1. Introduction
Oil production faces a decline in many oil fields in different parts of the world because of the withdrawal of large amounts of high-flowing oil. There is a necessity, therefore, to apply various methods to enhance oil recovery from existing oilfields worldwide. The extraction of heavy crude oil needs higher energy input due to its resistance to flow, high density, and low American Petroleum Institute (API) gravity of less than 20 [
1].
Viscosity is a measure of a fluid’s resistance to flow. It is one of the most important properties of a fluid and plays a very important role in the petroleum industry. The viscosity of a crude oil therefore affects the ability to pump it out of the ground. Small changes in viscosity can have a dramatic impact on the properties of petroleum fluids. Oil mobility can be improved by a reduction of oil viscosity; therefore, lowering the viscosity of crude oil is essential in the oil extraction industry. Thus, the discovery of new technologies that reduce the viscosity of crude oil with minimum requirements to raise its temperature is highly valued.
Microbial enhanced oil recovery (MEOR) has been proposed for many years as a cheap and effective alternative technique. MEOR is a good alternative in improving the recovery of crude oil from reservoir rocks by using microorganisms and their metabolic byproducts [
2]. Microbial enhanced oil recovery (MEOR) is used in the third phase of oil recovery from a well, which is the tertiary oil recovery stage [
3]. Many investigations of the MEOR technique have injected microorganisms or their products into oil reservoirs to improve the efficiency of oil recovery [
4,
5,
6]. Up to 50% of the residual oil can be extracted by this exceptionally low operating cost technology [
7]. Bacteria can ferment readily available organic compounds to produce gases such as CH
4, CO
2, and H
2. These gases, which are produced in situ, can increase the pressure of the reservoir and enhance oil recovery. In addition, they mobilize the crude oil through reducing its viscosity which enhances its recovery [
8].
This study aimed to demonstrate the potential of using bacterial isolates Bacillus species, Pseudomonas putida, and Nocardia cyriacigeorgica in the biotransformation of heavy crude oil (26° API) and to evaluate the viscosity reductions of the heavy crude oil to enhance its flow properties and increase its extraction efficiency from the reservoir.
2. Materials and Methods
2.1. Characterization of Soil and Oil Samples
Subsurface soil samples contaminated with heavy crude oil were collected from an oil-contaminated site in Libya, transferred to a microbiological laboratory of the Libyan Petroleum Institute, and stored at 4 °C until use. The soil was air-dried for 48 h and all samples mixed to obtain one homogenous sample. Then, soil was sieved to 2 mm to remove gravel and stones. Soil pH was determined using soil suspensions in water as described by [
9,
10]. Soil texture was analyzed using a Master Sizer 2000 (Malner International, London, UK). Mineralogy analysis was carried out using X-ray diffraction (XRD) (New York, NY, USA). Soil moisture content and water holding capacity were measured as described by [
11]. Maximum water holding capacity (WHC) was determined as described by [
12]. The petroleum products present in the soil samples were extracted using a Soxhlet Extraction System, New York, NY, USA (test method 35–40 °C) with dichloromethane to measure the total petroleum hydrocarbons (TPH) level. The heavy oil sample used in this study was obtained from Bouri oil field, Libya. The Bouri crude oil viscosity was measured using rotational viscometer, API gravity, and a CVM 3000 Viscometer (Shenzhen, China).
2.2. Isolation of Hydrocarbon Degrading Bacteria Using Heavy Crude Oil as Carbon Source
One gram of the homogenized soil sample was mixed with 10 mL of distilled water and vortexed thoroughly; 5 mL of the supernatant served as an inoculum for the first enrichment containing 50 mL of enrichment medium (g/L) (NH
4)
2SO
4 (1.5); NaH
2PO
4·H
2O (1.5); K
2HPO
4 (4.65); KCl (0.1); MgSO
4·7H
2O (0.2); yeast extract (0.5); peptone (0.5); casamino acid (0.5); and trace elements (2.0) (pH = 7.1), and varying Bouri crude oil concentrations of 1.0, 2.0, and 3.0% (
v/
v) were added. Cultures were incubated with shaking for 14 d at 37 and 55 °C for the purpose of isolating hydrocarbon-utilizing bacteria from oil contaminated soil. Three different isolates (
Nocardia cyriacigeorgica,
Bacillus species, and
Pseudomonas putida) showing the best growth on heavy crude oil were selected and transferred to a mineral medium containing (g/L) NaNO
3 (4); Na
2HPO
4 (0.5); KH
2PO
4 (1.5); KCl (0.1); MgSO
4·7H
2O (0.2); CaCl
2 (0.01); FeSO
4·7H
2O (0.0011); and yeast extract (0.1). The medium was supplemented with 1 mL of the trace elements solution [
13] and optimum concentration 2.0% (
v/
v) of crude oil. Then, 50 mL cultures were grown in 250 mL Erlenmeyer flasks and incubated with orbital shaking at 150 rpm for 21 d at 37 and 55 °C.
The three bacterial isolates with the ability to grow on target hydrocarbons were assessed further for growth rate, crude oil biotransformation, viscosity reduction, and oil recovery. Growth of the isolated bacterial strains on different concentrations of crude oil was evaluated by measuring culture absorbance at 600 nm using a spectrophotometer (Eppendorf Biophotometer plus, Hamburg, Germany).
2.3. Identification of Isolates by 16S rDNA Analysis
DNA was extracted from the bacterial colonies according to the protocol described by Sigma’s GenElute Bacterial Genomic Kit. Genomic DNA from hydrocarbon degraders were amplified by polymerase chain reaction (PCR) using the specific bacterial primers 27 f and 1525 r. These are universal primers that bind at the conserved 5 and 3′ ends of 16S rDNA of the eubacteria. PCR amplification was performed as described by [
14]. All reagents used were from Bioline, (London, UK). PCR was performed with a Biometra Thermal Cycler (Jena, Germany). In order to check the sizes of the PCR products, 4 μL of the PCR reactions were analyzed by 1.0% agarose gel electrophoresis in TAE (Tris acetic acid EDTA buffer, 1%
w/
v; 30 min at 100 V, 0.5× TBE), stained with ethidium bromide, and visualized under UV light using image analysis software (Bio-Rad Gel Doc, Flour-S multi-imager, Hertfordshire, UK), according to [
15]. The size of the amplified fragments was estimated by comparing with a 100 bp molecular size marker.
The PCR products were cleaned up using an ExoSAP-IT PCR clean-up kit, the protocol for which consists of a single pipetting step (enzyme mixture addition), a 15 min incubation at 37 °C followed by a further 15 min incubation at 80 °C for enzyme inactivation, and then the PCR for products were sequenced by Geneius Laboratories Ltd. (based on Newcastle University campus, UK) using Sanger sequencing based on a big dye terminator using an ABI 3730xl DNA sequence. Sequences were viewed using MEGA software version 4.0. A contiguous sequence using forward and reverse sequences was made, and the contiguous sequences were submitted into BLAST for comparison with known databases.
2.4. Sand-Packed Column Experiment
Five core samples and four sand packs were collected from the Sirte Basin and formation type (Dolomite), Concession 11 (Alghni Field, northeast of Libya), sand-packed model (4 cm in diameter; and 30 cm in length) were prepared with different grain size. The oil sample was heated to 65 °C for 960 h. K = 200 psi net confining stress, Q = 100 psi Helium. The dolomite sandstone core plugs were drilled in horizontal and vertical directions using a diamond core bit with water as the bit coolant and lubricant. The plug sample dolomite sandstone cores (absolute permeability 230 × 10−2 µm2) were extracted of hydrocarbons using toluene, leached of salt using methanol, and oven dried at 80 °C. A silver nitrate solution was used to ensure the removal of salts present in the pore spaces. The clean, dry samples were subjected to various analyses to determine permeability, porosity, and grain density values, and were saturated with filtered, sterilized formation water for 12 h in a desiccator under vacuum. The formation water was collected from one of the Libyan oil fields. The experimental procedures and equipment specifications were tested according to API RP 40 (recommended practice for core analysis).
A model of an oil-saturated dolomite formation column was used to mimic the reservoir condition. The crashed dolomite cores saturated with Bouri oil were placed in the packed column apparatus and heated in the oven at 50 °C, then were saturated with 5–6 pore volume formation water until no further oil was produced. The pore volume (PV) was calculated as the difference in the wet and dry weights of the core. The residual oil was calculated by measuring the amount of oil produced from water flooding. Then, 5–6 PV heavy crude oil was injected until no more water was produced (i.e., it reached the irreducible water saturation, (Swr)). The initial oil saturation of the cores was calculated volumetrically from the amount of injected oil and produced water. Further extra oil recovery was determined by injected 5–6 PV of the bacterial culture as a tertiary recovery.
The 24 h-grown isolates in Luria Bertani broth (LB) served as the inoculum for the packed column experiment, (OD600 = 0.764; 32.8 × 107 CFU/mL for Nocardia cyriacigeorgica, OD600 = 1.88; 21.8 × 107 CFU/mL for Bacillus species, and OD600 = 1.734; 47.3 × 107 CFU/mL for Pseudomonas putida). The bacterial inoculum was injected into the core and the column model was incubated for 12 weeks at 55 °C. A control experiment was performed without injection of the isolates at the same conditions. Core columns were incubated under anaerobic conditions and simulated reservoir conditions (pressure, temperature 55 °C, and flow rate).
Samples were taken to measure the viscosity reduction and oil recovery. The residual crude heavy oil in the core was measured from the volume of oil produced. The extra recovered oil was collected and measured using graduated tubes, and then analyzed by GC-FID for determining the biotransformation of heavy crude oil. The pH of the aqueous phase in the packed column was also measured before and during the incubation period.
2.5. Gas Chromatography Hydrocarbon Analysis
The collected oil fractions from the water flood before and after the incubation with bacteria strains were analyzed by GC. This was done to assess the effect of different bacterial species on the biodegradation of Bouri crude oil and to determine whether the biotransformation significantly depended on temperature as an important parameter.
The crude oil was extracted from core column after 12 weeks of incubation using liquid–liquid extraction techniques by a mixture of
n-hexane and dichloromethane (1:1) for GC analysis. The extracted solution was then dried through solvent-washed anhydrous sodium sulfate in Whatman #50 paper. The remaining organic phase of the extract was removed with a dichloromethane solvent and put in a sealed flask for subsequent analysis. Gas chromatography analyses were carried out on the initial sample (time = 0 day) and at the end of the experiments (time = 84 days) to evaluate the potential of biotransformation rate, as described by [
16,
17]. Total petroleum hydrocarbon and
n-alkane concentrations were determined using a DHA (Carbon Distribution) Model No: CP3800.
2.6. Bacterial Effect on Oil Viscosity Measurements
The viscosity density and specific gravity of the crude oil were determined at 50 °C by a digital viscometer SVM 3000 (Anton Paar Company, Graz, Austria). The percentage of oil viscosity reduction, μr, was calculated as:
where μbm and μam are the oil viscosity before microbial treatment and after microbial treatment, respectively. API gravity was determined by the following equation [
18]:
2.7. Rheological Investigations
Bouri crude oil was separated from the packed column using a separation technique, and the rheological study was performed using Rotational Viscometer (CV100) at 50 °C and 12 s
−1 shear rate to measure the viscosity of crude oil at 37 and 55 °C. The shear rate was increased gradually from 0 s
−1 to a larger shear, i.e., 50 s
−1. Viscosity was then measured as a function of shear rate and shear stress at different temperatures. The most common equation that describes such behavior is the Power Law model,
where
τ = shear stress, Pa,
= shear rate, s
−1, and
K = Apparent viscosity, Pascal.
3. Results
3.1. Characterization of Soil a Recovered Oil from the Core Modelnd Oil Samples
The heavy crude oil sample viscosity was determined as 17.193 and as 26.1 API. Soil pH was neutral to slightly alkaline (pH 7.6). Analyses of the soil samples indicated that the soil was sandy with high carbon content; however, the number of hydrocarbons degrading microorganisms present was 2.4 × 107 CFU g−1 dry soil. Moisture content of the soil was found to be 4.53 and 2.29 soil WHC.
The TPH of the soil sample was 6733.3 ppm. The mineral compositions of the soil samples measured by XRD showed that the soil samples contained Lepidocrocite, Microcline, and Wustite (
Table 1).
3.2. Core Samples Analysis
The core samples from Sirte Basin and formation type (Dolomite) were tested (date not shown). Lab experiments were performed by injecting live bacterial cells through core material (packed column).
3.3. Molecular-Based Identification of Bacterial Isolates
The isolates that could utilize heavy crude oil as a carbon source and showed maximum growth were isolated and selected for the study. The three bacterial isolates were characterized to the genus level based on the complete sequence of their 16S ribosomal RNA gene. The sequences of 16S rRNA were submitted to BLAST under accession numbers FM211694.1, AF282889.1, and AF526907, respectively, and the bacteria identified as shown in
Table 2. It was not possible to obtain full identification to the species level of all the bacterial isolates.
3.4. Effect of Temperature and Crude Oil Concentrations on the Growth of the 3 Bacterial Isolates
Pure cultures of the three selected bacteria were supplemented with different concentrations of crude oil −1.0, 2.0, and 3.0 (
v/
v) and incubated at 37 and 55 °C under controlled conditions. All species exhibited more favorable growth at temperatures of 55 °C than at 37 °C, and hydrocarbon concentrations at 2.0%. The optimum growth rates of the different isolates on 2.0% (
v/
v) of crude oil were calculated and showed that the
Bacillus species and
Pseudomonas putida grew quicker than the other isolate,
Nocardia cyriacigeorgica, on hydrocarbons of 2.0% (
v/
v) at 55 °C. The maximum specific growth rate (μmax) for the
Bacillus species was 1.88 h
−1,
Pseudomonas putida was 1.734 h
−1, and the
Nocardia cyriacigeorgica was 0.764 h
−1 (
Table 3). Therefore, both
Bacillus species and
Pseudomonas putida gave the highest growth rate and were selected for further investigation.
3.5. Oil Viscosity Reduction
Oil viscosity was used as an indicator of microbial-induced compositional change in the crude oil after microbial treatment.
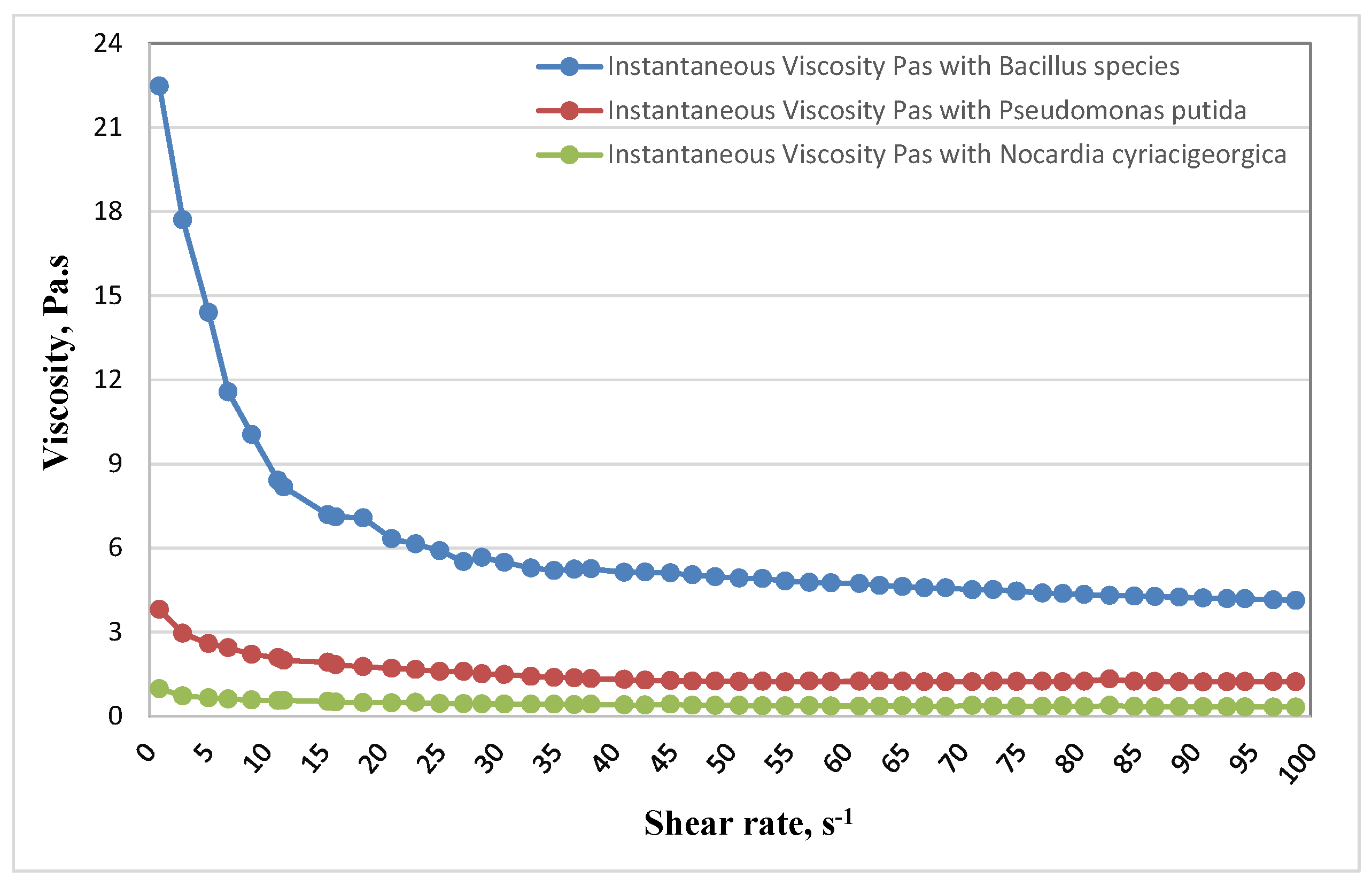
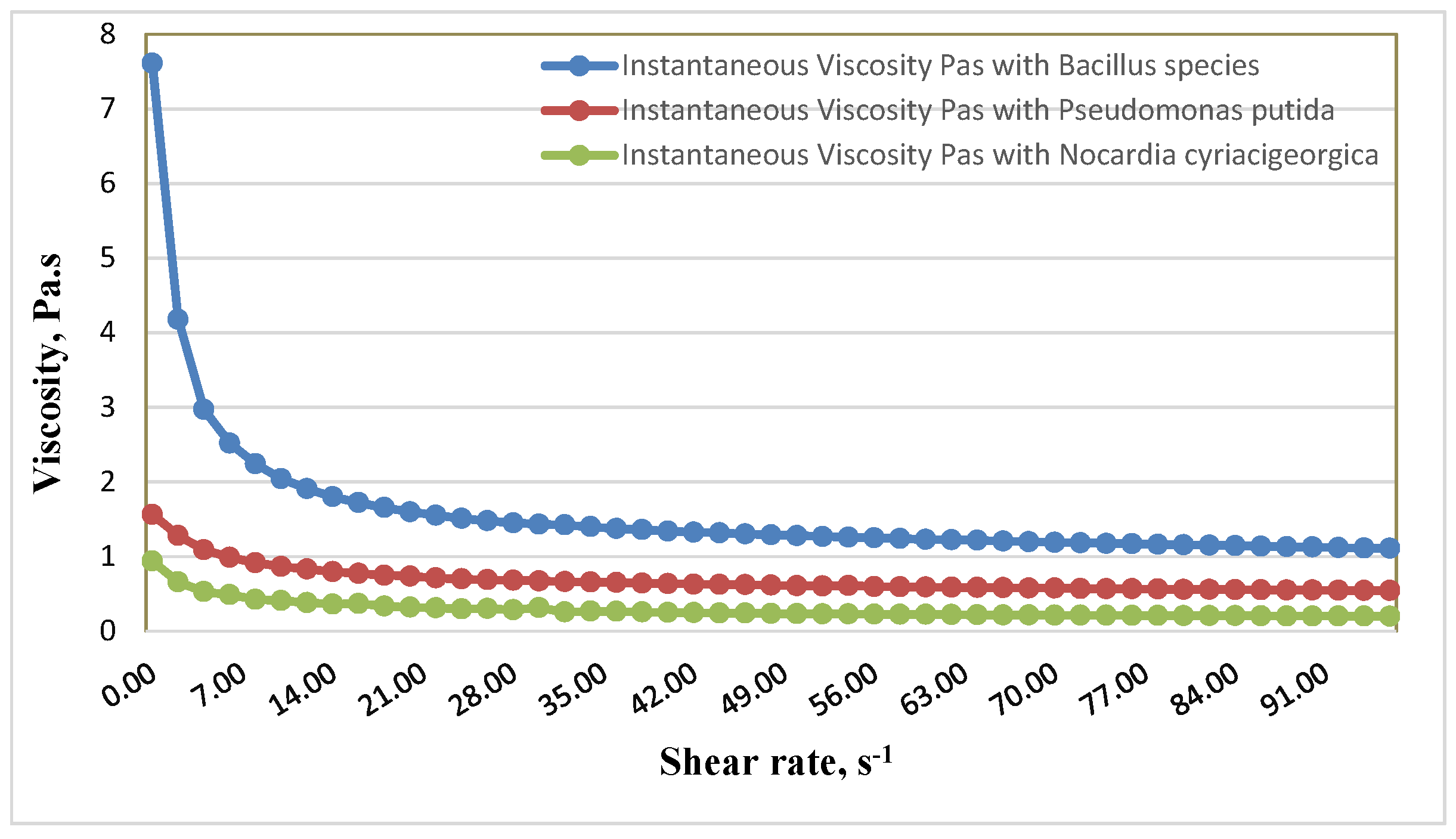
Table 4 represents the change in viscosity of crude oil after microbial treatment at different shear rate values. Two temperatures were covered in this study (37 and 55 °C) to examine the effect of temperature on the flow behavior of the heavy crude oil. The heavy crude oil viscosity decreases significantly from 13.827 to 3.597 Pas when the temperature changes from 37 to 55 °C.
Effect of Bacterial Isolates on Viscosity of Bouri Crude Oil
Specific quantitative lab procedures using a Rotational Viscometer (CV100) were carried out to measure the shift in rheological properties in treated (inoculated) samples. The reduction of oil viscosity to enhance the flow properties occurs due to effects of bacterial degradation of oil or excreting of components such as surfactants into the oil phase.
Figure 1 at 37 °C and
Figure 2 at 55 °C show a sharp decrease in viscosity at a shear rate below 3 s
−1, and after 50 s
−1 the viscosity remains steady.
The viscosity of crude oil treated with
Bacillus species was significantly decreased from 22.47 Pa.s at 0.8481 s
−1 to 4.127 Pa.s at 98.73 s
−1 at a temperature of 37 °C; comparing with the efficiency of the
Pseudomonas putida, the viscosity reduction was from 3.806 Pa.s at 0.7652 s
−1 to 1.226 Pa.s at 98.64 s
−1.
Nocardia cyriacigeorgica reduced oil viscosity from 0.9786 Pa.s at 0.8226 s
−1 to 0.3224 Pa.s at 98.79 s
−1 (
Figure 1).
At a temperature of 55 °C, the viscosity of crude oil treated with
Bacillus species was significantly decreased from 7.61 Pa.s at 0.885 s
−1 to 1.11 Pa.s at 96.74 s
−1. The result further showed that the
Bacillus species can be successfully used at high temperatures. The reduction in viscosity was decreased from 1.565 Pa.s at 0.7622 s
−1 to 0.5415 Pa.s at 98.67 s
−1 with
Pseudomonas putida.
Nocardia cyriacigeorgica reduced viscosity from 0.9406 Pa.s at 0.885 s
−1 to 0.1998 Pa.s at 96.74 s
−1 (
Figure 2).
There was no significant change of pH, indicating that no acidic byproducts were produced during the incubation period.
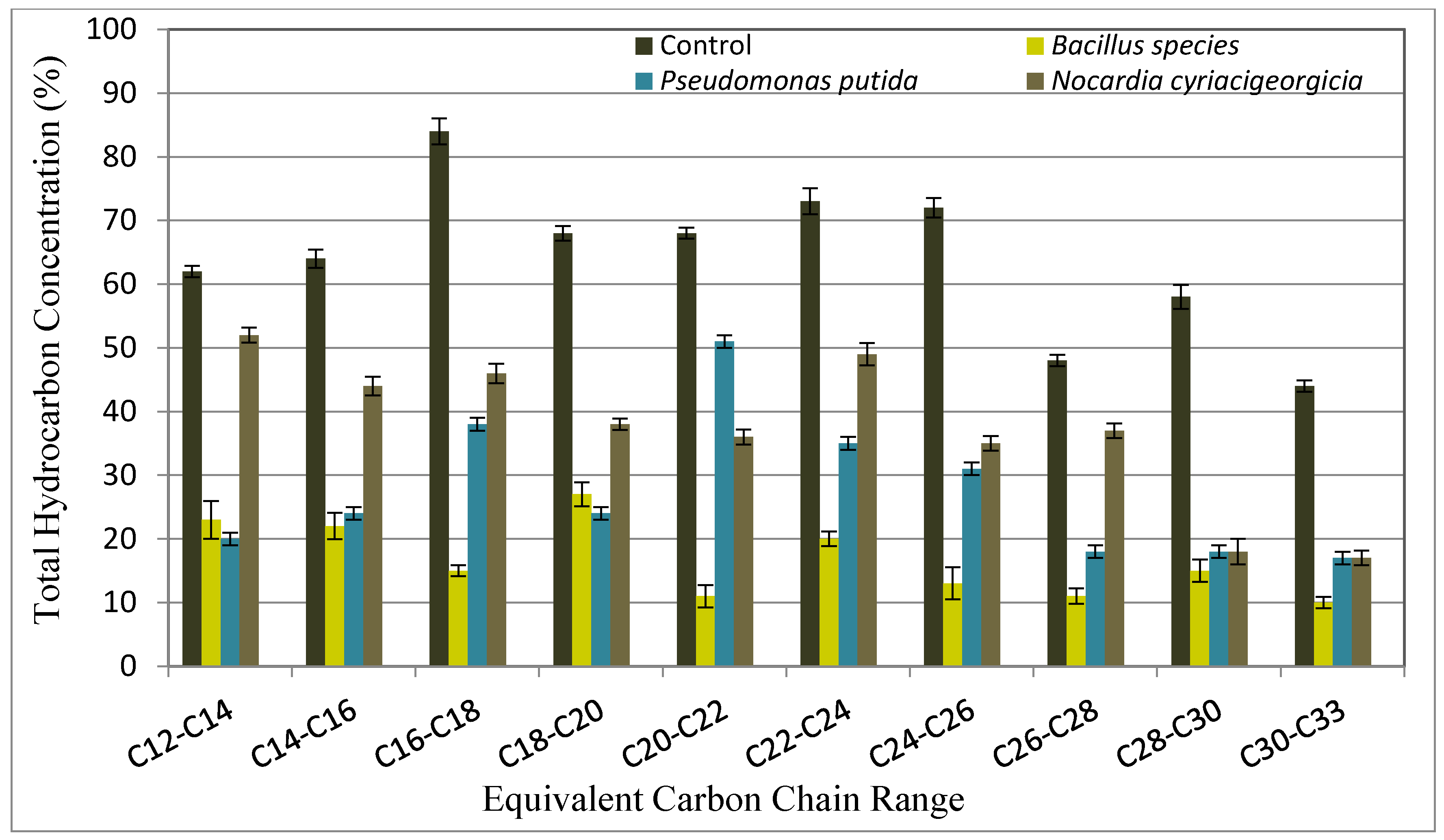
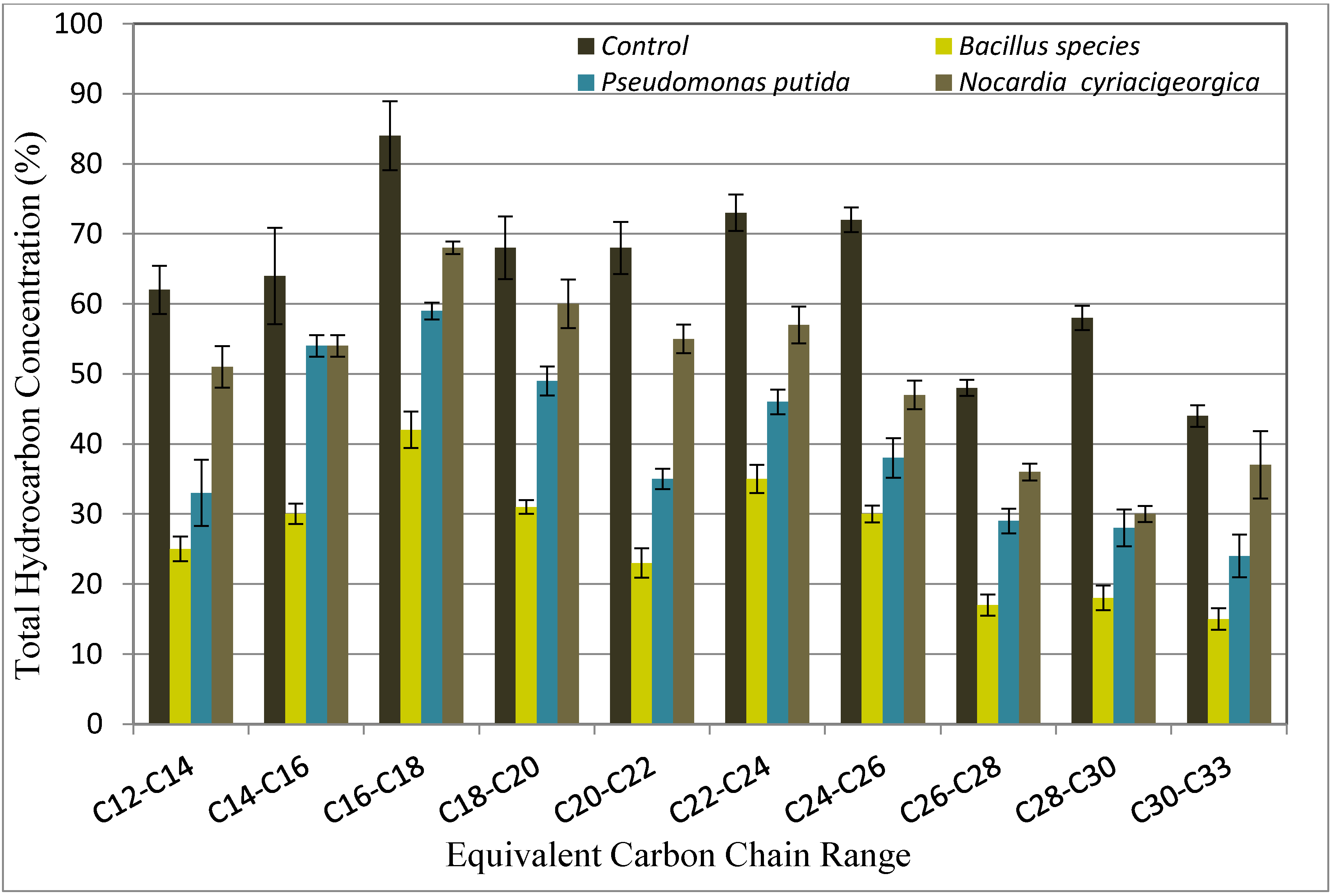
3.6. Crude Oil Transformation
The collected oil fractions from the flood water before and after the incubation with bacteria strains were analyzed by GC. The results of gas chromatograms indicated that there was a marked change in GC chromatograms for Bouri crude oil treated with three strains when compared with control untreated crude oil. A comparison of the profiles in
Figure 3 and
Figure 4 illustrates a decrease in the weight percentage of carbon number fractions before and after treatment with
Bacillus species,
Pseudomonas putida, and
Nocardia cyriacigeorgica.The analysis data of the Gas chromatograms confirmed that the biotransformation produced important changes in the composition of the Bouri crude oil. The microbial transformation of 2.0% Bouri crude oil at temperature 55 °C was higher than that obtained at temperature 37 °C.
At temperature 55 °C Bacillus species showed a significant decrease in carbon fraction (C12 to C33). A sharp reduction of heavy fraction (C22 to C33) with Pseudomonas putida treatment was observed; this was probably because this microbe utilized the heavy fraction before the light fraction. A similar trend was also observed in the case of treatment with Nocardia cyriacigeorgica, where the analysis showed 77% biotransformation of heavy crude oil for Bacillus species, and 61% for Pseudomonas putida and Nocardia cyriacigeorgica.
At temperatures of 37 °C, Bacillus species can degrade lighter hydrocarbons with a carbon chain length of (C12–C24) and heavier hydrocarbons with a carbon chain length of (C24–C33). However, Pseudomonas putida and Nocardia cyriacigeorgica exhibited better degradation on heavier hydrocarbons with carbon chain length of (C24–C33), the analysis showed 66% biotransformation of heavy crude oil for Bacillus species, 45% for Pseudomonas putida, and 16% with Nocardia cyriacigeorgica.
3.7. Recovered Oil from the Core Model
Dolomite sandstone cores were used to evaluate the potential of isolates in heavy oil recovery. After 12 weeks incubation, 7.9 to 11.38% additional oil-in-place was recovered. The oil initially in place (OIIP) in the core for Nocardia cyriacigeorgica was 11.4 mL (Soi = 63.6%). After injecting 5–8 PV of formation water, no more oil was produced and residual oil saturation (Soi) was about 20%, which resulted in the recovery of 50.87% of initial oil (OI), corresponding to 5.8 mL of the initial oil present in the core. The column was reinjected with 5–6 PV of brine that resulted in a total recovery of 58.77% (6.7 mL) of initial oil present in the core in which 7.9% (0.9 mL) was produced by the isolate.
With core treated by Pseudomonas putida, the oil initially in place (OIIP) was determined at 12.2 mL (Soi = 82.31%). The water flooding resulted in recovery of 54.09% (6.6 mL) of OIIP. Initial oil saturation (Soi) was calculated to be 63% after being injected with 5–6 PV of oil, which was indicated by the volume of water displaced. A total recovery of 64.75% (7.2 mL) of initial oil present in the core was recorded. An increase in oil recovery of 10.66% (0.6 mL) was detected after 12 week incubation.
The oil initially in place (OIIP) in the core for Bacillus species was determined, 12.3 mL, Soi = 84.02%. The water flooding resulted in recovery of 52.84% (6.5 mL) of OIIP. Initial oil saturation was calculated to be 63% after being injected with 5–6 PV of oil, which was indicated by the volume of water displaced, that resulted in a total recovery of 64.22% (7.9 mL) of initial oil present in the core. After 12 weeks an increase in oil recovery of 11.38% (1.4 mL) by Bacillus species was detected.
4. Discussions
Experiments were conducted on the growth of bacteria, adsorption of bacterial cells and metabolites on dolomite rocks, reduction in viscosity, and biotransformation of crude oil. In this study, three strains, Bacillus species, Pseudomonas putida, and Nocardia cyriacigeorgica, with potential for biotransforming heavy crude oil were isolated from heavy crude oil-contaminated soil samples collected from Libyan oil fields. The potential of three identified microbial genera for EOR was reported for the first time in Libya.
The reservoir factors such as temperature, salinity, pH, and pore size affect the effectiveness of microbes in the reservoir [
19]. The isolated strains were grown in pH between 6–8 and a crude oil range of 1.0–3.0%. The growth of the bacterial species was increased with increased crude oil concentration to an optimum at 2.0% concentration and only limited growth occurred when the concentration was further increased to 3.0%. Higher concentrations of hydrocarbons might inhibit biodegradation by limiting access to nutrient or oxygen supply or by its toxic effects [
20].
Experience has shown that, with careful planning and repeated sub-culturing, it is possible that these strains can grow at higher crude oil concentrations normally encountered in the reservoirs; however, this was not investigated further in this study.
Based on optimum temperatures for growth and crude oil concentration requirements, the optimum temperature of the three facultative anaerobes,
Bacillus species,
Pseudomonas putida and
Nocardia cyriacigeorgica, was 55 °C, while they did not grow at temperatures above 60 °C. This was similar to the optimum temperature range reported by Chen et al. [
21].
Methanobacterium showed optimum growth temperatures of 63 to 66 °C with a specific growth rate of 0.62 h
−1 [
22]. Many oil reservoirs have an initial temperature greater than 75 °C but due to continuous injection of water for oil recovery over time, the temperature becomes much less than the initial reservoir temperature as the field matures [
23]. The modeling work presented in the previous sections showed that the potential strains used can withstand both high crude oil concentration (2.0%) and high temperature conditions (55 °C). Lower growth rates were recorded at 37 °C with different concentrations of Bouri crude oil (1.0, 2.0, and 3.0%). Only the
Bacillus species gave good growth with crude oil concentrations of 2.0%.
Many of the bacteria strains used in MEOR usually experienced low efficiency when maintained at higher temperatures, but
Bacillus species and
Pseudomonas putida were able to exhibit good potential with crude oil concentrations of 2.0% at high temperature. However, what makes these strains better lies in their ability to grow at a high temperature of 55 °C, in comparison to many bacteria isolated for microbial enhanced oil recovery purposes. The microorganisms that are most used for MEOR field processes are species of
Bacillus and
Clostridium. These species produce spores, which are resistant stages, allowing them to survive in petroleum reservoirs and more stressful environmental conditions [
24].
Transformation of Hydrocarbon
The GC analysis showed that heavy oil that is trapped in an oil reservoir after primary and secondary recovery can be recovered by biotransforming the heavier fractions to lighter ones. The results have demonstrated that not all the alkanes were equally reduced as a difference was recorded (
Figure 3 and
Figure 4).
Comparing these results with other reports, the
Bacillus species isolated from a natural environment had the ability to degrade the heavy fraction of crude oil and change the chemical and physical properties of Bouri crude oil. Close strains such as
B. firmus and
B. halodurans were shown to result in the fractionation of higher
n-alkanes with carbon numbers up to C54 [
1].
The microbial degradation of oil and biosurfactants production are major factors taking place during microbial water injection, microbial activities, and interactions of metabolic products with oil and cores contributing to the enhancement of oil recovery [
25,
26]. The addition of
Thermoanerobacter brockii subsp.
Lactiethylicus 9801T was reported to degrade alkanes and produce important metabolites such as biosurfactants and gases which positively contribute to the oil recovery process [
27].
The percentage of heavier and lighter hydrocarbons was reduced to 77 and 63%, respectively, during the period of incubation (55 °C) with Bacillus species, and 66% on heavier fractions of Bouri crude oil at 37 °C.
Pseudomonas putida and
Nocardia cyriacigeorgica have decreased the concentration of heavier hydrocarbons C22+, to 61% at 55 °C. On the other hand, a slight decrease in lighter hydrocarbons of 45% and 16%, respectively, was recorded for both species at 37 °C. The crude oil utilization capability and degradation potential of both
Pseudomonas and
Bacillus sp. were reported in previous studies [
28,
29].
The heavy crude oil biodegradation and biotransformation were evident by the decrease in lighter and heavier compounds. Bacterial strains were found to destroy heavier fractions which led to an increased API of crude and tended to reduce viscosity, thus increasing the oil’s mobility and flow characteristics. Moreover, the degradation of alkanes was also very significant, probably from the effect of biosurfactants production (data not shown). These findings suggest that the bacterial strains used may be potential candidates for the biotransformation of heavy crude oil.
The isolated bacteria strains (
Pseudomonas putida,
Bacillus species, and
Nocardia cyriacigeorgica) significantly decreased the oil viscosity from 22.47 Pa.s at 0.8481 s
−1 to 4.127 Pa.s at 98.73 s
−1. According to [
30], using microbes for biodegrading long chain paraffins may have the following benefits: the amount of paraffin will be reduced, resulting in reduced paraffin precipitation or deposition problems along the production flow line, and as a result the viscosity of crude oil will be reduced, increasing its API gravity.
The microbial degradation of crude oil results in its dissolution away from the block and eventually being carried out in porous media while simultaneously resulting in the phenomenon of swept volume expansion that remarkably improves the oil displacement effect [
31].
The isolates were shown to be feasible for similar applications in a number of laboratory experiments reported before. However, they have not been widely used in heavy oil-saturated dolomite sandstone cores before and, since rock properties, such as mineral content, grain and pore size, porosity, permeability, under some physical and chemical conditions that resemble those experienced in an oil production reservoir can vary, such as high salinities and temperatures, this study highlights the novelty of using these strains, Bacillus species, Pseudomonas putida and Nocardia cyriacigeorgica, as excellent candidates that can reduce oil viscosity, leading to some biotransformation and an increase in oil recovery.
N. cyriacigeorgica was reported to degrade aliphatic and branched chain alkanes as well as alkylbenzene of oil in contaminated soils [
32], whereas in the current study this isolate showed the ability to biotransform heavy oil with an API gravity value of 26°, reducing its viscosity and increasing the oil recovery to 7.9%. In a bioaugmentation treatment to remove hydrocarbons,
Pseudomonas putida CB-100 enhanced TPH decontamination of contaminated aged soil; 40.6% of total petroleum hydrocarbons (TPH) was removed and 1.54 mg/kg rhamnolipid was reported produced [
33]. Our
P. putida utilized the heavy fraction of heavy crude oil and gave 61% of biotransformation, a reduction in viscosity from 1.565 Pa.s at 0.7622 s
−1 to 0.5415 Pa.s at 98.67 s, and, interestingly, increased the oil recovery by 10.66%.
Considerable oil recovery has been obtained by the biotransformation of heavy crude oil compared to the control experiment. The extra recovery measurements were based on the residual oil present in the core. The highest recovery of residual oil among different bacterial species was approximately 11.38% at 55 °C by
Bacillus species. Similar results were reported by [
34], where an additional recovery of 9.6% at 37 °C and 7.2% at 55 °C of crude oil 26 API occurred due to the combined effect of biosurfactants and the biotransforming ability of
Bacillus spp. In addition, 14.3% oil recovery for unfractured core models was recorded by [
35] using a different indigenous microorganism isolated from oil samples from the Payedar oilfield.
Oil recovery can generally be improved by 5% using microbial flooding solely [
36]. This approach has a significant effect of the fracture angle on performance of the process and has indicated that oil recovery was increased by 6–10% of initial oil in place using microbial mediated oil, depending on oil saturation at the start of the flooding [
37].
5. Conclusions
Local isolates of Pseudomonas putida, Bacillus species, and Nocardia cyriacigeorgica were able to grow at higher concentrations of heavy crude oil, effectively converting heavy fractions of crude oil to lighter fractions and indicating their potential application in microbial-enhanced oil recovery processes. Bacterial biotransformation of Bouri crude oil showed significant dependence on temperature as an important parameter.
Viscosity was studied versus shear rate and temperature. The control sample showed that the use of these bacteria can alter crude to a significant extent. The three isolates considerably improved oil production and reduced oil viscosity. Isolates Pseudomonas putida and Bacillus species were both demonstrated to be promising candidates for MEOR applications. The extra recovery of crude heavy oil in the sand-packed column experiments and migration of bacteria in porous sandstone cores further confirmed this.
In conclusion, MEOR is an eco-friendly cost-effective technique with various benefits over other chemical EOR methods and the isolated strains can result in additional oil as a part of the tertiary oil recovery process.