3.1. Body Compositions
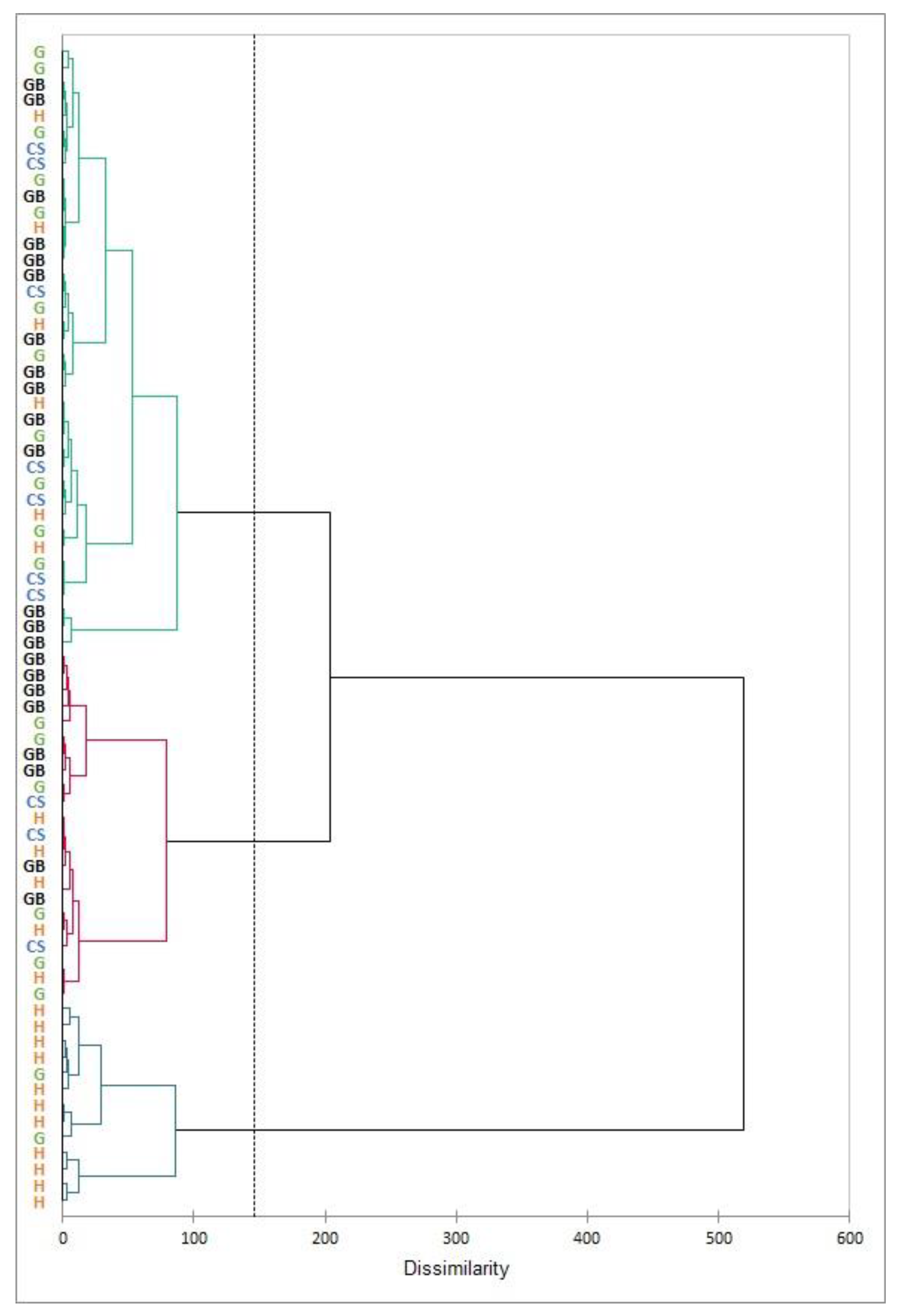
Chemical compositional data of ceramic bodies obtained by ICP-OES were treated statistically by hierarchical clustering analysis using eight variables (Na, Mg, Al, K, Ca, Ti, Mn, and Fe) and Euclidean distances between samples (Ward method) to group them. This multivariate statistical treatment (
Figure 3) revealed several differentiated groups whose compositions are summarized in
Table 1 (oxides were calculated by stoichiometry). Calcium was one of the elements whose content varied between groups and, together with other components, allowed the characterization of each group. The groups presented calcareous matrix whose composition ranged from 5 to 18% CaO. Group 3 presented lower average calcium proportion (7.5% CaO) than the other calcareous matrixes, but potassium, magnesium, and aluminum contents were higher. The other two groups with calcareous bodies (group 1 and 2) had similar calcium quantities, but different minor element contents. All studied decorated ceramics were produced with calcareous clay, only a less frequent group of slip-ware decorated with red/brown painted designs (not included in this paper) was made with non-calcareous clay (0.5–1% CaO) [
10]. The use of calcareous clays for manufacturing glazed pottery was expected in order to obtain light-colored bodies [
11].
The reference groups obtained by cluster analysis, according to the chemical composition of the bodies, could be related to the decoration types manufactured in Albarracin. Groups 1 and 2 corresponded to ceramics decorated with white or green glazes. Most of the cuerda-seca fragments were included inside group 1. Group 3 was mainly formed by honey-glazed objects, although some yellow fragments were also included in groups 1 and 2. This fact suggested that yellow-glazed ceramics could be manufactured with different raw materials or in different workshops.
According to chemical results, it seems that selection of the raw materials was performed depending on the decoration. In groups 1 and 2, it can be assumed that the same type of illitic clay was employed for the manufacture of the pottery. However, the raw materials used in the ceramic production of group 3 differed in the chemical composition from those employed for groups 1 and 2. Therefore, ceramics included in this last group can belong to another production workshop or even another area.
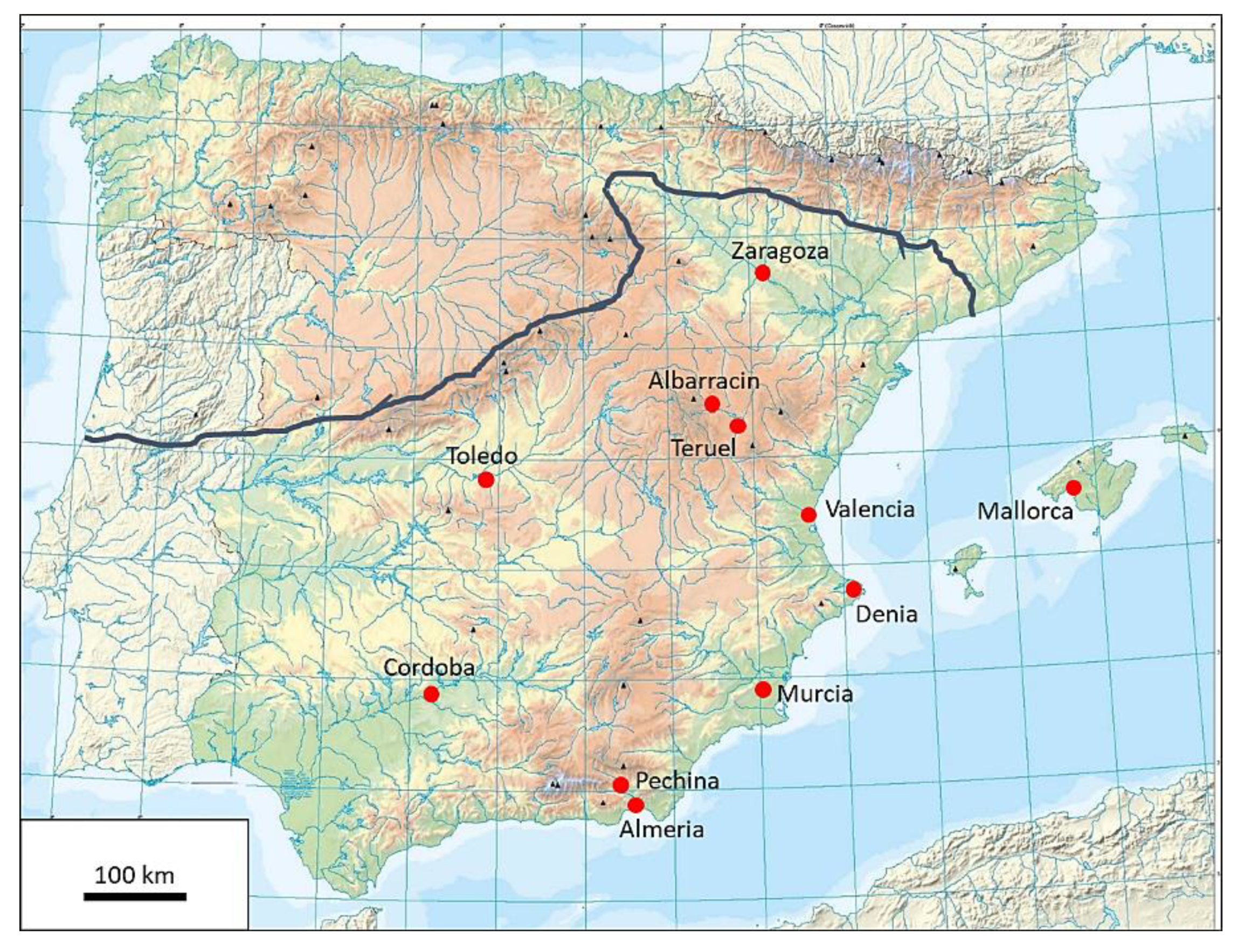
Comparing the results obtained from Albarracin ceramics with analysis of clay bodies of Islamic productions from other areas of the Iberian Peninsula [
4,
12,
13,
14,
15,
16], it can be pointed out that all the clays used in decorated ceramics were also Ca-rich (8–22% CaO) and had similar contents of iron (4–6% Fe
2O
3) and fluxing agents (4–5% Na
2O+K
2O). The use of calcareous clays for decorated pottery justified the buff colors of the bodies due to the growth of calcium silicates, which incorporated some iron atoms to their structure and decreased the content of iron oxides, responsible for red color [
11]. A great similarity was found when we compared these objects from Albarracin with other pottery productions from Muslim centers, such as Cordoba, Denia, Granada, Mallorca, Murcia, Pechina, or Zaragoza [
4,
13,
14,
15,
16]. For instance, the Islamic production from Zaragoza during the 11–12th centuries CE was studied [
13] and four reference groups were described, based on the type of body clays employed. In these workshops from Zaragoza, differences among the calcareous groups were also based on the amounts of alkalis and the groups were related to the final decoration and to the utility of the pottery. The selection of the raw materials implied great knowledge of their characteristics and properties.
In the south of al-Andalus, the production of ceramics was very popular with a great number of workshops. Cordoba, Denia, Granada, Murcia, and Pechina were important pottery centers during the 11th–13th centuries CE [
4,
14,
16]. The body composition of these glazed productions from Islamic workshops was very similar to ceramics from Albarracin with high contents of calcium (groups 1 and 2), although ceramics manufactured in some of them were richer in calcium content than Albarracin. The study carried out in the workshop of San Nicolas [
14], situated in Murcia and dating to the 10th century CE, revealed some differences with the pastes from Albarracin, but three different references groups can be also distinguished; these three groups were also related to the decoration to be applied. Again, this fact demonstrated the specialization of the potters on the selection of the material.
3.2. White Tin-Opacified Glazes
Different types of decoration on the Islamic pottery from Albarracin
Taifa: white tin-opacified glazes, colored glazes, and
cuerda seca were studied for their characterization. In the case of white tin-opacified glazed samples, the compositions of white glazes on the main side and the yellow-honey glazes on the secondary side of these fragments were studied. Further, brown and green decorations on the white glazes were analyzed in most ceramic samples. Tin-opacified glazes presented an average chemical composition that was very homogeneous in the main side, as can be seen in
Table 2. The major elements found in the white glazes were silicon (SiO
2 42.8%), lead (PbO 36.2%), and tin (SnO
2 7.0%), mixed with contents below 5% of aluminum, iron, alkalis (potassium and sodium) and calcium oxides. Glazes produced during the medieval period in the Iberian Peninsula were characterized by lead-rich contents. The low percentage of aluminum (Al
2O
3 < 4.5%) could be attributed to the use of small amounts of clay to produce the glaze, or also to the diffusion of aluminum from the paste to the glaze during firing [
17]. Iron and calcium were not added to the recipes of glazes as main components. Their very low proportions (FeO < 1% and CaO 2.86%) were also due to the migration of iron and calcium into the glazes. Higher contents of iron would induce yellow or brown colors in the glaze. Therefore, raw materials employed in the production of tin-opacified glazes were carefully chosen in order to obtain a white color of glaze.
Green and brown decorations on tin-glazed samples from Albarracin
Taifa did not differ in their compositions from the white ones, except for the presence of metallic oxides responsible for the colors (
Table 2). Analyses carried out on green areas were rich in copper oxide, and brown zones had higher contents of manganese oxide. The content of CuO was in the range of 1.4–3.4% and MnO varied between 1.2 and 6.0%. The presence of copper in brown decorations can be explained due to the thin brown design that made it difficult to determinate only the composition of brown areas without including green zones.
Opacity was accomplished by the presence of small crystals of cassiterite (SnO
2) within the glaze that reflected and scattered the light [
18]. These crystals were achieved by the addition of tin oxide to the raw materials of the glazes. During the heating (above 700 °C) of the mixture, the tin oxide was dissolved and began to recrystallize as cassiterite [
19]. The amount of tin, the size of tin oxide crystals, their distribution in the glaze, and the thickness of the glaze were factors with great influence on the grade of opacification [
12,
18,
19] and they were related to the process of manufacture. The average amount of tin oxide (7% SnO
2) employed in the studied samples was in good agreement to obtain enough opacity (5–10% SnO
2) without increasing too much the price of the product. The size of tin oxide crystals in Albarracin samples varied from 500 to 800 nm. These dimensions of the crystals indicated that high temperatures (above 800 °C) were reached during the firing process and the cooling occurred fast, as it was demonstrated in the literature [
19]. The homogeneous distribution of the cassiterite crystals suggested that the tin glazes were prepared by fritting the raw materials before applying them to the ceramic body [
15,
20]. Fritting was an extended method used since ancient times to obtain glasses and glazes, which consisted in melting the raw materials (lead, tin, and silica) previously to use them as components of the glazes. Finally, these fired materials were ground to powder before preparing the glaze suspension that was applied on the surface of the body. Besides the homogeneity of the fritted material, frits presented some advantages with respect to raw materials: they reduced the risk of contraction, avoiding cracking the surface, improved the maturation of the glaze, avoided bubbles, and allowed better control of the process [
19].
Only one tin-opacified glazed sample had also white glaze on the secondary side; the others had honey glazes on the secondary side, as it has been explained before. The composition of these secondary glazes revealed a higher heterogeneity between specimens (
Table 3), but it was possibly to classify them in two different subgroups. The first one included samples with contents of SiO
2 and PbO about 37% and 46% respectively, and the other group was composed by glazes with higher SiO
2 (45%) and lower PbO (37%) proportions. In both cases, the elements added as fluxes were lead and some alkalis with contents around 3–4%. Tin oxide was not present and the proportions of aluminum and iron oxides were increased regarding main tin-opacified glazes, which evidenced the use of clays and sand not as pure as those used to manufacture white glazes. The honey/yellowish color of the glazes was achieved by iron (>2% FeO) dissolved in the glaze.
Microstructure of glazes observed by SEM differed depending on the fragment side (
Figure 4). Tin-opacified glazes on the main side of the objects (
Figure 4a) had few inclusions, while on the secondary side (transparent glazes) were abundant inclusions with large sizes (often bigger than 60 μm) and well distributed through the glaze (
Figure 4b,c). In both cases, the inclusion compositions were the same: potassium feldspars and quartz. Those microstructural differences reflected that the materials for the main decoration were carefully chosen and probably fritted for producing the main glazes [
14].
Concerning thickness, it is outstanding that glazes of the main side were thicker than the coatings applied on the secondary side in all studied samples. On average, the thickness of the layers ranged from 100 to 200 μm for tin-opacified glazes, and from 50 to 100 μm for transparent glazes.
In the contact zone between clay body and glaze, several reactions occurred during firing that favored the migration of atoms between both phases. Diffusion of the elements produced the growth of Pb-rich feldspars crystals. The development of these crystals in the interface and the reaction degree between clay body and glaze were related to the type of clay body and the firing/cooling process [
17]. Not only in tin-opacified glazes from Albarracin but also in the transparent ones, the amount of crystals and the interaction between clay paste and glaze (interface thickness < 10 μm) was minimum. It was described that, when the glaze was applied on an already fired clay body, the diffusion between both phases was reduced, and consequently the number of crystals formed and the thickness of the interface were lower [
17,
21]. This feature indicated that tin-opacified glazes manufactured in the
Taifa of Albarracin were applied on a previously fired paste (biscuited) and a second firing process was carried out to produce the decoration.
If the results obtained for tin-opacified glazes of Albarracin production are compared with other workshops of the Islamic period in the Iberian Peninsula [
4,
14], it can be pointed out that the technology (firing process and opacity) seemed to be very similar and the glaze recipes differed only slightly. In all Iberian production centers, white opaque glazes were composed by a mixture of lead–silica with tin oxide contents from 5 to 10%. Each workshop had a characteristic lead/silica ratio that could help to differentiate the productions. Islamic ceramic centers in the south and the SE of the Peninsula had higher contents of lead (45–60% PbO and 30–42% SiO
2, in general; then, PbO/SiO
2 ratios from 2 to 1.1) than Albarracin (PbO/SiO
2 = 0.8) and Zaragoza samples (in the NE), except for some examples from the Vega of Granada [
16]. The low lead contents of these northeastern groups were compensated by higher amounts of alkaline components (2–5% K
2O); this fact allows the definition of these tin-opacified glazes as potassium-lead glazes. These differences were also revealed in the results obtained with the same methodology in our laboratory in samples from Pechina (9th–10th centuries CE), given by the Laboratoire de Céramologie (Lyon, France) (unpublished data). Those characteristics proved that white tin-opacified glazes manufactured in Albarracin and Zaragoza
Taifas could be differentiated by their chemical composition from other production areas.
3.3. Monochrome Colored Glazes
Colored glazes were used for coating always both ceramic sides. Yellow/honey glazed samples were decorated on both sides of the objects with the same color; however, green glazed fragments were decorated with either green or yellow glazes on the secondary side. Compositional analyses of green and yellow monochrome glazes (
Table 4) revealed that they were rich in lead and silicon, with minor quantities of iron, aluminum, calcium, and alkali oxides. Similar chemical compositions were determined on the secondary sides. Only slight compositional differences were observed among green and yellow glazes. Some of these differences were due to elements responsible of the color of glazes. Although the final color of glazes was also influenced by the body color and the metallic oxide added into the glaze [
12,
14,
17], the latter was the responsible of the compositional differences. Therefore, yellow/honey glazes had higher amounts of FeO (1.6–4% in yellow glazes, versus 0.6–1.7% in green glazes) and green coatings were richer in CuO proportions (1.2–4% in green glazes).
An important feature of these monochrome glazes was the fact that all green glazes included tin oxide in their composition (
Table 4). Tin was detected with EDS analysis and tin-oxide crystals were observed in the green glazes. Although the tin-oxide percentages were lower than white tin-opacified glazes (
Table 3), the proportion was enough to give a certain opacity. This characteristic was also highlighted in green glazes from Zaragoza
Taifa [
22].
Inclusions were occasional in the main monochrome glazes (
Figure 5), but they were always present in the secondary sides, which implied careful treatment of raw materials again, especially for the main sides. These inclusions were mainly quartz and feldspars. It is not easy to establish an average size of the inclusions because there was a lot of variation even inside the same specimen. The glaze thickness was quite constant in the same sample, but it varied significantly among different ceramic fragments. Additionally, not only the yellow/honey glazes but also the green glazes were thicker in the main side (100–150 μm for the yellow glazes, and 150–200 μm for the green glazes) than in the less important side (80–100 μm in the yellow glazes, and 100–150 μm in the green ones).
In these types of glazes, greater interactions between body and glaze were observed by SEM (
Figure 5a). Since the reaction between ceramic paste and glaze was related with the type of developed firing process [
12,
17,
21], it can be assumed that the thickness of the interface (35–40 μm) in monochrome glazes corresponded to a single firing process. This technique of manufacture justified the great amount of lead-potassium feldspar crystals that existed in the interface, and that favored the adhesion between both layers [
23]. In addition, this type of firing process enhanced the diffusion of certain elements (such as aluminum and iron) from the body to the glaze. Aluminum and iron were incorporated into the glaze and helped the stabilization of PbO-SiO
2 glazes. Therefore, the presence of aluminum and iron in the glazes can be attributed, on the one hand, to the raw materials of the glazes and, on the other hand, to the diffusion of components from the clay to the glaze during the firing process [
12,
17].
It can be pointed out that the composition of transparent yellow/honey glazes (
Table 4) was very similar to that of the glazes employed on the secondary side of white glazes, especially to subgroup 1 (
Table 3). Inclusions were abundant in both secondary-side glazes of yellow-honey glazed and white glazed ceramics.
The comparison of chemical compositions of the honey/yellow glazes from Albarracin with productions from other Islamic workshops [
4,
12,
16,
22] confirmed differences in lead/silicon ratios. Almost all productions whose workshops were situated in the south and SE part of the Iberian Peninsula had lead oxide content between 45–60% PbO. Lead percentages of glazes produced in Albarracin were among the lowest compared with the rest of the workshops. However, productions from Albarracin and Zaragoza
Taifas had in common that their ceramics were manufactured with a unique firing process, whereas the rest of the centers of production seemed to prepare monochrome glazes on biscuited ceramics.
3.4. Cuerda-Seca Decoration
Concerning cuerda-seca samples, we determined the composition of the black line which divided different areas of the drawing, and the glazes applied inside those areas.
The analyses of the black lines showed inhomogeneous compositions (
Table 5) that made it not realistic to establish average proportions. However, the high content of SiO
2 as major element in all the samples is remarkable, mixed with lead, manganese, aluminum, and iron. Manganese was the responsible for the black color of the line, probably in the form of different oxides. Lead could be added to enhance the adherence of the pigments to the paste [
24], because the black line was applied directly on the non-fired paste, as can be deduced by the thickness of the interface (
Figure 6).
The glazes used to fill the spaces between black lines were rich in silicon and lead (
Table 5). The green glazes had tin oxide in lower proportions than white tin-opacified glazes. Although there were not many
cuerda-seca samples with yellow and green glazes among the studied samples, and the yellow ones showed great variation in lead content, the average composition of the PbO was similar to the secondary yellow/honey glazes (subgroup 2,
Table 3) on green-and-brown decorated objects, and to green monochrome glazes (
Table 4). However, the silicon proportion of
cuerda-seca glazes differed more in both colored glazes.
Once again, the presence of copper and iron oxides with percentages around 2–3% contributed to the final color of the glazes. Only a sample showed
cuerda-seca decoration with honey and green-turquoise colors. In this case, the color was due to the presence of copper, but its green hue was shifted to turquoise because the glaze had alkaline-lead composition with a significant contribution of sodium (2.5% Na
2O) and potassium (2.9% K
2O) and lower lead content (23.3% PbO). These differences in green hues were already highlighted in
cuerda-seca ceramics of the 12th century CE produced in the Iberian Peninsula [
25].
Studies of pottery with
cuerda-seca glazes manufactured in the Iberian Peninsula during the 10th–12th centuries CE revealed the use of silicon and lead as main components [
6,
24,
25]. The amounts of lead decreased from the 10th century (43–55% PbO) to 12th century CE (34–43% PbO), mainly in productions from the south of al-Andalus [
25]. Nevertheless, samples manufactured during the 11th century CE in the NE workshop of Zaragoza were characterized by high lead proportions [
24]. Contrary to other types of decoration, pottery decorated with
cuerda seca produced in Albarracin in the 11th-12th centuries CE was more similar to the recipes employed in the south of the Peninsula with lead contents around 35–38% PbO.
Examinations by SEM of polished sections of
cuerda-seca glazes confirmed the presence of abundant inclusions scattered homogeneously in the glaze (
Figure 6). The inclusion sizes varied from 50 to 70 μm and their composition was mainly quartz. The use of such an amount of inclusions and their size, plus the tin-oxide content, could be responsible for the opacity. Opacification can be reached either with undissolved inclusions into the glazes or with tin-oxide crystals scattered in the glaze, and even with both of them [
26,
27].
Cuerda-seca ceramics manufactured in the same period in al-Andalus employed both techniques [
6,
24,
25], although only tin oxide was employed as an opacifier in later centuries. Therefore, the inclusions found in the glazes played an important role in the opacity of the glazes. The thickness of the glazes reached values higher than 200 μm and the thickness of the clay-glaze interfaces was also higher (>45 μm), which implied that a single firing technique was carried out during the pottery’s manufacture. The type of firing process (single or double) depended on the workshops; both techniques were used without a clear tendency. Single firing process was more frequent, especially in workshops such as Almeria and Zaragoza [
24,
25]. This characteristic was also followed in the studied production from Albarracin
Taifa.
As it has been explained before, the technology of opacification also underwent an evolution from a double method (tin oxide and inclusions) employed in the 10th century CE to the opacity technique due exclusively to tin oxide used in the 12th century CE. However, in our case, the double methodology was followed, as in the case of the southern workshops.
It seems clear that the
cuerda-seca technique underwent an evolution during the Islamic period with variations in recipes, firing processes, and methods of opacification. The evolution did not follow a regular extension to all the production centers, but we can highlight that Albarracin was influenced by the southern centers of al-Andalus. Although archaeological studies based on the decoration motifs of
cuerda seca during the period of
Taifas emphasized that the same type of vegetal designs was also employed in Zaragoza [
6], both influences could be possible taking in mind the good relationships with northern and southern
Taifas, and the wide commercial distribution of Islamic glazed ceramics [
28,
29].
3.5. Lead and Raw Materials for Glazes
Lead isotopes ratios were measured by ICP-QMS in a selection of representative glazed samples, because of the important information in lead sources that could be provided. Natural lead consists of four isotopes: three of them (
208Pb,
207Pb and
206Pb) come from the radioactive decay of U and Th, but the fourth one (
204Pb) has a non-radiogenic origin and natural lead changes its isotopic composition. The results of twenty-three samples from Albarracin, plus two samples from Islamic workshops of Zaragoza and one sample from Pechina are shown in
Figure 7.
The lead-isotope ratios of the samples, plotted as
208Pb/
206Pb vs.
207Pb/
206Pb and
206Pb/
204Pb vs.
207Pb/
206Pb ratios (
Figure 7), offered the possibility of establishing several groups and distinguishing different features of the used lead for the production of glazes. Samples from Albarracin can be clearly separated into two groups. One of the groups was related to the fragments with yellow/honey glazes (
Figure 7a,b, on the right of both plots). The second group was formed by white tin-opacified and green glazes from Albarracin (
Figure 7a,b, on the left of both plots). Moreover, these last two types of glazes showed slight differences between them, but both had in common being tin-opacified glazes. If we observed the results of the three samples from other areas (Zaragoza and Pechina), the white tin-opacified glaze from Pechina was grouped with the tin-opacified samples from Albarracin. However, the glazes from Zaragoza indicated differences in their source of lead, because both samples were closer to the honey glazes from Albarracin, and even the white tin-opacified glaze from Zaragoza (marked with a red circle in
Figure 7a,b) was unexpectedly similar to the transparent honey glazes from Albarracin.
Therefore, all of these glazed ceramics, manufactured in Islamic Albarracin, had lead isotope abundances very well characterized in two groups related to the type of glaze (tin-opacified or transparent) applied on the ceramics. This suggested diverse lead sources for the glaze raw materials depending on the type and quality of glaze; then, it could be related to the use of a type of commercial lead for producing tin-opacified glazes and another different lead product for preparing transparent glazes. The first one could be linked to obtaining a best-quality glaze or to the preparation (fritting) of tin-lead glazes. In later periods (16th century CE), differences in the commercial lead supplied to potters were documented: it was explained that the lead used to produce glazed cooking pots was cheaper than the lead acquired to prepare tin-opacified glazes [
30].
It is important to underline that this possible source or supplier of lead for tin-opacified glazes from Albarracin was quite similar to that for the white glaze from Pechina; as such it could perhaps come from one of the lead sources in the south/southeast of the Iberia Peninsula. However, lead used in Zaragoza workshops seemed to be different, or closer to that introduced in transparent glazes; slight differences could be highlighted between these two samples (honey and white) (
Figure 7a,b), but there are not enough results to find significant dissimilarities.