Evaluation of ACL Graft Remodeling and Prediction of Graft Insufficiency in Sequenced MRI—Two-Year Follow-Up
Abstract
Application Feature
Abstract
1. Introduction
2. Materials and Methods
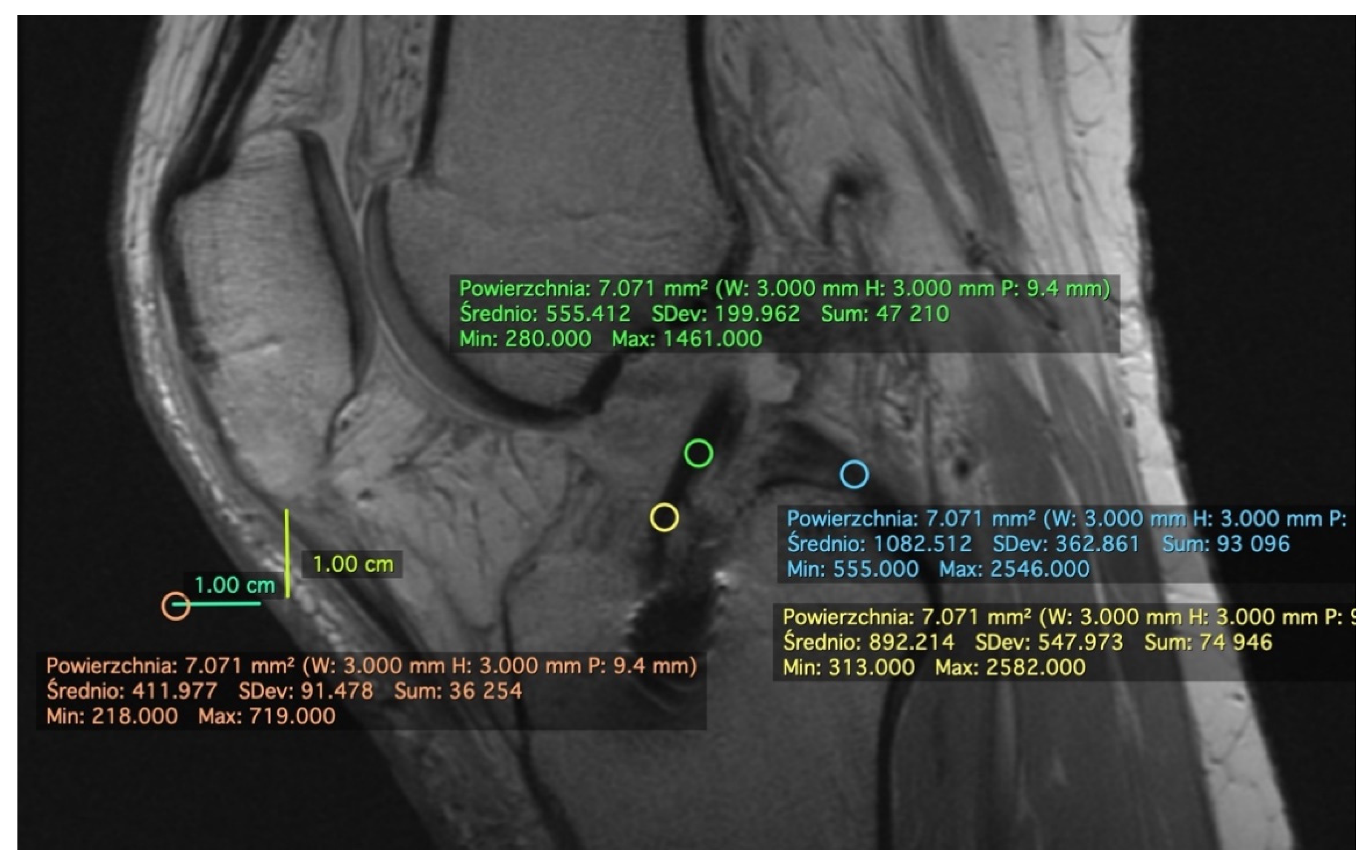
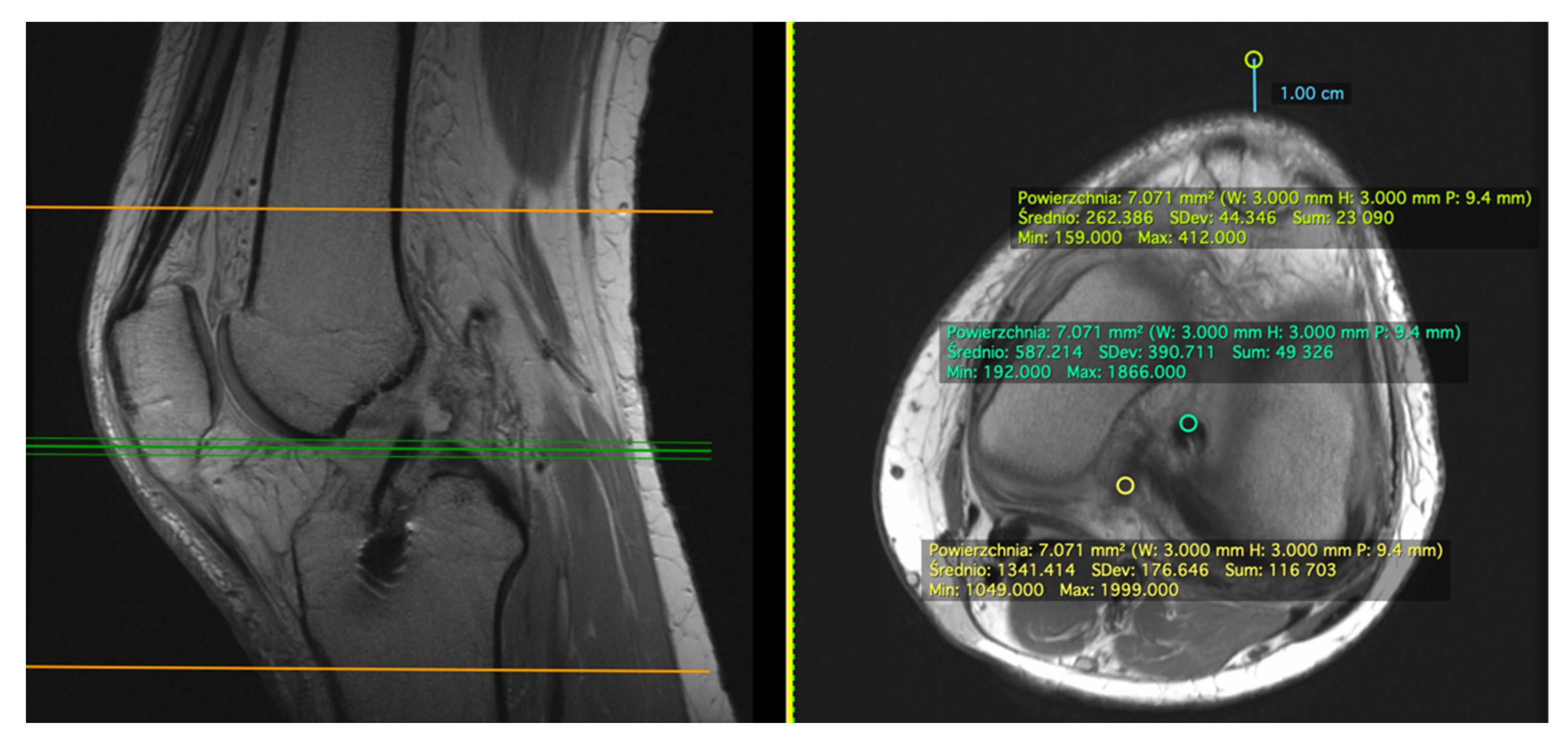
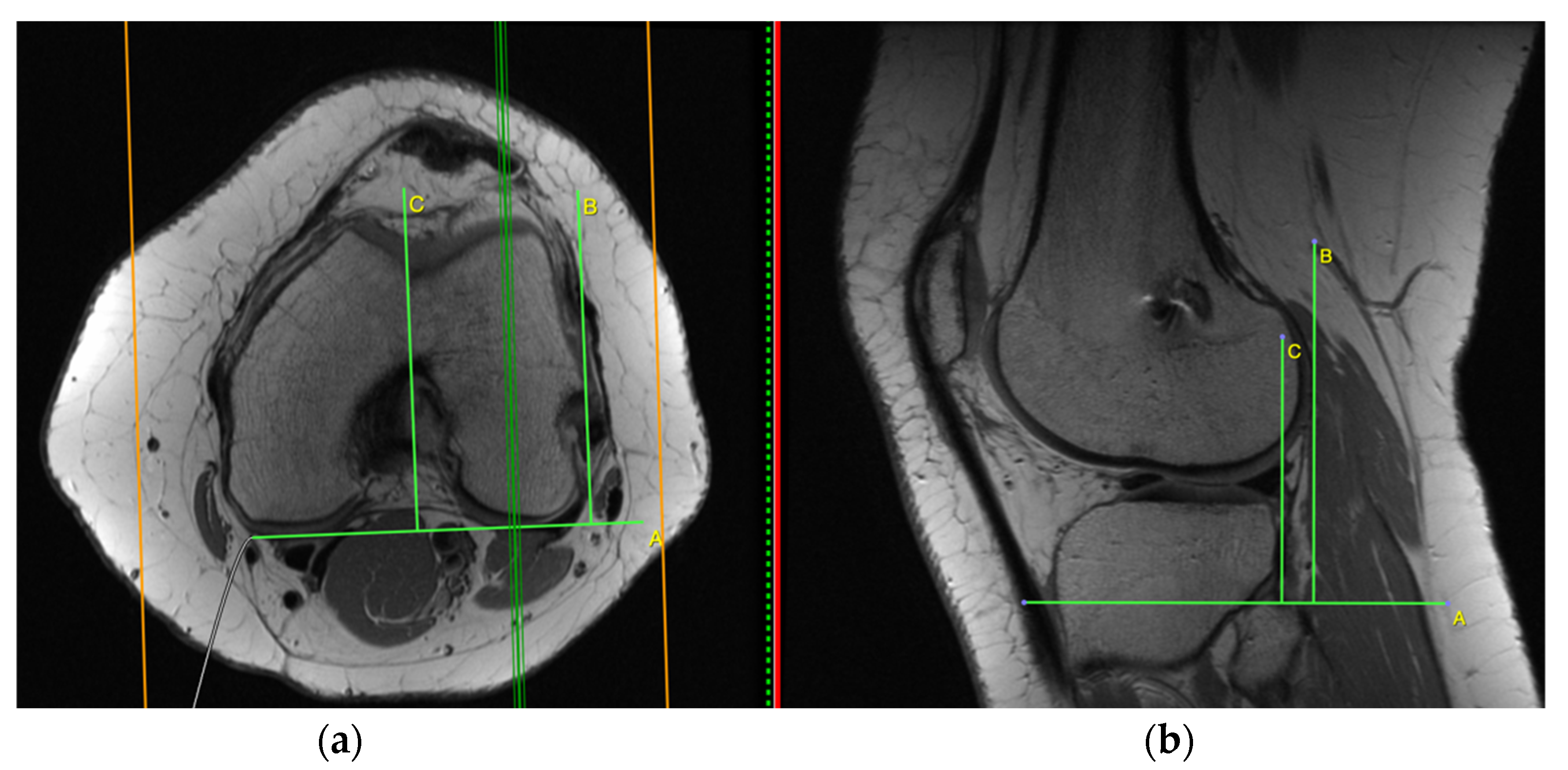
2.1. Measurement on the Sagittal Plane
2.2. Measurement on the Axial Plane
2.3. Standard Deviation (SD)
2.4. Indirect Signs of Anterior Cruciate Ligament (ACL) Graft Insufficiency
2.5. Data Analysis
Statistical Analyses
3. Results
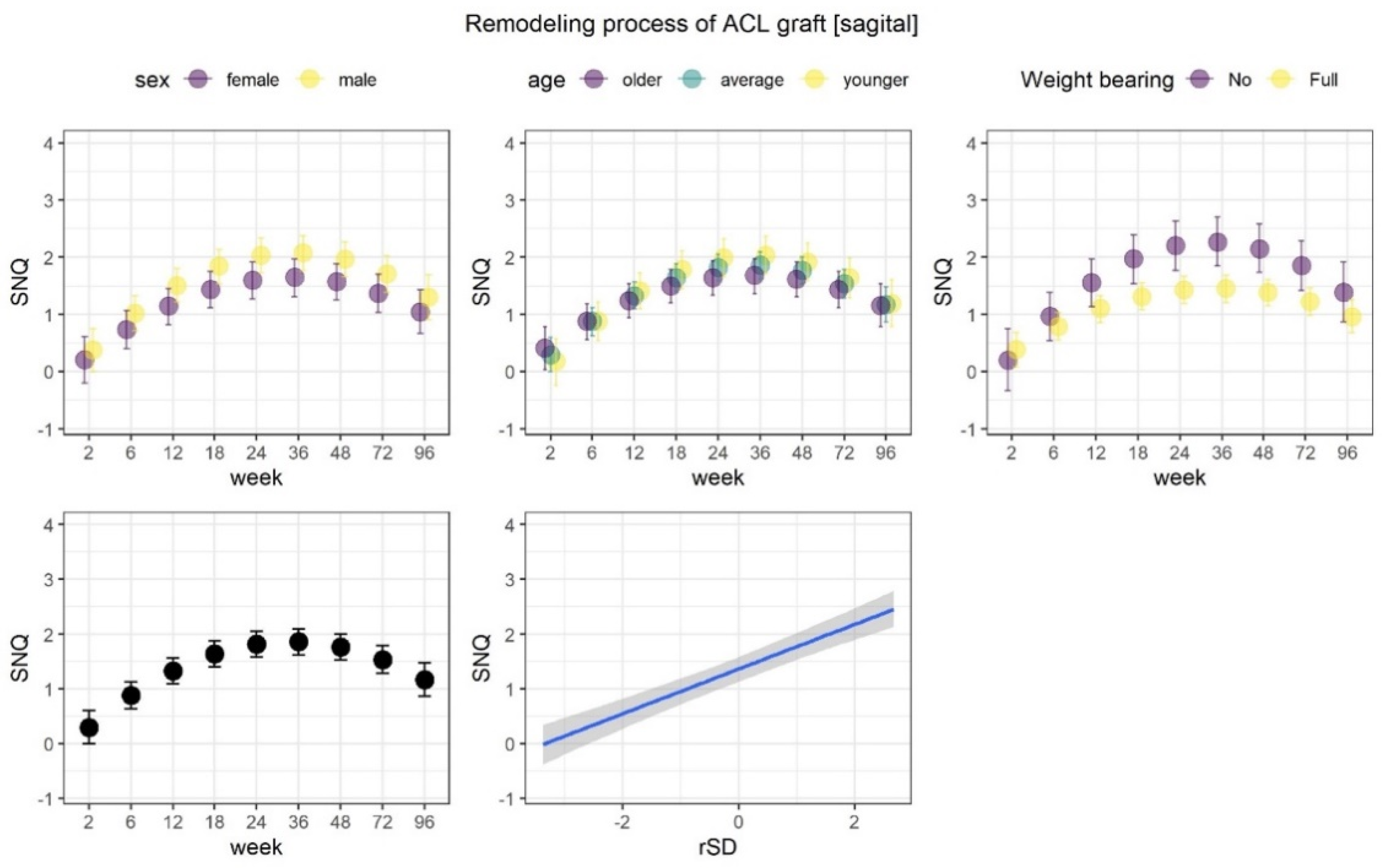
3.1. Remodeling Process of the ACL Graft
3.2. Indirect Signs of ACL Graft Insufficiency
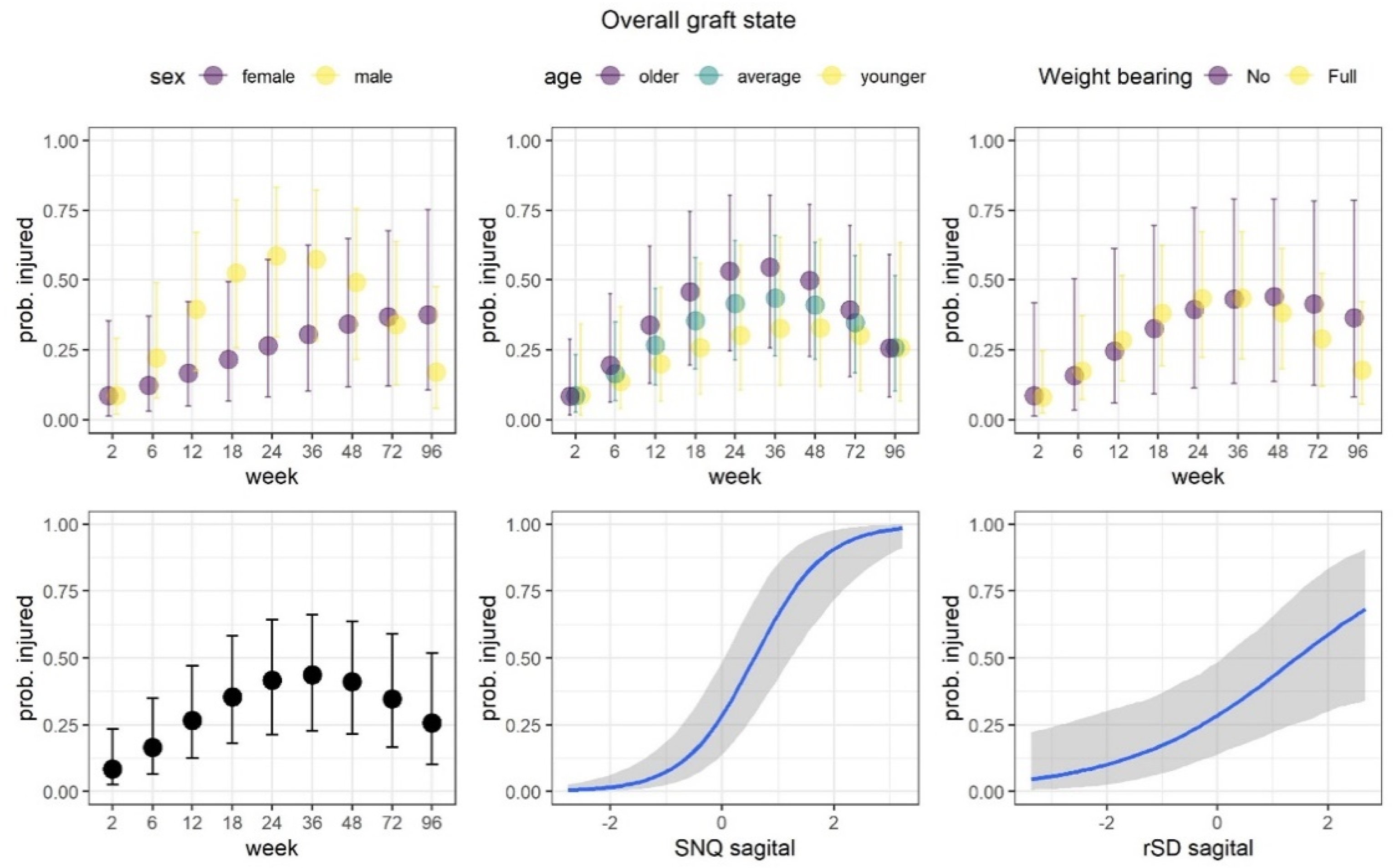
3.2.1. Overall Graft State
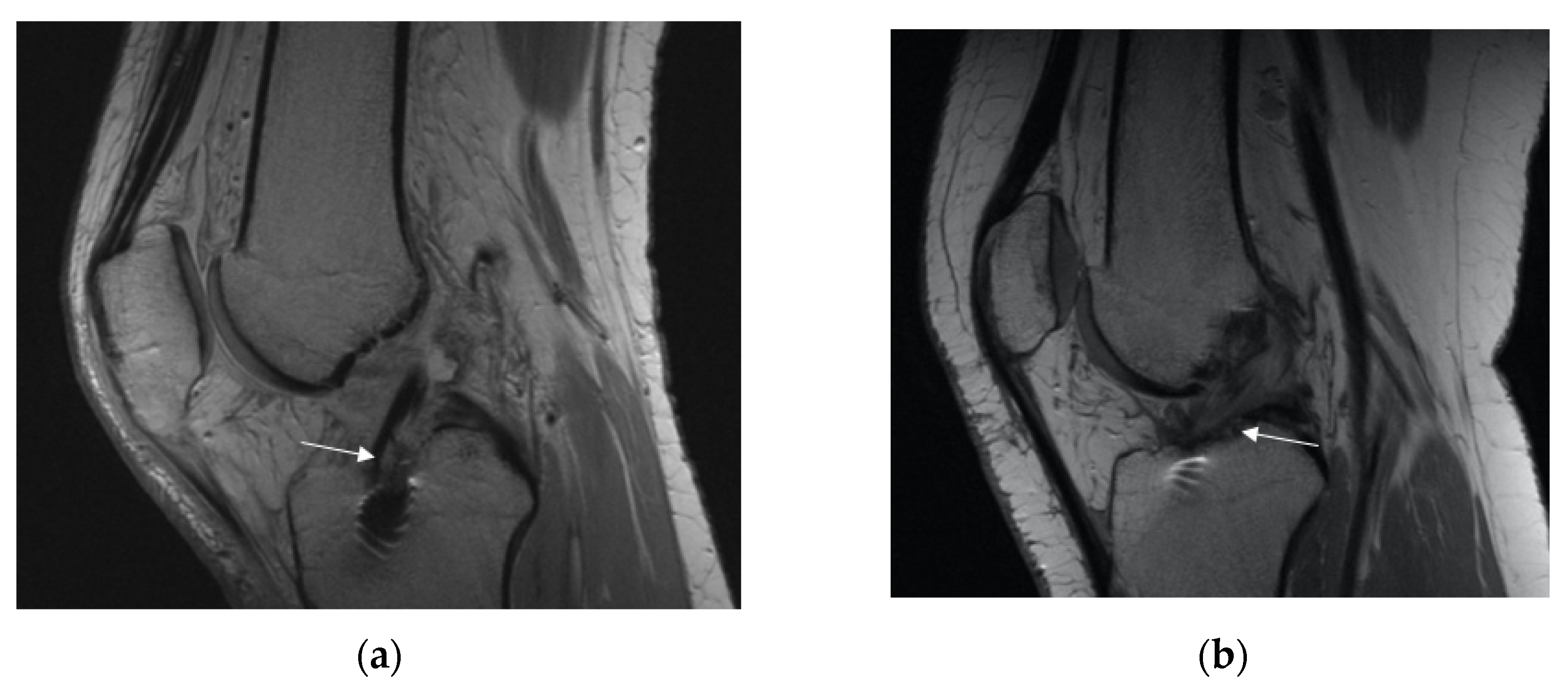
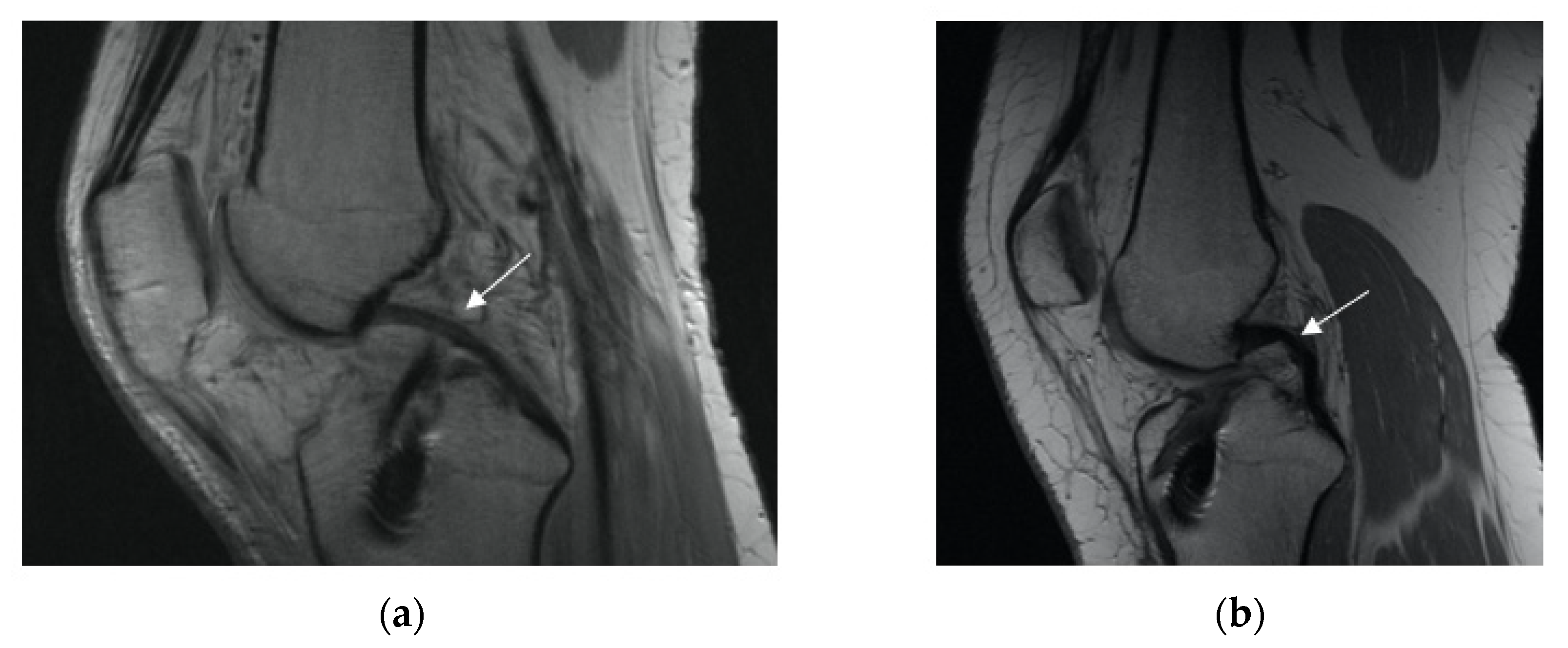
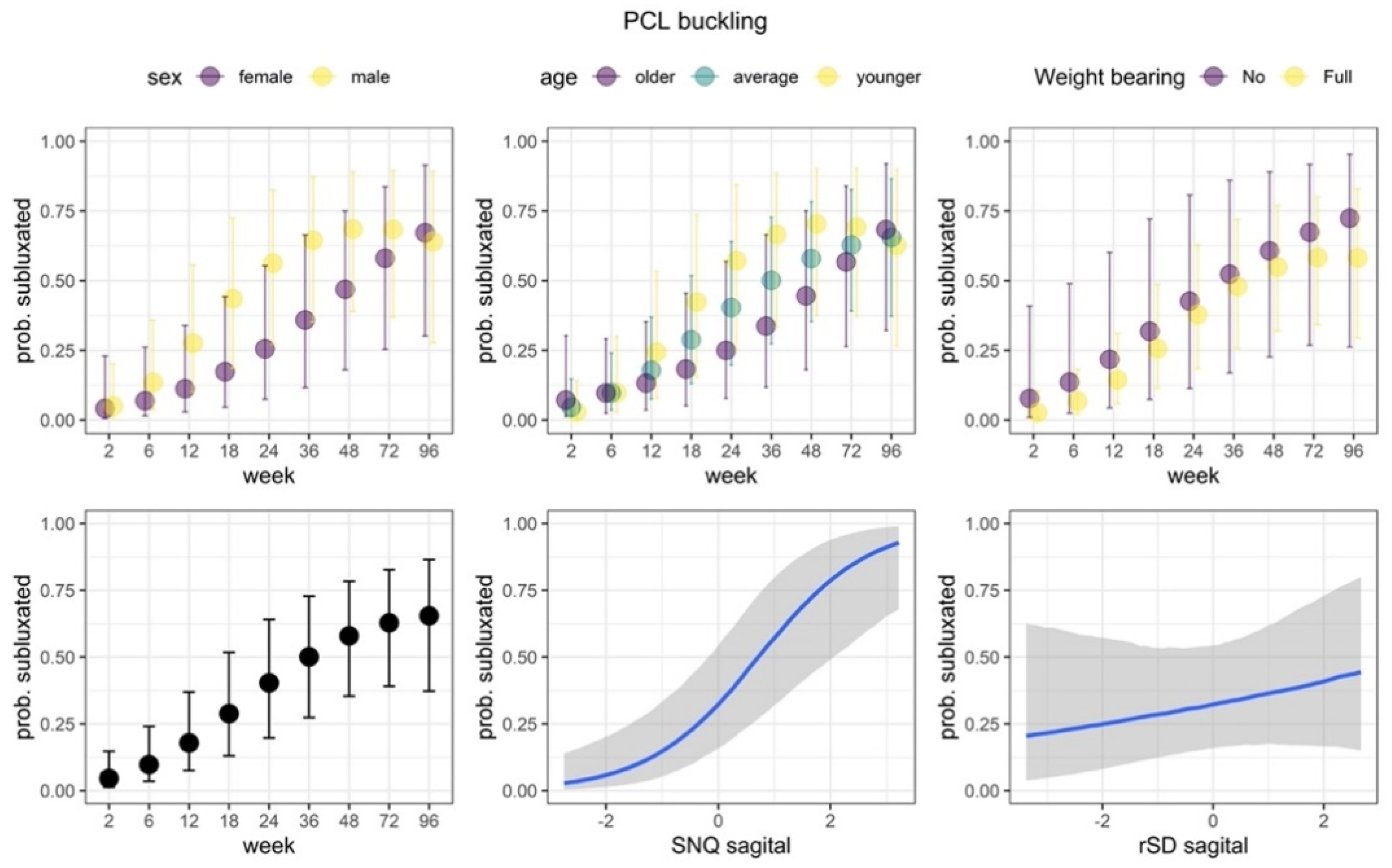
3.2.2. Posterior Cruciate Ligament (PCL) Buckling (Subluxation)
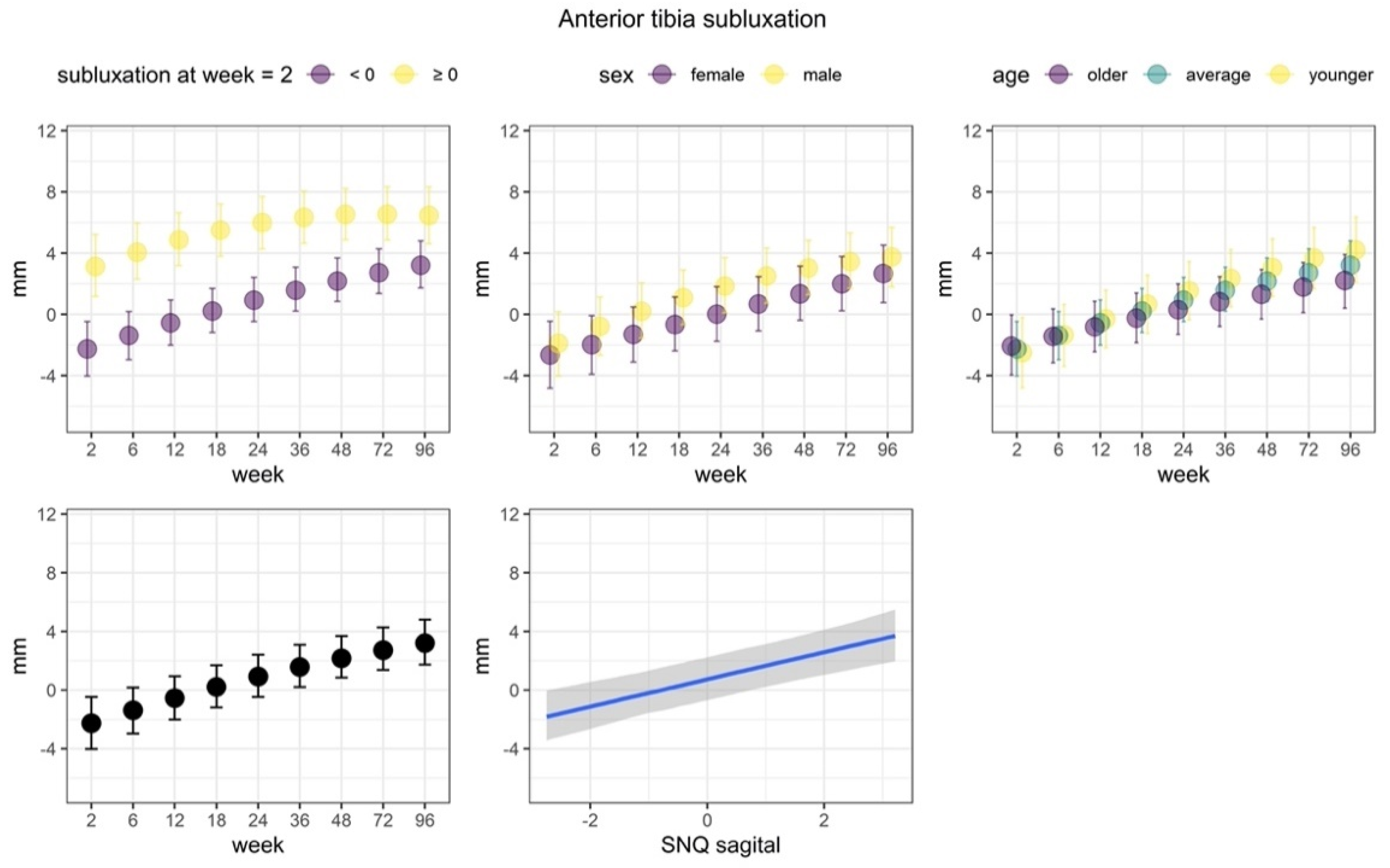
3.2.3. Anterior Tibia Subluxation
4. Discussion
- −
- Psychological readiness for sport;
- −
- No pain;
- −
- Clinically stable knee (Lachman test, Rollimetr/KT 1000);
- −
- Minimum ROM 0–130;
- −
- MRI—good signal of the graft;
- −
- Biomechanical evaluation: symmetrical muscle strength (on the level of pelvis and knee), good proprioception, movement analysis of simple jump and landing (always paying attention to pelvic stability and valgus knee position). If it is not possible to do an evaluation in the professional lab, a simple, functional test and video analysis based on a cell-phone would also work.
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lohmander, L.S.; Englund, P.M.; Dahl, L.L.; Roos, E.M. The Long-Term Consequence of Anterior Cruciate Ligament and Meniscus Injuries: Osteoarthritis. Am. J. Sports Med. 2007, 35, 1756–1769. [Google Scholar] [CrossRef]
- Logan, M.C.; Williams, A.; Lavelle, J.; Gedroyc, W.; Freeman, M. Tibiofemoral Kinematics Following Successful Anterior Cruciate Ligament Reconstruction Using Dynamic Multiple Resonance Imaging. Am. J. Sports Med. 2004, 32, 984–992. [Google Scholar] [CrossRef]
- Condello, V.; Zdanowicz, U.; Di Matteo, B.; Spalding, T.; Gelber, P.E.; Adravanti, P.; Heuberer, P.; Dimmen, S.; Sonnery-Cottet, B.; Hulet, C.; et al. Allograft Tendons Are a Safe and Effective Option for Revision ACL Reconstruction: A Clinical Review. Knee Surg. Sports Traumatol. Arthrosc. 2019, 27, 1771–1781. [Google Scholar] [CrossRef]
- Dong, S.; Xie, G.; Zhang, Y.; Shen, P.; Huangfu, X.; Zhao, J. Ligamentization of Autogenous Hamstring Grafts After Anterior Cruciate Ligament Reconstruction: Midterm Versus Long-Term Results. Am. J. Sports Med. 2015, 43, 1908–1917. [Google Scholar] [CrossRef]
- Claes, S.; Verdonk, P.; Forsyth, R.; Bellemans, J. The “Ligamentization” Process in Anterior Cruciate Ligament Reconstruction: What Happens to the Human Graft? A Systematic Review of the Literature. Am. J. Sports Med. 2011, 39, 2476–2483. [Google Scholar] [CrossRef]
- Biercevicz, A.M.; Miranda, D.L.; Machan, J.T.; Murray, M.M.; Fleming, B.C. In Situ, Noninvasive, T2*-Weighted MRI-Derived Parameters Predict Ex Vivo Structural Properties of an Anterior Cruciate Ligament Reconstruction or Bioenhanced Primary Repair in a Porcine Model. Am. J. Sports Med. 2013, 41, 560–566. [Google Scholar] [CrossRef]
- Biercevicz, A.M.; Akelman, M.R.; Fadale, P.D.; Hulstyn, M.J.; Shalvoy, R.M.; Badger, G.J.; Tung, G.A.; Oksendahl, H.L.; Fleming, B.C. MRI Volume and Signal Intensity of ACL Graft Predict Clinical, Functional, and Patient-Oriented Outcome Measures After ACL Reconstruction. Am. J. Sports Med. 2015, 43, 693–699. [Google Scholar] [CrossRef]
- Weiler, A.; Peters, G.; Mäurer, J.; Unterhauser, F.N.; Südkamp, N.P. Biomechanical Properties and Vascularity of an Anterior Cruciate Ligament Graft Can Be Predicted by Contrast-Enhanced Magnetic Resonance Imaging: A Two-Year Study in Sheep. Am. J. Sports Med. 2001, 29, 751–761. [Google Scholar] [CrossRef]
- Howell, S.M.; Clark, J.A.; Blasier, R.D. Serial Magnetic Resonance Imaging of Hamstring Anterior Cruciate Ligament Autografts during the First Year of Implantation: A Preliminary Study. Am. J. Sports Med. 1991, 19, 42–47. [Google Scholar] [CrossRef]
- Ahn, J.H.; Lee, S.H.; Choi, S.H.; Lim, T.K. Magnetic Resonance Imaging Evaluation of Anterior Cruciate Ligament Reconstruction Using Quadrupled Hamstring Tendon Autografts: Comparison of Remnant Bundle Preservation and Standard Technique. Am. J. Sports Med. 2010, 38, 1768–1777. [Google Scholar] [CrossRef]
- Vogl, T.J.; Schmitt, J.; Lubrich, J.; Hochmuth, K.; Diebold, T.; Del Tredici, K.; Südkamp, N. Reconstructed Anterior Cruciate Ligaments Using Patellar Tendon Ligament Grafts: Diagnostic Value of Contrast-Enhanced MRI in a 2-Year Follow-up Regimen. Eur. Radiol. 2001, 11, 1450–1456. [Google Scholar] [CrossRef] [PubMed]
- Pilson, D.; Decker, K.L. Compensation for Herbivory in Wild Sunflower: Response to Simulated Damage by the Head-Clipping Weevil. Ecology 2002, 83, 3097–3107. [Google Scholar] [CrossRef]
- Kruschke, J.K. Doing Bayesian Data Analysis: A Tutorial with R, JAGS, and Stan; Elsevier: Amsterdam, The Netherlands, 2015; ISBN 978-0-12-405916-0. [Google Scholar]
- Gelman, A.; Goodrich, B.; Gabry, J.; Vehtari, A. R-Squared for Bayesian Regression Models. Am. Stat. 2019, 73, 307–309. [Google Scholar] [CrossRef]
- Bürkner, P.-C. Brms: An R Package for Bayesian Multilevel Models Using. Stan. J. Stat. Soft. 2017, 80. [Google Scholar] [CrossRef]
- Van Dyck, P.; Zazulia, K.; Smekens, C.; Heusdens, C.H.W.; Janssens, T.; Sijbers, J. Assessment of Anterior Cruciate Ligament Graft Maturity With Conventional Magnetic Resonance Imaging: A Systematic Literature Review. Orthop. J. Sports Med. 2019, 7, 232596711984901. [Google Scholar] [CrossRef]
- Van Groningen, B.; van der Steen, M.C.; Janssen, D.M.; van Rhijn, L.W.; van der Linden, A.N.; Janssen, R.P.A. Assessment of Graft Maturity After Anterior Cruciate Ligament Reconstruction Using Autografts: A Systematic Review of Biopsy and Magnetic Resonance Imaging Studies. Arthrosc. Sports Med. Rehabil. 2020, 2, e377–e388. [Google Scholar] [CrossRef]
- Fukuda, H.; Asai, S.; Kanisawa, I.; Takahashi, T.; Ogura, T.; Sakai, H.; Takahashi, K.; Tsuchiya, A. Inferior Graft Maturity in the PL Bundle after Autograft Hamstring Double-Bundle ACL Reconstruction. Knee Surg. Sports Traumatol. Arthrosc. 2019, 27, 491–497. [Google Scholar] [CrossRef]
- Zaffagnini, S.; De Pasquale, V.; Marchesini Reggiani, L.; Russo, A.; Agati, P.; Bacchelli, B.; Marcacci, M. Electron Microscopy of the Remodelling Process in Hamstring Tendon Used as ACL Graft. Knee Surg. Sports Traumatol. Arthrosc. 2010, 18, 1052–1058. [Google Scholar] [CrossRef]
- Dziki, J.L.; Giglio, R.M.; Sicari, B.M.; Wang, D.S.; Gandhi, R.M.; Londono, R.; Dearth, C.L.; Badylak, S.F. The Effect of Mechanical Loading Upon Extracellular Matrix Bioscaffold-Mediated Skeletal Muscle Remodeling. Tissue Eng. Part A 2018, 24, 34–46. [Google Scholar] [CrossRef]
- van der List, J.P.; Mintz, D.N.; DiFelice, G.S. Postoperative Magnetic Resonance Imaging Following Arthroscopic Primary Anterior Cruciate Ligament Repair. Adv. Orthop. 2019, 2019, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Zaffagnini, S. Return to Sport after ACL Reconstruction: How, When and Why? A Narrative Review of Current Evidence. Joints 2015. [Google Scholar] [CrossRef]
- SIGASCOT Sports Committee; Grassi, A.; Vascellari, A.; Combi, A.; Tomaello, L.; Canata, G.L.; Zaffagnini, S. Return to Sport after ACL Reconstruction: A Survey between the Italian Society of Knee, Arthroscopy, Sport, Cartilage and Orthopaedic Technologies (SIGASCOT) Members. Eur. J. Orthop. Surg. Traumatol. 2016, 26, 509–516. [Google Scholar] [CrossRef]
- Nagelli, C.V.; Hewett, T.E. Should Return to Sport Be Delayed Until 2 Years After Anterior Cruciate Ligament Reconstruction? Biological and Functional Considerations. Sports Med. 2017, 47, 221–232. [Google Scholar] [CrossRef]
- Culvenor, A.G.; Patterson, B.E.; Guermazi, A.; Morris, H.G.; Whitehead, T.S.; Crossley, K.M. Accelerated Return to Sport After Anterior Cruciate Ligament Reconstruction and Early Knee Osteoarthritis Features at 1 Year: An Exploratory Study. PM&R 2018, 10, 349–356. [Google Scholar] [CrossRef]
| Time Post-Surgery | Rehabilitation Protocol | |
|---|---|---|
| GROUP 1—Full Weight Bearing Allowed Immediately after the Surgery | GROUP 2—No Weight Bearing after the Surgery for 3 Weeks | |
| Days 1–7 | Pain and swelling control | |
| Care for the dressing | ||
| Early range of motion exercises | ||
| Manual therapy | ||
| Quadriceps femoris muscle (QF) activation + hamstrings cocontraction exercises | ||
| Full weight bearing as tolerated | No weight bearing for 3 weeks | |
| Weeks 2–6 | Gradual increase of range of motion (ROM) 0–100 | |
| Hamstrings cocontraction exercises | ||
| Close kinetic chain QF exercises | ||
| Simple prioprioception exercises | ||
| Pelvic stabilization exercises | ||
| Scars mobilisation | ||
| Weeks 6–12 | Progression of above mentioned exercises | |
| Months 3–6 | Gradual implementation of open kinetic chain exercises | |
| Gradual implementation of dynamic exercises | ||
| Months 6–12 | Targeted exercices to eliminated any side-to-side differences in muscle strength | |
| Introducing a bike | ||
| Prioprioception exercises | ||
| Introducing jogging | ||
| Regaining proper biomechanics | ||
| ax PDWI | sag PDWI | |
|---|---|---|
| TR (ms) | 2800 | 2800 |
| TE (ms) | 35 | 35 |
| Slice thickness (mm) | 2 | 2 |
| Intersection gap (mm) | 2 | 2 |
| FOV (cm) | 16 × 16 | 16 × 16 |
| Matrix | 512 × 256 | 512 × 256 |
| Flip angle | 90 | 90 |
| Parameter | Remodeling Process of ACL Graft | ||||||
|---|---|---|---|---|---|---|---|
| SNQ at Sagittal Plane | SNQ at Axial Plane | ||||||
| Me | LI | UI | Me | LI | UI | ||
| β | Intercept | 0.07 | −0.11 | 0.25 | 0 | −0.18 | 0.19 |
| Week [L] | 0.68 | 0.46 | 0.88 | 0.96 | 0.73 | 1.18 | |
| Week [Q] | −0.95 | −1.15 | −0.73 | −0.67 | −0.9 | −0.43 | |
| rSD | 0.33 | 0.26 | 0.4 | 0.28 | 0.2 | 0.35 | |
| Sex | −0.14 | −0.29 | 0.01 | −0.13 | −0.28 | 0.03 | |
| Age | −0.07 | −0.22 | 0.09 | −0.05 | −0.22 | 0.1 | |
| Weight bearing | 0.2 | 0.02 | 0.37 | 0.05 | −0.13 | 0.23 | |
| Week [L] : Sex | −0.03 | −0.2 | 0.15 | 0.1 | −0.08 | 0.29 | |
| Week [Q] : Sex | 0.09 | −0.08 | 0.26 | 0.11 | −0.07 | 0.31 | |
| Week [L] : Age | −0.1 | −0.27 | 0.06 | −0.19 | −0.37 | −0.01 | |
| Week [Q] : Age | 0.2 | 0.01 | 0.37 | 0.27 | 0.07 | 0.47 | |
| Week [L] : Weight bearing | 0.24 | 0.02 | 0.45 | 0.26 | 0.05 | 0.47 | |
| Week [Q] : Weight bearing | −0.29 | −0.52 | −0.07 | 0.07 | −0.15 | 0.3 | |
| σ | Intercept | −0.52 | −0.65 | −0.41 | −0.52 | −0.65 | −0.4 |
| τ | β Intercept | 0.45 | 0.34 | 0.59 | 0.46 | 0.35 | 0.61 |
| σ Intercept | 0.25 | 0.11 | 0.41 | 0.31 | 0.17 | 0.47 | |
| α | 04.02 | 2.06 | 7.54 | 2.57 | 1.24 | 4.34 | |
| R^2 | Population level | 0.39 | 0.32 | 0.46 | 0.37 | 0.3 | 0.43 |
| Total | 0.6 | 0.55 | 0.64 | 0.57 | 0.51 | 0.62 | |
| Parameter | Overall Graft State | ||||||
|---|---|---|---|---|---|---|---|
| Sagittal | Axial | ||||||
| Me | LI | UI | Me | LI | UI | ||
| β | Intercept | −0.92 | −1.82 | −0.05 | −0.8 | −1.69 | 0.1 |
| Week [L] | 1.3 | 0.17 | 2.39 | 0.89 | −0.27 | 2.07 | |
| Week [Q] | −1.51 | −2.58 | −0.43 | −1.46 | −2.51 | −0.42 | |
| SNQ | 1.61 | 1.04 | 2.26 | 1.84 | 1.2 | 2.48 | |
| SD | 0.64 | 0.19 | 1.13 | 0.22 | −0.24 | 0.66 | |
| Sex | −0.3 | −1.08 | 0.48 | −0.49 | −1.27 | 0.26 | |
| Age | 0.27 | −0.51 | 1.04 | 0.2 | −0.57 | 0.97 | |
| Weight bearing | 0.06 | −0.8 | 0.95 | 0.18 | −0.71 | 1.05 | |
| Week [L] : Sex | 0.53 | −0.42 | 1.59 | 0.13 | −0.89 | 1.12 | |
| Week [Q] : Sex | 1.06 | 0.05 | 2.1 | 0.96 | −0.08 | 1.95 | |
| Week [L] : Age | 0.01 | −0.97 | 1.01 | −0.02 | −0.99 | 0.97 | |
| Week [Q] : Age | −0.53 | −1.54 | 0.4 | −0.79 | −1.8 | 0.15 | |
| Week [L] : Weight bearing | 0.44 | −0.68 | 1.54 | 0.59 | −0.49 | 1.72 | |
| Week [Q] : Weight bearing | 0.38 | −0.69 | 1.39 | 0.19 | −0.87 | 1.24 | |
| τ | β Intercept | 2.41 | 1.61 | 3.4 | 2.45 | 1.7 | 3.44 |
| Model accuracy | 0.91 | 0.9 | |||||
| Parameter | PCL Subluxation | ||||||
|---|---|---|---|---|---|---|---|
| Sagittal | Axial | ||||||
| Me | LI | UI | Me | LI | UI | ||
| β | Intercept | −0.73 | −1.67 | 0.18 | −0.64 | −1.66 | 0.36 |
| Week [L] | 3.58 | 2.43 | 4.73 | 3.52 | 2.3 | 4.74 | |
| Week [Q] | −0.86 | −1.93 | 0.25 | −1.01 | −2.07 | 0.04 | |
| SNQ | 1.03 | 0.51 | 1.6 | 1.12 | 0.6 | 1.67 | |
| SD | 0.18 | −0.28 | 0.64 | −0.33 | −0.79 | 0.11 | |
| Sex | −0.4 | −1.23 | 0.42 | −0.48 | −1.34 | 0.34 | |
| Age | −0.29 | −1.1 | 0.56 | −0.35 | −1.23 | 0.55 | |
| Weight bearing | 0.24 | −0.66 | 1.17 | 0.39 | −0.55 | 1.37 | |
| Week [L] : Sex | 0.16 | −0.87 | 1.23 | 0.02 | −1 | 1.1 | |
| Week [Q] : Sex | 0.74 | −0.2 | 1.74 | 0.7 | −0.26 | 1.71 | |
| Week [L] : Age | −0.32 | −1.37 | 0.71 | −0.2 | −1.3 | 0.92 | |
| Week [Q] : Age | 1.11 | 0.14 | 2.14 | 1.2 | 0.15 | 2.23 | |
| Week [L] : Weight bearing | −0.25 | −1.34 | 0.83 | −0.1 | −1.16 | 1.06 | |
| Week [Q] : Weight bearing | 0.4 | −0.63 | 1.41 | 0.32 | −0.76 | 1.39 | |
| τ | β Intercept | 2.64 | 1.84 | 3.69 | 2.86 | 2.01 | 3.88 |
| Model accuracy | 0.89 | 0.89 | |||||
| Parameter | Anterior Tibia Subluxation | ||||||
|---|---|---|---|---|---|---|---|
| Sagital | Axial | ||||||
| Me | LI | UI | Me | LI | UI | ||
| β | Intercept | 0.14 | −0.09 | 0.38 | 0.15 | −0.09 | 0.39 |
| Week [L] | 0.85 | 0.65 | 1.04 | 0.87 | 0.66 | 1.08 | |
| Week [Q] | −0.18 | −0.37 | 0 | −0.26 | −0.46 | −0.07 | |
| SNQ | 0.18 | 0.12 | 0.25 | 0.12 | 0.05 | 0.19 | |
| SD | 0.03 | −0.02 | 0.08 | −0.01 | −0.06 | 0.04 | |
| Sex | −0.15 | −0.35 | 0.06 | −0.17 | −0.39 | 0.05 | |
| Age | −0.11 | −0.3 | 0.09 | −0.12 | −0.33 | 0.09 | |
| PCLS at week 2 | −0.48 | −0.68 | −0.27 | −0.46 | −0.69 | −0.24 | |
| Weight bearing | 0.05 | −0.18 | 0.3 | 0.07 | −0.18 | 0.32 | |
| Week [L] : Sex | −0.04 | −0.18 | 0.1 | −0.05 | −0.19 | 0.1 | |
| Week [Q] : Sex | 0.1 | −0.02 | 0.23 | 0.11 | −0.02 | 0.24 | |
| Week [L] : Age | −0.24 | −0.39 | −0.09 | −0.21 | −0.36 | −0.05 | |
| Week [Q] : Age | 0.05 | −0.09 | 0.19 | 0.03 | −0.12 | 0.17 | |
| Week [L] : PCLS at week 2 | 0.21 | 0.07 | 0.35 | 0.17 | 0.03 | 0.31 | |
| Week [Q] : PCLS at week 2 | 0.08 | −0.05 | 0.2 | 0.09 | −0.04 | 0.23 | |
| Week [L] : Weight bearing | 0.15 | −0.04 | 0.34 | 0.21 | 0 | 0.41 | |
| Week [Q] : Weight bearing | 0 | −0.19 | 0.18 | −0.05 | −0.24 | 0.14 | |
| σ | Intercept | −1.04 | −1.19 | −0.88 | −1.01 | −1.16 | −0.85 |
| Week [L] | −0.5 | −0.8 | −0.2 | −0.42 | −0.71 | −0.12 | |
| Week [Q] | 0.38 | 0.06 | 0.71 | 0.55 | 0.23 | 0.86 | |
| PCLS at week 2 | 0.05 | −0.1 | 0.21 | 0.06 | −0.1 | 0.2 | |
| τ | β Intercept | 0.65 | 0.51 | 0.83 | 0.68 | 0.54 | 0.87 |
| σ Intercept | 0.42 | 0.29 | 0.57 | 0.39 | 0.27 | 0.54 | |
| α | −0.34 | −1.99 | 1.03 | 0.21 | −1.41 | 1.82 | |
| R^2 | Population level | 0.49 | 0.36 | 0.58 | 0.45 | 0.32 | 0.56 |
| Total | 0.83 | 0.81 | 0.84 | 0.82 | 0.79 | 0.83 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zdanowicz, U.; Ciszkowska-Łysoń, B.; Paśnik, M.; Drwięga, M.; Ratajczak, K.; Fulawka, K.; Lee, Y.C.; Śmigielski, R. Evaluation of ACL Graft Remodeling and Prediction of Graft Insufficiency in Sequenced MRI—Two-Year Follow-Up. Appl. Sci. 2021, 11, 5278. https://doi.org/10.3390/app11115278
Zdanowicz U, Ciszkowska-Łysoń B, Paśnik M, Drwięga M, Ratajczak K, Fulawka K, Lee YC, Śmigielski R. Evaluation of ACL Graft Remodeling and Prediction of Graft Insufficiency in Sequenced MRI—Two-Year Follow-Up. Applied Sciences. 2021; 11(11):5278. https://doi.org/10.3390/app11115278
Chicago/Turabian StyleZdanowicz, Urszula, Beata Ciszkowska-Łysoń, Marcin Paśnik, Michał Drwięga, Karol Ratajczak, Kamil Fulawka, Yoojin C. Lee, and Robert Śmigielski. 2021. "Evaluation of ACL Graft Remodeling and Prediction of Graft Insufficiency in Sequenced MRI—Two-Year Follow-Up" Applied Sciences 11, no. 11: 5278. https://doi.org/10.3390/app11115278
APA StyleZdanowicz, U., Ciszkowska-Łysoń, B., Paśnik, M., Drwięga, M., Ratajczak, K., Fulawka, K., Lee, Y. C., & Śmigielski, R. (2021). Evaluation of ACL Graft Remodeling and Prediction of Graft Insufficiency in Sequenced MRI—Two-Year Follow-Up. Applied Sciences, 11(11), 5278. https://doi.org/10.3390/app11115278






