Advances in Tissue Engineering for Disc Repair
Abstract
1. Introduction
2. Biological Approaches
2.1. Molecular Therapies
2.1.1. Growth Factors
2.1.2. Gene Therapy
2.1.3. Summary
2.2. Cell-Based Therapies
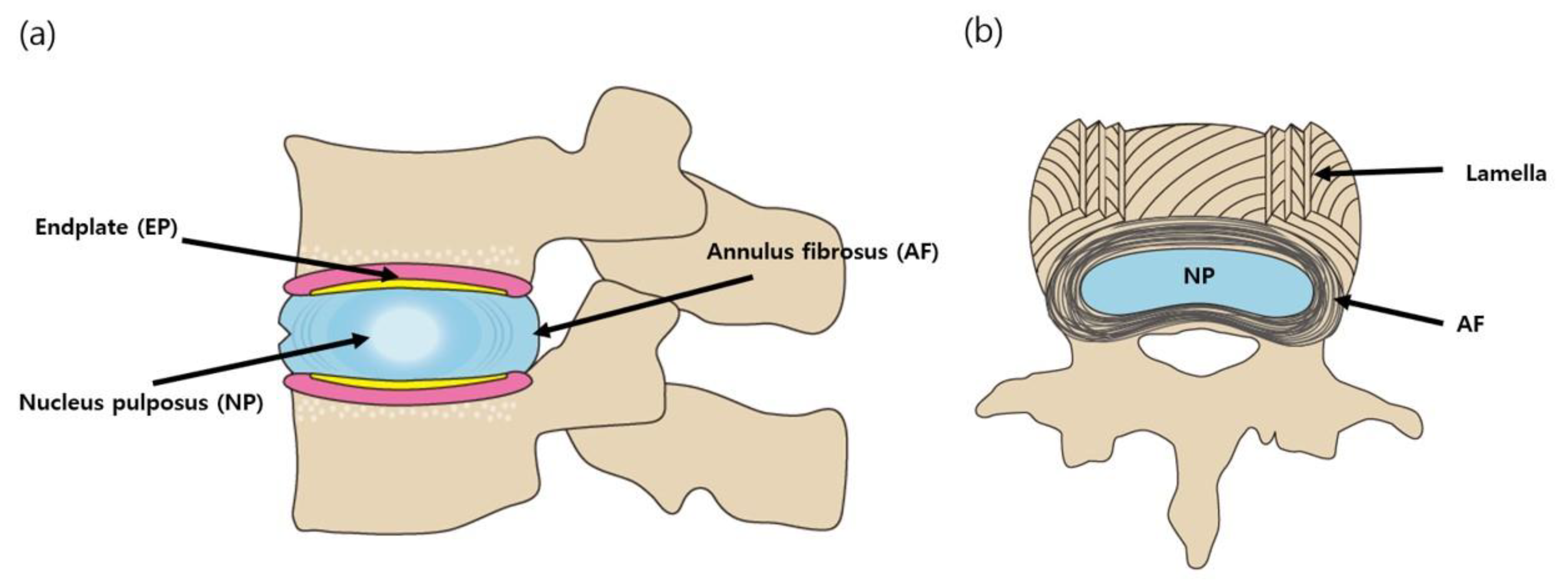
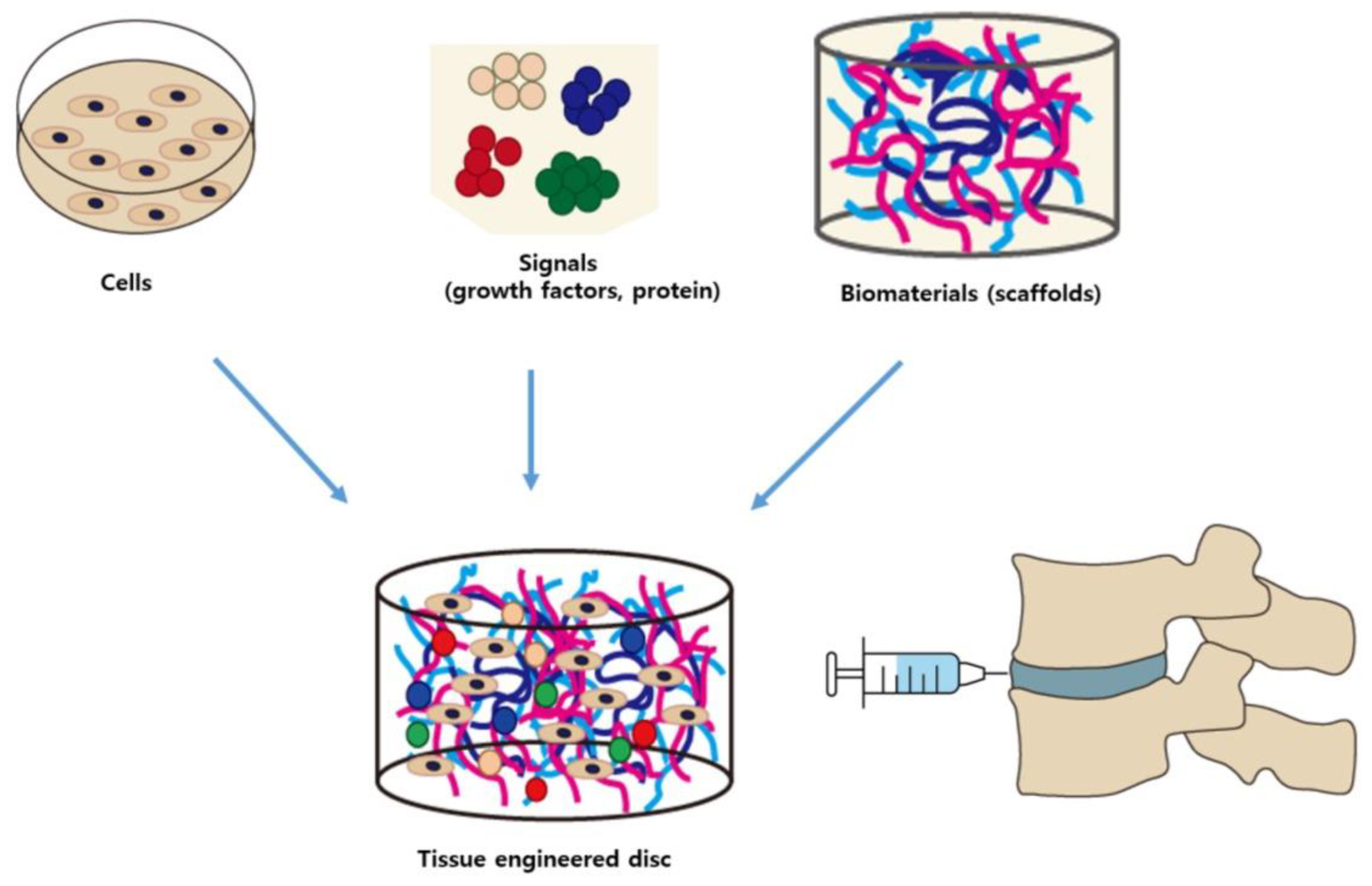
3. Tissue Engineering for IVD Regeneration
3.1. Biomaterials
3.2. Tissue Engineering for AF and NP Restoration and Maintenance
3.2.1. AF Regeneration and Tissue Engineering
3.2.2. NP Regeneration and Tissue Engineering
3.2.3. NP-AF Regeneration and Tissue Engineering
3.2.4. Summary
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Coric, D.; Pettine, K.; Sumich, A.; Boltes, M.O. Prospective study of disc repair with allogeneic chondrocytes Presented at the 2012 Joint Spine Section Meeting. J. Neurosurg. Spine 2013, 18, 85–95. [Google Scholar] [CrossRef]
- de Schepper, E.I.; Damen, J.; van Meurs, J.B.; Ginai, A.Z.; Popham, M.; Hofman, A.; Koes, B.W.; Bierma-Zeinstra, S.M. The association between lumbar disc degeneration and low back pain: The influence of age, gender, and individual radiographic features. Spine 2010, 35, 531–536. [Google Scholar] [CrossRef]
- Murray, C.J.; Lopez, A.D. Measuring the global burden of disease. N. Engl. J. Med. 2013, 369, 448–457. [Google Scholar] [CrossRef]
- Urban, J.P.; Roberts, S. Degeneration of the intervertebral disc. Arthritis Res. Ther. 2003, 5, 120–130. [Google Scholar] [CrossRef] [PubMed]
- Hoy, D.; Bain, C.; Williams, G.; March, L.; Brooks, P.; Blyth, F.; Woolf, A.; Vos, T.; Buchbinder, R. A systematic review of the global prevalence of low back pain. Arthritis Rheum. 2012, 64, 2028–2037. [Google Scholar] [CrossRef] [PubMed]
- Risbud, M.V.; Shapiro, I.M. Role of cytokines in intervertebral disc degeneration: Pain and disc content. Nat. Rev. Rheumatol. 2014, 10, 44–56. [Google Scholar] [CrossRef] [PubMed]
- Lotz, J.C.; Ulrich, J.A. Innervation, inflammation, and hypermobility may characterize pathologic disc degeneration: Review of animal model data. J. Bone Jt. Surg. 2006, 88 (Suppl. 2), 76–82. [Google Scholar] [CrossRef]
- Phillips, F.M.; Slosar, P.J.; Youssef, J.A.; Andersson, G.; Papatheofanis, F. Lumbar spine fusion for chronic low back pain due to degenerative disc disease: A systematic review. Spine 2013, 38, E409–E422. [Google Scholar] [CrossRef]
- Geisler, F.H.; McAfee, P.C.; Banco, R.J.; Blumenthal, S.L.; Guyer, R.D.; Holt, R.T.; Majd, M.E. Prospective, randomized, multicenter FDA IDE study of CHARITÉ artificial disc versus lumbar fusion: Effect at 5-year follow-up of prior surgery and prior discectomy on clinical outcomes following lumbar arthroplasty. SAS J. 2009, 3, 17–25. [Google Scholar] [CrossRef]
- Ghiselli, G.; Wang, J.C.; Bhatia, N.N.; Hsu, W.K.; Dawson, E.G. Adjacent segment degeneration in the lumbar spine. J. Bone Jt. Surg. 2004, 86, 1497–1503. [Google Scholar] [CrossRef] [PubMed]
- Zhao, R.; Liu, W.; Xia, T.; Yang, L. Disordered mechanical stress and tissue engineering therapies in intervertebral disc degeneration. Polymers 2019, 11, 1151. [Google Scholar] [CrossRef] [PubMed]
- Stergar, J.; Gradisnik, L.; Velnar, T.; Maver, U. Intervertebral disc tissue engineering: A brief review. Bosn. J. Basic Med. Sci. 2019, 19, 130–137. [Google Scholar] [CrossRef]
- Ju, D.G.; Kanim, L.E.; Bae, H.W. Intervertebral disc repair: Current concepts. Glob. Spine J. 2020, 10 (Suppl. 2), 130S–136S. [Google Scholar] [CrossRef] [PubMed]
- Wu, P.H.; Kim, H.S.; Jang, I.-T. Intervertebral disc diseases PART 2: A review of the current diagnostic and treatment strategies for intervertebral disc disease. Int. J. Mol. Sci. 2020, 21, 2135. [Google Scholar] [CrossRef]
- Griffith, J.F.; Wang, Y.-X.J.; Antonio, G.E.; Choi, K.C.; Yu, A.; Ahuja, A.T.; Leung, P.C. Modified Pfirrmann Grading System for lumbar intervertebral disc degeneration. Spine 2007, 32, E708–E712. [Google Scholar] [CrossRef]
- Dowdell, J.; Erwin, M.; Choma, T.; Vaccaro, A.; Iatridis, J.; Cho, S.K. intervertebral disk degeneration and repair. Neurosurgery 2017, 80, S46–S54. [Google Scholar] [CrossRef]
- Travascio, F.; Elmasry, S.; Asfour, S. Modeling the role of IGF-1 on extracellular matrix biosynthesis and cellularity in intervertebral disc. J. Biomech. 2014, 47, 2269–2276. [Google Scholar] [CrossRef]
- Feng, C.; Liu, H.; Yang, Y.; Huang, B.; Zhou, Y. Growth and differentiation factor-5 contributes to the structural and functional maintenance of the intervertebral disc. Cell. Physiol. Biochem. 2015, 35, 1–16. [Google Scholar] [CrossRef]
- Cho, H.; Lee, S.; Park, S.H.; Huang, J.; Hasty, K.A.; Kim, S.J. Synergistic effect of combined growth factors in porcine intervertebral disc degeneration. Connect. Tissue Res. 2013, 54, 181–186. [Google Scholar] [CrossRef] [PubMed]
- O’Connell, G.D.; Newman, I.B.; Carapezza, M.A. Effect of long-term osmotic loading culture on matrix synthesis from intervertebral disc cells. BioRes. Open Access 2014, 3, 242–249. [Google Scholar] [CrossRef]
- Sampat, S.R.; O’Connell, G.D.; Fong, J.V.; Alegre-Aguarón, E.; Ateshian, G.A.; Hung, C.T. Growth factor priming of synovium-derived stem cells for cartilage tissue engineering. Tissue Eng. Part A 2011, 17, 2259–2265. [Google Scholar] [CrossRef]
- Presciutti, S.M.; Paglia, D.N.; Karukonda, T.; Soung do, Y.; Guzzo, R.; Drissi, H.; Moss, I.L. PDGF-BB inhibits intervertebral disc cell apoptosis in vitro. J. Orthop. Res. 2014, 32, 1181–1188. [Google Scholar] [CrossRef] [PubMed]
- Imai, Y.; Miyamoto, K.; An, H.S.; Thonar, E.J.-M.A.; Andersson, G.B.J.; Masuda, K. Recombinant human osteogenic protein-1 upregulates proteoglycan metabolism of human anulus fibrosus and nucleus pulposus cells. Spine 2007, 32, 1303–1309. [Google Scholar] [CrossRef] [PubMed]
- Huang, K.Y.; Yan, J.J.; Hsieh, C.C.; Chang, M.S.; Lin, R.M. The in vivo biological effects of intradiscal recombinant human bone morphogenetic protein-2 on the injured intervertebral disc: An animal experiment. Spine 2007, 32, 1174–1180. [Google Scholar] [CrossRef]
- Walsh, A.J.L.; Bradford, D.S.; Lotz, J.C. In vivo growth factor treatment of degenerated intervertebral discs. Spine 2004, 29, 156–163. [Google Scholar] [CrossRef] [PubMed]
- Liang, H.; Ma, S.-Y.; Feng, G.; Shen, F.H.; Li, X.J. Therapeutic effects of adenovirus-mediated growth and differentiation factor-5 in a mice disc degeneration model induced by annulus needle puncture. Spine J. 2010, 10, 32–41. [Google Scholar] [CrossRef]
- O’Connell, G.D.; Leach, J.K.; Klineberg, E.O. Tissue engineering a biological repair strategy for lumbar disc herniation. BioRes. Open Access 2015, 4, 431–445. [Google Scholar] [CrossRef]
- Yan, J.; Yang, S.; Sun, H.; Guo, D.; Wu, B.; Ji, F.; Zhou, D. Effects of releasing recombinant human growth and differentiation factor-5 from poly(lactic-co-glycolic acid) microspheres for repair of the rat degenerated intervertebral disc. J. Biomater. Appl. 2014, 29, 72–80. [Google Scholar] [CrossRef] [PubMed]
- Han, I.; Ropper, A.E.; Konya, D.; Kabatas, S.; Toktas, Z.; Aljuboori, Z.; Zeng, X.; Chi, J.H.; Zafonte, R.; Teng, Y.D. Biological approaches to treating intervertebral disk degeneration: Devising stem cell therapies. Cell Transplant. 2015, 24, 2197–2208. [Google Scholar] [CrossRef]
- Roh, E.; Darai, A.; Kyung, J.; Choi, H.; Kwon, S.; Bhujel, B.; Kim, K.; Han, I. Genetic therapy for intervertebral disc degeneration. Int. J. Mol. Sci. 2021, 22, 1579. [Google Scholar] [CrossRef]
- Risbud, M.V.; Schoepflin, Z.R.; Mwale, F.; Kandel, R.A.; Grad, S.; Iatridis, J.C.; Sakai, D.; Hoyland, J.A. Defining the phenotype of young healthy nucleus pulposus cells: Recommendations of the Spine Research Interest Group at the 2014 annual ORS meeting. J. Orthop. Res. 2015, 33, 283–293. [Google Scholar] [CrossRef]
- Sakai, D.; Andersson, G.B.J. Stem cell therapy for intervertebral disc regeneration: Obstacles and solutions. Nat. Rev. Rheumatol. 2015, 11, 243–256. [Google Scholar] [CrossRef]
- Sheyn, D.; Ben-David, S.; Tawackoli, W.; Zhou, Z.; Salehi, K.; Bez, M.; De Mel, S.; Chan, V.; Roth, J.; Avalos, P.; et al. Human iPSCs can be differentiated into notochordal cells that reduce intervertebral disc degeneration in a porcine model. Theranostics 2019, 9, 7506–7524. [Google Scholar] [CrossRef]
- Chen, S.; Emery, S.E.; Pei, M. Coculture of synovium-derived stem cells and nucleus pulposus cells in serum-free defined medium with supplementation of transforming growth factor-beta1: A potential application of tissue-specific stem cells in disc regeneration. Spine 2009, 34, 1272–1280. [Google Scholar] [CrossRef] [PubMed]
- Leckie, S.K.; Sowa, G.A.; Bechara, B.P.; Hartman, R.A.; Coelho, J.P.; Witt, W.T.; Dong, Q.D.; Bowman, B.W.; Bell, K.M.; Vo, N.V.; et al. Injection of human umbilical tissue–derived cells into the nucleus pulposus alters the course of intervertebral disc degeneration in vivo. Spine J. 2013, 13, 263–272. [Google Scholar] [CrossRef]
- Chang, E.-A.; Jin, S.-W.; Nam, M.-H.; Kim, S.-D. human induced pluripotent stem cells: Clinical significance and applications in neurologic diseases. J. Korean Neurosurg. Soc. 2019, 62, 493–501. [Google Scholar] [CrossRef] [PubMed]
- Yim, R.L.-H.; Lee, J.T.-Y.; Bow, C.H.; Meij, B.; Leung, V.; Cheung, K.M.; Vavken, P.; Samartzis, D. A Systematic review of the safety and efficacy of mesenchymal stem cells for disc degeneration: Insights and future directions for regenerative therapeutics. Stem Cells Dev. 2014, 23, 2553–2567. [Google Scholar] [CrossRef]
- Salzig, D.; Schmiermund, A.; Gebauer, E.; Fuchsbauer, H.-L.; Czermak, P. Influence of porcine intervertebral disc matrix on stem cell differentiation. J. Funct. Biomater. 2011, 2, 155–172. [Google Scholar] [CrossRef] [PubMed]
- Vadalà, G.; Sowa, G.; Hubert, M.; Gilbertson, L.G.; Denaro, V.; Kang, J.D. Mesenchymal stem cells injection in degenerated intervertebral disc: Cell leakage may induce osteophyte formation. J. Tissue Eng. Regen. Med. 2011, 6, 348–355. [Google Scholar] [CrossRef]
- Shi, P.; Chee, A.; Liu, W.; Chou, P.-H.; Zhu, J.; An, H.S. Therapeutic effects of cell therapy with neonatal human dermal fibroblasts and rabbit dermal fibroblasts on disc degeneration and inflammation. Spine J. 2019, 19, 171–181. [Google Scholar] [CrossRef] [PubMed]
- Teixeira, G.Q.; Pereira, C.L.; Ferreira, J.R.; Maia, A.F.; Gomez-Lazaro, M.; Barbosa, M.A.; Neidlinger-Wilke, C.; Goncalves, R.M. Immunomodulation of human mesenchymal stem/stromal cells in intervertebral disc degeneration: Insights from a proinflammatory/degenerative ex vivo model. Spine 2018, 43, e673–e682. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Wang, Y.; Deng, G.; Ma, J.; Huang, X.; Yu, J.; Xi, Y.; Ye, X. Transplantation of hypoxic-preconditioned bone mesenchymal stem cells retards intervertebral disc degeneration via enhancing implanted cell survival and migration in rats. Stem Cells Int. 2018, 2018, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Maidhof, R.; Rafiuddin, A.; Chowdhury, F.; Jacobsen, T.; Chahine, N.O. Timing of mesenchymal stem cell delivery impacts the fate and therapeutic potential in intervertebral disc repair. J. Orthop. Res. 2017, 35, 32–40. [Google Scholar] [CrossRef]
- Hang, D.; Li, F.; Che, W.; Wu, X.; Wan, Y.; Wang, J.; Zheng, Y. One-stage positron emission tomography and magnetic resonance imaging to assess mesenchymal stem cell survival in a canine model of intervertebral disc degeneration. Stem Cells Dev. 2017, 26, 1334–1343. [Google Scholar] [CrossRef] [PubMed]
- Steffen, F.; Smolders, L.A.; Roentgen, A.M.; Bertolo, A.; Stoyanov, J. Bone marrow-derived mesenchymal stem cells as autologous therapy in dogs with naturally occurring intervertebral disc disease: Feasibility, safety, and preliminary results. Tissue Eng. Part C Methods 2017, 23, 643–651. [Google Scholar] [CrossRef] [PubMed]
- Noriega, D.C.; Ardura, F.; Hernández-Ramajo, R.; Martín-Ferrero, M.; Sánchez-Lite, I.; Toribio, B.; Alberca, M.; García, V.; Moraleda, J.M.; Sánchez, A.; et al. Intervertebral disc repair by allogeneic mesenchymal bone marrow cells: A randomized controlled trial. Transplantation 2017, 101, 1945–1951. [Google Scholar] [CrossRef]
- Centeno, C.; Markle, J.; Dodson, E.; Stemper, I.; Williams, C.J.; Hyzy, M.; Ichim, T.; Freeman, M. Treatment of lumbar degenerative disc disease-associated radicular pain with culture-expanded autologous mesenchymal stem cells: A pilot study on safety and efficacy. J. Transl. Med. 2017, 15, 1–12. [Google Scholar] [CrossRef]
- Kumar, H.; Ha, D.-H.; Lee, E.-J.; Park, J.H.; Shim, J.H.; Ahn, T.-K.; Kim, K.-T.; Ropper, A.E.; Sohn, S.; Kim, C.-H.; et al. Safety and tolerability of intradiscal implantation of combined autologous adipose-derived mesenchymal stem cells and hyaluronic acid in patients with chronic discogenic low back pain: 1-year follow-up of a phase I study. Stem Cell Res. Ther. 2017, 8, 1–14. [Google Scholar] [CrossRef]
- Pettine, K.A.; Suzuki, R.K.; Sand, T.T.; Murphy, M.B. Autologous bone marrow concentrate intradiscal injection for the treatment of degenerative disc disease with three-year follow-up. Int. Orthop. 2017, 41, 2097–2103. [Google Scholar] [CrossRef]
- Tschugg, A.; Michnacs, F.; Strowitzki, M.; Meisel, H.J.; Thomé, C. A prospective multicenter phase I/II clinical trial to evaluate safety and efficacy of NOVOCART Disc plus autologous disc chondrocyte transplantation in the treatment of nucleotomized and degenerative lumbar disc to avoid secondary disease: Study protocol for a randomized controlled trial. Trials 2016, 17, 1–10. [Google Scholar] [CrossRef]
- Grunhagen, T.; Shirazi-Adl, A.; Fairbank, J.C.; Urban, J.P. Intervertebral disk nutrition: A review of factors influencing concentrations of nutrients and metabolites. Orthop. Clin. N. Am. 2011, 42, 465–477. [Google Scholar] [CrossRef]
- Krock, E.; Rosenzweig, D.H.; Haglund, L. The inflammatory milieu of the degenerate disc: Is mesenchymal stem cell-based therapy for intervertebral disc repair a feasible approach? Curr. Stem Cell Res. Ther. 2015, 10, 317–328. [Google Scholar] [CrossRef]
- Wuertz, K.; Haglund, L. Inflammatory mediators in intervertebral disk degeneration and discogenic pain. Glob. Spine J. 2013, 3, 175–184. [Google Scholar] [CrossRef] [PubMed]
- Alkhatib, B.; Rosenzweig, D.H.; Krock, E.; Roughley, P.J.; Beckman, L.; Steffen, T.; Weber, M.H.; Ouellet, J.A.; Haglund, L. Acute mechanical injury of the human intervertebral disc: Link to degeneration and pain. Eur. Cells Mater. 2014, 28, 98–111. [Google Scholar] [CrossRef]
- Binch, A.L.A.; Cole, A.A.; Breakwell, L.M.; Michael, A.L.R.; Chiverton, N.; Cross, A.K.; Le Maitre, C.L. Expression and regulation of neurotrophic and angiogenic factors during human intervertebral disc degeneration. Arthritis Res. Ther. 2014, 16, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Moure, J.; Moore, C.A.; Kim, K.; Karim, A.; Smith, K.; Barbosa, Z.; Van Eps, J.; Rameshwar, P.; Weiner, B. Novel therapeutic strategies for degenerative disc disease: Review of cell biology and intervertebral disc cell therapy. SAGE Open Med. 2018, 6, 2050312118761674. [Google Scholar] [CrossRef]
- Bowles, R.D.; Setton, L.A. Biomaterials for intervertebral disc regeneration and repair. Biomaterials 2017, 129, 54–67. [Google Scholar] [CrossRef]
- Liang, C.-Z.; Li, H.; Tao, Y.-Q.; Peng, L.-H.; Gao, J.-Q.; Wu, J.-J.; Li, F.-C.; Hua, J.-M.; Chen, Q.-X. Dual release of dexamethasone and TGF-β3 from polymeric microspheres for stem cell matrix accumulation in a rat disc degeneration model. Acta Biomater. 2013, 9, 9423–9433. [Google Scholar] [CrossRef]
- Francisco, A.T.; Mancino, R.J.; Bowles, R.D.; Brunger, J.M.; Tainter, D.M.; Chen, Y.-T.; Richardson, W.J.; Guilak, F.; Setton, L.A. Injectable laminin-functionalized hydrogel for nucleus pulposus regeneration. Biomaterials 2013, 34, 7381–7388. [Google Scholar] [CrossRef]
- Henriksson, H.B.; Svanvik, T.; Jonsson, M.; Hagman, M.; Horn, M.; Lindahl, A.; Brisby, H. Transplantation of human mesenchymal stems cells into intervertebral discs in a xenogeneic porcine model. Spine 2009, 34, 141–148. [Google Scholar] [CrossRef] [PubMed]
- Leckie, A.E.; Akens, M.K.; Woodhouse, K.A.; Yee, A.J.; Whyne, C.M. Evaluation of thiol-modified hyaluronan and elastin-like polypeptide composite augmentation in early-stage disc degeneration: Comparing 2 minimally invasive techniques. Spine 2012, 37, E1296–E1303. [Google Scholar] [CrossRef]
- Malhotra, N.R.; Han, W.M.; Beckstein, J.; Cloyd, J.; Chen, W.; Elliott, D.M. An injectable nucleus pulposus implant restores compressive range of motion in the ovine disc. Spine 2012, 37, E1099–E1105. [Google Scholar] [CrossRef]
- Gupta, P.K.; Chullikana, A.; Rengasamy, M.; Shetty, N.; Pandey, V.; Agarwal, V.; Wagh, S.Y.; Vellotare, P.K.; Damodaran, D.; Viswanathan, P.; et al. Efficacy and safety of adult human bone marrow-derived, cultured, pooled, allogeneic mesenchymal stromal cells (Stempeucel®): Preclinical and clinical trial in osteoarthritis of the knee joint. Arthritis Res. Ther. 2016, 18, 1–18. [Google Scholar] [CrossRef]
- Park, Y.-B.; Ha, C.-W.; Lee, C.-H.; Yoon, Y.C. Cartilage regeneration in osteoarthritic patients by a composite of allogeneic umbilical cord blood-derived mesenchymal stem cells and hyaluronate hydrogel: Results from a clinical trial for safety and proof-of-concept with 7 years of extended follow-up. Stem Cells Transl. Med. 2017, 6, 613–621. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, T.A.; Dare, E.V.; Hincke, M. Fibrin: A versatile scaffold for tissue engineering applications. Tissue Eng. Part B Rev. 2008, 14, 199–215. [Google Scholar] [CrossRef] [PubMed]
- Davis, H.; Miller, S.; Case, E.; Leach, J. Supplementation of fibrin gels with sodium chloride enhances physical properties and ensuing osteogenic response. Acta Biomater. 2011, 7, 691–699. [Google Scholar] [CrossRef]
- Park, S.-H.; Cho, H.; Gil, E.S.; Mandal, B.B.; Min, B.-H.; Kaplan, D.L. Silk-fibrin/hyaluronic acid composite gels for nucleus pulposus tissue regeneration. Tissue Eng. Part A 2011, 17, 2999–3009. [Google Scholar] [CrossRef] [PubMed]
- Park, S.-H.; Gil, E.S.; Mandal, B.B.; Cho, H.S.; Kluge, J.A.; Min, B.-H.; Kaplan, D.L. Annulus fibrosus tissue engineering using lamellar silk scaffolds. J. Tissue Eng. Regen. Med. 2012, 6 (Suppl. 3), s24–s33. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Ramay, H.R.; Hauch, K.D.; Xiao, D.; Zhang, M. Chitosan–alginate hybrid scaffolds for bone tissue engineering. Biomaterials 2005, 26, 3919–3928. [Google Scholar] [CrossRef]
- Kim, I.-Y.; Seo, S.-J.; Moon, H.-S.; Yoo, M.-K.; Park, I.-Y.; Kim, B.-C.; Cho, C.-S. Chitosan and its derivatives for tissue engineering applications. Biotechnol. Adv. 2008, 26, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Kranenburg, H.-J.C.; Meij, B.P.; Onis, D.; Van Der Veen, A.J.; Saralidze, K.; Smolders, L.A.; Huizinga, J.G.; Knetsch, M.L.W.; Luijten, P.R.; Visser, F.; et al. Design, synthesis, imaging, and biomechanics of a softness-gradient hydrogel nucleus pulposus prosthesis in a canine lumbar spine model. J. Biomed. Mater. Res. Part B Appl. Biomater. 2012, 100, 2148–2155. [Google Scholar] [CrossRef] [PubMed]
- Kumar, D.; Gerges, I.; Tamplenizza, M.; Lenardi, C.; Forsyth, N.R.; Liu, Y. Three-dimensional hypoxic culture of human mesenchymal stem cells encapsulated in a photocurable, biodegradable polymer hydrogel: A potential injectable cellular product for nucleus pulposus regeneration. Acta Biomater. 2014, 10, 3463–3474. [Google Scholar] [CrossRef]
- Durdag, E.; Ayden, O.; Albayrak, S.; Atci, I.B.; Armagan, E. Fragmentation to epidural space: First documented complication of Gelstix(TM.). Turk. Neurosurg. 2014, 24, 602–605. [Google Scholar]
- Penolazzi, L.; Pozzobon, M.; Bergamin, L.S.; D’Agostino, S.; Francescato, R.; Bonaccorsi, G.; De Bonis, P.; Cavallo, M.; Lambertini, E.; Piva, R. Extracellular matrix from decellularized Wharton’s jelly improves the behavior of cells from degenerated intervertebral disc. Front. Bioeng. Biotechnol. 2020, 8, 262. [Google Scholar] [CrossRef] [PubMed]
- Ishiguro, H.; Kaito, T.; Yarimitsu, S.; Hashimoto, K.; Okada, R.; Kushioka, J.; Chijimatsu, R.; Takenaka, S.; Makino, T.; Sakai, Y.; et al. Intervertebral disc regeneration with an adipose mesenchymal stem cell-derived tissue-engineered construct in a rat nucleotomy model. Acta Biomater. 2019, 87, 118–129. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Wang, J.; Fang, W.; Tao, Y.; Zhao, T.; Xia, K.; Liang, C.; Hua, J.; Li, F.; Chen, Q. Genipin cross-linked type II collagen/chondroitin sulfate composite hydrogel-like cell delivery system induces differentiation of adipose-derived stem cells and regenerates degenerated nucleus pulposus. Acta Biomater. 2018, 71, 496–509. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Wang, J.; Huang, X.; Fang, W.; Tao, Y.; Zhao, T.; Liang, C.; Hua, J.; Chen, Q.; Li, F. Injectable decellularized nucleus pulposus-based cell delivery system for differentiation of adipose-derived stem cells and nucleus pulposus regeneration. Acta Biomater. 2018, 81, 115–128. [Google Scholar] [CrossRef]
- Choy, A.T.H.; Chan, B.P. A Structurally and Functionally Biomimetic Biphasic Scaffold for Intervertebral Disc Tissue Engineering. PLoS ONE 2015, 10, e0131827. [Google Scholar] [CrossRef] [PubMed]
- Chik, T.K.; Chooi, W.H.; Li, Y.Y.; Ho, F.C.; Cheng, H.W.; Choy, T.H.; Sze, K.Y.; Luk, K.K.D.; Cheung, K.M.C.; Chan, B.P. Bioengineering a Multicomponent Spinal Motion Segment Construct-A 3D Model for complex tissue engineering. Adv. Healthc. Mater. 2014, 4, 99–112. [Google Scholar] [CrossRef]
- Martin, J.T.; Milby, A.H.; Chiaro, J.A.; Kim, D.H.; Hebela, N.M.; Smith, L.J.; Elliott, D.M.; Mauck, R.L. Translation of an engineered nanofibrous disc-like angle-ply structure for intervertebral disc replacement in a small animal model. Acta Biomater. 2014, 10, 2473–2481. [Google Scholar] [CrossRef] [PubMed]
- Sivan, S.; Roberts, S.; Urban, J.; Menage, J.; Bramhill, J.; Campbell, D.; Franklin, V.; Lydon, F.; Merkher, Y.; Maroudas, A.; et al. Injectable hydrogels with high fixed charge density and swelling pressure for nucleus pulposus repair: Biomimetic glycosaminoglycan analogues. Acta Biomater. 2014, 10, 1124–1133. [Google Scholar] [CrossRef] [PubMed]
- Jeong, C.G.; Francisco, A.T.; Niu, Z.; Mancino, R.L.; Craig, S.L.; Setton, L.A. Screening of hyaluronic acid–poly(ethylene glycol) composite hydrogels to support intervertebral disc cell biosynthesis using artificial neural network analysis. Acta Biomater. 2014, 10, 3421–3430. [Google Scholar] [CrossRef] [PubMed]
- Peng, Y.; Huang, D.; Liu, S.; Li, J.; Qing, X.; Shao, Z. Biomaterials-induced stem cells specific differentiation into intervertebral disc lineage cells. Front. Bioeng. Biotechnol. 2020, 8, 56. [Google Scholar] [CrossRef]
- Ishihara, H.; Warensjo, K.; Roberts, S.; Urban, J.P. Proteoglycan synthesis in the intervertebral disk nucleus: The role of extracellular osmolality. Am. J. Physiol. 1997, 272 Pt 1, C1499–C1506. [Google Scholar] [CrossRef]
- Schollum, M.L.; Robertson, P.A.; Broom, N.D. ISSLS prize winner: Microstructure and mechanical disruption of the lumbar disc annulus: Part I: A microscopic investigation of the translamellar bridging network. Spine 2008, 33, 2702–2710. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Fairbank, J.C.T.; Roberts, S.; Urban, J.P.G. The elastic fiber network of the anulus fibrosus of the normal and scoliotic human intervertebral disc. Spine 2005, 30, 1815–1820. [Google Scholar] [CrossRef]
- Nakamichi, R.; Ito, Y.; Inui, M.; Onizuka, N.; Kayama, T.; Kataoka, K.; Suzuki, H.; Mori, M.; Inagawa, M.; Ichinose, S.; et al. Mohawk promotes the maintenance and regeneration of the outer annulus fibrosus of intervertebral discs. Nat. Commun. 2016, 7, 12503. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Xu, B.; Yang, Q.; Li, X.; Ma, X.; Xia, Q.; Zhang, Y.; Zhang, C.; Wu, Y.; Zhang, Y. Comparison of decellularization protocols for preparing a decellularized porcine annulus fibrosus scaffold. PLoS ONE 2014, 9, e86723. [Google Scholar] [CrossRef] [PubMed]
- Pirvu, T.; Blanquer, S.B.; Benneker, L.M.; Grijpma, D.W.; Richards, R.G.; Alini, M.; Eglin, D.; Grad, S.; Li, Z. A combined biomaterial and cellular approach for annulus fibrosus rupture repair. Biomaterials 2015, 42, 11–19. [Google Scholar] [CrossRef]
- Zhu, C.; Li, J.; Liu, C.; Zhou, P.; Yang, H.; Li, B. Modulation of the gene expression of annulus fibrosus-derived stem cells using poly(ether carbonate urethane)urea scaffolds of tunable elasticity. Acta Biomater. 2016, 29, 228–238. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.-C.; Su, W.-Y.; Yang, S.-H.; Gefen, A.; Lin, F.-H. In situ forming hydrogels composed of oxidized high molecular weight hyaluronic acid and gelatin for nucleus pulposus regeneration. Acta Biomater. 2013, 9, 5181–5193. [Google Scholar] [CrossRef]
- Choi, U.Y.; Joshi, H.P.; Payne, S.; Kim, K.T.; Kyung, J.W.; Choi, H.; Cooke, M.J.; Kwon, S.Y.; Roh, E.J.; Sohn, S.; et al. An Injectable Hyaluronan–Methylcellulose (HAMC) Hydrogel combined with Wharton’s jelly-derived mesenchymal Stromal cells (WJ-MSCs) promotes degenerative disc repair. Int. J. Mol. Sci. 2020, 21, 7391. [Google Scholar] [CrossRef] [PubMed]
- Gan, Y.; Li, P.; Wang, L.; Mo, X.; Song, L.; Xu, Y.; Zhao, C.; Ouyang, B.; Tu, B.; Luo, L.; et al. An interpenetrating network-strengthened and toughened hydrogel that supports cell-based nucleus pulposus regeneration. Biomaterials 2017, 136, 12–28. [Google Scholar] [CrossRef]
- Bridgen, D.T.; Fearing, B.V.; Jing, L.; Sanchez-Adams, J.; Cohan, M.C.; Guilak, F.; Chen, J.; Setton, L.A. Regulation of human nucleus pulposus cells by peptide-coupled substrates. Acta Biomater. 2017, 55, 100–108. [Google Scholar] [CrossRef]
- Wan, S.; Borland, S.; Richardson, S.M.; Merry, C.L.; Saiani, A.; Gough, J.E. Self-assembling peptide hydrogel for intervertebral disc tissue engineering. Acta Biomater. 2016, 46, 29–40. [Google Scholar] [CrossRef] [PubMed]
- Nesti, L.J.; Li, W.-J.; Shanti, R.M.; Jiang, Y.J.; Jackson, W.; Freedman, B.A.; Kuklo, T.R.; Giuliani, J.R.; Tuan, R.S. Intervertebral disc tissue engineering using a Novel Hyaluronic Acid–Nanofibrous Scaffold (HANFS) amalgam. Tissue Eng. Part A 2008, 14, 1527–1537. [Google Scholar] [CrossRef]
- Park, S.-H.; Gil, E.S.; Cho, H.; Mandal, B.B.; Tien, L.W.; Min, B.-H.; Kaplan, D.L. Intervertebral disk tissue engineering using biphasic silk composite scaffolds. Tissue Eng. Part A 2012, 18, 447–458. [Google Scholar] [CrossRef]
- Yang, J.; Wang, L.; Zhang, W.; Sun, Z.; Li, Y.; Yang, M.; Zeng, D.; Peng, B.; Zheng, W.; Jiang, X.; et al. Reverse reconstruction and bioprinting of bacterial cellulose-based functional total intervertebral disc for therapeutic implantation. Small 2018, 14, 1702582. [Google Scholar] [CrossRef] [PubMed]
- Chan, L.K.; Leung, V.Y.; Tam, V.; Lu, W.W.; Sze, K.; Cheung, K.M. Decellularized bovine intervertebral disc as a natural scaffold for xenogenic cell studies. Acta Biomater. 2013, 9, 5262–5272. [Google Scholar] [CrossRef] [PubMed]
- Hensley, A.; Rames, J.; Casler, V.; Rood, C.; Walters, J.; Fernandez, C.; Gill, S.; Mercuri, J.J. Decellularization and characterization of a whole intervertebral disk xenograft scaffold. J. Biomed. Mater. Res. Part A 2018, 106, 2412–2423. [Google Scholar] [CrossRef]
- Da Costa, R.C.; De Decker, S.; Lewis, M.J.; Volk, H.; Canine Spinal Cord Injury Consortium (CANSORT-SCI). Diagnostic imaging in intervertebral disc disease. Front. Vet. Sci. 2020, 7, 588338. [Google Scholar] [PubMed]
| Author | Cell Line | Effect |
|---|---|---|
| 2019, Shi et al. [40] 2019, Sheyn et al. [33] | Neonatal human dermal fibroblasts Rabbit, in vivo Notochordal cells from human iPSCs | Increased regeneration markers Reduction of disc degeneration in a porcine model |
| 2018, Teixeria et al. [41] | Human BM-MSC Bovine, ex-vivo | Promoted cell migration and increased inflammatory cytokine expression |
| 2018, Wang et al. [42] | Rat BM-MSC Rat, in vivo | Hypoxic pre-treatment of BM-MSC with CoCl2 enhanced migration, decreased apoptosis, increased disc height, MSC numbers in the NP and AF, and extracellular matrix production |
| 2017, Maidhof et al. [43] | Allogeneic rat BM-MSC Rat, in vivo | Cell therapies administered at an early stage of injury or disease progression may have greater chances of mitigating matrix loss |
| 2017, Hang et al. [44] | Autologous canine BM-MSC Canine, in vivo | PET was more reliable than MRI for quantifying implanted BM-MSC survival |
| 2017, Steffen et al. [45] | Autologous canine BM-MSC Canine, in vivo | Successful injection of BM-MSC into lumbosacral discs of naturally IVD-degenerative canines |
| 2017, Noriega et al. [46] | Allogeneic BM-MSC Clinical trial (N = 24, follow-up: 12 months) | Significant VAS and ODI reductions, improvement on MRI |
| 2017, Centeno et al. [47] | Autologous BM-MSC Clinical trial (N = 33, follow-up: 72 months) | Disc bulging reduction on MRI, pain and function improvement |
| 2017, Kumar et al. [48] | Autologous AD-MSC Clinical trial (N = 10, follow-up: 12 months) | Combined implantation of AD-MSC and hyaluronic acid in discogenic back pain is safe and tolerable |
| 2017, Pettine et al. [49] | Autologous BM-MSC Clinical trial (N = 26, follow-up: 36 months) | Evidence for the safety and feasibility of intradiscal BM concentrate therapy |
| 2016, Tschugg et al. [50] | Autologous disc chondrocyte Clinical trial (N = 120, follow-up: 48 months) | Ongoing study |
| Author | Materials | Effect |
|---|---|---|
| 2020, Penolazzi et al. [74] | Decellularized Wharton’s jelly matrix from human umbilical cord as ECM-based scaffold | Promoted cell differentiation toward a discogenic phenotype, positively affected the expression of regulators of IVD homeostasis |
| 2019, Ishiguro et al. [75] | AD-MSC-Tissue engineered construct Rat | Regenerative efficacy was investigated structurally and biomechanically up to 6 months after implantation |
| 2018, Zhou et al. [76] | Type II collagen/chondroitin sulfate (CS) composite hydrogel-like adipose-derived stem cell delivery system | Minimally invasive approach to promote the regeneration of degenerated NP |
| 2018, Zhou et al. [77] | Injectable decellularized NP-based cell delivery system (NPCS) | The mechanical properties of the NPCS system were similar to those of fresh NP; Biocompatible; It induced NP-like differentiation and ECM synthesis |
| 2015, Choy et al. [78] | Collagen-glycosaminoglycan (GAG) co-precipitate and multiple lamellae of a photo-crosslinked collagen membrane | A biphasic scaffold comprising 10 AF-like lamellae had the best mechanical performance and elastic compliance |
| 2015, Chik et al. [79] | Collagen-GAG coprecipitate MSC and contracted collagen gel, MSC | Spinal motion segment tissue engineering. Provided a 3D model for studying tissue maturation and functional remodeling |
| 2014, Martin et al. [80] | Electrospun poly scaffold with cell-seeded hydrogels and disc-like angle-ply structure | Optimized the design of functional disc replacement in vivo |
| 2014, Sivan et al. [81] | Biomimetic GAG analogue based on sulphonate-containing polymer | Provided intrinsic swelling pressure which could maintain disc hydration and height |
| 2014, Jeong et al. [82] | Hyaluronic acid-poly(ethylene glycol) composite hydrogel | Highest number of NP and AF cells on HA-PEG hydrogels from lower molecular weight HA |
| Type | Advantages | Disadvantages | ||
|---|---|---|---|---|
| Growth factor | GDF-5, IGF-1, TGF-β, bFGF, OP-1 | Stimulation of ECM production | Short half-life Need repeated injection | |
| Gene therapy | Virus mediated Non-virus mediated RNAi CRISPR/Cas9 | Long-lasting and timeless effects | Safety concerns Ethical concerns Significant cost | |
| Stem cell | ESCs | Differentiation into three germ layers Self-renewal and high replication | Immune rejection concern Ethical concerns | |
| Potential for tumor formation | ||||
| iPSCs | Less ethical concerns than ESCs Patient-specific Autologous | Need method standardization Potential for tumor formation Need validation for safety | ||
| MSCs | Bone marrow, Adipose tissue, umbilical cord Wharton’s jelly Synovial membrane | Less ethical concerns than ESCs and iPSCs | Less cell proliferation Limit differentiation potential | |
| Tissue engineering | Combination: stem cells, biomaterials, and growth factors | Ideal constructs | Need validation for biodegradation, biocompatibility, and optimal | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, C.K.; Heo, D.H.; Chung, H.; Roh, E.J.; Darai, A.; Kyung, J.W.; Choi, H.; Kwon, S.Y.; Bhujel, B.; Han, I. Advances in Tissue Engineering for Disc Repair. Appl. Sci. 2021, 11, 1919. https://doi.org/10.3390/app11041919
Lee CK, Heo DH, Chung H, Roh EJ, Darai A, Kyung JW, Choi H, Kwon SY, Bhujel B, Han I. Advances in Tissue Engineering for Disc Repair. Applied Sciences. 2021; 11(4):1919. https://doi.org/10.3390/app11041919
Chicago/Turabian StyleLee, Chang Kyu, Dong Hwa Heo, Hungtae Chung, Eun Ji Roh, Anjani Darai, Jae Won Kyung, Hyemin Choi, Su Yeon Kwon, Basanta Bhujel, and Inbo Han. 2021. "Advances in Tissue Engineering for Disc Repair" Applied Sciences 11, no. 4: 1919. https://doi.org/10.3390/app11041919
APA StyleLee, C. K., Heo, D. H., Chung, H., Roh, E. J., Darai, A., Kyung, J. W., Choi, H., Kwon, S. Y., Bhujel, B., & Han, I. (2021). Advances in Tissue Engineering for Disc Repair. Applied Sciences, 11(4), 1919. https://doi.org/10.3390/app11041919








