Co(II) Complex of Quercetin–Spectral, Anti-/Pro-Oxidant and Cytotoxic Activity in HaCaT Cell Lines
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Synthesis
2.3. Spectral Studies
2.4. Anti-/Pro-Oxidant Studies
2.5. Cell Viability Test
3. Results
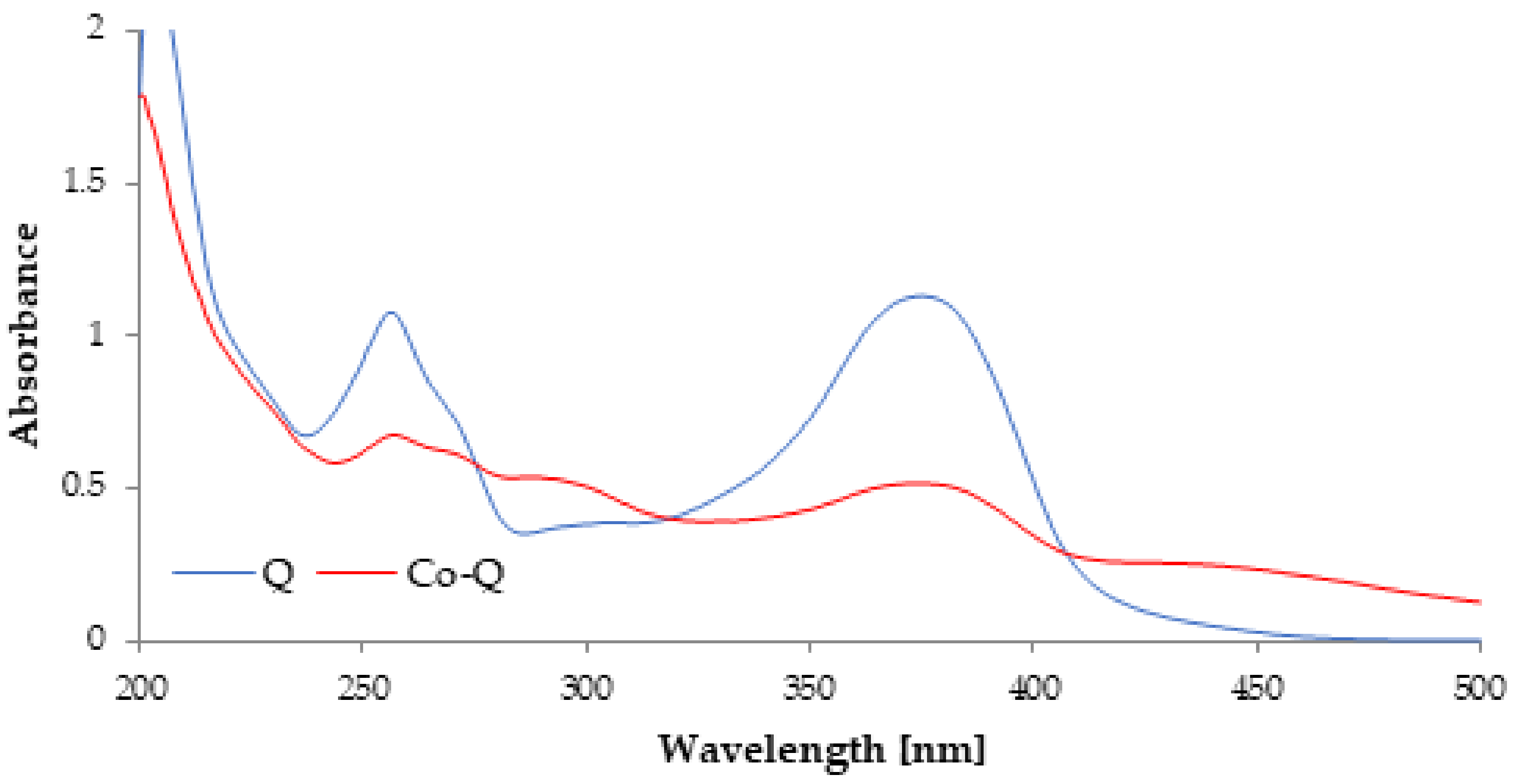
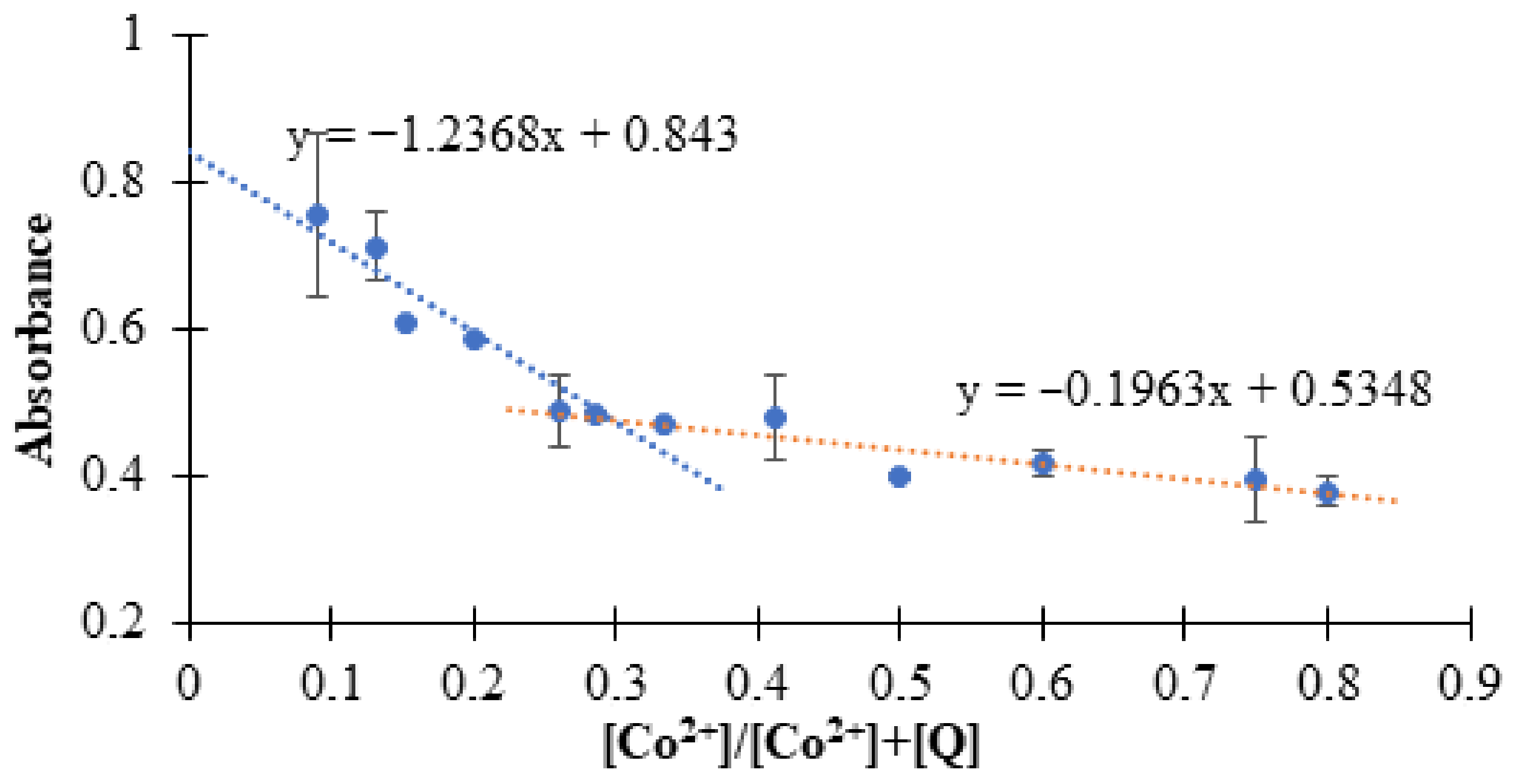
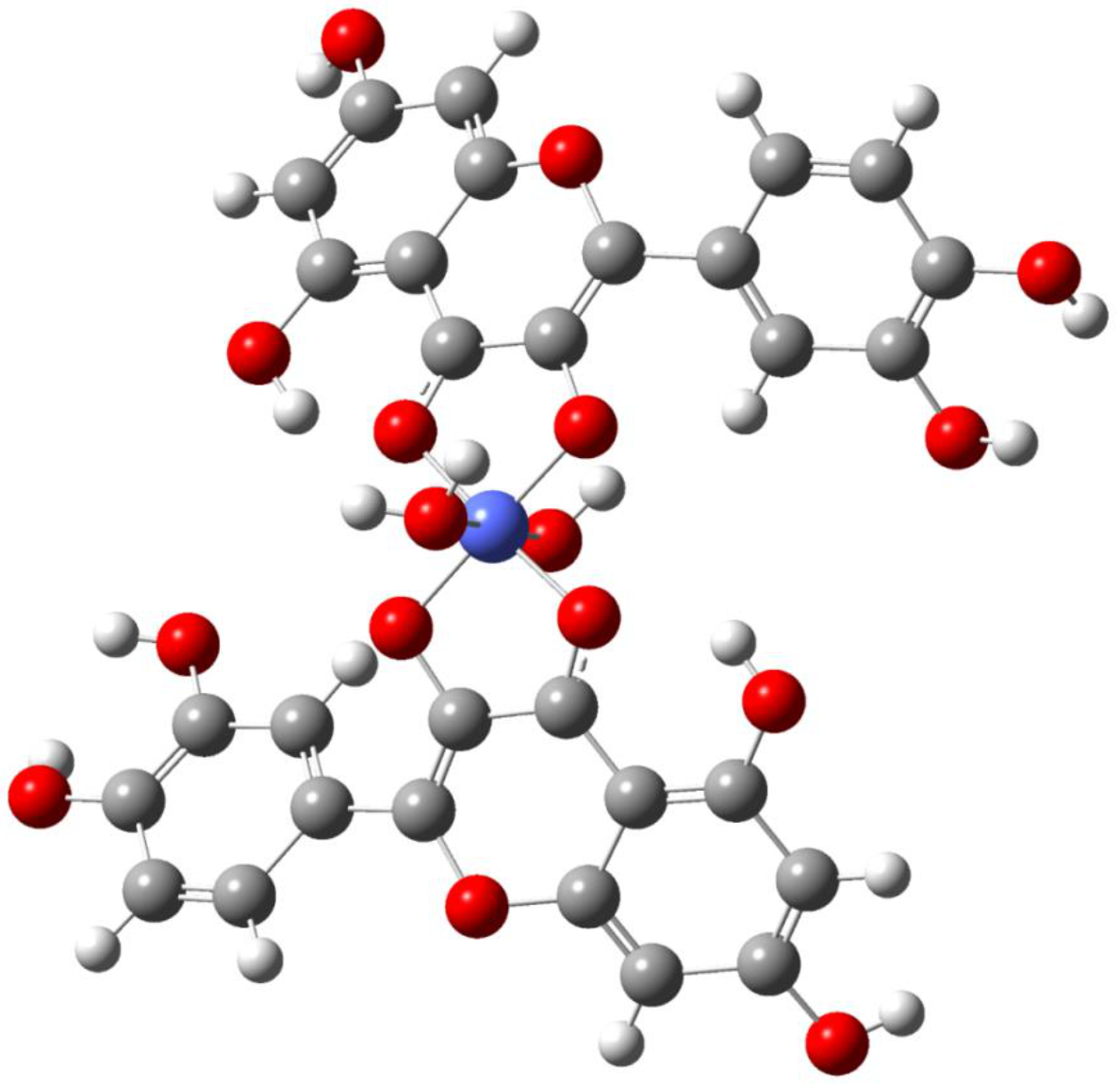
3.1. Spectral Analysis
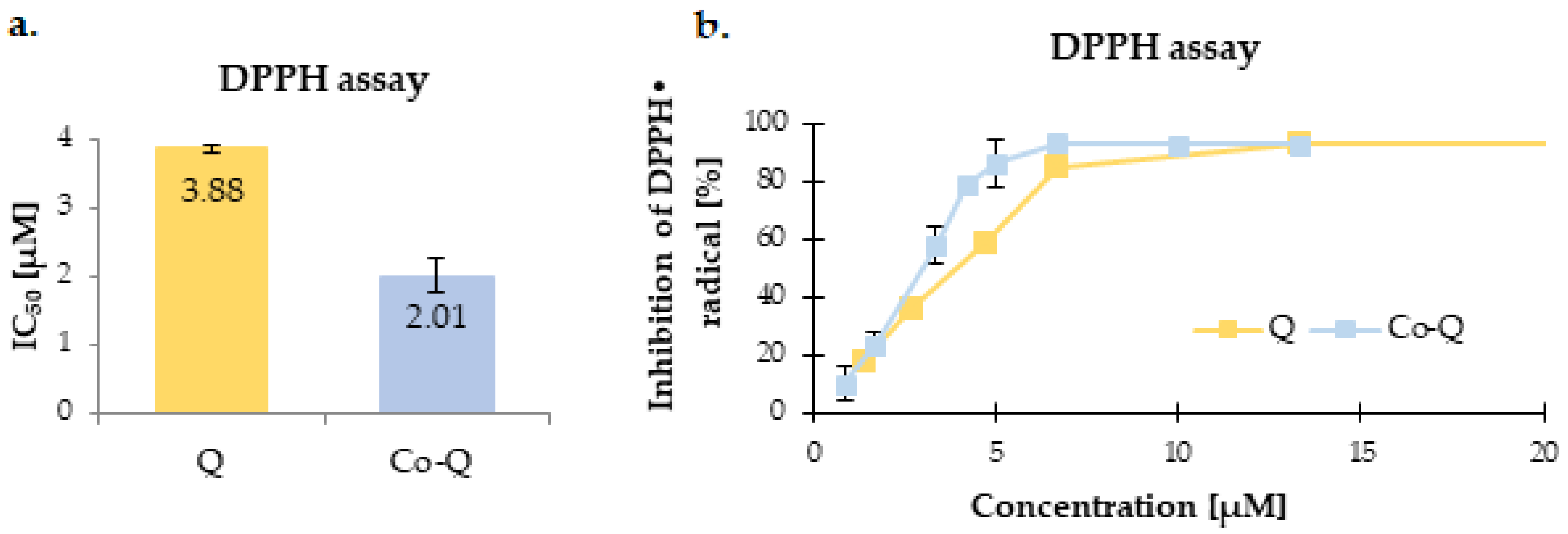
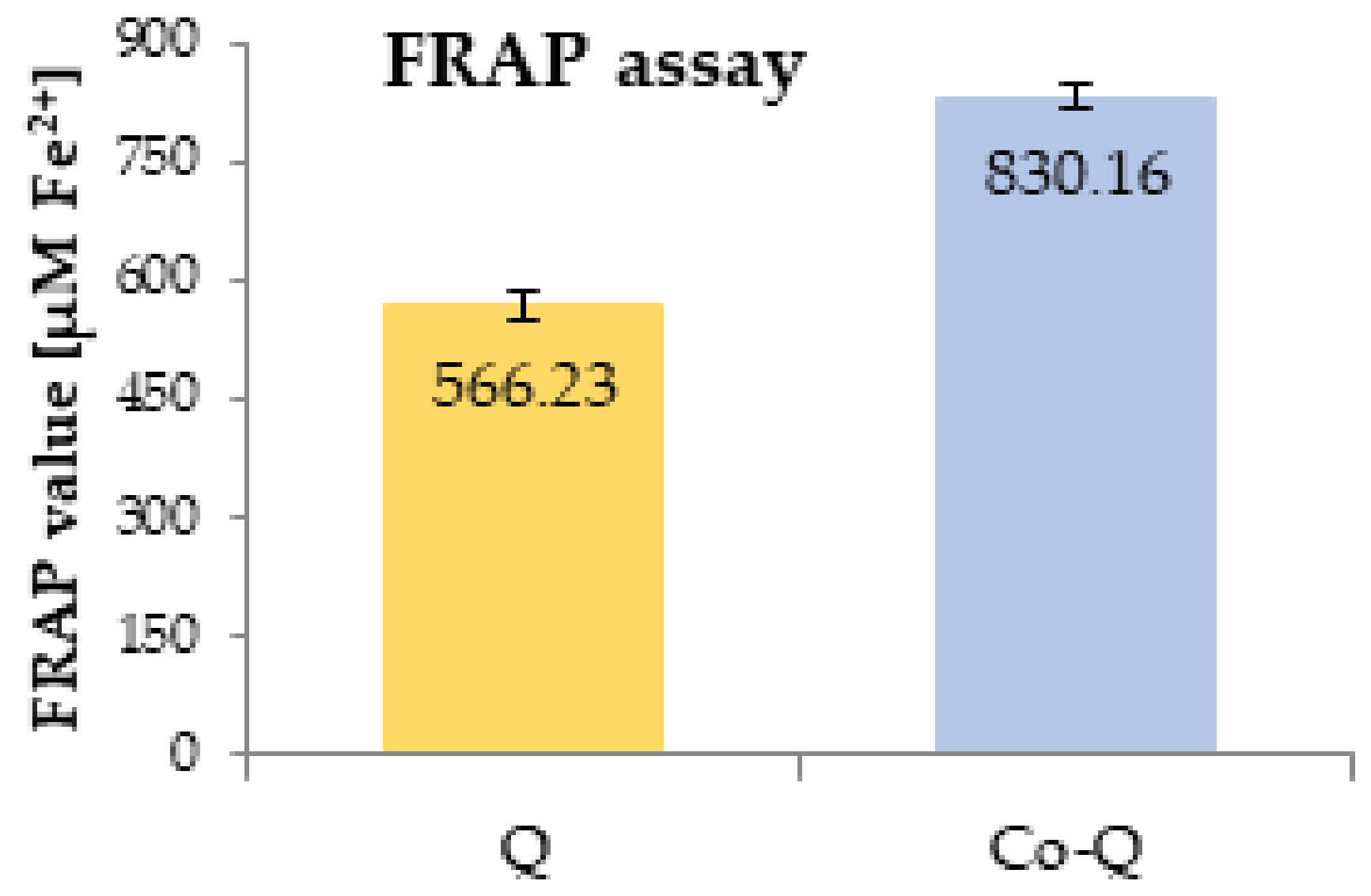
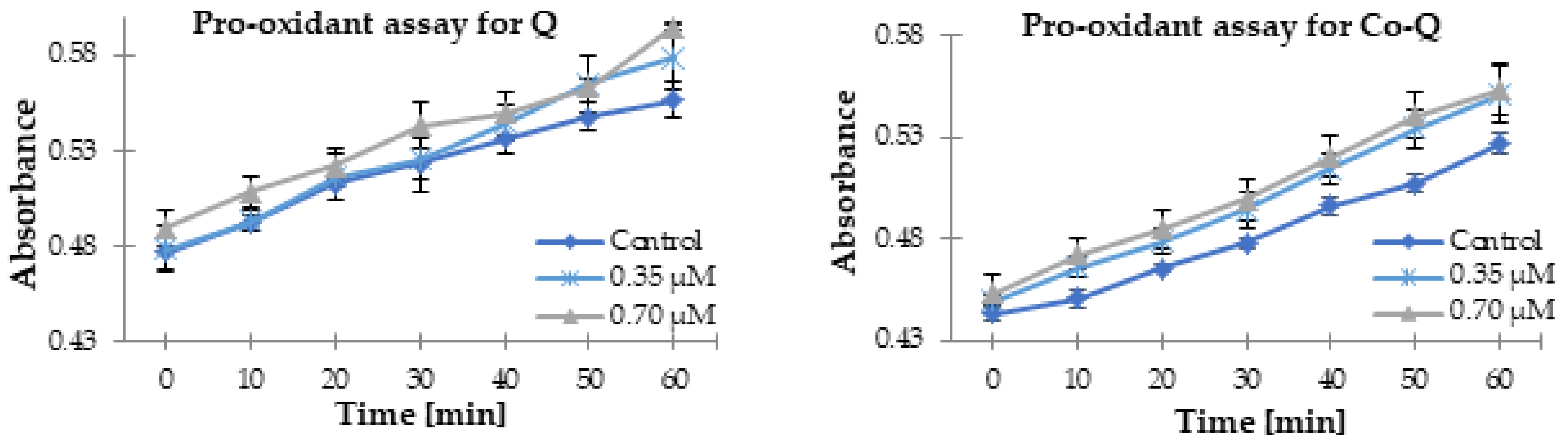
3.2. Antioxidant Activity
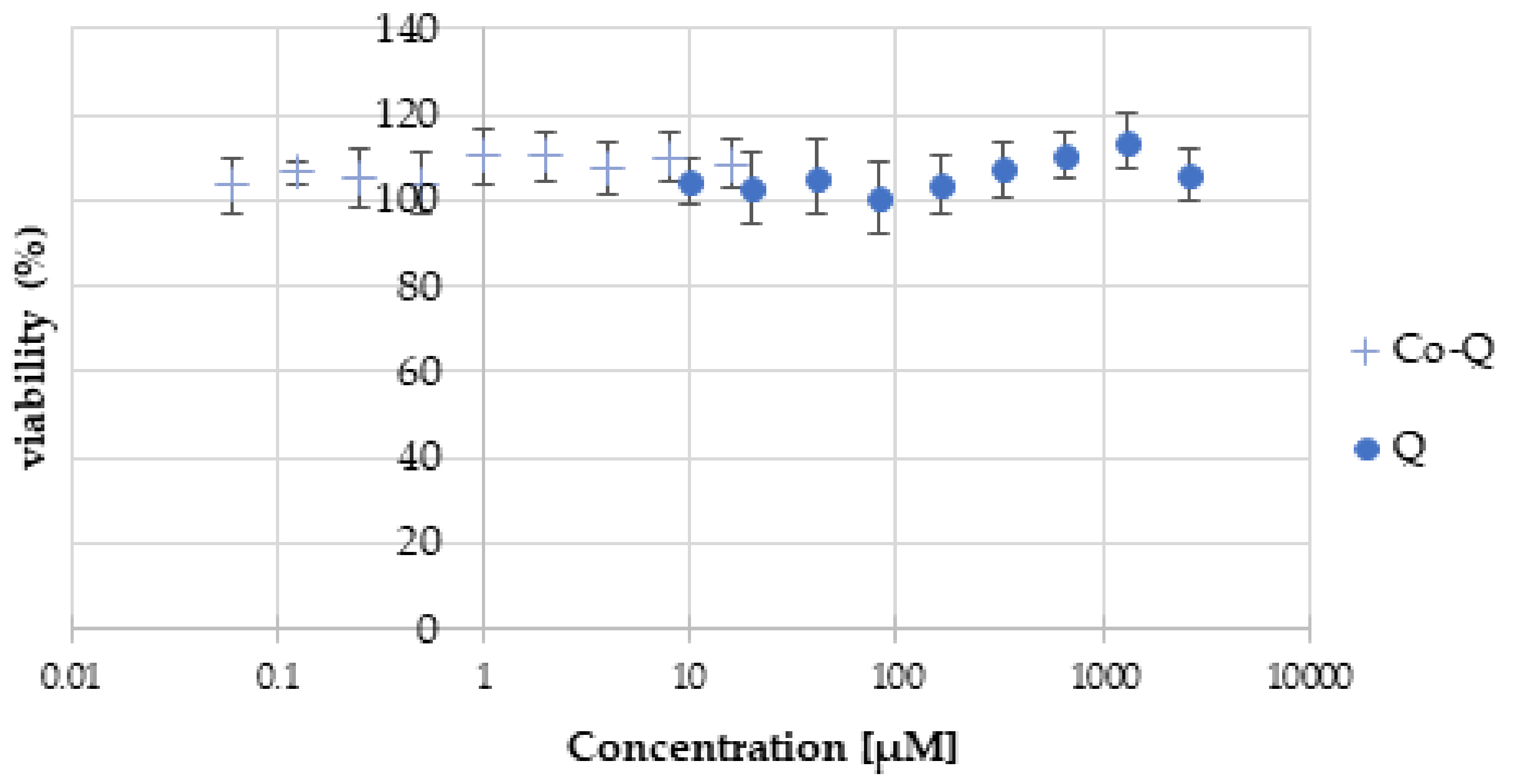
3.3. Cell Viability
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Singh, P.; Arif, Y.; Bajguz, A.; Hayat, S. The role of quercetin in plants. Plant Phys. Biochem. 2021, 166, 10–19. [Google Scholar] [CrossRef] [PubMed]
- Rauf, A.; Imran, M.; Khan, I.A.; Ur-Rehman, M.; Gilani, S.A.; Mehmood, Z.; Mubarak, M.S. Anticancer potential of quercetin: A comprehensive review. Phytother. Res. 2018, 32, 2109–2130. [Google Scholar] [CrossRef] [PubMed]
- David, A.V.A.; Arulmoli, R.; Parasuraman, S. Overviews of Biological Importance of Quercetin: A Bioactive Flavonoid. Pharmacogn. Rev. 2016, 10, 84–89. [Google Scholar]
- Kumar, R.; Vijayalakshmi, S.; Nadanasabapathi, S. Health Benefits of Quercetin. Def. Life Sci. J. 2017, 2, 142–151. [Google Scholar] [CrossRef] [Green Version]
- Shoskes, D.A.; Nickel, J.C. Quercetin for Chronic Prostatitis/Chronic Pelvic Pain Syndrome. Urol. Clin. N. Am. 2011, 38, 279–284. [Google Scholar] [CrossRef]
- Salvamani, S.; Gunasekaran, B.; Shaharuddin, N.A.; Ahmad, S.A.; Shukor, M.Y. Antiartherosclerotic Effects of Plant Flavonoids. Biomed Res. Int. 2014, 2014, 480258. [Google Scholar] [CrossRef] [Green Version]
- Sultana, B.; Anwar, F. Flavonols (kaempeferol, quercetin, myricetin) contents of selected fruits, vegetables and medicinal plants. Food Chem. 2008, 108, 879–884. [Google Scholar] [CrossRef]
- Batiha, G.E.-S.; Beshbishy, A.M.; Ikram, M.; Mulla, Z.S.; El-Hack, M.E.A.; Taha, A.E.; Algammal, A.M.; Elewa, Y.H.A. The Pharmacological Activity, Biochemical Properties, and Pharmacokinetics of the Major Natural Polyphenolic Flavonoid: Quercetin. Foods 2020, 9, 374. [Google Scholar] [CrossRef] [Green Version]
- Murade, V.; Hase, D.; Deshmukh, K.; Pansambal, S. A comprehensive review of phytopharmacology of Ricinus Communis (Linn.). Int. J. Phytopharm. 2014, 5, 334. [Google Scholar]
- Liguori, I.; Russo, G.; Curcio, F.; Bulli, G.; Aran, L.; DELLA Morte, D.; Gargiulo, G.; Testa, G.; Cacciatore, F.; Bonaduce, D.; et al. Oxidative stress, aging, and diseases. Clin. Interv. Aging 2018, 13, 757–772. [Google Scholar] [CrossRef] [Green Version]
- Schnekenburger, M.; Diederich, M. Nutritional Epigenetic Regulators in the Field of Cancer: New Avenues for Chemopreventive Approaches. In Epigenetic Cancer Therapy; Academic Press: Cambridge, MA, USA, 2015; pp. 393–425. [Google Scholar]
- Salehi, B.; Machin, L.; Monzote, L.; Sharifi-Rad, J.; Ezzat, S.M.; Salem, M.A.; Merghany, R.; Mahdy, N.M.; Kılıç, C.S.; Sytar, O.; et al. Therapeutic Potential of Quercetin: New Insights and Perspectives for Human Health. ACS Omega 2020, 5, 11849–11872. [Google Scholar] [CrossRef]
- Haenen, G.R.; Paquay, J.B.; Korthouwer, R.E.; Bast, A. Peroxynitrite Scavenging by Flavonoids. Biochem. Biophys. Res. Commun. 1997, 236, 591–593. [Google Scholar] [CrossRef]
- Kumar, S.; Pandey, A.K. Chemistry and Biological Activities of Flavonoids: An Overview. Sci. World J. 2013, 2013, 1–16. [Google Scholar] [CrossRef] [Green Version]
- Raza, A.; Xu, X.; Xia, L.; Xia, C.; Tang, J.; Ouyang, Z. Quercetin-Iron Complex: Synthesis, Characterization, Antioxidant, DNA Binding, DNA Cleavage, and Antibacterial Activity Studies. J. Fluoresc. 2016, 26, 2023–2031. [Google Scholar] [CrossRef]
- Trifunschi, S.; Munteanu, M.F. Synthesis, Characterization and Antioxidant Activity of Cooper-Quercetin Complex and Iron-Quercetin Complex. Rev. Chim. 2018, 69, 2621–2624. [Google Scholar] [CrossRef]
- Trifunschi, S.; Chim, D.A. Synthesis, characterization and antioxidant activity of Co (II) and Cd (II) complexes with quercetin. Rev. Chim. 2016, 67, 2422–2424. [Google Scholar]
- Mallick, M.S.; Maitra, R.; Roy, R.; Ghosh, S.; Mallick, S. Synthesis characterization and study of antioxidant activity of luteolin–magnesium complex. UJPSR 2016, 2, 2454–3756. [Google Scholar]
- Alper, P.; Erkisa, M.; Genckal, H.M.; Sahin, S.; Ulukaya, E.; Ari, F. Synthesis, characterization, anticancer and antioxidant activity of new nickel(II) and copper(II) flavonoid complexes. J. Mol. Struct. 2019, 1196, 783–792. [Google Scholar] [CrossRef]
- Bukhari, S.B.; Memon, S.; Tahir, M.M.; Bhanger, M. Synthesis, characterization and investigation of antioxidant activity of cobalt–quercetin complex. J. Mol. Struct. 2008, 892, 39–46. [Google Scholar] [CrossRef]
- Bukhari, S.B.; Memon, S.; Mahroof-Tahir, M.; Bhanger, M.I. Synthesis, characterization and antioxidant activity copper-quercetin complex. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2009, 71, 1901–1906. [Google Scholar] [CrossRef]
- Ravichandran, R.; Rajendran, M.; Devapiriam, D. Structural characterization and physicochemical properties of quercetin–Pb complex. J. Coord. Chem. 2014, 67, 1449–1462. [Google Scholar] [CrossRef]
- Tong, L.; Zhang, J.; Liu, Q.; Zhai, G. Sb(IIII)-Quercetin Complex: Synthesis, Characterization and Antioxidant Activity. Life Sci. J. 2016, 13, 127–133. [Google Scholar]
- Tan, J.; Wang, B.; Zhu, L. Hydrolytic cleavage of DNA by quercetin manganese(II) complexes. Colloids Surf. B Biointerfaces 2007, 55, 149–152. [Google Scholar]
- Lee, Y.-H.; Tuyet, P.-T. Synthesis and biological evaluation of quercetin–zinc (II) complex for anti-cancer and anti-metastasis of human bladder cancer cells. Vitr. Cell. Dev. Biol.-Anim. 2019, 55, 395–404. [Google Scholar] [CrossRef]
- Massi, A.; Bortolini, O.; Ragno, D.; Bernardi, T.; Sacchetti, G.; Tacchini, M.; Risi, C. De Research Progress in the Modification of Quercetin Leading to Anticancer Agents. Molecules 2017, 22, 1270. [Google Scholar] [CrossRef]
- Tabassum, S.; Zaki, M.; Afzal, M.; Arjmand, F. New modulated design and synthesis of quercetin–CuII/ZnII–Sn2IV scaffold as anticancer agents: In vitro DNA binding profile, DNA cleavage pathway and Topo-I activity. Dalton Trans. 2013, 42, 10029. [Google Scholar] [CrossRef]
- Zhou, J.; Wang, L.; Wang, J.; Tang, N. Antioxidative and anti-tumour activities of solid quercetin metal(II) complexes. Transit. Met. Chem. 2001, 26, 57–63. [Google Scholar] [CrossRef]
- Mateus, P.G.; Wolf, V.G.; Borges, M.S.; Ximenes, V.F. Chapter 9—Quercetin: Prooxidant Effect and Apoptosis in Cancer. Stud. Nat. Prod. Chem. 2018, 58, 265–288. [Google Scholar]
- Malacaria, L.; Corrente, G.; Beneduci, A.; Furia, E.; Marino, T.; Mazzone, G. A Review on Coordination Properties of Al(III) and Fe(III) toward Natural Antioxidant Molecules: Experimental and Theoretical Insights. Molecules 2021, 26, 2603. [Google Scholar] [CrossRef]
- Glade, M.J.; Meguid, M.M. A glance at… antioxidant and anti-inflammatory properties of dietary cobalt. Nutrition 2018, 46, 62–66. [Google Scholar] [CrossRef]
- Chang, E.L.; Simmers, C.; Knight, D.A. Cobalt Complexes as Antiviral and Antibacterial Agents. Pharmaceuticals 2010, 3, 1711–1728. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Czarnek, K.; Terpiłowska, S.; Siwicki, A.K. Selected aspects of the action of cobalt ions in the human body. Cent. Eur. J. Immunol. 2015, 40, 236–242. [Google Scholar] [CrossRef] [PubMed]
- Ajibade, T.O.; Oyagbemi, A.A.; Omobowale, T.; Asenuga, E.R.; Adigun, K.O. Quercetin and Vitamin C Mitigate Cobalt Chloride-Induced Hypertension through Reduction in Oxidative Stress and Nuclear Factor Kappa Beta (NF-Kb) Expression in Experimental Rat Model. Biol. Trace Elem. Res. 2016, 175, 347–359. [Google Scholar] [CrossRef] [PubMed]
- Repetto, G.; del Peso, A.; Zurita, J.L. Neutral red uptake assay for the estimation of cell viability/cytotoxicity. Nat. Protoc. 2008, 3, 1125–1131. [Google Scholar] [CrossRef]
- Kedare, S.B.; Singh, R.P. Genesis and development of DPPH method of antioxidant assay. J. Food Sci. Technol. 2011, 48, 412–422. [Google Scholar] [CrossRef] [Green Version]
- Rice-Evans, C.A.; Diplock, A.T.; Symons, M.C.R. Techniques in Free Radical Research; Elsevier: New York, NY, USA, 1991. [Google Scholar]
- Zeraik, M.L.; Petrônio, M.S.; Coelho, D.; Regasini, L.O.; Silva, D.; Da Fonseca, L.M.; Machado, S.A.S.; Bolzani, V.; Ximenes, V.F. Improvement of Pro-Oxidant Capacity of Protocatechuic Acid by Esterification. PLoS ONE 2014, 9, e110277. [Google Scholar] [CrossRef] [Green Version]
- Kalinowska, M.; Świderski, G.; Matejczyk, M.; Lewandowski, W. Spectroscopic, thermogravimetric and biological studies of Na(I), Ni(II) and Zn(II) complexes of quercetin. J. Therm. Anal. Calorim. 2016, 126, 141–148. [Google Scholar] [CrossRef] [Green Version]
- Lewandowski, W.; Kalinowska, M.; Lewandowska, H. The influence of metals on the electronic system of biologically important ligands. Spectroscopic study of benzoates, salicylates, nicotinates and isoorotates. Review. J. Inorg. Biochem. 2005, 99, 1407–1423. [Google Scholar] [CrossRef]
- Shastrala, K.; Kalam, S.; Damerakonda, K.; Sheshagiri, S.B.B.; Kumar, H.; Guda, R.; Kasula, M.; Bedada, S.K. Synthesis, characterization, and pharmacological evaluation of some metal complexes of quercetin as P-gp inhibitors. Futur. J. Pharm. Sci. 2021, 7, 1–13. [Google Scholar] [CrossRef]
- Spiegel, M.; Kapusta, K.; Kołodziejczyk, W.; Saloni, J.; Żbikowska, B.; Hill, G.A.; Sroka, Z. Antioxidant Activity of Selected Phenolic Acids–Ferric Reducing Antioxidant Power Assay and QSAR Analysis of the Structural Features. Molecules 2020, 25, 3088. [Google Scholar] [CrossRef]
- Foti, M.C. Use and Abuse of the DPPH• Radical. J. Agric. Food Chem. 2015, 63, 8765–8776. [Google Scholar] [CrossRef]
- Litwinienko, G.; Ingold, K.U. Abnormal Solvent Effects on Hydrogen Atom Abstractions. 1. The Reactions of Phenols with 2,2-Diphenyl-1-picrylhydrazyl (dpph•) in Alcohols. J. Org. Chem. 2003, 68, 3433–3438. [Google Scholar] [CrossRef] [Green Version]
- Litwinienko, G.; Ingold, K.U. Abnormal solvent effects on hydrogen atom abstraction. 2. Resolution of the curcumin antioxidant controversy. The role of sequential proton loss electron transfer. J. Org. Chem. 2004, 69, 5888–5896. [Google Scholar] [CrossRef]
- Kalinowska, M.; Gołębiewska, E.; Mazur, L.; Lewandowska, H.; Pruszyński, M.; Świderski, G.; Wyrwas, M.; Pawluczuk, N.; Lewandowski, W. Crystal Structure, Spectroscopic Characterization, Antioxidant and Cytotoxic Activity of New Mg(II) and Mn(II)/Na(I) Complexes of Isoferulic Acid. Materials 2021, 14, 3236. [Google Scholar] [CrossRef]
- Suganthy, N.; Devi, K.P.; Nabavi, S.F.; Braidy, N. Bioactive effects of quercetin in the central nervous system: Focusing on the mechanisms of actions. Biomed. Pharmacother. 2016, 84, 892–908. [Google Scholar] [CrossRef]
- Suematsu, N.; Hosoda, M.; Fujimori, K. Protective effects of quercetin against hydrogen peroxide-induced apoptosis in human neuronal SH-SY5Y cells. Neurosci. Lett. 2011, 504, 223–227. [Google Scholar] [CrossRef]
- Xu, D.; Hu, M.-J.; Wang, Y.-Q.; Cui, Y.-L. Antioxidant Activities of Quercetin and Its Complexes for Medicinal Application. Molecules 2019, 24, 1123. [Google Scholar] [CrossRef] [Green Version]
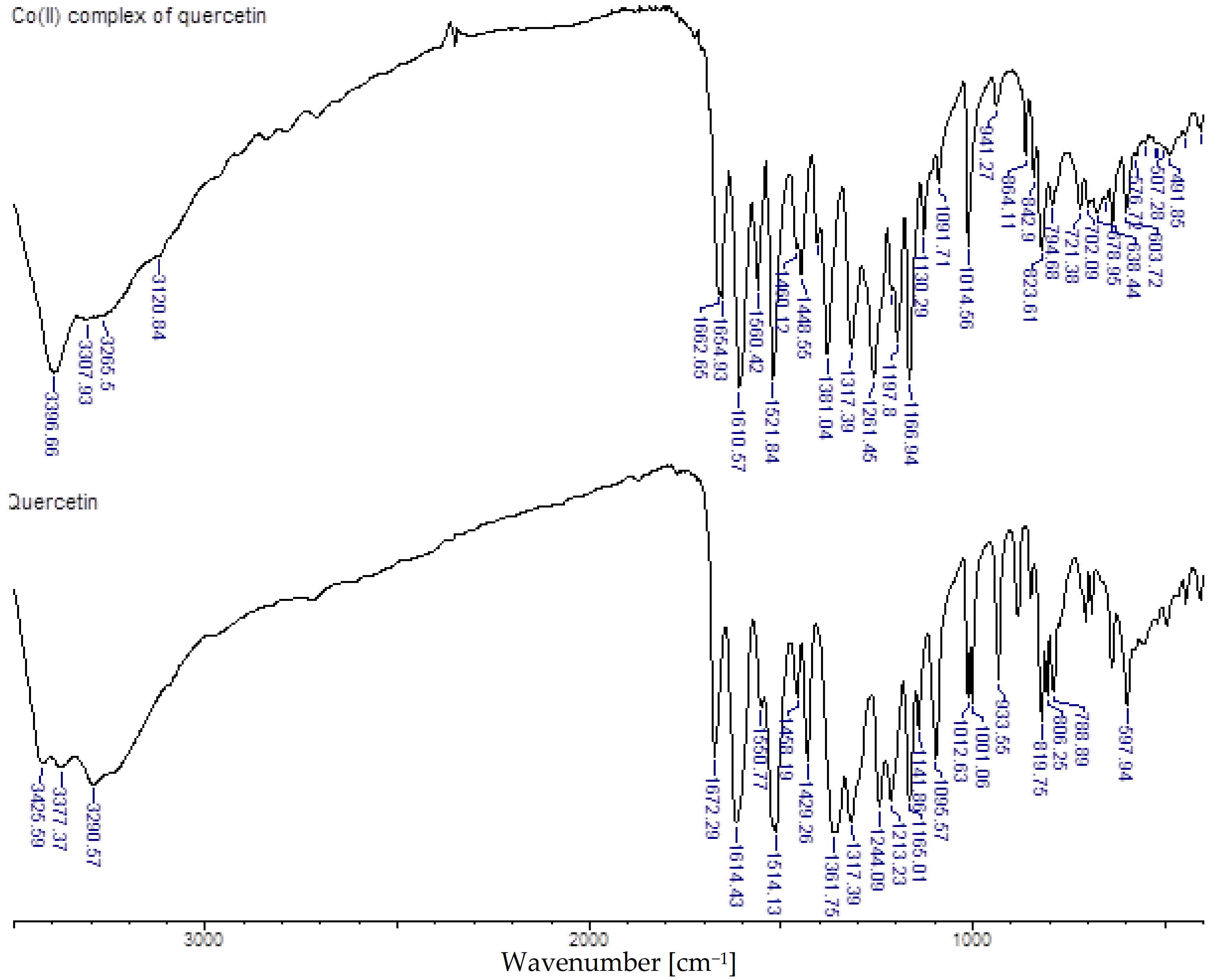
| Assignment | Quercetin | Co(II)-Quercetin | ||
|---|---|---|---|---|
| IR | Int. | IR | Int. | |
| ν(OH) * | 3426–3291 | s | 3397–3266 | s |
| ν(C=O) | 1672 | s | 1663, 1655 | s |
| ν ring | 1614 | vs | 1611 | vs |
| ν ring | 1514 | vs | 1522 | vs |
| ν(C-O) catechol group | - | 1460 | m | |
| ν ring | 1429 | s | 1449 | s |
| ν(C-O) catechol group | - | 1422 | s | |
| β(C-OH) | 1362 | vs | - | |
| β(CH) | 1317 | s | 1317 | s |
| ν(C-O-C) | 1244 | vs | 1261 | vs |
| β(OH) | 1213 | vs | 1210 | m |
| ν(C-CO-C) + β(C-CO-C) | 1165 | vs | 1167 | s |
| ν(Co(II)-O) | 604 | w | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kalinowska, M.; Lewandowska, H.; Pruszyński, M.; Świderski, G.; Gołębiewska, E.; Gryko, K.; Braun, J.; Borkowska, M.; Konieczna, M.; Lewandowski, W. Co(II) Complex of Quercetin–Spectral, Anti-/Pro-Oxidant and Cytotoxic Activity in HaCaT Cell Lines. Appl. Sci. 2021, 11, 9244. https://doi.org/10.3390/app11199244
Kalinowska M, Lewandowska H, Pruszyński M, Świderski G, Gołębiewska E, Gryko K, Braun J, Borkowska M, Konieczna M, Lewandowski W. Co(II) Complex of Quercetin–Spectral, Anti-/Pro-Oxidant and Cytotoxic Activity in HaCaT Cell Lines. Applied Sciences. 2021; 11(19):9244. https://doi.org/10.3390/app11199244
Chicago/Turabian StyleKalinowska, Monika, Hanna Lewandowska, Marek Pruszyński, Grzegorz Świderski, Ewelina Gołębiewska, Kamila Gryko, Julia Braun, Monika Borkowska, Magda Konieczna, and Włodzimierz Lewandowski. 2021. "Co(II) Complex of Quercetin–Spectral, Anti-/Pro-Oxidant and Cytotoxic Activity in HaCaT Cell Lines" Applied Sciences 11, no. 19: 9244. https://doi.org/10.3390/app11199244
APA StyleKalinowska, M., Lewandowska, H., Pruszyński, M., Świderski, G., Gołębiewska, E., Gryko, K., Braun, J., Borkowska, M., Konieczna, M., & Lewandowski, W. (2021). Co(II) Complex of Quercetin–Spectral, Anti-/Pro-Oxidant and Cytotoxic Activity in HaCaT Cell Lines. Applied Sciences, 11(19), 9244. https://doi.org/10.3390/app11199244










