Vitamin D and Primary Ciliary Dyskinesia: A Topic to Be Further Explored
Abstract
1. Introduction
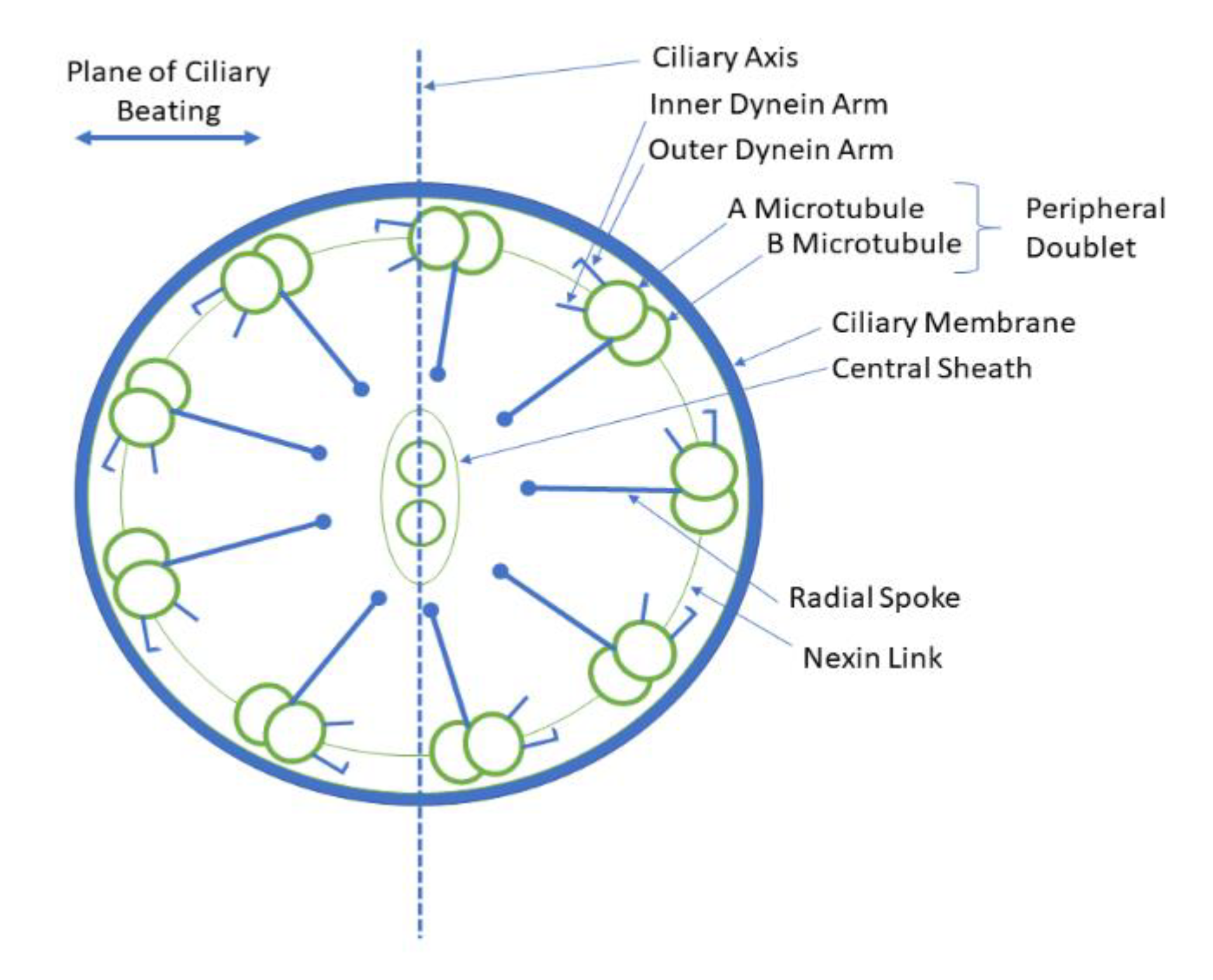
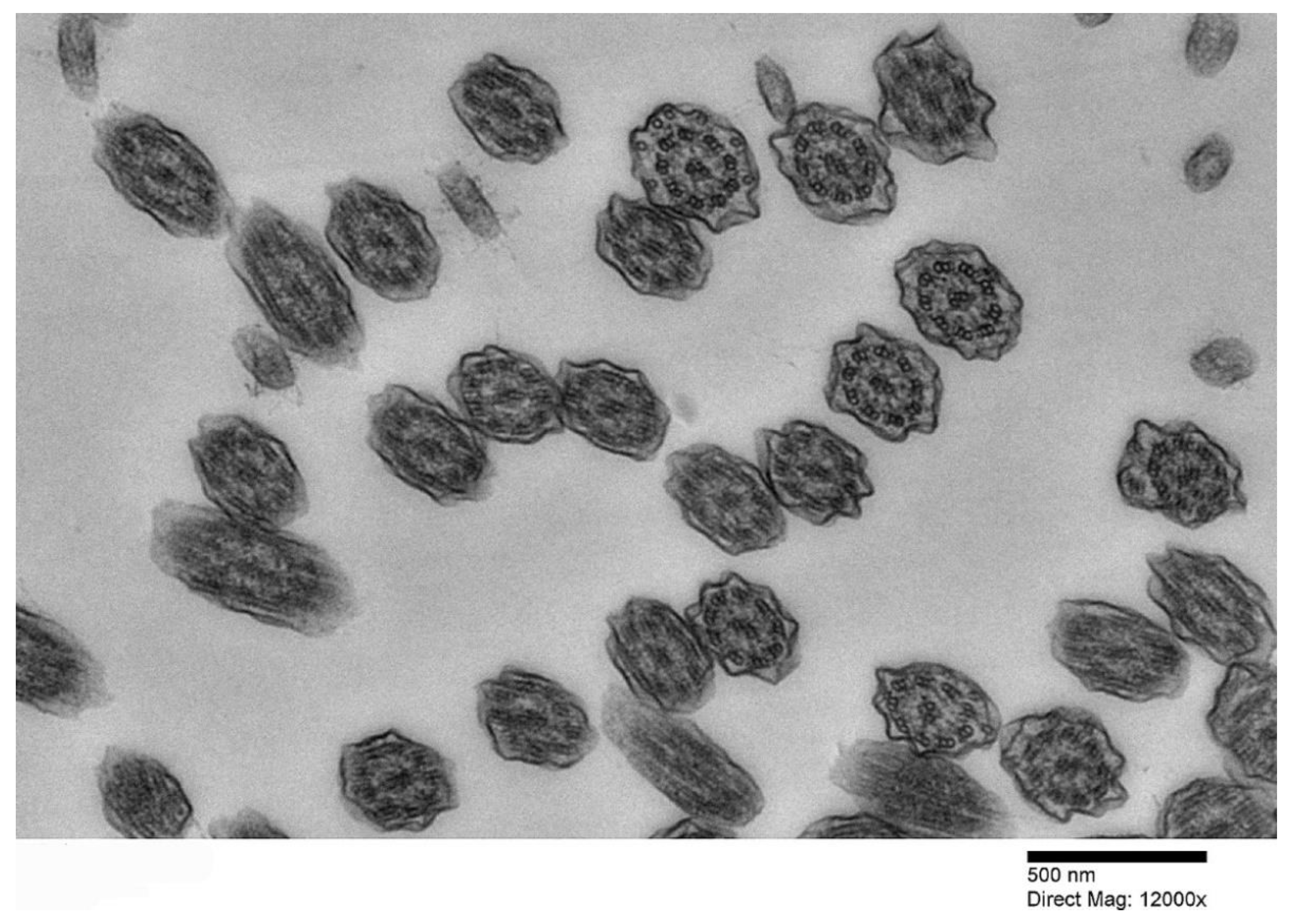
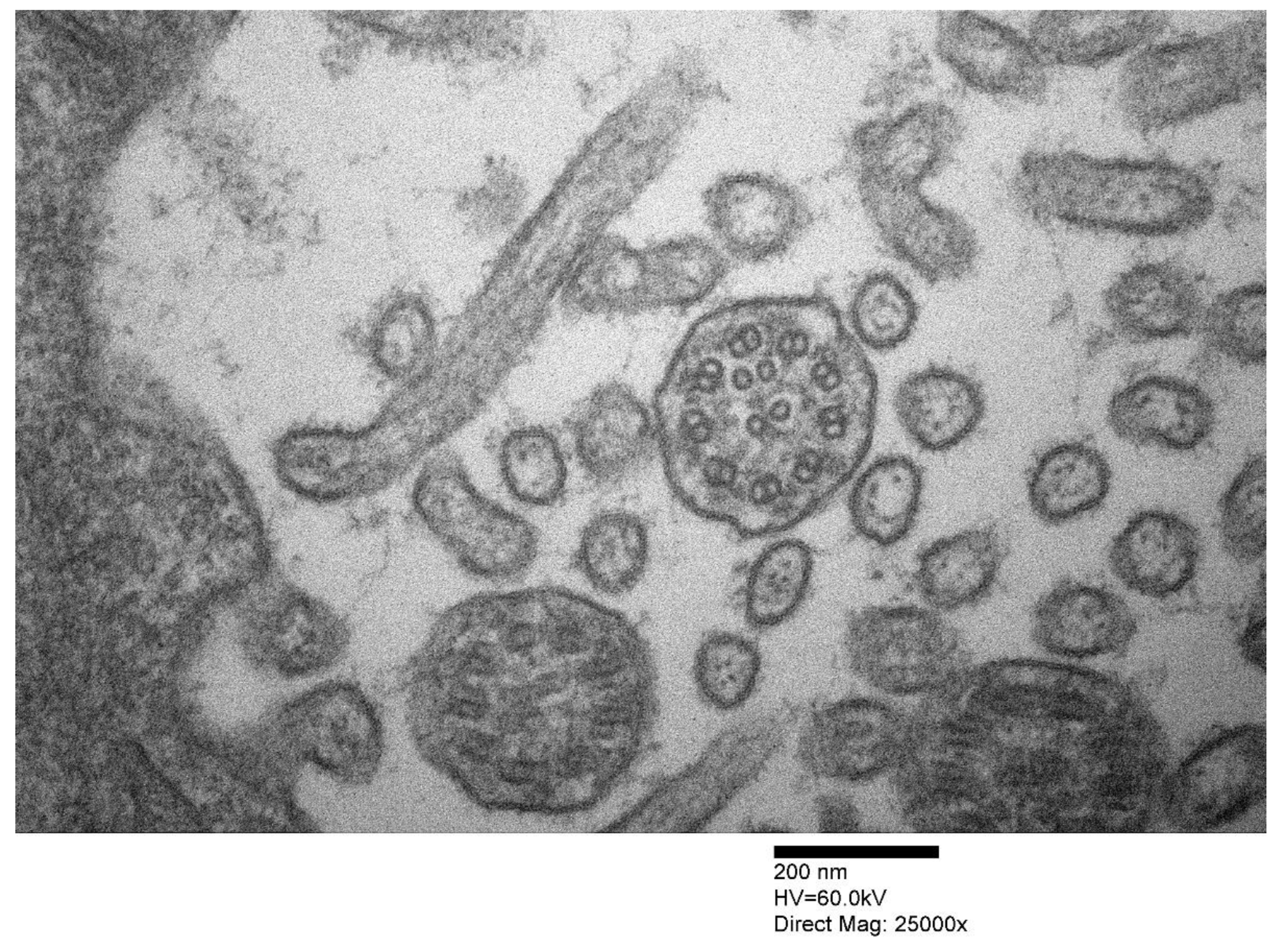
2. PCD under the Lens
3. Vitamin D
4. Vitamin D Role in PCD
5. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bhatt, R.; Hogg, C. Primary ciliary dyskinesia: A major player in a bigger game. Breathe 2020, 16, 200047. [Google Scholar] [CrossRef]
- Legendre, M.; Zaragosi, L.-E.; Mitchison, H.M. Motile cilia and airway disease. Semin. Cell Dev. Biol. 2021, 110, 19–33. [Google Scholar] [CrossRef] [PubMed]
- Lee, L.; Ostrowski, L.E. Motile cilia genetics and cell biology: Big results from little mice. Cell. Mol. Life Sci. 2021, 78, 769–797. [Google Scholar] [CrossRef] [PubMed]
- Sironen, A.; Shoemark, A.; Patel, M.; Loebinger, M.R.; Mitchison, H.M. Sperm defects in primary ciliary dyskinesia and related causes of male infertility. Cell. Mol. Life Sci. 2019, 77, 2029–2048. [Google Scholar] [CrossRef] [PubMed]
- Tsang, K.W.; Tipoe, G.L.; Mak, J.C.; Sun, J.; Wong, M.; Leung, R.; Tan, K.C.; MedStat, C.K.M.; Ho, J.C.; Ho, P.L.; et al. Ciliary central microtubular orientation is of no clinical significance in bronchiectasis. Respir. Med. 2005, 99, 290–297. [Google Scholar] [CrossRef][Green Version]
- Tsang, K.W.; Zheng, L.; Tipoe, G. Ciliary assessment in bronchiectasis. Respirol. 2000, 5, 91–98. [Google Scholar] [CrossRef]
- Marthin, J.K.; Petersen, N.; Skovgaard, L.T.; Nielsen, K.G. Lung function in patients with primary ciliary dyskinesia: A cross-sectional and 3-decade longitudinal study. Am. J. Respir. Crit. Care Med. 2010, 181, 1262–1268. [Google Scholar] [CrossRef]
- Wallmeier, J.; Nielsen, K.G.; Kuehni, C.E.; Lucas, J.S.; Leigh, M.W.; Zariwala, M.A.; Omran, H. Motile ciliopathies. Nat. Rev. Dis. Prim. 2020, 6, 77. [Google Scholar] [CrossRef]
- Sergi, C.M. Pathology of Childhood and Adolescence; Springer Nature: Berlin, Germany, 2020. [Google Scholar]
- Cave, D.; Ross, D.B.; Bahitham, W.; Chan, A.; Sergi, C.; Adatia, I. Mitochondrial DNA depletion syndrome-an unusual reason for interstage attrition after the modified stage 1 norwood operation. Congenit. Heart Dis. 2011, 8, E20–E23. [Google Scholar] [CrossRef]
- Postema, M.C.; Carrion-Castillo, A.; Fisher, S.E.; Vingerhoets, G.; Francks, C. The genetics of situs inversus without primary ciliary dyskinesia. Sci. Rep. 2020, 10, 3677. [Google Scholar] [CrossRef]
- Stern, B.M.; Sharma, G. Ciliary Dysfunction. In StatPearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, January 2021. [Google Scholar]
- Leigh, M.W.; Horani, A.; Kinghorn, B.; O’Connor, M.G.; Zariwala, M.A.; Knowles, M.R. Primary ciliary dyskinesia (PCD): A genetic disorder of motile cilia. Transl. Sci. Rare Dis. 2019, 4, 51–75. [Google Scholar] [CrossRef]
- Chen, X.; Deng, S.; Xia, H.; Yuan, L.; Xu, H.; Tang, S.; Deng, H. Identification of a CCDC114 variant in a Han-Chinese patient with situs inversus. Exp. Ther. Med. 2020, 20, 3336–3342. [Google Scholar] [CrossRef] [PubMed]
- Guo, Z.; Chen, W.; Wang, L.; Qian, L. Clinical and genetic spectrum of children with primary ciliary dyskinesia in China. J. Pediatr. 2020, 225, 157–165.e5. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; He, Y.; Cai, G.; Xiao, F.; Yang, J.; Li, Q.; Chen, X. CCDC114 is mutated in patient with a complex phenotype combining primary ciliary dyskinesia, sensorineural deafness, and renal disease. J. Hum. Genet. 2019, 64, 39–48. [Google Scholar] [CrossRef]
- Fedick, A.M.; Jalas, C.; Treff, N.R.; Knowles, M.R.; Zariwala, M.A. Carrier frequencies of eleven mutations in eight genes associated with primary ciliary dyskinesia in the Ashkenazi Jewish population. Mol. Genet. Genom. Med. 2014, 3, 137–142. [Google Scholar] [CrossRef]
- Knowles, M.R.; Leigh, M.W.; Ostrowski, L.E.; Huang, L.; Carson, J.L.; Hazucha, M.J.; Yin, W.; Berg, J.S.; Davis, S.D.; Dell, S.D.; et al. Exome sequencing identifies mutations in CCDC114 as a cause of primary ciliary dyskinesia. Am. J. Hum. Genet. 2013, 92, 99–106. [Google Scholar] [CrossRef] [PubMed]
- Noone, P.G.; Leigh, M.W.; Sannuti, A.; Minnix, S.L.; Carson, J.L.; Hazucha, M.; Zariwala, M.A.; Knowles, M.R. Primary ciliary dyskinesia: Diagnostic and phenotypic features. Am. J. Respir. Crit. Care Med. 2004, 169, 459–467. [Google Scholar] [CrossRef]
- Rayner, C.F.; Rutman, A.; Dewar, A.; Cole, P.J.; Wilson, R. Ciliary disorientation in patients with chronic upper respiratory tract inflammation. Am. J. Respir. Crit. Care Med. 1995, 151, 800–804. [Google Scholar] [CrossRef] [PubMed]
- Rayner, C.F.; Rutman, A.; Dewar, A.; Greenstone, M.A.; Cole, P.J.; Wilson, R. Ciliary disorientation alone as a cause of primary ciliary dyskinesia syndrome. Am. J. Respir. Crit. Care Med. 1996, 153, 1123–1129. [Google Scholar] [CrossRef]
- Rutman, A.; Cullinan, P.; Woodhead, M.; Cole, P.J.; Wilson, R. Ciliary disorientation: A possible variant of primary ciliary dyskinesia. Thorax 1993, 48, 770–771. [Google Scholar] [CrossRef][Green Version]
- Rutland, J.; De Iongh, R.U. Random ciliary orientation. N. Engl. J. Med. 1990, 323, 1681–1684. [Google Scholar] [CrossRef]
- Sergi, C.M. Transmission Electron Microscopy of Pediatric Small Round Blue Cell Tumors Microscopy Science: Last Approaches on Educational Programs and Applied Research; Formatex Research Center: Badajoz, Spain, 2018. [Google Scholar]
- Knowles, M.R.; Daniels, L.A.; Davis, S.D.; Zariwala, M.A.; Leigh, M.W. Primary ciliary dyskinesia. recent advances in diagnostics, genetics, and characterization of clinical disease. Am. J. Respir. Crit. Care Med. 2013, 188, 913–922. [Google Scholar] [CrossRef]
- Bricmont, N.; Benchimol, L.; Poirrier, A.-L.; Grignet, C.; Seaton, C.; Chilvers, M.A.; Seghaye, M.-C.; Louis, R.; Lefebvre, P.; Kempeneers, C. Nasal brushing sampling and processing using digital high speed ciliary videomicroscopy—Adaptation for the COVID-19 pandemic. J. Vis. Exp. 2020, 10, e61949. [Google Scholar] [CrossRef] [PubMed]
- DeLuca, H.F. History of the discovery of vitamin D and its active metabolites. BoneKEy Rep. 2014, 3, 479. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Shen, F.; Petryk, A.; Tang, J.; Chen, X.; Sergi, C. “English Disease”: Historical notes on rickets, the bone–lung link and child neglect issues. Nutrients 2016, 8, 722. [Google Scholar] [CrossRef] [PubMed]
- Sergi, C.; Linderkamp, O. Pathological case of the month: Classic rickets in a setting of significant psychosocial deprivation. Arch Pediatr. Adolesc. Med. 2001, 155, 967–968. Available online: https://www.ncbi.nlm.nih.gov/pubmed/11483129 (accessed on 31 December 2020). [CrossRef]
- Pinzon, R.T.; Pradana, A.A.W. Vitamin D deficiency among patients with COVID-19: Case series and recent literature review. Trop. Med. Health 2020, 48, 102. [Google Scholar] [CrossRef] [PubMed]
- Wise, J. Covid-19: Evidence is lacking to support vitamin D’s role in treatment and prevention. BMJ 2020, 371, m4912. [Google Scholar] [CrossRef]
- Mariani, J.; Giménez, V.M.M.; Bergam, I.; Tajer, C.; Antonietti, L.; Inserra, F.; Ferder, L.; Manucha, W. Association between vitamin D deficiency and COVID-19 incidence, complications, and mortality in 46 countries: An ecological study. Health Secur. 2020. [Google Scholar] [CrossRef]
- Santaolalla, A.; Beckmann, K.; Kibaru, J.; Josephs, D.; Van Hemelrijck, M.; Irshad, S. Association between Vitamin D and novel SARS-CoV-2 respiratory dysfunction—A scoping review of current evidence and its implication for COVID-19 pandemic. Front. Physiol. 2020, 11, 564387. [Google Scholar] [CrossRef]
- Morris, H.A.; Turner, A.G.; Anderson, P.H. Vitamin-D regulation of bone mineralization and remodelling during growth. Front Biosci. 2012, 4, 677–689. Available online: http://www.ncbi.nlm.nih.gov/pubmed/22201904 (accessed on 31 December 2020). [CrossRef]
- Saggese, G.; Vierucci, F.; Boot, A.M.; Czech-Kowalska, J.; Weber, G.; Camargo, C.A.; Mallet, E.; Fanos, M.; Shaw, N.J.; Holick, M.F. Vitamin D in childhood and adolescence: An expert position statement. Eur. J. Nucl. Med. Mol. Imaging 2015, 174, 565–576. [Google Scholar] [CrossRef]
- Vojinovic, J.; Cimaz, R. Vitamin D—Update for the pediatric rheumatologists. Pediatr. Rheumatol. 2015, 13, 18. [Google Scholar] [CrossRef]
- Kalra, S.; Aggarwal, S. Vitamin D deficiency: Diagnosis and patient centred management. J. Pak. Med. Assoc. 2015, 65, 569–573. Available online: http://www.ncbi.nlm.nih.gov/pubmed/26028397 (accessed on 31 December 2020). [PubMed]
- Kitanaka, S. Vitamin D dependency and its treatment. Clin. Calcium 2016, 26, 277–283. [Google Scholar]
- Biver, E.; Calmy, A.; Aubry-Rozier, B.; Birkhäuser, M.; Bischoff-Ferrari, H.A.; Ferrari, S.; Frey, D.; Kressig, R.W.; Lamy, O.; Lippuner, K.; et al. Diagnosis, prevention, and treatment of bone fragility in people living with HIV: A position statement from the swiss association against osteoporosis. Osteoporos. Int. 2019, 30, 1125–1135. [Google Scholar] [CrossRef] [PubMed]
- Ceballos, M.E.; Carvajal, C.; Jaramillo, J.; Dominguez, A.; González, G. Vitamin D and bone mineral density in HIV newly diagnosed therapy-naive patients without any secondary causes of osteoporosis. Calcif. Tissue Int. 2019, 104, 42–49. [Google Scholar] [CrossRef] [PubMed]
- Messina, O.D.; Wilman, M.V.; Neira, L.F.V. Nutrition, osteoarthritis and cartilage metabolism. Aging Clin. Exp. Res. 2019, 31, 807–813. [Google Scholar] [CrossRef]
- Atteritano, M.; Mirarchi, L.; Venanzi-Rullo, E.; Santoro, D.; Iaria, C.; Catalano, A.; Lasco, A.; Arcoraci, V.; Gullo, A.L.; Bitto, A.; et al. Vitamin D status and the relationship with bone fragility fractures in HIV-infected patients: A case control study. Int. J. Mol. Sci. 2018, 19, 119. [Google Scholar] [CrossRef]
- Cascio, A.; Colomba, C.; Di Carlo, P.; Serra, N.; Re, G.L.; Gambino, A.; Casto, A.L.; Guglielmi, G.; Veronese, N.; Lagalla, R.; et al. Low bone mineral density in HIV-positive young Italians and migrants. PLoS ONE 2020, 15, e0237984. [Google Scholar] [CrossRef]
- Kepron, C.; Pollanen, M.S. Rickets or abuse? A histologic comparison of rickets and child abuse-related fractures. Forensic Sci. Med. Pathol. 2015, 11, 78–87. [Google Scholar] [CrossRef]
- Chesney, R.W.; Mehls, O.; Anast, C.S.; Brown, E.; Hammerman, M.R.; Portale, A.; Fallon, M.D.; Mahan, J.; Alfrey, A.C. Renal osteodystrophy in children: The role of vitamin D, phosphorus, and parathyroid hormone. Am. J. Kidney Dis. 1986, 7, 275–284. [Google Scholar] [CrossRef]
- Pettifor, J.M.; Prentice, A. The role of vitamin D in paediatric bone health. Best Pract. Res. Clin. Endocrinol. Metab. 2011, 25, 573–584. [Google Scholar] [CrossRef] [PubMed]
- Prentice, A. Nutritional rickets around the world. J. Steroid Biochem. Mol. Biol. 2013, 136, 201–206. [Google Scholar] [CrossRef]
- Prentice, A. Vitamin D deficiency: A global perspective. Nutr. Rev. 2008, 66 (Suppl. 2), S153–S164. [Google Scholar] [CrossRef]
- Prentice, A.; Schoenmakers, I.; Laskey, M.A.; de Bono, S.; Ginty, F.; Goldberg, G.R. Nutrition and bone growth and development. Proc. Nutr. Soc. 2006, 65, 348–360. Available online: http://www.ncbi.nlm.nih.gov/pubmed/17181901 (accessed on 31 December 2020). [CrossRef] [PubMed]
- Chesney, R.W. Vitamin D and the magic mountain: The anti-infectious role of the vitamin. J. Pediatr. 2010, 156, 698–703. [Google Scholar] [CrossRef] [PubMed]
- Beck-Nielsen, S.S. Rickets in Denmark. Dan. Med. J. 2012, 59, B4384. Available online: http://www.ncbi.nlm.nih.gov/pubmed/22293055 (accessed on 31 December 2020). [PubMed]
- Alles, M.S.; Eussen, S.R.; Van Der Beek, E.M. Nutritional challenges and opportunities during the weaning period and in young childhood. Ann. Nutr. Metab. 2014, 64, 284–293. [Google Scholar] [CrossRef] [PubMed]
- Ameen, S.; Staub, L.; Ulrich, S.; Vock, P.; Ballmer, F.; Anderson, S.E. Harris lines of the tibia across centuries: A comparison of two populations, medieval and contemporary in Central Europe. Skelet. Radiol. 2004, 34, 279–284. [Google Scholar] [CrossRef] [PubMed]
- Assiri, A.; Saeed, A.; Sarkhy, A.A.; Mouzan, M.I.E.; Matary, W.E. Celiac disease presenting as rickets in Saudi children. Ann. Saudi Med. 2013, 33, 49–51. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.F. High Prevalence of vitamin D inadequacy and implications for health. Mayo Clin. Proc. 2006, 81, 353–373. [Google Scholar] [CrossRef]
- Kaganov, B.; Caroli, M.; Mazur, A.; Singhal, A.; Vania, A. Suboptimal micronutrient intake among children in Europe. Nutrients 2015, 7, 3524–3535. [Google Scholar] [CrossRef] [PubMed]
- Pettifor, J.M. Vitamin D&/or calcium deficiency rickets in infants & children: A global perspective. Indian J. Med. Res. 2008, 127, 245–249. Available online: http://www.ncbi.nlm.nih.gov/pubmed/18497438 (accessed on 31 December 2020). [PubMed]
- Vahlquist, B. Two-century perspective of some major nutritional deficiency diseases in childhood. Acta Paediatr. Scand. 1975, 64, 161–171. Available online: http://www.ncbi.nlm.nih.gov/pubmed/805508 (accessed on 31 December 2020). [PubMed]
- Wandel, M. Nutrition-related diseases and dietary change among third world immigrants in northern Europe. Nutr. Health 1993, 9, 117–133. [Google Scholar] [CrossRef]
- Alpert, P.T.; Shaikh, U. The effects of vitamin D deficiency and insufficiency on the endocrine and paracrine systems. Biol. Res. Nurs. 2007, 9, 117–129. [Google Scholar] [CrossRef]
- Cruz, J.A.A. Nutrition and osteoporosis: Facts and uncertainties about calcium and vitamin D recommendations. Forum Nutr. 2003, 56, 178–181. Available online: http://www.ncbi.nlm.nih.gov/pubmed/15806851 (accessed on 31 December 2020).
- Calvo, M.S.; Uribarri, J. Public health impact of dietary phosphorus excess on bone and cardiovascular health in the general population. Am. J. Clin. Nutr. 2013, 98, 6–15. [Google Scholar] [CrossRef]
- Cashman, K.D. Vitamin D in childhood and adolescence. Postgrad. Med. J. 2007, 83, 230–235. [Google Scholar] [CrossRef]
- Sundaram, M.E.; Coleman, L.A. Vitamin D and influenza. Adv. Nutr. 2012, 3, 517–525. [Google Scholar] [CrossRef] [PubMed]
- DeLuca, H.F. Vitamin D: Historical Overview. Vitam. Horm. 2016, 100, 1–20. [Google Scholar] [CrossRef]
- Ogunkolade, B.W.; Boucher, B.J.; Fairclough, P.D.; Hitman, G.A.; Dorudi, S.; Jenkins, P.J.; Bustin, S.A. Expression of 25-hydroxyvitamin D-1-α-hydroxylase mRNA in individuals with colorectal cancer. Lancet 2002, 359, 1831–1832. [Google Scholar] [CrossRef]
- Bah, S.Y.; Dickinson, P.; Forster, T.; Kampmann, B.; Ghazal, P. Immune oxysterols: Role in mycobacterial infection and inflammation. J. Steroid Biochem. Mol. Biol. 2017, 169, 152–163. [Google Scholar] [CrossRef]
- Zittermann, A.; Pilz, S.; Hoffmann, H.; März, W. Vitamin D and airway infections: A European perspective. Eur. J. Med. Res. 2016, 21, 14. [Google Scholar] [CrossRef]
- Gleeson, M. Immunological aspects of sport nutrition. Immunol. Cell Biol. 2015, 94, 117–123. [Google Scholar] [CrossRef]
- Neme, A.; Nurminen, V.; Seuter, S.; Carlberg, C. The vitamin D-dependent transcriptome of human monocytes. J. Steroid Biochem. Mol. Biol. 2016, 164, 180–187. [Google Scholar] [CrossRef]
- Chen, S.; Sims, G.P.; Chen, X.X.; Gu, Y.Y.; Chen, S.; Lipsky, P.E. Modulatory effects of 1,25-dihydroxyvitamin D3 on human B cell differentiation. J. Immunol. 2007, 179, 1634–1647. [Google Scholar] [CrossRef]
- Manolagas, S.C.; Provvedini, D.M.; Tsoukas, C.D. Interactions of 1,25-dihydroxyvitamin D3 and the immune system. Mol. Cell. Endocrinol. 1985, 43, 113–122. [Google Scholar] [CrossRef]
- Haavaldsen, R.; Egnund, K.M.; Nicolaysen, R. Studies in calcium metabolism in rats. II. The interaction of vitamin D and the endogenous factor. Acta Physiol. Scand. 1956, 36, 108–113. [Google Scholar] [CrossRef] [PubMed]
- Haavaldsen, R.; Nicolaysen, R. Studies in calcium metabolism in rats. I. A long term study in rats given an optimal diet with and without vitamin, D. Acta Physiol. Scand. 1956, 36, 102–107. [Google Scholar] [CrossRef]
- Nicolaysen, R. The influence of vitamin D on the absorption of calcium from the intestine of rats: Experiments with isolated loops. Acta Physiol. Scand. 1951, 22, 260–266. [Google Scholar] [CrossRef]
- Sasaki, K.; Shiba, K.; Nakamura, A.; Kawano, N.; Satouh, Y.; Yamaguchi, H.; Morikawa, M.; Shibata, D.; Yanase, R.; Jokura, K.; et al. Calaxin is required for cilia-driven determination of vertebrate laterality. Commun. Biol. 2019, 2, 226. [Google Scholar] [CrossRef] [PubMed]
- Auguste, Y.; Delague, V.; Desvignes, J.-P.; Longepied, G.; Gnisci, A.; Besnier, P.; Levy, N.; Beroud, C.; Megarbane, A.; Metzler-Guillemain, C.; et al. Loss of calmodulin- and radial-spoke-associated complex protein CFAP251 leads to immotile spermatozoa lacking mitochondria and infertility in men. Am. J. Hum. Genet. 2018, 103, 413–420. [Google Scholar] [CrossRef] [PubMed]
- Van Hemelrijck, M.; Shanmugalingam, T.; Bosco, C.; Wulaningsih, W.; Rohrmann, S. The association between circulating IGF1, IGFBP3, and calcium: Results from NHANES III. Endocr. Connect. 2015, 4, 187–195. [Google Scholar] [CrossRef] [PubMed]
- Upadhyaya, A.; Baraban, M.; Wong, J.; Matsudaira, P.; Van Oudenaarden, A.; Mahadevan, L. Power-limited contraction dynamics of vorticella convallaria: An ultrafast biological spring. Biophys. J. 2008, 94, 265–272. [Google Scholar] [CrossRef]
- Plattner, H.; Hentschel, J. Sub-second cellular dynamics: Time-resolved electron microscopy and functional correlation. Adv. Virus Res. 2006, 255, 133–176. [Google Scholar] [CrossRef]
- Gordon, R.E.; Williams, K.B.; Puszkin, S. Immune localization of calmodulin in the ciliated cells of hamster tracheal epithelium. J. Cell Biol. 1982, 95, 57–63. [Google Scholar] [CrossRef]
- Schrumpf, J.A.; Ninaber, D.K.; Van Der Does, A.M.; Hiemstra, P.S. TGF-β1 impairs vitamin D-induced and constitutive airway epithelial host defense mechanisms. J. Innate Immun. 2019, 12, 74–89. [Google Scholar] [CrossRef]
- Zdrenghea, M.T.; Makrinioti, H.; Bagacean, C.; Bush, A.; Johnston, S.L.; Stanciu, L.A. Vitamin D modulation of innate immune responses to respiratory viral infections. Rev. Med. Virol. 2017, 27, e1909. [Google Scholar] [CrossRef]
- Sun, J. Dietary vitamin D, vitamin D receptor, and microbiome. Curr. Opin. Clin. Nutr. Metab. Care 2018, 21, 471–474. [Google Scholar] [CrossRef]
- Hall, S.C.; Agrawal, D.K. Vitamin D and bronchial asthma: An overview of data from the past 5 years. Clin. Ther. 2017, 39, 917–929. [Google Scholar] [CrossRef]
- Fawaz, L.; Mrad, M.F.; Kazan, J.M.; Sayegh, S.; Akika, R.; Khoury, S.J. Comparative effect of 25(OH)D3 and 1,25(OH)2D3 on Th17 cell differentiation. Clin. Immunol. 2016, 166–167, 59–71. [Google Scholar] [CrossRef] [PubMed]
- Schedel, M.; Jia, Y.; Michel, S.; Takeda, K.; Domenico, J.; Joetham, A.; Ning, F.; Strand, M.; Han, J.; Wang, M.; et al. 1,25D3 prevents CD8+Tc2 skewing and asthma development through VDR binding changes to the Cyp11a1 promoter. Nat. Commun. 2016, 7, 10213. [Google Scholar] [CrossRef] [PubMed]
- Urashima, M.; Segawa, T.; Okazaki, M.; Kurihara, M.; Wada, Y.; Ida, H. Randomized trial of vitamin D supplementation to prevent seasonal influenza A in schoolchildren. Am. J. Clin. Nutr. 2010, 91, 1255–1260. [Google Scholar] [CrossRef] [PubMed]
- Camargo, C.A.; Ganmaa, D.; Frazier, A.L.; Kirchberg, F.F.; Stuart, J.J.; Kleinman, K.; Sumberzul, N.; Rich-Edwards, J.W. Randomized trial of vitamin D Supplementation and risk of acute respiratory infection in mongolia. Pediatrics 2012, 130, e561–e567. [Google Scholar] [CrossRef] [PubMed]
- Xiao, L.; Xing, C.; Yang, Z.; Xu, S.; Wang, M.; Du, H.; Liu, K.; Huang, Z. Vitamin D supplementation for the prevention of childhood acute respiratory infections: A systematic review of randomised controlled trials. Br. J. Nutr. 2015, 114, 1026–1034. [Google Scholar] [CrossRef]
- Sergi, C.; Shen, F.; Bouma, G. Intraepithelial lymphocytes, scores, mimickers and challenges in diagnosing gluten-sensitive enteropathy (celiac disease). World J. Gastroenterol. 2017, 23, 573–589. [Google Scholar] [CrossRef] [PubMed]
- Pifferi, M.; Bush, A.; Pioggia, G.; Caramella, D.; Tartarisco, G.; Di Cicco, M.; Zangani, M.; Chinellato, I.; Maggi, F.; Tezza, G.; et al. Evaluation of pulmonary disease using static lung volumes in primary ciliary dyskinesia. Thorax 2012, 67, 993–999. [Google Scholar] [CrossRef][Green Version]
- Valerio, G.; Giallauria, F.; Montella, S.; Vaino, N.; Vigorito, C.; Mirra, V.; Santamaria, F. Cardiopulmonary assessment in primary ciliary dyskinesia. Eur. J. Clin. Investig. 2011, 42, 617–622. [Google Scholar] [CrossRef]
- Marino, L.; Harris, A.; Johnstone, C.; Friend, A.; Newell, C.; Miles, E.; Lucas, J.; Calder, P.; Walker, W. Characterising the nutritional status of children with primary ciliary dyskinesia. Clin. Nutr. 2019, 38, 2127–2135. [Google Scholar] [CrossRef] [PubMed]
- Kyle, U.G.; Bosaeus, I.; De Lorenzo, A.D.; Deurenberg, P.; Elia, M.; Gómez, J.M.; Heitmann, B.L.; Kent-Smith, L.; Melchior, J.-C.; Pirlich, M.; et al. Bioelectrical impedance analysis—Part II: Utilization in clinical practice. Clin. Nutr. 2004, 23, 1430–1453. [Google Scholar] [CrossRef]
- Kyle, U.G.; Bosaeus, I.; De Lorenzo, A.D.; Deurenberg, P.; Elia, M.; Gomez, J.M.; Heitmann, B.L.; Kent-Smith, L.; Melchior, J.C.; Pirlich, M.; et al. Bioelectrical impedance analysis? Part I: Review of principles and methods. Clin. Nutr. 2004, 23, 1226–1243. [Google Scholar] [CrossRef] [PubMed]
- Cohen-Cymberknoh, M.; Simanovsky, N.; Hiller, N.; Hillel, A.G.; Shoseyov, D.; Kerem, E. Differences in disease expression between primary ciliary dyskinesia and cystic fibrosis with and without pancreatic insufficiency. Chest 2014, 145, 738–744. [Google Scholar] [CrossRef]
- Mirra, V.; Caffarelli, C.; Maglione, M.; Valentino, R.; Perruolo, G.; Mazzarella, C.; Di Micco, L.L.; Montella, S.; Santamaria, F. Hypovitaminosis D: A novel finding in primary ciliary dyskinesia. Ital. J. Pediatr. 2015, 41, 14–16. [Google Scholar] [CrossRef]
- Khan, A.; Sergi, C. SAMHD1 as the potential link between SARS-CoV-2 infection and neurological complications. Front. Neurol. 2020, 11. [Google Scholar] [CrossRef]
- Burnett, M.L.; Sergi, C.M. Face masks are beneficial regardless of the level of infection in the fight against COVID-19. Disaster Med. Public Health Prep. 2020, 14, e47–e50. [Google Scholar] [CrossRef]
- Sergi, C.M.; Chiu, B. Targeting NLRP3 inflammasome in an animal model for coronavirus disease 2019 (COVID-19) caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). J. Med. Virol. 2021, 93, 669–670. [Google Scholar] [CrossRef]
- Dwyer, J.T.; Dietz, W.H.; Hass, G.; Suskind, R. Risk of nutritional rickets among vegetarian children. Arch. Pediatr. Adolesc. Med. 1979, 133, 134–140. [Google Scholar] [CrossRef] [PubMed]
- Dagnelie, P.C.; Vergote, F.J.; Van Staveren, W.A.; Berg, H.V.D.; Dingjan, P.G.; Hautvast, J.G. High prevalence of rickets in infants on macrobiotic diets. Am. J. Clin. Nutr. 1990, 51, 202–208. [Google Scholar] [CrossRef] [PubMed]
- Green, T.J.; Li, W.; Barr, S.I.; Jahani, M.; Chapman, G.E.; Green, T. Vitamin D supplementation is associated with higher serum 25OHD in Asian and white infants living in Vancouver, Canada. Matern. Child. Nutr. 2012, 11, 253–259. [Google Scholar] [CrossRef] [PubMed]
- Pettifor, J.M. Nutritional rickets: Deficiency of vitamin D, calcium, or both? Am. J. Clin. Nutr. 2004, 80 (Suppl. 6), 1725S–1729S. [Google Scholar] [CrossRef] [PubMed]
- Koch, H.-C.; Burmeister, W. Vitamin D-status bei afrikanischen und asiatischen diplomatenkindern und -jugendlichen in Deutschland. Klin. Pädiatrie 1993, 205, 416–420. [Google Scholar] [CrossRef] [PubMed]
- Boyle, I.T. Bones for the future. Acta Paediatr. 1991, 80, 58–65. [Google Scholar] [CrossRef]
- Heald, F.P.; Rosebrough, R.H.; Jacobson, M.S. Nutrition and the adolescent: An update. J. Adolesc. Health Care 1980, 1, 142–151. [Google Scholar] [CrossRef]
- Goel, K.; Logan, R.; Arneil, G.; Sweet, E.; Warren, J.; Shanks, R. Florid and subclinical rickets among immigrant children in glasgow. Lancet 1976, 307, 1141–1145. [Google Scholar] [CrossRef]
- Ford, J.A.; Davidson, D.C.; McIntosh, W.B.; Fyfe, W.M.; Dunnigan, M.G. Neonatal rickets in Asian immigrant population. Br. Med. J. 1973, 3, 211–212. [Google Scholar] [CrossRef]
- Holmes, A.M.; Enoch, B.A.; Taylor, J.L.; Jones, M.E. Occult rickets and osteomalacia amongst the Asian immigrant population. QJM Int. J. Med. 1973, 42, 125–149. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sergi, C.M. Vitamin D and Primary Ciliary Dyskinesia: A Topic to Be Further Explored. Appl. Sci. 2021, 11, 3818. https://doi.org/10.3390/app11093818
Sergi CM. Vitamin D and Primary Ciliary Dyskinesia: A Topic to Be Further Explored. Applied Sciences. 2021; 11(9):3818. https://doi.org/10.3390/app11093818
Chicago/Turabian StyleSergi, Consolato M. 2021. "Vitamin D and Primary Ciliary Dyskinesia: A Topic to Be Further Explored" Applied Sciences 11, no. 9: 3818. https://doi.org/10.3390/app11093818
APA StyleSergi, C. M. (2021). Vitamin D and Primary Ciliary Dyskinesia: A Topic to Be Further Explored. Applied Sciences, 11(9), 3818. https://doi.org/10.3390/app11093818





