Additive Manufacturing of Bone Scaffolds Using PolyJet and Stereolithography Techniques
Abstract
1. Introduction
2. Materials and Methods
2.1. Design and Development of 3D-Printed Scaffold Structures
2.2. Permeability Analysis of 3D-Printed Scaffold Structures
2.2.1. Surface Energy Calculation
2.2.2. Numerical Calculations of Permeability
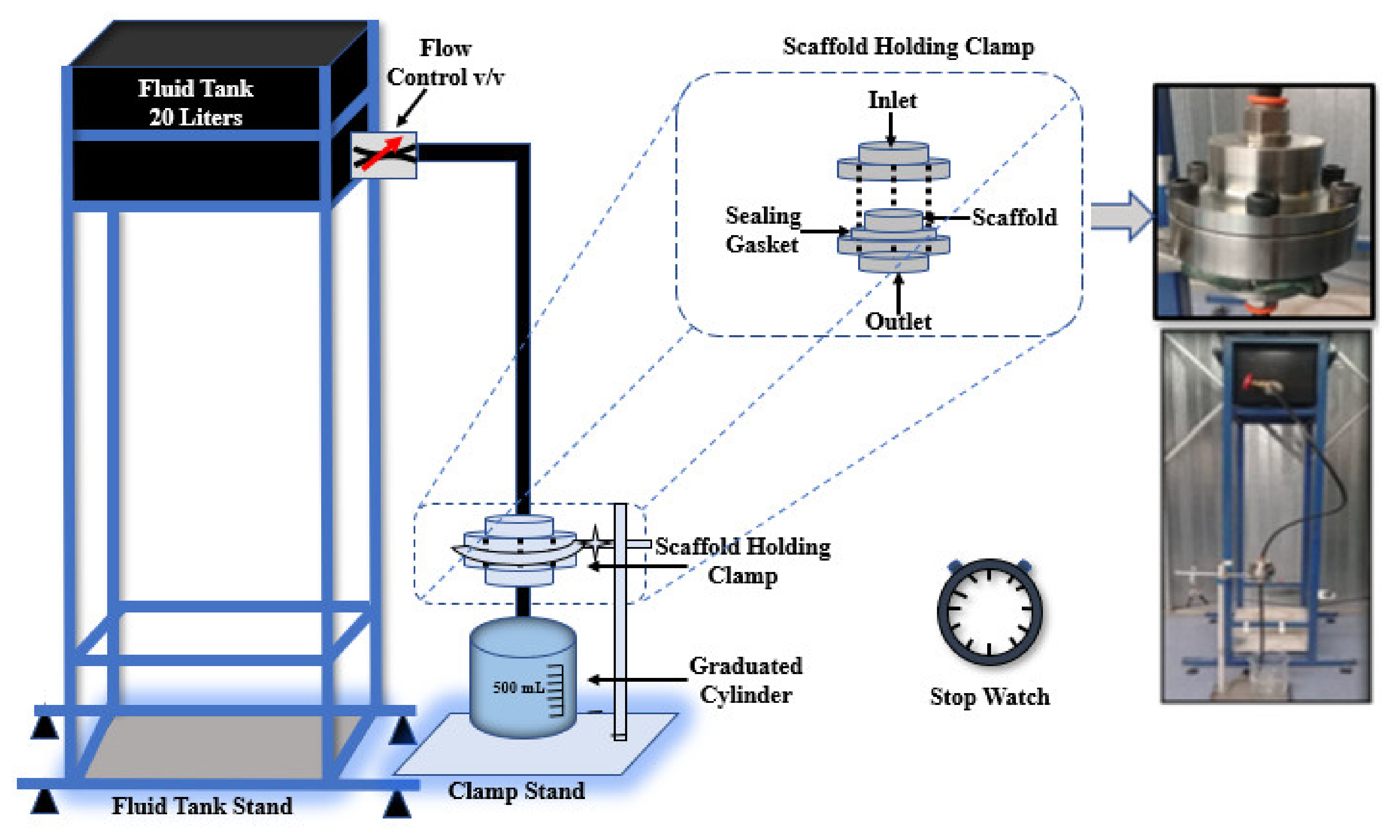
2.2.3. Experimental Measurements of Permeability
2.3. Design and 3D Printing of Standard Samples for Mechanical Testing
2.4. Mechanical Testing of Standard Blocks and 3D-Printed Scaffold Structures
3. Results
3.1. Contact Angle and Surface Energy Analyses
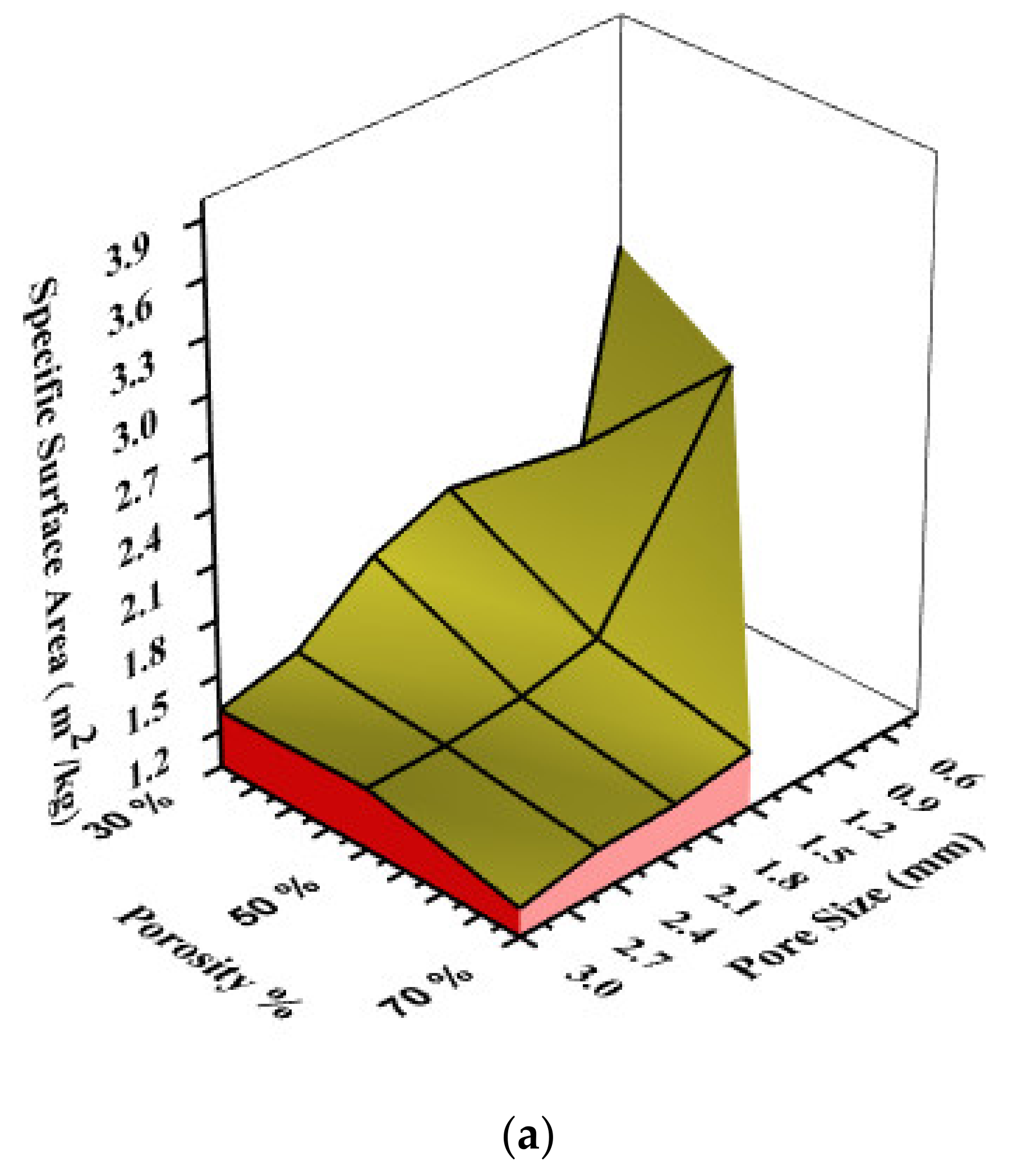
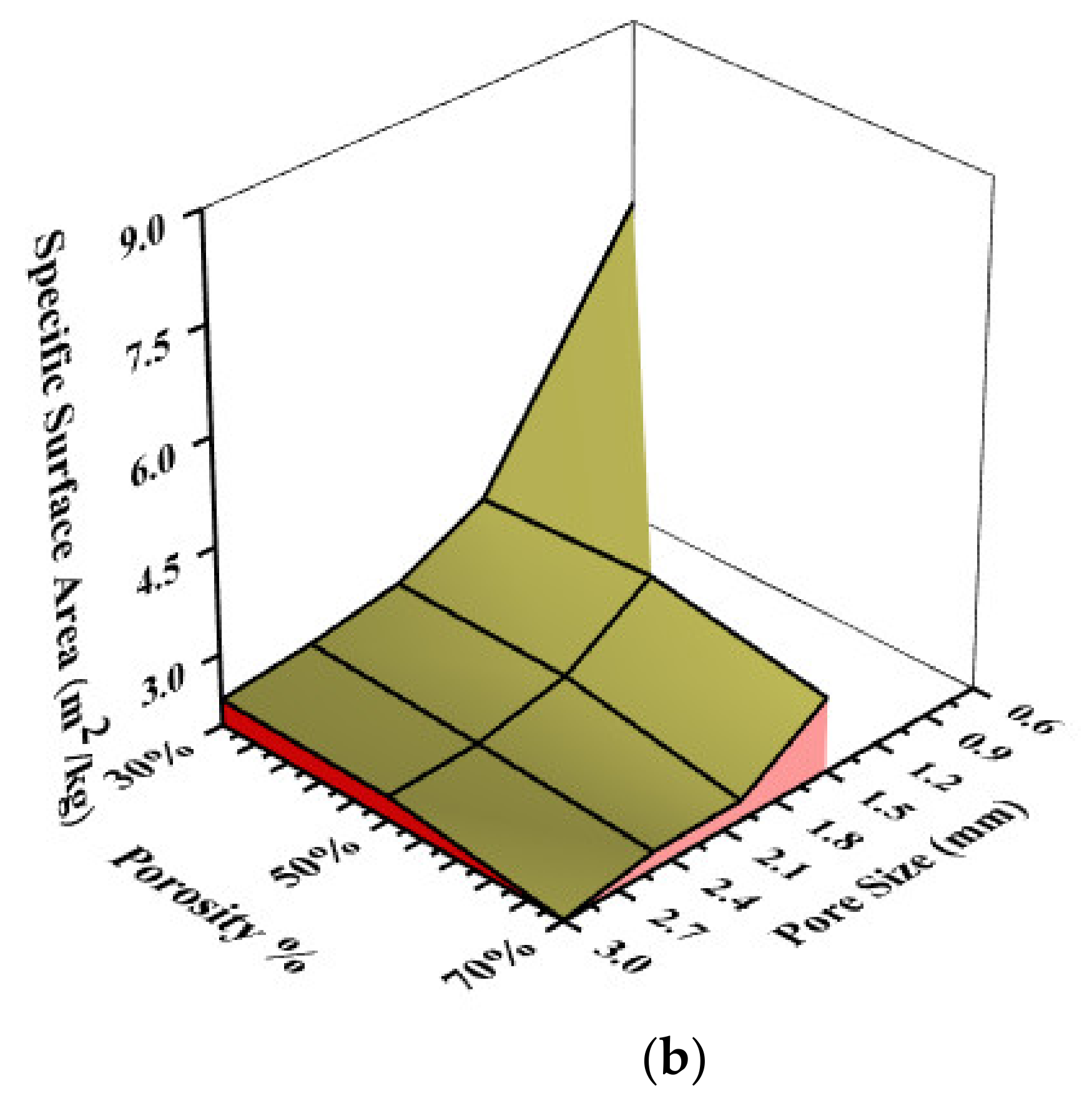
3.2. Numerically Calculated Permeability
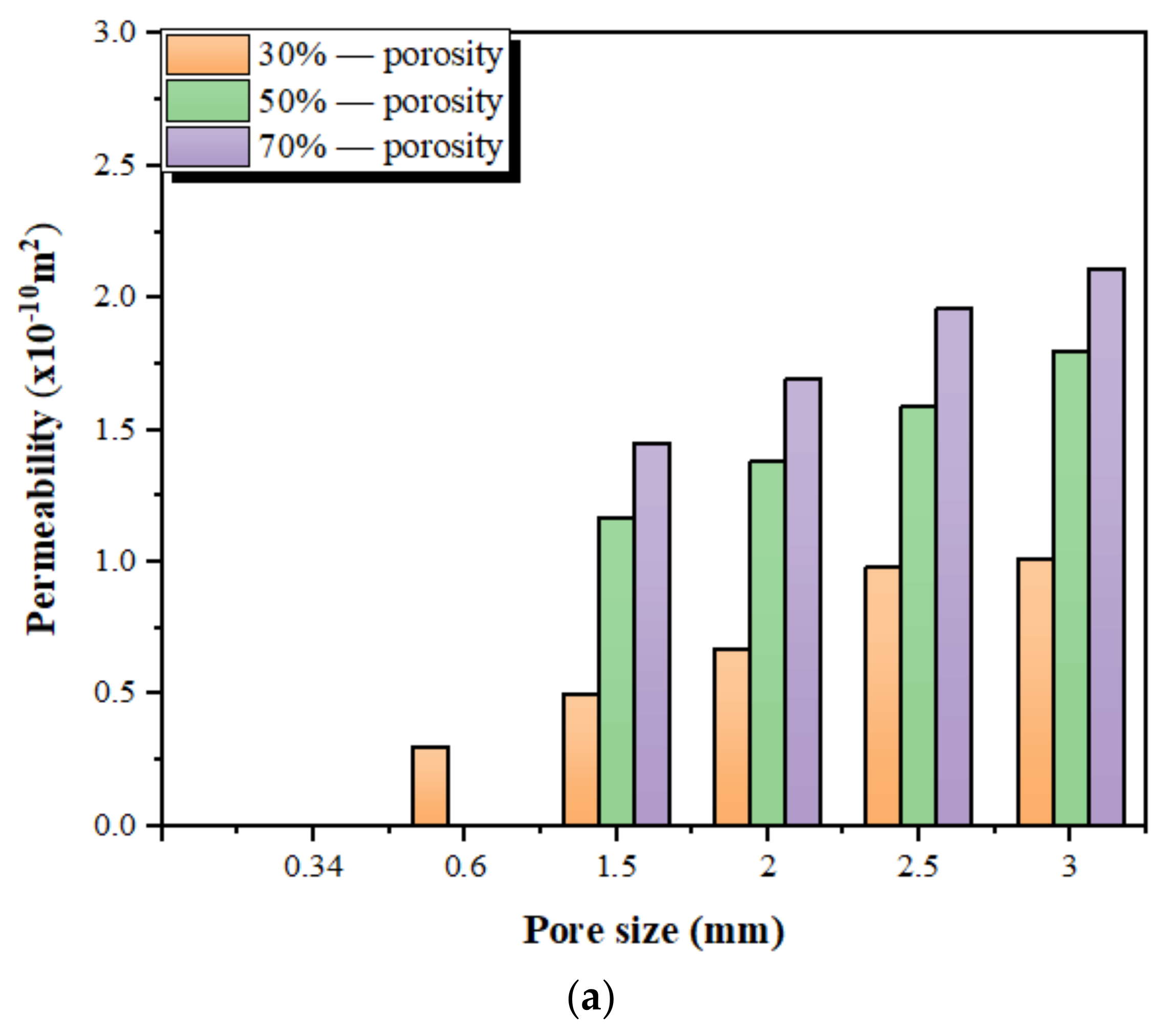
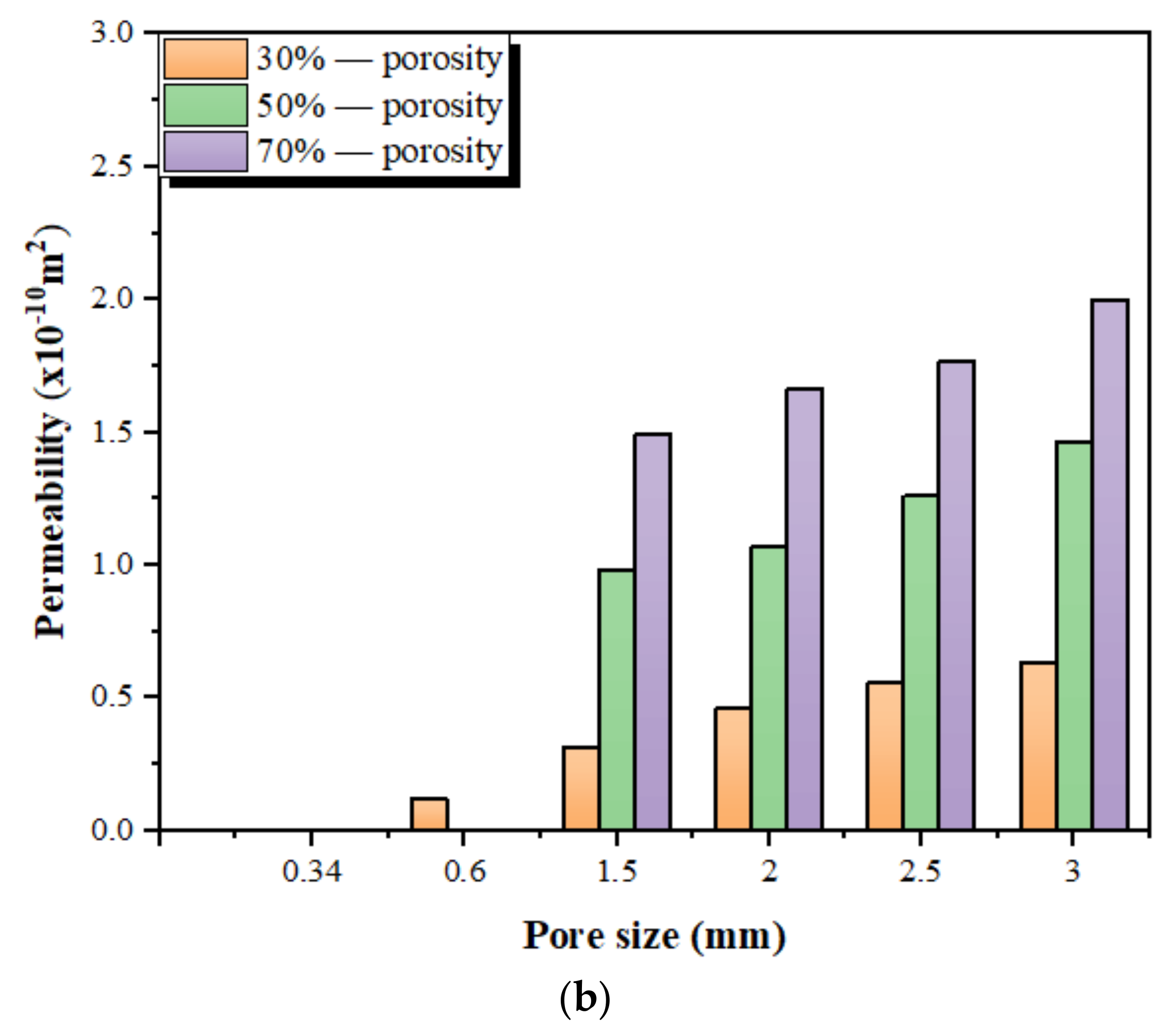
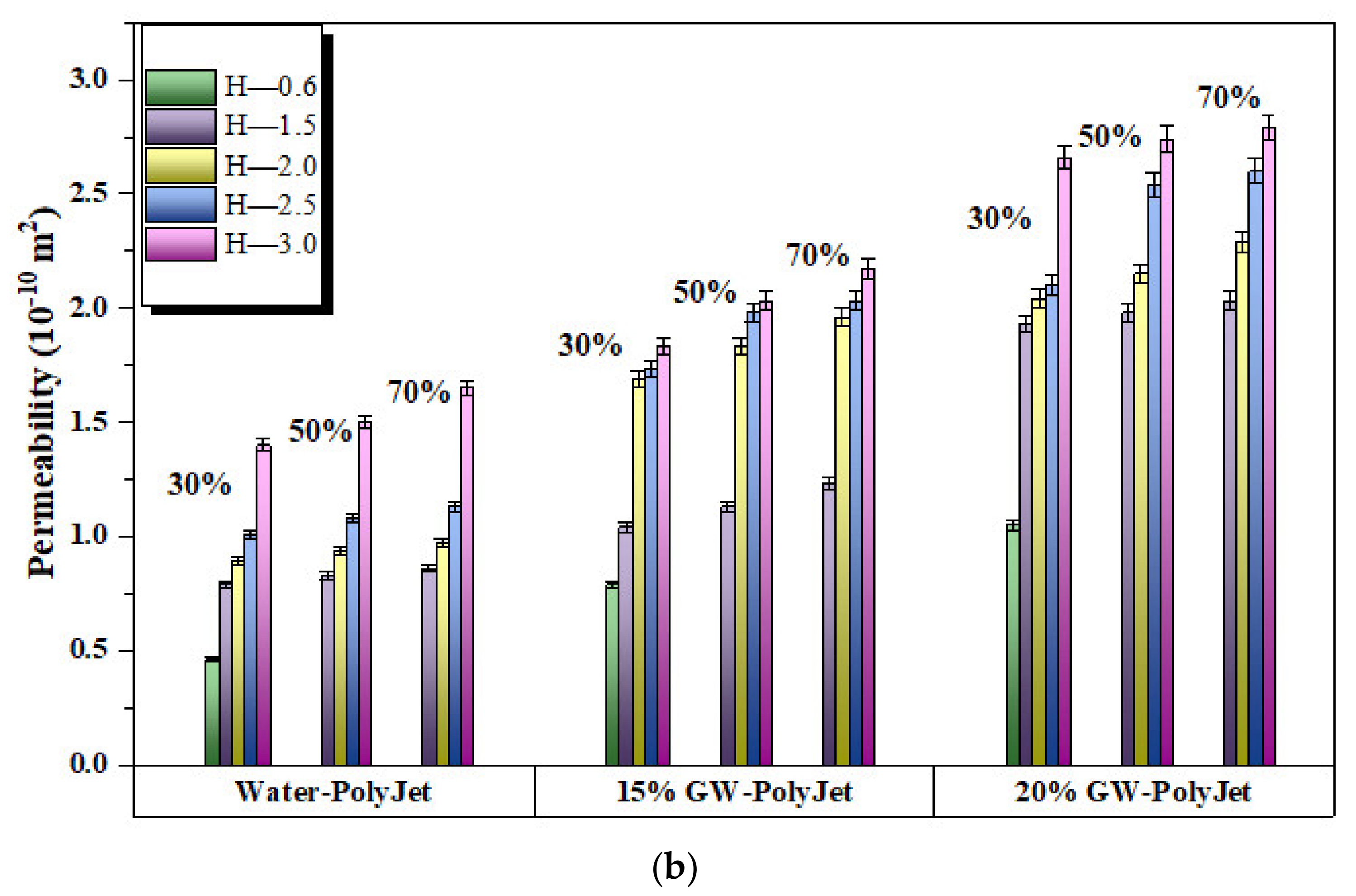
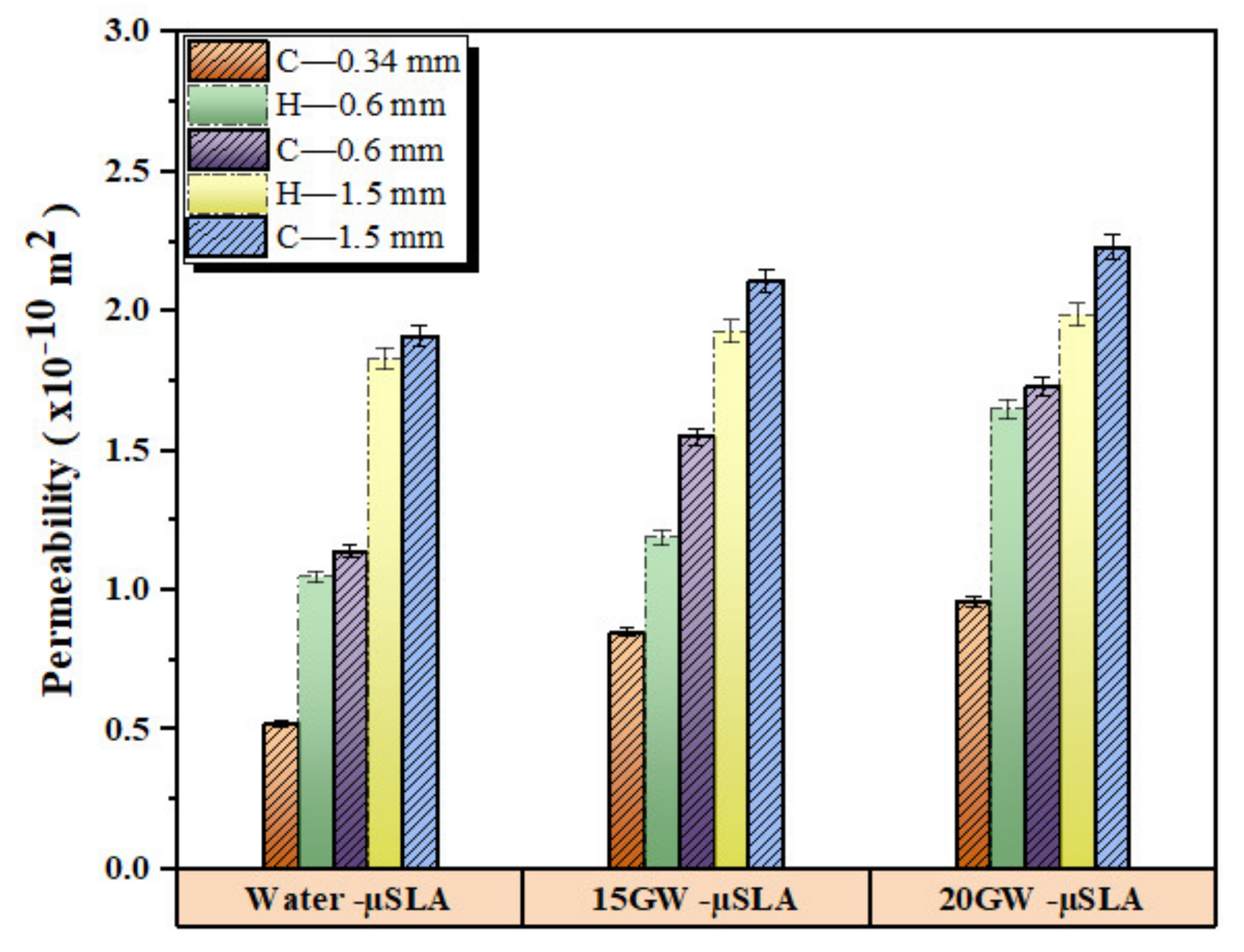
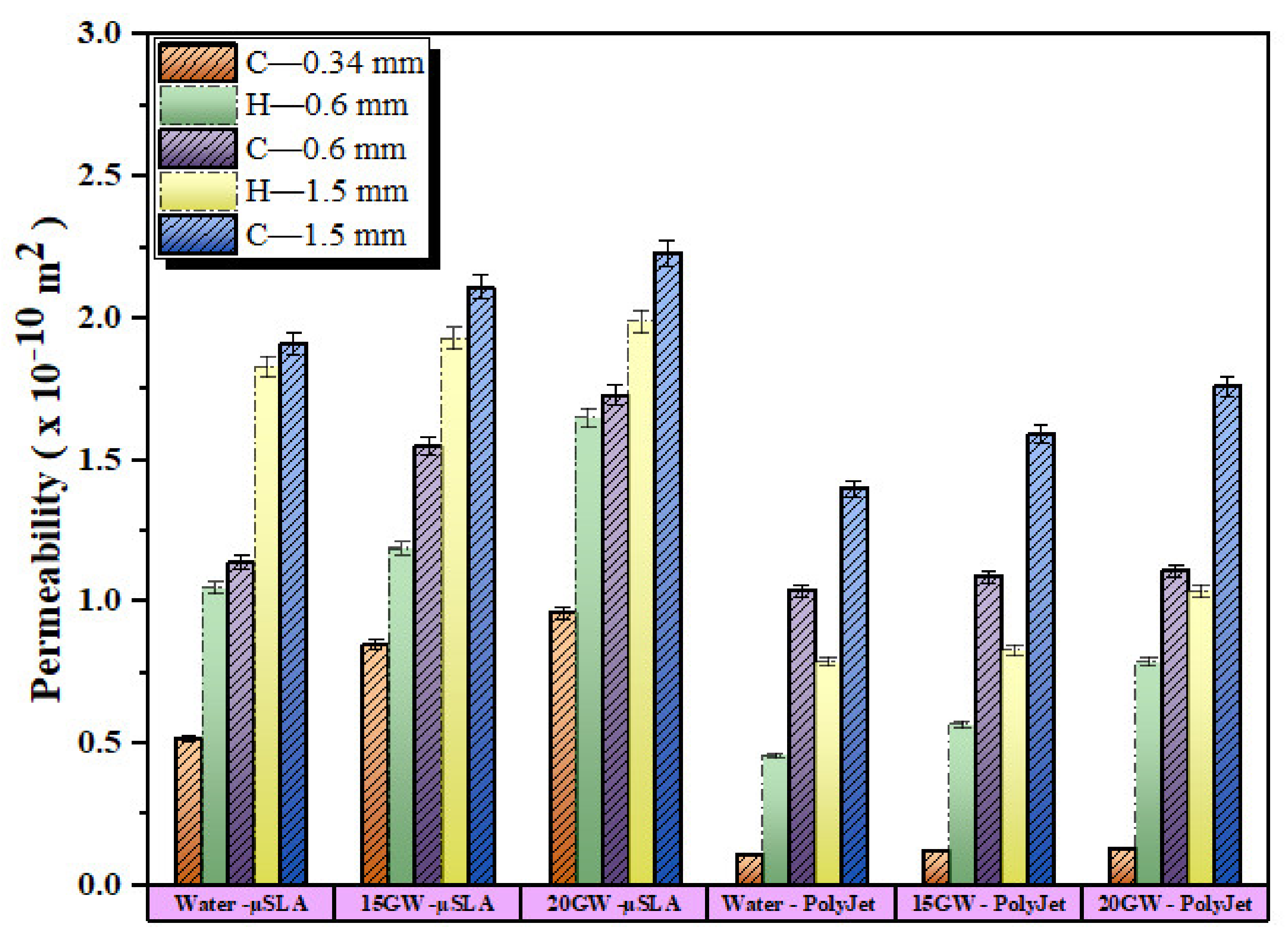
3.3. Experimentally Measured Permeability
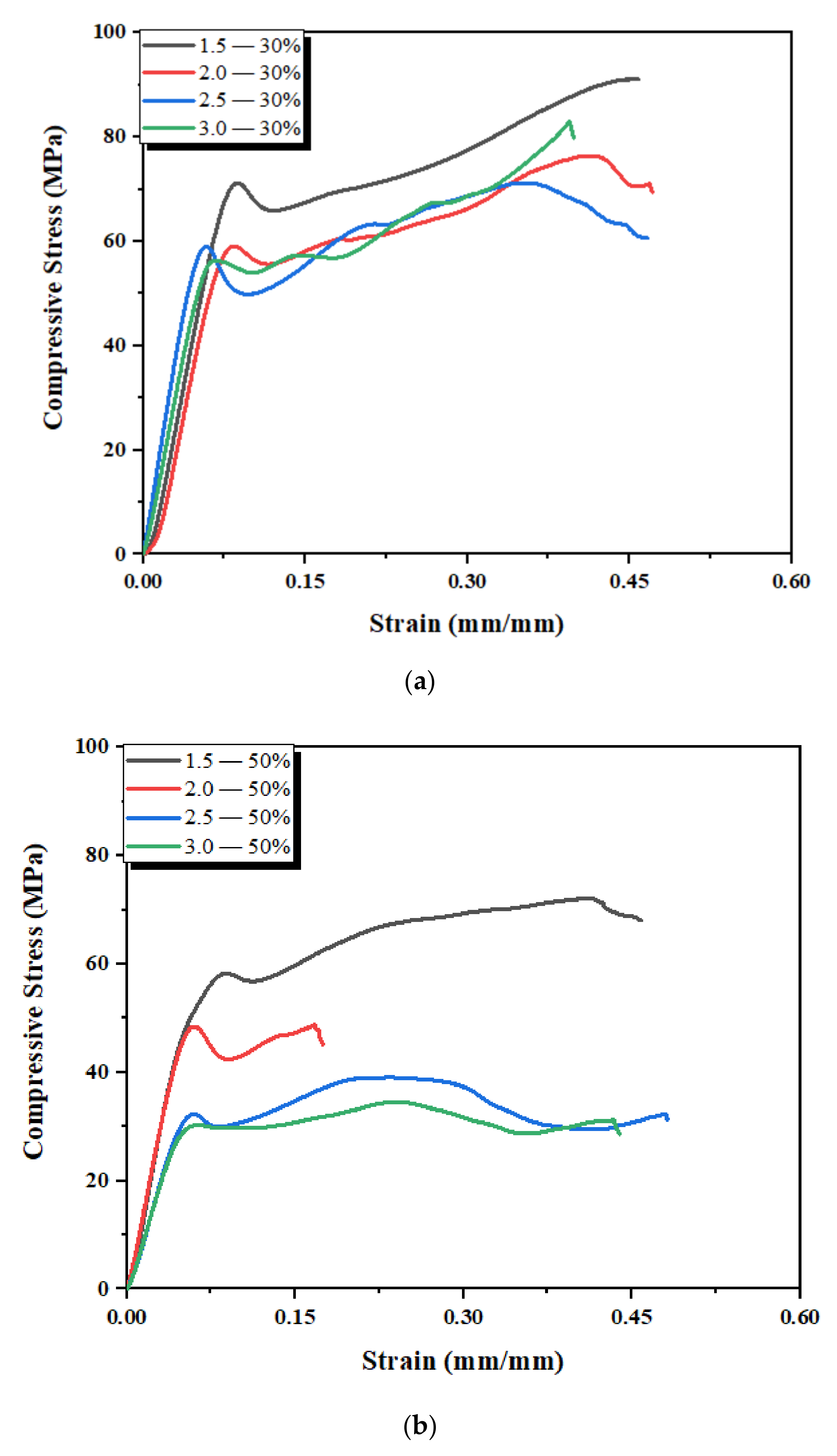
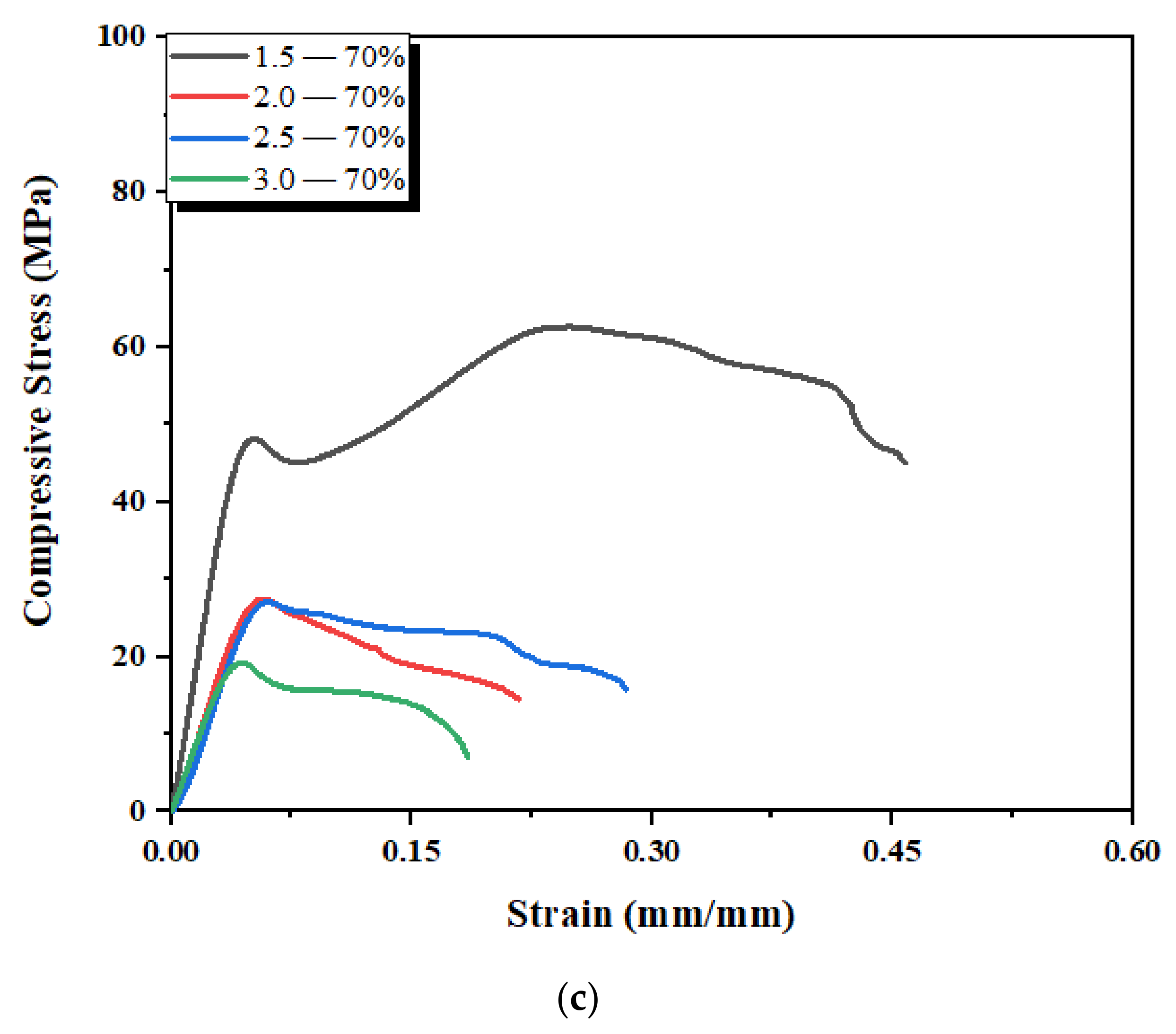
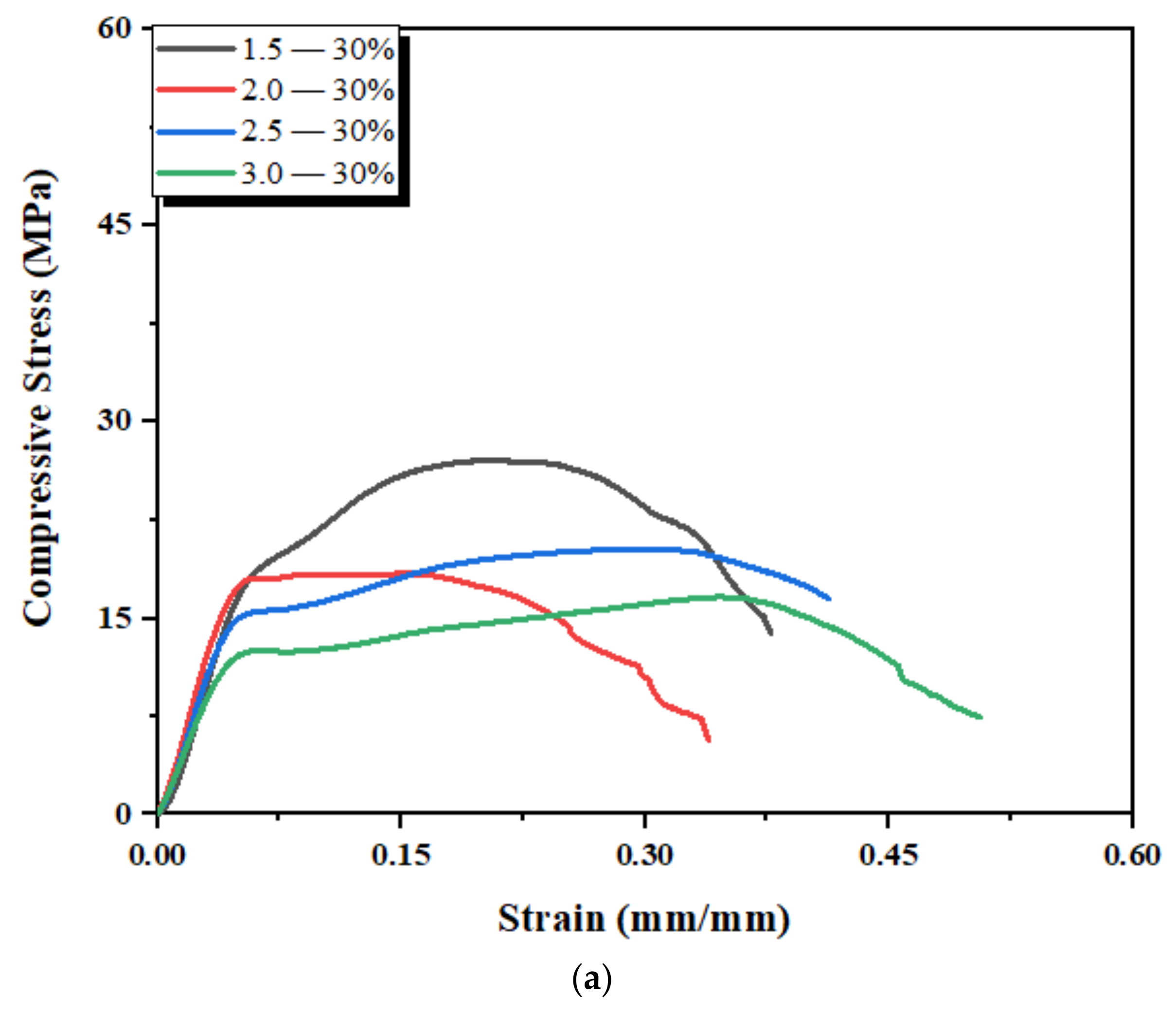
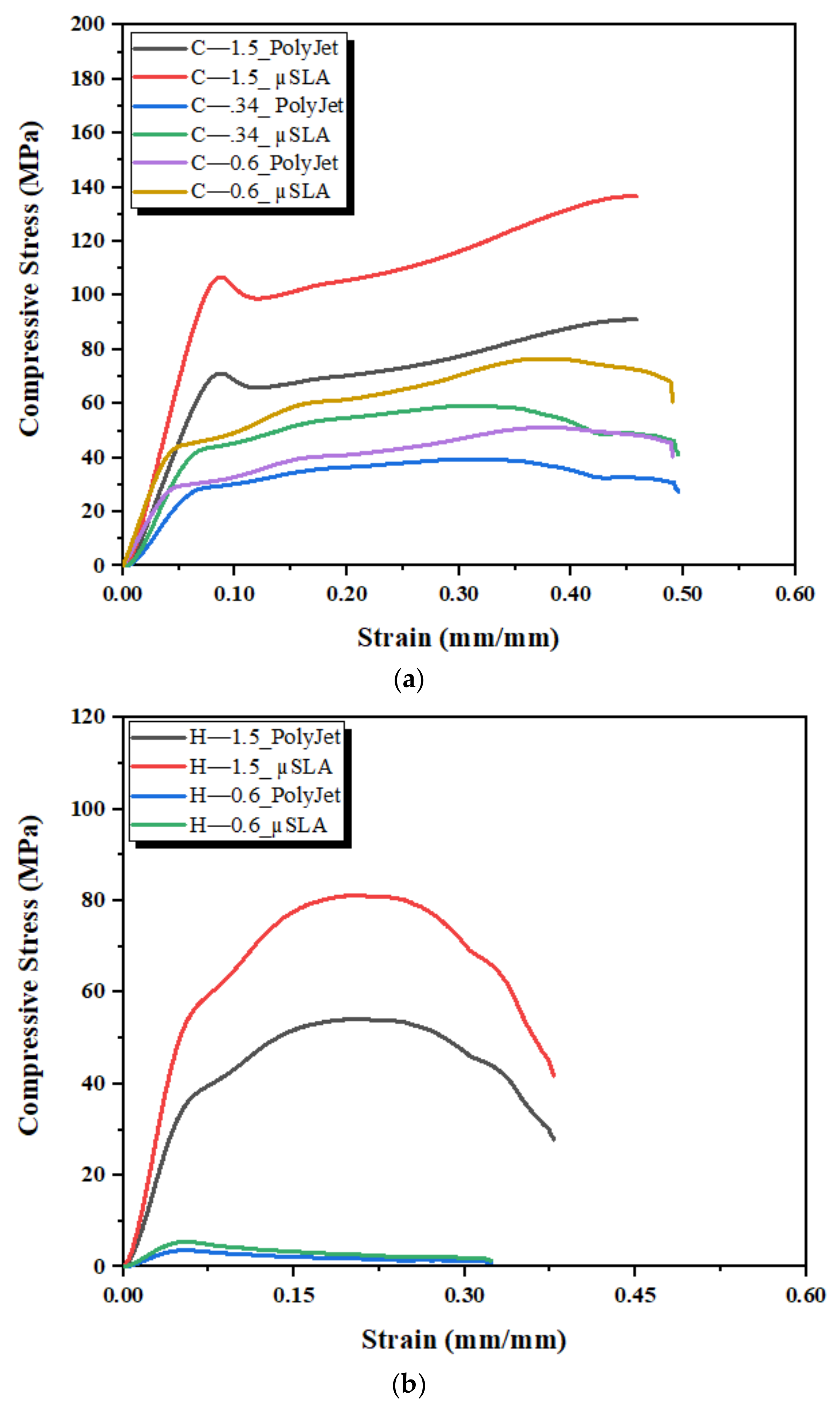
3.4. Mechanical Properties of 3D-Printed Scaffold Structures
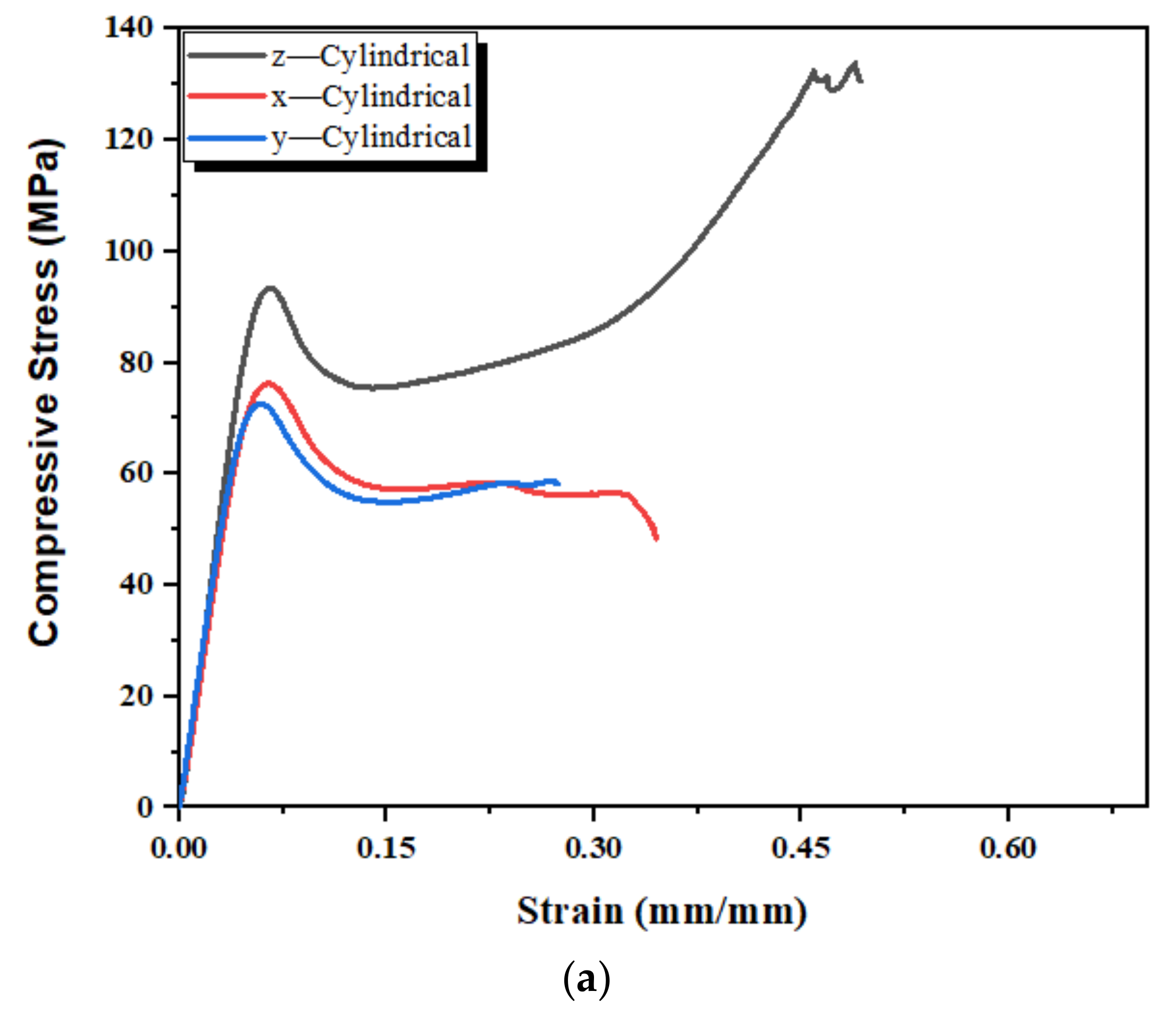
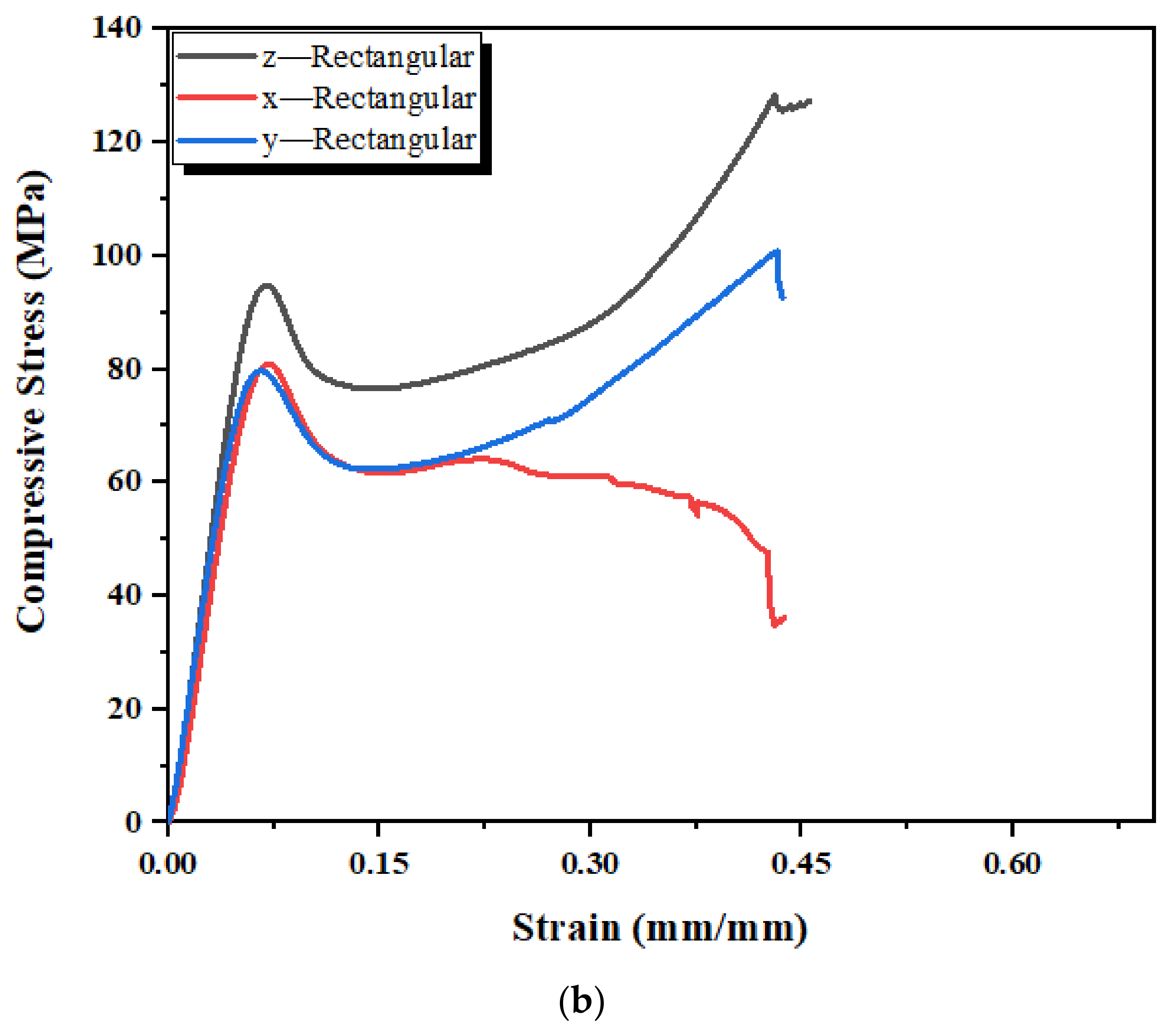
3.5. Mechanical Properties of 3D-Printed Standard Samples
4. Discussions
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- El-Rashidy, A.A.; Roether, J.A.; Harhaus, L.; Kneser, U.; Boccaccini, A.R. Regenerating bone with bioactive glass scaffolds: A review of in vivo studies in bone defect models. Acta Biomater. 2017, 62, 1–28. [Google Scholar] [CrossRef]
- Du, X.; Fu, S.; Zhu, Y. 3D printing of ceramic-based scaffolds for bone tissue engineering: An overview. J. Mater. Chem. B 2018, 6, 4397–4412. [Google Scholar] [CrossRef] [PubMed]
- Campana, V.; Milano, G.; Pagano, E.; Barba, B.; Salonna, G.; Lattanzi, W.; Logroscino, G. Bone substitutes in orthopaedic surgery: From basic science to clinical practice. J. Mater. Sci. Mater. Med. 2014, 25, 2445–2461. [Google Scholar] [CrossRef] [PubMed]
- Syahrom, A.; Abdul Kadir, M.R.; Abdullah, J.; Öchsner, A. Permeability studies of artificial and natural cancellous bone structures. Med. Eng. Phys. 2013, 35, 792–799. [Google Scholar] [CrossRef] [PubMed]
- Zhao, F.; Vaughan, T.J.; Mcnamara, L.M. Multiscale fluid–structure interaction modelling to determine the mechanical stimulation of bone cells in a tissue engineered scaffold. Biomech. Model. Mechanobiol. 2015, 14, 231–243. [Google Scholar] [CrossRef]
- Murugan, R.; Ramakrishna, S. Development of nanocomposites for bone grafting. Compos. Sci. Technol. 2005, 65, 2385–2406. [Google Scholar] [CrossRef]
- Yunus Basha, R.; Sampath, S.K.; Doble, M. Design of biocomposite materials for bone tissue regeneration. Mater. Sci. Eng. C 2015, 57, 452–463. [Google Scholar] [CrossRef] [PubMed]
- Bajaj, P.; Schweller, R.M.; Khademhosseini, A.; West, J.L.; Bashir, R. 3D biofabrication strategies for tissue engineering and regenerative medicine. Annu. Rev. Biomed. Eng. 2014, 16, 247–276. [Google Scholar] [CrossRef] [PubMed]
- Chocholata, P.; Kulda, B.; Babuska, V. Fabrication of scaffolds for bone-tissue regeneration. Materials 2019, 14, 568. [Google Scholar] [CrossRef]
- Gotman, I.; Ben-David, D.; Unger, R.E.; Böse, T.; Gutmanas, E.Y.; Kirkpatrick, C.J. Mesenchymal stem cell proliferation and differentiation on load-bearing trabecular Nitinol scaffolds. Acta Biomater. 2013, 9, 8440–8448. [Google Scholar] [CrossRef]
- Liu, B.; Lin, P.; Shen, Y.; Dong, Y. Porous bioceramics reinforced by coating gelatin. J. Mater. Sci. Mater. Med. 2008, 19, 1203–1207. [Google Scholar] [CrossRef]
- Hutmacher, D.W. Scaffolds in tissue engineering bone and cartilage. Biomaterials 2000, 21, 2529–2543. [Google Scholar] [CrossRef]
- Wu, S.; Liu, X.; Yeung, K.W.K.; Yang, X. Biomimetic porous scaffolds for bone tissue engineering. Mater. Sci. Eng. R Rep. 2014, 80, 1–36. [Google Scholar] [CrossRef]
- Warnke, P.H. Rapid prototyping: Porous titanium alloy scaffolds produced by selective laser melting for bone tissue engineering. Tissue Eng. Part C Methods 2009, 15, 115–124. [Google Scholar] [CrossRef] [PubMed]
- Van Bael, S. The effect of pore geometry on the in vitro biological behavior of human periosteum-derived cells seeded on selective laser-melted Ti6Al4V bone scaffolds. Acta Biomater. 2012, 8, 2824–2834. [Google Scholar] [CrossRef] [PubMed]
- Sun, W.; Hu, X. Reasoning Boolean operation-based modeling for heterogeneous objects. CAD Comput. Aided Des. 2002, 34, 481–488. [Google Scholar] [CrossRef]
- Choy, S.Y.; Sun, C.N.; Leong, K.F.; Wei, J. Compressive properties of functionally graded lattice structures manufactured by selective laser melting. Mater. Des. 2017, 131, 112–120. [Google Scholar] [CrossRef]
- O’Brien, F.J.; Harley, B.A.; Waller, M.A.; Yannas, I.V.; Gibson, L.J.; Prendergast, P.J. The effect of pore size on permeability and cell attachment in collagen scaffolds for tissue engineering. Technol. Health Care 2007, 15, 3–17. [Google Scholar] [CrossRef]
- Melchels, F.P.W.; Barradas, A.M.C.; Van Blitterswijk, C.A.; De Boer, J.; Feijen, J.; Grijpma, D.W. Effects of the architecture of tissue engineering scaffolds on cell seeding and culturing. Acta Biomater. 2010, 6, 4208–4217. [Google Scholar] [CrossRef]
- Jessica, M.K.; Scott, J.H. Differential effects of designed scaffold permeability on chondrogenesis by chondrocytes and bone marrow stromal cells. Biomaterials 2010, 31, 279–287. [Google Scholar]
- Furumoto, T.; Koizumi, A.; Alkahari, M.R.; Anayama, R.; Hosokawa, A.; Tanaka, R.; Ueda, T. Permeability and strength of a porous metal structure fabricated by additive manufacturing. J. Mater. Process. Technol. 2015, 219, 10–16. [Google Scholar] [CrossRef]
- Syahrom, A.; Abdul Kadir, M.R.; Abdullah, J.; Öchsner, A. Permeability study of cancellous bone and its idealised structures. Med. Eng. Phys. 2015, 37, 77–86. [Google Scholar] [CrossRef] [PubMed]
- Ochoa, I.; Sanz-Herrera, A.; Sanz-Herrera, J.M.; Doblaré, M.; Yunos, D.M.; Boccaccini, A.R. Permeability evaluation of 45S5 Bioglass®-based scaffolds for bone tissue engineering. J. Biomech. 2009, 42, 257–260. [Google Scholar] [CrossRef]
- Sandino, C.; Kroliczek, P.; McErlain, D.D.; Boyd, S.K. Predicting the permeability of trabecular bone by micro-computed tomography and finite element modeling. J. Biomech. 2014, 47, 3129–3134. [Google Scholar] [CrossRef] [PubMed]
- Malachanne, E.; Dureisseix, D.; Cañadas, P.; Jourdan, F. Experimental and numerical identification of cortical bone permeability. J. Biomech. 2008, 41, 721–725. [Google Scholar] [CrossRef]
- Chor, M.V.; Li, W. A permeability measurement system for tissue engineering scaffolds. Meas. Sci. Technol. 2007, 18, 208–216. [Google Scholar] [CrossRef]
- Grimm, M.J.; Williams, J.L. Measurements of permeability in human calcaneal trabecular bone. J. Biomech. 1997, 30, 743–745. [Google Scholar] [CrossRef]
- Beno, T.; Yoon, Y.J.; Cowin, S.C.; Fritton, S.P. Estimation of bone permeability using accurate microstructural measurements. J. Biomech. 2006, 39, 2378–2387. [Google Scholar] [CrossRef]
- Kohles, S.S.; Roberts, J.B.; Upton, M.L.; Wilson, C.G.; Bonassar, L.J.; Schlichting, A.L. Direct perfusion measurements of cancellous bone anisotropic permeability. J. Biomech. 2001, 34, 1197–1202. [Google Scholar] [CrossRef]
- Swider, P.; Conroy, M.; Pedrono, A.; Ambard, D.; Mantell, S.; Soballe, K. Use of high-resolution MRI for investigation of fluid flow and global permeability in a material with interconnected porosity. J. Biomech. 2007, 40, 2112–2118. [Google Scholar] [CrossRef]
- Barba, D.; Alabort, E.; Reed, R.C. Synthetic bone: Design by additive manufacturing. Acta Biomater. 2019, 97, 637–656. [Google Scholar] [CrossRef] [PubMed]
- Melchels, F.P.W.; Bertoldi, K.; Gabbrielli, R.; Velders, A.H.; Feijen, J.; Grijpma, D.W. Mathematically defined tissue engineering scaffold architectures prepared by stereolithography. Biomaterials 2010, 31, 6909–6916. [Google Scholar] [CrossRef]
- Brennan, S.A.; Brabazon, D.; O’Byrne, J.M. Effect of vibration on the shear strength of impacted bone graft in revision hip surgery. J. Bone Jt. Surg. Ser. B 2011, 93-B, 755–759. [Google Scholar] [CrossRef]
- Mcmenamin, P.G.; Quayle, M.R.; Mchenry, C.R.; Adams, J.W. The production of anatomical teaching resources using three-dimensional (3D) printing technology. Anat. Sci. Educ. 2014, 7, 479–486. [Google Scholar] [CrossRef] [PubMed]
- Msallem, B.; Sharma, N.; Cao, S.; Halbeisen, F.S.; Zeilhofer, H.F.; Thieringer, F.M. Evaluation of the Dimensional Accuracy of 3D-Printed Anatomical Mandibular Models Using FFF, SLA, SLS, MJ, and BJ Printing Technology. J. Clin. Med. 2020, 9, 817. [Google Scholar] [CrossRef] [PubMed]
- Kim, C.G.; Han, K.S.; Lee, S.; Kim, M.C.; Kim, S.Y.; Nah, J. Fabrication of Biocompatible Polycaprolactone–Hydroxyapatite Composite Filaments for the FDM 3D Printing of Bone Scaffolds. Appl. Sci. 2021, 11, 6351. [Google Scholar]
- Surface Energy Methods Summary. Available online: https://www.jascoint.co.jp/products/contact-angle/pdf/ap72.pdf (accessed on 1 May 2021).
- Kruczek, B. Carman–Kozeny Equation. In Encyclopedia of Membranes; Drioli, E., Giorno, L., Eds.; Springer: Berlin/Heidelberg, Germany, 2014. [Google Scholar]
- Heinl, P.; Müller, L.; Körner, C.; Singer, R.F. Cellular Ti-6Al-4V structures with interconnected macro porosity for bone implants fabricated by selective electron beam melting. Acta Biomater. 2008, 4, 1536–1544. [Google Scholar] [CrossRef] [PubMed]
- Zein, I.; Hutmacher, D.W.; Tan, K.C.; Teoh, S.H. Fused deposition modeling of novel scaffold architectures for tissue engineering applications. Biomaterials 2002, 23, 1169–1185. [Google Scholar] [CrossRef]
- Seitz, H.; Rieder, W.; Irsen, S.; Leukers, B.; Tille, C. Three-dimensional printing of porous ceramic scaffolds for bone tissue engineering. J. Biomed. Mater. Res. Part B Appl. Biomater. 2005, 74, 782–788. [Google Scholar] [CrossRef]
- Liu, Y.; Lu, Y.; Tian, X.; Cui, G.; Zhao, Y.; Yang, Q.; Yu, S.; Xing, G.; Zhang, B. Segmental bone regeneration using an rhBMP-2-loaded gelatin/nanohydroxyapatite/fibrin scaffold in a rabbit model. Biomaterials 2009, 30, 6276–6285. [Google Scholar] [CrossRef]
- Rosenson, R.S.; McCormick, A.; Uretz, E.F. Distribution of blood viscosity values and biochemical correlates in healthy adults. Clin. Chem. 1996, 72, 1189–1195. [Google Scholar] [CrossRef]
- ASTM D695-15; Standard Test Method for Compressive Properties of Rigid Plastics. ASTM International 8 January 2008. Available online: https://www.astm.org/Standards/D695.htm (accessed on 1 May 2021).
- Nauman, E.A.; Fong, K.E.; Keaveny, T.M. Dependence of Intertrabecular Permeability on Flow Direction and Anatomic Site. Ann. Biomed. Eng. 1999, 27, 517–524. [Google Scholar] [CrossRef] [PubMed]
- Beaudoin, A.J.; Mihalko, W.M.; Krause, W.R. Finite element modelling of polymethylmethacrylate flow through cancellous bone. J. Biomech. 1991, 24, 127–129. [Google Scholar] [PubMed]
- Ochia, R.S.; Ching, R.P. Hydraulic resistance and permeability in human lumbar vertebral bodies. J. Biomech. Eng. 2002, 124, 533–537. [Google Scholar] [CrossRef]
- Wu, Y.C.; Lee, T.M.; Chiu, K.H.; Shaw, S.Y.; Yang, C.Y. A comparative study of the physical and mechanical properties of three natural corals based on the criteria for bone-tissue engineering scaffolds. J. Mater. Sci. Mater. Med. 2009, 20, 1273–1280. [Google Scholar] [CrossRef]
- Lipowiecki, M.; Ryvolova, M.; Tottosi, A.; Kolmer, N.; Naher, S.; Brennan, S.; Vazquez, M.; Brabazon, D. Permeability of rapid prototyped artificial bone scaffold structures. J. Biomed. Mater. Res. Part A 2014, 102, 4127–4135. [Google Scholar]
- Li, Y.; Yang, C.; Zhao, H.; Qu, S.; Li, Y. New developments of ti-based alloys for biomedical applications. Materials 2014, 7, 1709–1800. [Google Scholar] [CrossRef]
- Zaharin, H.A.; Rani, A.M.A.; Azam, I.F.; Ginta, T.L.; Salliha, N.; Ahmad, A.; Yunus, N.A.; Zulkifi, T.Z.A. Effect of Unit Cell Type and Pore Size on Porosity and Mechanical Behavior of Additively Manufactured Ti6Al4V Scaffolds. Materials 2018, 11, 2402. [Google Scholar] [CrossRef] [PubMed]




| Pore Size mm | Porosity Level | |||||
|---|---|---|---|---|---|---|
| 30% | 50% | 70% | ||||
| 0.2 | C * | H * | C * | H * | C * | H * |
| 0.34 | C | H * | C * | H * | C * | H * |
| 0.6 | C | H | C | H * | C * | H * |
| 1.5 | C | H | C | H | C | H |
| 2.0 | C | H | C | H | C | H |
| 2.5 | C | H | C | H | C | H |
| 3.0 | C | H | C | H | C | H |
| Test Fluid | Tap Water | 15% Glycerol–Water | 20% Glycerol–Water |
|---|---|---|---|
| Density | 998 | 1045 | 1060 |
| Pressure (Pa) | 7833 | 8192 | 8318 |
| Test Liquids | Contact Angle ° | Surface Energy mN/m | ||
|---|---|---|---|---|
| VeroClear | PlasWhite | VeroClear | PlasWhite | |
| Tap Water | 67.0 | 69.1 | 41.7 | 38.5 |
| 15% Glycerol–Water | 70.0 | 72.6 | 40.0 | 37.9 |
| 20% Glycerol–Water | 72.9 | 73.7 | 39.8 | 37.1 |
| Permeability (×10−10 m2) | PolyJet | µSLA | ||
|---|---|---|---|---|
| C | H | C | H | |
| Measured | 1.05–2.83 | 0.46–2.75 | 0.52–2.23 | 1.05–1.99 |
| Theoretical | 0.3–2.11 | 0.12–2.0 | 0.3–2.11 | 0.12–2.0 |
| Pore Size (mm) | Elastic Modulus | Yield Strength | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| (GPa) | (MPa) | |||||||||||
| 30% | 50% | 70% | 30% | 50% | 70% | |||||||
| C | H | C | H | C | H | C | H | C | H | C | H | |
| 0.34 | 0.4 | - | - | - | - | - | 28 | - | - | - | - | - |
| 0.6 | 0.5 | 0.4 | 0.5 | - | - | - | 30 | 25 | 29 | - | - | - |
| 1.5 | 1.3 | 0.6 | 1.1 | 0.7 | 1.0 | 0.9 | 72 | 42 | 60 | 40 | 48 | 38 |
| 2.0 | 0.9 | 0.5 | 0.8 | 0.6 | 0.7 | 0.5 | 60 | 37 | 50 | 33 | 28 | 19 |
| 2.5 | 0.7 | 0.4 | 0.6 | 0.2 | 0.5 | 0.2 | 58 | 30 | 34 | 13 | 26 | 8 |
| 3.0 | 0.7 | 0.3 | 0.5 | 0.2 | 0.4 | 0.2 | 56 | 25 | 30 | 10 | 18 | 7.5 |
| Mechanical Properties | Pore Shape | Pore Size (mm) | ||
|---|---|---|---|---|
| 0.34 | 0.6 | 1.5 | ||
| Elastic Modulus (MPa) | C | 600 | 800 | 1900 |
| H | - | 600 | 900 | |
| Yield strength (MPa) | C | 42 | 45 | 108 |
| H | - | 38 | 63 | |
| Mechanical Properties (MPa) | Rectangular | Cylindrical | ||||
|---|---|---|---|---|---|---|
| x-Direction | y-Direction | z-Direction | x-Direction | y-Direction | z-Direction | |
| Elastic Modulus | 1400 | 1300 | 1600 | 1300 | 1200 | 1600 |
| Yield Strength | 82 | 80 | 96 | 78 | 74 | 100 |
| Anatomic Region | Material Used | Fluid | Permeability m2 | Ref |
|---|---|---|---|---|
| Cancellous bone | Simulated cancellous bone | Simulated blood | 10−11 to 10−7 | [22] |
| Human Vertebral body | Human cadaveric bone | Deionized water | 80.5 × 10−10 | [46] |
| Human Proximal femur | 27.6 × 10−10 | |||
| Human Calcaneus | 35.4 × 10−10 | |||
| Human, proximal tibia | Simulated cancellous bone | Simulated PMMA | 76.8 × 10−10 | [47] |
| Human calcaneal | Human cadaveric bone | Raw linseed oil | 4–109.7 × 10−10 | [27] |
| Human cadaveric lumbar | Human cadaveric bone | Extra virgin olive oil | 4.899 × 10−10 | [48] |
| Human vertebral body lumbar | 10.7–109 × 10−10 | |||
| Human lumbar | 215–743 × 10−10 | |||
| Corals | Coral samples | Water | 0.12–4.46 × 10−9 | [49] |
| µSLA 3D-printed | Accura 60 resin | Water | 1.84–41.9 × 10−10 | [50] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rasheed, S.; Lughmani, W.A.; Obeidi, M.A.; Brabazon, D.; Ahad, I.U. Additive Manufacturing of Bone Scaffolds Using PolyJet and Stereolithography Techniques. Appl. Sci. 2021, 11, 7336. https://doi.org/10.3390/app11167336
Rasheed S, Lughmani WA, Obeidi MA, Brabazon D, Ahad IU. Additive Manufacturing of Bone Scaffolds Using PolyJet and Stereolithography Techniques. Applied Sciences. 2021; 11(16):7336. https://doi.org/10.3390/app11167336
Chicago/Turabian StyleRasheed, Shummaila, Waqas Akbar Lughmani, Muhannad Ahmed Obeidi, Dermot Brabazon, and Inam Ul Ahad. 2021. "Additive Manufacturing of Bone Scaffolds Using PolyJet and Stereolithography Techniques" Applied Sciences 11, no. 16: 7336. https://doi.org/10.3390/app11167336
APA StyleRasheed, S., Lughmani, W. A., Obeidi, M. A., Brabazon, D., & Ahad, I. U. (2021). Additive Manufacturing of Bone Scaffolds Using PolyJet and Stereolithography Techniques. Applied Sciences, 11(16), 7336. https://doi.org/10.3390/app11167336








