Effects of Leukocyte-Platelet-Rich Fibrin (L–PRF) on Pain, Soft Tissue Healing, Growth Factors, and Cytokines after Third Molar Extraction: A Randomized, Split-Mouth, Double-Blinded Clinical Trial
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Outcomes
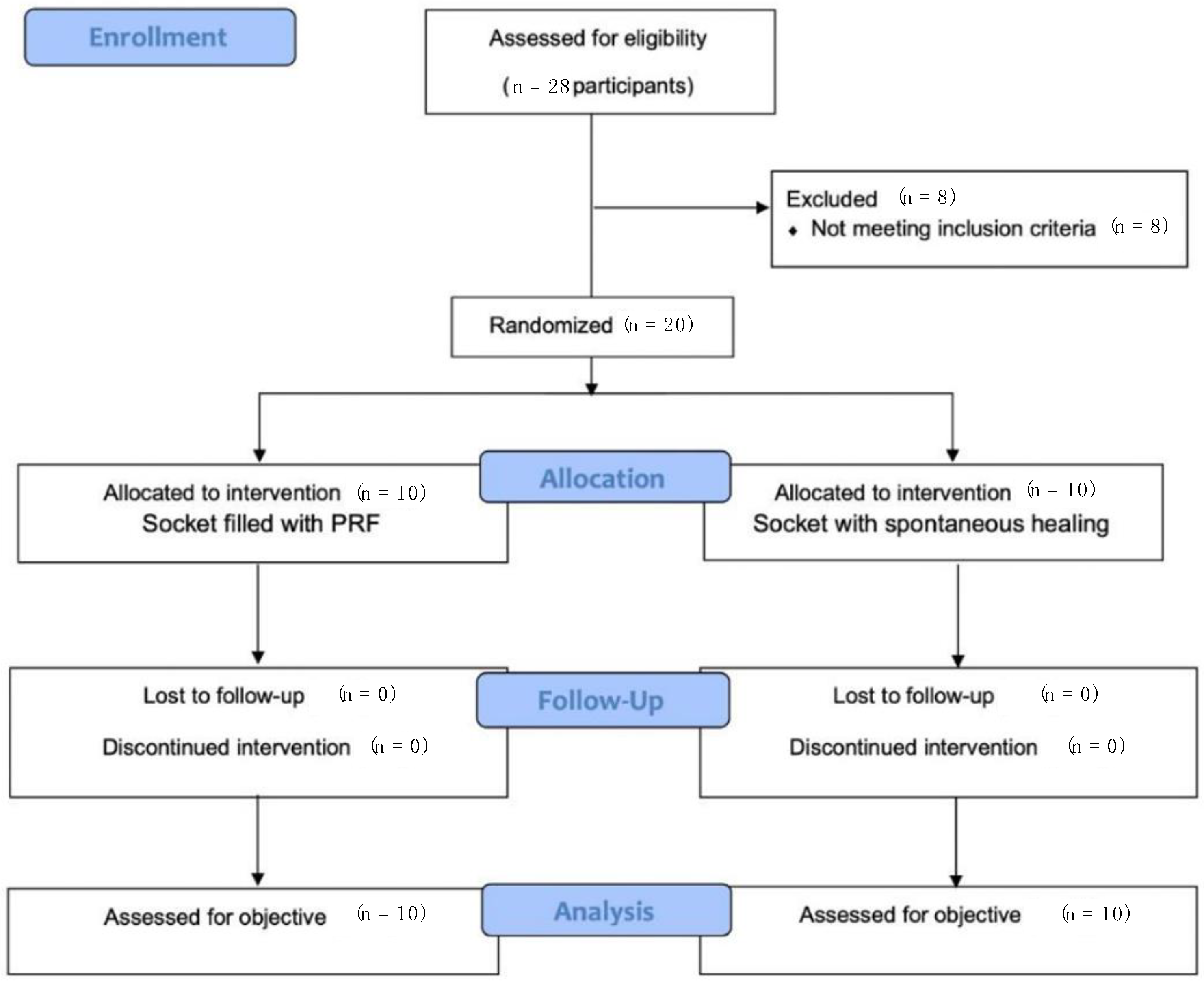
2.2. Sample Calculation and Randomization
2.3. Inclusion and Exclusion Criteria
2.4. Participant Selection
2.5. Pre-Surgical Procedures
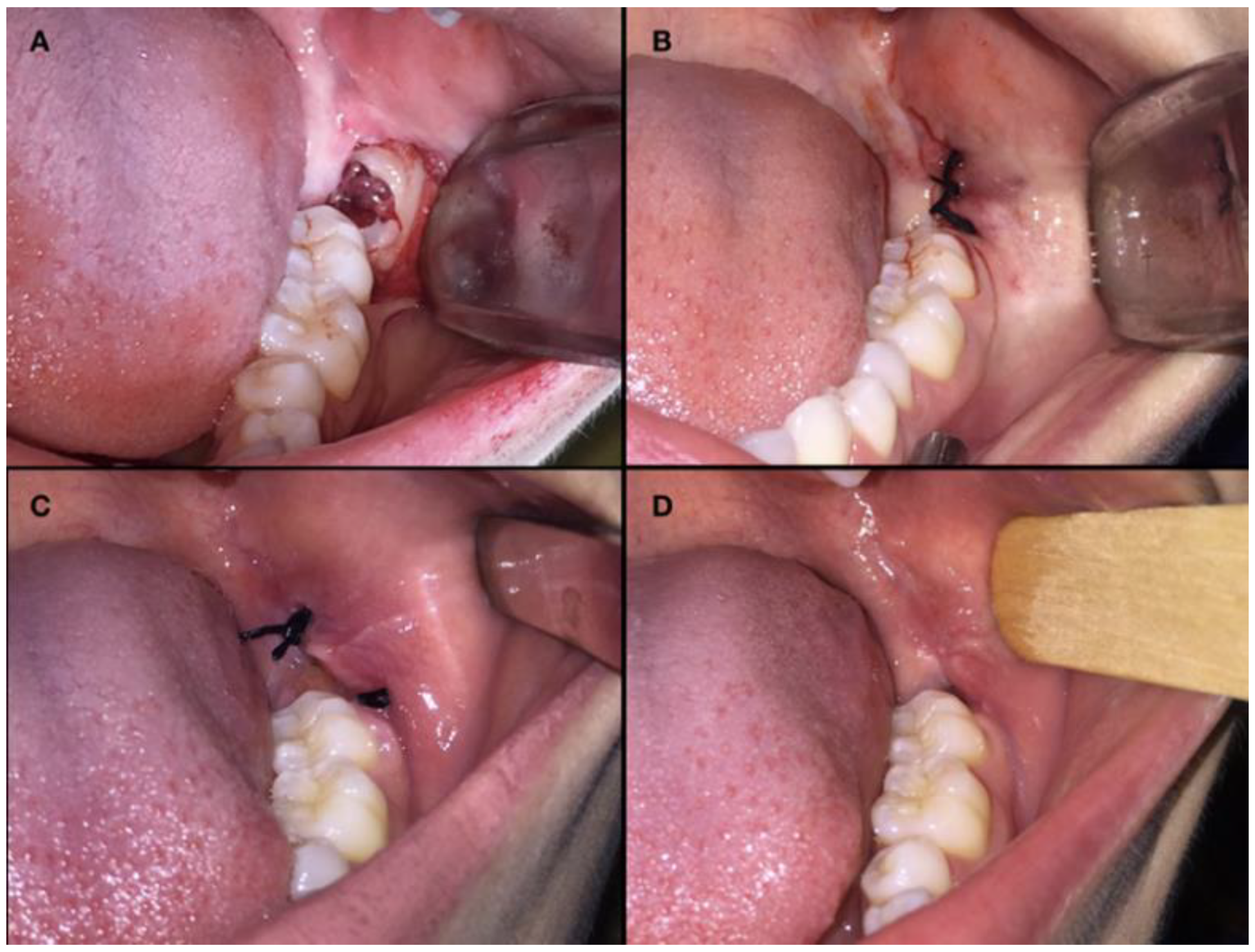
2.6. Surgical Procedures
2.7. Clinical Measurements
2.8. Quantification of Cytokines and Growth Factors
2.9. Statistical Analysis
3. Results
3.1. Clinical Evaluation
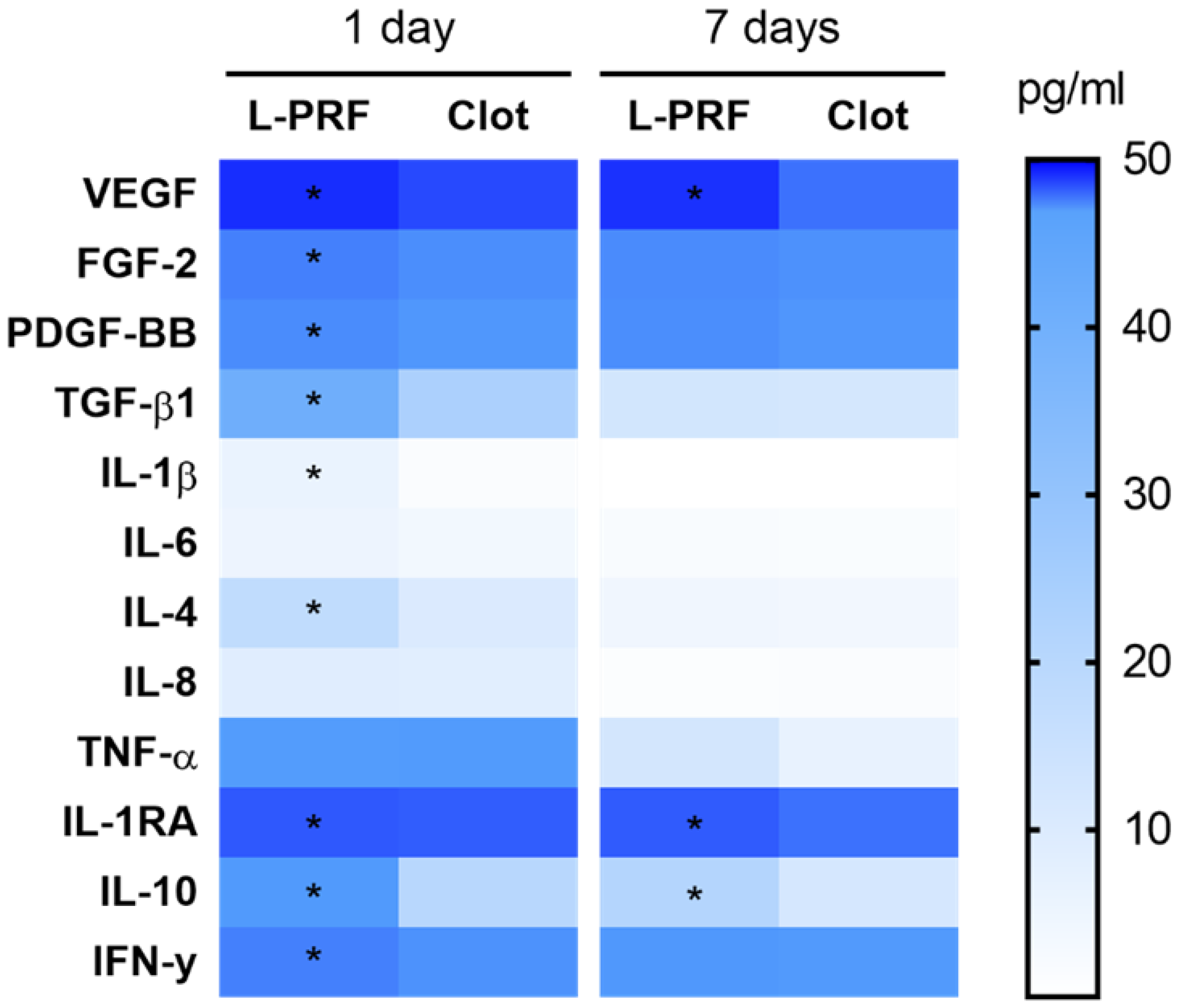
3.2. Biochemical Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Osunde, O.D.; Adebola, R.A.; Saheeb, B.D. A comparative study of the effect of suture-less and multiple suture techniques on inflammatory complications following third molar surgery. Int. J. Oral Maxillofac. Surg. 2012, 41, 1275–1279. [Google Scholar] [CrossRef]
- Kaewkumnert, S.; Phithaksinsuk, K.; Changpoo, C.; Nochit, N.; Muensaiyat, Y.; Wilaipornsawai, S.; Piriyaphokai, U.; Powcharoen, W. Comparison of intraosseous and submucosal dexamethasone injection in mandibular third molar surgery: A split-mouth randomized clinical trial. Int. J. Oral Maxillofac. Surg. 2020, 49, 529–535. [Google Scholar] [CrossRef] [PubMed]
- Zandi, M.; Amini, P.; Keshavarz, A. Effectiveness of cold therapy in reducing pain, trismus, and oedema after impacted mandibular third molar surgery: A randomized, self-controlled, observer-blind, split-mouth clinical trial. Int. J. Oral Maxillofac. Surg. 2016, 45, 118–123. [Google Scholar] [CrossRef] [PubMed]
- Afat, İ.M.; Akdoğan, E.T.; Gönül, O. Effects of Leukocyte-and Platelet-Rich Fibrin Alone and Combined With Hyaluronic Acid on Pain, Edema, and Trismus after Surgical Extraction of Impacted Mandibular Third Molars. J. Oral Maxillofac. Surg. 2018, 76, 926–932. [Google Scholar] [CrossRef] [PubMed]
- Ritto, F.G.; Pimentel, T.; Canellas, J.V.S.; Junger, B.; Cruz, M.; Medeiros, P.J. Randomized double-blind clinical trial evaluation of bone healing after third molar surgery with the use of leukocyte- and platelet-rich fibrin. Int. J. Oral Maxillofac. Surg. 2019, 48, 1088–1093. [Google Scholar] [CrossRef]
- Ghanaati, S.; Booms, P.; Orlowska, A.; Kubesch, A.; Lorenz, J.; Rutkowski, J.; Landes, C.; Sader, R.; Kirkpatrick, C.; Choukroun, J. Advanced platelet-rich fibrin: A new concept for cell-based tissue engineering by means of inflammatory cells. J. Oral Implantol. 2014, 40, 679–689. [Google Scholar] [CrossRef] [PubMed]
- Kang, Y.H.; Jeon, S.H.; Park, J.Y.; Chung, J.H.; Choung, Y.H.; Choung, H.W.; Kim, E.S.; Choung, P.H. Platelet-rich fibrin is a Bioscaffold and reservoir of growth factors for tissue regeneration. Tissue Eng. Part A 2011, 17, 349–359. [Google Scholar] [CrossRef]
- Dohan, D.M.; Choukroun, J.; Diss, A.; Dohan, S.L.; Dohan, A.J.; Mouhyi, J.; Gogly, B. Platelet-rich fibrin (PRF): A second-generation platelet concentrate. Part III: Leucocyte activation: A new feature for platelet concentrates? Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 2006, 101, e51–e55. [Google Scholar] [CrossRef]
- Lourenco, E.S.; Mourao, C.; Leite, P.E.C.; Granjeiro, J.M.; Calasans-Maia, M.D.; Alves, G.G. The in vitro release of cytokines and growth factors from fibrin membranes produced through horizontal centrifugation. J. Biomed. Mater. Res. Part A 2018, 106, 1373–1380. [Google Scholar] [CrossRef]
- de Almeida Barros Mourão, C.F.; de Mello-Machado, R.C.; Javid, K.; Moraschini, V. The use of leukocyte- and platelet-rich fibrin in the management of soft tissue healing and pain in post-extraction sockets: A randomized clinical trial. J. Craniomaxillofac. Surg. 2020, 48, 452–457. [Google Scholar] [CrossRef]
- Lektemur Alpan, A.; Torumtay Cin, G. PRF improves wound healing and postoperative discomfort after harvesting subepithelial connective tissue graft from palate: A randomized controlled trial. Clin. Oral Investig. 2020, 24, 425–436. [Google Scholar] [CrossRef] [PubMed]
- Miron, R.J.; Xu, H.; Chai, J.; Wang, J.; Zheng, S.; Feng, M.; Zhang, X.; Wei, Y.; Chen, Y.; Mourão, C.; et al. Comparison of platelet-rich fibrin (PRF) produced using 3 commercially available centrifuges at both high (~700 g) and low (~200 g) relative centrifugation forces. Clin. Oral Investig. 2020, 24, 1171–1182. [Google Scholar] [CrossRef]
- Ahmed, M.; Shama, A.A.; Hamdy, R.; Ezz, M. Bioresorbable versus titanium space-maintaining mesh in maxillary sinus floor elevation: A split-mouth study. Int. J. Oral Maxillofac. Surg. 2017, 46, 1178–1187. [Google Scholar] [CrossRef] [PubMed]
- Taschieri, S.; Testori, T.; Corbella, S.; Weinstein, R.; Francetti, L.; Di Giancamillo, A.; Del Fabbro, M. Platelet-rich plasma and deproteinized bovine bone matrix in maxillary sinus lift surgery: A split-mouth histomorphometric evaluation. Implant Dent. 2015, 24, 592–597. [Google Scholar] [CrossRef] [PubMed]
- Schulz, K.F.; Altman, D.G.; Moher, D. CONSORT 2010 Statement: Updated guidelines for reporting parallel group randomised trials. BMJ 2010, 340, c332. [Google Scholar] [CrossRef]
- Kawase, T.; Mubarak, S.; Mourão, C.F. The Platelet Concentrates Therapy: From the Biased Past to the Anticipated Future. Bioengineering 2020, 7, 82. [Google Scholar] [CrossRef]
- Peterson, L.J. Principles of management of impacted teeth. In Contemporary Oral and Maxillofacial Surgery, 3rd ed.; Mosby: St. Louis, MO, USA, 1998; pp. 215–248. [Google Scholar]
- Ghanaati, S.; Mourão, C.F.; Adam, E.H.; Sader, R.; Zadeh, H.H.; Al-Maawi, S. The role of centrifugation process in the preparation of therapeutic blood concentrates: Standardization of the protocols to improve reproducibility. Int. J. Growth Factors Stem Cells Dent. 2019, 2, 41–44. [Google Scholar] [CrossRef]
- Landry, R.G. Effectiveness of Benzydamine HC1 in the Treatment of Periodontal Post-Surgical Patients; Faculty of Dentistry, University of Toronto: Toronto, ON, Canada, 1985. [Google Scholar]
- Huskisson, E.; Jones, J.; Scott, P.J. Application of visual-analogue scales to the measurement of functional capacity. Rheumatology 1976, 15, 185–187. [Google Scholar] [CrossRef]
- Frey-Law, L.A.; Lee, J.E.; Wittry, A.M.; Melyon, M. Pain rating schema: Three distinct subgroups of individuals emerge when rating mild, moderate, and severe pain. J. Pain Res. 2013, 7, 13–23. [Google Scholar] [CrossRef]
- Hinkle, D.E.; Wiersma, W.; Jurs, S.G. Applied Statistics for the Behavioral Sciences; Houghton Mifflin College Division: Boston, MA, USA, 2003; Volume 663. [Google Scholar]
- Biesbrock, A.; Yeh, C.H. Relationship of Surface Epithelium Concentrations of IL-1α and IL-1β to Cinical Inflammation during Experimental Gingivitis. In Assessment of Oral Health; Karger Publishers: Berlin, Germany, 2000; Volume 17, pp. 20–31. [Google Scholar]
- Perkins, M.A.; Osterhues, M.A.; Farage, M.A.; Robinson, M.K. A noninvasive method to assess skin irritation and compromised skin conditions using simple tape adsorption of molecular markers of inflammation. Skin Res. Technol. 2001, 7, 227–237. [Google Scholar] [CrossRef] [PubMed]
- Jaedicke, K.M.; Preshaw, P.M.; Taylor, J.J. Salivary cytokines as biomarkers of periodontal diseases. Periodontology 2000, 70, 164–183. [Google Scholar] [CrossRef]
- Nunes, Q.M.; Li, Y.; Sun, C.; Kinnunen, T.K.; Fernig, D.G. Fibroblast growth factors as tissue repair and regeneration therapeutics. PeerJ 2016, 4, e1535. [Google Scholar] [CrossRef]
- Bao, P.; Kodra, A.; Tomic-Canic, M.; Golinko, M.S.; Ehrlich, H.P.; Brem, H. The role of vascular endothelial growth factor in wound healing. J. Surg. Res. 2009, 153, 347–358. [Google Scholar] [CrossRef]
- Eshwar, S.S.P.; Victor, D.J.; Sangeetha, S.; Prakash, P. Platelet Rich Plasma in Periodontal Therapy. J. Pharm. Sci. Res. 2017, 9, 965–971. [Google Scholar]
- Zhang, Y.; Alexander, P.B.; Wang, X.-F. TGF-β family signaling in the control of cell proliferation and survival. Cold Spring Harb. Perspect. Biol. 2017, 9, a022145. [Google Scholar] [CrossRef] [PubMed]
- Miron, R.J.; Chai, J.; Zhang, P.; Li, Y.; Wang, Y.; Mourão, C.; Sculean, A.; Fujioka Kobayashi, M.; Zhang, Y. A novel method for harvesting concentrated platelet-rich fibrin (C-PRF) with a 10-fold increase in platelet and leukocyte yields. Clin. Oral Investig. 2019, 24, 2819–2828. [Google Scholar] [CrossRef] [PubMed]
- Belstrøm, D.; Damgaard, C.; Könönen, E.; Gürsoy, M.; Holmstrup, P.; Gürsoy, U.K. Salivary cytokine levels in early gingival inflammation. J. Oral Microbiol. 2017, 9, 1364101. [Google Scholar] [CrossRef]
- Kitamura, Y.; Watanabe, T.; Nakamura, M.; Isobe, K.; Kawabata, H.; Uematsu, K.; Okuda, K.; Nakata, K.; Tanaka, T.; Kawase, T. Platelet Counts in Insoluble Platelet-Rich Fibrin Clots: A Direct Method for Accurate Determination. Front Bioeng. Biotechnol. 2018, 6, 4. [Google Scholar] [CrossRef]
- Kawase, T.; Nagata, M.; Okuda, K.; Ushiki, T.; Fujimoto, Y.; Watanabe, M.; Ito, A.; Nakata, K. Platelet-Rich Fibrin Extract: A Promising Fetal Bovine Serum Alternative in Explant Cultures of Human Periosteal Sheets for Regenerative Therapy. Int. J. Mol. Sci. 2019, 20, 1053. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Chen, J.; Huang, Y.; Pan, Q.; Nie, M. Local Application of Platelet-Rich Fibrin During Lower Third Molar Extraction Improves Treatment Outcomes. J. Oral Maxillofac. Surg. 2017, 75, 2497–2506. [Google Scholar] [CrossRef] [PubMed]
- Sirintawat, N.; Sawang, K.; Chaiyasamut, T.; Wongsirichat, N. Pain measurement in oral and maxillofacial surgery. J. Dent. Anesth. Pain Med. 2017, 17, 253–263. [Google Scholar] [CrossRef] [PubMed]
- Lesaffre, E.; Philstrom, B.; Needleman, I.; Worthington, H. The design and analysis of split-mouth studies: What statisticians and clinicians should know. Stat. Med. 2009, 28, 3470–3482. [Google Scholar] [CrossRef] [PubMed]

| Gender | n = 20 (Participants) |
|---|---|
| Male | 6 |
| Female | 14 |
| Age (years), mean ± SD | 23 ± 3.28 |
| Education Level | |
| High school diploma | 14 (9 female and 5 male) |
| Bachelor’s degree | 6 (5 female and 1 male) |
| Extraction indication | n = 40 (dental extractions) |
| Previous pericoronitis | 17 |
| Dental caries | 9 |
| Orthodontic treatment | 14 |
| VEGF | TGF–β1 | FGFb | PDGF–bb | TNF–α | IL–10 | IL–6 | IL–4 | IL–1β | IL–8 | IL–1RA | IFN–γ | ||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Clot | |||||||||||||
| Leucocytes (WBC) | Spearman ρ | −0.432 | 0.0365 | 0.335 | 0.054 | −0.032 | −0.275 | 0.450 | −0.481 | 0.098 | −0.429 | −0.372 | −0.243 |
| p value | 0.212 | 0.924 | 0.341 | 0.884 | 0.933 | 0.435 | 0.191 | 0.159 | 0.789 | 0.216 | 0.286 | 0.494 | |
| Platelets | Spearman ρ | 0.079 | 0.382 | −0.468 | −0.395 | 0.110 | 0.176 | 0.123 | 0.175 | 0.132 | 0.129 | 0.1963 | 0.060 |
| p value | 0.899 | 0.272 | 0.173 | 0.256 | 0.764 | 0.620 | 0.734 | 0.620 | 0.713 | 0.719 | 0.578 | 0.871 | |
| L–PRF | |||||||||||||
| Leucocytes (WBC) | Spearman ρ | −0.561 | 0.465 | −0.226 | −0.287 | −0.440 | −0.440 | 0.000 | −0.142 | −0.218 | −0.258 | 0.017 | 0.278 |
| p value | 0.096 | 0.177 | 0.529 | 0.420 | 0.202 | 0.203 | 1.009 | 0.694 | 0.553 | 0.467 | 1.000 | 0.431 | |
| Platelets | Spearman ρ | 0.769 * | 0.634 | 0.012 | 0.049 | 0.430 | 0.021 | −0.389 | −0.135 | −0.359 | 0.051 | −0.638 | −0.490 |
| p value | 0.040 | 0.050 | 0.980 | 0.899 | 0.212 | 0.957 | 0.264 | 0.708 | 0.308 | 0.889 | 0.067 | 0.154 |
| VEGF | TGF–b1 | FGFb | PDGF–bb | TNF–alfa | IL–10 | IL–6 | IL–4 | IL–1b | IL–8 | IL–1RA | IFN–Gamma | ||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| VAS (day one) | Coefficient | −0.002922 | 0.3448 | −2.08 | 0.3448 | 0.3501 | 0.0003067 | −2.669 | 0.9531 | −5.03 | 2.771 | 0.02966 | −0.1704 |
| Multiple R2 = 0.6538 | SE | 1.74609 × 1011 | 8.689 × 1014 | 1.352 × 1014 | 1.071 × 1015 | 7.551 × 1014 | 3.43 × 1010 | 7.609 × 1015 | 2.202 × 1014 | 2,514 × 1015 | 4,265 × 1015 | 7,81449 × 1012 | 1,59 × 1014 |
| 95% CI | −4.12 × 1011 to 4.12 × 1012 | −2.055 × 1015 to 2.055 × 1015 | −3.196 × 1014 to 3.196 × 1014 | −2.533 × 1015 to 2.533 × 1015 | −1.785 × 1015 to 1.785 × 1015 | −8.11 × 1010 to 8.11 × 1010 | −1.799 × 1016 to 1.799 × 1016 | −5.207 × 1014 to 5.207 × 1014 | −5.946 × 1015 to 5.946 × 1015 | −1.009 × 1016 to 1.009 × 1016 | −1.84 × 1012 to 1.84 × 1012 | −3.760 × 1014 to 3.760 × 1014 | |
| p value | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | |
| VAS (day 2) | Coefficient | 0.01323 | −0.916 | −1.279 | −0.916 | 0.8711 | 0.0001924 | −2.471 | 2.224 | −10.74 | 4.307 | 0.006912 | −0.2313 |
| Multiple R2 = 0.5957 | SE | 2.98372 × 1011 | 1.485 × 1015 | 2.31 × 1014 | 1.83 × 1015 | 1.29 × 1015 | 58,631,858,055 | 1.3 × 1016 | 3.763 × 1014 | 4.297 × 1015 | 7.288 × 1015 | 1.33534 × 1013 | 2.717 × 1014 |
| 95% CI | −70,553,7947,738 to 705,537,947,738 | −3.511 × 1015 to 3.511 × 1015 | −5.462 × 1014 to 5.462 × 1014 | −4.328 × 1015 to 4.328 × 1015 | −3.051 × 1015 to 3.051 × 1015 | −138,642,313,474 to 138642313474 | −3.074 × 1016 to 3.074 × 1016 | −8.898 × 1014 to 8.898 × 1014 | −1.016 × 1016 to 1.016 × 1016 | −1.723 × 1016 to 1.723 × 1016 | −315,75,789,661,657 to 31,575,789,661,657 | −6.424 × 1014 to 6.424 × 1014 | |
| p value | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | |
| VAS (day 3) | Coefficient | −0.008301 | −0.3741 | −2.558 | −0.3741 | 0.6477 | −0.000004932 | −4.217 | 3.077 | −9.134 | 2.942 | 0.02786 | −0.2079 |
| Multiple R2 = 0.5957 | SE | 1.78067 × 1011 | 8.862 × 1014 | 1.378 × 1014 | 1.092 × 1015 | 7.7 × 1014 | 34,991,205,207 | 7.759 × 1015 | 2.246 × 1014 | 2.564 × 1015 | 4.35 × 1015 | 7.96925 × 1012 | 1.621 × 1014 |
| 95% CI | −4.21 × 1011 to 4.21 × 1011 | −2.095 × 1015 to 2.095 × 1015 | −3.259 × 1014 to 3.259 × 1014 | −2.583 × 1015 to 2.583 × 1015 | −1.821 × 1015 to 1.821 × 1015 | −8.27 × 1010 to 8.27 × 1010 | −1.835 × 1016 to 1.835 × 1016 | −5.310 × 1014 to 5.310 × 1014 | −6.063 × 1015 to 6.063 × 1015 | −1.028 × 1016 to 1.028 × 1016 | −1.88 × 1013 to 1.88 × 1013 | −3.834 × 1014 to 3.834 × 1014 | |
| p value | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | |
| CI (day 7) | Coefficient | −0.003013 | 0.02395 | −0.06562 | 0.02394 | 0.2534 | 0.00002793 | −1.158 | 2.91 | −5.612 | −0.6447 | −0.006342 | −0.0268 |
| Multiple R2 = 0.6824 | SE | 1.25912 × 1011 | 6.266 × 1014 | 9.74692 × 1013 | 7.724 × 1014 | 5.445 × 1014 | 24742518484 | 5.487 × 1015 | 1.588 × 1014 | 1.813 × 1015 | 3.076 × 1015 | 5.63511 × 1012 | 1.147 × 1014 |
| 95% CI | −2.97 × 1011 to 2.97 × 1011 | −1.482 × 1015 to 1.482 × 1015 | −2.305 × 1014 to 2.305 × 1014 | −1.826 × 1015 to 1.826 × 1015 | −1.287 × 1015 to 1.287 × 1015 | −5.85 × 1010 to 5.85 × 1010 | −1.297 × 1016 to 1.297 × 1016 | −3.755 × 1014 to 3.755 × 1014 | −4.287 × 1015 to 4.287 × 1015 | −7.273 × 1015 to 7.273 × 1015 | −1.3 × 1013 to 1.3 × 1013 | −2.711 × 1014 to 2.711 × 1014 | |
| p value | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | |
| CI (day 14) | Coefficient | 0.002542 | −0.1547 | 0.03387 | −0.1547 | −0.226 | 0.00008631 | −0.1141 | −2.935 | 6.486 | −0.4847 | 0.009534 | 0.0623 |
| Multiple R2 = 0.6667 | SE | 1.10432 × 1011 | 5.496 × 1014 | 8.54861 × 1013 | 6.774 × 1014 | 4.775 × 1014 | 21,700,624,254 | 4.812 × 1015 | 1.393 × 1014 | 1.59 × 1015 | 2.697 × 1015 | 4.94232 × 1012 | 1.006 × 1014 |
| 95% CI | −2.61 × 1011 to 2.61 × 1011 | −1.300 × 1015 to 1.300 × 1015 | −2.021 × 1014 to 2.021 × 1014 | −1.602 × 1015 to 1.602 × 1015 | −1.129 × 1015 to 1.129 × 1015 | −5.13 × 1010 to 5.13 × 1010 | −1.138 × 1016 to 1.138 × 1016 | −3.293 × 1014 to 3.293 × 1014 | −3.760 × 1015 to 3.760 × 1015 | −6.378 × 1015 to 6.378 × 1015 | −1.16 × 1013 to 1.16 × 1013 | −2.378 × 1014 to 2.378 × 1014 | |
| p-value | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | |
| SPID 24–48 | Coefficient | −0.3877 | 30.26 | −19.21 | 30.26 | −12.5 | 0.002744 | −4.739 | −30.49 | 137 | −36.86 | 0.5459 | 1.462 |
| Multiple R2 = 0.7039 | SE | 7.68153 × 1012 | 3.823 × 1016 | 5.946 × 1015 | 4.712 × 1016 | 3.322 × 1016 | 1.50947 × 1012 | 3.347 × 1017 | 9.688 × 1015 | 1.106 × 1017 | 1.876 × 1017 | 3.438 × 1014 | 6.995 × 1015 |
| 95% CI | −1.82 × 1013 to 1.82 × 1013 | −9.039 × 1016 to 9.039 × 1016 | −1.406 × 1016 to 1.406 × 1016 | −1.114 × 1017 to 1.114 × 1017 | −7.855 × 1016 to 7.855 × 1016 | −3.56 × 1012 to 3.56 × 1012 | −7.915 × 1017 to 7.915 × 1017 | −2.291 × 1016 to 2.291 × 1016 | −2.616 × 1017 to 2.616 × 1017 | −4.437 × 1017 to 4.437 × 1017 | −8.129 × 1014 to 8.129 × 1014 | −1.654 × 1016 to 1.654 × 1016 | |
| p value | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | |
| SPID 24–72 | Coefficient | −0.2586 | 47.51 | −7.727 | 47.51 | −19.65 | 0.01022 | 32.42 | −81.46 | 235.5 | −40.96 | 0.5892 | 2.362 |
| Multiple R2 = 0.7411 | SE | 1.26831 × 1013 | 6.312 × 1016 | 9.818 × 1015 | 7.78 × 1016 | 5.485 × 1016 | 2.49231 × 1012 | 5.527 × 1017 | 1.6 × 1016 | 1.826 × 1017 | 3.098 × 1017 | 5.676 × 1014 | 1.155 × 1016 |
| 95% CI | −2.99 × 1013 to2.99 × 1013 | −1.492 × 1017 to 1.492 × 1017 | −2.322 × 1016 to 2.322 × 1016 | −1.840 × 1017 to 1.840 × 1017 | −1.297 × 1017 to 1.297 × 1017 | −5.89 × 1012 to 5.89 × 1012 | −1.307 × 1018 to 1.307 × 1018 | −3.782 × 1016 to 3.782 × 1016 | −4.319 × 1017 to 4.319 × 1017 | −7.326 × 1017 to 7.326 × 1017 | −1.342 × 1015 to 1.342 × 1015 | −2.731 × 1016 to 2.731 × 1016 | |
| p value | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | |
| SPID 24–168 | Coefficient | −0.1601 | 69.64 | −28.6 | 69.64 | −15.58 | 0.0581 | 57.51 | −133.7 | 136.4 | 116.9 | 1.578 | −4.764 |
| Multiple R2 = 0.6888 | SE | 2.91903 × 1013 | 1.453 × 1017 | 2.26 × 1016 | 1.791 × 1017 | 1.262 × 1017 | 5.73606 × 1012 | 1.272 × 1018 | 3.681 × 1016 | 4.203 × 1017 | 7.13 × 1017 | 1.306 × 1015 | 2.658 × 1016 |
| 95% CI | −6.9 × 1013 to 6.9 × 1013 | −3.435 × 1017 to 3.435 × 1017 | −5.343 × 1016 to 5.343 × 1016 | −4.234 × 1017 to 4.234 × 1017 | −2.985 × 1017 to 2.985 × 1017 | −1.35 × 1012 to 1.35 × 1012 | −3.008 × 1018 to 3.008 × 1018 | −8.705 × 1016 to 8.705 × 1016 | −9.939 × 1017 to 9.939 × 1017 | −1.686 × 1018 to 1.686 × 1018 | −3.089 × 1015 to 3.089 × 1015 | −6.285 × 1016 to 6.285 × 1016 | |
| p value |
| VEGF | TGF–b1 | FGFb | PDGF–bb | TNF–alfa | IL–10 | IL–6 | IL–4 | IL–1b | IL–8 | IL–1RA | IFN–Gamma | ||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| VAS (day one) | Coefficient | 0.02764 | −0.1627 | 0.1696 | −2.716 | −0.04526 | 0.04117 | 2.914 | −0.4218 | −3.725 | 0.8876 | 0.0283 | −0.0944 |
| Multiple R2 = 0.6538 | SE | 2.26727 × 1012 | 6.864 × 1014 | 1.84 × 1014 | 4.094 | 6.66499 × 1013 | 3.52915 × 1012 | 6.25 × 1015 | 2.292 × 1015 | 3.893 × 1015 | 2.153 × 1015 | 5.89863 × 1012 | 7.88918 × 1013 |
| 95% CI | −5.36 × 1012 to 5.36 × 1012 | −1.62 × 1015 to 1.623 × 1015 | −4.351 × 1014 to 4.351 × 1014 | −12.40 to 6.965 | −1.576 × 1014 to 1.576 × 1014 | −8.34 × 1012 to 8.34 × 1012 | −1.478 × 1016 to 1.478 × 1016 | −5.421 × 1015 to 5.421 × 1015 | −9.205 × 1015 to 9.205 × 1015 | −5.091 × 1015 to 5.091 × 1015 | −1.39 × 1012 to 1.39 × 1012 | −1.865 × 1014 to 1.865 × 1014 | |
| p value | >0.9999 | >0.9999 | >0.9999 | 0.5283 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | |
| VAS (day 2) | Coefficient | 0.001511 | 0.03459 | 2.165 | −2.875 | 0.2745 | −0.01392 | −0.4092 | −0.05736 | −0.1734 | 0.4868 | 0.00836 | −0.03179 |
| Multiple R2 = 0.5957 | SE | 2.34162 × 1012 | 7.089 × 1014 | 1.901 × 1014 | 4.175 | 6.88358 × 1013 | 3.64489 × 1012 | 6.455 × 1015 | 2.368 × 1015 | 4.02 × 1015 | 2.224 × 1015 | 6.09207 × 1012 | 8.14791 × 1013 |
| 95% CI | −5.54 × 1012 to 5.54 × 1012 | −1.676 × 1015 to 1.676 × 1015 | −4.494 × 1014 to 4.494 × 1014 | −12.75 to 6.998 | −1.628 × 1014 to 1.628 × 1014 | −8.62 × 1012 to 8.62 × 1012 | −1.526 × 1016 to 1.526 × 1016 | −5.599 × 1015 to 5.599 × 1015 | −9.507 × 1015 to 9.507 × 1015 | −5.258 × 1015 to 5.258 × 1015 | −1.44 × 1012 to 1.44 × 1012 | −1.92 × 1014 to 1.92 × 1014 | |
| p value | >0.9999 | >0.9999 | >0.9999 | 0.5133 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | |
| VAS (day 3) | Coefficient | 0.0009442 | 0.04703 | 0.2057 | −1.876 | 0.131 | −0.01446 | 0.5434 | −0.2135 | −0.776 | 0.9408 | 0.002328 | −0.03558 |
| Multiple R2 = 0.5957 | SE | 1.17081 × 1012 | 3.544 × 1014 | 9.50296 × 1013 | 1.64 | 3.44179 × 1013 | 1.82244 × 1012 | 3.228 × 1015 | 1.184 × 1015 | 2.01 × 1015 | 1.112 × 1015 | 3.04604 × 1012 | 4.07395 × 1013 |
| 95% CI | −2.77 × 1012 to 2.77 × 1012 | −8.381 × 1014 to 8.381 × 1014 | −2.247 × 1014 to 2.247 × 1014 | −5.755 to 2.003 | −8.13 × 1014 to 8.13 × 1014 | −4.30 × 1012 to 4.30 × 1012 | −7.632 × 1015 to 7.632 × 1015 | −2.799 × 1015 to 2.799 × 1015 | −4.753 × 1015 to 4.753 × 1015 | −2.629 × 1015 to 2.629 × 1015 | −7.20 × 1012 to 7.20 × 1012 | −9.63 × 1014 to 9.63 × 1014 | |
| p value | >0.9999 | >0.9999 | >0.9999 | 0.2903 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | |
| CI (day 7) | Coefficient | −0.01202 | −0.4186 | −2.129 | 1.224 | −0.1411 | 0.01456 | 4.754 | 0.4722 | −2.011 | 0.8682 | 0.0071 | −0.05192 |
| Multiple R2 = 0.6824 | SE | 1.09519 × 1012 | 3.315 × 1014 | 8.8892 × 1013 | 2.042 | 3.2195 × 1013 | 1.70474 × 1012 | 3.019 × 1015 | 1.107 × 1015 | 1.88 × 1015 | 1.04 × 1015 | 2.84931 × 1012 | 3.81083 × 1013 |
| 95% CI | −2.59 × 1012 to 2.59 × 1012 | −7.840 × 1014 to 7.840 × 1014 | −2.102 × 1014 to 2.102 × 1014 | −3.604 to 6.052 | −7.62 × 1013 to 7.62 × 1013 | −4.03 × 1012 to 4.03 × 1012 | −7.139 × 1015 to 7.139 × 1015 | −2.619 × 1015 to 2.619 × 1015 | −4.446 × 1015 to 4.446 × 1015 | −2.459 × 1015 to 2.459 × 1015 | −6.73 × 1012 to 6.730 × 1012 | −9.0 × 1013 to 9.0 × 1013 | |
| p value | >0.9999 | >0.9999 | >0.9999 | 0.567795 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | |
| CI (day 14) | Coefficient | −0.009527 | −0.2259 | 0.3207 | 0.7161 | −0.04191 | 0.0129 | 2.573 | 0.5418 | −0.2613 | −0.1487 | −0.006896 | 0.009277 |
| Multiple R2 = 0.6667 | SE | 5.85406 × 1011 | 1.772 × 1014 | 4.75148 × 1013 | 1.046 | 1.72089 × 1013 | 9.11222 × 1011 | 1.614 × 1015 | 5.919 × 1014 | 1.005 × 1015 | 5.56 × 1014 | 1.52302 × 1012 | 2.03698 × 1013 |
| 95% CI | −1.38 × 1011 to 1.38 × 1011 | −4.190 × 1014 to 4.190 × 1014 | −1.124 × 1014 to 1.124 × 1014 | −1.757 to 3.189 | −4.06 × 1013 to 4.06 × 1013 | −2.15 × 1012 to 2.15 × 1012 | −3.816 × 1015 to 3.816 × 1015 | −1.400 × 1015 to 1.400 × 1015 | −2.377 × 1015 to 2.377 × 1015 | −1.315 × 1015 to 1.315 × 1015 | −3.60 × 1012 to 3.60 × 1012 | −4.81 × 1013 to 4.81 × 1013 | |
| p value | >0.9999 | >0.9999 | >0.9999 | 0.5156 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | |
| SPID 24–48 | Coefficient | 0.627 | −4.734 | −47.89 | 3.795 | −7.674 | 1.322 | 79.75 | −8.746 | −85.24 | 9.621 | 0.4785 | −1.502 |
| Multiple R2 = 0.7039 | SE | 4.65977 × 1013 | 1.411 × 1016 | 3.782 × 1015 | 103.1 | 1.37 × 1015 | 7.25323 × 1013 | 1.285 × 1017 | 4.712 × 1016 | 8.001 × 1016 | 4.425 × 1016 | 1.212 × 1014 | 1.621 × 1015 |
| 95% CI | −1.102 × 1014 to 1.102 × 1014 | −3.336 × 1016 to 3.336 × 1016 | −8.943 × 1015 to 8.943 × 1015 | −240.1 to 247.7 | −3.239 × 1015 to 3.239 × 1015 | −1.715 × 1014 to 1.715 × 1014 | −3.038 × 1017 to 3.038 × 1017 | −1.114 × 1017 to 1.114 × 1017 | −1.892 × 1017 to 1.892 × 1017 | −1.046 × 1017 to 1.046 × 1017 | −2.867 × 1014 to 2.867 × 1014 | −3.834 × 1015 to 3.834 × 1015 | |
| p value | >0.9999 | >0.9999 | >0.9999 | 0.9717 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | |
| SPID 24–72 | Coefficient | 1.268 | −9.767 | −48.76 | −16.37 | −11.9 | 2.658 | 136.6 | −13.74 | −156 | 8.344 | 1.102 | −2.914 |
| Multiple R2 = 0.7411 | SE | 7.94774 × 1013 | 2.406 × 1016 | 6.451 × 1015 | 175.2 | 2.336 × 1015 | 1.237 × 1014 | 2.191 × 1017 | 8.036 × 1016 | 1.365 × 1017 | 7.548 × 1016 | 2.068 × 1014 | 2.765 × 1015 |
| 95% CI | −1.879 × 1014 to 1.879 × 1014 | −5.689 × 1016 to 5.689 × 1016 | −1.525 × 1016 to 1.525 × 1016 | −430.6 to 397.8 | −5.525 × 1015 to 5.525 × 1015 | −2.925 × 1014 to 2.925 × 1014 | −5.181 × 1017 to 5.181 × 1017 | −1.900 × 1017 to 1.900 × 1017 | −3.227 × 1017 to 3.227 × 1017 | −1.785 × 1017 to 1.785 × 1017 | −4.889 × 1014 to 4.889 × 1014 | −6.539 × 1015 to 6.539 × 1015 | |
| p value | >0.9999 | >0.9999 | >0.9999 | 0.9282 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | |
| SPID 24–168 | Coefficient | 3.921 | −25.38 | −32.48 | −277.1 | −16.25 | 6.61 | 416.4 | −54.23 | −513.6 | 93.56 | 3.819 | −11.98 |
| Multiple R2 = 0.6888 | SE | 2.767 × 1014 | 8.378 × 1016 | 2.246 × 1016 | 536.4 | 8.135 × 1015 | 4,308 × 1014 | 7.629 × 1017 | 2.798 × 1017 | 4.752 × 1017 | 2.628 × 1017 | 7.2 × 1014 | 9.63 × 1015 |
| 95% CI | −6.544 × 1014 to 6.544 × 1014 | −1.981 × 1017 to 1.981 × 1017 | −5.312 × 1016 to 5.312 × 1016 | −1546 to 991.3 | −1.924 × 1016 to 1.924 × 1016 | −1.019 × 1015 to 1.019 × 1015 | −1.804 × 1018 to 1.804 × 1018 | −6.617 × 1017 to 6.617 × 1017 | −1.124 × 1018 to 1.124 × 1018 | −6.215 × 1017 to 6.215 × 1017 | −1.703 × 1015 to 1.703 × 1015 | −2.277 × 1016 to 2.277 × 1016 | |
| p value | >0.9999 | >0.9999 | >0.9999 | 0.6213 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 | >0.9999 |
| Outcome Measure | VEGF | TGF–β1 | FGFb | PDGF–bb | TNF–α | IL–10 | IL–6 | IL–4 | IL–1β | IL–8 | IL–1RA | IFN–γ | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Clot | |||||||||||||
| VAS (day 1) | Spearman ρ | −0.013 | 0.317 | 0.299 | 0.317 | −0.316 | 0.068 | −0.273 | −0.209 | −0.050 | −0.387 | 0.046 | −0.300 |
| p value | 0.957 | 0.173 | 0.200 | 0.173 | 0.175 | 0.776 | 0.244 | 0.378 | 0.836 | 0.092 | 0.847 | 0.199 | |
| VAS (day 2) | Spearman ρ | 0.074 | 0.042 | 0.107 | 0.042 | −0.274 | −0.107 | −0.191 | −0.152 | −0.044 | −0.055 | −0.133 | −0.299 |
| p value | 0.758 | 0.859 | 0.655 | 0.859 | 0.243 | 0.653 | 0.421 | 0.521 | 0.853 | 0.819 | 0.576 | 0.200 | |
| VAS (day 3) | Spearman ρ | −0.020 | −0.127 | −0.077 | −0.127 | −0.268 | −0.069 | −0.037 | −0.002 | 0.191 | −0.008 | 0.008 | −0.292 |
| p value | 0.933 | 0.594 | 0.748 | 0.594 | 0.254 | 0.773 | 0.876 | 0.994 | 0.420 | 0.975 | 0.972 | 0.211 | |
| CI (day 7) | Spearman ρ | −0.182 | −0.045 | −0.010 | −0.045 | 0.234 | 0.222 | −0.036 | −0.027 | −0.197 | −0.146 | 0.098 | 0.234 |
| p value | 0.443 | 0.850 | 0.967 | 0.850 | 0.321 | 0.346 | 0.882 | 0.910 | 0.405 | 0.540 | 0.682 | 0.321 | |
| CI (day 14) | Spearman ρ | −0.349 | 0.091 | 0.172 | 0.091 | 0.404 | 0.326 | 0.163 | 0.057 | 0.089 | −0.076 | 0.442 | 0.366 |
| p value | 0.132 | 0.703 | 0.469 | 0.703 | 0.077 | 0.161 | 0.492 | 0.810 | 0.710 | 0.752 | 0.051 | 0.113 | |
| SPID24−48 | Spearman ρ | −0.136 | 0.116 | 0.105 | 0.116 | −0.002 | 0.146 | −0.031 | −0.060 | 0.021 | −0.194 | 0.090 | −0.017 |
| p value | 0.567 | 0.626 | 0.659 | 0.626 | 0.992 | 0.538 | 0.895 | 0.803 | 0.930 | 0.413 | 0.707 | 0.942 | |
| SPID24-72 | Spearman ρ | −0.096 | 0.214 | 0.212 | 0.214 | −0.081 | 0.143 | −0.129 | −0.144 | −0.055 | −0.260 | 0.035 | −0.095 |
| p value | 0.686 | 0.365 | 0.368 | 0.365 | 0.734 | 0.547 | 0.588 | 0.545 | 0.819 | 0.268 | 0.884 | 0.691 | |
| SPID24-196 | Spearman ρ | −0.159 | 0.276 | 0.320 | 0.276 | −0.062 | 0.197 | −0.162 | −0.191 | −0.065 | −0.299 | 0.120 | −0.086 |
| p value | 0.502 | 0.238 | 0.168 | 0.238 | 0.797 | 0.405 | 0.495 | 0.421 | 0.784 | 0.200 | 0.613 | 0.720 | |
| L-PRF | |||||||||||||
| VAS (day 1) | Spearman ρ | −0.228 | −0.122 | 0.147 | −0.403 | −0.199 | 0.278 | −0.300 | −0.250 | −0.138 | −0.379 | 0.031 | −0.240 |
| p value | 0.333 | 0.609 | 0.536 | 0.078 | 0.400 | 0.235 | 0.198 | 0.288 | 0.560 | 0.100 | 0.896 | 0.309 | |
| VAS (day 2) | Spearman ρ | −0.050 | −0.243 | −0.302 | −0.524 * | −0.154 | 0.207 | −0.165 | −0.138 | 0.093 | −0.223 | 0.072 | −0.301 |
| p value | 0.833 | 0.301 | 0.195 | 0.018 | 0.516 | 0.381 | 0.488 | 0.563 | 0.697 | 0.344 | 0.763 | 0.198 | |
| VAS (day 3) | Spearman ρ | 0.120 | −0.208 | −0.479 * | −0.709 * | −0.091 | −0.003 | −0.249 | −0.216 | −0.135 | −0.215 | 0.013 | −0.172 |
| p value | 0.615 | 0.378 | 0.033 | 0.009 | 0.704 | 0.989 | 0.289 | 0.360 | 0.570 | 0.363 | 0.957 | 0.468 | |
| CI (day 7) | Spearman ρ | −0.171 | 0.202 | −0.328 | 0.516 * | 0.040 | −0.105 | 0.106 | 0.042 | −0.067 | 0.025 | 0.042 | 0.247 |
| p value | 0.470 | 0.392 | 0.159 | 0.020 | 0.868 | 0.660 | 0.655 | 0.861 | 0.778 | 0.915 | 0.862 | 0.294 | |
| CI (day 14) | Spearman ρ | −0.348 | 0.362 | −0.174 | 0.522 * | −0.150 | −0.098 | 0.309 | 0.117 | 0.214 | 0.261 | 0.166 | 0.261 |
| p value | 0.133 | 0.117 | 0.463 | 0.018 | 0.529 | 0.681 | 0.184 | 0.622 | 0.365 | 0.265 | 0.485 | 0.265 | |
| SPID24-48 | Spearman ρ | −0.229 | 0.134 | −0.133 | 0.078 | −0.185 | 0.073 | −0.069 | −0.086 | −0.104 | −0.153 | 0.076 | 0.068 |
| p-value | 0.332 | 0.572 | 0.575 | 0.743 | 0.434 | 0.759 | 0.774 | 0.719 | 0.662 | 0.520 | 0.750 | 0.776 | |
| SPID24-72 | Spearman ρ | −0.261 | 0.043 | −0.110 | −0.003 | −0.174 | 0.204 | −0.133 | −0.112 | −0.080 | −0.250 | 0.076 | −0.053 |
| p value | 0.267 | 0.856 | 0.646 | 0.990 | 0.464 | 0.388 | 0.575 | 0.637 | 0.738 | 0.287 | 0.750 | 0.824 | |
| SPID24-196 | Spearman ρ | −0.258 | −0.083 | 0.073 | −0.288 | −0.179 | 0.277 | −0.309 | -0.256 | −0.188 | −0.382 | −0.008 | −0.215 |
| p value | 0.273 | 0.727 | 0.759 | 0.218 | 0.451 | 0.238 | 0.185 | 0.275 | 0.428 | 0.096 | 0.975 | 0.363 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
da Silva, M.T.; de Almeida Barros Mourão, C.F.; Mello-Machado, R.C.; Montemezzi, P.; de Lima Barbosa, R.; Sartoretto, S.C.; Leite, P.E.C.; Javid, K.; Kawase, T.; Alves, G.G.; et al. Effects of Leukocyte-Platelet-Rich Fibrin (L–PRF) on Pain, Soft Tissue Healing, Growth Factors, and Cytokines after Third Molar Extraction: A Randomized, Split-Mouth, Double-Blinded Clinical Trial. Appl. Sci. 2021, 11, 1666. https://doi.org/10.3390/app11041666
da Silva MT, de Almeida Barros Mourão CF, Mello-Machado RC, Montemezzi P, de Lima Barbosa R, Sartoretto SC, Leite PEC, Javid K, Kawase T, Alves GG, et al. Effects of Leukocyte-Platelet-Rich Fibrin (L–PRF) on Pain, Soft Tissue Healing, Growth Factors, and Cytokines after Third Molar Extraction: A Randomized, Split-Mouth, Double-Blinded Clinical Trial. Applied Sciences. 2021; 11(4):1666. https://doi.org/10.3390/app11041666
Chicago/Turabian Styleda Silva, Madelaine Torres, Carlos Fernando de Almeida Barros Mourão, Rafael Coutinho Mello-Machado, Pietro Montemezzi, Renata de Lima Barbosa, Suelen Cristina Sartoretto, Paulo Emílio Correa Leite, Kayvon Javid, Tomoyuki Kawase, Gutemberg Gomes Alves, and et al. 2021. "Effects of Leukocyte-Platelet-Rich Fibrin (L–PRF) on Pain, Soft Tissue Healing, Growth Factors, and Cytokines after Third Molar Extraction: A Randomized, Split-Mouth, Double-Blinded Clinical Trial" Applied Sciences 11, no. 4: 1666. https://doi.org/10.3390/app11041666
APA Styleda Silva, M. T., de Almeida Barros Mourão, C. F., Mello-Machado, R. C., Montemezzi, P., de Lima Barbosa, R., Sartoretto, S. C., Leite, P. E. C., Javid, K., Kawase, T., Alves, G. G., & Calasans-Maia, M. D. (2021). Effects of Leukocyte-Platelet-Rich Fibrin (L–PRF) on Pain, Soft Tissue Healing, Growth Factors, and Cytokines after Third Molar Extraction: A Randomized, Split-Mouth, Double-Blinded Clinical Trial. Applied Sciences, 11(4), 1666. https://doi.org/10.3390/app11041666









