The Biocatalytic Degradation of Organic Dyes Using Laccase Immobilized Magnetic Nanoparticles
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Synthesis of Fe3O4 Magnetic Nanoparticles
2.3. Synthesis of Copper Ferrite (CuFe2O4) Magnetic Nanoparticles
2.4. Immobilization of Laccase on Synthesized Nanoparticles
2.5. Assay for Laccase Activity
2.6. The Stability of Free and Immobilized Laccase
2.6.1. Temperature and pH Effect
2.6.2. Storage Stability
2.6.3. Kinetic Parameters
2.7. Biodegradation of DR23 Dye
3. Results and Discussion
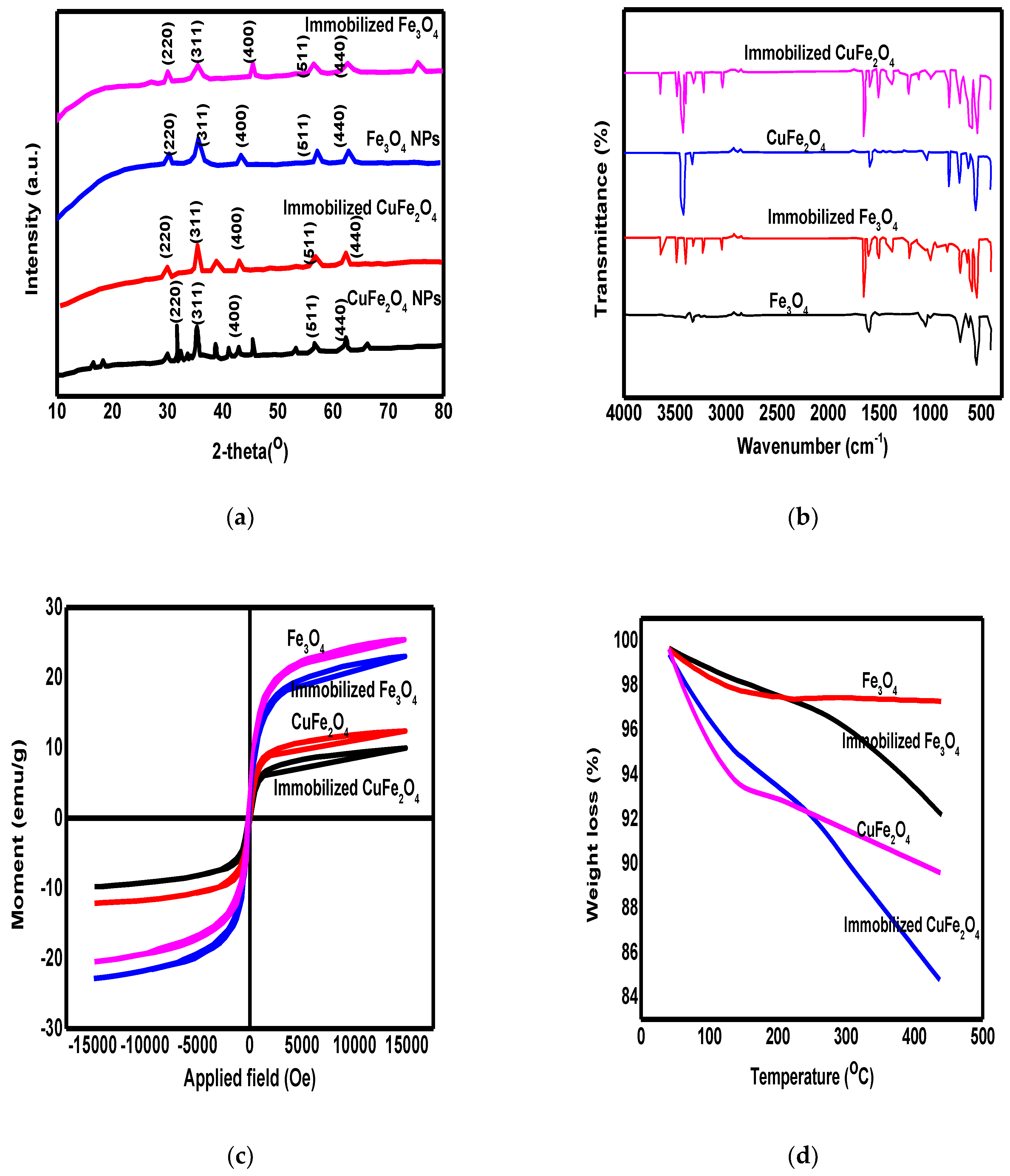
3.1. The Characterization of Synthesized Materials
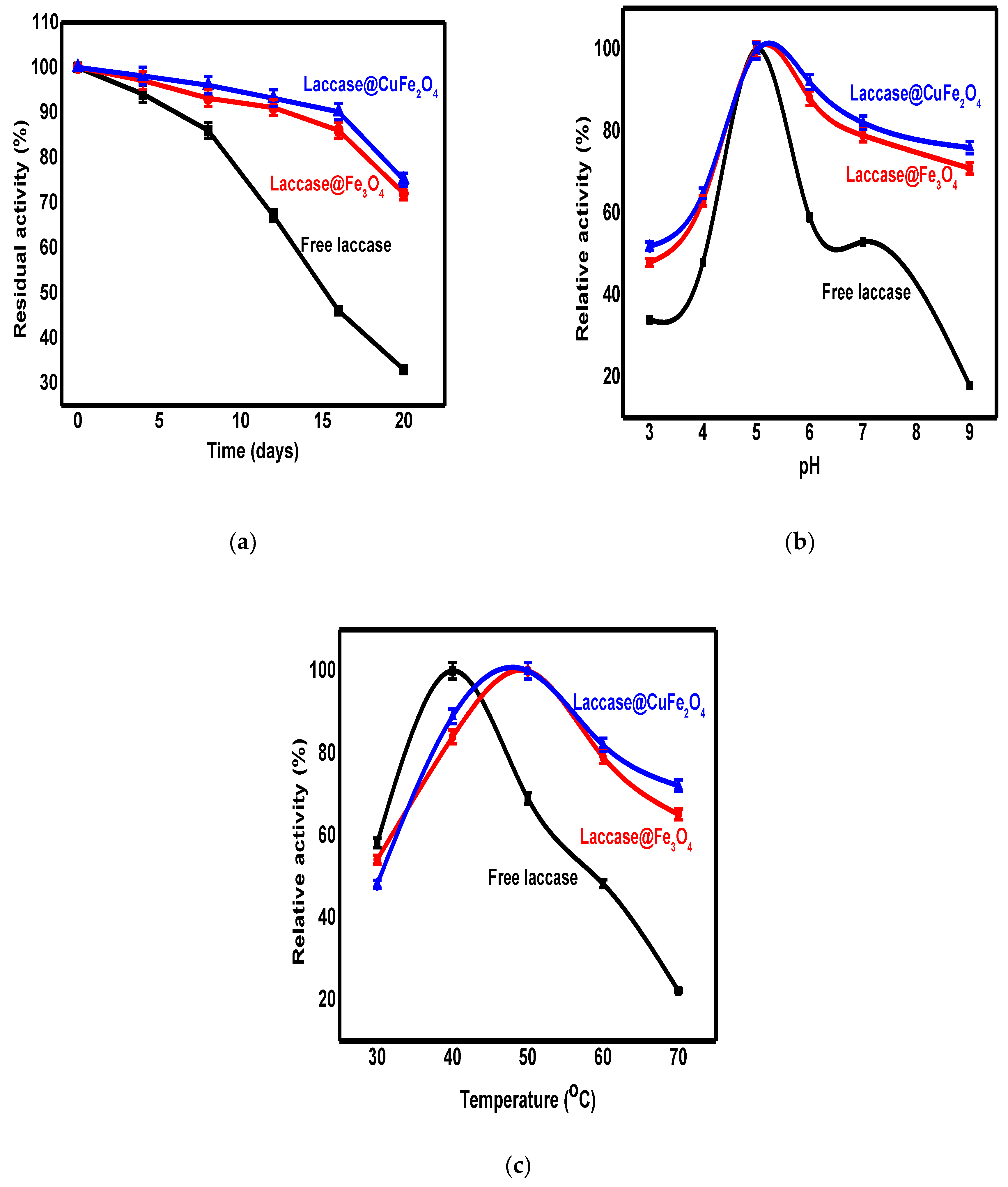
3.2. The Stability of Free and Immobilized Laccase
3.2.1. Storage Stability of Free and Immobilized Laccase
3.2.2. The pH Effect
3.2.3. The Temperature Effect
3.2.4. Kinetic Parameters
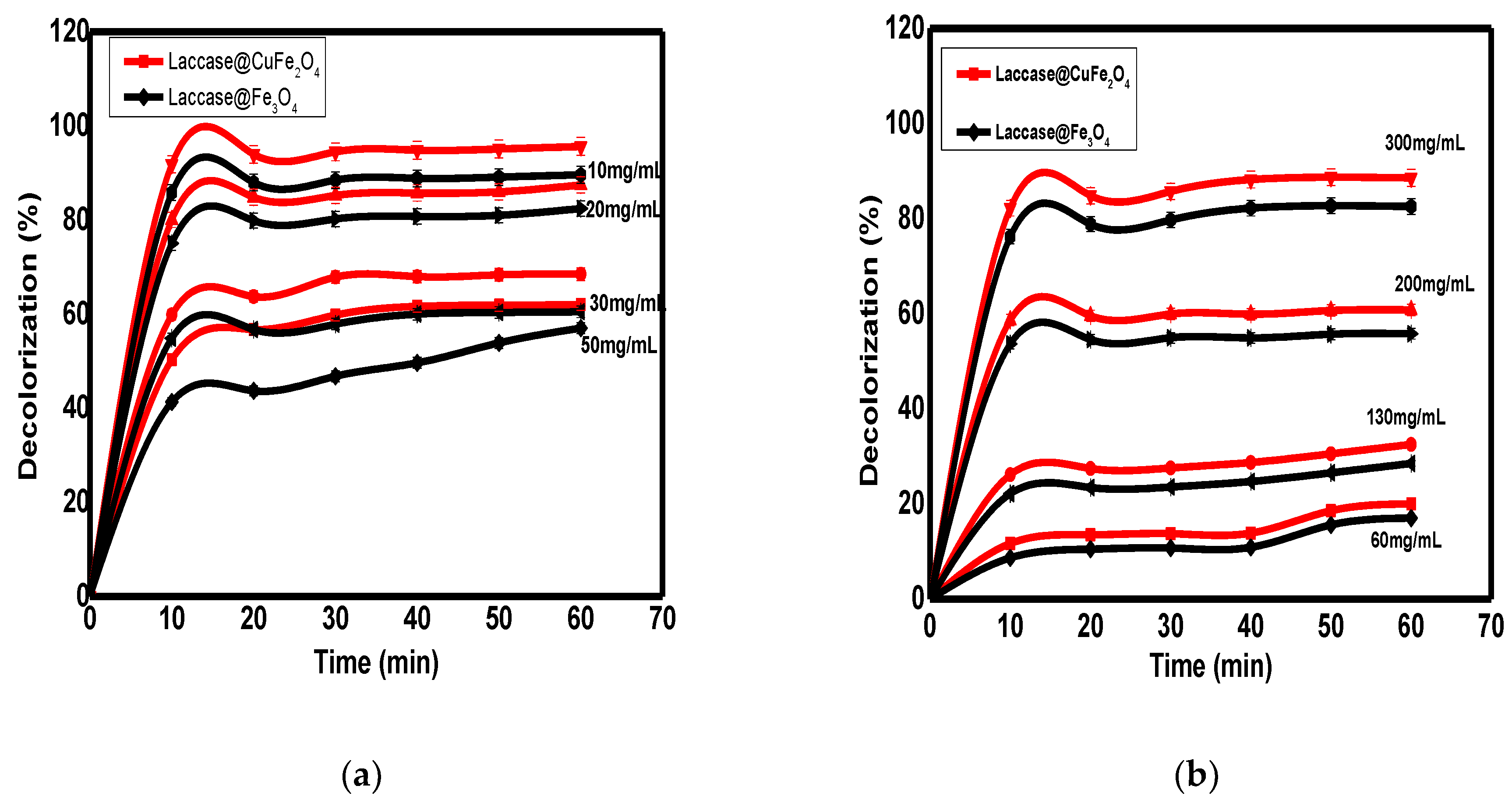
3.3. Biodegradation of DR23 Dye
3.3.1. Effect of Dye Concentration
3.3.2. Effect of Nanobiocatalyst Dosage
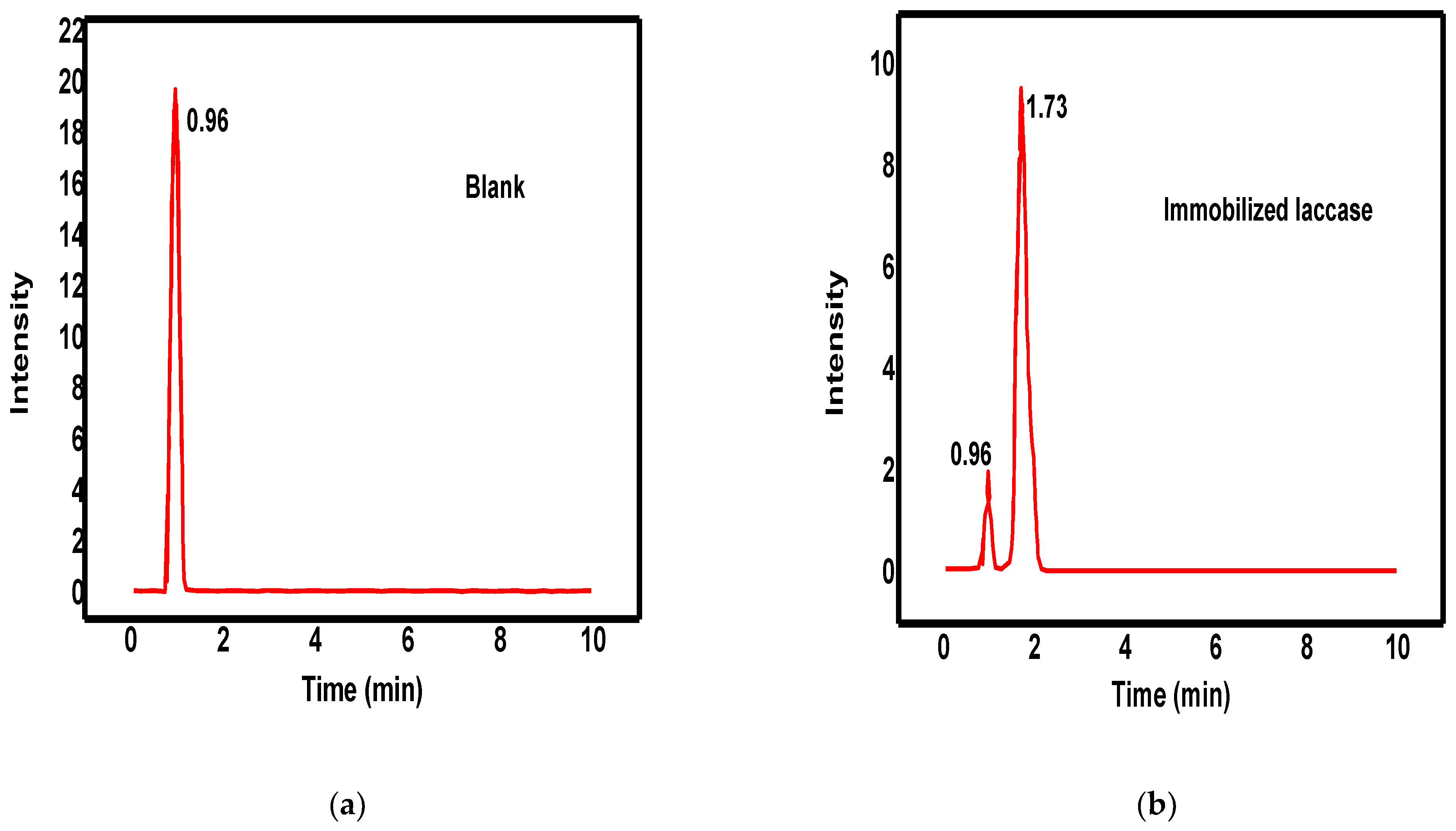
3.3.3. The Mechanism of DR23 Biodegradation
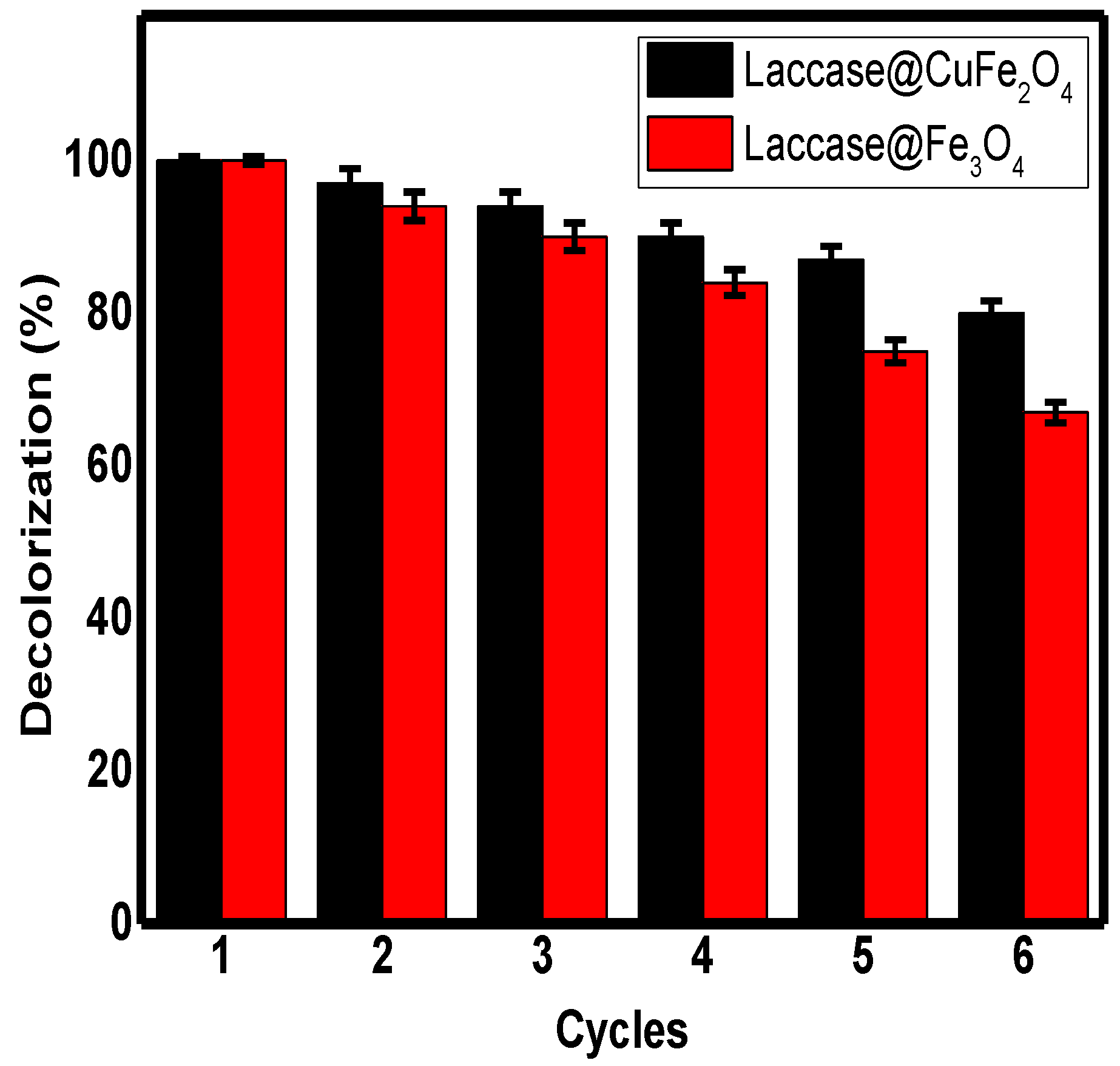
3.3.4. Reusability
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kour, D.; Kaur, T.; Devi, R.; Yadav, A.; Singh, M.; Joshi, D.; Singh, J.; Suyal, D.C.; Kumar, A.; Rajput, V.D. Beneficial micro-biomes for bioremediation of diverse contaminated environments for environmental sustainability: Present status and future challenges. Environ. Sci. Pollut. Res. 2021, 28, 24917–24939. [Google Scholar] [CrossRef] [PubMed]
- Overdahl, K.E.; Gooden, D.; Bobay, B.; Getzinger, G.J.; Stapleton, H.M.; Ferguson, P.L. Characterizing azobenzene disperse dyes in commercial mixtures and children’s polyester clothing. Environ. Pollut. 2021, 287, 117299. [Google Scholar] [CrossRef] [PubMed]
- Alderete, B.L.; da Silva, J.; Godoi, R.; da Silva, F.R.; Taffarel, S.R.; da Silva, L.P.; Garcia, A.L.H.; Júnior, H.M.; de Amorim, H.L.N.; Picada, J.N. Evaluation of toxicity and mutagenicity of a synthetic effluent containing azo dye after Advanced Oxidation Process treatment. Chemosphere 2021, 263, 128291. [Google Scholar] [CrossRef]
- Rafiq, A.; Ikram, M.; Ali, S.; Niaz, F.; Khan, M.; Khan, Q.; Maqbool, M. Photocatalytic degradation of dyes using semiconductor photocatalysts to clean industrial water pollution. J. Ind. Eng. Chem. 2021, 97, 111–128. [Google Scholar] [CrossRef]
- Kishor, R.; Purchase, D.; Saratale, G.D.; Ferreira, L.F.R.; Bilal, M.; Iqbal, H.M.; Bharagava, R.N. Environment friendly degradation and detoxification of Congo red dye and textile industry wastewater by a newly isolated Bacillus cohnni (RKS9). Environ. Technol. Innov. 2021, 22, 101425. [Google Scholar] [CrossRef]
- Legerská, B.; Chmelová, D.; Ondrejovič, M. Decolourization and detoxification of monoazo dyes by laccase from the white-rot fungus Trametes versicolor. J. Biotechnol. 2018, 285, 84–90. [Google Scholar] [CrossRef] [PubMed]
- Morsi, R.; Al-Maqdi, K.A.; Bilal, M.; Iqbal, H.; Khaleel, A.; Shah, I.; Ashraf, S.S. Immobilized Soybean Peroxidase Hybrid Bi-ocatalysts for Efficient Degradation of Various Emerging Pollutants. Biomolecules 2021, 11, 904. [Google Scholar] [CrossRef]
- Aslam, S.; Asgher, M.; Khan, N.A.; Bilal, M. Immobilization of Pleurotus nebrodensis WC 850 laccase on glutaraldehyde cross-linked chitosan beads for enhanced biocatalytic degradation of textile dyes. J. Water Process. Eng. 2021, 40, 101971. [Google Scholar] [CrossRef]
- Svetozarević, M.; Šekuljica, N.; Knežević-Jugović, Z.; Mijin, D. Agricultural waste as a source of peroxidase for wastewater treatment: Insight in kinetics and process parameters optimization for anthraquinone dye removal. Environ. Technol. Innov. 2021, 21, 101289. [Google Scholar] [CrossRef]
- Zhang, K.; Yang, W.; Liu, Y.; Zhang, K.; Chen, Y.; Yin, X. Laccase immobilized on chitosan-coated Fe3O4 nanoparticles as reusable biocatalyst for degradation of chlorophenol. J. Mol. Struct. 2020, 1220, 128769. [Google Scholar] [CrossRef]
- Tavares, A.P.; Silva, C.G.; Dražić, G.; Silva, A.; Loureiro, J.M.; Faria, J. Laccase immobilization over multi-walled carbon nanotubes: Kinetic, thermodynamic and stability studies. J. Colloid Interface Sci. 2015, 454, 52–60. [Google Scholar] [CrossRef]
- Mehandia, S.; Sharma, S.; Arya, S.K. Immobilization of laccase on chitosan-clay composite beads to improve its catalytic efficiency to degrade industrial dyes. Mater. Today Commun. 2020, 25, 101513. [Google Scholar] [CrossRef]
- Mohammadi, M.; As’Habi, M.A.; Salehi, P.; Yousefi, M.; Nazari, M.; Brask, J. Immobilization of laccase on epoxy-functionalized silica and its application in biodegradation of phenolic compounds. Int. J. Biol. Macromol. 2018, 109, 443–447. [Google Scholar] [CrossRef]
- García-Morales, R.; Garcia, A.; Orona-Navar, C.; Osma, J.F.; Nigam, K.; Ornelas-Soto, N. Biotransformation of emerging pollutants in groundwater by laccase from P. sanguineus CS43 immobilized onto titania nanoparticles. J. Environ. Chem. Eng. 2018, 6, 710–717. [Google Scholar] [CrossRef]
- Olshansky, Y.; Masaphy, S.; Root, R.A.; Rytwo, G. Immobilization of Rhus vernicifera laccase on sepiolite; effect of chitosan and copper modification on laccase adsorption and activity. Appl. Clay Sci. 2018, 152, 143–147. [Google Scholar] [CrossRef]
- Bansal, M.; Kumar, D.; Chauhan, G.S.; Kaushik, A. Preparation, characterization and trifluralin degradation of lac-case-modified cellulose nanofibers. Mater. Sci. Energy Technol. 2018, 1, 29–37. [Google Scholar]
- Jiang, X.; Yu, Y.; Li, X.; Kong, X.Z. High yield preparation of uniform polyurea microspheres through precipitation polymeri-zation and their application as laccase immobilization support. Chem. Eng. J. 2017, 328, 1043–1050. [Google Scholar] [CrossRef]
- Le, T.T.; Murugesan, K.; Lee, C.-S.; Vu, C.H.; Chang, Y.-S.; Jeon, J.-R. Degradation of synthetic pollutants in real wastewater using laccase encapsulated in core–shell magnetic copper alginate beads. Bioresour. Technol. 2016, 216, 203–210. [Google Scholar] [CrossRef] [PubMed]
- Alsaiari, N.; Alzahrani, F.; Katubi, K.; Amari, A.; Rebah, F.; Tahoon, M. Polyethylenimine-Modified Magnetic Chitosan for the Uptake of Arsenic from Water. Appl. Sci. 2021, 11, 5630. [Google Scholar] [CrossRef]
- Katubi, K.M.; Alzahrani, F.M.; Alsaiari, N.S.; Amari, A.; Rebah, F.B.; Tahoon, M.A. Partially Reduced Graphene Oxide Mod-ified with Polyacrylonitrile for the Removal of Sm3+ from Water. Processes 2021, 9, 818. [Google Scholar] [CrossRef]
- Alzahrani, F.; Alsaiari, N.; Katubi, K.; Amari, A.; Ben Rebah, F.; Tahoon, M. Synthesis of Polymer-Based Magnetic Nanocomposite for Multi-Pollutants Removal from Water. Polymers 2021, 13, 1742. [Google Scholar] [CrossRef]
- Amari, A.; Elboughdiri, N.; Ghernaout, D.; Lajimi, R.H.; Alshahrani, A.M.; Tahoon, M.A.; Rebah, F.B. Multifunctional cross-linked chitosan/nitrogen-doped graphene quantum dot for wastewater treatment. Ain Shams Eng. J. 2021, in press. [Google Scholar] [CrossRef]
- Alsaiari, N.S.; Katubi, K.M.M.; Alzahrani, F.M.; Siddeeg, S.M.; Tahoon, M.A. The application of nanomaterials for the elec-trochemical detection of antibiotics: A review. Micromachines 2021, 12, 308. [Google Scholar] [CrossRef]
- Siddeeg, S.M.; Alsaiari, N.S.; Tahoon, M.A.; Rebah, F.B. The application of nanomaterials as electrode modifiers for the elec-trochemical detection of ascorbic acid. Int. J. Electrochem. Sci. 2020, 15, 3327–3346. [Google Scholar] [CrossRef]
- Rani, M.; Shanker, U.; Chaurasia, A.K. Catalytic potential of laccase immobilized on transition metal oxides nanomaterials: Degradation of alizarin red S dye. J. Environ. Chem. Eng. 2017, 5, 2730–2739. [Google Scholar] [CrossRef]
- Amari, A.; Alzahrani, F.M.; Alsaiari, N.S.; Katubi, K.M.; Rebah, F.B.; Tahoon, M.A. Magnetic metal organic framework im-mobilized laccase for wastewater decolorization. Processes 2021, 9, 774. [Google Scholar] [CrossRef]
- Siddeeg, S.M.; Tahoon, M.A.; Mnif, W.; Ben Rebah, F. Iron Oxide/Chitosan Magnetic Nanocomposite Immobilized Manganese Peroxidase for Decolorization of Textile Wastewater. Processes 2019, 8, 5. [Google Scholar] [CrossRef] [Green Version]
- Solé, M.; Müller, I.; Pecyna, M.J.; Fetzer, I.; Harms, H.; Schlosser, D. Differential Regulation by Organic Compounds and Heavy Metals of Multiple Laccase Genes in the Aquatic Hyphomycete Clavariopsis aquatica. Appl. Environ. Microbiol. 2012, 78, 4732–4739. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guo, L.-Q.; Lin, S.-X.; Zheng, X.-B.; Huang, Z.-R.; Lin, J.-F. Production, purification and characterization of a thermostable laccase from a tropical white-rot fungus. World J. Microbiol. Biotechnol. 2010, 27, 731–735. [Google Scholar] [CrossRef]
- Maity, D.; Agrawal, D. Synthesis of iron oxide nanoparticles under oxidizing environment and their stabilization in aqueous and non-aqueous media. J. Magn. Magn. Mater. 2007, 308, 46–55. [Google Scholar] [CrossRef]
- Gholinejad, M.; Karimi, B.; Mansouri, F. Synthesis and characterization of magnetic copper ferrite nanoparticles and their catalytic performance in one-pot odorless carbon-sulfur bond formation reactions. J. Mol. Catal. A Chem. 2014, 386, 20–27. [Google Scholar] [CrossRef]
- Pavithradevi, S.; Suriyanarayanan, N.; Boobalan, T. Synthesis, structural, dielectric and magnetic properties of polyol assisted copper ferrite nano particles. J. Magn. Magn. Mater. 2017, 426, 137–143. [Google Scholar] [CrossRef]
- Nadar, S.S.; Rathod, V.K. A co-immobilization of pectinase and cellulase onto magnetic nanoparticles for antioxidant extraction from waste fruit peels. Biocatal. Agric. Biotechnol. 2019, 17, 470–479. [Google Scholar] [CrossRef]
- Kurian, J.; Mathew, M.J. Structural, optical and magnetic studies of CuFe2O4, MgFe2O4 and ZnFe2O4 nanoparticles prepared by hydrothermal/solvothermal method. J. Magn. Magn. Mater. 2018, 451, 121–130. [Google Scholar] [CrossRef]
- Jamatia, R.; Gupta, A.; Pal, A.K. Superparamagnetic Copper Ferrite Nanoparticles Catalyzed One Step Regioselective Synthesis of Dibenzodiazepinones via Ligand and Base Free Ullmann Type Coupling Reaction. Chem. Sel. 2016, 1, 852–860. [Google Scholar] [CrossRef]
- Muley, A.B.; Thorat, A.S.; Singhal, R.S.; Babu, K.H. A tri-enzyme co-immobilized magnetic complex: Process details, kinetics, thermodynamics and applications. Int. J. Biol. Macromol. 2018, 118, 1781–1795. [Google Scholar] [CrossRef]
- Datta, R.S.; Gutteridge, A.; Swanton, C.; Maley, C.C.; Graham, T.A. Modelling the evolution of genetic instability during tumour progression. Evol. Appl. 2012, 6, 20–33. [Google Scholar] [CrossRef] [PubMed]
- Mohajershojaei, K.; Khosravi, A.; Mahmoodi, N.M. Decolorization of dyes using laccase enzyme from single and binary sys-tems. Desalination Water Treat. 2014, 52, 1895–1902. [Google Scholar] [CrossRef]
- Sayahi, E.; Ladhari, N.; Mechichi, T.; Sakli, F. Azo dyes decolourization by the laccase fromTrametes trogii. J. Text. Inst. 2015, 107, 1478–1482. [Google Scholar] [CrossRef]
- Katuri, K.P.; Mohan, S.V.; Sridhar, S.; Pati, B.; Sarma, P. Laccase-membrane reactors for decolorization of an acid azo dye in aqueous phase: Process optimization. Water Res. 2009, 43, 3647–3658. [Google Scholar] [CrossRef]
- Ramírez-Montoya, L.A.; Hernández-Montoya, V.; Montes-Morán, M.A.; Jáuregui-Rincón, J.; Cervantes, F.J. Decolorization of dyes with different molecular properties using free and immobilized laccases from Trametes versicolor. J. Mol. Liq. 2015, 212, 30–37. [Google Scholar] [CrossRef]
- Singh, R.L.; Singh, P.K.; Singh, R.P. Enzymatic decolorization and degradation of azo dyes–A review. Int. Biodeterior. Biodegrad. 2015, 104, 21–31. [Google Scholar] [CrossRef]
- Guerrero, E.; Aburto, P.; Terrés, E.; Villegas, O.; González, E.; Zayas, T.; Hernández, F.; Torres, E. Improvement of catalytic efficiency of chloroperoxidase by its covalent immobilization on SBA-15 for azo dye oxidation. J. Porous Mater. 2012, 20, 387–396. [Google Scholar] [CrossRef]
- Alsaiari, N.; Amari, A.; Katubi, K.; Alzahrani, F.; Rebah, F.; Tahoon, M. Innovative Magnetite Based Polymeric Nanocomposite for Simultaneous Removal of Methyl Orange and Hexavalent Chromium from Water. Processes 2021, 9, 576. [Google Scholar] [CrossRef]
- Siddeeg, S.M.; Amari, A.; Tahoon, M.A.; Alsaiari, N.S.; Rebah, F.B. Removal of meloxicam, piroxicam and Cd+2 by Fe3O4/SiO2/glycidyl methacrylate-S-SH nanocomposite loaded with laccase. Alex. Eng. J. 2020, 59, 905–914. [Google Scholar] [CrossRef]
- Alzahrani, F.M.; Alsaiari, N.S.; Katubi, K.M.; Amari, A.; Elkhaleefa, A.M.; Rebah, F.B.; Tahoon, M.A. Magnetic Nitro-gen-Doped Porous Carbon Nanocomposite for Pb (II) Adsorption from Aqueous Solution. Molecules 2021, 26, 4809. [Google Scholar] [CrossRef] [PubMed]
- Yang, D.; Wang, X.; Shi, J.; Wang, X.; Zhang, S.; Han, P.; Jiang, Z. In situ synthesized rGO–Fe3O4 nanocomposites as enzyme immobilization support for achieving high activity recovery and easy recycling. Biochem. Eng. J. 2016, 105, 273–280. [Google Scholar] [CrossRef]
- Gomaa, E.A.; Tahoon, M.A.; Negm, A. Aqueous micro-solvation of Li+ ions: Thermodynamics and energetic studies of Li+-(H2O)n (n = 1–6) structures. J. Mol. Liq. 2017, 241, 595–602. [Google Scholar] [CrossRef]
- Tahoon, M.; Gomaa, E.; Suleiman, M. Aqueous micro-hydration of Na+ (H2O)n = 1–7 clusters: DFT study. Open Chem. 2019, 17, 260–269. [Google Scholar]
- Gomaa, E.A.; Tahoon, M.A. Ion association and solvation behavior of copper sulfate in binary aqueous–methanol mixtures at different temperatures. J. Mol. Liq. 2016, 214, 19–23. [Google Scholar] [CrossRef]

| System | Vmax (U/mL) | Km (mg/mL) | R2 |
|---|---|---|---|
| Laccase@CuFe2O4 | 34.80 | 3.64 | 0.95 |
| Laccase@Fe3O4 | 30.93 | 3.76 | 0.97 |
| Free laccase | 33.60 | 3.65 | 0.96 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alsaiari, N.S.; Amari, A.; Katubi, K.M.; Alzahrani, F.M.; Harharah, H.N.; Rebah, F.B.; Tahoon, M.A. The Biocatalytic Degradation of Organic Dyes Using Laccase Immobilized Magnetic Nanoparticles. Appl. Sci. 2021, 11, 8216. https://doi.org/10.3390/app11178216
Alsaiari NS, Amari A, Katubi KM, Alzahrani FM, Harharah HN, Rebah FB, Tahoon MA. The Biocatalytic Degradation of Organic Dyes Using Laccase Immobilized Magnetic Nanoparticles. Applied Sciences. 2021; 11(17):8216. https://doi.org/10.3390/app11178216
Chicago/Turabian StyleAlsaiari, Norah Salem, Abdelfattah Amari, Khadijah Mohammedsaleh Katubi, Fatimah Mohammed Alzahrani, Hamed N. Harharah, Faouzi Ben Rebah, and Mohamed A. Tahoon. 2021. "The Biocatalytic Degradation of Organic Dyes Using Laccase Immobilized Magnetic Nanoparticles" Applied Sciences 11, no. 17: 8216. https://doi.org/10.3390/app11178216
APA StyleAlsaiari, N. S., Amari, A., Katubi, K. M., Alzahrani, F. M., Harharah, H. N., Rebah, F. B., & Tahoon, M. A. (2021). The Biocatalytic Degradation of Organic Dyes Using Laccase Immobilized Magnetic Nanoparticles. Applied Sciences, 11(17), 8216. https://doi.org/10.3390/app11178216










