A Systematic Review of the Effects of Football Playing on Changes in Serum Brain-Derived Neurotrophic Factor Level
Abstract
1. Introduction
2. Materials and Methods
2.1. Design
2.2. Eligibility Criteria
2.3. Information Sources and Search
2.4. Data Extraction
2.5. Data Items
2.6. Methodological Assessment
3. Results
3.1. Study Identification and Selection
3.2. Methodological Quality
3.3. Characteristics of Individual Studies
3.4. Main Outcomes of Studies
4. Discussion
Guidelines for Future Research
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Szuhany, K.L.; Bugatti, M.; Otto, M.W. A Meta-Analytic Review of the Effects of Exercise on Brain-Derived Neurotrophic Factor. J. Psychiatr. Res. 2015, 60, 56–64. [Google Scholar] [CrossRef]
- Jiménez-Maldonado, A.; Rentería, I.; García-Suárez, P.C.; Moncada-Jiménez, J.; Freire-Royes, L.F. The Impact of High-Intensity Interval Train. on Brain Derived Neurotrophic Factor in Brain: A Mini-Review. Front. Neurosci. 2018, 12, 1–9. [Google Scholar] [CrossRef]
- Bathina, S.; Das, U.N. Brain-Derived Neurotrophic Factor and Its Clinical Implications. Arch. Med. Sci. 2015, 11, 1164–1178. [Google Scholar] [CrossRef]
- Fernandes, J.; Arida, R.M.; Gomez-Pinilla, F. Physical Exercise as an Epigenetic Modulator of Brain Plasticity and Cognition. Neurosci. Biobehav. Rev. 2017, 80, 443–456. [Google Scholar] [CrossRef]
- Tapia-Arancibia, L.; Aliaga, E.; Silhol, M.; Arancibia, S. New Insights into Brain BDNF Function in Normal Aging and Alzheimer Disease. Brain Res. Rev. 2008, 59, 201–220. [Google Scholar] [CrossRef] [PubMed]
- Babaei, P.; Damirchi, A.; Mehdipoor, M.; Tehrani, B.S. Long Term Habitual Exercise Is Associated with Lower Resting Level of Serum BDNF. Neurosci. Lett. 2014, 566, 304–308. [Google Scholar] [CrossRef]
- Nofuji, Y.; Suwa, M.; Moriyama, Y.; Nakano, H.; Ichimiya, A.; Nishichi, R.; Sasaki, H.; Radak, Z.; Kumagai, S. Decreased Serum Brain-Derived Neurotrophic Factor in Trained Men. Neurosci. Lett. 2008, 437, 29–32. [Google Scholar] [CrossRef] [PubMed]
- Zoladz, J.A.; Pilc, A.; Majerczak, J.; Grandys, M.; Zapart-Bukowska, J.; Duda, K. Endurance Training Increases Plasma Brain-Derived Neurotrophic Factor Concentration in Young Healthy Men. J. Physiol. Pharmacol. 2008, 59 (Suppl. 7), 119–132. [Google Scholar]
- Radak, Z.; Suzuki, K.; Higuchi, M.; Balogh, L.; Boldogh, I.; Koltai, E. Physical Exercise, Reactive Oxygen Species and Neuroprotection. Free Radic. Biol. Med. 2016, 98, 187–196. [Google Scholar] [CrossRef] [PubMed]
- Kahlert, S.; Zündorf, G.; Reiser, G. Glutamate-Mediated Influx of Extracellular Ca2+ Is Coupled with Reactive Oxygen Species Generation in Cultured Hippocampal Neurons but Not in Astrocytes. J. Neurosci. Res. 2005, 79, 262–271. [Google Scholar] [CrossRef] [PubMed]
- Bergersen, L.H. Lactate Transport and Signaling in the Brain: Potential Therapeutic Targets and Roles in Body—Brain Interaction. J. Cereb. Blood Flow Metab. 2015, 35, 176–185. [Google Scholar] [CrossRef] [PubMed]
- Knaepen, K.; Goekint, M.; Heyman, E.M.; Meeusen, R. Neuroplasticity—Exercise-Induced Response of Peripheral Brain-Derived Neurotrophic Factor: A Systematic Review of Experimental Studies in Human Subjects. Sports Med. 2010, 40, 765–801. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, A.C.; Lasmar, R.P.; Caramelli, P. Effects of Soccer Heading on Brain Structure and Function. Front Neurol. 2016, 7, 38. [Google Scholar] [CrossRef]
- Hashimoto, T.; Fukui, K.; Takeuchi, H.; Yokota, S.; Kikuchi, Y.; Tomita, H.; Taki, Y.; Kawashima, R. Effects of the BDNF Val66Met Polymorphism on Gray Matter Volume in Typically Developing Children and Adolescents. Cerebral Cortex 2016, 26, 1795–1803. [Google Scholar] [CrossRef]
- Cabral-Santos, C.; Castrillón, C.I.M.; Miranda, R.A.T.; Monteiro, P.A.; Inoue, D.S.; Campos, E.Z.; Hofmann, P.; Lira, F.S. Inflammatory Cytokines and BDNF Response to High-Intensity Intermittent Exercise: Effect the Exercise Volume. Front. Physiol. 2016, 7, 1–8. [Google Scholar] [CrossRef]
- Saucedo Marquez, C.M.; Vanaudenaerde, B.; Troosters, T.; Wenderoth, N. High-Intensity Interval Training Evokes Larger Serum BDNF Levels Compared with Intense Continuous Exercise. J. Appl. Physiol. 2015, 119, 1363–1373. [Google Scholar] [CrossRef] [PubMed]
- Heyman, E.; Gamelin, F.-X.; Goekint, M.; Piscitelli, F.; Roelands, B.; Leclair, E.; Di Marzo, V.; Meeusen, R. Intense Exercise Increases Circulating Endocannabinoid and BDNF Levels in Humans—Possible Implications for Reward and Depression. Psychoneuroendocrinology 2012, 37, 844–851. [Google Scholar] [CrossRef] [PubMed]
- Rojas-Valverde, D. Potential Role of Cannabidiol (CBD) on Sport Recovery: A Narrative Review. Front. Physiol. 2021, 12, 722550. [Google Scholar] [CrossRef]
- Iaia, F.M.; Ermanno, R.; Bangsbo, J. High-Intensity Training in Football. Int. J. Sports Physiol. Perform. 2009, 4, 291–306. [Google Scholar] [CrossRef]
- Schimpchen, J.; Gopaladesikan, S.; Meyer, T. The Intermittent Nature of Player Physical Output in Professional Football Matches: An Analysis of Sequences of Peak Intensity and Associated Fatigue Responses. Eur. J. Sport Sci. 2021, 21, 793–802. [Google Scholar] [CrossRef]
- Oliva-Lozano, J.M.; Rojas-Valverde, D.; Gómez-Carmona, C.D.; Fortes, V.; Pino-Ortega, J. Impact of Contextual Variables on The Representative External Load Profile of Spanish Professional Soccer Match-Play: A Full Season Study. Eur. J. Sport Sci. 2020, 21, 497–506. [Google Scholar] [CrossRef] [PubMed]
- Junge, A.; Dvorak, J.; Rösch, D.; Graf-Baumann, T.; Chomiak, J.; Peterson, L. Psychological and Sport-Specific Characteristics of Football Players. Am. J. Sports Med. 2000, 28, S22–S28. [Google Scholar] [CrossRef]
- Terpstra, A.R.; Vasquez, B.P.; Colella, B.; Tartaglia, M.C.; Tator, C.H.; Mikulis, D.; Davis, K.D.; Wennberg, R.; Green, R.E.A. Comprehensive Neuropsychiatric and Cognitive Characterization of Former Professional Football Players: Implications for Neurorehabilitation. Front. Neurol. 2019, 10, 712. [Google Scholar] [CrossRef] [PubMed]
- Piepmeier, A.T.; Etnier, J.L. Brain-Derived Neurotrophic Factor (BDNF) as a Potential Mechanism of the Effects of Acute Exercise on Cognitive Performance. J. Sport Health Sci. 2015, 4, 14–23. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 Statement: An Updated Guideline for Reporting Systematic Reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]
- Rico-González, M.; Pino-Ortega, J.; Clemente, F.; Los Arcos, A. Guidelines for Performing Systematic Reviews in Sports Science. Biol. Sport 2022, 39, 463–471. [Google Scholar] [CrossRef]
- Khan, K.S.; Kunz, R.; Kleijnen, J.; Antes, G. Five Steps to Conducting a Systematic Review. J. R. Soc. Med. 2003, 96, 118–121. [Google Scholar] [CrossRef]
- Fernández-Rodríguez, R.; Álvarez-Bueno, C.; Martínez-Ortega, I.A.; Martínez-Vizcaíno, V.; Mesas, A.E.; Notario-Pacheco, B. Immediate Effect of High-Intensity Exercise on Brain-Derived Neurotrophic Factor in Healthy Young Adults: A Systematic Review and Meta-Analysis. J. Sport Health Sci. 2021, in press. [Google Scholar] [CrossRef]
- de Azevedo, K.P.M.; de Oliveira, V.H.; de Medeiros, G.C.B.S.; de Sousa Mata, Á.N.; García, D.Á.; Martínez, D.G.; Leitão, J.C.; Knackfuss, M.I.; Piuvezam, G. The Effects of Exercise on BDNF Levels in Adolescents: A Systematic Review with Meta-Analysis. Int. J. Environ. Res. Public Health 2020, 17, 6056. [Google Scholar] [CrossRef]
- Prictor, M.; Hill, S. Cochrane Consumers and Communication Review Group: Leading the Field on Health Communication Evidence. J. Evid.-Based Med. 2013, 6, 216–220. [Google Scholar] [CrossRef] [PubMed]
- de Azevedo, K.P.M.; de Oliveira Segundo, V.H.; de Medeiros, G.C.B.S.; de Sousa Mata, Á.N.; García, D.Á.; de Carvalho Leitão, J.C.G.; Knackfuss, M.I.; Piuvezam, G. Effects of Exercise on the Levels of BDNF and Executive Function in Adolescents. Medicine 2019, 98, e16445. [Google Scholar] [CrossRef]
- O’Reilly, M.; Caulfield, B.; Ward, T.; Johnston, W.; Doherty, C. Wearable Inertial Sensor Systems for Lower Limb Exercise Detection and Evaluation: A Systematic Review. Sports Med. 2018, 48, 1221–1246. [Google Scholar] [CrossRef]
- Murtagh, C.F.; Brownlee, T.E.; Rienzi, E.; Roquero, S.; Moreno, S.; Huertas, G.; Lugioratto, G.; Baumert, P.; Turner, D.C.; Lee, D.; et al. The Genetic Profile of Elite Youth Soccer Players and Its Association with Power and Speed Depends on Maturity Status. PLoS ONE 2020, 15, e0234458. [Google Scholar] [CrossRef]
- Bamaç, B.; Tamer, G.S.; Colak, T.; Colak, E.; Seyrek, E.; Duman, C.; Colak, S.; Ozbek, A. Effects of repeatedly heading a soccer ball on serum levels of two neurotrophic factors of brain tissue, BDNF and NGF, in professional soccer players. Biol. Sport 2011, 28, 177–181. [Google Scholar] [CrossRef]
- Roh, H.-T.; So, W.-Y.; Cho, S.-Y.; Suh, S.-H. Effects of Fluid Ingestion on Brain-Derived Neurotrophic Factor and Cognition during Exercise in the Heat. J. Hum. Kinet. 2017, 58, 73–86. [Google Scholar] [CrossRef][Green Version]
- Yang, J.S.; Yoo, S.H.; Cho, S.Y.; Roh, H.T. Effects of Acute Soccer Game on Serum Levels of Neurotrophins and Neurocognitive Functions in Male Adolescents. J. Life Sci. 2012, 22, 1444–1450. [Google Scholar] [CrossRef]
- Williams, R.A.; Cooper, S.B.; Dring, K.J.; Hatch, L.; Morris, J.G.; Sunderland, C.; Nevill, M.E. Effect of Football Activity and Physical Fitness on Information Processing, Inhibitory Control and Working Memory in Adolescents. BMC Public Health 2020, 20, 1398. [Google Scholar] [CrossRef] [PubMed]
- Hunter, L.E.; Freudenberg-Hua, Y.; Davies, P.; Kim, M.; Fleysher, R.; Stewart, W.F.; Lipton, R.B.; Lipton, M.L. BDNF Val66Met Positive Players Demonstrate Diffusion Tensor Imaging Consistent with Impaired Myelination Associated with High Levels of Soccer Heading: Indication of a Potential Gene-Environment Interaction Mechanism. Front. Neurol. 2019, 10, 1297. [Google Scholar] [CrossRef]
- Yang, C.; Li, Y.; Du, M.; Chen, Z. Recent Advances in Ultrasound-Triggered Therapy. J. Drug Target 2019, 27, 33–50. [Google Scholar] [CrossRef]
- Dinoff, A.; Herrmann, N.; Swardfager, W.; Lanctôt, K.L. The Effect of Acute Exercise on Blood Concentrations of Brain-Derived Neurotrophic Factor in Healthy Adults: A Meta-Analysis. Eur. J. Neurosci. 2017, 46, 1635–1646. [Google Scholar] [CrossRef]
- Feter, N.; Alt, R.; Dias, M.G.; Rombaldi, A.J. How Do Different Physical Exercise Parameters Modulate Brain-Derived Neurotrophic Factor in Healthy and Non-Healthy Adults? A Systematic Review, Meta-Analysis and Meta-Regression. Sci. Sports 2019, 34, 293–304. [Google Scholar] [CrossRef]
- Rasmussen, P.; Brassard, P.; Adser, H.; Pedersen, M.V.; Leick, L.; Hart, E.; Secher, N.H.; Pedersen, B.K.; Pilegaard, H. Evidence for a Release of Brain-Derived Neurotrophic Factor from the Brain during Exercise. Exp. Physiol. 2009, 94, 1062–1069. [Google Scholar] [CrossRef] [PubMed]
- Schiffer, T.; Schulte, S.; Sperlich, B.; Achtzehn, S.; Fricke, H.; Strüder, H.K. Lactate Infusion at Rest Increases BDNF Blood Concentration in Humans. Neurosci. Lett. 2011, 488, 234–237. [Google Scholar] [CrossRef] [PubMed]
- Deveci, S.Ş.; Matur, Z.; Kesim, Y.Y.; Senturk, G.G.; Sargın-Kurt, G.G.; Ugur, S.A.; Öge, A.E. Effect of the Brain-Derived Neurotrophic Factor Gene Val66Met Polymorphism on Sensory-Motor Integration during a Complex Motor Learning Exercise. Brain Res. 2020, 1732, 146652. [Google Scholar] [CrossRef]

| Item | Inclusion Criteria | Exclusion Criteria |
|---|---|---|
| Population | Soccer players of any age and sex without injury or illness reported | Athletes from other sports (basketball, futsal, etc.) |
| Intervention/exposure | Measures were performed during soccer training or competition | Measures were performed in another context |
| Comparison/control | Not necessary | - |
| Outcome | Values related to BDNF were reported | No BDNF-related values were provided |
| Study design | - | - |
| Other | Only original and full-text studies written in English | Written in a language other than English. Article types other than the original (e.g., reviews, letters to editors, trial registrations, proposals for protocols, editorials, book chapters, and conference abstracts). |
| Reference | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | Quality |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Murtagh et al. [33] | X | X | X | X | X | X | X | X | X | X | 10 |
| Babaei et al. [6] | X | X | X | - | X | X | X | - | X | - | 7 |
| Bamaç et al. [34] | X | X | X | X | X | X | X | X | X | - | 9 |
| Roh et al. [35] | X | X | X | X | X | X | X | X | X | - | 9 |
| Yang, Jung-Su et al. [36] | X | X | X | X | X | X | X | - | X | - | 8 |
| Williams et al. [37] | X | X | X | X | X | X | X | X | X | X | 10 |
| Hunter et al. [38] | X | X | X | X | X | X | X | X | X | X | 10 |
| Ref. | Aim | Sample | Design | Methodological Information | Output Variables | Main Outcomes |
|---|---|---|---|---|---|---|
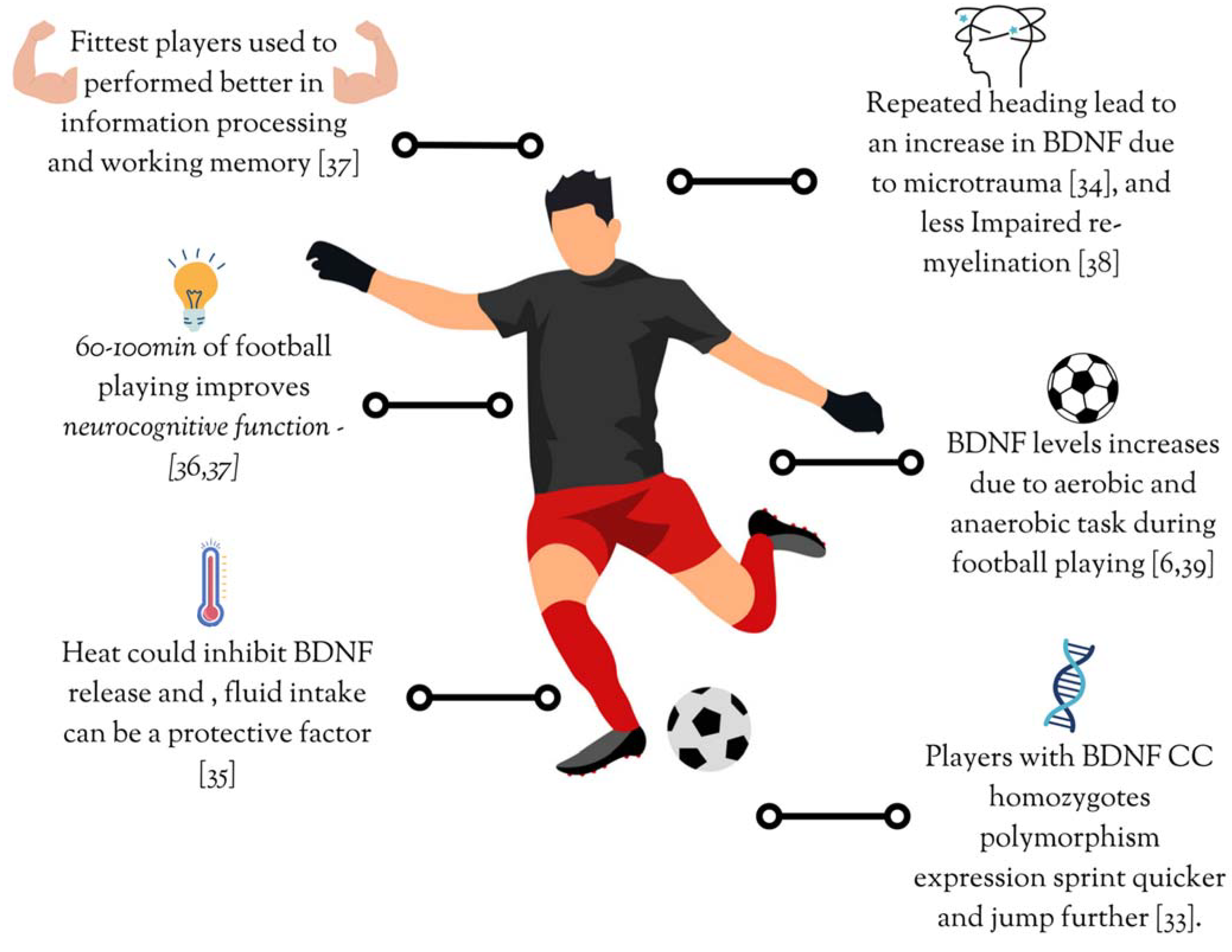
| Murtagh et al. [33] | Association of multiple single nucleotide polymorphisms (SNPs) with athlete status and power/speed performance in elite male youth football players (ESP) and control participants (CON) at different stages of maturity | 535 elite football players (8–23 years) and 151 healthy adults (9–26 years) | Cross-sectional comparative | Relationship between allele and genotype frequency distribution and the field tests performance | Genotype and allele frequency distribution Bilateral vertical and horizontal countermovement jump 10–20 m Sprints 505 Agility test | Elite football players presented higher ACTN3 (XX), PPARA (C allele), AGT (GG), and NOS3 (T allele) frequency distribution. Football players with BDNF (CC) homozygotes sprint quicker and jump farther than players who are A allele and T allele carriers. |
| Babaei et al. [6] | Evaluate the basal BDNF level and memory performance, also the responsiveness of BDNF regulation system to acute aerobic and anaerobic training in athletes and sedentary groups | Study 1: 25 Master football players (45–65 years) and 22 sedentary adults Study 2: 19 Master football players (45–65 years) and 20 sedentary adults | Study 1: Cross-sectional comparative Study 2: Quasi-experimental | Study 1 (Longitudinal comparative): Basal serum BDNF, platelets, and memory performance comparison between groups Study 2: Test results change after a single bout of aerobic (shuttle run, n = 20) and anaerobic n = 19, Rast test exercise | BDNF Platelets Memory task | Study 1: Athletes had higher basal BDNF levels, better memory. Study 2: An increase in BDNF level after aerobic and anaerobic tasks in both groups |
| Bamaç et al. [34] | Determine the effects of heading training on serum nerve growth factor (NGF) and BDNF levels in football players | 17 Professional male football players (age 24 ± 4.4 years) | Cross-sectional comparative | Players performed 15 headings after corner kicks (30–35 m far) in about 20–25 min | BDNF NGF | BDNF and NGF levels increased after repeated heading training due to microtrauma |
| Roh et al. [35] | Investigate the effects of fluid ingestion during exercise in different environments on the serum brain-derived neurotrophic factor and cognition among athletes | Ten collegiate male football and rugby players | Cross-sectional comparative | Players performed running tests (60 min each) in four conditions: a. Thermoneutral temperature at 18 °C b. High ambient temperature at 32 °C without fluid ingestion c. High ambient temperature at 32 °C with water ingestion d. High ambient temperature at 32 °C with sports drink ingestion | BDNF Stroop Colour and Word Test scores IGF-1 Plasma f-TRP | Running performed in a thermoneutral environment improved cognitive function and presented higher BDNF levels than those performed in hot environments. Liquid intake may counteract the negative heat effects on BDNF exertion. |
| Yang, Jung-Su et al. [36] | Investigate the effects of acute football match on serum levels of neurotrophins and neurocognitive function | 15 Healthy male adolescents | Cross-sectional comparative | Participants were evaluated three times: before, after treatment and two hours after treatment; the two treatments (100 min) were football match vs. self-study | BDNF NGF IGF-1 Stroop Colour and Word Test | Stroop test scores, BDNF, NGF, and IGF-1 increased after the football match and were higher than self-study conditions |
| Williams et al. [37] | Examine the effect of an acute bout of outdoor football activity on information processing, inhibitory control, working memory, and circulating BDNF in adolescents | 36 Adolescents (16 girls) | Counterbalanced crossover | Two conditions were 60 min football session and 60 min sitting | BDNF levels Stroop Test Sternberg Paradigm | BDNF level was not affected by football activity or physical fitness The high-fit group presented better Stroop task and Sternberg paradigm results |
| Hunter et al. [38] | Examine the potential effect modifying role of the BDNF Val66Met polymorphism on the association of football heading with white matter microstructure | 312 Football players | Longitudinal follow-up comparative | Met allele expression and reported sub-concussive heading in football was assessed throughout two years | BDNF Val66Met polymorphism Self-reported total heading in the prior 12 months Diffusion tensor imaging | BDNF Val66Met (+) football players with long-term exposure to high levels of heading exhibit less low radial diffusivity (impaired remyelination) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gutiérrez-Vargas, R.; Ugalde-Ramírez, A.; Rico-González, M.; Pino-Ortega, J.; González-Hernández, J.; Rojas-Valverde, D. A Systematic Review of the Effects of Football Playing on Changes in Serum Brain-Derived Neurotrophic Factor Level. Appl. Sci. 2021, 11, 11828. https://doi.org/10.3390/app112411828
Gutiérrez-Vargas R, Ugalde-Ramírez A, Rico-González M, Pino-Ortega J, González-Hernández J, Rojas-Valverde D. A Systematic Review of the Effects of Football Playing on Changes in Serum Brain-Derived Neurotrophic Factor Level. Applied Sciences. 2021; 11(24):11828. https://doi.org/10.3390/app112411828
Chicago/Turabian StyleGutiérrez-Vargas, Randall, Alexis Ugalde-Ramírez, Markel Rico-González, José Pino-Ortega, Juan González-Hernández, and Daniel Rojas-Valverde. 2021. "A Systematic Review of the Effects of Football Playing on Changes in Serum Brain-Derived Neurotrophic Factor Level" Applied Sciences 11, no. 24: 11828. https://doi.org/10.3390/app112411828
APA StyleGutiérrez-Vargas, R., Ugalde-Ramírez, A., Rico-González, M., Pino-Ortega, J., González-Hernández, J., & Rojas-Valverde, D. (2021). A Systematic Review of the Effects of Football Playing on Changes in Serum Brain-Derived Neurotrophic Factor Level. Applied Sciences, 11(24), 11828. https://doi.org/10.3390/app112411828










