Temporomandibular Disorders and Bruxism in Patients Attending a Tinnitus Clinic
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Sample
2.2. Tinnitus Severity and Side
2.3. Depression and Anxiety Severity
2.4. TMD and Bruxism Prevalence
2.5. Statistical Analysis
3. Results
3.1. Demographic Features of the Study Sample
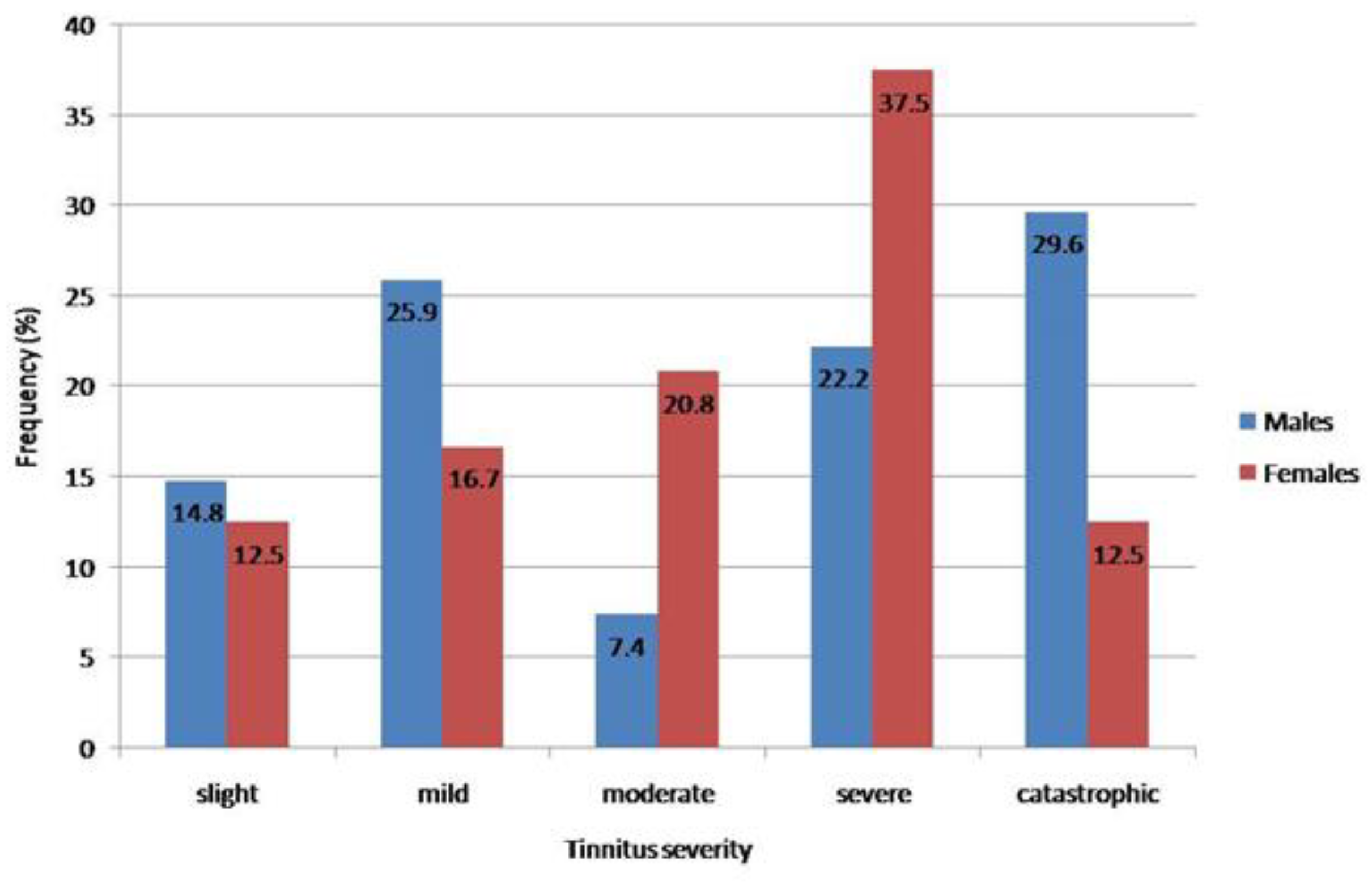
3.2. Tinnitus Severity by Gender and Age
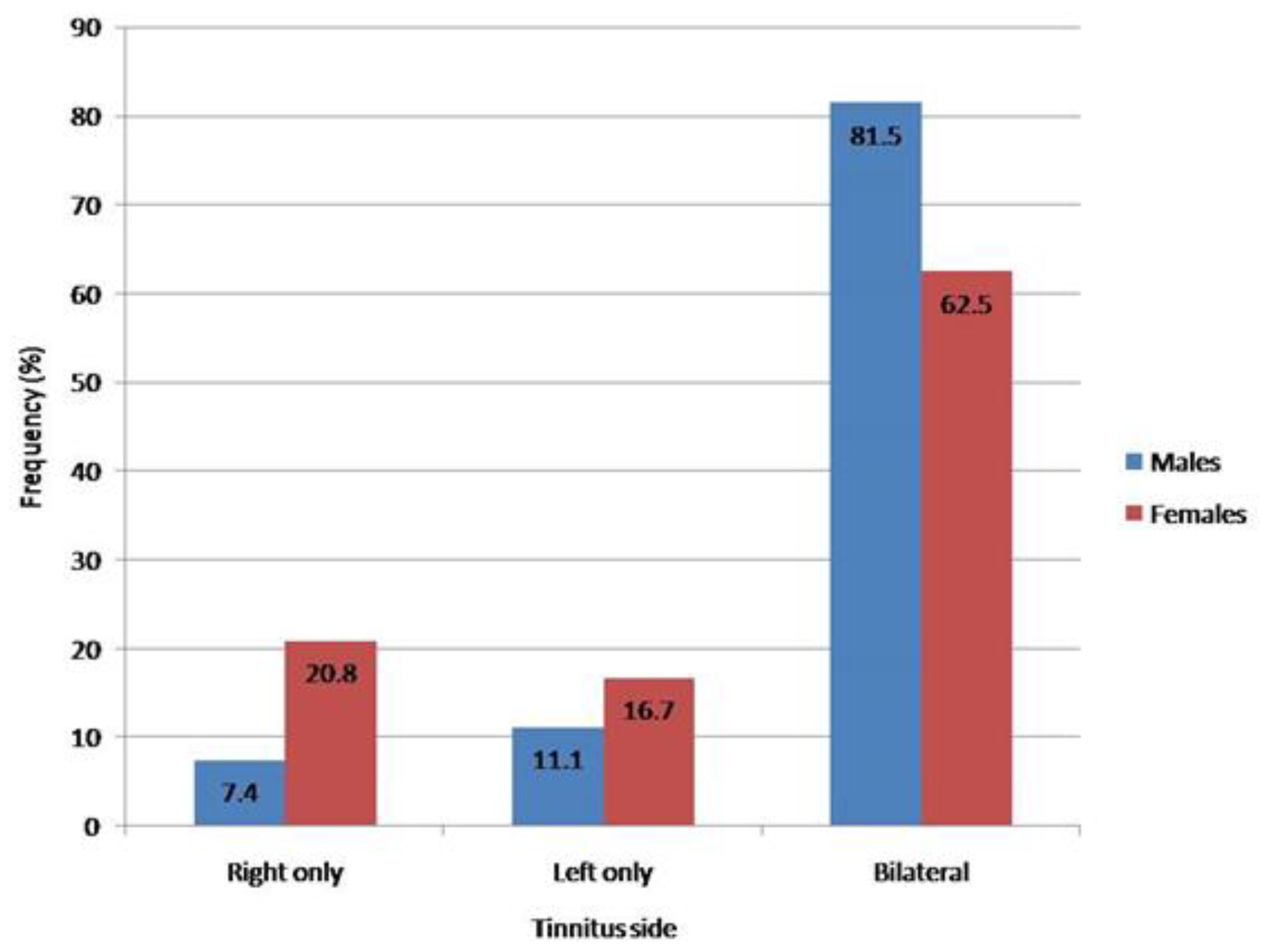
3.3. Tinnitus Side
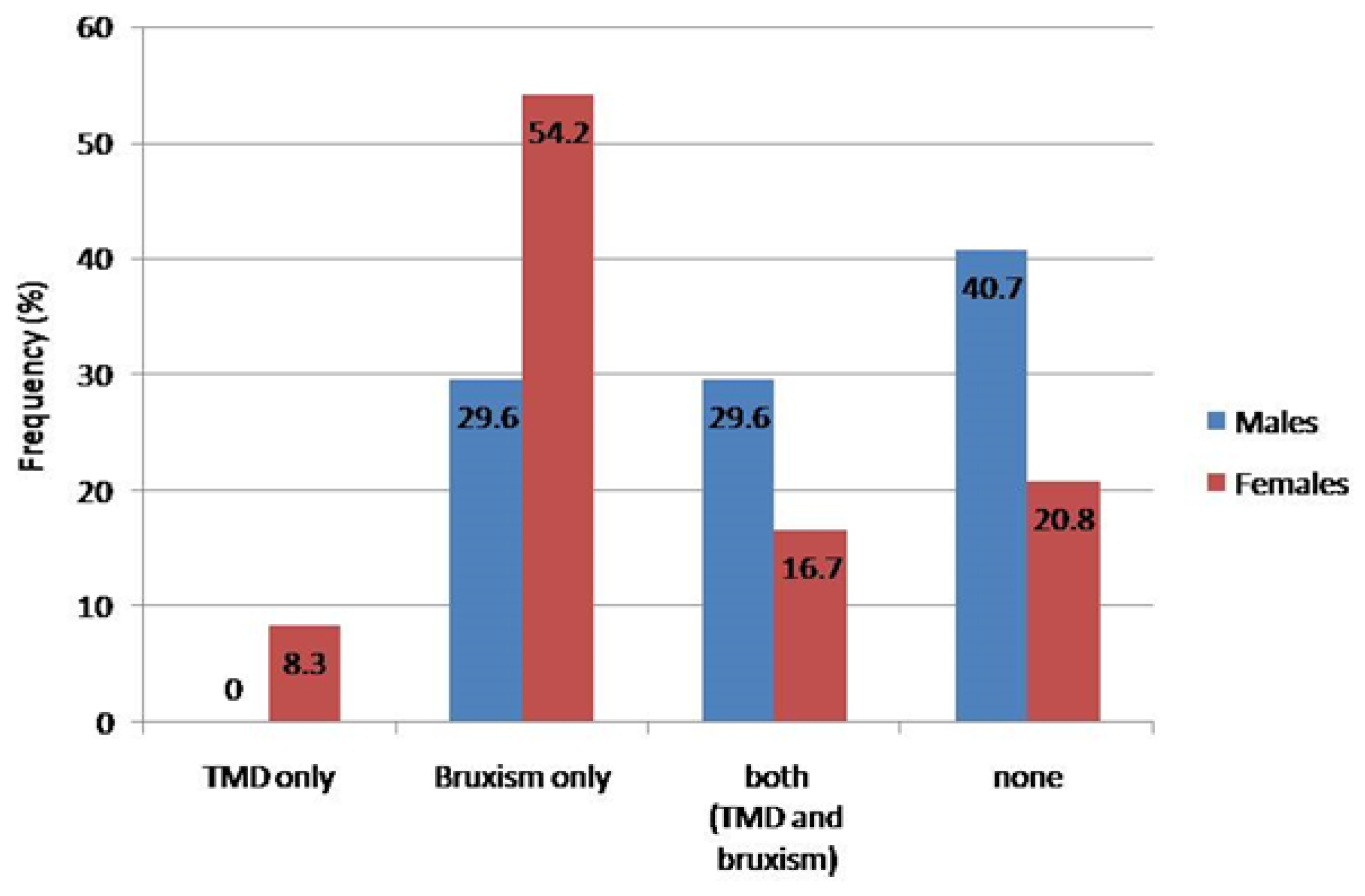
3.4. Prevalence of Bruxism and TMD
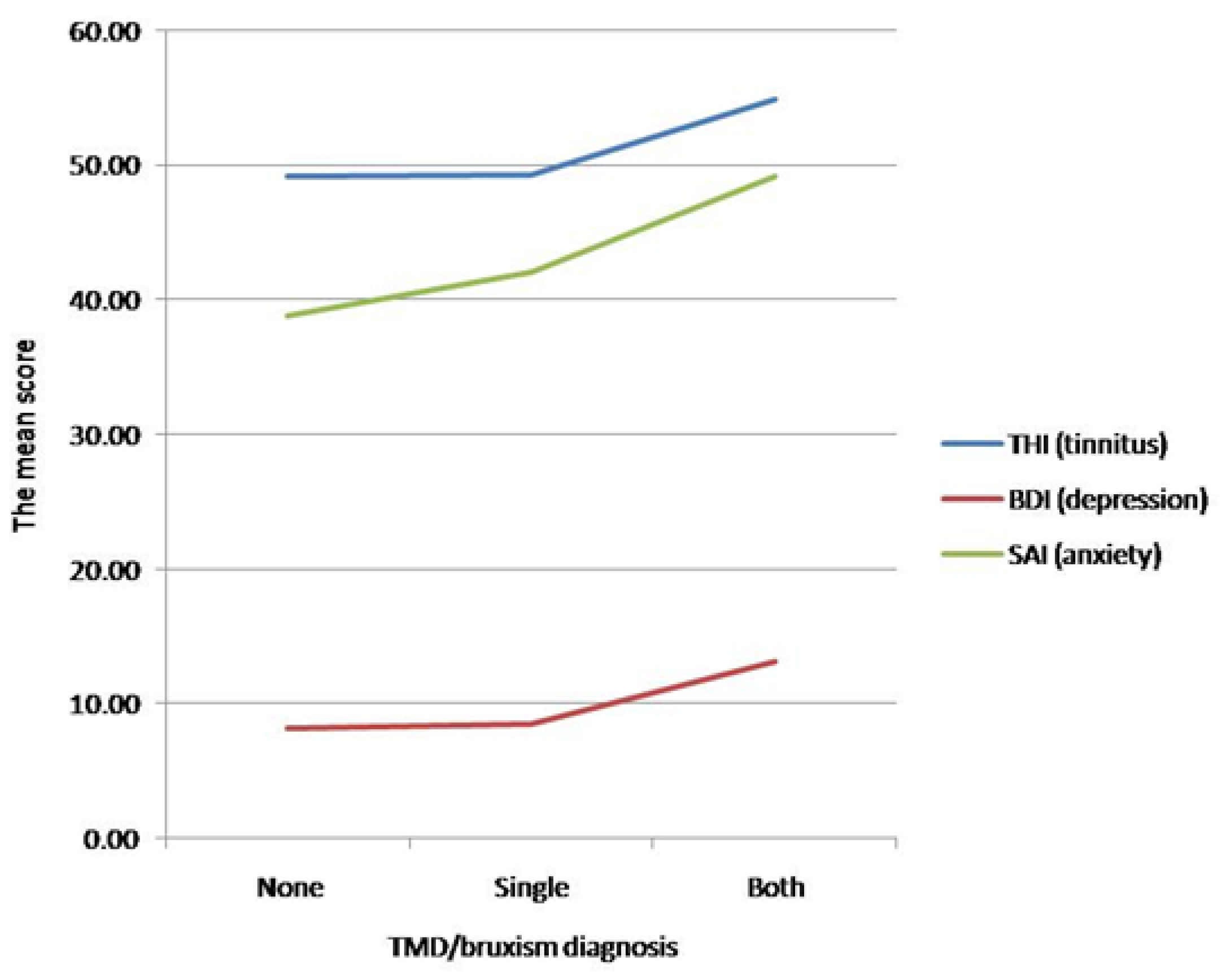
3.5. Tinnitus/Depression/Anxiety Scores and TMD/Bruxism
3.6. TMJ Clicking
3.7. Factors Associated with Tinnitus Severity
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Frantzeska, K.; Fulya, Ö. Temporomandibular Disorders: Fundamental Questions and Answers. Turk. J. Orthod. 2020, 33, 246–252. [Google Scholar]
- National Institute of Dental and Craniofacial Research: Facial Pain. Available online: https://www.nidcr.nih.gov/research/data-statistics/facial-pain (accessed on 1 May 2022).
- Mijiritsky, E.; Winocur, E.; Emodi Perelman, A.; Friedman-Rubin, P.; Dahar, E.; Reiter, S.H. Tinnitus in Temporomandibular Disorders: Axis I and II Findings According to the Diagnostic Criteria for Temporomandibular Disorders. J. Oral Facial Pain Headache 2020, 34, 265–272. [Google Scholar] [CrossRef] [PubMed]
- Špalj, S.; Šlaj, M.; Athanasiou, A.E.; Žak, I.; Šimunović, M.; Šlaj, M. Temporomandibular disorders and orthodontic treatment need in orthodontically untreated children and adolescents. Coll. Antropol. 2015, 39, 151–158. [Google Scholar] [PubMed]
- Schiffman, E.; Ohrbach, R.; Truelove, E.; Look, J.; Anderson, G.; Goulet, J.P. Diagnostic Criteria for Temporomandibular Disorders (DC/TMD) for Clinical and Research Applications: Recommendations of the International RDC/TMD Consortium Network and Orofacial Pain Special Interest Group. J. Oral Facial Pain Headache 2014, 28, 6–27. [Google Scholar] [CrossRef]
- Miettinen, O.; Lahti, S.; Sipilä, K. Psychosocial aspects of temporomandibular disorders and oral health-related quality-of-life. Acta Odontol. Scand. 2012, 70, 331–336. [Google Scholar] [CrossRef] [Green Version]
- Conti, P.C.; dos Santos, C.N.; Kogawa, E.M.; de Castro Ferreira Conti, A.C.; de Araujo Cdos, R. The treatment of painful temporomandibular joint clicking with oral splints: A randomized clinical trial. J. Am. Dent. Assoc. 2006, 137, 1108–1114. [Google Scholar] [CrossRef]
- Reissmann, D.R.; John, M.T. Is temporomandibular joint (TMJ) clicking a risk factor for pain in the affected TMJ. Schmerz 2007, 21, 131–138. [Google Scholar]
- Emodi, P.A.; Lobbezoo, F.; Zar, A.; Friedman, R.P.; Van Selms, M.; Winocur, E. Self-Reported bruxism and associated factors in Israeli adolescents. J. Oral Rehabil. 2016, 43, 443–450. [Google Scholar] [CrossRef]
- Raphael, K.G.; Santiago, V.; Lobbezoo, F. Is Bruxism a Disorder or a Behaviour? Rethinking the International Consensus on Defining and Grading of Bruxism. J. Oral Rehabil. 2016, 43, 791–798. [Google Scholar] [CrossRef] [Green Version]
- Manfredini, D.; Winocur, E.; Guarda-Nardini, L.; Paesani, D.; Lobbezoo, F. Epidemiology of bruxism in adults: A systematic review of the literature. J. Orofac. Pain 2013, 27, 99–110. [Google Scholar] [CrossRef] [Green Version]
- Lavigne, J.; Huynh, N.; Kato, T.; Okura, K.; Adachi, K.; Yao, D.; Sessle, B. Genesis of sleep bruxism: Motor and autonomic-cardiac interactions. Arch. Oral Biol. 2007, 52, 381–384. [Google Scholar] [CrossRef] [PubMed]
- Manfredini, D.; Restrepo, C.; Diaz-Serrano, K.; Winocur, E.; Lobbezoo, F. Prevalence of SB in children: A systematic review of the literature. J. Oral Rehabil. 2013, 40, 631–642. [Google Scholar] [CrossRef] [PubMed]
- De Luca Canto, G.; Singh, V.; Conti, P.; Dick, B.D.; Gozal, D.; Major, P.W.; Flores-Mir, C. Association between SB and psychosocial factors in children and adolescents: A systematic review. Clin. Pediatr. 2015, 54, 469–478. [Google Scholar] [CrossRef] [PubMed]
- Van Selms, M.K.; Visscher, C.M.; Naeije, M.; Lobbezoo, F. Bruxism and associated factors among Dutch adolescents. Community Dent. Oral Epidemiol. 2013, 41, 353–363. [Google Scholar] [CrossRef]
- Yadav, S.; Yang, Y.; Dutra, E.H.; Robinson, J.L.; Wadhwa, S. Temporomandibular Joint Disorders in Older Adults. J. Am. Geriatr. Soc. 2018, 66, 1213–1217. [Google Scholar] [CrossRef] [PubMed]
- Vavrina, J.; Vavrina, J. Bruxism: Classification, Diagnostics and Treatment. Praxis 2020, 109, 973–978. [Google Scholar] [CrossRef]
- Yap, A.U.; Chua, A.P. Sleep bruxism: Current knowledge and contemporary management. J. Conserv. Dent. 2016, 19, 383–389. [Google Scholar] [CrossRef]
- Meirelles, L.; Cunha Matheus Rodrigues Garcia, R. Influence of bruxism and splint therapy on tongue pressure against teeth. Cranio 2016, 34, 100–104. [Google Scholar] [CrossRef]
- Lobbezoo, F.; Ahlberg, J.; Raphael, K.G.; Wetselaar, P.; Glaros, A.G.; Kato, T.; Santiago, V.; Winocur, E.; De Laat, A.; De Leeuw, R.; et al. International consensus on the assessment of bruxism: Report of a work in progress. J. Oral Rehabil. 2018, 45, 837–844. [Google Scholar] [CrossRef]
- Levine, A.R.; Oron, Y. Tinnitus. In Handbook of Clinical Neurology; Elsevier: Amsterdam, The Netherlands, 2015; Volume 129, Chapter 23. [Google Scholar]
- Esmaili, A.; Renton, J. A Review of Tinnitus. Aust. J. Gen. Pract. 2018, 47, 205–208. [Google Scholar] [CrossRef] [Green Version]
- Fernandes, G.; de Siqueira, J.T.T.; Gonçalves, D.G.; Camparis, C.M. Association between painful temporomandibular disorders, sleep bruxism and tinnitus. Braz. Oral Res. 2014, 28, 1–7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Levine, R.A.; Nam, E.C.; Oron, Y.; Melcher, J.R. Evidence for a tinnitus subgroup responsive to somatosensory based treatment modalities. Prog. Brain Res. 2007, 166, 195–207. [Google Scholar] [PubMed]
- Levine, R.A. Somatic (craniocervical) tinnitus and the dorsal cochlear nucleus hypothesis. Am. J. Otolaryngol. 1999, 20, 351–362. [Google Scholar] [CrossRef]
- Mottaghi, A.; Menéndez-Díaz, I.; Cobo, J.L.; González-Serrano, J.; Cobo, T. Is There a Higher Prevalence of Tinnitus in Patients with Temporomandibular Disorders? Syst. Rev. Meta-Anal. J. Oral Rehabil. 2019, 46, 76–86. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Camparis, C.M.; Formigoni, G.; Teixeira, M.J.; De Siqueira, J.T.T. Clinical evaluation of tinnitus in patients with sleep bruxism: Prevalence and characteristics. J. Oral Rehabil. 2005, 32, 808–814. [Google Scholar] [CrossRef] [PubMed]
- Oron, Y.; Shushan, S.; Kreitler, S.; Roth, Y. A Hebrew adaptation of the tinnitus handicap inventory. Int. J. Audiol. 2011, 50, 426–430. [Google Scholar] [CrossRef] [PubMed]
- Gil, S.; Gilbar, O. Hopelessness among cancer patients. J. Psychosoc. Oncol. 2001, 19, 21–33. [Google Scholar] [CrossRef]
- Teichman, Y.; Melnick, H. The Hebrew Manual for the State-Trait Anxiety Inventory; Ramot: Tel-Aviv, Israel, 1980. [Google Scholar]
- McCormack, A.; Edmondson-Jones, M.; Somerset, S.; Hall, D. A systematic review of the reporting of tinnitus prevalence and severity. Hear. Res. 2016, 337, 70–79. [Google Scholar] [CrossRef]
- Andersson, G.; Strömgren, T.; Ström, L.; Lyttkens, L. Randomized Controlled Trial of Internet-Based Cognitive Behavior Therapy for Distress Associated with Tinnitus. Psychosom. Med. 2002, 64, 810–816. [Google Scholar]
- Beukes, E.W.; Baguley, D.M.; Jacquemin, L.; Lourenco, M.P.C.G.; Allen, P.M.; Onozuka, J.; Stockdale, D.; Kaldo, V.; Andersson, G.; Manchaiah, V. Changes in Tinnitus Experiences during the COVID-19 Pandemic. Front. Public Health 2020, 8, 592878. [Google Scholar] [CrossRef]
- Choi, E.P.H.; Hui, B.P.H.; Wan, E.Y.F. Depression and Anxiety in Hong Kong during COVID-19. Int. J. Environ Res. Public Health 2020, 17, 3740. [Google Scholar] [CrossRef] [PubMed]
- Rauso, R.; Chirico, F.; Federico, F.; Nicoletti, G.F.; Colella, G.; Fragola, R.; Pafundi, P.C.; Tartaro, G. Maxillo-facial reconstruction following cancer ablation during COVID-19 pandemic in southern Italy. Oral Oncol. 2021, 115, 105114. [Google Scholar] [CrossRef] [PubMed]
- Allevi, A.; Dionisio, U.; Baciliero, U.; Balercia, P.; Beltramini, G.A.; Bertossi, D.; Bozzetti, A.; Califano, L.; Cascone, P.; Colombo, L.; et al. Impact of COVID-19 epidemic on maxillofacial surgery in Italy. Br. J. Oral Maxillofac. Surg. 2020, 58, 692–697. [Google Scholar] [CrossRef] [PubMed]
- Di Bartolomeo, M.; Pellacani, A.; Negrello, S.; Chiarini, L.; Anesi, A. Emerging challenges and possible strategies in maxillo-facial and oral surgery during the COVID-19 pandemic. J. Oral Sci. 2020, 62, 452–454. [Google Scholar] [CrossRef] [PubMed]
| Diagnosis | Gender | n | Prevalence (%) | p-Value |
|---|---|---|---|---|
| Bruxism | Male | 16 | 59.3 | 0.234 |
| Female | 18 | 75.0 | ||
| Total | 34 | 66.7 | ||
| TMD | Male | 8 | 29.6 | 0.712 |
| Female | 6 | 25.0 | ||
| Total | 14 | 27.5 |
| Severity Scores | TMD/Bruxism Diagnosis * | n | Mean | ±SD | p-Value # |
|---|---|---|---|---|---|
| THI (tinnitus) | Single | 23 | 49.22 | 25.504 | 0.829 |
| Both | 12 | 54.83 | 30.361 | ||
| None | 16 | 49.13 | 29.285 | ||
| BDI (depression) | Single | 23 | 8.48 | 6.251 | 0.260 |
| Both | 12 | 13.17 | 13.790 | ||
| None | 16 | 8.13 | 7.210 | ||
| SAI (anxiety) | Single | 23 | 42.00 | 10.353 | 0.045 |
| Both | 12 | 49.17 | 11.731 | ||
| None | 16 | 38.75 | 10.459 |
| Diagnosis | Gender | n | % |
|---|---|---|---|
| Clicks | Male | 22 | 81.48 |
| Female | 14 | 58.33 | |
| Total | 36 | 70.59 | |
| No clicks | Male | 5 | 18.52 |
| Female | 10 | 41.67 | |
| Total | 15 | 29.41 |
| Variable | Standardized Coefficients | p-Value * |
|---|---|---|
| Gender | 0.045 | 0.695 |
| Age | −0.196 | 0.089 |
| Depression score | 0.524 | 0.001 |
| Anxiety score | 0.171 | 0.234 |
| Dental wear | 0.119 | 0.371 |
| Tongue indentations | 0.323 | 0.025 |
| TMD/Bruxism | 0.291 | 0.027 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Peleg, O.; Haddad, L.; Kleinman, S.; Sella Tunis, T.; Wasserman, G.; Mijiritsky, E.; Oron, Y. Temporomandibular Disorders and Bruxism in Patients Attending a Tinnitus Clinic. Appl. Sci. 2022, 12, 4970. https://doi.org/10.3390/app12104970
Peleg O, Haddad L, Kleinman S, Sella Tunis T, Wasserman G, Mijiritsky E, Oron Y. Temporomandibular Disorders and Bruxism in Patients Attending a Tinnitus Clinic. Applied Sciences. 2022; 12(10):4970. https://doi.org/10.3390/app12104970
Chicago/Turabian StylePeleg, Oren, Lama Haddad, Shlomi Kleinman, Tatiana Sella Tunis, Gilad Wasserman, Eitan Mijiritsky, and Yahav Oron. 2022. "Temporomandibular Disorders and Bruxism in Patients Attending a Tinnitus Clinic" Applied Sciences 12, no. 10: 4970. https://doi.org/10.3390/app12104970
APA StylePeleg, O., Haddad, L., Kleinman, S., Sella Tunis, T., Wasserman, G., Mijiritsky, E., & Oron, Y. (2022). Temporomandibular Disorders and Bruxism in Patients Attending a Tinnitus Clinic. Applied Sciences, 12(10), 4970. https://doi.org/10.3390/app12104970







