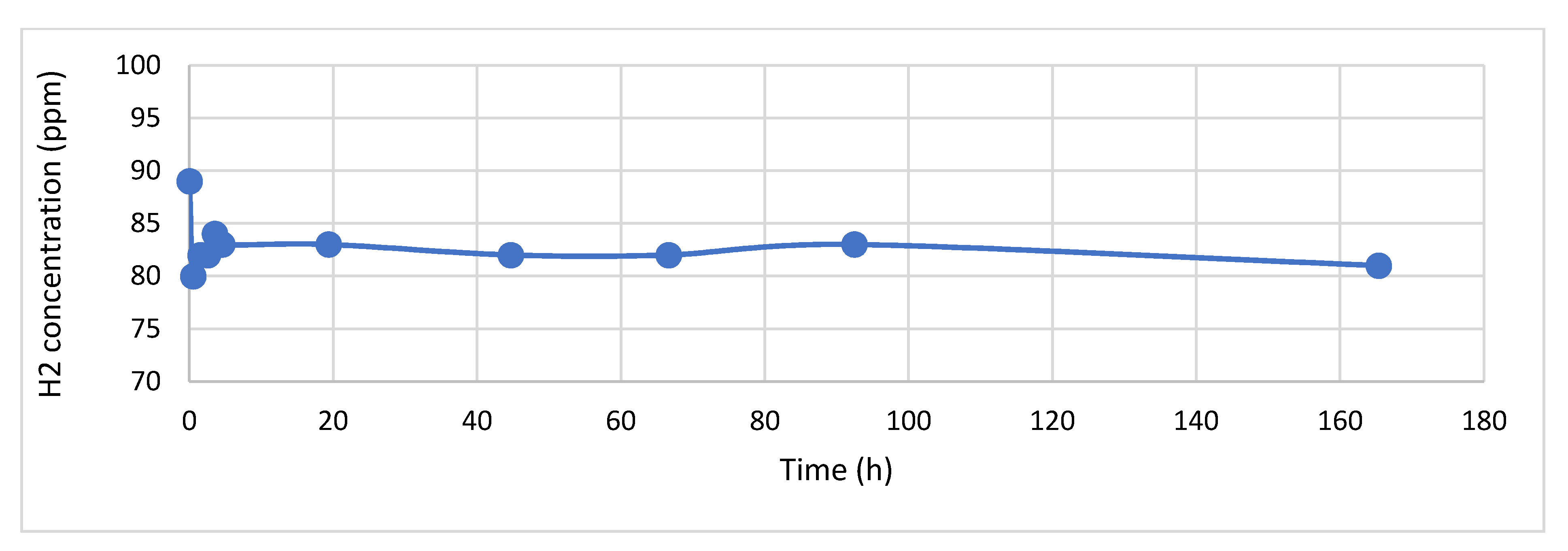
2.2. Sensor Stability
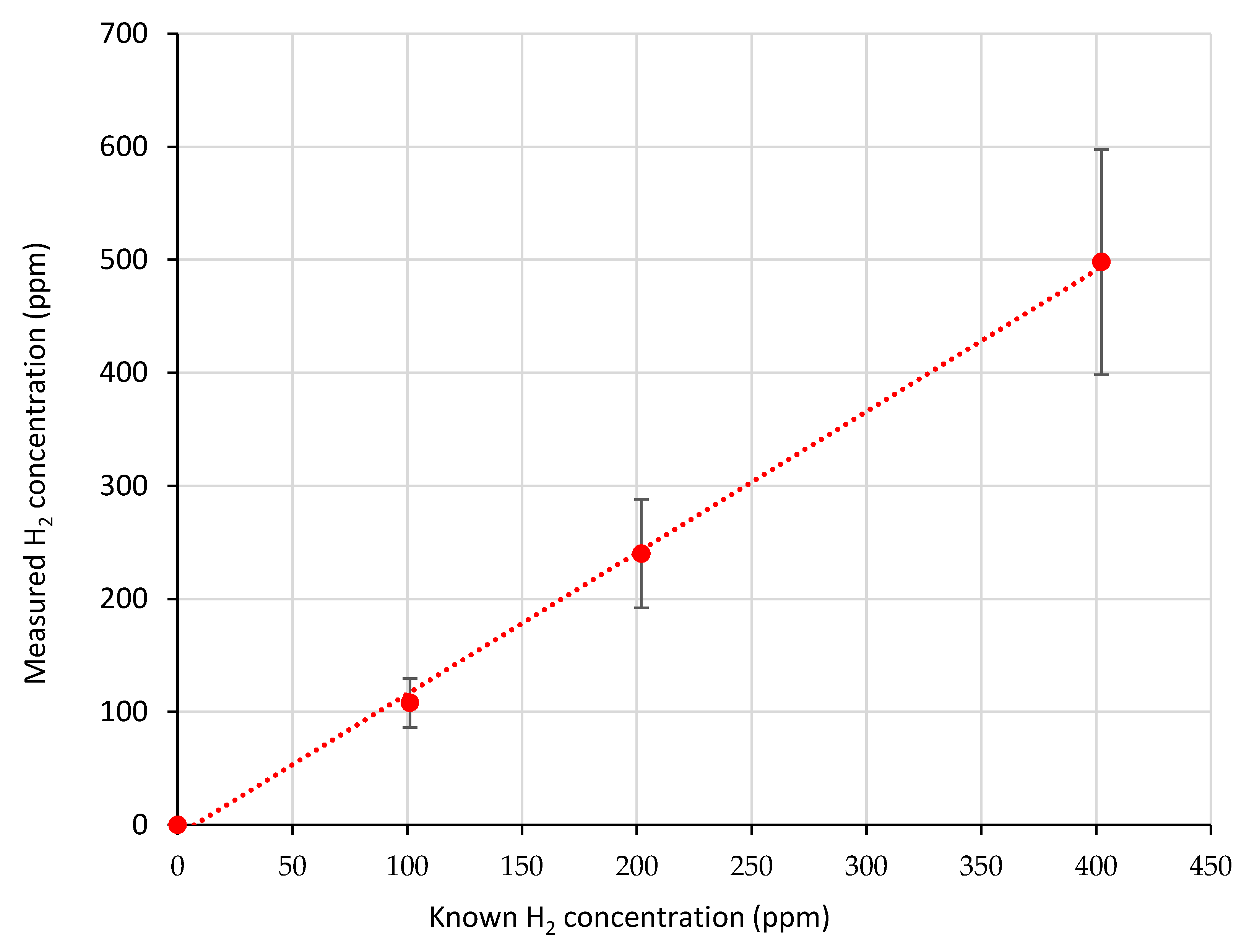
2.3. Sensor Linearity and Effect of Methane
2.4. Field Measurements
3. Results and Discussion
3.1. Sensor Response and Stability
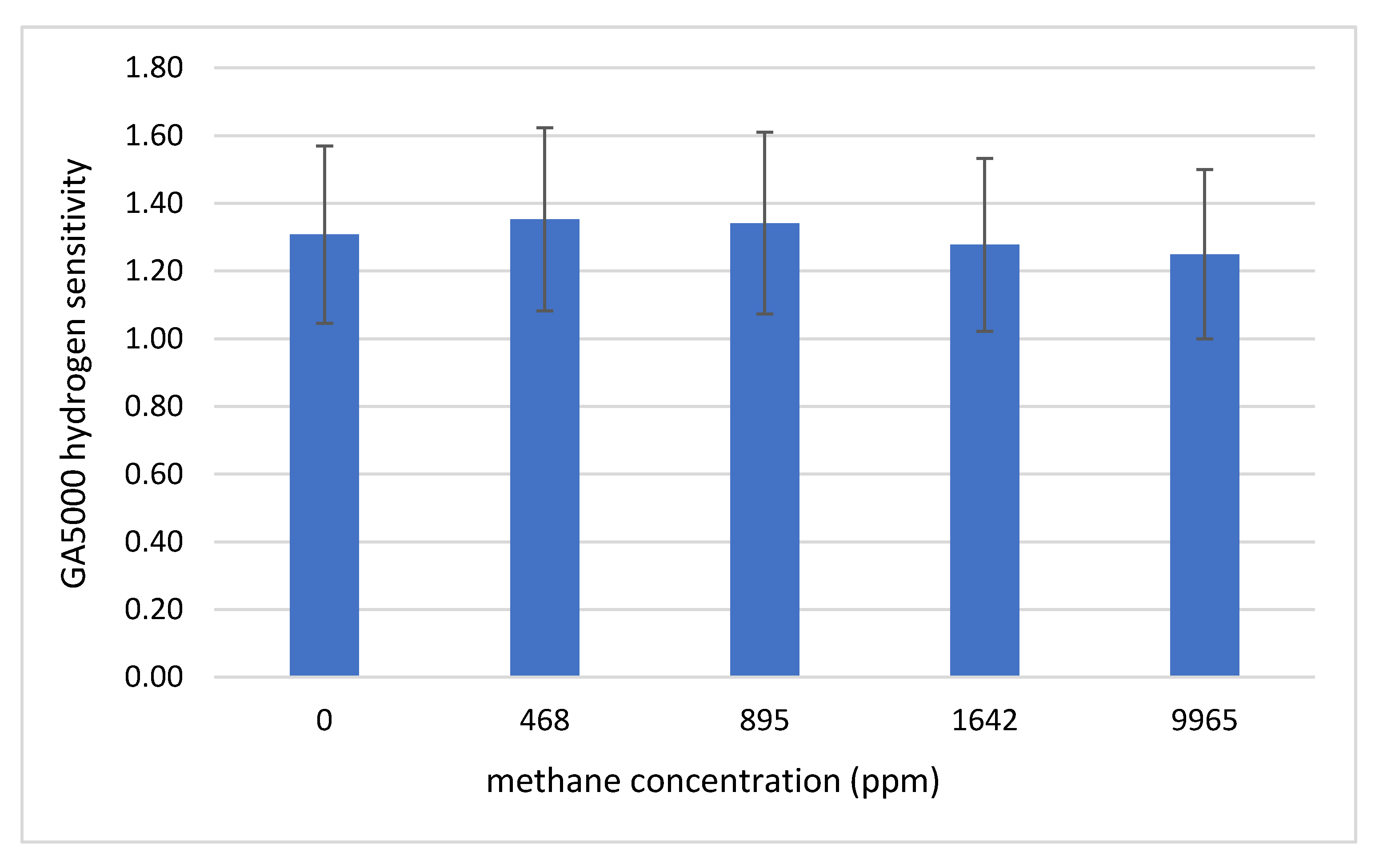
3.2. Effect of Methane Concentration
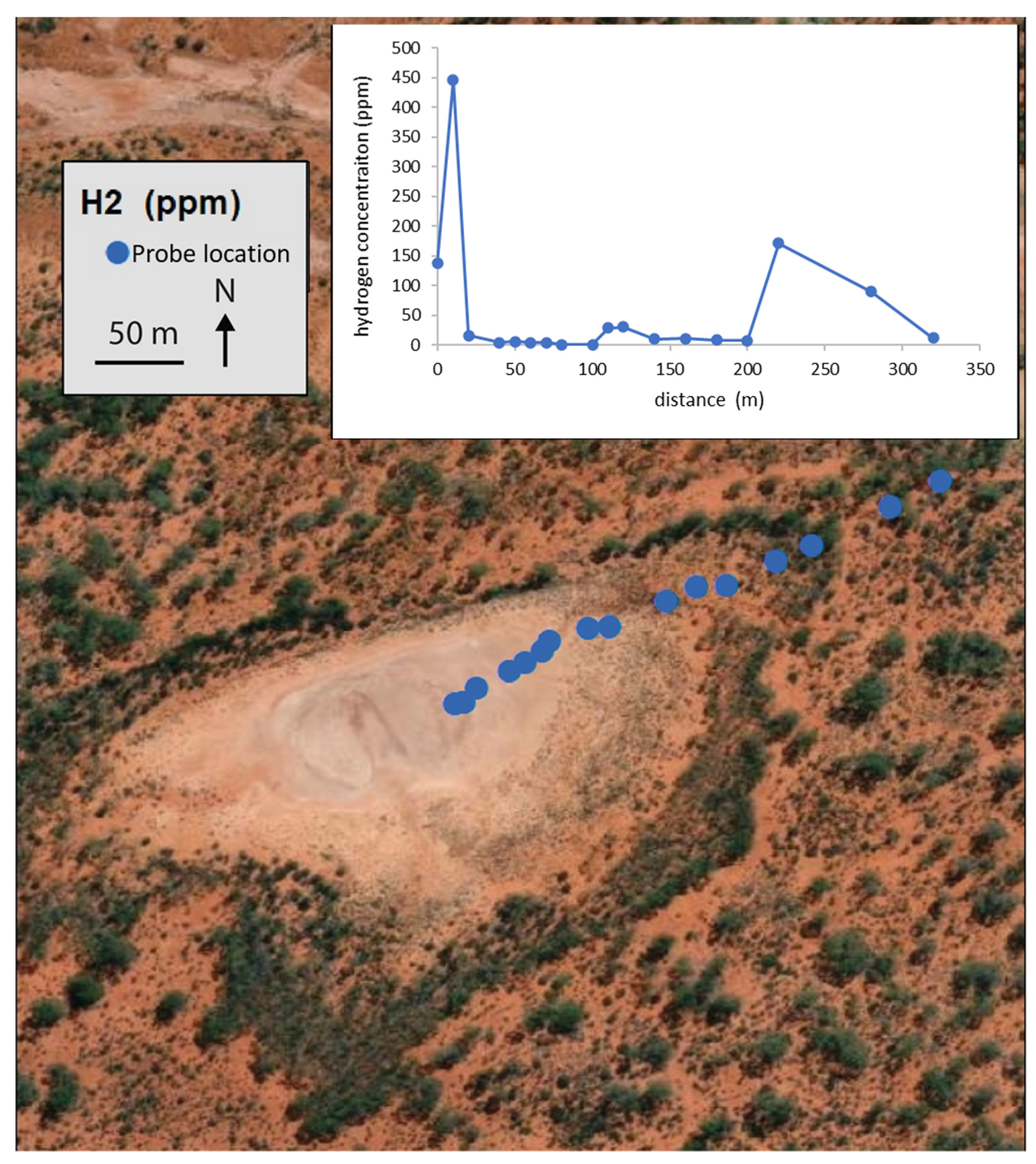
3.3. Hydrogen Seep Detection
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Global Hydrogen Review 2021; IEA: Paris, France, 2021.
- The Future of Hydrogen; IEA: Paris, France, 2019.
- Gaucher, E. New Perspectives in the Industrial Exploration for Native Hydrogen. Elements 2020, 16, 8–9. [Google Scholar] [CrossRef] [Green Version]
- Deville, E.; Prinzhofer, A. Hydrogène Naturel. La Prochaine Révolution Énergétique? Belin Éditeur: Paris, France, 2015. [Google Scholar]
- Prinzhofer, A.; Tahara Cissé, C.S.; Diallo, A.B. Discovery of a large accumulation of natural hydrogen in Bourakebougou (Mali). Int. J. Hydrog. Energy 2018, 43, 19315–19326. [Google Scholar] [CrossRef]
- Van Dijk, J.; Affinito, V.; Atena, R.; Caputi, A.; Cestari, A.; D’Elia, S.; Giancipoli, N.; Lanzellotti, M.; Lazzari, M.; Oriolo, N.; et al. Cento Anni di Ricerca Petrolifera: L’Alta Val d’Agri: Basilicata, Italia meridionale. In Proceedings of the Atti del 1° Congresso dell’Ordine dei Geologi di Basilicata,“Ricerca, Sviluppo ed Utilizzo delle Fonti Fossili: Il Ruolo del Geologo”, Potenza, Italy, 30 November–2 December 2012. [Google Scholar]
- Link, W.K. Significance of Oil and Gas Seeps in World Oil Exploration1. AAPG Bull. 1952, 36, 1505–1540. [Google Scholar]
- Larin, N.; Zgonnik, V.; Rodina, S.; Deville, E.; Prinzhofer, A.; Larin, V.N. Natural Molecular Hydrogen Seepage Associated with Surficial, Rounded Depressions on the European Craton in Russia. Nat. Resour. Res. 2015, 24, 369–383. [Google Scholar] [CrossRef]
- Moretti, I.; Brouilly, E.; Loiseau, K.; Prinzhofer, A.; Deville, E. Hydrogen Emanations in Intracratonic Areas: New Guide Lines for Early Exploration Basin Screening. Geosciences 2021, 11, 145. [Google Scholar] [CrossRef]
- Prinzhofer, A.; Moretti, I.; Françolin, J.; Pacheco, C.; D’Agostino, A.; Werly, J.; Rupin, F. Natural hydrogen continuous emission from sedimentary basins: The example of a Brazilian H2-emitting structure. Int. J. Hydrog. Energy 2019, 44, 5676–5685. [Google Scholar] [CrossRef]
- Zgonnik, V.; Beaumont, V.; Deville, E.; Larin, N.; Pillot, D.; Farrell, K.M. Evidence for natural molecular hydrogen seepage associated with Carolina bays (surficial, ovoid depressions on the Atlantic Coastal Plain, Province of the USA). Prog. Earth Planet. Sci. 2015, 2, 31. [Google Scholar] [CrossRef] [Green Version]
- Frery, E.; Langhi, L.; Maison, M.; Moretti, I. Natural hydrogen seeps identified in the North Perth Basin, Western Australia. Int. J. Hydrog. Energy 2021, 46, 31158–31173. [Google Scholar] [CrossRef]
- Dugamin, E.; Truche, L.; Donze, F. Natural Hydrogen Exploration Guide. 2019. Available online: https://www.researchgate.net/publication/330728855_Natural_Hydrogen_Exploration_Guide (accessed on 15 November 2021).
- Moretti, I.; Prinzhofer, A.; Françolin, J.; Pacheco, C.; Rosanne, M.; Rupin, F.; Mertens, J. Long-term monitoring of natural hydrogen superficial emissions in a brazilian cratonic environment. Sporadic large pulses versus daily periodic emissions. Int. J. Hydrog. Energy 2021, 46, 3615–3628. [Google Scholar] [CrossRef]
- Chauhan, P.S.; Bhattacharya, S. Hydrogen gas sensing methods, materials, and approach to achieve parts per billion level detection: A review. Int. J. Hydrog. Energy 2019, 44, 26076–26099. [Google Scholar] [CrossRef]
- Hübert, T.; Boon-Brett, L.; Black, G.; Banach, U. Hydrogen sensors—A review. Sens. Actuators B Chem. 2011, 157, 329–352. [Google Scholar] [CrossRef]
- Sharma, B.; Sharma, A.; Kim, J.-S. Recent advances on H2 sensor technologies based on MOX and FET devices: A review. Sens. Actuators B Chem. 2018, 262, 758–770. [Google Scholar] [CrossRef]
- Korotcenkov, G.; Han, S.D.; Stetter, J.R. Review of Electrochemical Hydrogen Sensors. Chem. Rev. 2009, 109, 1402–1433. [Google Scholar] [CrossRef] [PubMed]
- Avetisov, V.; Bjoroey, O.; Wang, J.; Geiser, P.; Paulsen, K.G. Hydrogen Sensor Based on Tunable Diode Laser Absorption Spectroscopy. Sensors 2019, 19, 5313. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Adler-Golden, S.M.; Goldstein, N.; Bien, F.; Matthew, M.W.; Gersh, M.E.; Cheng, W.K.; Adams, F.W. Laser Raman sensor for measurement of trace-hydrogen gas. Appl. Opt. 1992, 31, 831–835. [Google Scholar] [CrossRef]
- GA5000. Available online: https://www.airmet.com.au/assets/documents/product/301/GA5000-Datasheet.pdf (accessed on 13 June 2022).
- Government of Western Australia Department of Mines, Industry Regulation and Safety. 1:500,000 State Interpreted Bedrock Geology of Western Australia, 2020; Government of Western Australia Department of Mines, Industry Regulation and Safety: Perth, Western Australia, 2020. [Google Scholar]
- Government of Western Australia Department of Mines, Industry Regulation and Safety. MINEDEX Database; Government of Western Australia Department of Mines, Industry Regulation and Safety: Perth, Western Australia, 2021. [Google Scholar]
- Brunet, F. Hydrothermal Production of H2 and Magnetite From Steel Slags: A Geo-Inspired Approach Based on Olivine Serpentinization. Front. Earth Sci. 2019, 7, 17. [Google Scholar] [CrossRef] [Green Version]
- Charlou, J.L.; Donval, J.P.; Fouquet, Y.; Jean-Baptiste, P.; Holm, N. Geochemistry of high H2 and CH4 vent fluids issuing from ultramafic rocks at the Rainbow hydrothermal field (36°14′ N, MAR). Chem. Geol. 2002, 191, 345–359. [Google Scholar] [CrossRef]
- Zgonnik, V. The occurrence and geoscience of natural hydrogen: A comprehensive review. Earth-Sci. Rev. 2020, 203, 103140. [Google Scholar] [CrossRef]
- Myagkiy, A.; Moretti, I.; Brunet, F. Space and time distribution of subsurface H2 concentration in so-called “fairy circles”: Insight from a conceptual 2-D transport model. BSGF-Earth Sci. Bull. 2020, 191, 13. [Google Scholar] [CrossRef]
- Klein, F.; Tarnas, J.D.; Bach, W. Abiotic Sources of Molecular Hydrogen on Earth. Elements 2020, 16, 19–24. [Google Scholar] [CrossRef]
- Halas, P.; Dupuy, A.; Franceschi, M.; Bordmann, V.; Fleury, J.-M.; Duclerc, D. Hydrogen gas in circular depressions in South Gironde, France: Flux, stock, or artefact? Appl. Geochem. 2021, 127, 104928. [Google Scholar] [CrossRef]
- Lefeuvre, N.; Truche, L.; Donzé, F.-V.; Ducoux, M.; Barré, G.; Fakoury, R.-A.; Calassou, S.; Gaucher, E.C. Native H2 Exploration in the Western Pyrenean Foothills. Geochem. Geophys. Geosyst. 2021, 22, e2021GC009917. [Google Scholar] [CrossRef]
- Lewan, M.D. Experiments on the role of water in petroleum formation. Geochim. Cosmochim. 1997, 61, 3691–3723. [Google Scholar] [CrossRef]
- Li, X.; Krooss, B.M.; Weniger, P.; Littke, R. Molecular hydrogen (H2) and light hydrocarbon gases generation from marine and lacustrine source rocks during closed-system laboratory pyrolysis experiments. J. Anal. Appl. Pyrolysis 2017, 126, 275–287. [Google Scholar] [CrossRef]
- Lorant, F.; Behar, F. Late Generation of Methane from Mature Kerogens. Energy Fuels 2002, 16, 412–427. [Google Scholar] [CrossRef]
- Hirose, T.; Kawagucci, S.; Suzuki, K. Mechanoradical H2 generation during simulated faulting: Implications for an earthquake-driven subsurface biosphere. Geophys. Res. Lett. 2011, 38. [Google Scholar] [CrossRef] [Green Version]
- Kameda, J.; Saruwatari, K.; Tanaka, H. H2 generation in wet grinding of granite and single-crystal powders and implications for H2 concentration on active faults. Geophys. Res. Lett. 2003, 30. [Google Scholar] [CrossRef]
- Kita, I.; Matsuo, S.; Wakita, H. H2 generation by reaction between H2O and crushed rock: An experimental study on H2 degassing from the active fault zone. J. Geophys. Res. Solid Earth 1982, 87, 10789–10795. [Google Scholar] [CrossRef]
- Sato, M.; Sutton, A.J.; McGee, K.A. Anomalous hydrogen emissions from the San Andreas fault observed at the Cienega Winery, central California. Pure Appl. Geophys. 1984, 122, 376–391. [Google Scholar] [CrossRef]

| Composition of Cylinder | A | B | C |
|---|---|---|---|
| Nitrogen | 100% | 95% | 99% |
| Hydrogen | 0% | 5% | 0% |
| Methane | 0% | 0% | 1% |
| Experiment Number | Cylinder Flow Rates (mL/min) | Theoretical Manifold H2 Concentration (ppm) | Theoretical Manifold CH4 Concentration (ppm) | ||
|---|---|---|---|---|---|
| A | B | C | |||
| 1 | 1000 | 0 | 0 | 0.0 | 0.0 |
| 2 | 1000 | 0 | 50 | 0.0 | 469.8 |
| 3 | 1000 | 0 | 100 | 0.0 | 897.5 |
| 4 | 1000 | 0 | 200 | 0.0 | 1647.2 |
| 5 | 1000 | 2 | 0 | 99.8 | 0.0 |
| 6 | 1000 | 2 | 50 | 95.1 | 468.9 |
| 7 | 1000 | 2 | 100 | 90.9 | 895.9 |
| 8 | 1000 | 2 | 200 | 83.4 | 1644.4 |
| 9 | 1000 | 4 | 0 | 199.2 | 0.0 |
| 10 | 1000 | 4 | 50 | 189.9 | 468.1 |
| 11 | 1000 | 4 | 100 | 181.4 | 894.2 |
| 12 | 1000 | 4 | 200 | 166.5 | 1641.7 |
| 13 | 1000 | 8 | 0 | 396.8 | 0.0 |
| 14 | 1000 | 8 | 50 | 378.3 | 466.3 |
| 15 | 1000 | 8 | 100 | 361.5 | 891.0 |
| 16 | 1000 | 8 | 200 | 331.9 | 1636.2 |
| 17 | 0 | 0 | 1000 | 0.0 | 10,000.0 |
| 18 | 0 | 2 | 1000 | 101.2 | 9979.8 |
| 19 | 0 | 4 | 1000 | 202.0 | 9959.6 |
| 20 | 0 | 8 | 1000 | 402.4 | 9919.5 |
| Methane Concentration | Gradient | R2 |
|---|---|---|
| 0 ppm | 1.31 ± 0.27 | 0.9998 |
| 468 ± 2 ppm | 1.35 ± 0.28 | 0.9999 |
| 895 ± 3 ppm | 1.34 ± 0.27 | 0.999 |
| 1642 ± 6 ppm | 1.28 ± 0.26 | 0.9994 |
| 9965 ± 40 ppm | 1.25 ± 0.26 | 0.9986 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mainson, M.; Heath, C.; Pejcic, B.; Frery, E. Sensing Hydrogen Seeps in the Subsurface for Natural Hydrogen Exploration. Appl. Sci. 2022, 12, 6383. https://doi.org/10.3390/app12136383
Mainson M, Heath C, Pejcic B, Frery E. Sensing Hydrogen Seeps in the Subsurface for Natural Hydrogen Exploration. Applied Sciences. 2022; 12(13):6383. https://doi.org/10.3390/app12136383
Chicago/Turabian StyleMainson, Mederic, Charles Heath, Bobby Pejcic, and Emanuelle Frery. 2022. "Sensing Hydrogen Seeps in the Subsurface for Natural Hydrogen Exploration" Applied Sciences 12, no. 13: 6383. https://doi.org/10.3390/app12136383
APA StyleMainson, M., Heath, C., Pejcic, B., & Frery, E. (2022). Sensing Hydrogen Seeps in the Subsurface for Natural Hydrogen Exploration. Applied Sciences, 12(13), 6383. https://doi.org/10.3390/app12136383