Polyphenoloxidase (PPO): Effect, Current Determination and Inhibition Treatments in Fresh-Cut Produce
Abstract
:Featured Application
Abstract
1. Introduction
1.1. Fresh-Cut Fruits and Vegetables
1.2. Main Deterioration Aspects in Minimal Processing
2. PPO Effect on Fresh-Cut Produce and Its Mechanism
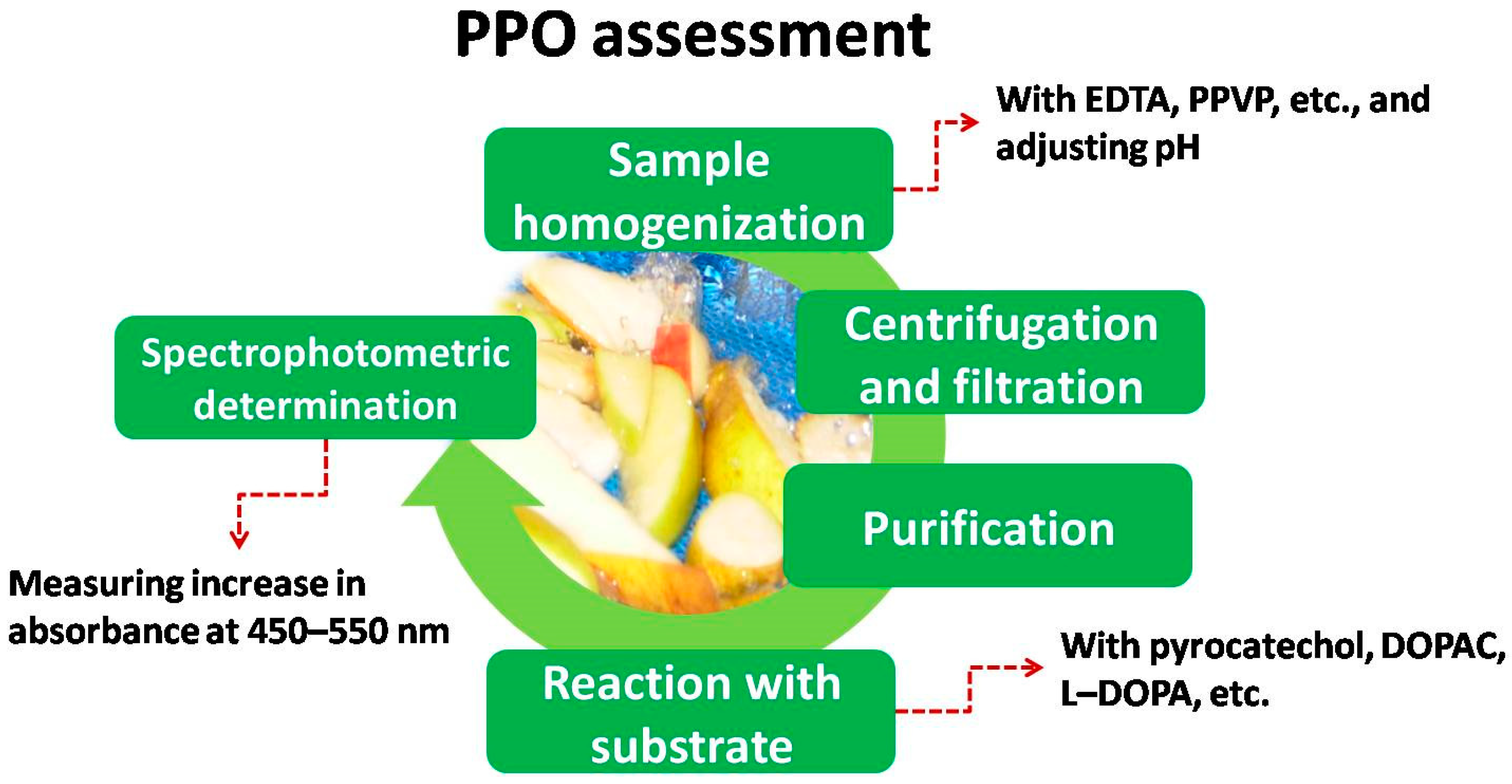
3. PPO Activity Assessment
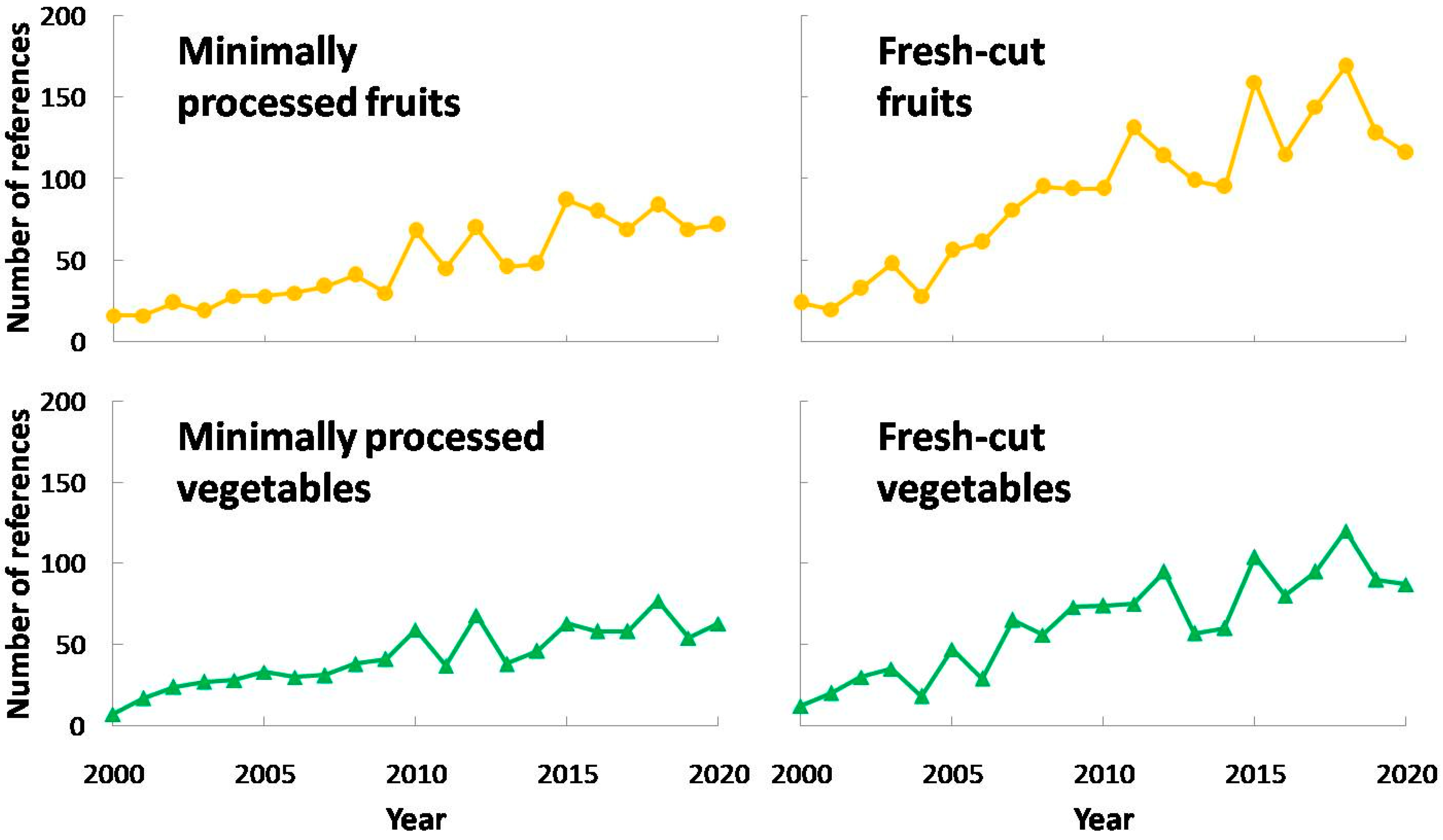
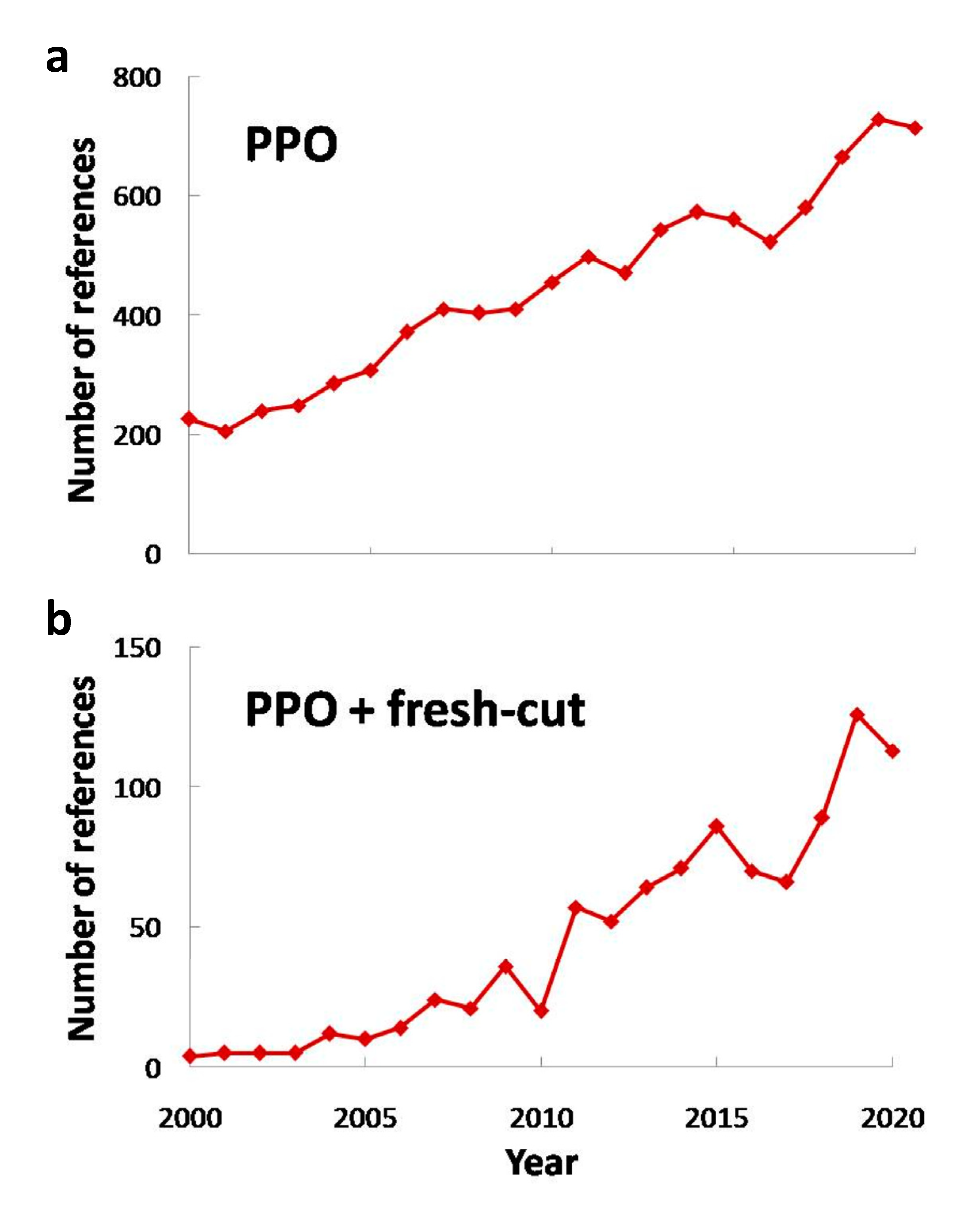
4. Current Studies Focused on PPO Activity
4.1. PPO Distribution
4.2. Fruit and Vegetables Characterizationand Processing
4.3. PPO Evolution in Fresh-Cut Produce
4.4. Influence on PPO Activity
4.4.1. Temperature
4.4.2. pH
4.4.3. Cell Integrity
4.4.4. Pre-Harvest Conditions
4.4.5. Microbial Growth and Decay
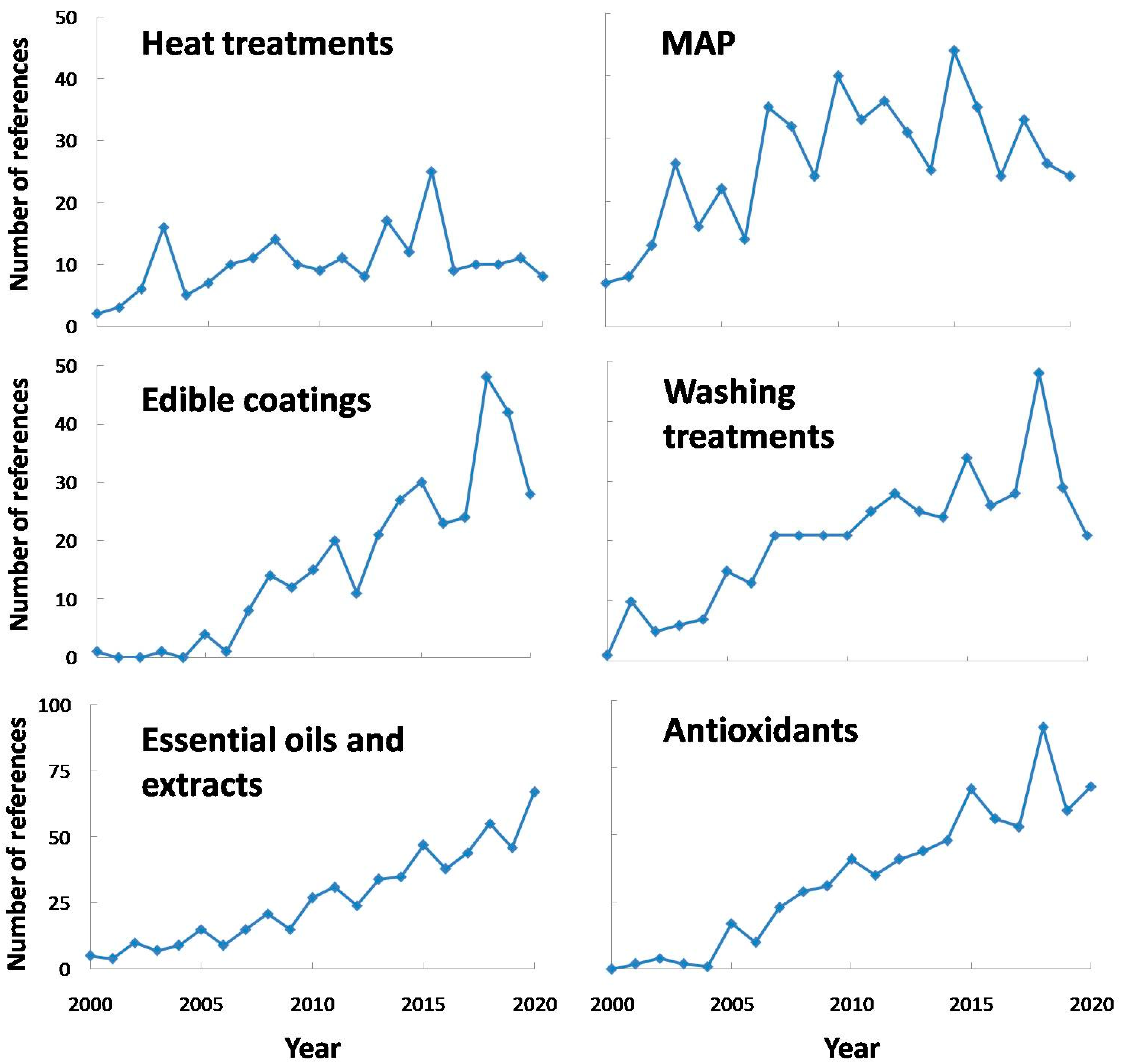
5. Current Treatments to Avoid or Reduce PPO Activity in Fresh-Cut Produce
5.1. Heat Treatments
5.2. Modified Atmosphere Packaging and Controlled Atmosphere
5.2.1. Traditional MAP or CA
5.2.2. High O2 Partial Pressure
5.3. Calcium Washing Treatments
5.4. Antioxidants and Antimicrobials
5.4.1. Ascorbic Acid
5.4.2. Citric Acid
5.4.3. Cysteine and Its Derivatives
5.4.4. Salicylic Acid
5.4.5. Whey Permeate
5.4.6. Hydrogen Peroxide
5.4.7. Ozonated Water
5.4.8. Neutral Electrolyzed Oxidizing Water (NEW)
5.4.9. Other Washing Treatments
5.5. Edible Coatings
5.6. Extracts and Essential Oils
5.7. Electromagnetic Radiations/Ultrasonication
5.7.1. Gamma Irradiation
5.7.2. Pulsed Light
5.7.3. High Intensity Light (HIL)
5.7.4. Ultraviolet Treatments
5.7.5. Ultrasonication
5.8. High-Pressure Carbon Dioxide (HPCD) and Argon (HPA)
6. Conclusions and Future Needs
- The importance of pre-harvest conditions on PPO activity (among other enzymes) and quality deterioration. In that sense, many authors have pointed out the strong influence of these conditions in many aspects related to PPO activity, such as PPO or polyphenol content, firmness, etc.
- The specific characterization of cultivars in order to predict their performance during minimal processing, specifically regarding PPO and polyphenol content. This way, many authors have selected the most suitable fruit or vegetable cultivars to obtain high-quality fresh-cut produce.
- Selection and optimization of treatments to keep quality of minimally processed fruits or vegetables. As explained in previous sections, many works have pointed out the importance of using optimum conditions for each treatment (for instance, the right % CO2 percentage in MAP or the suitable temperature and time treatment for blanching), both to improve the effectiveness of the treatment to decrease PPO activity and to avoid undesirable effects. It should have noted that, in some cases (especially when different conservation techniques are combined), an intermediary solution should be found.
- The combined use of conservative techniques. As many studies have proved, the use of two or three techniques can present additive or synergistic effects when it comes to PPO inhibition, due to the direct inhibition of the enzyme or indirect positive effects (such as microbial control or turgence promotion, which also results in a decrease in PPO activity). In that sense, the MAP and washing treatments seem to be suitable techniques to be combined with other innovative ones, such as irradiation, the use of active packaging, etc.
- Sustainability and circular economy. In most cases, optimization of operating parameters in most conservative techniques is necessary, not only in order to increase the effectiveness of each method, but also to reduce costs and develop industrial-scale strategies. Thus, the use of hurdle technologies can contribute to mild conditions, implying lower costs (especially concerning temperature or reagent addition). Another interesting aspect is the possible contribution to circular economy, by using some wastes to obtain interesting product for minimal processing (such as natural extracts or essential oils, among others).
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Nomenclature
| AA | Ascorbic Acid |
| CA | Controlled atmosphere |
| DOPAC | 3,4-Dihydroxyphenylacetic acid |
| EDTA | Ethylenediaminetetraacetic acid |
| GT | Green tea extract |
| HIL | High intensity light |
| HPA | High pressure argon |
| HPCD | High pressure carbon dioxide |
| MBTH | 3-Methyl-2-Benzothiazolinone Hydrazone |
| MAP | Modified atmosphere packaging |
| NEW | Neutral electrolyzed oxidizing water |
| POD | Peroxidase |
| PAL | Phenylalanine ammonia lyase |
| PMSF | Phenylmethylsulphonyl fluoride |
| PPO | Polyphenol oxidase |
| PVP | Polyvinyl pyrrolidone |
| PVPP | Polyvinyl polypyrrolidone |
| SA | Salicylic acid |
| US | Ultrasound |
| UV | Ultraviolet |
References
- Giovenzana, V.; Casson, A.; Beghi, R.; Pampuri, A.; Fiorindo, I.; Tugnolo, A.; Guidetti, R. Evaluation of consumer domestic habits on the environmental impact of ready-to-eat and minimally processed fresh-cut lamb’s lettuce. Sustain. Prod. Consum. 2021, 28, 925–935. [Google Scholar] [CrossRef]
- Baselice, A.; Colantuoni, F.; Lass, D.A.; Nardone, G.; Stasi, A. Trends in EU consumers’ attitude towards fresh-cut fruit and vegetables. Food Qual. Prefer. 2017, 59, 87–96. [Google Scholar] [CrossRef]
- Nogales-Delgado, S.; Fuentes-Pérez, M.C.; Bohoyo-Gil, D. Cultivar characterization of stone fruits for their minimal processing. J. Food Sci. Technol. 2015, 52. [Google Scholar] [CrossRef] [Green Version]
- Fuentes-Pérez, M.D.C.; Nogales-Delgado, S.; Ayuso, M.C.; Bohoyo-Gil, D. Different peach cultivars and their suitability for minimal processing. Czech J. Food Sci. 2014, 32, 2193–2201. [Google Scholar] [CrossRef] [Green Version]
- Nogales-Delgado, S.; Fernández-León, A.M.; Delgado-Adámez, J.; Hernández-Méndez, M.T.; Bohoyo Gil, D. Effects of several sanitisers for improving quality attributes of minimally processed fragaria vesca strawberry. Czech J. Food Sci. 2013, 31, 49–54. [Google Scholar] [CrossRef] [Green Version]
- Nogales-Delgado, S.; Fuentes-Pérez, M.C.; Ayuso-Yuste, C.; Bohoyo-Gil, D. Study of different nectarine cultivars and their suitability for fresh-cut processing. Int. J. Food Sci. Technol. 2014, 49, 114–120. [Google Scholar] [CrossRef]
- SCOPUS Scopus Preview. Available online: https://www.scopus.com/home.uri (accessed on 30 July 2021).
- Rico, D.; Martín-Diana, A.B.; Barat, J.M.; Barry-Ryan, C. Extending and measuring the quality of fresh-cut fruit and vegetables: A review. Trends Food Sci. Technol. 2007, 18, 373–386. [Google Scholar] [CrossRef] [Green Version]
- Hodges, D.M.; Toivonen, P.M.A. Quality of fresh-cut fruits and vegetables as affected by exposure to abiotic stress. Postharvest Biol. Technol. 2008, 48, 155–162. [Google Scholar] [CrossRef]
- Li-Qin, Z.; Jie, Z.; Shu-Hua, Z.; Lai-Hui, G. Inhibition of browning on the surface of peach slices by short-term exposure to nitric oxide and ascorbic acid. Food Chem. 2009, 114, 174–179. [Google Scholar] [CrossRef]
- Toivonen, P.M.A.; Brummell, D.A. Biochemical bases of appearance and texture changes in fresh-cut fruit and vegetables. Postharvest Biol. Technol. 2008, 48, 1–14. [Google Scholar] [CrossRef]
- Oms-Oliu, G.; Rojas-Graü, M.A.; González, L.A.; Varela, P.; Soliva-Fortuny, R.; Hernando, M.I.H.; Munuera, I.P.; Fiszman, S.; Martín-Belloso, O. Recent approaches using chemical treatments to preserve quality of fresh-cut fruit: A review. Postharvest Biol. Technol. 2010, 57, 139–148. [Google Scholar] [CrossRef]
- Moreno, J.; Chiralt, A.; Escriche, I.; Serra, J.A. Effect of blanching/osmotic dehydration combined methods on quality and stability of minimally processed strawberries. Food Res. Int. 2000, 33, 609–616. [Google Scholar] [CrossRef]
- Cefola, M.; D’Antuono, I.; Pace, B.; Calabrese, N.; Carito, A.; Linsalata, V.; Cardinali, A. Biochemical relationships and browning index for assessing the storage suitability of artichoke genotypes. Food Res. Int. 2012, 48, 397–403. [Google Scholar] [CrossRef]
- Martin-Diana, A.B.; Rico, D.; Barry-Ryan, C.; Frias, J.M.; Mulcahy, J.; Henehan, G.T.M. Calcium lactate washing treatments for salad-cut Iceberg lettuce: Effect of temperature and concentration on quality retention parameters. Food Res. Int. 2005, 38, 729–740. [Google Scholar] [CrossRef]
- Peng, L.; Yang, S.; Li, Q.; Jiang, Y.; Joyce, D.C. Hydrogen peroxide treatments inhibit the browning of fresh-cut Chinese water chestnut. Postharvest Biol. Technol. 2008, 47, 260–266. [Google Scholar] [CrossRef]
- Rico, D.; Martín-Diana, A.B.; Barry-Ryan, C.; Frías, J.M.; Henehan, G.T.M.; Barat, J.M. Optimisation of steamer jet-injection to extend the shelflife of fresh-cut lettuce. Postharvest Biol. Technol. 2008, 48, 431–442. [Google Scholar] [CrossRef] [Green Version]
- Bico, S.L.S.; Raposo, M.F.J.; Morais, R.M.S.C.; Morais, A.M.M.B. Combined effects of chemical dip and/or carrageenan coating and/or controlled atmosphere on quality of fresh-cut banana. Food Control 2009, 20, 508–514. [Google Scholar] [CrossRef]
- Chung, H.S.; Moon, K.D. Browning characteristics of fresh-cut “Tsugaru” apples as affected by pre-slicing storage atmospheres. Food Chem. 2009, 114, 1433–1437. [Google Scholar] [CrossRef]
- Martin-Diana, A.B.; Rico, D.; Frias, J.; Mulcahy, J.; Henehan, G.T.M.; Barry-Ryan, C. Whey permeate as a bio-preservative for shelf life maintenance of fresh-cut vegetables. Innov. Food Sci. Emerg. Technol. 2006, 7, 112–123. [Google Scholar] [CrossRef] [Green Version]
- Degl’Innocenti, E.; Pardossi, A.; Tognoni, F.; Guidi, L. Physiological basis of sensitivity to enzymatic browning in “lettuce”, “escarole” and “rocket salad” when stored as fresh-cut products. Food Chem. 2007, 104, 209–215. [Google Scholar] [CrossRef]
- Martín-Diana, A.B.; Rico, D.; Barry-Ryan, C. Green tea extract as a natural antioxidant to extend the shelf-life of fresh-cut lettuce. Innov. Food Sci. Emerg. Technol. 2008, 9, 593–603. [Google Scholar] [CrossRef] [Green Version]
- Albanese, D.; Cinquanta, L.; Di Matteo, M. Effects of an innovative dipping treatment on the cold storage of minimally processed Annurca apples. Food Chem. 2007, 105, 1054–1060. [Google Scholar] [CrossRef]
- Panadare, D.; Rathod, V.K. Extraction and purification of polyphenol oxidase: A review. Biocatal. Agric. BioTechnol. 2018, 14, 431–437. [Google Scholar] [CrossRef]
- Cabezas-Serrano, A.B.; Amodio, M.L.; Colelli, G. Effect of solution pH of cysteine-based pre-treatments to prevent browning of fresh-cut artichokes. Postharvest Biol. Technol. 2013, 75, 17–23. [Google Scholar] [CrossRef]
- Bi, X.; Wu, J.; Zhang, Y.; Xu, Z.; Liao, X. High pressure carbon dioxide treatment for fresh-cut carrot slices. Innov. Food Sci. Emerg. Technol. 2011, 12, 298–304. [Google Scholar] [CrossRef]
- Chauhan, O.P.; Raju, P.S.; Ravi, N.; Singh, A.; Bawa, A.S. Effectiveness of ozone in combination with controlled atmosphere on quality characteristics including lignification of carrot sticks. J. Food Eng. 2011, 102, 43–48. [Google Scholar] [CrossRef]
- Barbagallo, R.N.; Chisari, M.; Caputa, G. Effects of calcium citrate and ascorbate as inhibitors of browning and softening in minimally processed “Birgah” eggplants. Postharvest Biol. Technol. 2012, 73, 107–114. [Google Scholar] [CrossRef]
- Mishra, B.B.; Gautam, S.; Sharma, A. Browning of fresh-cut eggplant: Impact of cutting and storage. Postharvest Biol. Technol. 2012, 67, 44–51. [Google Scholar] [CrossRef]
- Chen, Z.; Zhu, C.; Zhang, Y.; Niu, D.; Du, J. Effects of aqueous chlorine dioxide treatment on enzymatic browning and shelf-life of fresh-cut asparagus lettuce (Lactuca sativa L.). Postharvest Biol. Technol. 2010, 58, 232–238. [Google Scholar] [CrossRef]
- Chisari, M.; Todaro, A.; Barbagallo, R.N.; Spagna, G. Salinity effects on enzymatic browning and antioxidant capacity of fresh-cut baby Romaine lettuce (Lactuca sativa L. cv. Duende). Food Chem. 2010, 119, 1502–1506. [Google Scholar] [CrossRef]
- Luna, M.C.; Tudela, J.A.; Martínez-Sánchez, A.; Allende, A.; Marín, A.; Gil, M.I. Long-term deficit and excess of irrigation influences quality and browning related enzymes and phenolic metabolism of fresh-cut iceberg lettuce (Lactuca sativa L.). Postharvest Biol. Technol. 2012, 73, 37–45. [Google Scholar] [CrossRef]
- Du, J.; Fu, Y.; Wang, N. Effects of aqueous chlorine dioxide treatment on browning of fresh-cut lotus root. LWT Food Sci. Technol. 2009, 42, 654–659. [Google Scholar] [CrossRef]
- Oms-Oliu, G.; Aguiló-Aguayo, I.; Martín-Belloso, O.; Soliva-Fortuny, R. Effects of pulsed light treatments on quality and antioxidant properties of fresh-cut mushrooms (Agaricus bisporus). Postharvest Biol. Technol. 2010, 56, 216–222. [Google Scholar] [CrossRef]
- Tanhaş, E.; Martin, E.; Korucu, E.N.; Dirmenci, T. Effect of aqueous extract, hydrosol, and essential oil forms of some endemic Origanum L. (Lamiaceae) taxa on polyphenol oxidase activity in fresh-cut mushroom samples. J. Food Process. Preserv. 2020, 44, e14726. [Google Scholar] [CrossRef]
- Cabezas-Serrano, A.B.; Amodio, M.L.; Cornacchia, R.; Rinaldi, R.; Colelli, G. Suitability of five different potato cultivars (Solanum tuberosum L.) to be processed as fresh-cut products. Postharvest Biol. Technol. 2009, 53, 138–144. [Google Scholar] [CrossRef]
- Zhang, Z.; Yao, Y.; Shi, Q.; Zhao, J.; Fu, H.; Wang, Y. Effects of radio-frequency-assisted blanching on the polyphenol oxidase, microstructure, physical characteristics, and starch content of potato. LWT 2020, 125, 109357. [Google Scholar] [CrossRef]
- Li, L.; Bai, J.; Wu, M.; Zhao, M.; Wang, R.; Guo, M.; Liu, H.; Liu, T. Studies on browning inhibition technology and mechanisms of fresh-cut potato. J. Food Process. Preserv. 2017, 41, 1–8. [Google Scholar] [CrossRef]
- Li, J.; Wang, H.; Lu, Y.; Mao, T.F.; Xiong, J.; He, S.L.; Liu, H. Inhibitory effect of tartary buckwheat seedling extracts and associated flavonoid compounds on the polyphenol oxidase activity in potatoes (Solanum tuberosum L.). J. Integr. Agric. 2019, 18, 2173–2182. [Google Scholar] [CrossRef]
- Preczenhak, A.P.; Orsi, B.; Lima, G.P.P.; Tezotto-Uliana, J.V.; Minatel, I.O.; Kluge, R.A. Cysteine enhances the content of betalains and polyphenols in fresh-cut red beet. Food Chem. 2019, 286, 600–607. [Google Scholar] [CrossRef]
- Latorre, M.E.; Narvaiz, P.; Rojas, A.M.; Gerschenson, L.N. Effects of gamma irradiation on bio-chemical and physico-chemical parameters of fresh-cut red beet (Beta vulgaris L. var. conditiva) root. J. Food Eng. 2010, 98, 178–191. [Google Scholar] [CrossRef]
- Barbagallo, R.N.; Chisari, M.; Patané, C. Polyphenol oxidase, total phenolics and ascorbic acid changes during storage of minimally processed “California Wonder” and “Quadrato d’Asti” sweet peppers. LWT Food Sci. Technol. 2012, 49, 192–196. [Google Scholar] [CrossRef]
- Jang, J.H.; Moon, K.D. Inhibition of polyphenol oxidase and peroxidase activities on fresh-cut apple by simultaneous treatment of ultrasound and ascorbic acid. Food Chem. 2011, 124, 444–449. [Google Scholar] [CrossRef]
- Luo, Y.; Lu, S.; Zhou, B.; Feng, H. Dual effectiveness of sodium chlorite for enzymatic browning inhibition and microbial inactivation on fresh-cut apples. LWT Food Sci. Technol. 2011, 44, 1621–1625. [Google Scholar] [CrossRef]
- Zambrano-Zaragoza, M.L.; Gutiérrez-Cortez, E.; Del Real, A.; González-Reza, R.M.; Galindo-Pérez, M.J.; Quintanar-Guerrero, D. Fresh-cut Red Delicious apples coating using tocopherol/mucilage nanoemulsion: Effect of coating on polyphenol oxidase and pectin methylesterase activities. Food Res. Int. 2014, 62, 974–983. [Google Scholar] [CrossRef]
- Villamil-Galindo, E.; Van de Velde, F.; Piagentini, A.M. Extracts from strawberry by-products rich in phenolic compounds reduce the activity of apple polyphenol oxidase. LWT 2020, 133, 110097. [Google Scholar] [CrossRef]
- Teixeira, G.H.A.; Durigan, J.F.; Alves, R.E.; O’Hare, T.J. Use of modified atmosphere to extend shelf life of fresh-cut carambola (Averrhoa carambola L. cv. Fwang Tung). Postharvest Biol. Technol. 2007, 44, 80–85. [Google Scholar] [CrossRef] [Green Version]
- Teixeira, G.H.A.; Durigan, J.F.; Ferraudo, A.S.; Alves, R.E.; O’Hare, T.J. Multivariate analysis of fresh-cut carambola slices stored under different temperatures. Postharvest Biol. Technol. 2012, 63, 91–97. [Google Scholar] [CrossRef]
- Kanjanapongkul, K.; Baibua, V. Effects of ohmic pasteurization of coconut water on polyphenol oxidase and peroxidase inactivation and pink discoloration prevention. J. Food Eng. 2021, 292, 110268. [Google Scholar] [CrossRef]
- Aquino-Bolaños, E.N.; Mercado-Silva, E. Effects of polyphenol oxidase and peroxidase activity, phenolics and lignin content on the browning of cut jicama. Postharvest Biol. Technol. 2004, 33, 275–283. [Google Scholar] [CrossRef]
- Kuwar, U.; Sharma, S.; Tadapaneni, V.R.R. Aloe Vera Gel and Honey-Based Edible Coatings Combined with Chemical Dip as a Safe Means for Quality Maintenance and Shelf Life Extension of Fresh-Cut Papaya. J. Food Qual. 2015, 38, 347–358. [Google Scholar] [CrossRef]
- Koukounaras, A.; Diamantidis, G.; Sfakiotakis, E. The effect of heat treatment on quality retention of fresh-cut peach. Postharvest Biol. Technol. 2008, 48, 30–36. [Google Scholar] [CrossRef]
- Serra, S.; Anthony, B.; Masia, A.; Giovannini, D.; Musacchi, S. Determination of Biochemical Composition in Peach by Di ff erent Flesh Color and Textural Typologies. Foods 2020, 9, 1452. [Google Scholar] [CrossRef]
- Xiao, C.; Zhu, L.; Luo, W.; Song, X.; Deng, Y. Combined action of pure oxygen pretreatment and chitosan coating incorporated with rosemary extracts on the quality of fresh-cut pears. Food Chem. 2010, 121, 1003–1009. [Google Scholar] [CrossRef]
- Xiao, Z.; Luo, Y.; Luo, Y.; Wang, Q. Combined effects of sodium chlorite dip treatment and chitosan coatings on the quality of fresh-cut d’Anjou pears. Postharvest Biol. Technol. 2011, 62, 319–326. [Google Scholar] [CrossRef]
- Martín-Diana, A.B.; Rico, D.; Barry-Ryan, C.; Frías, J.M.; Henehan, G.T.M.; Barat, J.M. Efficacy of steamer jet-injection as alternative to chlorine in fresh-cut lettuce. Postharvest Biol. Technol. 2007, 45, 97–107. [Google Scholar] [CrossRef] [Green Version]
- Yingsanga, P.; Srilaong, V.; Kanlayanarat, S.; Noichinda, S.; McGlasson, W.B. Relationship between browning and related enzymes (PAL, PPO and POD) in rambutan fruit (Nephelium lappaceum Linn.) cvs. Rongrien and See-Chompoo. Postharvest Biol. Technol. 2008, 50, 164–168. [Google Scholar] [CrossRef]
- Ayaz, F.A.; Demir, O.; Torun, H.; Kolcuoglu, Y.; Colak, A. Characterization of polyphenoloxidase (PPO) and total phenolic contents in medlar (Mespilus germanica L.) fruit during ripening and over ripening. Food Chem. 2008, 106, 291–298. [Google Scholar] [CrossRef]
- Mayer, A.M. Polyphenol oxidases in plants and fungi: Going places? A review. Phytochemistry 2006, 67, 2318–2331. [Google Scholar] [CrossRef]
- Han, Q.; Liu, F.; Hao, Y.; Ni, Y. Characterization of membrane-bound polyphenol oxidase from Granny Smith apple (Malus × domestica Borkh.). Int. J. Biol. Macromol. 2020, 158, 977–984. [Google Scholar] [CrossRef]
- Scuderi, D.; Restuccia, C.; Chisari, M.; Barbagallo, R.N.; Caggia, C.; Giuffrida, F. Salinity of nutrient solution influences the shelf-life of fresh-cut lettuce grown in floating system. Postharvest Biol. Technol. 2011, 59, 132–137. [Google Scholar] [CrossRef]
- Debelo, H.; Li, M.; Ferruzzi, M.G. Processing influences on food polyphenol profiles and biological activity. Curr. Opin. Food Sci. 2020, 32, 90–102. [Google Scholar] [CrossRef]
- Dovene, A.K.; Wang, L.; Bokhary, S.U.F.; Madebo, M.P.; Zheng, Y.; Jin, P. Eff ect of Cutting Styles on Quality and Antioxidant Activity of Stored Fresh-Cut Sweet Potato. Foods Artic. 2019, 8, 674. [Google Scholar]
- Landi, M.; Degl’Innocenti, E.; Guglielminetti, L.; Guidi, L. Role of ascorbic acid in the inhibition of polyphenol oxidase and the prevention of browning in different browning-sensitive Lactuca sativa var. capitata (L.) and Eruca sativa (Mill.) stored as fresh-cut produce. J. Sci. Food Agric. 2013, 93, 1814–1819. [Google Scholar] [CrossRef] [PubMed]
- Peng, L.; Jiang, Y. Exogenous salicylic acid inhibits browning of fresh-cut Chinese water chestnut. Food Chem. 2006, 94, 535–540. [Google Scholar] [CrossRef]
- Abreu, M.; Beirão-da-Costa, S.; Gonçalves, E.M.; Beirão-da-Costa, M.L.; Moldão-Martins, M. Use of mild heat pre-treatments for quality retention of fresh-cut “Rocha” pear. Postharvest Biol. Technol. 2003, 30, 153–160. [Google Scholar] [CrossRef]
- Olivas, G.I.; Mattinson, D.S.; Barbosa-Cánovas, G.V. Alginate coatings for preservation of minimally processed “Gala” apples. Postharvest Biol. Technol. 2007, 45, 89–96. [Google Scholar] [CrossRef]
- Gomes, M.H.; Fundo, J.F.; Santos, S.; Amaro, A.L.; Almeida, D.P.F. Hydrogen ion concentration affects quality retention and modifies the effect of calcium additives on fresh-cut “Rocha” pear. Postharvest Biol. Technol. 2010, 58, 239–246. [Google Scholar] [CrossRef]
- Gomes, M.H.; Fundo, J.F.; Poças, M.F.; Almeida, D.P.F. Quality changes in fresh-cut “Rocha” pear as affected by oxygen levels in modified atmosphere packaging and the pH of antibrowning additive. Postharvest Biol. Technol. 2012, 74, 62–70. [Google Scholar] [CrossRef]
- Gancarz, M. At harvest prediction of the susceptibility of potato varieties to blackspot after impact over long-term storage. Postharvest Biol. Technol. 2018, 142, 93–98. [Google Scholar] [CrossRef]
- Krochmal-Marczak, B.; Sawicka, B.; Krzysztofik, B.; Danilčenko, H.; Jariene, E. The effects of temperature on the quality and storage stalibity of sweet potato (Ipomoea batatas L. [Lam]) grown in Central Europe. Agronomy 2020, 10, 1665. [Google Scholar] [CrossRef]
- Lufu, R.; Ambaw, A.; Opara, U.L. Water loss of fresh fruit: Influencing pre-harvest, harvest and postharvest factors. Sci. Hortic. 2020, 272, 109519. [Google Scholar] [CrossRef]
- Grudzińska, M.; Boguszewska-Mańkowska, D.; Zarzyńska, K. Drought stress during the growing season: Changes in reducing sugars, starch content and respiration rate during storage of two potato cultivars differing in drought sensitivity. J. Agron. Crop Sci. 2021, 1–12. [Google Scholar] [CrossRef]
- Caleb, O.J.; Fawole, O.A.; Mphahlele, R.R.; Opara, U.L. Impact of preharvest and postharvest factors on changes in volatile compounds of pomegranate fruit and minimally processed arils—Review. Sci. Hortic. 2015, 188, 106–114. [Google Scholar] [CrossRef]
- Marques Silva, F.V.; Sulaiman, A. Polyphenoloxidase in Fruit and Vegetables: Inactivation by Thermal and Non-thermal Processes; Melton, L., Shahidi, F., Varelis, P.B.T.-E., Eds.; Academic Press: Oxford, UK, 2019; pp. 287–301. ISBN 978-0-12-814045-1. [Google Scholar]
- Mostafidi, M.; Sanjabi, M.R.; Shirkhan, F.; Zahedi, M.T. A review of recent trends in the development of the microbial safety of fruits and vegetables. Trends Food Sci. Technol. 2020, 103, 321–332. [Google Scholar] [CrossRef]
- Ma, L.; Zhang, M.; Bhandari, B.; Gao, Z. Recent developments in novel shelf life extension technologies of fresh-cut fruits and vegetables. Trends Food Sci. Technol. 2017, 64, 23–38. [Google Scholar] [CrossRef] [Green Version]
- Ukuku, D.O.; Geveke, D.J.; Chau, L.; Niemira, B.A. Microbial safety and overall quality of cantaloupe fresh-cut pieces prepared from whole fruit after wet steam treatment. Int. J. Food Microbiol. 2016, 231, 86–92. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Putnik, P.; Roohinejad, S.; Greiner, R.; Granato, D.; Bekhit, A.E.D.A.; Bursać Kovačević, D. Prediction and modeling of microbial growth in minimally processed fresh-cut apples packaged in a modified atmosphere: A review. Food Control 2017, 80, 411–419. [Google Scholar] [CrossRef]
- Giannakourou, M.C.; Tsironi, T.N. Application of Processing and Packaging Hurdles for Fresh-Cut Fruits and Vegetables Preservation. Foods 2021, 10, 830. [Google Scholar] [CrossRef]
- Maringgal, B.; Hashim, N.; Mohamed Amin Tawakkal, I.S.; Muda Mohamed, M.T. Recent advance in edible coating and its effect on fresh/fresh-cut fruits quality. Trends Food Sci. Technol. 2020, 96, 253–267. [Google Scholar] [CrossRef]
- Zhang, L.; Lu, Z.; Yu, Z.; Gao, X. Preservation of fresh-cut celery by treatment of ozonated water. Food Control 2005, 16, 279–283. [Google Scholar] [CrossRef]
- Gómez-López, V.M.; Ragaert, P.; Ryckeboer, J.; Jeyachchandran, V.; Debevere, J.; Devlieghere, F. Shelf-life of minimally processed cabbage treated with neutral electrolysed oxidising water and stored under equilibrium modified atmosphere. Int. J. Food Microbiol. 2007, 117, 91–98. [Google Scholar] [CrossRef]
- Tinello, F.; Lante, A. Recent advances in controlling polyphenol oxidase activity of fruit and vegetable products. Innov. Food Sci. Emerg. Technol. 2018, 50, 73–83. [Google Scholar] [CrossRef]
- Iqbal, A.; Murtaza, A.; Hu, W.; Ahmad, I.; Ahmed, A.; Xu, X. Activation and inactivation mechanisms of polyphenol oxidase during thermal and non-thermal methods of food processing. Food Bioprod. Process. 2019, 117, 170–182. [Google Scholar] [CrossRef]
- Del Valle, J.M.; Aránguiz, V.; Léon, H. Effects of blanching and calcium infiltration on PPO activity, texture, microstructure and kinetics of osmotic dehydration of apple tissue. Food Res. Int. 1998, 31, 557–569. [Google Scholar] [CrossRef]
- Shrestha, L.; Kulig, B.; Moscetti, R.; Massantini, R.; Pawelzik, E.; Hensel, O.; Sturm, B. Optimisation of physical and chemical treatments to control browning development and enzymatic activity on fresh-cut apple slices. Foods 2020, 9, 76. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Luo, Z.; Li, D.; Xie, J.; Feng, S.; Wang, Y. Effects of Heat Treatment on Quality and Browning of Fresh-Cut Sugarcane. J. Food Process. Preserv. 2015, 39, 688–696. [Google Scholar] [CrossRef]
- Oliveira, M.; Abadias, M.; Usall, J.; Torres, R.; Teixidó, N.; Viñas, I. Application of modified atmosphere packaging as a safety approach to fresh-cut fruits and vegetables—A review. Trends Food Sci. Technol. 2015, 46, 13–26. [Google Scholar] [CrossRef]
- Saxena, A.; Bawa, A.S.; Raju, P.S. Phytochemical changes in fresh-cut jackfruit (Artocarpus heterophyllus L.) bulbs during modified atmosphere storage. Food Chem. 2009, 115, 1443–1449. [Google Scholar] [CrossRef]
- Wang, D.; Li, D.; Xu, Y.; Li, L.; Belwal, T.; Zhang, X.; Luo, Z. Elevated CO2 alleviates browning development by modulating metabolisms of membrane lipids, proline, and GABA in fresh-cut Asian pear fruit. Sci. Hortic. 2021, 281, 109932. [Google Scholar] [CrossRef]
- Wang, D.; Li, W.; Li, D.; Li, L.; Luo, Z. Effect of high carbon dioxide treatment on reactive oxygen species accumulation and antioxidant capacity in fresh-cut pear fruit during storage. Sci. Hortic. 2021, 281, 109925. [Google Scholar] [CrossRef]
- You, Y.; Jiang, Y.; Duan, X.; Su, X.; Song, L.; Liu, H.; Sun, J.; Yang, H. Browning inhibition and quality maintenance of fresh-cut chinese water chestnut by anoxia treatment. J. Food Process. Preserv. 2007, 31, 595–606. [Google Scholar] [CrossRef]
- Thivya, P.; Bhosale, Y.K.; Anandakumar, S.; Hema, V.; Sinija, V.R. Development of active packaging film from sodium alginate/carboxymethyl cellulose containing shallot waste extracts for anti-browning of fresh-cut produce. Int. J. Biol. Macromol. 2021, 188, 790–799. [Google Scholar] [CrossRef]
- Gorny, J.R.; Hess-Pierce, B.; Cifuentes, R.A.; Kader, A.A. Quality changes in fresh-cut pear slices as affected by controlled atmospheres and chemical preservatives. Postharvest Biol. Technol. 2002, 24, 271–278. [Google Scholar] [CrossRef] [Green Version]
- Rocculi, P.; Romani, S.; Dalla Rosa, M. Evaluation of physico-chemical parameters of minimally processed apples packed in non-conventional modified atmosphere. Food Res. Int. 2004, 37, 329–335. [Google Scholar] [CrossRef]
- Limbo, S.; Piergiovanni, L. Shelf life of minimally processed potatoes: Part 1. Effects of high oxygen partial pressures in combination with ascorbic and citric acids on enzymatic browning. Postharvest Biol. Technol. 2006, 39, 254–264. [Google Scholar] [CrossRef]
- Aguayo, E.; Requejo-Jackman, C.; Stanley, R.; Woolf, A. Effects of calcium ascorbate treatments and storage atmosphere on antioxidant activity and quality of fresh-cut apple slices. Postharvest Biol. Technol. 2010, 57, 52–60. [Google Scholar] [CrossRef]
- Xing, Y.; Li, X.; Xu, Q.; Jiang, Y.; Yun, J.; Li, W. Effects of chitosan-based coating and modified atmosphere packaging (MAP) on browning and shelf life of fresh-cut lotus root (Nelumbo nucifera Gaerth). Innov. Food Sci. Emerg. Technol. 2010, 11, 684–689. [Google Scholar] [CrossRef]
- Arias, E.; López-Buesa, P.; Oria, R. Extension of fresh-cut “Blanquilla” pear (Pyrus communis L.) shelf-life by 1-MCP treatment after harvest. Postharvest Biol. Technol. 2009, 54, 53–58. [Google Scholar] [CrossRef]
- Li, L.; Li, C.; Sun, J.; Xin, M.; Yi, P.; He, X.; Sheng, J.; Zhou, Z.; Ling, D.; Zheng, F.; et al. Synergistic effects of ultraviolet light irradiation and high-oxygen modified atmosphere packaging on physiological quality, microbial growth and lignification metabolism of fresh-cut carrots. Postharvest Biol. Technol. 2021, 173. [Google Scholar] [CrossRef]
- Martín-Diana, A.B.; Rico, D.; Frías, J.M.; Barat, J.M.; Henehan, G.T.M.; Barry-Ryan, C. Calcium for extending the shelf life of fresh whole and minimally processed fruits and vegetables: A review. Trends Food Sci. Technol. 2007, 18, 210–218. [Google Scholar] [CrossRef]
- Perez-Cabrera, L.; Chafer, M.; Chiralt, A.; Gonzalez-Martinez, C. Effectiveness of antibrowning agents applied by vacuum impregnation on minimally processed pear. LWT Food Sci. Technol. 2011, 44, 2273–2280. [Google Scholar] [CrossRef]
- Abbott, J.A.; Saftner, R.A.; Gross, K.C.; Vinyard, B.T.; Janick, J. Consumer evaluation and quality measurement of fresh-cut slices of “Fuji,” “Golden Delicious,” “GoldRush,” and “Granny Smith” apples. Postharvest Biol. Technol. 2004, 33, 127–140. [Google Scholar] [CrossRef] [Green Version]
- Jiang, Y.; Pen, L.; Li, J. Use of citric acid for shelf life and quality maintenance of fresh-cut Chinese water chestnut. J. Food Eng. 2004, 63, 325–328. [Google Scholar] [CrossRef]
- Amodio, M.L.; Cabezas-Serrano, A.B.; Peri, G.; Colelli, G. Post-cutting quality changes of fresh-cut artichokes treated with different anti-browning agents as evaluated by image analysis. Postharvest Biol. Technol. 2011, 62, 213–220. [Google Scholar] [CrossRef]
- Gómez-López, V.M.; Ragaert, P.; Jeyachchandran, V.; Debevere, J.; Devlieghere, F. Shelf-life of minimally processed lettuce and cabbage treated with gaseous chlorine dioxide and cysteine. Int. J. Food Microbiol. 2008, 121, 74–83. [Google Scholar] [CrossRef] [PubMed]
- Zhou, D.; Li, L.; Wu, Y.; Fan, J.; Ouyang, J. Salicylic acid inhibits enzymatic browning of fresh-cut Chinese chestnut (Castanea mollissima) by competitively inhibiting polyphenol oxidase. Food Chem. 2015, 171, 19–25. [Google Scholar] [CrossRef]
- Altunkaya, A. Effect of whey protein concentrate on phenolic profile and browning of fresh-cut lettuce (Lactuca Sativa). Food Chem. 2011, 128, 754–760. [Google Scholar] [CrossRef]
- Botondi, R.; Barone, M.; Grasso, C. A review into the effectiveness of ozone technology for improving the safety and preserving the quality of fresh-cut fruits and vegetables. Foods 2021, 10, 748. [Google Scholar] [CrossRef]
- Rico, D.; Martín-Diana, A.B.; Frías, J.M.; Henehan, G.T.; Barry-Ryan, C. Effect of ozone and calcium lactate treatments on browning and texture properties of fresh-cut lettuce. J. Sci. Food Agric. 2006, 86, 2179–2188. [Google Scholar] [CrossRef]
- Calder, B.L.; Skonberg, D.I.; Davis-Dentici, K.; Hughes, B.H.; Bolton, J.C. The Effectiveness of Ozone and Acidulant Treatments in Extending the Refrigerated Shelf Life of Fresh-Cut Potatoes. J. Food Sci. 2011, 76, 492–498. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Jiang, X.; Chen, Y.; Lin, M.; Tang, J.; Fang, L.; Li, M.; Hung, Y.; Lin, H. Recent trends and applications of electrolyzed oxidizing water. Food Chem. 2021, 130873. [Google Scholar] [CrossRef]
- Rico, D.; Martín-Diana, A.B.; Barry-Ryan, C.; Frías, J.M.; Henehan, G.T.M.; Barat, J.M. Use of neutral electrolysed water (EW) for quality maintenance and shelf-life extension of minimally processed lettuce. Innov. Food Sci. Emerg. Technol. 2008, 9, 37–48. [Google Scholar] [CrossRef] [Green Version]
- Gómez-López, V.M.; Rajkovic, A.; Ragaert, P.; Smigic, N.; Devlieghere, F. Chlorine dioxide for minimally processed produce preservation: A review. Trends Food Sci. Technol. 2009, 20, 17–26. [Google Scholar] [CrossRef]
- Cheng, D.; Wang, G.; Tang, J.; Yao, C.; Li, P.; Song, Q.; Wang, C. Inhibitory effect of chlorogenic acid on polyphenol oxidase and browning of fresh-cut potatoes. Postharvest Biol. Technol. 2020, 168, 111282. [Google Scholar] [CrossRef]
- Rojas-Graü, M.A.; Soliva-Fortuny, R.; Martín-Belloso, O. Edible coatings to incorporate active ingredients to fresh-cut fruits: A review. Trends Food Sci. Technol. 2009, 20, 438–447. [Google Scholar] [CrossRef]
- Yousuf, B.; Qadri, O.S.; Srivastava, A.K. Recent developments in shelf-life extension of fresh-cut fruits and vegetables by application of different edible coatings: A review. LWT Food Sci. Technol. 2018, 89, 198–209. [Google Scholar] [CrossRef]
- Pen, L.T.; Jiang, Y.M. Effects of chitosan coating on shelf life and quality of fresh-cut Chinese water chestnut. LWT Food Sci. Technol. 2003, 36, 359–364. [Google Scholar] [CrossRef]
- Qi, H.; Hu, W.; Jiang, A.; Tian, M.; Li, Y. Extending shelf-life of Fresh-cut “Fuji” apples with chitosan-coatings. Innov. Food Sci. Emerg. Technol. 2011, 12, 62–66. [Google Scholar] [CrossRef]
- Montero-Calderón, M.; Rojas-Graü, M.A.; Martín-Belloso, O. Effect of packaging conditions on quality and shelf-life of fresh-cut pineapple (Ananas comosus). Postharvest Biol. Technol. 2008, 50, 182–189. [Google Scholar] [CrossRef]
- Supapvanich, S.; Prathaan, P.; Tepsorn, R. Browning inhibition in fresh-cut rose apple fruit cv. Taaptimjaan using konjac glucomannan coating incorporated with pineapple fruit extract. Postharvest Biol. Technol. 2012, 73, 46–49. [Google Scholar] [CrossRef]
- Bata Gouda, M.H.; Zhang, C.; Peng, S.; Kong, X.; Chen, Y.; Li, H.; Li, X.; Luo, H.; Yu, L. Combination of sodium alginate-based coating with L-cysteine and citric acid extends the shelf-life of fresh-cut lotus root slices by inhibiting browning and microbial growth. Postharvest Biol. Technol. 2021, 175, 111502. [Google Scholar] [CrossRef]
- Lee, M.K. Inhibitory effect of banana polyphenol oxidase during ripening of banana by onion extract and Maillard reaction products. Food Chem. 2007, 102, 146–149. [Google Scholar] [CrossRef]
- Yu, L.; Shi, H. Effect of two mulberry (Morus alba L.) leaf polyphenols on improving the quality of fresh-cut cantaloupe during storage. Food Control 2021, 121, 107624. [Google Scholar] [CrossRef]
- Yousuf, B.; Wu, S.; Siddiqui, M.W. Incorporating essential oils or compounds derived thereof into edible coatings: Effect on quality and shelf life of fresh/fresh-cut produce. Trends Food Sci. Technol. 2021, 108, 245–257. [Google Scholar] [CrossRef]
- Lu, Z.; Yu, Z.; Gao, X.; Lu, F.; Zhang, L. Preservation effects of gamma irradiation on fresh-cut celery. J. Food Eng. 2005, 67, 347–351. [Google Scholar] [CrossRef]
- Zhang, L.; Lu, Z.; Lu, F.; Bie, X. Effect of γ irradiation on quality-maintaining of fresh-cut lettuce. Food Control 2006, 17, 225–228. [Google Scholar] [CrossRef]
- Zhan, L.; Li, Y.; Hu, J.; Pang, L.; Fan, H. Browning inhibition and quality preservation of fresh-cut romaine lettuce exposed to high intensity light. Innov. Food Sci. Emerg. Technol. 2012, 14, 70–76. [Google Scholar] [CrossRef]
- Gómez, P.L.; Alzamora, S.M.; Castro, M.A.; Salvatori, D.M. Effect of ultraviolet-C light dose on quality of cut-apple: Microorganism, color and compression behavior. J. Food Eng. 2010, 98, 60–70. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, Y.; Zhou, L.; Liao, X. A comparative study of inactivation of peach polyphenol oxidase and carrot polyphenol oxidase induced by high-pressure carbon dioxide. Int. J. Food Sci. Technol. 2010, 45, 2297–2305. [Google Scholar] [CrossRef]
- Wu, Z.S.; Zhang, M.; Wang, S. Effects of high pressure argon treatments on the quality of fresh-cut apples at cold storage. Food Control 2012, 23, 120–127. [Google Scholar] [CrossRef]


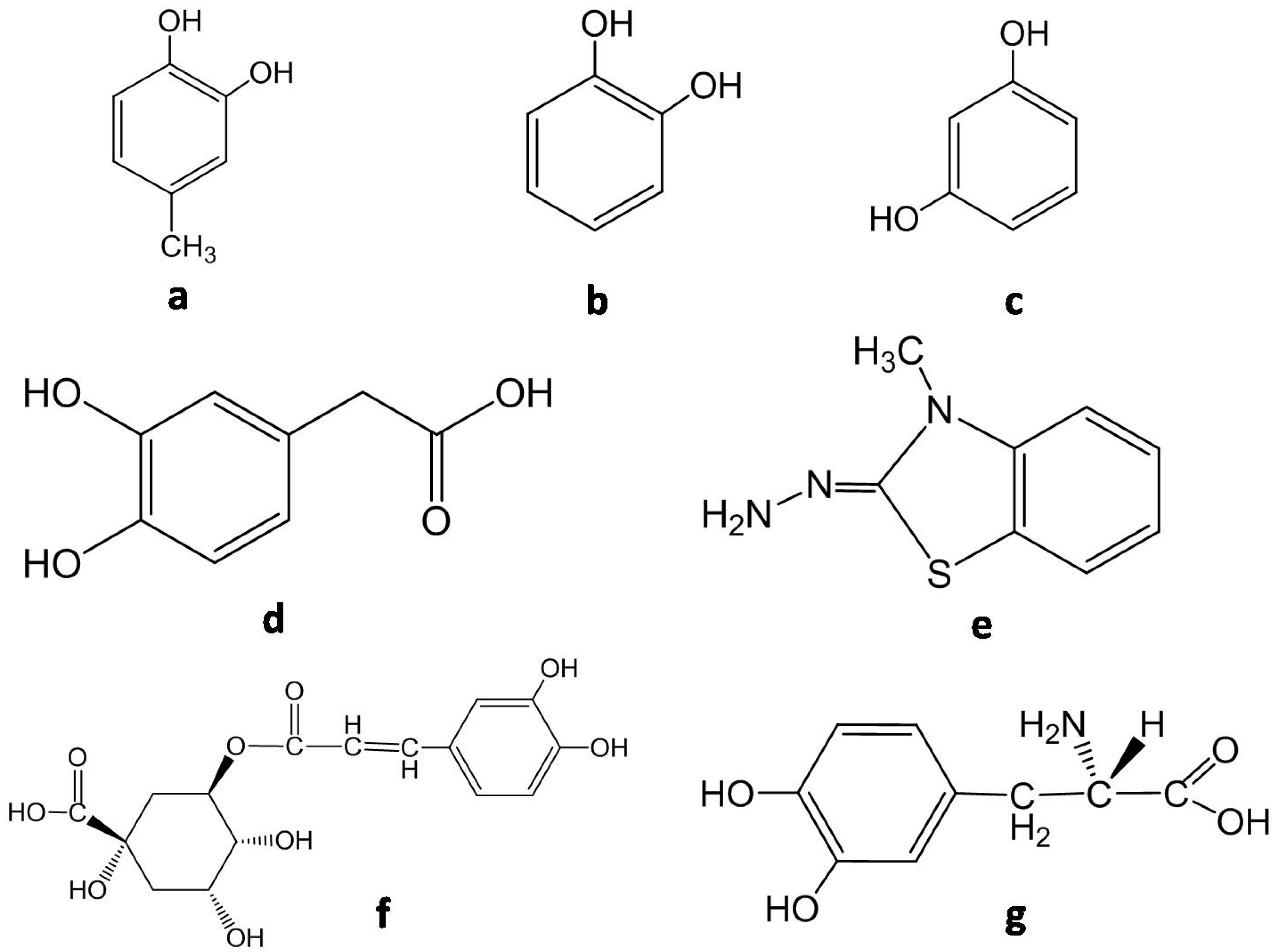

| PPO Extraction | Spectrophotometric Determination | ||||
|---|---|---|---|---|---|
| Produce | Reference | Substrate Solution | pH | Wavelength (nm) | |
| Artichoke | [14] | PVP and acetate buffer (pH 5.6) | Chlorogenic acid | 6.0 | 410 |
| Artichoke | [25] | Benzamidine, ascorbic acid, PMSF, triton and phosphate buffer (pH 7.0) | MBTH | 4.5 | 400 |
| Carrot | [26] | PVPP and phosphate buffer (pH 6.5) | Catechol | 6.5 | 420 |
| Carrot | [27] | Phosphate buffer (pH 7.0) | Catechol | 7.0 | 410 |
| Eggplant | [28] | Citrate phosphate buffer (pH 7.0) | DOPAC, MBTH, methanol | 7.0 | 466 |
| Eggplant | [29] | PVP, PVPP, triton, ascorbic acid and phosphate buffer (pH 6.8) | 4-methylcatechol | 6.8 | 420 |
| Lettuce | [15] | PVPP and phosphate buffer (pH 6.5) | Catechol | 6.5 | 400 |
| Lettuce | [30] | PVPP and phosphate buffer (pH 7.0) | Catechol | 7.0 | 420 |
| Lettuce | [31] | Acetone, MBTH and citrate phosphate buffer (pH 7.5) | DOPAC | 7.0 | 505 |
| Lettuce | [32] | Phosphate buffer (pH 7.0) | MBTH | 7.0 | 467 |
| Lotus root | [33] | PVPP and phosphate buffer (pH 7.0) | Catechol | 7.0 | 410 |
| Mushroom | [34] | PVPP and NaCl (pH 6.5) | Catechol | 7.0 | 400 |
| Mushroom | [35] | PVPP, triton and phosphate buffer (pH 6.8) | Catechol | 6.8 | 420 |
| Potato | [36] | PVPP and phosphate buffer (pH 6.0) | 4-methylcatechol | 6.0 | 410 |
| Potato | [37] | Phosphate buffer (pH 6.5) | Catechol | 6.5 | 410 |
| Potato | [38] | PBS (pH 6.8) | Catechol | 5.5 | 405 |
| Potato | [39] | PVP, triton and phosphate buffer (pH 6.8) | Catechol | 7.0 | 475 |
| Red beet | [40] | NaCl, PVP and phosphate-citrate buffer (pH 6.5) | Pyrocatechol | 6.5 | 420 |
| Red beet | [41] | NaCl and phosphate buffer (pH 6.0) | Pyrocatechol | 7.0 | 420 |
| Sweet peppers | [42] | Acetone and citrate phosphate buffer (pH 7.5) | DOPAC | 7.0 | 505 |
| PPO Extraction | Spectrophotometric Determination | ||||
|---|---|---|---|---|---|
| Produce | Reference | Substrate Solution | pH | Wavelength (nm) | |
| Apple | [19] | PVPP and phosphate buffer (pH 7.0) | Cathecol | 5.8 | 420 |
| Apple | [43] | PVPP and phosphate buffer (pH 5.0) | 4-methylcatechol | 5.0 | 494 |
| Apple | [44] | Triton and phosphate buffer (pH 7.2) | Chlorogenic acid | 5.2 | 420 |
| Apple | [45] | PVPP, triton and Phosphate buffer (pH 7.0) | Citrate-phosphate | 5.0 | 420 |
| Apple | [46] | Potassium phosphate buffer (pH 7.0) | Sodium acetate | 5.5 | 405 |
| Banana | [18] | PVPP, triton and phosphate buffer (pH 6.5) | Catechol | 6.5 | 420 |
| Carambola | [47] | PVPP, KCl and phosphate buffer (pH 6.8) | Catechol | 7.2 | 410 |
| Carambola | [48] | PVPP, KCl and phosphate buffer (pH 6.8) | Catechol | 7.2 | 410 |
| Coconut water | [49] | Phosphate buffer (pH 5.5) | Pyrocatechol | 5.5 | 470 |
| Jicama | [50] | PVPP and phosphate buffer (pH 7.0) | Catechol | 7.0 | 420 |
| Papaya | [51] | Sodium phosphate buffer (pH 6.5) | Catechol | 6.5 | 420 |
| Peach | [52] | PMSF, PVPP, triton and phosphate buffer (pH 6.8) | L-Dopa | 6.8 | 475 |
| Peach | [10] | EDTA, MgCl2, PMSF, PVPP, triton and phosphate buffer (pH 7.0) | Resorcinol | 7.0 | 500 |
| Peach | [53] | EDTA, PVPP and phosphate buffer (pH 7.0) | -- | 7.0 | 420 |
| Pear | [54] | Dihydrogen phosphate buffer (pH 6.8) | Catechol | 6.8 | 398 |
| Pear | [55] | Triton and phosphate buffer (pH 7.2) | Chlorogenic acid | 4.2 | 420 |
| Strawberry | [13] | PVPP, triton and phosphate buffer (pH 6.5) | Catechol | 6.5 | 420 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nogales-Delgado, S. Polyphenoloxidase (PPO): Effect, Current Determination and Inhibition Treatments in Fresh-Cut Produce. Appl. Sci. 2021, 11, 7813. https://doi.org/10.3390/app11177813
Nogales-Delgado S. Polyphenoloxidase (PPO): Effect, Current Determination and Inhibition Treatments in Fresh-Cut Produce. Applied Sciences. 2021; 11(17):7813. https://doi.org/10.3390/app11177813
Chicago/Turabian StyleNogales-Delgado, Sergio. 2021. "Polyphenoloxidase (PPO): Effect, Current Determination and Inhibition Treatments in Fresh-Cut Produce" Applied Sciences 11, no. 17: 7813. https://doi.org/10.3390/app11177813
APA StyleNogales-Delgado, S. (2021). Polyphenoloxidase (PPO): Effect, Current Determination and Inhibition Treatments in Fresh-Cut Produce. Applied Sciences, 11(17), 7813. https://doi.org/10.3390/app11177813







