Functional Food Product Based on Nanoselenium-Enriched Lactobacillus casei against Cadmium Kidney Toxicity
Abstract
1. Introduction
2. Materials and Methods
2.1. Eco-Friendly Biosynthesis and Characterization of SeNPs
2.1.1. Production of Purified NanoSelenium Particles (SeNPs)
2.1.2. Production of Lacto-NanoSelenium Particles (LSeNPs)
2.2. Animal and Experimental Design
- Control group—received orally by gavage during the experiment only water;
- Cadmium group (Cd)—received orally 5 mg/kg CdCl2;
- 0.1 SeNPs + Cd—received orally by gavage 0.1 mg/kg of SeNPs together with 5 mg/kg of CdCl2
- 0.2 SeNPs + Cd—received orally by gavage 0.2 mg/kg of SeNPs together with 5 mg/kg of CdCl2
- 0.4 SeNPs + Cd—received 0.4 mg/kg of SeNPs together with 5 mg/kg of CdCl2
- 0.1 LSeNPs + Cd—received orally by gavage 0.1 mg/kg of LSeNPs together with 5 mg/kg of CdCl2
- 0.2 LSeNPs + Cd—received orally by gavage 0.2 mg/kg of LSeNPs together with 5 mg/kg of CdCl2
- 0.4 LSeNPs + Cd—received 0.4 mg/kg of LSeNPs together with 5 mg/kg of CdCl2
2.3. Blood Creatinine Level
2.4. Antioxidant Capacity of Mice Plasma—TEAC Assay
2.5. Histopathology Analysis
2.6. Immunohistochemical Analysis
2.7. RT-PCR Analysis
2.8. Statistical Analysis
3. Results and Discussion
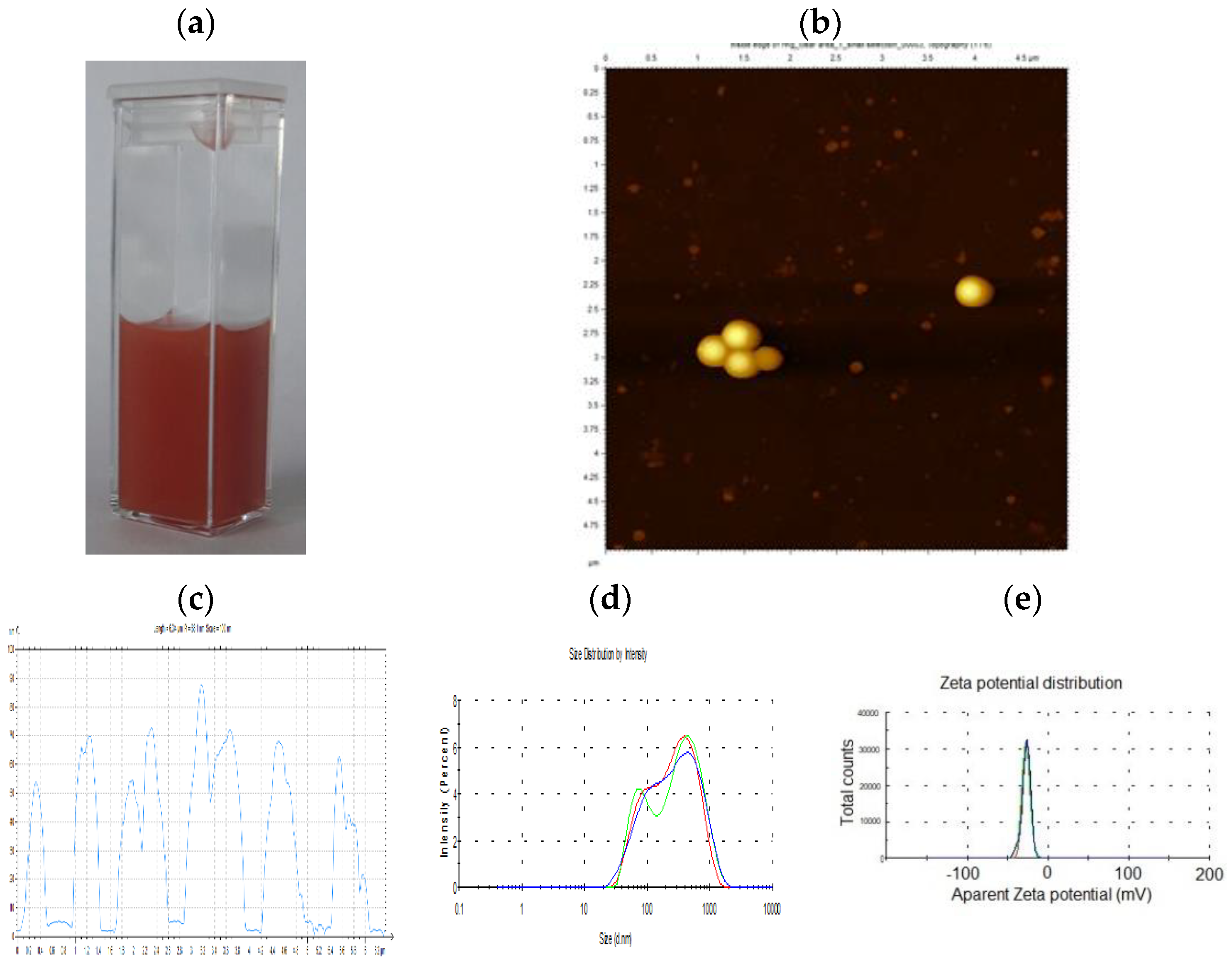
3.1. Physico-Chemical Characterization of SeNPs
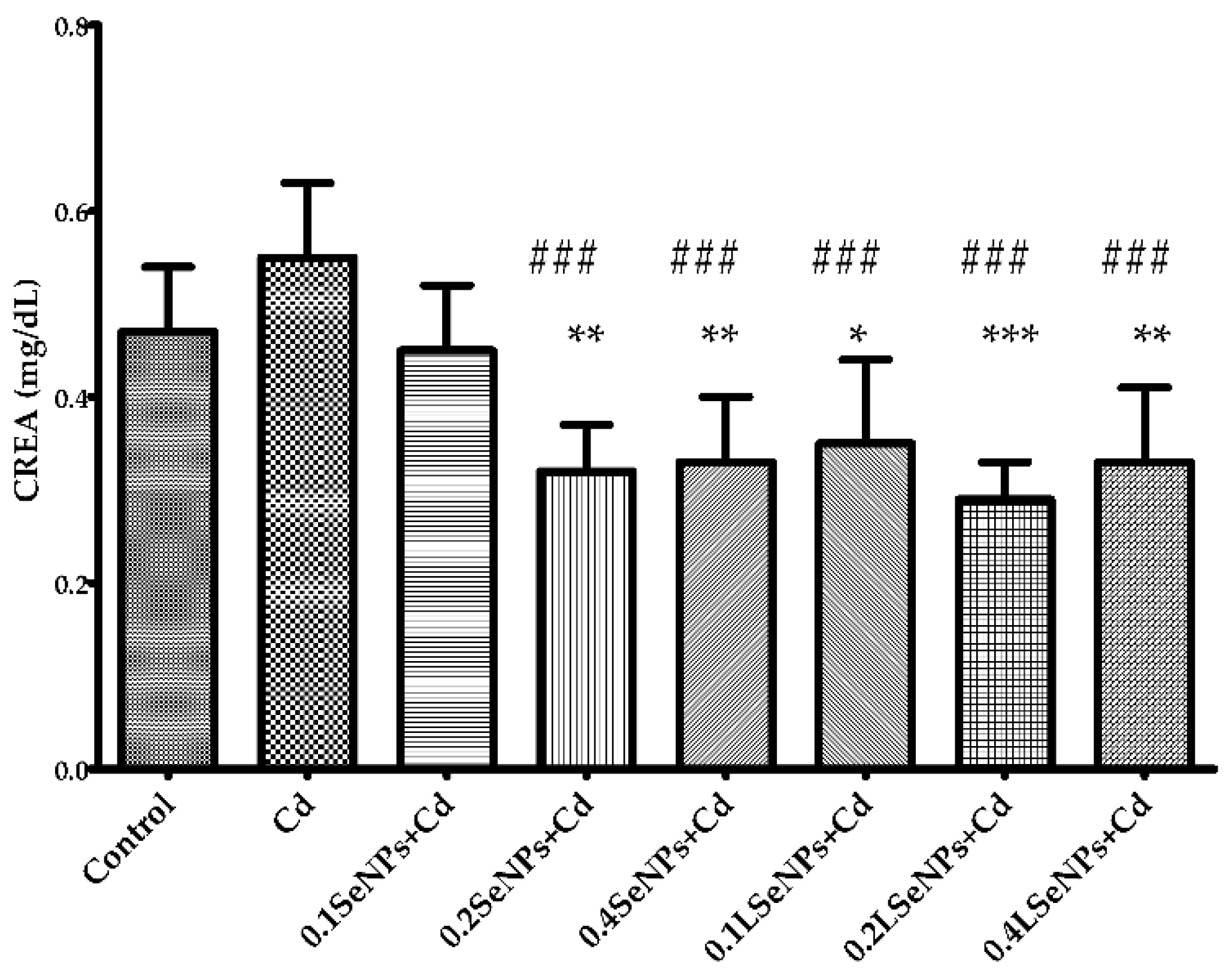
3.2. Effect of SeNPs on Blood Creatinine Level
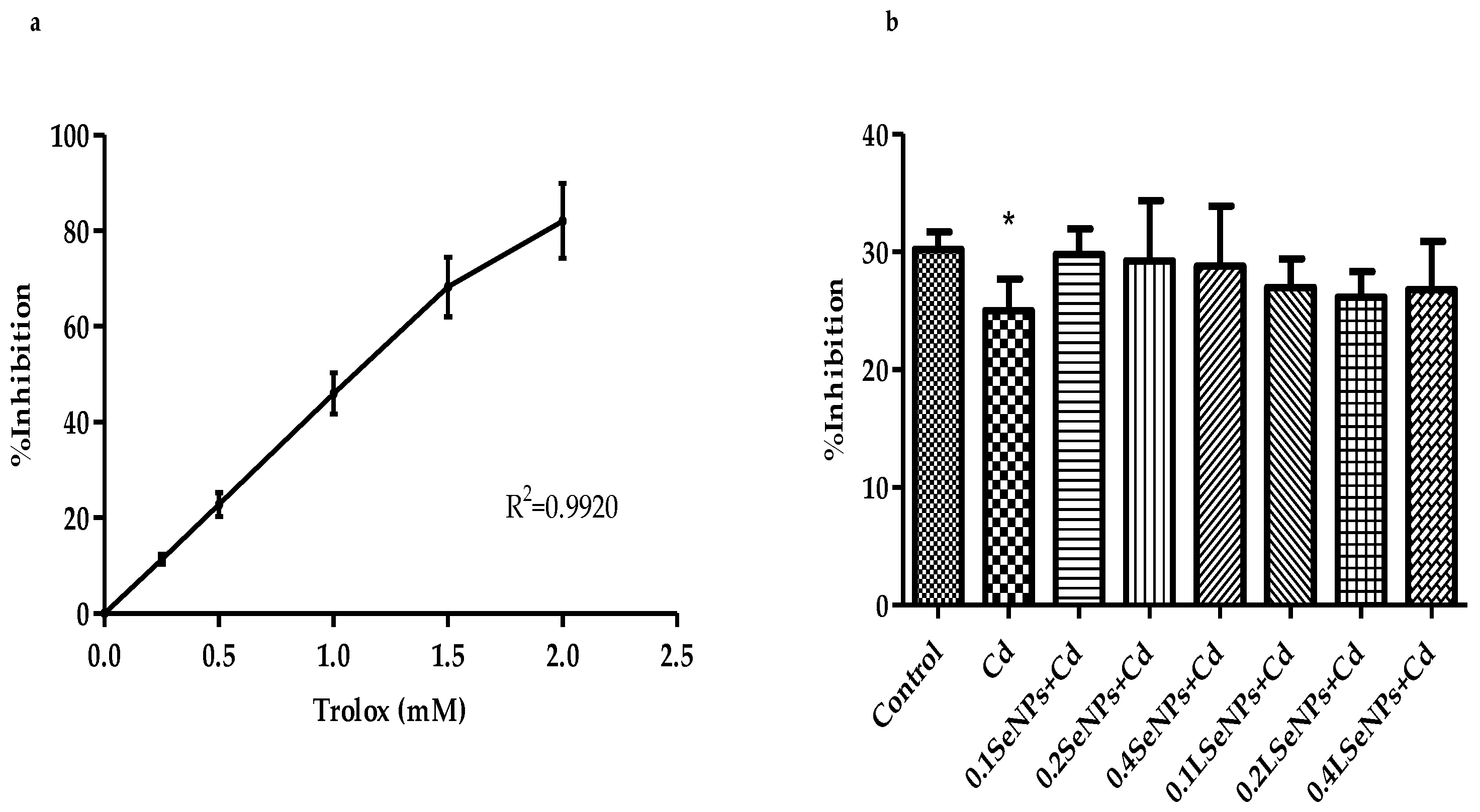
3.3. Antioxidant Capacity of Mice Plasma—TEAC Assay
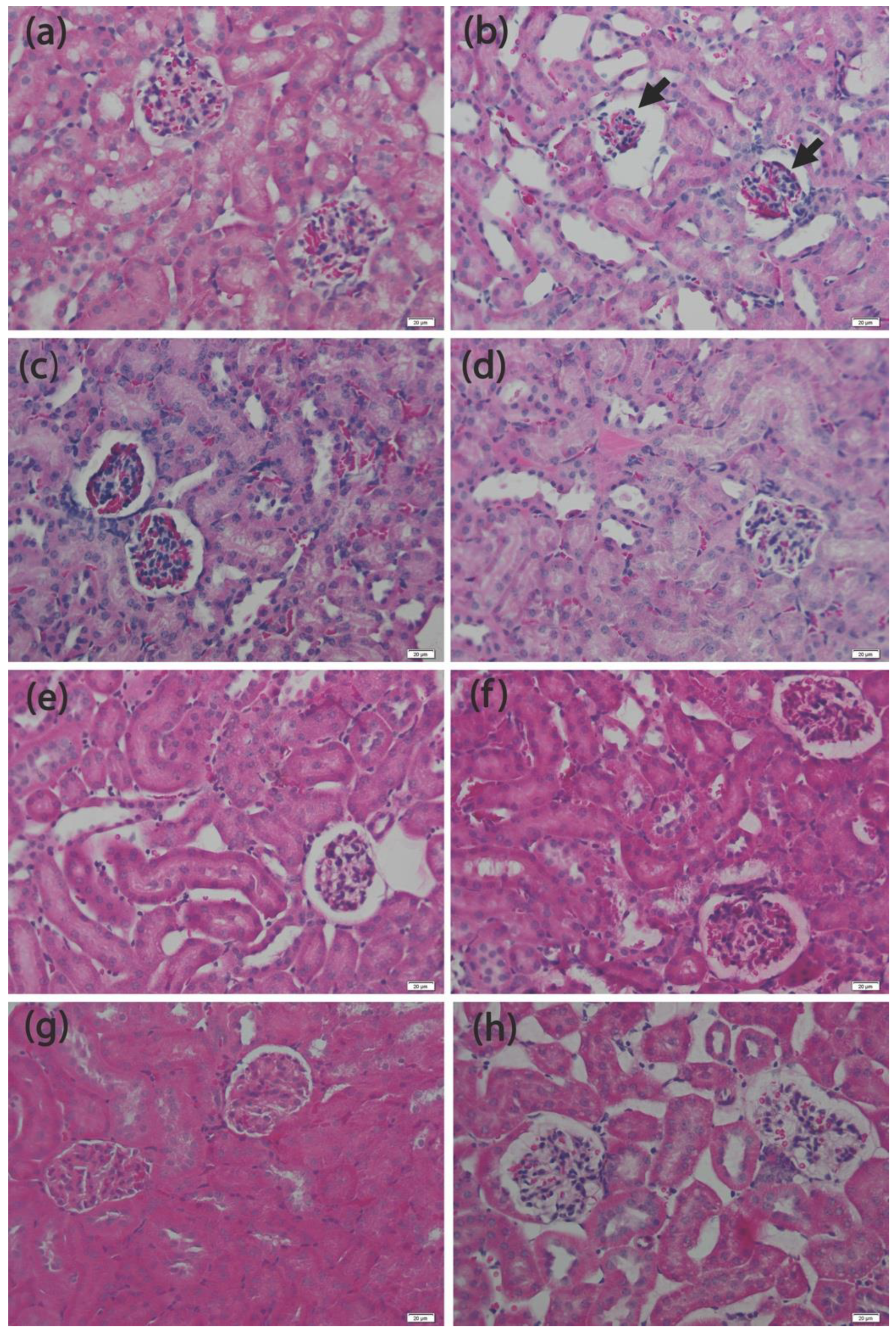
3.4. Histopathology Analysis
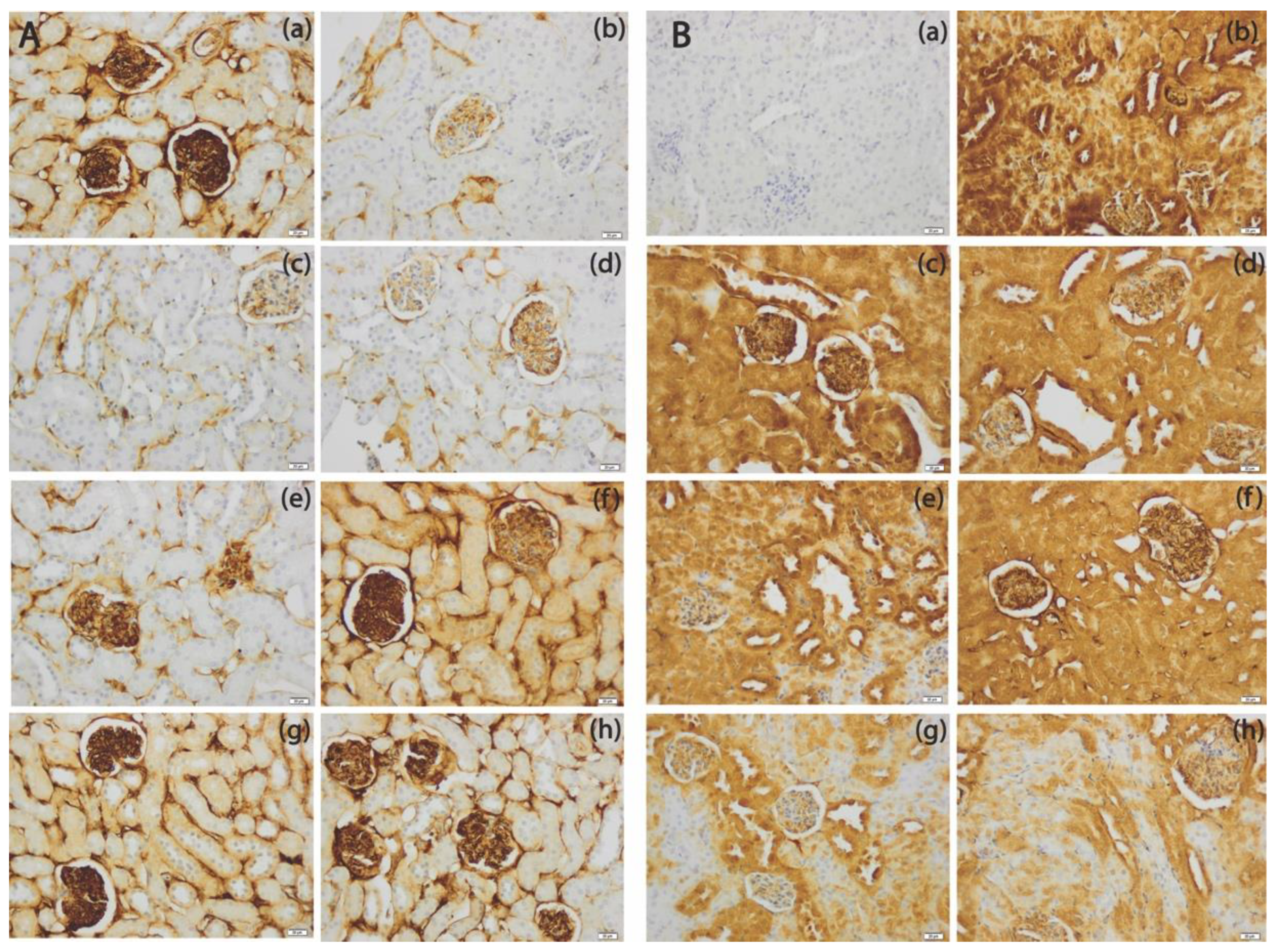
3.5. The SeNPs Prevent Apoptosis in Renal Parenchyma Induced by Cadmium
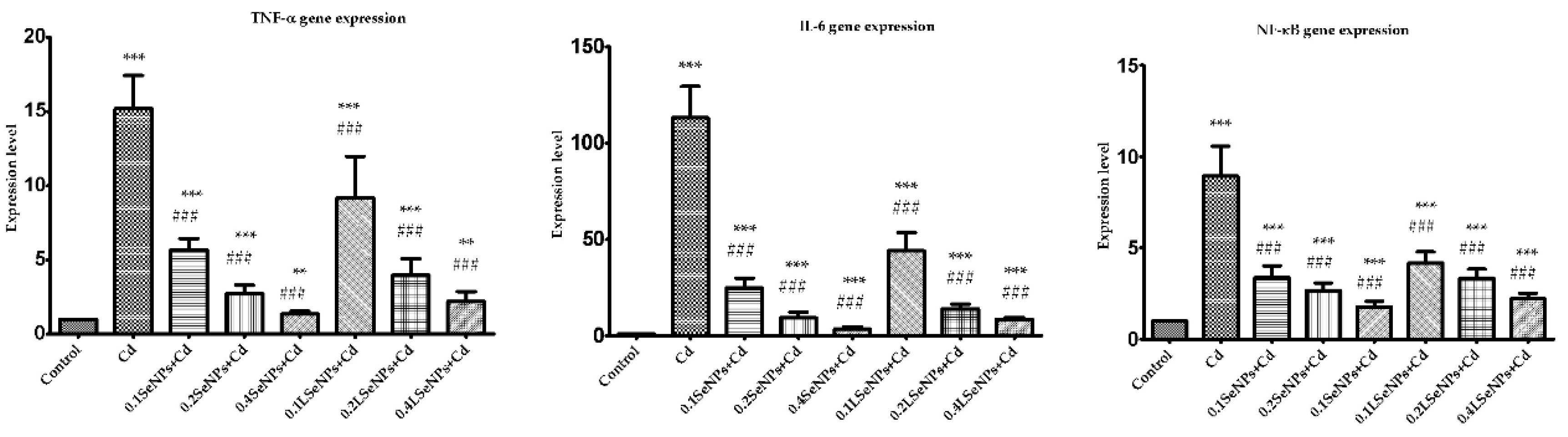
3.6. The SeNPs Prevent Inflammation in Kidney Parenchyma Induced by Cadmium
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Genchi, G.; Sinicropi, M.S.; Lauria, G.; Carocci, A.; Catalano, A. Review The Effects of Cadmium Toxicity. Int. J. Environ. Res. Public Health 2020, 17, 3782. [Google Scholar] [CrossRef]
- Cai, K.; Yu, Y.; Zhang, M.; Kim, K. Concentration, Source, and Total Health Risks of Cadmium in Multiple Media in Densely Populated Areas, China. Int. J. Environ. Res. Public Health 2019, 16, 2269. [Google Scholar] [CrossRef]
- Xiao, R.; Guo, D.; Ali, A.; Mi, S.; Liu, T.; Ren, C.; Li, R.; Zhang, Z. Accumulation, Ecological-Health Risks Assessment, and Source Apportionment of Heavy Metals in Paddy Soils: A Case Study in Hanzhong, Shaanxi, China. Environ. Pollut. 2019, 248, 349–357. [Google Scholar] [CrossRef] [PubMed]
- Rao, Z.X.; Huang, D.Y.; Wu, J.S.; Zhu, Q.H.; Zhu, H.-H.; Xu, C.; Xiong, J.; Wang, H.; Duan, M.M. Distribution and Availability of Cadmium in Profile and Aggregatesof a Paddy Soil with 30-Year Fertilization and Its Impact on Cd Accumulation in Rice Plant. Environ. Pollut. 2018, 239, 198–204. [Google Scholar] [CrossRef] [PubMed]
- Daisley, B.A.; Monachese, M.; Trinder, M.; Bisanz, J.E.; Chmiel, J.A.; Burton, J.P.; Reid, G. Immobilization of Cadmium and Lead by Lactobacillus Rhamnosus GR-1 Mitigates Apical-Tobasolateral Heavy Metal Translocation in a Caco-2 Model of the Intestinal Epithelium. Gut. Microbes. 2019, 10, 321–333. [Google Scholar] [CrossRef] [PubMed]
- Satarug, S. Dietary Cadmium Intake and Its Effects on Kidneys. Toxics 2018, 6, 15. [Google Scholar] [CrossRef]
- Tinkov, A.A.; Gritsenko, V.A.; Skalnaya, M.G.; Cherkasov, S.V.; Aaseth, J.; Skalny, A.V. Gut as a Target for Cadmium Toxicity. Environ. Pollut. 2018, 235, 429–434. [Google Scholar] [CrossRef]
- Arroyo, V.S.; Flores, K.M.; Ortiz, L.B.; Gómez-Quiroz, L.E.; Gutiérrez-Ruiz, M.C. Liver and Cadmium Toxicity. J. Drug. Metab. Toxicol. 2012, S5:001, 1–7. [Google Scholar] [CrossRef]
- Flora, S.J.S.; Pachauri, V. Chelation in Metal Intoxication. Int. J. Environ. Res. Public Health 2010, 7, 2745–2788. [Google Scholar] [CrossRef] [PubMed]
- Mandel, J.S.; McLaughlin, J.K.; Schlehofer, B.; Mellemgaard, A.; Helmuert, U.; Linbald, R.; McCredie, M.; Adami, U. International Renal-Cell Cancer Study. IV. Occupation. Int. J. Cancer 1995, 61, 601–605. [Google Scholar] [CrossRef]
- Zalups, R.K.; Ahmad, S. Molecular Handling of Cadmium in Transporting Epithelia. Toxicol. Appl. Pharmacol. 2003, 186, 163–188. [Google Scholar] [CrossRef]
- Klaassen, C.D.; Liu, J. Role of Metallothionein in Cadmium-Induced Hepatotoxicity and Nephrotoxicity. Drug. Metab. Rev. 1997, 29, 79–102. [Google Scholar] [CrossRef]
- Cavalu, S.; Simon, V. Proteins Adsorption to Orthopedic Biomaterials-Vibrational Spectroscopic Evidence. J. Optoelectron. Adv. Mater. 2007, 9, 3297–3302. [Google Scholar]
- Duan, H.; Yu, L.; Tian, F.; Zhai, Q.; Fan, L.; Chen, W. Gut Microbiota: A Target for Heavy Metal Toxicity and a Probiotic Protective Strategy Review. Sci. Total. Environ. 2020, 742, 140429. [Google Scholar] [CrossRef] [PubMed]
- Inbaraj, B.S.; Chen, B.-H. In Vitro Removal of Toxic Heavy Metals by Poly(Υ-Glutamic Acid)-Coated Superparamagnetic Nanoparticles. Int. J. Nanomed. 2012, 7, 4419–4432. [Google Scholar]
- Xia, S.K.; Chen, L.; Liang, J.Q. Enriched Selenium and Its Effects on Growth and Biochemical Composition in Lactobacillus Bulgaricus. J. Agric. Food Chem. 2007, 55, 2413–2417. [Google Scholar] [CrossRef]
- Elsanhoty, R.M.; Al-Turki, I.A.; Ramadan, M.F. Application of Lactic Acid Bacteria in Removing Heavy Metals and Aflatoxin B1 from Contaminated Water. Water Sci. Technol. 2016, 74, 625–638. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, F.; Halttunen, T.; Tahvonen, R.; Salminen, S. Probiotic Bacteria as Potential Detoxification Tools: Assessing Their Heavy Metal Binding Isotherms. Can. J. Microbiol. 2016, 52, 877–885. [Google Scholar] [CrossRef] [PubMed]
- Calomme, M.; Hu, J.; Van den Branden, K.; Vanden Berghe, D.A. Seleno-Lactobacillus. An Organic Selenium Source. Biol. Trace Elem. Res. 1995, 47, 379–383. [Google Scholar] [CrossRef] [PubMed]
- Eszenyi, P.; Sztrik, A.; Babka, B.; Prokisch, J. Elemental, Nano-Sized (100–500 Nm) Selenium Production by Probiotic Lactic Acid Bacteria. Int. J. Biosci. Biochem. Bioinform. 2011, 1, 148–152. [Google Scholar] [CrossRef]
- Eswayah, A.S.; Smith, T.J.; Gardiner, P.H.E. Microbial Transformations of Selenium Species of Relevance to Bioremediation. Appl. Environ. Microbiol. 2016, 82, 4848–4859. [Google Scholar] [CrossRef] [PubMed]
- Prokisch, J.; Zommara, M. Process for Producing Elemental Selenium Nanospheres. U.S. Patent 20100189634, 23 August 2011. [Google Scholar]
- El-Boshy, M.E.; Risha, E.F.; Abdelhamid, F.M.; Mubarak, M.S.; Hadda, T.B. Protective Effects of Selenium against Cadmium Inducedhematological Disturbances, Immunosuppressive, Oxidative Stress Andhepatorenal Damage in Rats. JTEMB 2015, 29, 104–110. [Google Scholar] [CrossRef]
- El-Ramady, H.R.; Domokos-Szabolcsy, É.; Abdalla, N.A.; Alshaal, T.A.; Shalaby, T.A.; Sztrik, A.; Prokisch, J.; Fári, M. Selenium and Nano-Selenium in Agroecosystems. Environ. Chem. Lett. 2014, 12, 495–510. [Google Scholar] [CrossRef]
- Kieliszek, M.; Błażejak, S. Current Knowledge on the Importance of Selenium in Food for Living Organisms: A Review. Molecules 2016, 21, 609. [Google Scholar] [CrossRef]
- Zwolak, I. The Role of Selenium in Arsenic and Cadmium Toxicity: An Updated Review of Scientific Literature. Biol. Trace Elem. Res. 2020, 193, 44–63. [Google Scholar] [CrossRef]
- Rayman, M.P. The Use of High-Selenium Yeast to Raise Selenium Status: How Does It Measure Up? Br. J. Nutr. 2014, 92, 557–573. [Google Scholar] [CrossRef]
- Ingale, A.G.; Chaudhari, A.N. Biogenic Synthesis of Nanoparticles and Potential Applications: An Eco-Friendly Approach. J. Nanomed. Nanotechnol. 2013, 4, 1–7. [Google Scholar] [CrossRef]
- Chen, J.; He, W.; Zhu, X.; Yang, S.; Yu, T.; Ma, W. Epidemiological Study of Kidney Health in an Area with High Levels of Soil Cadmium and Selenium: Does Selenium Protect against Cadmium-Induced Kidney Injury? Sci. Total. Environ. 2020, 698, 134106. [Google Scholar] [CrossRef] [PubMed]
- Skröder, H.; Hawkesworth, S.; Kippler, M.; El Arifeen, S.; Wagatsuma, Y.; Moore, S.E.; Vahter, M. Kidney Function and Blood Pressure in Preschool-Aged Children Exposed to Cadmium and Arsenic—Potential Alleviation by Selenium. Environ. Res. 2015, 140, 205–213. [Google Scholar] [CrossRef] [PubMed]
- Wei, X.L.; He, J.R.; Cen, Y.L.; Su, Y.; Chen, L.J.; Lin, Y.; Wu, B.H.; Su, F.X.; Tang, L.Y.; Ren, Z.F. Modified Effect of Urinary Cadmium on Breast Cancer Risk by Selenium. Clin. Chim. Acta 2015, 438, 80–85. [Google Scholar] [CrossRef] [PubMed]
- Zhai, Q.; Tian, F.; Zhao, J.; Zhang, H.; Narbad, A.; Chen, W. Oral Administration of Probiotics Inhibits Absorption of the Heavy Metal Cadmium by Protecting the Intestinal Barrier. Appl. Environ. Microbiol. 2016, 82. [Google Scholar] [CrossRef]
- Yang, J.; Hou, B.; Wang, J.; Tian, B.; Bi, J.; Wang, N.; Li, X.; Huang, X. Nanomaterials for the Removal of Heavy Metals from Wastewater. Nanomaterials 2019, 9, 424. [Google Scholar] [CrossRef] [PubMed]
- Benko, I.; Nagy, G.; Tanczos, B.; Ungvari, E.; Sztrik, A.; Eszenyi, P.; Prokisch, J.; Banfalvi, G. Subacute Toxicity of Nano-Selenium Compared to Other Selenium Species in Mice. Environ. Toxicol. Chem. 2012, 31, 2812–2820. [Google Scholar] [CrossRef] [PubMed]
- Ren, X.M.; Wang, G.G.; Xu, D.Q.; Luo, K.; Liu, Y.X.; Zhong, Y.H.; Cai, Y.Q. The Protection of Selenium on Cadmium-Induced Inhibition of Spermatogenesis via Activating Testosterone Synthesis in Mice. Food Chem. Toxicol. 2012, 50, 3521–3529. [Google Scholar] [CrossRef]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant Activity Applying an Improved ABTS Radical Cation Decolorization Assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of Relative Gene Expression Data Using Real-Time Quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Cavalu, S.; Kamel, E.; Laslo, V.; Fritea, L.; Costea, T.; Antoniac, I.V.; Vasile, E.; Antoniac, A.; Semenescu, A.; Mohan, A.; et al. Eco-Friendly, Facile and Rapid Way for Synthesis of Selenium Nanoparticles. Production, Structural and Morphological Characterisation. Rev. Chim. 2018, 68, 2963–2966. [Google Scholar] [CrossRef]
- Cavalu, S.; Antoniac, I.V.; Fritea, L.; Mates, I.M.; Milea, C.; Laslo, V.; Vicas, S.; Mohan, A. Surface Modifications of the Titanium Mesh for Cranioplasty Using Selenium Nanoparticles Coating. J. Adhes. Sci. Tech. 2018, 32, 2509–2522. [Google Scholar] [CrossRef]
- Xu, C.; Guo, Y.; Qiao, L.; Ma, L.; Cheng, Y.; Roman, A. Biogenic Synthesis of Novel Functionalized Selenium Nanoparticles by Lactobacillus Casei ATCC 393 and Its Protective Effects on Intestinal Barrier Dysfunction Caused by Enterotoxigenic Escherichia Coli K88. Front. Microbiol. 2018, 9, 1129. [Google Scholar] [CrossRef]
- Wallin, M.; Sallsten, G.; Lundh, T.; Barregard, L. Low-Level Cadmium Exposure and Effects on Kidney Function. Occup. Environ. Med. 2014, 71, 848–854. [Google Scholar] [CrossRef]
- He, Y.; Chen, S.; Liu, Z.; Cheng, C.; Li, H.; Wang, M. Toxicity of Selenium Nanoparticles in Male Sprague–Dawley Rats at Supranutritional and Nonlethal Levels. Life Sci. 2014, 115, 44–51. [Google Scholar] [CrossRef] [PubMed]
- Rubio, C.P.; Hernández-Ruiz, J.; Martinez-Subiela, S.; Tvarijonaviciute, A.; Ceron, J.J. Spectrophotometric Assays for Total Antioxidant Capacity (TAC) in Dog Serum: An Update. BMC Vet. Res. 2016, 12, 1–7. [Google Scholar] [CrossRef]
- Katalinica, V.; Modun, D.; Music, I.; Boban, M. Gender Differences in Antioxidant Capacity of Rat Tissues Determined by 2,2V-Azinobis (3-Ethylbenzothiazoline 6-Sulfonate; ABTS) and Ferric Reducing Antioxidant Power (FRAP) Assays. Comp. Biochem. Physiol. Part C 2005, 140, 47–52. [Google Scholar] [CrossRef]
- Ognjanovic, B.I.; Pavlovic, S.Z.; Maletic, S.D.; Zikic, R.V.; Stajn, A.S.; Radojicic, R.M.; Saicic, Z.S.; Petrovic, V.M. Protective Influence of Vitamin E on Antioxidant Defense System in the Blood of Rats Treated with Cadmium. Physiol. Res. 2003, 52, 563–570. [Google Scholar] [PubMed]
- El-Sharaky, A.S.; Newairy, A.A.; Badreldeen, M.M.; Eweda, S.M.; Sheweita, S.A. Protective Role of Selenium against Renal Toxicity Induced by Cadmium in Rats. Toxicology 2007, 235, 185–193. [Google Scholar] [CrossRef]
- Hasani, M.; Djalalinia, S.; Khazdooz, M.; Asayesh, H.; Zarei, M.; Gorabi, A.M.; Ansari, H.; Qorbani, M.; Heshmat, R. Effect of Selenium Supplementation on Antioxidant Markers: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Hormones 2019, 18, 451–462. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Habeebu, S.S.; Liu, Y.; Klaassen, C.D. Acute CdMT Injection Is Not a Good Model to Study Chronic Cd Nephropathy: Comparison of Chronic CdCl2 and CdMT Exposure with Acute CdMT Injection in Rats. Toxicol. Appl. Pharmacol. 1998, 153, 48–58. [Google Scholar] [CrossRef]
- Thijssen, S.; Maringwa, J.; Faes, C.; Lambrichts, I.; Van Kerkhove, E. Chronic Exposure of Mice to Environmentally Relevant, Low Doses of Cadmium Leads to Early Renal Damage, Not Predicted by Blood or Urine Cadmium Levels. Toxicology 2007, 229, 145–156. [Google Scholar] [CrossRef]
- Damek-Poprawa, M.; Sawicka-Kapusta, K. Damage to the Liver, Kidney, and Testis with Reference to Burden of Heavy Metals in Yellow-Necked Mice from Areas around Steelworks and Zinc Smelters in Poland. Toxicology 2003, 186, 1–10. [Google Scholar] [CrossRef]
- Bernard, A.; Lauwerys, R. Early Markers of Cadmium Nephrotoxicity: Biological Significance and Predictive Value. Toxicol. Environ. Chem. 1990, 27, 65–72. [Google Scholar] [CrossRef]
- Bernard, A. Biomarkers of Metal Toxicity in Population Studies: Research Potential and Interpretation Issues. J. Toxicol. Environ. Health A 2008, 71, 1259–1265. [Google Scholar] [CrossRef]
- Park, J.D.; Cherrington, N.J.; Klaassen, C.D. Intestinal Absorption of Cadmium Is Associated with Divalent Metal Transporter 1 in Rats. Toxicol. Sci. 2002, 68, 288–294. [Google Scholar] [CrossRef]
- Johri, N.; Jacquillet, G.; Unwin, R. Heavy Metal Poisoning: The Effects of Cadmium on the Kidney. Biometals 2010, 23, 783–792. [Google Scholar] [CrossRef]
- Chen, L.; Lei, L.; Jin, T.; Nordberg, M.; Nordberg, G.F. Plasma Metallothionein Antibody, Urinary Cadmium, and Renal Dysfunction in a Chinese Type 2 Diabetic Population. Diabetes Care 2006, 29, 2682–2687. [Google Scholar] [CrossRef]
- Gan, L.; Liu, Q.; Xu, H.B.; Zhu, Y.S.; Yang, X.L. Effects of Selenium Overexposure on Glutathione Peroxidase and Thioredoxin Reductase Gene Expressions and Activities. Biol. Trace Elem. Res. 2002, 89, 165–175. [Google Scholar] [CrossRef]
- Verzola, D.; Bertolotto, M.B.; Villaggio, B.; Ottonello, L.; Dallegri, F.; Frumento, G.; Berruti, V.; Gandolfo, M.T.; Garibotto, G.; Deferran, G. Taurine Prevents Apoptosis Induced by High Glucose in Human Tuble Renal Cells. J. Investig. Med. 2002, 50, 443–451. [Google Scholar] [CrossRef]
- Messner, B.; Turkcan, A.; Ploner, C.; Laufer, G.; Bernhard, D. Cadmium Overkill: Autophagy, Apoptosis and Necrosis Signalling in Endothelial Cells Exposed to Cadmium. Cell. Mol. Life Sci. 2016, 73, 1699–1713. [Google Scholar] [CrossRef]
- Oltval, Z.N.; Milliman, C.L. Korsmeyer SJ Bcl-2 Heterodimerizes in Vivo with a Conserved Homolog, Bax, that Accelerates Programed Cell Death. Cell 1993, 74, 609–619. [Google Scholar] [CrossRef]
- Kinne-Saffran, E.; Hulseweh, M.; Pfaff, C.; Kinne, R.K. Inhibition of Na, K-ATPase by Cadmium: Different Mechanisms in Different Species. Toxicol. Appl. Pharmacol. 1993, 121, 22–29. [Google Scholar] [CrossRef]
- Wang, Y.; Fang, J.; Leonard, S.S.; Rao, K.M. Cadmium Inhibits the Electron Transfer Chain and Induces Reactive Oxygen Species. Free Radic. Biol. Med. 2004, 36, 1434–1443. [Google Scholar] [CrossRef]
- Stohs, S.J.; Bagchi, D.; Hassoun, E.; Bagchi, M. Oxidative Mechanisms in the Toxicity of Chromium and Cadmium Ions. J. Environ. Pathol. Toxicol. Oncol. 2000, 19, 201–213. [Google Scholar] [CrossRef] [PubMed]
- Tang, W.; Shaikh, Z.A. Renal Cortical Mitochondrial Dysfunction upon Cadmium Metallothionein Administration to Sprague-Dawley Rats. J. Toxicol. Environ. Health A 2001, 63, 221–235. [Google Scholar] [CrossRef] [PubMed]
- Thevenod, F. Nephrotoxicity and the Proximal Tubules. Insights from Cadmium. Nephron Physiol. 2003, 93, 87–93. [Google Scholar] [CrossRef]
- Desagher, S.; Martinou, J.C. Mitochondria as the Central Control Point of Apoptosis. Trends. Cell. Biol. 2000, 10, 369–377. [Google Scholar] [CrossRef]
- Thévenod, F.; Friedmann, J.M.; Katsen, A.D.; Hauser, I.A. Up-Regulation of Multidrug Resistance P-Glycoprotein via Nuclear Factor-KappaB Activation Protects Kidney Proximal Tubule Cells from Cadmium- and Reactive Oxygen Species-Induced Apoptosis. J. Biol. Chem. 2000, 275, 1887–1896. [Google Scholar] [CrossRef]
- Tzirogiannis, K.N.; Panoutsopoulos, G.I.; Demonakou, M.D.; Hereti, R.I.; Alexandropoulou, K.M.; Basayannis, A.C.; Mykoniatis, M.G. Time-Course of Cadmium-Induced Acute Hepatotoxicity in the Rat Liver: The Role of Apoptosis. Arch. Toxicol. 2003, 77, 694–701. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wu, Y.; Luo, K.; Liu, Y.; Zhou, M.; Yan, S.; Shi, H.; Cai, Y. The Protective Effects of Selenium on Cadmium-Induced Oxidative Stress and Apoptosis via Mitochondria Pathway in Mice Kidney. Food Chem. Toxicol. 2013, 58, 61–67. [Google Scholar] [CrossRef]
- Kayama, F.; Yoshida, T.; Elwell, M.R.; Luster, M.I. Cadmium-Induced Renal Damage and Proinflammatory Cytokines: Possible Role of IL-6 in Tubular Epithelial Cell Regeneration. Toxicol. Appl. Pharmacol. 1995, 134, 26–34. [Google Scholar] [CrossRef]
- Shaikh, Z.A.; Vu, T.; Zaman, K. Oxidative Stress as a Mechanism of Chronic Cadmium Hepatotoxicity and Nephrotoxicity and Protection by Antioxidants. Toxicol. Appl. Pharmacol. 1999, 154, 256–263. [Google Scholar] [CrossRef]
- Kataranovski, M.; Kataranovski, D.; Savic, D.; Jovcic, G.; Bogdanovic, Z.; Jovanovic, T. Granulocyte and Plasma Cytokine Activity in Acute Cadmium Intoxicationin Rats. Physiol. Res. 1998, 47, 453–461. [Google Scholar]
- Kataranovski, M.; Popovic, S.; Kataranovski, D. Differential Effects of in Vivocadmium Administration on Lymphocytes and Granulocytes in Rats. Vet. Hum. Toxicol. 1999, 41, 200–204. [Google Scholar]
- Tan, S.; Chi, Q.; Liu, T.; Sun, Z.; Min, Y.; Zhang, Z.; Li, S. Alleviation Mechanisms of Selenium on Cadmium-Spiked Neutrophil Injury to Chicken. Biol. Trace Elem. Res. 2017, 178, 301–309. [Google Scholar] [CrossRef]
| Target | Sense | Antisense |
|---|---|---|
| NF-ĸB 65 | 5′CTTGGCAACAGCACAGACC3′ | 5′GAGAAGTCCATGTCCGCAAT3′ |
| TNF-α | 5′CTGTAGCCCACGTCGTAGC3′ | 5′TTGAGATCCATGCCGTTG3′ |
| IL-6 | 5′AAAGAGTTGTGCAATGGCAATTCT3′ | 5′AAGTGCATCATCGTTGTTCATACA3′ |
| GAPDH | 5′CGACTTCAACAGCAACTCCCACTCTTCC3′ | 5′TGGGTGGTCCAGGGTTTCTTACTCCTT3′ |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vicas, S.I.; Laslo, V.; Timar, A.V.; Balta, C.; Herman, H.; Ciceu, A.; Gharbia, S.; Rosu, M.; Mladin, B.; Fritea, L.; et al. Functional Food Product Based on Nanoselenium-Enriched Lactobacillus casei against Cadmium Kidney Toxicity. Appl. Sci. 2021, 11, 4220. https://doi.org/10.3390/app11094220
Vicas SI, Laslo V, Timar AV, Balta C, Herman H, Ciceu A, Gharbia S, Rosu M, Mladin B, Fritea L, et al. Functional Food Product Based on Nanoselenium-Enriched Lactobacillus casei against Cadmium Kidney Toxicity. Applied Sciences. 2021; 11(9):4220. https://doi.org/10.3390/app11094220
Chicago/Turabian StyleVicas, Simona Ioana, Vasile Laslo, Adrian Vasile Timar, Cornel Balta, Hildegard Herman, Alina Ciceu, Sami Gharbia, Marcel Rosu, Bianca Mladin, Luminita Fritea, and et al. 2021. "Functional Food Product Based on Nanoselenium-Enriched Lactobacillus casei against Cadmium Kidney Toxicity" Applied Sciences 11, no. 9: 4220. https://doi.org/10.3390/app11094220
APA StyleVicas, S. I., Laslo, V., Timar, A. V., Balta, C., Herman, H., Ciceu, A., Gharbia, S., Rosu, M., Mladin, B., Fritea, L., Cavalu, S., Cotoraci, C., Prokisch, J., Puschita, M., Pop, C., Miutescu, E., & Hermenean, A. (2021). Functional Food Product Based on Nanoselenium-Enriched Lactobacillus casei against Cadmium Kidney Toxicity. Applied Sciences, 11(9), 4220. https://doi.org/10.3390/app11094220











