A Pilot Proteomic Study of Normal Human Tears: Leptin as a Potential Biomarker of Metabolic Disorders
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Clinical Tests
2.2.1. OSDI Questionnaire
2.2.2. FTBUT
2.2.3. CFS
2.2.4. Schirmer 1 Test without Anesthesia
2.2.5. TER
2.2.6. NITBUT
2.3. Tear Samples Collection and Analysis of Metabolic Protein Concentrations
2.4. Statistical Analysis
3. Results
3.1. Demographics and Clinical Features of the Study Population
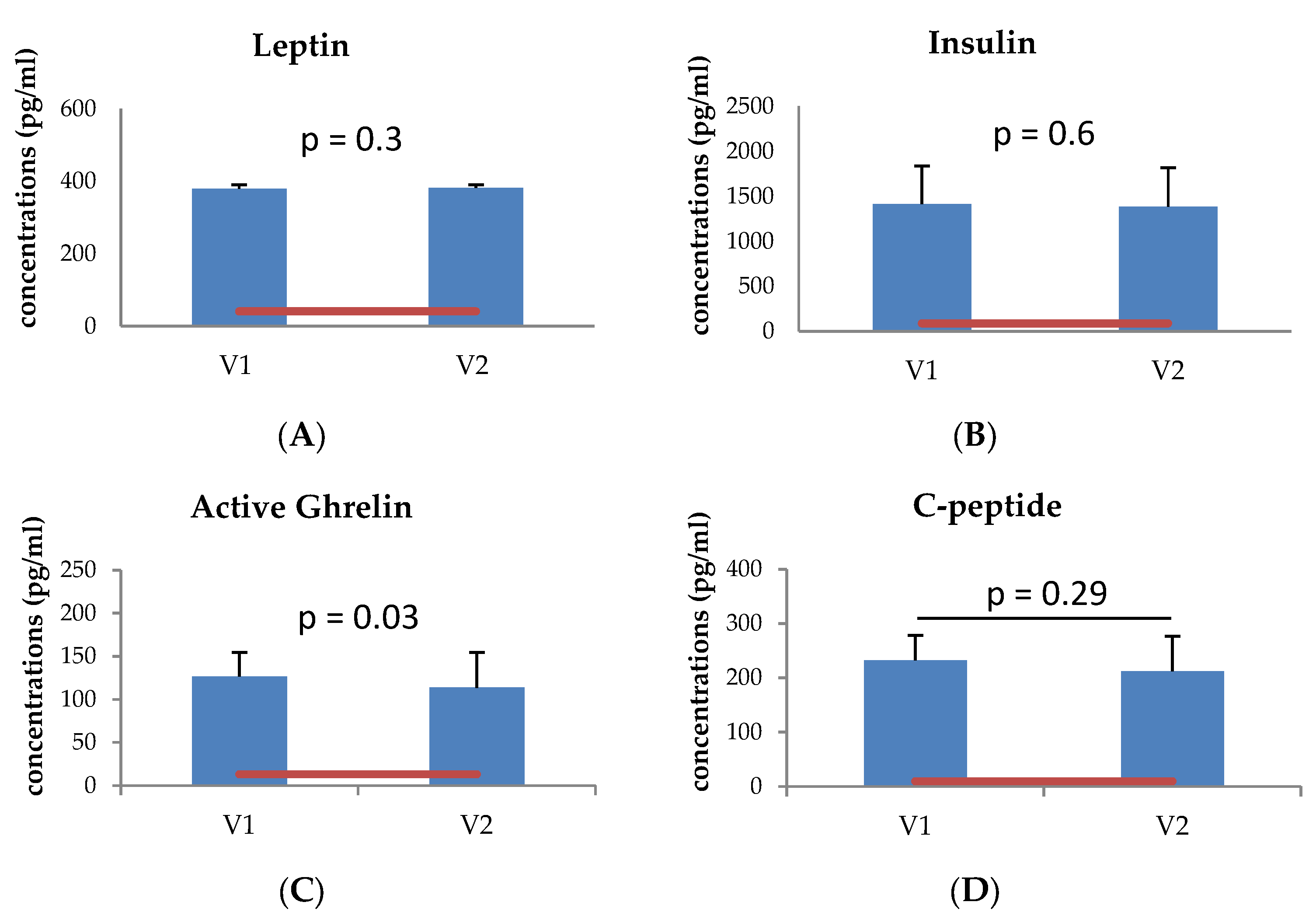
3.2. Metabolic Protein Analysis of the Tear Fluid Samples
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Magni, F.; Van Der Burgt, Y.E.M.; Chinello, C.; Mainini, V.; Gianazza, E.; Squeo, V.; Deelder, A.M.; Kienle, M.G. Biomarkers discovery by peptide and protein profiling in biological fluids based on functionalized magnetic beads purification and mass spectrometry. Blood Transfus. 2010, 8, s92–s97. [Google Scholar]
- Hagan, S.; Tomlinson, A. Tear Fluid Biomarker Profiling: A Review of Multiplex Bead Analysis. Ocul. Surf. 2013, 11, 219–235. [Google Scholar] [CrossRef]
- Hagan, S.; Martin, E.; Enríquez-de-Salamanca, A. Tear fluid biomarkers in ocular and systemic disease: Potential use for predictive, preventive and personalised medicine. EPMA J. 2016, 7, 15. [Google Scholar] [CrossRef]
- Martin, E.; Oliver, K.M.; Pearce, E.I.; Tomlinson, A.; Simmons, P.; Hagan, S. Effect of tear supplements on signs, symptoms and inflammatory markers in dry eye. Cytokine 2018, 105, 37–44. [Google Scholar] [CrossRef]
- Roda, M.; Corazza, I.; Bacchi Reggiani, M.L.; Pellegrini, M.; Taroni, L.; Giannaccare, G.; Versura, P. Dry Eye Disease and Tear Cytokine Levels-A Meta-Analysis. Int. J. Mol. Sci. 2020, 21, 3111. [Google Scholar] [CrossRef]
- Tamhane, M.; Cabrera-Ghayouri, S.; Abelian, G.; Viswanath, V. Review of Biomarkers in Ocular Matrices: Challenges and Opportunities. Pharm. Res. 2019, 36, 1–35. [Google Scholar] [CrossRef]
- Balasubramanian, S.A.; Wasinger, V.C.; Pye, D.C.; Willcox, M.D.P. Preliminary identification of differentially expressed tear proteins in keratoconus. Mol. Vis. 2013, 19, 2124–2134. [Google Scholar] [PubMed]
- Shinde, V.; Hu, N.; Renuse, S.; Mahale, A.; Pandey, A.; Eberhart, C.; Stone, D.; Al-Swailem, S.A.; Maktabi, A.; Chakravarti, S. Mapping Keratoconus Molecular Substrates by Multiplexed High-Resolution Proteomics of Unpooled Corneas. OMICS J. Integr. Biol. 2019, 23, 583–597. [Google Scholar] [CrossRef]
- De Almeida Borges, D.; Alborghetti, M.R.; Franco PaesLeme, A.; Ramos Domingues, R.; Duarte, B.; Veiga, M.; Trindade Ferrer, M.; VianaWanzeler, A.C.; LeiteArieta, C.E.; Alves, M. Tear proteomic profile in three distinct ocular surface diseases: Keratoconus, pterygium, and dry eye related to graft-versus-host disease. Clin. Proteom. 2020, 17, 42. [Google Scholar] [CrossRef] [PubMed]
- Kishazi, E.; Dor, M.; Eperon, S.; Oberic, A.; Hamedani, M.; Turck, N. Thyroid-associated orbitopathy and tears: A proteomics study. J. Proteom. 2018, 170, 110–116. [Google Scholar] [CrossRef] [PubMed]
- Csősz, É.; Deák, E.; Kalló, G.; Csutak, A.; Tőzsér, J. Diabetic retinopathy: Proteomic approaches to help the differential diagnosis and to understand the underlying molecular mechanisms. J. Proteom. 2017, 150, 351–358. [Google Scholar] [CrossRef]
- Winiarczyk, M.; Kaarniranta, K.; Winiarczyk, S.; Adaszek, Ł.; Winiarczyk, D.; Mackiewicz, J. Tear film proteome in age-related macular degeneration. Graefes Arch. Clin. Exp. Ophthalmol. 2018, 256, 1127–1139. [Google Scholar] [CrossRef]
- Jung, J.W.; Han, S.J.; Song, M.K.; Kim, T.; Kim, E.K.; Min, Y.H.; Cheong, J.; Seo, K.Y. Tear Cytokines as Biomarkers for Chronic Graft-versus-Host Disease. Biol. Blood Marrow Transplant. 2015, 21, 2079–2085. [Google Scholar] [CrossRef] [PubMed]
- Cocho, L.; Fernández, I.; Calonge, M.; de la Maza, M.S.; Rovira, M.; Stern, M.E.; Garcia-Vazquez, C.; Enríquez-de-Salamanca, A. Prehematopoietic Stem Cell Transplantation Tear Cytokines as Potential Susceptibility Biomarkers for Ocular Chronic Graft-Versus-Host Disease. Investig. Ophthalmol. Vis. Sci. 2017, 58, 4836–4846. [Google Scholar] [CrossRef]
- O’Leary, O.E.; Schoetzau, A.; Amruthalingam, L.; Geber-Hollbach, N.; Plattner, K.; Jenoe, P.; Schmidt, A.; Ullmer, C.; Drawnel, F.M.; Fauser, S.; et al. Tear Proteomic Predictive Biomarker Model for Ocular Graft Versus Host Disease Classification. Transl. Vis. Sci. Technol. 2020, 9, 3. [Google Scholar] [CrossRef] [PubMed]
- Cicalini, I.; Rossi, C.; Pieragostino, D.; Agnifili, L.; Mastropasqua, L.; di Ioia, M.; De Luca, G.; Onofrj, M.; Federici, L.; Del Boccio, P. Integrated Lipidomics and Metabolomics Analysis of Tears in Multiple Sclerosis: An Insight into Diagnostic Potential of Lacrimal Fluid. Int. J. Mol. Sci. 2019, 20, 1265. [Google Scholar] [CrossRef] [PubMed]
- Sancesario, G.; Bernardini, S. AD biomarker discovery in CSF and in alternative matrices. Clin. Biochem. 2019, 72, 52–57. [Google Scholar] [CrossRef] [PubMed]
- Zetterberg, H.; Compta, Y. A tear fluid proteome of Parkinson’s disease. Parkinsonism Relat. Disord. 2019, 63, 1–2. [Google Scholar] [CrossRef]
- Nättinen, J.; Jylhä, A.; Aapola, U.; Mäkinen, P.; Beuerman, R.; Pietilä, J.; Vaajanen, A.; Uusitalo, H. Age-associated changes in human tear proteome. Clin. Proteom. 2019, 16, 11. [Google Scholar] [CrossRef]
- Lee, S.Y.; Han, S.J.; Nam, S.M.; Yoon, S.C.; Ahn, J.M.; Kim, T.; Kim, E.K.; Seo, K.Y. Analysis of Tear Cytokines and Clinical Correlations in Sjögren Syndrome Dry Eye Patients and Non–Sjögren Syndrome Dry Eye Patients. Am. J. Ophthalmol. 2013, 156, 247–253. [Google Scholar] [CrossRef]
- Baldini, C.; Ferro, F.; Elefante, E.; Bombardieri, S. Biomarkers for Sjögren’s syndrome. Biomark. Med. 2018, 12, 275–286. [Google Scholar] [CrossRef]
- Amil-Bangsa, N.; Mohd-Ali, B.; Ishak, B.; Abdul-Aziz, C.N.; Ngah, N.; Hashim, H.; Ghazali, A. Total Protein Concentration and Tumor Necrosis Factor α in Tears of Nonproliferative Diabetic Retinopathy. Optom. Vis. Sci. 2019, 96, 934–939. [Google Scholar] [CrossRef]
- Yazdani, M.; Elgstøen, K.B.P.; Rootwelt, H.; Shahdadfar, A.; Utheim, Ø.A.; Utheim, T.P. Tear Metabolomics in Dry Eye Disease: A Review. Int. J. Mol. Sci. 2019, 20, 3755. [Google Scholar] [CrossRef]
- Balashova, E.E.; Maslov, D.L.; Lokhov, P.G. A Metabolomics Approach to Pharmacotherapy Personalization. J. Pers. Med. 2018, 8, 28. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.X.; Wang, S.; Kuo, C.; Tsai, I. Metabolome analysis for investigating host-gut microbiota interactions. J. Formos. Med. Assoc. 2019, 118, S10–S22. [Google Scholar] [CrossRef]
- Chen, L.; Gao, Y.; Wang, L.Z.; Cheung, N.; Tan, G.S.W.; Cheung, G.C.M.; Beuerman, R.W.; Wong, T.Y.; Chan, E.C.Y.; Zhou, L. Recent advances in the applications of metabolomics in eye research. Anal. Chim. Acta 2018, 1037, 28–40. [Google Scholar] [CrossRef]
- Pieragostino, D.; Agnifili, L.; Cicalini, I.; Calienno, R.; Zucchelli, M.; Mastropasqua, L.; Sacchetta, P.; Del Boccio, P.; Rossi, C. Tear Film Steroid Profiling in Dry Eye Disease by Liquid Chromatography Tandem Mass Spectrometry. Int. J. Mol. Sci. 2017, 18, 1349. [Google Scholar] [CrossRef]
- Sağlik, A.; Koyuncu, İ.; Soydan, A.; Sağlik, F.; Gönel, A. Tear Organic Acid Analysis After Corneal Collagen Crosslinking in Keratoconus. Eye Contact Lens 2020, 46, S122–S128. [Google Scholar] [CrossRef] [PubMed]
- Byambajav, M.; Collier, A.; Xinhua, S.; Koshy, Z.; Hagan, S. Tear Fluid Inflammatory Cytokines and Metabolic Proteins Levels in Type 2 Diabetes-Related Dry Eye. Investig. Ophthalmol. Vis. Sci. 2020, 61, 39, ARVO meeting abstract. [Google Scholar]
- Rocha, E.M.; Cunha, D.A.; Carneiro, E.M.; Boschero, A.C.; Saad, M.J.A.; Velloso, L.A. Identification of insulin in the tear film and insulin receptor and IGF-1 receptor on the human ocular surface. Investig. Ophthalmol. Vis. Sci. 2002, 43, 963–967, ARVO meeting abstract. [Google Scholar]
- Turgut, B.; Kurt, J.; İlhan, N.; Koca, S.S.; Demir, T.; Çeliker, Ü. Serum and Tear Leptin Levels in Patients with Allergic Conjunctivitis. Turkiye Klinikleri J. Med. Sci. 2010, 30, 233–238. [Google Scholar] [CrossRef][Green Version]
- Hao, R.; Liu, Y.; Li, X. Leptin’s concentration in tears and dry eye: A clinical observational study. Int. J. Ophthalmol. 2021, 14, 83–88. [Google Scholar] [CrossRef] [PubMed]
- Ali, S.; Daniel, J. Drucker Benefits and limitations of reducing glucagon action for the treatment of type 2 diabetes. Am. J. Physiol. Endocrinol. Metab. 2009, 296, 415–421. [Google Scholar] [CrossRef] [PubMed]
- Yi, J.; Warunek, D.; Craft, D. Degradation and Stabilization of Peptide Hormones in Human Blood Specimens. PLoS ONE 2015, 10, e0134427. [Google Scholar] [CrossRef]
- Jones, A.G.; Hattersley, A.T. The clinical utility of C-peptide measurement in the care of patients with diabetes. Diabet. Med. 2013, 30, 803–817. [Google Scholar] [CrossRef]
- Schiffman, R.M.; Christianson, M.D.; Jacobsen, G.; Hirsch, J.D.; Reis, B.L. Reliability and validity of the Ocular Surface Disease Index. Arch. Ophthalmol. 2000, 118, 615–621. [Google Scholar] [CrossRef]
- Grubbs, J.; Tolleson-Rinehart, S.; Huynh, K.; Davis, R. A Review of Quality of Life Measures in Dry Eye Questionnaires. Cornea 2014, 33, 215–218. [Google Scholar] [CrossRef]
- Efron, N. Grading scales for contact lens complications. Ophthalmic Physiol. Opt. J. Br. Coll. Ophthalmic Opt. 1998, 18, 182–186. [Google Scholar] [CrossRef]
- Bron, A.J.; Smith, J.A.; Calonge, M. Methodologies to Diagnose and Monitor Dry Eye Disease: Report of the Diagnostic Methodology Subcommittee of the International Dry Eye WorkShop. Ocul. Surf. 2007, 5, 108–152. [Google Scholar]
- Enríquez-de-Salamanca, A.; Castellanos, E.; Stern, M.E.; Fernández, I.; Carreño, E.; García-Vázquez, C.; Herreras, J.M.; Calonge, M. Tear cytokine and chemokine analysis and clinical correlations in evaporative-type dry eye disease. Mol. Vis. 2010, 16, 862–873. [Google Scholar]
- González-García, M.J.; Murillo, G.M.; Pinto-Fraga, J.; García, N.; Fernández, I.; Maldonado, M.J.; Calonge, M.; Enríquez-de-Salamanca, A. Clinical and tear cytokine profiles after advanced surface ablation refractive surgery: A six-month follow-up. Exp. Eye Res. 2020, 193, 107976. [Google Scholar] [CrossRef]
- Pinto-Fraga, J.; Enríquez-de-Salamanca, A.; Calonge, M.; González-García, M.J.; López-Miguel, A.; López-de la Rosa, A.; García-Vázquez, C.; Calder, V.; Stern, M.E.; Fernández, I. Severity, therapeutic, and activity tear biomarkers in dry eye disease: An analysis from a phase III clinical trial. Ocul. Surf. 2018, 16, 368–376. [Google Scholar] [CrossRef]
- Helsel, D.R.; Cohn, T.A. Estimation of descriptive statistics for multiply censored water quality data. Water Resour. Res. 1988, 24, 1997–2004. [Google Scholar] [CrossRef]
- Lopatka, M.; Barcaru, A.; Sjerps, M.J.; Vivó-Truyols, G. Leveraging probabilistic peak detection to estimate baseline drift in complex chromatographic samples. J. Chromatogr. A 2016, 1431, 122–130. [Google Scholar] [CrossRef] [PubMed]
- Portney, L.G.; Watkins, M.P. Validity of Measurements. In Foundations of Clinical Research: Applications to Practice, 2nd ed.; Portney, L.G., Watkins, M.P., Eds.; Prentice Hall Health: Hoboken, NJ, USA, 2000; pp. 79–112. [Google Scholar]
- Chan, Y.H. Biostatistics 104: Correlational analysis. Singap. Med. J. 2003, 44, 614–619. [Google Scholar]
- Sato, T.; Nakamura, Y.; Shiimura, Y.; Ohgusu, H.; Kangawa, K.; Kojima, M. Structure, regulation and function of ghrelin. J. Biochem. 2012, 151, 119–128. [Google Scholar] [CrossRef]
- Giezenaar, C.; Hutchison, A.; Luscombe-Marsh, N.; Chapman, I.; Horowitz, M.; Soenen, S. Effect of Age on Blood Glucose and Plasma Insulin, Glucagon, Ghrelin, CCK, GIP, and GLP-1 Responses to Whey Protein Ingestion. Nutrients 2017, 10, 2. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zou, X.; Cai, X.; Liu, W.; Chen, L.; Zhang, R.; Zhou, L.; Gong, S.; Gao, Y.; Li, S.; et al. Urinary C-peptide/Creatinine Ratio: A Useful Biomarker of Insulin Resistance and Refined Classification of Type 2 Diabetes Mellitus. J. Diabetes 2021. [Google Scholar] [CrossRef] [PubMed]
- Landgraf, W.; Owens, D.R.; Frier, B.M.; Zhang, M.; Bolli, G.B. Fasting C-peptide, a biomarker for hypoglycaemia risk in insulin-naïve people with type 2 diabetes initiating basal insulin glargine 100 U./mL. Diabetes Obes. Metab. 2020, 22, 315–323. [Google Scholar] [CrossRef]
- Wang, Y.; Wan, H.; Chen, Y.; Xia, F.; Zhang, W.; Wang, C.; Fang, S.; Zhang, K.; Li, Q.; Wang, N.; et al. Association of C-peptide with diabetic vascular complications in type 2 diabetes. Diabetes Metab. 2020, 46, 33–40. [Google Scholar] [CrossRef]
- Dysinger, M.; Marusov, G.; Fraser, S. Quantitative analysis of four protein biomarkers: An automated microfluidic cartridge-based method and its comparison to colorimetric ELISA. J. Immunol. Methods 2017, 451, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Carreño, E.; Enríquez-de-Salamanca, A.; Tesón, M.; García-Vázquez, C.; Stern, M.E.; Whitcup, S.M.; Calonge, M. Cytokine and chemokine levels in tears from healthy subjects. Acta Ophthalmol. 2010, 88, e250–e258. [Google Scholar] [CrossRef] [PubMed]
- Tesón, M.; González-García, M.J.; López-Miguel, A.; Enríquez-de-Salamanca, A.; Martín-Montañez, V.; Benito, M.J.; Mateo, M.E.; Stern, M.E.; Calonge, M. Influence of a Controlled Environment Simulating an In-Flight Airplane Cabin on Dry Eye Disease. Investig. Ophthalmol. Vis. Sci. 2013, 54, 2093–2099. [Google Scholar] [CrossRef] [PubMed]


| N | Age | Sex | OSDI | fTBUT | Schirmer | CFS | Test Eye | |||
|---|---|---|---|---|---|---|---|---|---|---|
| OD | OS | OD | OS | OD | OS | |||||
| 1 | 38 | M | 15.9 | 6.93 | 16.67 | 26 | 22 | 1 | 2 | OS |
| 2 | 26 | M | 9.09 | 15 | 16.34 | 32 | 17 | 3 | 2 | OS |
| 3 | 27 | F | 0 | 6.02 | 10.25 | 30 | 30 | 4 | 0 | OD |
| 4 | 33 | M | 4.16 | 4.6 | 5.44 | 15 | 6 | 2 | 4 | OD |
| 5 | 21 | F | 2.08 | 11.06 | 11.23 | 33 | 31 | 0 | 0 | OD |
| 6 | 26 | F | 2.5 | 11.63 | 11.37 | 35 | 35 | 0 | 0 | OD |
| 7 | 25 | F | 0 | 5.26 | 7.94 | 28 | 25 | 0 | 0 | OD |
| 8 | 18 | F | 0 | 14.22 | 13.77 | 16 | 15 | 0 | 0 | OS |
| 9 | 19 | F | 0 | 6.1 | 15.44 | 21 | 12 | 0 | 0 | OD |
| 10 | 32 | M | 2.27 | 13.47 | 16.94 | 28 | 26 | 0 | 0 | OD |
| 11 | 25 | M | 0 | 8.08 | 7.99 | 35 | 35 | 1 | 0 | OD |
| 12 | 38 | M | 10 | 11.09 | 13.53 | 35 | 35 | 0 | 0 | OS |
| 13 | 18 | F | 9.09 | 8.17 | 14.9 | 15 | 11 | 0 | 1 | OS |
| 14 | 18 | F | 9.09 | 7.06 | 10.53 | 35 | 29 | 0 | 0 | OS |
| 15 | 21 | F | 2.08 | 9.6 | 12.17 | 10 | 8 | 0 | 0 | OS |
| 16 | 21 | F | 6.25 | 12 | 13.67 | 35 | 35 | 0 | 0 | OS |
| 17 | 21 | M | 4.16 | 6.67 | 13.33 | 35 | 35 | 0 | 0 | OS |
| Biomarker | Visit | Number Detected out of 17 Samples and Percentage Detection (%) | 95% CI | |
|---|---|---|---|---|
| Inf. | Sup. | |||
| Leptin | V1 | 17 (100%) | 77.8 | 100 |
| V2 | 17 (100%) | 77.8 | 100 | |
| Insulin | V1 | 17 (100%) | 77.8 | 100 |
| V2 | 17 (100%) | 77.8 | 100 | |
| Active Ghrelin | V1 | 17 (100%) | 77.8 | 100 |
| V2 | 17 (100%) | 77.8 | 100 | |
| C-peptide | V1 | 17 (100%) | 77.8 | 100 |
| V2 | 17 (100%) | 77.8 | 100 | |
| GIP | V1 | 12 (70.6%) | 44 | 88.6 |
| V2 | 9 (52.6%) | 28.5 | 76.1 | |
| Biomarker | N | CV (%) | 95% CI | CVm (%) | |
|---|---|---|---|---|---|
| Inf. | Sup. | ||||
| Leptin | 17 | 1.09 | 0.63 | 1.55 | <15 |
| Insulin | 17 | 25.62 | 13.16 | 38.06 | <15 |
| Active Ghrelin | 17 | 3.51 | 1.67 | 5.34 | <15 |
| C-peptide | 17 | 1.71 | 0.87 | 2.55 | <15 |
| GIP | 17 | 60.92 | 39.01 | 82.82 | <15 |
| Biomarker | ICC | Reproducibility Rating |
|---|---|---|
| Leptin | 0.70 | strong |
| Insulin | 0.15 | poor |
| Active Ghrelin | 0.24 | poor |
| C-peptide | 0.43 | low |
| GIP | 0.29 | low |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Byambajav, M.; Arroyo-del Arroyo, C.; Enríquez-de-Salamanca, A.; Fernández, I.; Martin, E.; Hagan, S. A Pilot Proteomic Study of Normal Human Tears: Leptin as a Potential Biomarker of Metabolic Disorders. Appl. Sci. 2021, 11, 5755. https://doi.org/10.3390/app11125755
Byambajav M, Arroyo-del Arroyo C, Enríquez-de-Salamanca A, Fernández I, Martin E, Hagan S. A Pilot Proteomic Study of Normal Human Tears: Leptin as a Potential Biomarker of Metabolic Disorders. Applied Sciences. 2021; 11(12):5755. https://doi.org/10.3390/app11125755
Chicago/Turabian StyleByambajav, Mungunshur, Cristina Arroyo-del Arroyo, Amalia Enríquez-de-Salamanca, Itziar Fernández, Eilidh Martin, and Suzanne Hagan. 2021. "A Pilot Proteomic Study of Normal Human Tears: Leptin as a Potential Biomarker of Metabolic Disorders" Applied Sciences 11, no. 12: 5755. https://doi.org/10.3390/app11125755
APA StyleByambajav, M., Arroyo-del Arroyo, C., Enríquez-de-Salamanca, A., Fernández, I., Martin, E., & Hagan, S. (2021). A Pilot Proteomic Study of Normal Human Tears: Leptin as a Potential Biomarker of Metabolic Disorders. Applied Sciences, 11(12), 5755. https://doi.org/10.3390/app11125755







