Application in Nanomedicine of Manganese-Zinc Ferrite Nanoparticles
Abstract
:1. Introduction
2. Synthetic Strategies of Mn-Zn Ferrites
2.1. Co-Precipitation Method
2.2. Thermal Decomposition Method
2.3. Microemulsion Method
2.4. Hydrothermal Method
2.5. Sol-Gel Method
3. In Vitro Applications of Mn-Zn Ferrites
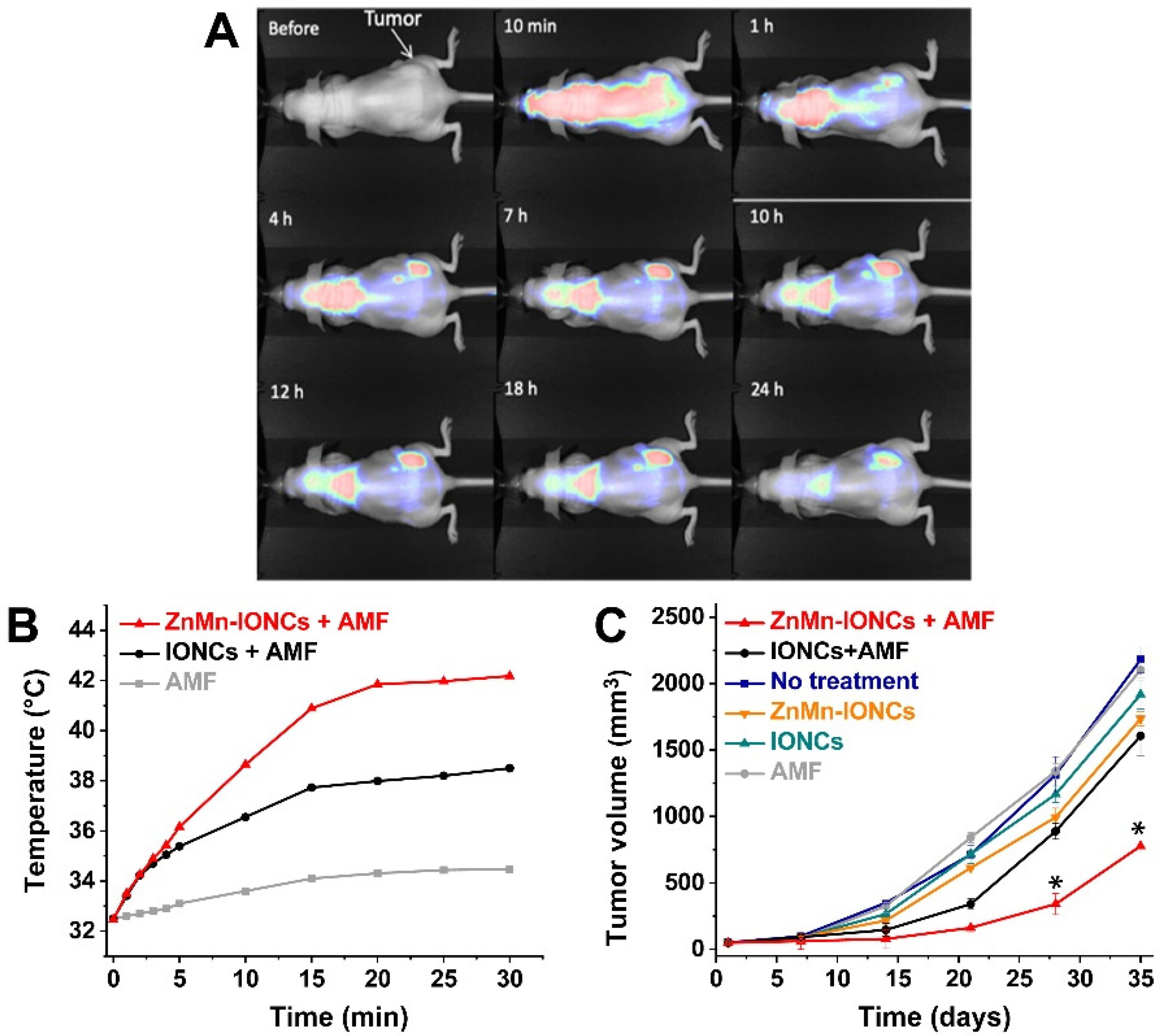
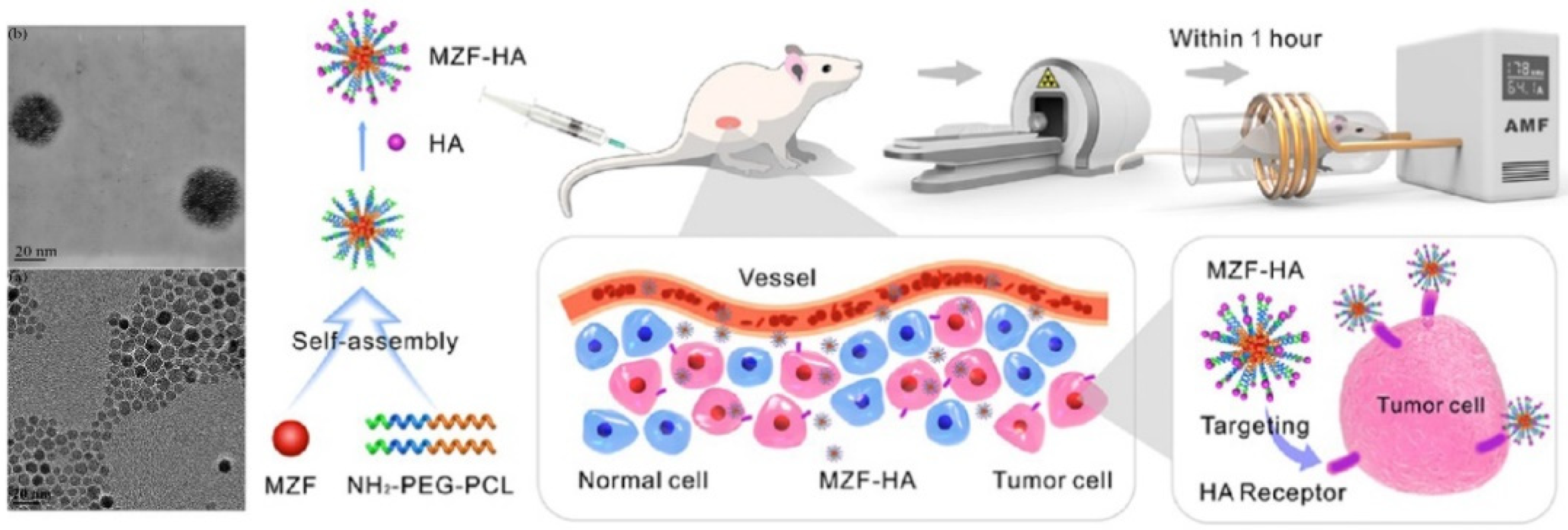
4. In Vivo Applications of Mn-Zn Ferrites
5. Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Doane, T.L.; Burda, C. The unique role of nanoparticles in nanomedicine: Imaging, drug delivery and therapy. Chem. Soc. Rev. 2012, 41, 2885–2911. [Google Scholar] [CrossRef]
- Wong, X.Y.; Sena-Torralba, A.; Alvarez-Diduk, R.; Muthoosamy, K.; Merkoci, A. Nanomaterials for nanotheranostics: Tuning their properties according to disease needs. ACS Nano 2020, 14, 2585–2627. [Google Scholar] [CrossRef]
- Lin, X.M.; Samia, A.C.S. Synthesis, assembly and physical properties of magnetic nanoparticles. J. Magn. Magn. Mater. 2006, 305, 100–109. [Google Scholar] [CrossRef]
- Zhang, Y.; Kohler, N.; Zhang, M.Q. Surface modification of superparamagnetic magnetite nanoparticles and their intracellular uptake. Biomaterials 2002, 23, 1553–1561. [Google Scholar] [CrossRef]
- Figuerola, A.; Di Corato, R.; Manna, L.; Pellegrino, T. From iron oxide nanoparticles towards advanced iron-based inorganic materials designed for biomedical applications. Pharmacol. Res. 2010, 62, 126–143. [Google Scholar] [CrossRef]
- Gobbo, O.L.; Sjaastad, K.; Radomski, M.W.; Volkov, Y.; Prina-Mello, A. Magnetic nanoparticles in cancer theranostics. Theranostics 2015, 5, 1249–1263. [Google Scholar] [CrossRef] [PubMed]
- Dadfar, S.M.; Roemhild, K.; Drude, N.I.; von Stillfried, S.; Knuchel, R.; Kiessling, F.; Lammers, T. Iron oxide nanoparticles: Diagnostic, therapeutic and theranostic applications. Adv. Drug Deliv. Rev. 2019, 138, 302–325. [Google Scholar] [CrossRef] [PubMed]
- Doaga, A.; Cojocariu, A.M.; Amin, W.; Heib, F.; Bender, P.; Hempelmann, R.; Caltun, O.F. Synthesis and characterizations of manganese ferrites for hyperthermia applications. Mater. Chem. Phys. 2013, 143, 305–310. [Google Scholar] [CrossRef]
- Andersen, H.L.; Saura-Muzquiz, M.; Granados-Miralles, C.; Canevet, E.; Lock, N.; Christensen, M. Crystalline and magnetic structure-property relationship in spinel ferrite nanoparticles. Nanoscale 2018, 10, 14902–14914. [Google Scholar] [CrossRef] [Green Version]
- Gazeau, F.; Levy, M.; Wilhelm, C. Optimizing magnetic nanoparticle design for nanothermotherapy. Nanomedicine 2008, 3, 831–844. [Google Scholar] [CrossRef]
- Bante-Guerra, J.; Macias, J.D.; Caballero-Aguilar, L.; Vales-Pinzon, C.; Alvarado-Gil, J.J. Infrared thermography analysis of thermal diffusion induced by RF magnetic field on agar phantoms loaded with magnetic nanoparticles. In Proceedings of the SPIE Conference on Energy-Based Treatment of Tissue and Assessment VII, San Francisco, CA, USA, 3–4 February 2013. [Google Scholar]
- Jang, J.T.; Nah, H.; Lee, J.H.; Moon, S.H.; Kim, M.G.; Cheon, J. Critical enhancements of MRI contrast and hyperthermic effects by dopant-controlled magnetic nanoparticles. Angew. Chem. Int. Ed. 2009, 48, 1234–1238. [Google Scholar] [CrossRef]
- Lee, J.H.; Huh, Y.M.; Jun, Y.; Seo, J.; Jang, J.; Song, H.T.; Kim, S.; Cho, E.J.; Yoon, H.G.; Suh, J.S.; et al. Artificially engineered magnetic nanoparticles for ultra-sensitive molecular imaging. Nat. Med. 2006, 13, 95–99. [Google Scholar] [CrossRef]
- Thakur, P.; Chahar, D.; Taneja, S.; Bhalla, N.; Thakur, A. A review on MnZn ferrites: Synthesis, characterization and applications. Ceram. Int. 2020, 46, 15740–15763. [Google Scholar] [CrossRef]
- Massart, R. Preparation of aqueous magnetic liquids in alkaline and acidic media. IEEE Trans. Magn. 1981, 17, 1247–1248. [Google Scholar] [CrossRef]
- Sun, T.; Borrasso, A.; Liu, B.; Dravid, V. Synthesis and Characterization of nanocrystalline zinc manganese ferrite. J. Am. Ceram. Soc. 2011, 94, 1490–1495. [Google Scholar] [CrossRef]
- Sugimoto, M. The past, present, and future of ferrites. J. Am. Ceram. Soc. 2004, 82, 269–280. [Google Scholar] [CrossRef]
- Lu, A.-H.; Salabas, E.L.; Schueth, F. Magnetic nanoparticles: Synthesis, protection, functionalization, and application. Angew. Chem. Int. Ed. 2007, 46, 1222–1244. [Google Scholar] [CrossRef]
- Babes, L.; Denizot, B.; Tanguy, G.; Le Jeune, J.J.; Jallet, P. Synthesis of iron oxide nanoparticles used as MRI contrast agents: A parametric study. J. Colloid Interface Sci. 1999, 212, 474–482. [Google Scholar] [CrossRef]
- De Mello, L.B.; Varanda, L.C.; Sigoli, F.A.; Mazali, I.O. Co-precipitation synthesis of (Zn-Mn)-co-doped magnetite nanoparticles and their application in magnetic hyperthermia. J. Alloys Compd. 2018, 779, 698–705. [Google Scholar] [CrossRef]
- Veena Gopalan, E.; Al-Omari, I.A.; Malini, K.A.; Joy, P.A.; Sakthi Kumar, D.; Yoshida, Y.; Anantharaman, M.R. Impact of zinc substitution on the structural and magnetic properties of chemically derived nanosized manganese zinc mixed ferrites. J. Magn. Magn. Mater. 2008, 321, 1092–1099. [Google Scholar] [CrossRef] [Green Version]
- Kareem, S.H.; Ati, A.A.; Shamsuddin, M.; Lee, S.L. Nanostructural, morphological and magnetic studies of PEG/Mn(1−x)Zn(x)Fe2O4 nanoparticles synthesized by co-precipitation. Ceram. Int. 2015, 41, 11702–11709. [Google Scholar] [CrossRef]
- Nasrin, S.; Chowdhury, F.U.Z.; Hoque, S.M. Study of hydrodynamic size distribution and hyperthermia temperature of chitosan encapsulated zinc-substituted manganese nano ferrites suspension. Phys. B Condens. Matter 2019, 561, 54–63. [Google Scholar] [CrossRef]
- Patade, S.R.; Andhare, D.D.; Khedkar, M.V.; Jadhav, S.A.; Jadhav, K.M. Synthesis and characterizations of magnetically inductive Mn–Zn spinel ferrite nanoparticles for hyperthermia applications. J. Mater. Sci. Mater. Electron. 2021, 32, 13685–13692. [Google Scholar] [CrossRef]
- Latorre-Esteves, M.; Cortés, A.; Torres-Lugo, M.; Rinaldi, C. Synthesis and characterization of carboxymethyl dextran-coated Mn/Zn ferrite for biomedical applications. J. Magn. Magn. Mater. 2009, 321, 3061–3066. [Google Scholar] [CrossRef]
- Salehpour, F.; Khorramdin, A.; Shokrollahi, H.; Pezeshki, A.; Mirzaei, F.; Nader, N.D. Synthesis of Zn-doped manganese ferrite nanoparticles via coprecipitation method for magnetic resonance imaging contrast agent. J. Nanotechnol. Eng. Med. 2014, 5, 041002. [Google Scholar] [CrossRef]
- Raland, R.D.; Borah, J.P. Efficacy of heat generation in CTAB coated Mn doped ZnFe2O4 nanoparticles for magnetic hyperthermia. J. Phys. D Appl. Phys. 2016, 50, 035001. [Google Scholar] [CrossRef]
- Arulmurugan, R.; Jeyadevan, B.; Vaidyanathan, G.; Sendhilnathan, S. Effect of zinc substitution on Co–Zn and Mn–Zn ferrite nanoparticles prepared by co-precipitation. J. Magn. Magn. Mater. 2004, 288, 470–477. [Google Scholar] [CrossRef]
- Hejase, H.; Hayek, S.S.; Qadri, S.; Haik, Y. MnZnFe nanoparticles for self-controlled magnetic hyperthermia. J. Magn. Magn. Mater. 2012, 324, 3620–3628. [Google Scholar] [CrossRef]
- Zeng, H.; Rice, P.M.; Wang, S.X.; Sun, S. Shape-Controlled synthesis and shape-induced texture of MnFe2O4 nanoparticles. J. Am. Chem. Soc. 2004, 126, 11458–11459. [Google Scholar] [CrossRef]
- Orsini, N.J.; Milic, M.M.; Torres, T.E. Zn- and (Mn, Zn)-substituted versus unsubstituted magnetite nanoparticles: Structural, magnetic and hyperthermic properties. Nanotechnology 2020, 31, 225707. [Google Scholar] [CrossRef]
- Pardo, A.; Pelaz, B.; Gallo, J.; Bañobre-López, M.; Parak, W.J.; Barbosa, S.; del Pino, P.; Taboada, P. Synthesis, characterization, and evaluation of superparamagnetic doped ferrites as potential therapeutic nanotools. Chem. Mater. 2020, 32, 2220–2231. [Google Scholar] [CrossRef]
- Jeun, M.; Moon, S.J.; Kobayashi, H.; Shin, H.Y.; Tomitaka, A.; Kim, Y.J.; Takemura, Y.; Paek, S.H.; Park, K.H.; Chung, K.-W.; et al. Effects of Mn concentration on the ac magnetically induced heating characteristics of superparamagnetic MnxZn1−xFe2O4 nanoparticles for hyperthermia. Appl. Phys. Lett. 2010, 96, 202511. [Google Scholar] [CrossRef]
- Muller, B.W.; Muller, R.H. Particle size distributions and particle size alterations in microemulsions. J. Pharm. Sci. 1984, 73, 919–922. [Google Scholar] [CrossRef] [PubMed]
- Makovec, D.; Kodre, A.; Arčon, I.; Drofenik, M. Structure of manganese zinc ferrite spinel nanoparticles prepared with co-precipitation in reversed microemulsions. J. Nanoparticle Res. 2008, 11, 1145–1158. [Google Scholar] [CrossRef] [Green Version]
- Rath, C.; Sahu, K.K.; Anand, S.; Date, S.K.; Mishra, N.C.; Das, R.P. Preparation and characterization of nanosize Mn-Zn ferrite. Magn. Magn. Mater. 1999, 202, 77–84. [Google Scholar] [CrossRef]
- Zahraei, M.; Monshi, A.; Morales, M.D.; Shahbazi-Gahrouei, D.; Amirnasr, M.; Behdadfar, B. Hydrothermal synthesis of fine stabilized superparamagnetic nanoparticles of Zn2+ substituted manganese ferrite. J. Magn. Magn. Mater. 2015, 393, 429–436. [Google Scholar] [CrossRef]
- Kaewmanee, T.; Wannapop, S.; Phuruangrat, A.; Thongtem, T.; Wiranwetchayan, O.; Promnopas, W.; Sansongsiri, S.; Thongtem, S. Effect of oleic acid content on manganese-zinc ferrite properties. Inorg. Chem. Commun. 2019, 103, 87–92. [Google Scholar] [CrossRef]
- Seyyed Ebrahimi, S.A.; Masoudpanah, S.M. Effects of pH and citric acid content on the structure and magnetic properties of MnZn ferrite nanoparticles synthesized by a sol–gel autocombustion method. J. Magn. Magn. Mater. 2014, 357, 77–81. [Google Scholar] [CrossRef]
- Thota, S.; Kashyap, S.C.; Sharma, S.K.; Reddy, V.R. Micro Raman, Mossbauer and magnetic studies of manganese substituted zinc ferrite nanoparticles: Role of Mn. J. Phys. Chem. Solids 2016, 91, 136–144. [Google Scholar] [CrossRef]
- Qu, Y.; Li, J.; Ren, J.; Leng, J.; Lin, C.; Shi, D. Enhanced magnetic fluid hyperthermia by micellar magnetic nanoclusters composed of MnxZn1−xFe2O4 nanoparticles for induced tumor cell apoptosis. ACS Appl. Mater. Interfaces 2014, 6, 16867–16879. [Google Scholar] [CrossRef]
- Guo, T.; Dou, F.; Lin, M.; Huang, J.; Zhou, C.; Zhang, J.; Yu, H.; Jiang, X.; Ye, J.; Shi, Y.; et al. Biological characteristics and carrier functions of pegylated manganese zinc ferrite nanoparticles. J. Nanomater. 2019, 2019, 1–10. [Google Scholar] [CrossRef]
- Sobhani, T.; Shahbazi-Gahrouei, D.; Rostami, M.; Zahraei, M.; Farzadniya, A. Assessment of manganese-zinc ferrite nanoparticles as a novel magnetic resonance imaging contrast agent for the detection of 4T1 breast cancer cells. Med. Signals Sens. 2019, 9, 245–251. [Google Scholar] [CrossRef]
- Leng, J.; Li, J.; Ren, J.; Deng, L.; Lin, C. Star–block copolymer micellar nanocomposites with Mn, Zn-doped nano-ferrite as superparamagnetic MRI contrast agent for tumor imaging. Mater. Lett. 2015, 152, 185–188. [Google Scholar] [CrossRef]
- Herynek, V.; Turnovcova, K.; Galisova, A.; Kaman, O.; Marekova, D.; Koktan, J.; Vosmanska, M.; Kosinova, L.; Jendelova, P. Manganese-zinc ferrites: Safe and efficient nanolabels for cell imaging and tracking in vivo. ChemistryOpen 2019, 8, 155–165. [Google Scholar] [CrossRef]
- Sun, Y.; Yan, C.; Xie, J.; Yan, D.; Hu, K.; Huang, S.; Liu, J.; Zhang, Y.; Gu, N.; Xiong, F. High-Performance Worm-like Mn-Zn Ferrite Theranostic Nanoagents and the Application on Tumor Theranostics. ACS Appl. Mater. Interfaces 2019, 11, 29536–29548. [Google Scholar] [CrossRef] [PubMed]
- Albarqi, H.A.; Demessie, A.A.; Sabei, F.Y.; Moses, A.S.; Hansen, M.N.; Dhagat, P.; Taratula, O.R.; Taratula, O. Systemically Delivered Magnetic Hyperthermia for Prostate Cancer Treatment. Pharmaceutics 2020, 12, 1020. [Google Scholar] [CrossRef]
- Wang, Y.; Zou, L.; Qiang, Z.; Jiang, J.; Zhu, Z.; Ren, J. Enhancing targeted cancer treatment by combining hyperthermia and radiotherapy using Mn-Zn ferrite magnetic nanoparticles. ACS Biomater. Sci. Eng. 2020, 6, 3550–3562. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Slavu, L.M.; Rinaldi, R.; Di Corato, R. Application in Nanomedicine of Manganese-Zinc Ferrite Nanoparticles. Appl. Sci. 2021, 11, 11183. https://doi.org/10.3390/app112311183
Slavu LM, Rinaldi R, Di Corato R. Application in Nanomedicine of Manganese-Zinc Ferrite Nanoparticles. Applied Sciences. 2021; 11(23):11183. https://doi.org/10.3390/app112311183
Chicago/Turabian StyleSlavu, Laura Maria, Rosaria Rinaldi, and Riccardo Di Corato. 2021. "Application in Nanomedicine of Manganese-Zinc Ferrite Nanoparticles" Applied Sciences 11, no. 23: 11183. https://doi.org/10.3390/app112311183
APA StyleSlavu, L. M., Rinaldi, R., & Di Corato, R. (2021). Application in Nanomedicine of Manganese-Zinc Ferrite Nanoparticles. Applied Sciences, 11(23), 11183. https://doi.org/10.3390/app112311183





