The Present and Future Role of Microfluidics for Protein and Peptide-Based Therapeutics and Diagnostics
Abstract
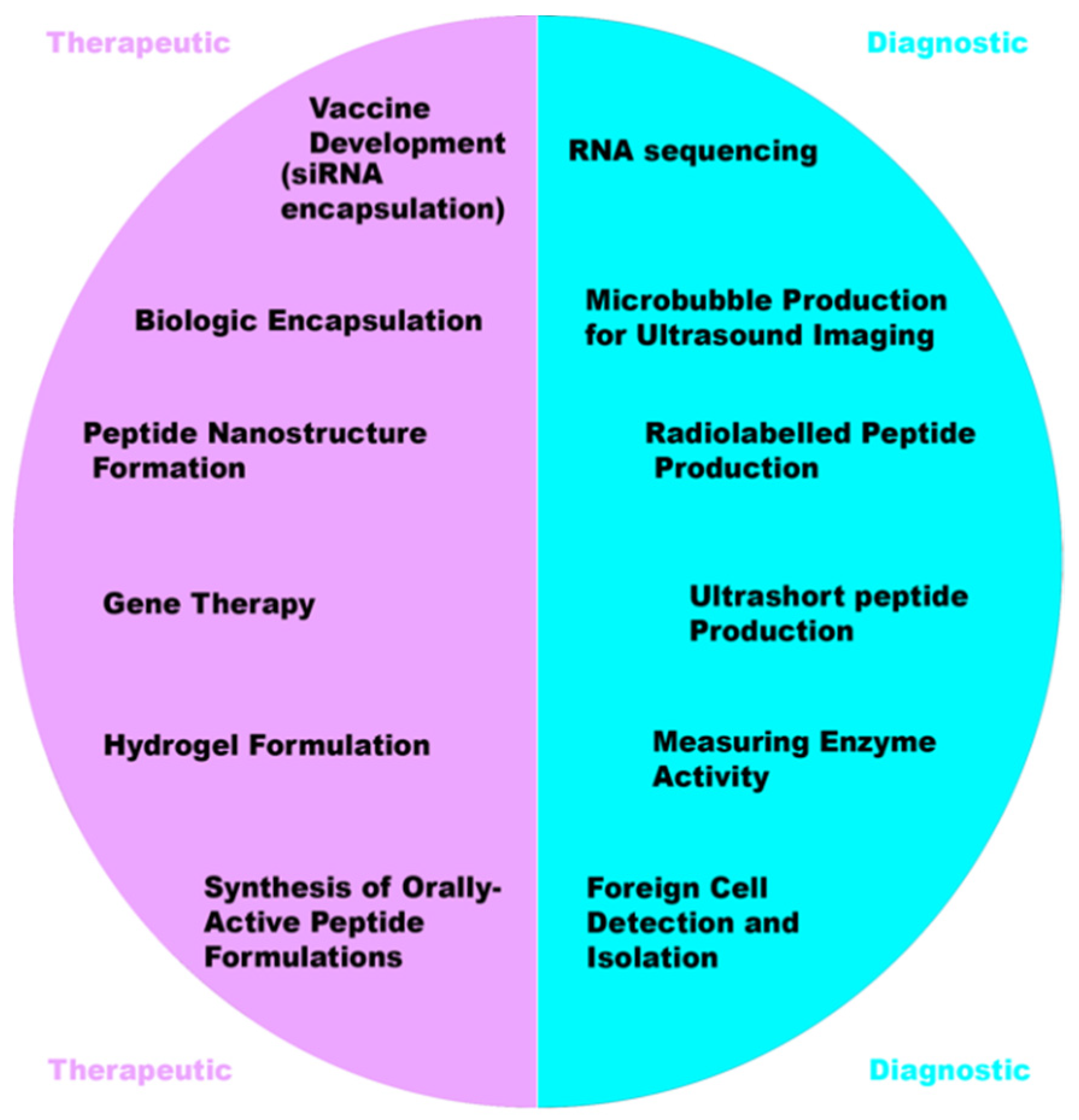
1. Introduction
- Assist in delivering an active pharmaceutical ingredient (API), both as carrier material and as a functionalized epitope, to provide region-specific delivery [17].
- Act as the API within a formulation;
- Provide unique binding regions to assist with the isolation of specific receptors [18];
- Enhance the quality of diagnostic imaging techniques [19].
Microfluidics
2. Therapeutic Applications of Peptides
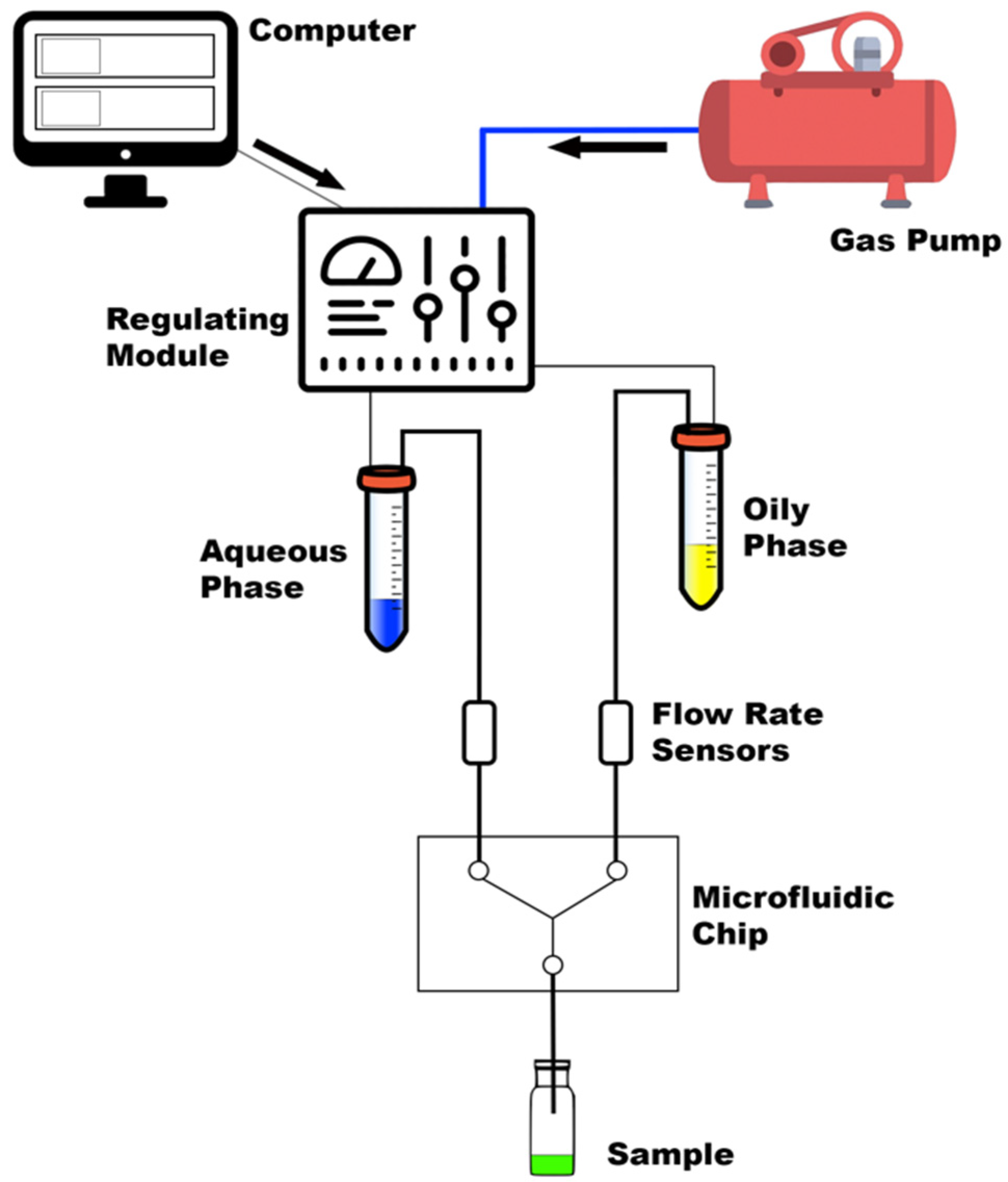
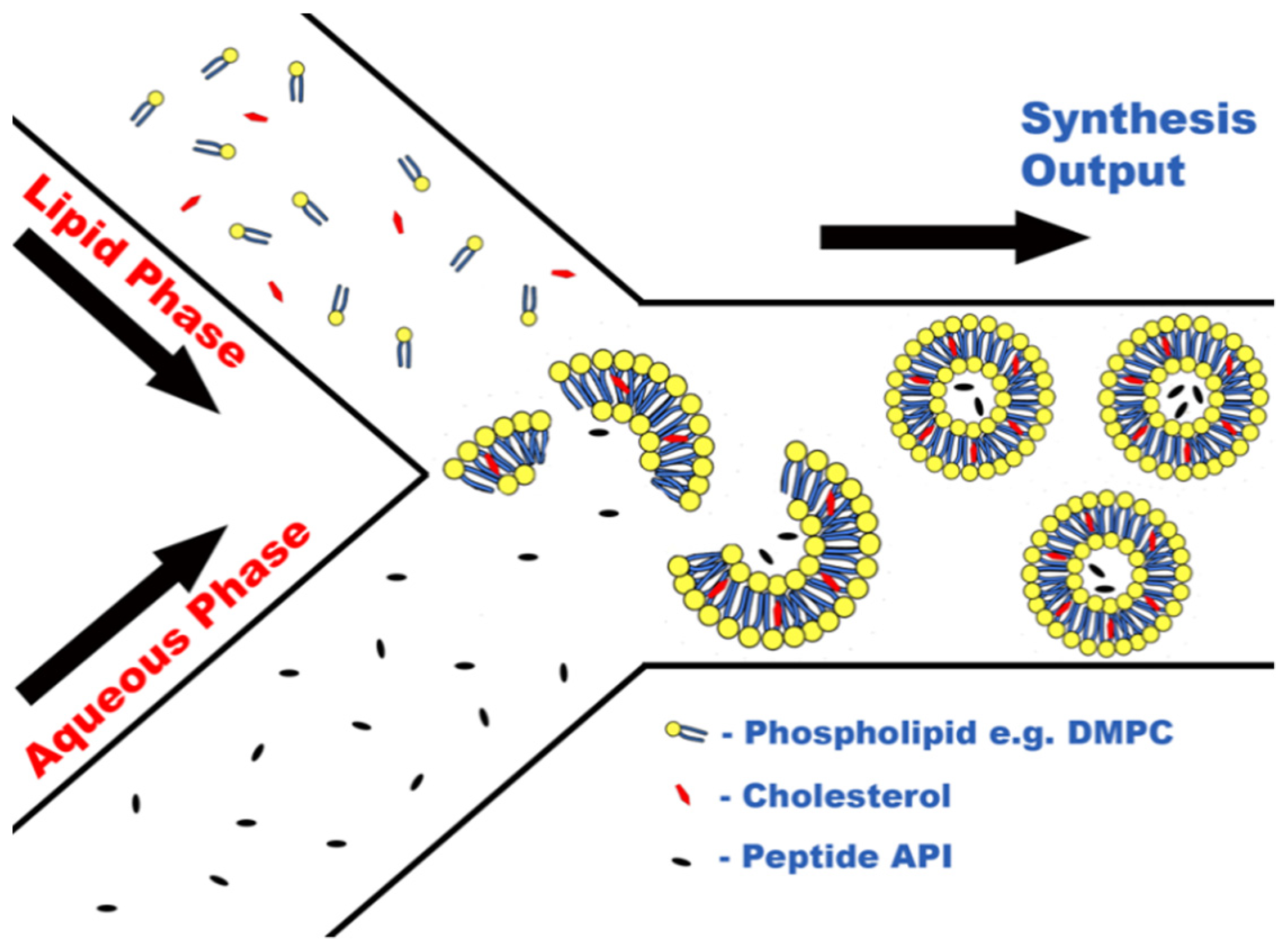
2.1. Peptide Encapsulation
2.2. Hydrogels and Peptide Antibiotics
2.3. Gene Therapy
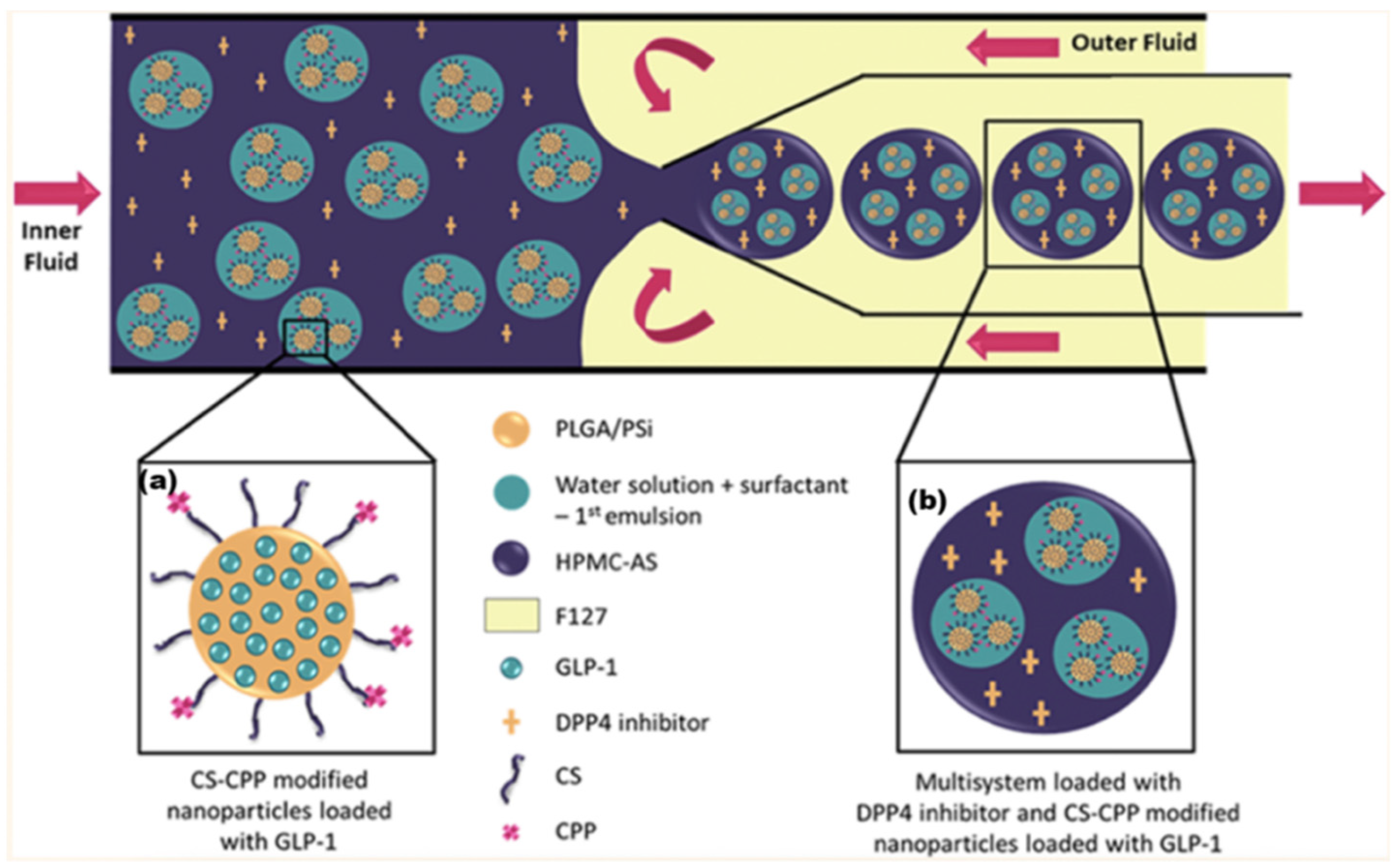
2.4. Oral Administration of Peptide-Based Medicines
3. Microfluidics as a Diagnostic Platform
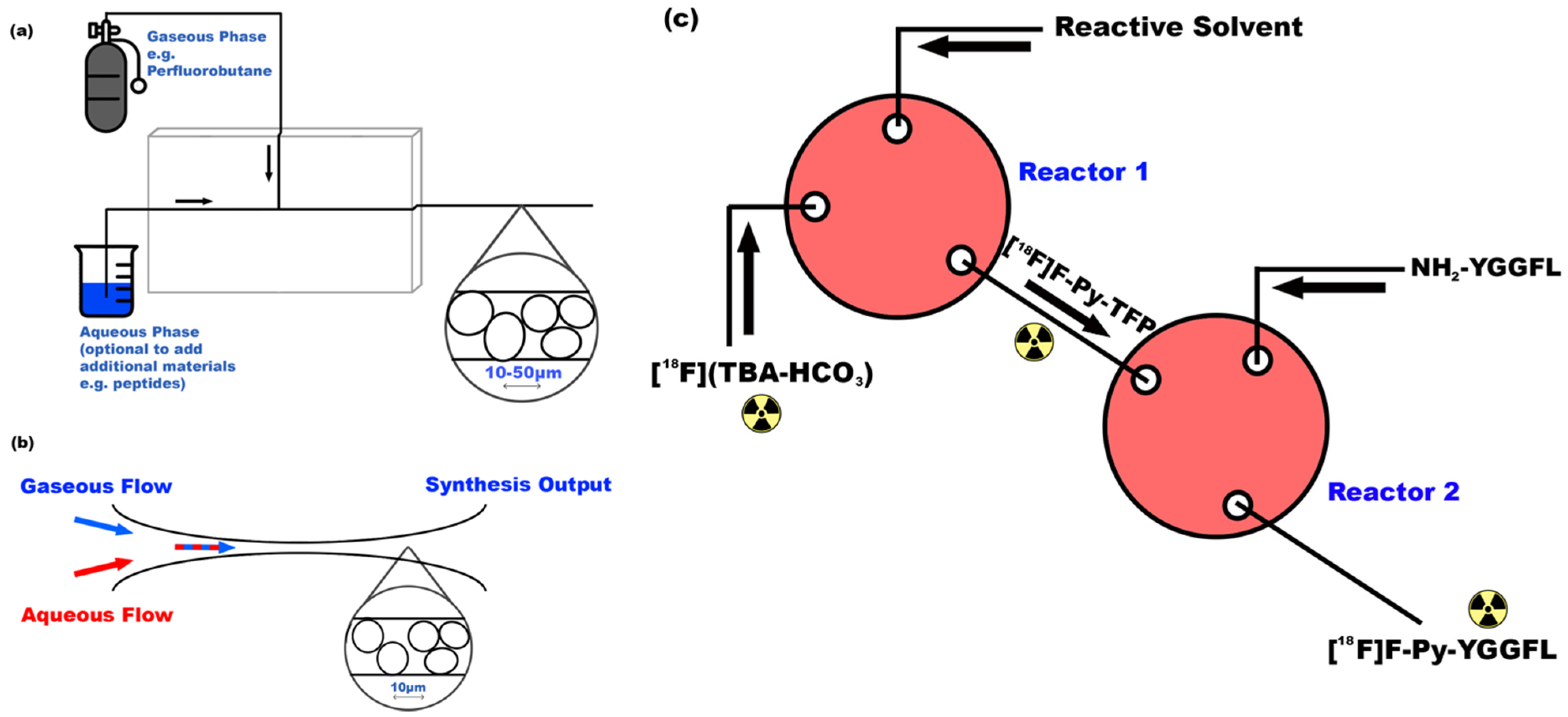
3.1. Microfluidics for Peptide Imaging
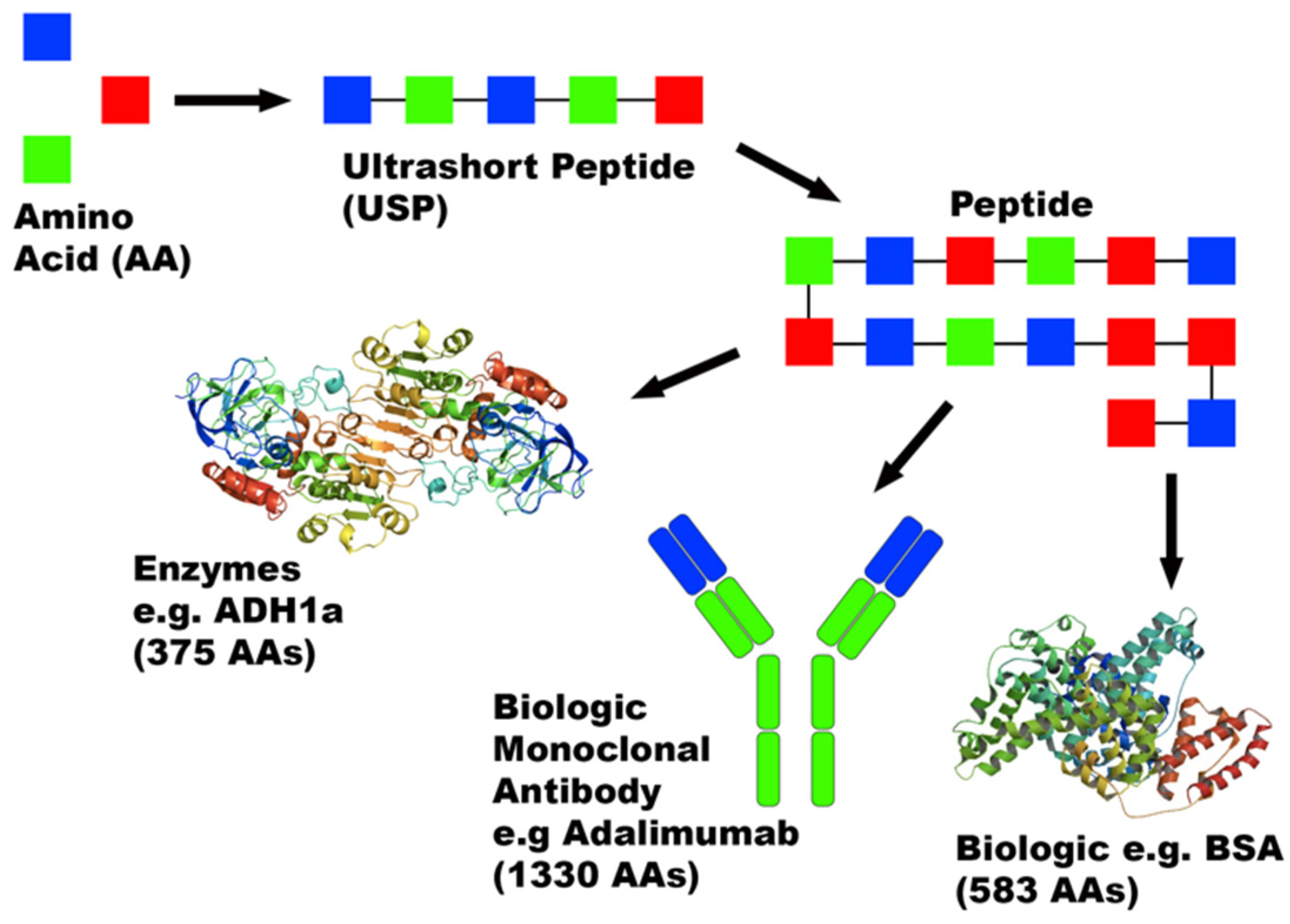
3.2. USP Diagnostics
3.3. Enzyme Activity and Biomarker Detection
3.4. Peptide-Conjugated Microbeads
4. Future Perspectives
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Polypeptides and Proteins. 3 January 2021. Available online: https://bio.libretexts.org/@go/page/3426 (accessed on 29 April 2021).
- Osorio, D.; Rondón-Villarreal, P.; Torres, R. Peptides: A package for data mining of antimicrobial peptides. Small 2015, 7, 4–14. [Google Scholar] [CrossRef]
- Marqus, S.; Pirogova, E.; Piva, T.J. Evaluation of the use of therapeutic peptides for cancer treatment. J. Biomed. Sci. 2017, 24, 21. [Google Scholar] [CrossRef] [PubMed]
- Hedges, J.B.; Ryan, K.S. Biosynthetic Pathways to Nonproteinogenic α-Amino Acids. Chem. Rev. 2020, 120, 3161–3209. [Google Scholar] [CrossRef] [PubMed]
- Marchese, L.; Nascimento, J.; Damasceno, F.; Bringaud, F.; Michels, P.; Silber, A. The Uptake and Metabolism of Amino Acids, and Their Unique Role in the Biology of Pathogenic Trypanosomatids. Pathogens 2018, 7, 36. [Google Scholar] [CrossRef] [PubMed]
- Fosgerau, K.; Hoffmann, T. Peptide therapeutics: Current status and future directions. Drug Discov. Today 2015, 20, 122–128. [Google Scholar] [CrossRef] [PubMed]
- Vllasaliu, D.; Thanou, M.; Stolnik, S.; Fowler, R. Recent advances in oral delivery of biologics: Nanomedicine and physical modes of delivery. Expert Opin. Drug Deliv. 2018, 15, 759–770. [Google Scholar] [CrossRef]
- Meade, E.; Slattery, M.A.; Garvey, M. Bacteriocins, Potent Antimicrobial Peptides and the Fight against Multi Drug Resistant Species: Resistance Is Futile? Antibiotics 2020, 9, 32. [Google Scholar] [CrossRef]
- Roudi, R.; Syn, N.L.; Roudbary, M. Antimicrobial Peptides As Biologic and Immunotherapeutic Agents against Cancer: A Comprehensive Overview. Front. Immunol. 2017, 8, 1320. [Google Scholar] [CrossRef]
- Cutone, A.; Rosa, L.; Ianiro, G.; Lepanto, M.S.; Bonaccorsi Di Patti, M.C.; Valenti, P.; Musci, G. Lactoferrin’s Anti-Cancer Properties: Safety, Selectivity, and Wide Range of Action. Biomolecules 2020, 10, 456. [Google Scholar] [CrossRef]
- Hazam, P.K.; Jerath, G.; Kumar, A.; Chaudhary, N.; Ramakrishnan, V. Effect of tacticity-derived topological constraints in bactericidal peptides. Biochim. Biophys. Acta Biomembr. 2017, 1859, 1388–1395. [Google Scholar] [CrossRef]
- Otieno, B.A.; Krause, C.E.; Jones, A.L.; Kremer, R.B.; Rusling, J.F. Cancer Diagnostics via Ultrasensitive Multiplexed Detection of Parathyroid Hormone-Related Peptides with a Microfluidic Immunoarray. Anal. Chem. 2016, 88, 9269–9275. [Google Scholar] [CrossRef]
- Kovalainen, M.; Mönkäre, J.; Riikonen, J.; Pesonen, U.; Vlasova, M.; Salonen, J.; Lehto, V.-P.; Järvinen, K.; Herzig, K.-H. Novel Delivery Systems for Improving the Clinical Use of Peptides. Pharmacol. Rev. 2015, 67, 541–561. [Google Scholar] [CrossRef]
- Hosseini, S.F.; Ramezanzade, L.; McClements, D.J. Recent advances in nanoencapsulation of hydrophobic marine bioactives: Bioavailability, safety, and sensory attributes of nano-fortified functional foods. Trends Food Sci. Technol. 2021, 109, 322–339. [Google Scholar] [CrossRef]
- Turecek, P.L.; Bossard, M.J.; Schoetens, F.; Ivens, I.A. PEGylation of Biopharmaceuticals: A Review of Chemistry and Nonclinical Safety Information of Approved Drugs. J. Pharm. Sci. 2016, 105, 460–475. [Google Scholar] [CrossRef] [PubMed]
- Cao, M.; Xing, R.; Chang, R.; Wang, Y.; Yan, X. Peptide-coordination self-assembly for the precise design of theranostic nanodrugs. Coord. Chem. Rev. 2019, 397, 14–27. [Google Scholar] [CrossRef]
- Knauer, N.; Pashkina, E.; Apartsin, E. Topological Aspects of the Design of Nanocarriers for Therapeutic Peptides and Proteins. Pharmaceutics 2019, 11, 91. [Google Scholar] [CrossRef] [PubMed]
- Sibener, L.V.; Fernandes, R.A.; Kolawole, E.M.; Carbone, C.B.; Liu, F.; McAffee, D.; Birnbaum, M.E.; Yang, X.; Su, L.F.; Yu, W.; et al. Isolation of a Structural Mechanism for Uncoupling T Cell Receptor Signaling from Peptide-MHC Binding. Cell 2018, 174, 672–687. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Li, Y.; Liu, T.; Li, Z.; Zhang, X.; Chen, X. Peptide-based imaging agents for cancer detection. Adv. Drug Deliv. Rev. 2017, 110–111, 38–51. [Google Scholar] [CrossRef]
- Convery, N.; Gadegaard, N. 30 years of microfluidics. Micro Nano Eng. 2019, 2, 76–91. [Google Scholar] [CrossRef]
- Fei, S.; Hwang, W.; Mukherjee, A.; Chakrabarty, K. Defect-Oriented Testing and Diagnosis of Digital Microfluidicsbased Biochips. In Proceedings of the IEEE International Conference on Test, Austin, TX, USA, 8 November 2005. [Google Scholar]
- Gogoi, S.; Gogoi, S.B. Review on microfluidic studies for EOR application. J. Pet. Explor. Prod. Technol. 2019, 9, 2263–2277. [Google Scholar] [CrossRef]
- Song, L.; Gao, W.; Rahmat-Samii, Y. 3-D Printed Microfluidics Channelizing Liquid Metal for Multipolarization Reconfigurable Extended E-Shaped Patch Antenna. IEEE Trans. Antennas Propag. 2020, 68, 6867–6878. [Google Scholar] [CrossRef]
- Feng, J.; Yuan, J.; Cho, S.K. Micropropulsion by an acoustic bubble for navigating microfluidic spaces. Lab Chip 2015, 15, 1554–1562. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Korolj, A.; Lai, B.F.L.; Radisic, M. Advances in organ-on-a-chip engineering. Nat. Rev. Mater. 2018, 3, 257–278. [Google Scholar] [CrossRef]
- Ren, K.; Zhou, J.; Wu, H. Materials for Microfluidic Chip Fabrication. Acc. Chem. Res. 2013, 46, 2396–2406. [Google Scholar] [CrossRef] [PubMed]
- Gupta, N.; Liu, J.R.; Patel, B.; Solomon, D.E.; Vaidya, B.; Gupta, V. Microfluidics-based 3D cell culture models: Utility in novel drug discovery and delivery research. Bioeng. Transl. Med. 2016, 1, 63–81. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Yan, S.; Miao, C.; Li, L.; Shi, W.; Liu, X.; Luo, Y.; Liu, T.; Lin, B.; Wu, W.; et al. Paper Microfluidics for Cell Analysis. Adv. Healthc. Mater. 2019, 8, 1801084. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Z.; Yang, C.J. Hydrogel Droplet Microfluidics for High-Throughput Single Molecule/Cell Analysis. Acc. Chem. Res. 2017, 50, 22–31. [Google Scholar] [CrossRef]
- Pento, J.T. Monoclonal antibodies for the treatment of cancer. Anticancer Res. 2017, 37, 5935–5939. [Google Scholar]
- Hildebrand, D.; Metz-Zumaran, C.; Jaschkowitz, G.; Heeg, K. Silencing SOCS1 via Liposome-Packed siRNA Sustains TLR4-Ligand Adjuvant. Front. Immunol. 2019, 10, 1279. [Google Scholar] [CrossRef]
- Le, T.T.; Andreadakis, Z.; Kumar, A.; Román, R.G.; Tollefsen, S.; Saville, M.; Mayhew, S. The COVID-19 vaccine development landscape. Nat. Rev. Drug Discov. 2020, 19, 305–306. [Google Scholar] [CrossRef]
- Koutsopoulos, S. Self-assembling peptide nanofiber hydrogels in tissue engineering and regenerative medicine: Progress, design guidelines, and applications. J. Biomed. Mater. Res. Part A 2016, 104, 1002–1016. [Google Scholar] [CrossRef]
- Zhang, W.; Taheri-Ledari, R.; Hajizadeh, Z.; Zolfaghari, E.; Ahghari, M.R.; Maleki, A.; Hamblin, M.R.; Tian, Y. Enhanced activity of vancomycin by encapsulation in hybrid magnetic nanoparticles conjugated to a cell-penetrating peptide. Nanoscale 2020, 12, 3855–3870. [Google Scholar] [CrossRef] [PubMed]
- McClements, D.J. Encapsulation, protection, and delivery of bioactive proteins and peptides using nanoparticle and microparticle systems: A review. Adv. Colloid Interface Sci. 2018, 253, 1–22. [Google Scholar] [CrossRef]
- Wong, C.Y.; Al-Salami, H.; Dass, C.R. The role of chitosan on oral delivery of peptide-loaded nanoparticle formulation. J. Drug Target. 2018, 26, 551–562. [Google Scholar] [CrossRef] [PubMed]
- Ross, C.; Taylor, M.; Fullwood, N.; Allsop, D. Liposome delivery systems for the treatment of Alzheimer’s disease. Int. J. Nanomed. 2018, 13, 8507–8522. [Google Scholar] [CrossRef] [PubMed]
- Ibaraki, H.; Kanazawa, T.; Kurano, T.; Oogi, C.; Takashima, Y.; Seta, Y. Anti-RelA siRNA-Encapsulated Flexible Liposome with Tight Junction-Opening Peptide as a Non-invasive Topical Therapeutic for Atopic Dermatitis. Biol. Pharm. Bull. 2019, 42, 1216–1225. [Google Scholar] [CrossRef] [PubMed]
- Lindqvist, A.; Rip, J.; van Kregten, J.; Gaillard, P.J.; Hammarlund-Udenaes, M. In vivo functional evaluation of increased brain delivery of the opioid peptide DAMGO by Glutathione-PEGylated liposomes. Pharm. Res. 2016, 33, 177–185. [Google Scholar] [CrossRef]
- Oshima, A.; Sumitomo, K. Vesicle fusion with bilayer lipid membrane controlled by electrostatic interaction. Biochem. Biophys. Rep. 2017, 11, 58–63. [Google Scholar] [CrossRef]
- Forbes, N.; Hussain, M.T.; Briuglia, M.L.; Edwards, D.P.; Horst, J.H.T.; Szita, N.; Perrie, Y. Rapid and scale-independent microfluidic manufacture of liposomes entrapping protein incorporating in-line purification and at-line size monitoring. Int. J. Pharm. 2019, 556, 68–81. [Google Scholar] [CrossRef]
- Zhang, H. Thin-Film Hydration Followed by Extrusion Method for Liposome Preparation. Methods Mol. Biol. 2017, 1522, 17–22. [Google Scholar] [CrossRef]
- Corey, L.; Mascola, J.R.; Fauci, A.S.; Collins, F.S. A strategic approach to COVID-19 vaccine R&D. Science 2020, 368, 948–950. [Google Scholar] [CrossRef] [PubMed]
- Leung, A.K.K.; Tam, Y.Y.C.; Chen, S.; Hafez, I.M.; Cullis, P.R. Microfluidic Mixing: A General Method for Encapsulating Macromolecules in Lipid Nanoparticle Systems. J. Phys. Chem. B 2015, 119, 8698–8706. [Google Scholar] [CrossRef]
- Moon, H.-S.; Je, K.; Min, J.-W.; Park, D.; Han, K.-Y.; Shin, S.-H.; Park, W.-Y.; Yoo, C.E.; Kim, S.-H. Inertial-ordering-assisted droplet microfluidics for high-throughput single-cell RNA-sequencing. Lab Chip 2018, 18, 775–784. [Google Scholar] [CrossRef] [PubMed]
- Taylor, N.; Elbaum-Garfinkle, S.; Vaidya, N.; Zhang, H.; Stone, H.A.; Brangwynne, C.P. Biophysical characterization of organelle-based RNA/protein liquid phases using microfluidics. Soft Matter 2016, 12, 9142–9150. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.-H.; Lin, C.-C.; Thomas, J.L.; Chan, C.-K.; Lin, H.-Y. Epitope recognition of magnetic peptide-imprinted chitosan composite nanoparticles for the extraction of CRISPR/dCas9a proteins from transfected cells. Nanotechnology 2021, 32, 18LT02. [Google Scholar] [CrossRef]
- Roozbehani, M.; Falak, R.; Mohammadi, M.; Hemphill, A.; Razmjou, E.; Meamar, A.R.; Masoori, L.; Khoshmirsafa, M.; Moradi, M.; Gharavi, M.J. Characterization of a multi-epitope peptide with selective MHC-binding capabilities encapsulated in PLGA nanoparticles as a novel vaccine candidate against Toxoplasma gondii infection. Vaccine 2018, 36, 6124–6132. [Google Scholar] [CrossRef]
- Gessner, I.; Neundorf, I. Nanoparticles Modified with Cell-Penetrating Peptides: Conjugation Mechanisms, Physicochemical Properties, and Application in Cancer Diagnosis and Therapy. Int. J. Mol. Sci. 2020, 21, 2536. [Google Scholar] [CrossRef]
- Field, L.D.; Delehanty, J.B.; Chen, Y.; Medintz, I.L. Peptides for Specifically Targeting Nanoparticles to Cellular Organelles: Quo Vadis? Acc. Chem. Res. 2015, 48, 1380–1390. [Google Scholar] [CrossRef]
- Liu, K.; Li, H.; Williams, G.R.; Wu, J.; Zhu, L.-M. pH-responsive liposomes self-assembled from electrosprayed microparticles, and their drug release properties. Colloids Surfaces A Physicochem. Eng. Asp. 2018, 537, 20–27. [Google Scholar] [CrossRef]
- Schiller, J.L.; Lai, S.K. Tuning Barrier Properties of Biological Hydrogels. ACS Appl. Bio Mater. 2020, 3, 2875–2890. [Google Scholar] [CrossRef]
- Chan, K.H.; Tay, J.J.J. Advancement of Peptide Nanobiotechnology via Emerging Microfluidic Technology. Micromachines 2019, 10, 627. [Google Scholar] [CrossRef] [PubMed]
- Cui, T.; Li, X.; He, S.; Xu, D.; Yin, L.; Huang, X.; Deng, S.; Yue, W.; Zhong, W. Instant Self-Assembly Peptide Hydrogel Encapsulation with Fibrous Alginate by Microfluidics for Infected Wound Healing. ACS Biomater. Sci. Eng. 2020, 6, 5001–5011. [Google Scholar] [CrossRef] [PubMed]
- Jain, R.; Khandelwal, G.; Roy, S. Unraveling the Design Rules in Ultrashort Amyloid-Based Peptide Assemblies toward Shape-Controlled Synthesis of Gold Nanoparticles. Langmuir 2019, 35, 5878–5889. [Google Scholar] [CrossRef] [PubMed]
- Gireesha, B.; Sowmya, G.; Gorla, R.S.R. Nanoparticle shape effect on the thermal behaviour of moving longitudinal porous fin. Proc. Inst. Mech. Eng. Part N J. Nanomater. Nanoeng. Nanosyst. 2020, 234, 115–121. [Google Scholar] [CrossRef]
- Parsley, N.C.; Smythers, A.L.; Hicks, L.M. Implementation of Microfluidics for Antimicrobial Susceptibility Assays: Issues and Optimization Requirements. Front. Cell. Infect. Microbiol. 2020, 10, 547177. [Google Scholar] [CrossRef] [PubMed]
- Di Santo, R.; Digiacomo, L.; Palchetti, S.; Palmieri, V.; Perini, G.; Pozzi, D.; Papi, M.; Caracciolo, G. Microfluidic manufacturing of surface-functionalized graphene oxide nanoflakes for gene delivery. Nanoscale 2019, 11, 2733–2741. [Google Scholar] [CrossRef] [PubMed]
- Imani, R.; Emami, S.H.; Faghihi, S. Synthesis and characterization of an octaarginine functionalized graphene oxide nano-carrier for gene delivery applications. Phys. Chem. Chem. Phys. 2015, 17, 6328–6339. [Google Scholar] [CrossRef]
- Araújo, F.; Shrestha, N.; Shahbazi, M.A.; Liu, D.; Herranz-Blanco, B.; Mäkilä, E.M.; Salonen, J.J.; Hirvonen, J.T.; Granja, P.L.; Sarmento, B.; et al. Microfluidic Assembly of a Multifunctional Tailorable Composite System Designed for Site Specific Combined Oral Delivery of Peptide Drugs. ACS Nano 2015, 9, 8291–8302. [Google Scholar] [CrossRef]
- Unni, E.; Sternbach, N.; Goren, A. Using the Medication Adherence Reasons Scale (MAR-Scale) to identify the reasons for non-adherence across multiple disease conditions. Patient Prefer. Adherence 2019, 13, 993–1004. [Google Scholar] [CrossRef]
- Cheang, J.Y.; Moyle, P.M. Glucagon-Like Peptide-1 (GLP-1)-Based Therapeutics: Current Status and Future Opportunities beyond Type 2 Diabetes. ChemMedChem 2018, 13, 662–671. [Google Scholar] [CrossRef]
- Fiabane, J.; Prentice, P.; Pancholi, K. High Yielding Microbubble Production Method. BioMed Res. Int. 2016, 2016, 3572827. [Google Scholar] [CrossRef]
- Pulsipher, K.W.; Hammer, D.A.; Lee, D.; Sehgal, C.M. Engineering Theranostic Microbubbles Using Microfluidics for Ultrasound Imaging and Therapy: A Review. Ultrasound Med. Biol. 2018, 44, 2441–2460. [Google Scholar] [CrossRef]
- Angilè, F.E.; Vargo, K.B.; Sehgal, C.M.; Hammer, D.A.; Lee, D. Recombinant Protein-Stabilized Monodisperse Microbubbles with Tunable Size Using a Valve-Based Microfluidic Device. Langmuir 2014, 30, 12610–12618. [Google Scholar] [CrossRef]
- Cumming, R.C.; Olberg, D.E.; Sutcliffe, J.L. Rapid18F-radiolabeling of peptides from [18F]fluoride using a single microfluidics device. RSC Adv. 2014, 4, 49529–49534. [Google Scholar] [CrossRef]
- Kesch, C.; Kratochwil, C.; Mier, W.; Kopka, K.; Giesel, F.L. 68Ga or18F for Prostate Cancer Imaging? J. Nucl. Med. 2017, 58, 687–688. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Tian, M.; Zhang, H. Microfluidics for Synthesis of Peptide-Based PET Tracers. BioMed Res. Int. 2013, 2013, 1–8. [Google Scholar] [CrossRef]
- Ni, M.; Tresset, G.; Iliescu, C.; Hauser, C.A.E. Ultrashort Peptide Theranostic Nanoparticles by Microfluidic-Assisted Rapid Solvent Exchange. IEEE Trans. NanoBiosci. 2020, 19, 627–632. [Google Scholar] [CrossRef]
- Miles, J.J.; Tan, M.P.; Dolton, G.; Edwards, E.S.J.; Galloway, S.A.E.; Laugel, B.; Clement, M.; Makinde, J.; Ladell, K.; Matthews, K.K.; et al. Peptide mimic for influenza vaccination using nonnatural combinatorial chemistry. J. Clin. Investig. 2018, 128, 1569–1580. [Google Scholar] [CrossRef] [PubMed]
- Srisa-Art, M.; Noblitt, S.D.; Krummel, A.T.; Henry, C.S. IR-Compatible PDMS microfluidic devices for monitoring of enzyme kinetics. Anal. Chim. Acta 2018, 1021, 95–102. [Google Scholar] [CrossRef] [PubMed]
- Grant, J.; Goudarzi, S.H.; Mrksich, M. High-Throughput Enzyme Kinetics with 3D Microfluidics and Imaging SAMDI Mass Spectrometry. Anal. Chem. 2018, 90, 13096–13103. [Google Scholar] [CrossRef]
- Briones, J.C.; Espulgar, W.V.; Koyama, S.; Yoshikawa, H.; Park, J.; Naito, Y.; Kumanogoh, A.; Tamiya, E.; Takamatsu, H.; Saito, M. A Microfluidic Platform for Single Cell Fluorometric Granzyme B Profiling. Theranostics 2020, 10, 123–132. [Google Scholar] [CrossRef] [PubMed]
- Hwang, H.K.; Kim, H.-I.; Kim, S.H.; Choi, J.; Kang, C.M.; Kim, K.S.; Lee, W.J. Prognostic impact of the tumor-infiltrating regulatory T-cell (Foxp3+)/activated cytotoxic T lymphocyte (granzyme B+) ratio on resected left-sided pancreatic cancer. Oncol. Lett. 2016, 12, 4477–4484. [Google Scholar] [CrossRef]
- Varillas, J.I.; Zhang, J.; Chen, K.; Barnes, I.I.; Liu, C.; George, T.J.; Fan, Z.H. Microfluidic Isolation of Circulating Tumor Cells and Cancer Stem-Like Cells from Patients with Pancreatic Ductal Adenocarcinoma. Theranostics 2019, 9, 1417–1425. [Google Scholar] [CrossRef]
- Safa, N.; Vaithiyanathan, M.; Sombolestani, S.; Charles, S.; Melvin, A.T. Population-based analysis of cell-penetrating peptide uptake using a microfluidic droplet trapping array. Anal. Bioanal. Chem. 2019, 411, 2729–2741. [Google Scholar] [CrossRef]
- Suwatthanarak, T.; Thiodorus, I.A.; Tanaka, M.; Shimada, T.; Takeshita, D.; Yasui, T.; Baba, Y.; Okochi, M. Microfluidic-based capture and release of cancer-derived exosomes via peptide–nanowire hybrid interface. Lab Chip 2021, 21, 597–607. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.; Liao, X.; Tian, Y.; Li, G. Exosome separation using microfluidic systems: Size-based, immunoaffinity-based and dynamic methodologies. Biotechnol. J. 2017, 12, 1600699. [Google Scholar] [CrossRef] [PubMed]
- Panagiotara, A.; Markou, A.; Lianidou, E.S.; Patrinos, G.P.; Katsila, T. Exosomes: A Cancer Theranostics Road Map. Public Health Genom. 2017, 20, 116–125. [Google Scholar] [CrossRef]
- Tang, M.Y.H.; Shum, H.C. One-step immunoassay of C-reactive protein using droplet microfluidics. Lab Chip 2016, 16, 4359–4365. [Google Scholar] [CrossRef] [PubMed]
- Cho, C.-F.; Ghotmi, Y.; Fazden, C.; Wolfe, J.; Bononi, F.; Murrell, E.; Luyt, L.; Viapiano, M.; Pentelute, B.; Chiocca, E.A.; et al. DDIS-19. Novel Peptide Homing to Glioma-Specific Isoform of Brevican Selectively Targets Malignant Brain Tumors. Neuro-Oncol. 2017, 19, vi62–vi63. [Google Scholar] [CrossRef]
- Qi, J.; Li, B.; Wang, X.; Fu, L.; Luo, L.; Chen, L. Rotational Paper-Based Microfluidic-Chip Device for Multiplexed and Simultaneous Fluorescence Detection of Phenolic Pollutants Based on a Molecular-Imprinting Technique. Anal. Chem. 2018, 90, 11827–11834. [Google Scholar] [CrossRef] [PubMed]
- Plunkett, A.; Tong, J. Sepsis in children. BMJ 2015, 350, h3017. [Google Scholar] [CrossRef] [PubMed]
- Na, W.; Nam, D.; Lee, H.; Shin, S. Rapid molecular diagnosis of infectious viruses in microfluidics using DNA hydrogel formation. Biosens. Bioelectron. 2018, 108, 9–13. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; He, L.; Li, W.; Li, H.; Wong, W.-M.; Ramakrishna, S.; Wu, W. Functional self-assembling peptide nanofiber hydrogel for peripheral nerve regeneration. Regen. Biomater. 2017, 4, 21–30. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, M.; Halperin-Sternfeld, M.; Grinberg, I.; Adler-Abramovich, L. Injectable Alginate-Peptide Composite Hydrogel as a Scaffold for Bone Tissue Regeneration. Nanomaterials 2019, 9, 497. [Google Scholar] [CrossRef] [PubMed]
| Glass/Silicon | PDMS | Hydrogels | Paper |
|---|---|---|---|
| Advantages | |||
| <100 nm channel dimensions possible [26] | Easy and cheap to synthesize | Can mimic extracellular environment | Regions can be modified to be hydrophobic |
| Reduces cost of processes, such as capillary electrophoresis [26] | High level of adaptability | Useful for cell encapsulation for 3D cultures [27] | Useful for controlling liquid absorption |
| Low permeability to oxygen | Oxygen permeability allows for cell culture assays [27] | Low cost [28] | |
| Resistant to organic solvents | Allows nanometer-resolution from 3D printing | External pump, e.g., gas not necessarily required [28] | |
| High elasticity | |||
| Disadvantages | |||
| High-cost of initial microfabrication | Low compatibility with organic solvents | Low-level resolution [29] | Low scope of detection Properties |
| Valve fabrication is difficult | Absorbent for small hydrophobic molecules | Poor for microfabrication methods | Unsuitable for liquids with low surface tension |
| Cannot be used for cell cultures | Limited applications for MFs | ||
| Microfluidics | Unilamellar Vesicle Fusion | Thin-Film Hydration |
|---|---|---|
| Advantages | ||
| Allows synthesis at room temperature | Simple method | Simple method |
| Good control over PDI and particle size | Process can be controlled via electrostatic manipulation [40] | Acceptable encapsulation efficiencies for smaller peptides |
| Easy method to scale-up | ||
| High encapsulation efficiencies [41] | ||
| Continuous process | ||
| Disadvantages | ||
| Material interactions with MF chip | Less control over PDI and particle size | Less control over PDI and particle size |
| Requires initial high-cost system | Affected greatly by temperature | Produces heterogeneous particle population that requires extrusion/sonication [42] |
| Batch process | ||
| Condition | Associated Peptide Biomarker(s) |
|---|---|
| Leukemia | Transforming growth factor ß1 (TGF-ß1) |
| Melanoma | Caveolin-1 CD63 CTLA4 |
| Alzheimer’s disease | Apolipoprotein E (APOE) |
| Diabetes prediabetes | Hemoglobin A1c (HbA1c) |
| Breast cancer | HER2 Tissue polypeptide antigen * |
| HIV/AIDS | Plasmodium falciparum histidine-rich-protein 2 (PfHRP2) * |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Weaver, E.; Uddin, S.; Cole, D.K.; Hooker, A.; Lamprou, D.A. The Present and Future Role of Microfluidics for Protein and Peptide-Based Therapeutics and Diagnostics. Appl. Sci. 2021, 11, 4109. https://doi.org/10.3390/app11094109
Weaver E, Uddin S, Cole DK, Hooker A, Lamprou DA. The Present and Future Role of Microfluidics for Protein and Peptide-Based Therapeutics and Diagnostics. Applied Sciences. 2021; 11(9):4109. https://doi.org/10.3390/app11094109
Chicago/Turabian StyleWeaver, Edward, Shahid Uddin, David K. Cole, Andrew Hooker, and Dimitrios A. Lamprou. 2021. "The Present and Future Role of Microfluidics for Protein and Peptide-Based Therapeutics and Diagnostics" Applied Sciences 11, no. 9: 4109. https://doi.org/10.3390/app11094109
APA StyleWeaver, E., Uddin, S., Cole, D. K., Hooker, A., & Lamprou, D. A. (2021). The Present and Future Role of Microfluidics for Protein and Peptide-Based Therapeutics and Diagnostics. Applied Sciences, 11(9), 4109. https://doi.org/10.3390/app11094109








