Cyanobacterial Potential for Restoration of Loess Surfaces through Artificially Induced Biocrusts
Abstract
1. Introduction
2. Materials and Methods
2.1. Screening of Cyanobacterial Strains for Toxin, Biomass and Polysaccharide Production
2.1.1. Isolation and Cultivation of Cyanobacterial Strains
2.1.2. Toxicity Assessment
2.1.3. Quantification of Biomass Production
2.1.4. Polysaccharide Production
2.1.5. Viscosity of the Medium after Cultivation
2.1.6. Statistical Analysis
2.2. Ex Situ Particle Trapping and Stabilization Study
2.2.1. Microscopic Examination of Developed Biocrusts
2.2.2. Chl a Analysis
3. Results
3.1. Screening of Cyanobacterial Strains for Toxin, Biomass and Polysaccharide Production
3.1.1. Toxicity Assessment
3.1.2. Quantification of Biomass Production
3.1.3. Polysaccharide Production
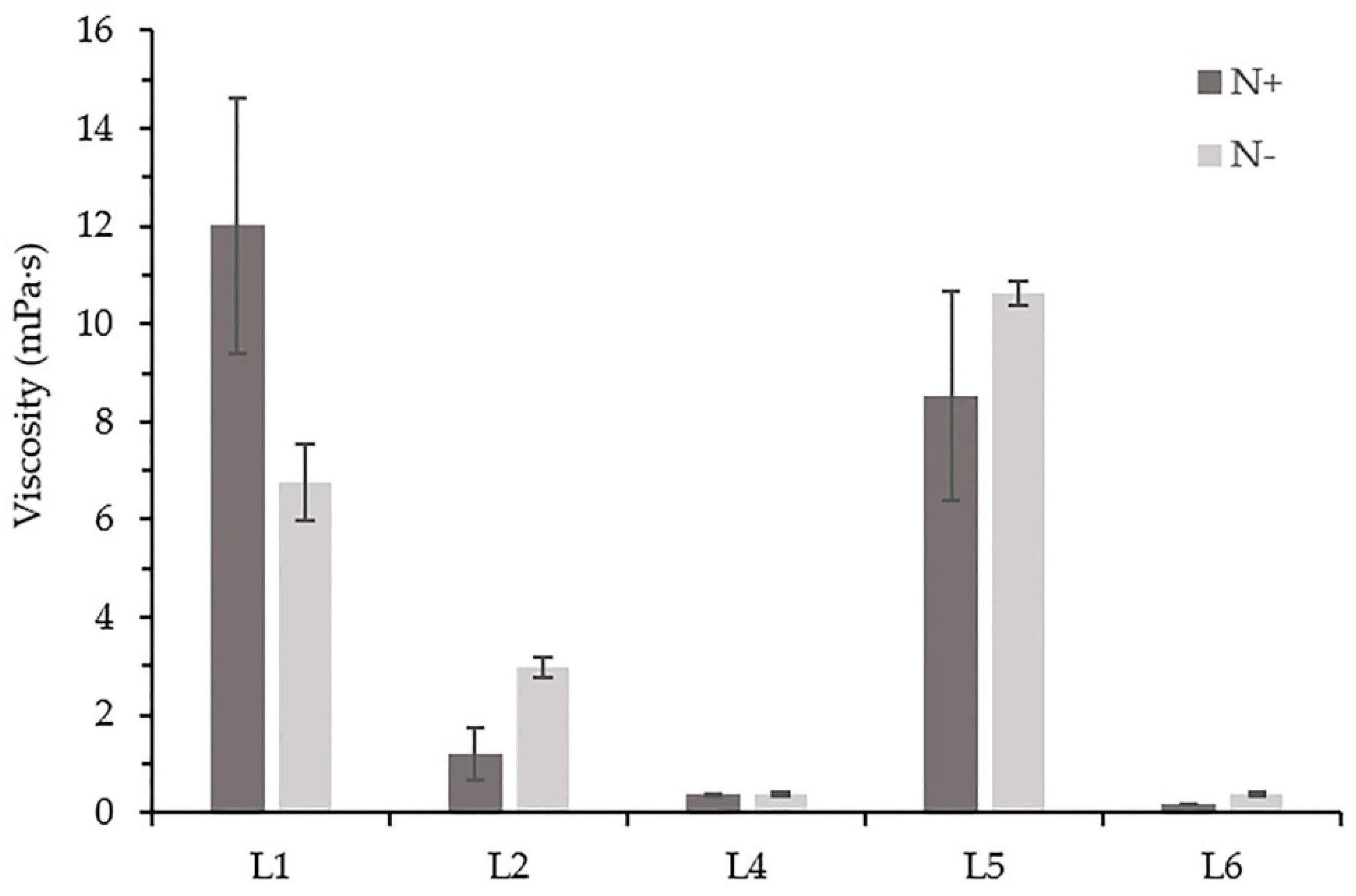
3.1.4. Viscosity of the Cell-Free Medium
3.1.5. Statistical Analyses
3.2. Ex Situ Particle Trapping and Stabilization Study
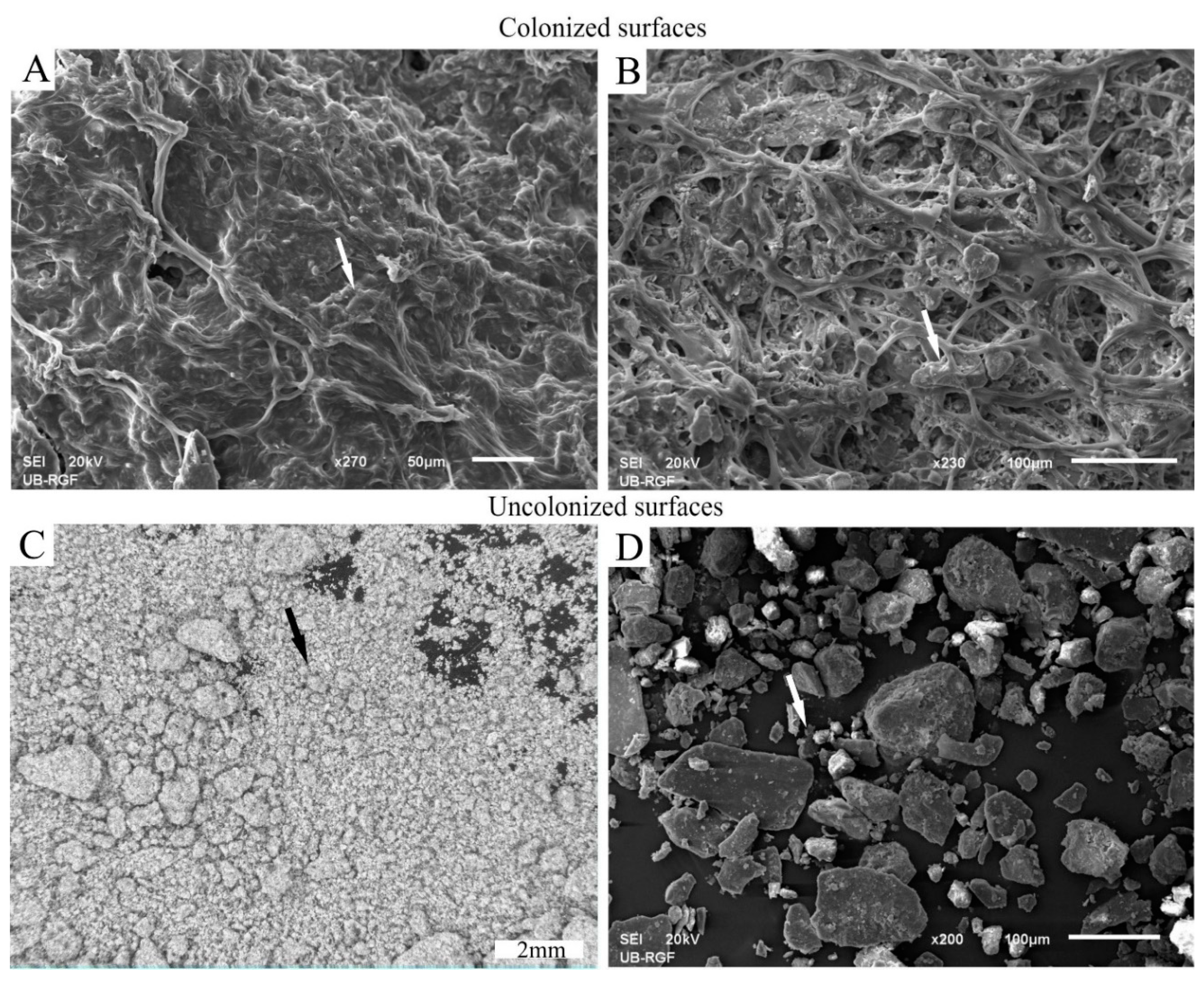
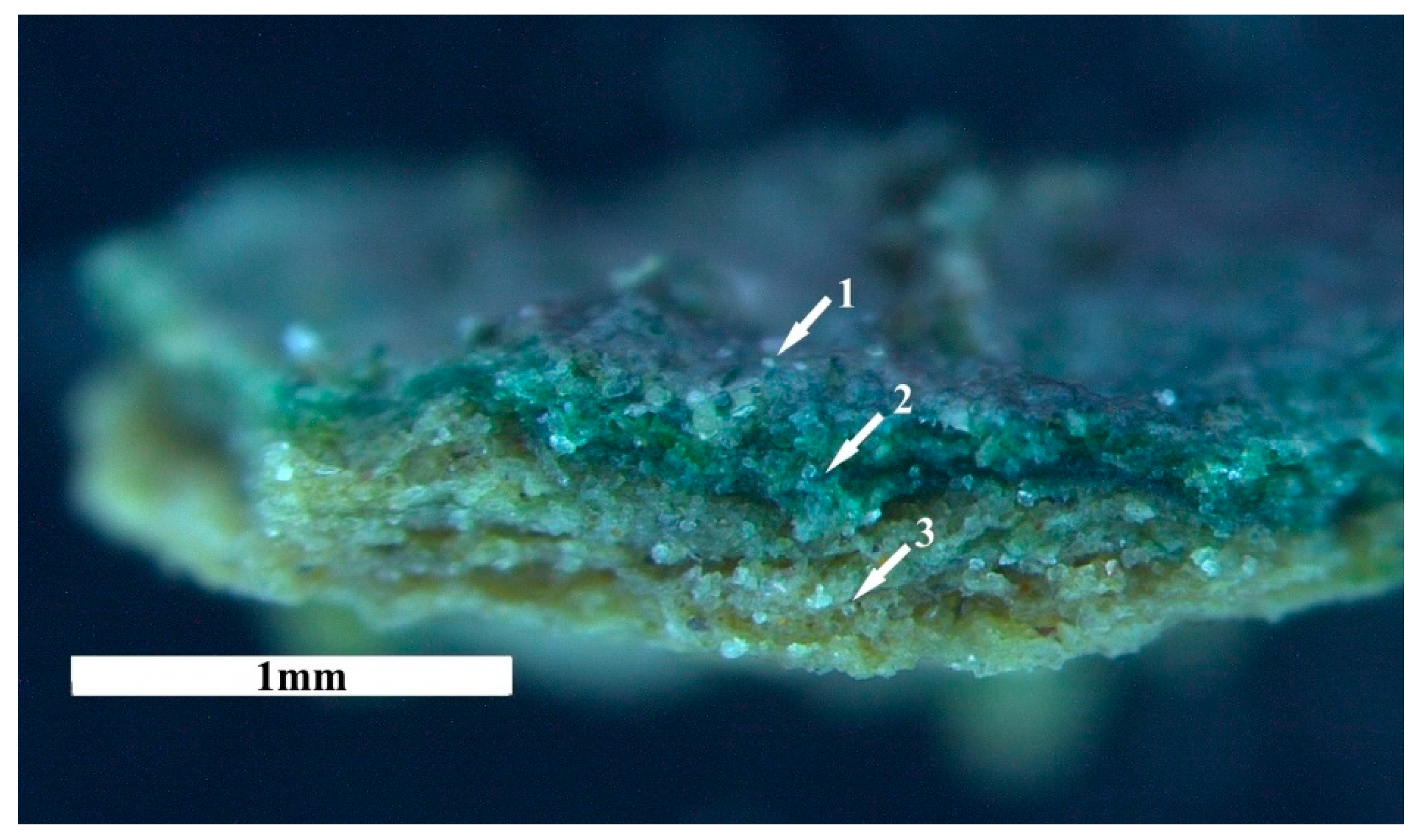
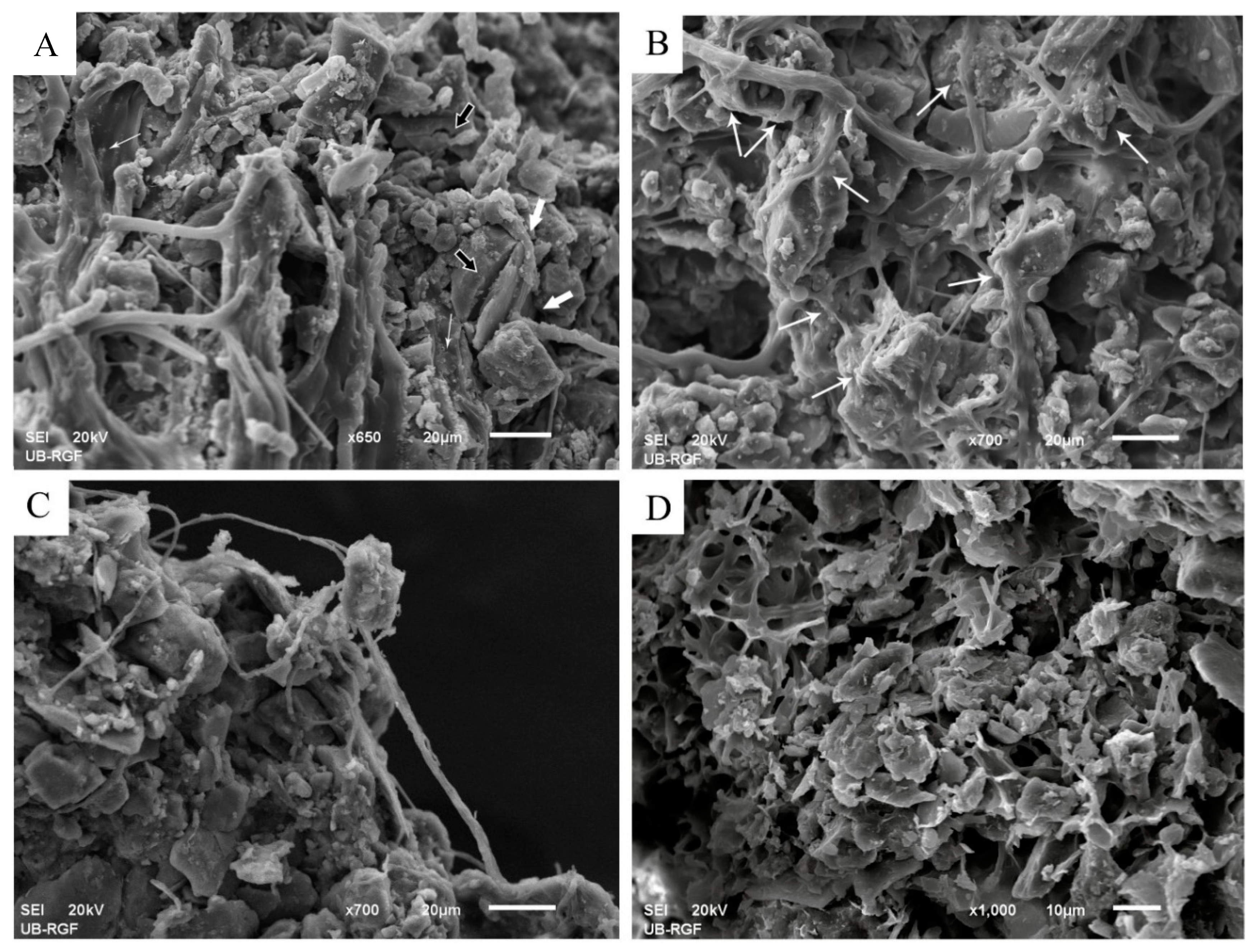
3.2.1. Development and Morphology of Biocrusts
3.2.2. Stabilization of Loess Particles by Cyanobacteria
Trapping of Particles
Accumulation and Stabilization of Particles
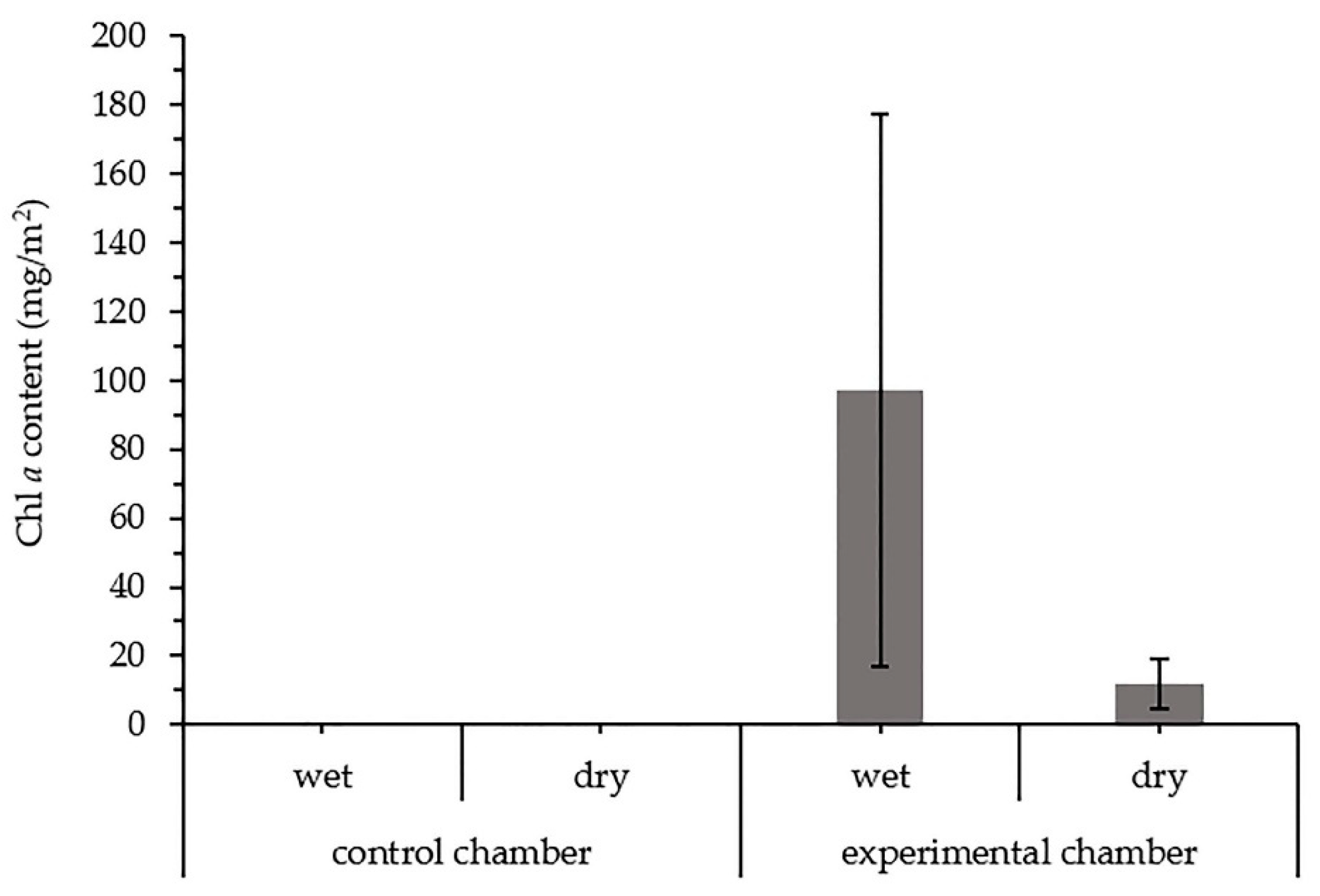
3.2.3. Chlorophyll a in the Biocrusts
4. Discussion
4.1. Production of Toxin, Biomass and Polysaccharide by the Cyanobacterial Strains
4.1.1. Toxicity
4.1.2. Production of Biomass
4.1.3. Production of Polysaccharides
4.2. Particle Trapping and Stabilization by Cyanobacteria
4.3. Strain Selection Criteria and Perspectives on the Application of Cyanobacteria in the Restoration of Exposed Loess Sediments
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Pécsi, M. The role of principles and methods in loess-paleosol investigations. GeoJournal 1995, 36, 117–131. [Google Scholar] [CrossRef]
- Pécsi, M. Loess is not just the accumulation of dust. Quat. Int. 1990, 7, 1–21. [Google Scholar] [CrossRef]
- Smalley, I.J.; Marković, S.B.; Svirčev, Z. Loess is [almost totally formed by] the accumulation of dust. Quat. Int. 2011, 240, 4–11. [Google Scholar] [CrossRef]
- Guo, Z.T.; Ruddiman, W.T.; Hao, Q.Z.; Wu, H.B.; Qiao, Y.S.; Zhu, R.X.; Peng, S.Z.; Wei, J.J.; Yuan, B.Y.; Liu, T.S. Onset of Asian desertification by 22 Myr ago inferred from loess deposits in China. Nature 2002, 416, 159–163. [Google Scholar] [CrossRef]
- Markovic, S.B.; Stevens, T.; Kukla, G.J.; Hambach, U.; Fitzsimmons, K.E.; Gibbard, P.; Buggle, B.; Zech, M.; Guo, Z.T.; Hao, Q.Z.; et al. Danube loess stratigraphy—Towards a pan-European loess stratigraphic model. Earth Sci. Rev. 2015, 148, 228–258. [Google Scholar] [CrossRef]
- Obreht, I.; Zeeden, C.; Hambach, U.; Veres, D.; Markovic, S.B.; Lehmkuhl, F. A critical reevaluation of palaeoclimate proxy records from loess in the Carpathian Basin. Earth Sci. Rev. 2019, 190, 498–520. [Google Scholar] [CrossRef]
- Sprafke, T.; Obreht, I. Loess: Rock, sediment or soil: What is missing for its definition? Quat Int. 2016, 399, 198–207. [Google Scholar] [CrossRef]
- Smalley, I.J.; Marković, S.B. Loessification and hydroconsolidation: There is a connection. Catena 2014, 117, 94–99. [Google Scholar] [CrossRef]
- Svirčev, Z.; Marković, S.B.; Stevens, T.; Codd, G.A.; Smalley, I.J.; Simeunović, J.; Obreht, I.; Dulić, T.; Pantelić, D.; Hambach, U. Importance of biological loess crusts for loess formation in semi-arid environments. Quat. Int. 2013, 296, 206–215. [Google Scholar] [CrossRef]
- Svirčev, Z.; Dulić, T.; Obreht, I.; Codd, G.A.; Lehmkuhl, F.; Marković, S.B.; Hambach, U.; Meriluoto, J. Cyanobacteria and loess-an underestimated interaction. Plant Soil. 2019. [Google Scholar] [CrossRef]
- United Nations Convention to Combat Desertification (UNCCD). United Nations Convention to Combat Desertification in Those Countries Experiencing Serious Drought and/or Desertification Particularly in Africa; United Nations: Paris, France, 1994. [Google Scholar]
- Burrell, A.L.; Evans, J.P.; De Kauwe, M.G. Anthropogenic climate change has driven over 5 million km2 of drylands towards desertification. Nat. Commun. 2020, 11, 3853. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Zhang, G.; Zhang, Y.; Guan, X.; Wei, Y.; Guo, R. Global desertification vulnerability to climate change and human activities. Land Degrad. Dev. 2020, 31, 1380–1391. [Google Scholar] [CrossRef]
- Maestre, F.T.; Quero, J.L.; Gotelli, N.J.; Escudero, A.; Ochoa, V.; Delgado-Baquerizo, M.; García-Gómez, M.; Bowker, M.A.; Soliveres, S.; Escolar, C.; et al. Plant species richness and ecosystem multifunctionality in global drylands. Science 2012, 335, 214–218. [Google Scholar] [CrossRef] [PubMed]
- United Nations Convention to Combat Desertification (UNCCD). Desertification, Land Degradation and Drought (DLDD): Some Global Facts and Figures; United Nations: Bonn, Germany, 2015. [Google Scholar]
- Rashki, A.; Eriksson, P.G.; Rautenbach, C.D.; Kaskaoutis, D.G.; Grote, W.; Dykstra, J. Assessment of chemical and mineralogical characteristics of airborne dust in the Sistan region, Iran. Chemosphere 2013, 90, 227–236. [Google Scholar] [CrossRef]
- Mostern, R. Loess is More: The Spatial and Ecological History of Erosion on China’s Northwest Frontier. J. Econ. Soc. Hist. Orient 2019, 62, 560–598. [Google Scholar] [CrossRef]
- Sweeney, M.R.; Mason, J.A. Mechanisms of dust emission from Pleistocene loess deposits, Nebraska, USA. J. Geophys. Res. Earth Surf. 2013, 118, 1–12. [Google Scholar] [CrossRef]
- Munson, S.; Belnap, J.; Okin, G.S. Responses of wind erosion to climate-induced vegetation changes on the Colorado Plateau. Proc. Natl. Acad. Sci. USA 2011, 108, 3854–3859. [Google Scholar] [CrossRef]
- National Development and Reform Commission (NDRC); Ministry of Water Resources (MWR); Ministry of Agriculture (MA); State Forestry Administration (SFA). People’s Republic of China. In Programming for Comprehensive Management of the Loess Plateau (2010–2030); 2010; pp. 4–14. [Google Scholar]
- Shi, H.; Shao, M. Soil and water loss from the Loess Plateau in China. J. Arid Environ. 2000, 45, 9–20. [Google Scholar] [CrossRef]
- Cai, Q. The relationships between soil erosion and human activities on the Loess Plateau. In Proceedings of the 12th ISCO Conference, Beijing, China, 26–31 May 2002. [Google Scholar]
- Goudie, A.S. Desert dust and human health disorders. Environ. Int. 2014, 63, 101–113. [Google Scholar] [CrossRef]
- Raghimi, M.; Ramezani Mojaveri, M. Investigation of esophageal cancer with medical geology aspect in Golestan Province, Iran. Chin. J. Geochem. 2006, 25, 58–59. [Google Scholar] [CrossRef]
- Jabbari, A.; Besharat, S.; Semnani, S. Role of silis in esophageal cancer. World J. Gastroenterol. 2008, 14, 3106–3107. [Google Scholar] [CrossRef] [PubMed]
- Cao, S.; Chen, L.; Shankman, D.; Wang, C.; Wang, X.; Zhang, H. Excessive reliance on afforestation in China’s arid and semi-arid regions: Lessons in ecological restoration. Earth-Sci. Rev. 2011, 104, 240–245. [Google Scholar] [CrossRef]
- Ostwald, M.; Moberg, J.; Persson, M.; Xu, J. The Chinese Grain for Green Program-assessing the sequestered carbon from the land reform. In Proceedings of the World Rewable Energy Congress, Linköping, Sweden, 8–13 May 2011. [Google Scholar]
- Gao, L.; Bowker, M.A.; Xu, M.; Sun, H.; Tuo, D.; Zhao, Y. Biological soil crusts decrease erodibility by modifying inherent soil properties on the Loess Plateau, China. Soil Biol. Biochem. 2017, 105, 49–58. [Google Scholar] [CrossRef]
- Cao, S.; Chen, L.; Yu, X. Impact of China’s Grain for Green Project on the landscape of vulnerable arid and semiarid agricultural regions: A case study in northern Shaanxi Province. J. Appl. Ecol. 2009, 46, 536–543. [Google Scholar] [CrossRef]
- Wang, X.; Zhang, C.; Hasi, E.; Dong, Z. Has the Three Norths Forest Shelterbelt Program solved the desertification and dust storm problems in arid and semiarid China? J. Arid Environ. 2010, 74, 13–22. [Google Scholar] [CrossRef]
- Lamb, D.; Erskine, P.D.; Parrotta, J.A. Restoration of degraded tropical forest landscapes. Science 2005, 310, 1628–1632. [Google Scholar] [CrossRef]
- Thomas, D.G. Science and the desertification debate. J. Arid Environ. 1997, 37, 599–608. [Google Scholar] [CrossRef]
- Chen, Y.; Tang, H. Desertification in North China: Background, anthropogenic impacts and failures in combating it. Land Degrad. Develop. 2005, 16, 367–376. [Google Scholar] [CrossRef]
- Normile, D. Getting at the roots of killer dust storms. Science 2007, 317, 314–316. [Google Scholar] [CrossRef]
- Lan, S.; Zhang, Q.; Wu, L.; Liu, Y.; Zhang, D.; Hu, C. Artificially accelerating the reversal of desertification: Cyanobacterial inoculation facilitates the succession of vegetation communities. Environ. Sci. Technol. 2014, 48, 307–315. [Google Scholar] [CrossRef]
- Zhao, Y.; Xu, M. Runoff and soil loss from revegetated grasslands in the Hilly Loess Plateau Region, China: Influence of biocrust patches and plant canopies. J. Hydrol. Eng. 2013, 18, 387–393. [Google Scholar] [CrossRef]
- Eldridge, D.J.; Greene, R.S. Microbiotic soil crusts: A review of their roles in soil and ecological processes in the rangelands of Australia. Aust. J. Soil Res. 1994, 32, 389–415. [Google Scholar] [CrossRef]
- Williams, A.J.; Buck, B.J.; Beyene, M.A. Biological soil crusts in the Mojave Desert, USA: Micromorphology and pedogenesis. Soil Sci. Soc. Am. J. 2012, 76, 1685–1695. [Google Scholar] [CrossRef]
- Svirčev, Z.; Nikolić, B.; Vukić, V.; Marković, S.; Gavrilov, M.; Ian, S.; Obreht, I.; Vukotić, B.; Meriluoto, J. Loess and life out of Earth? Quat. Int. 2016, 399, 208–217. [Google Scholar] [CrossRef]
- Malam Issa, O.; Trichet, J.; De’farge, C.; Coute, A.; Valentin, C. Morphology and microstructure of microbiotic soil crusts on a tiger bush sequence (Niger, Sahel). Catena 1999, 37, 175–196. [Google Scholar] [CrossRef]
- Malam Issa, O.; Le Bissonnais, Y.; De´farge, C.; Trichet, J. Role of a cyanobacterial cover on structural stability of sandy soils in the Sahelian part of western Niger. Geoderma 2001, 101, 15–30. [Google Scholar] [CrossRef]
- Xiao, B.; Wang, Q.H.; Zhao, Y.G.; Shao, M.A. Artificial culture of biological soil crusts and its effects on overland flow and infiltration under simulated rainfall. Appl. Soil Ecol. 2011, 48, 11–17. [Google Scholar] [CrossRef]
- Lan, S.B.; Wu, L.; Zhang, D.L.; Hu, C.X. Assessing level of development and successional stages in biological soil crusts with biological indicators. Microb. Ecol. 2013, 66, 394–403. [Google Scholar] [CrossRef]
- Maestre, F.T.; Bowker, M.A.; Cantón, Y.; Castillo-Monroy, A.P.; Cortina, J.; Escolar, C.; Escudero, A.; Lázaro, R.; Martínez, I. Ecology and functional roles of biological soil crusts in semi-arid ecosystems of Spain. J. Arid Environ. 2011, 75, 1282–1291. [Google Scholar] [CrossRef]
- Belnap, J. The world at your feet: Desert biological soil crusts. Fron. Ecol. Environ. 2003, 1, 181–189. [Google Scholar] [CrossRef]
- Wang, W.B.; Liu, Y.D.; Li, D.H.; Hu, C.X.; Rao, B.Q. Feasibility of cyanobacterial inoculation for biological soil crusts formation in desert area. Soil Biol. Biochem. 2009, 41, 926–929. [Google Scholar] [CrossRef]
- Li, X.R.; Tian, F.; Jia, R.L.; Zhang, Z.S.; Liu, L.C. Do biological soil crusts determine vegetation changes in sandy deserts? Implications for managing artificial vegetation. Hydrol. Process. 2010, 24, 3621–3630. [Google Scholar] [CrossRef]
- Hu, C.X.; Liu, Y.D. Primary succession of Algal community structure in desert soil. Acta Bot. Sin. 2003, 45, 917–924. [Google Scholar]
- Chiquoine, L.P. Restoration of Biological Soil Crust on Disturbed Gypsiferous Soils in Lake Mead National Recreation Area, Eastern Mojave Desert. Master’s Thesis, Las Vegas, University of Nevada, Reno, NV, USA, 2012. [Google Scholar]
- LeQuire, E. Biological soil crusts: A crucial component of arid ecosystems. J. Fire Sci. Brief 2009, 85, 1–6. [Google Scholar]
- Malam Issa, O.; Défarge, C.; Le Bissonais, Y.; Marin, B.; Duval, O.; Bruand, A.; D’Acqui, L.P.; Nordenberg, S.; Annerman, M. Effects of the inoculation of cyanobacteria on the microstructure and the structural stability of a tropical soil. Plant Soil 2007, 290, 209–219. [Google Scholar] [CrossRef]
- Chen, L.; Xie, Z.; Hu, C.; Li, D.; Wang, G.; Liu, Y. Man-made desert algal crusts as affected by environmental factors in Inner Mongolia, China. J. Arid Environ. 2006, 67, 521–527. [Google Scholar] [CrossRef]
- Xie, Z.; Liu, Y.; Hu, C.; Chen, L.; Li, D. Relationships between the biomass of algal crusts in fields and their compressive strength. Soil Biol. Biochem. 2007, 39, 567–572. [Google Scholar] [CrossRef]
- Lan, S.B.; Wu, L.; Zhang, D.L.; Hu, C.X.; Liu, Y.D. Effects of drought and salt stresses on man-made cyanobacterial crusts. Eur. J. Soil Biol. 2010, 46, 381–386. [Google Scholar] [CrossRef]
- Wu, Y.; Rao, B.; Wu, P.; Liu, Y.; Li, G.; Li, D. Development of artificially induced biological soil crusts in fields and their effects on top soil. Plant Soil 2013, 370, 115–124. [Google Scholar] [CrossRef]
- Rozenstein, O.; Zaady, E.; Katra, I.; Karnieli, A.; Adamowski, J.; Yizhaq, H. The effect of sand grain size on the development of cyanobacterial biocrusts. Aeolian Res. 2014, 15, 217–226. [Google Scholar] [CrossRef]
- Mugnai, G.; Rossi, R.; Felde, V.J.M.N.L.; Colesie, C.; Büdel, B.; Peth, S.; Kaplan, A.; De Philippis, R. Development of the polysaccharidic matrix in biocrusts induced by a cyanobacterium inoculated in sand microcosms. Biol. Fertil. Soils. 2017, 54, 27–40. [Google Scholar] [CrossRef]
- Mugnai, G.; Rossi, F.; Felde, V.J.M.N.L.; Colesie, C.; Büdel, B.; Peth, S.; Kaplan, A.; De Philippis, R. The potential of the cyanobacterium Leptolyngbya ohadii as inoculum for stabilizing bare sandy substrates. Soil Biol. Biochem. 2018, 127, 318–328. [Google Scholar] [CrossRef]
- Park, C.H.; Li, X.R.; Zhao, Y.; Jia, R.L.; Hur, J.S. Rapid development of cyanobacterial crust in the field for combating desertification. PLoS ONE 2017, 12, e0179903. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Zhu, Q.; Li, P.; Zhao, L.; Wang, L.; Zheng, X.; Ma, H. Effects of artificially cultivated biological soil crusts on soil nutrients and biological activities in the Loess Plateau. J. Arid Land. 2014, 6, 742–752. [Google Scholar] [CrossRef]
- Xiao, B.; Zhao, Y.; Wang, B.; Li, C. Development of artificial moss-dominated biological soil crusts and their effects on runoff and soil water content in a semi-arid environment. J. Arid Environ. 2015, 11, 75–83. [Google Scholar] [CrossRef]
- Belnap, J.; Lange, L.O. Biological Soil Crusts: Structure, Function and Management; Springer Verlag: Berlin, Germany, 2003. [Google Scholar]
- Garcia-Pichel, F.; Pringault, O. Cyanobacteria track water in desert soils. Nature 2001, 413, 380–381. [Google Scholar] [CrossRef]
- Rao, B.Q.; Wu, P.P.; Dauta, A.; Li, D.H.; Liu, Y.D. Effects of UV-B radiation on growth and ultrastructures of cyanobacterial crusts under greenhouse conditions. Acta Sci. Circum. 2011, 31, 649–657. [Google Scholar]
- Rossi, F.; De Philippis, R. Role of Cyanobacterial Exopolysaccharides in phototrophic biofilms and in complex microbial mats. Life 2015, 5, 1218–1238. [Google Scholar] [CrossRef]
- De Philippis, R.; Vincenzini, M. Exocellular polysaccharides from cyanobacteria and their possible applications. FEMS Microbiol. Rev. 1998, 22, 151–175. [Google Scholar] [CrossRef]
- Mazor, G.; Kidron, G.J.; Vonshak, A.; Abeliovich, A. The role of cyanobacterial exopolysaccharides in structuring desert microbial crusts. FEMS Microbiol. Ecol. 1996, 21, 121–130. [Google Scholar] [CrossRef]
- Potts, M. Desiccation tolerance: A simple process? Trends Microbiol. 2001, 9, 553–559. [Google Scholar] [CrossRef]
- Rippka, R. Isolation and purification of Cyanobacteria. Meth. Enzymol. 1988, 167, 3–27. [Google Scholar]
- Komárek, J.; Anagnostidis, K. Cyanoprokaryota 1. Chroococcales. In Süsswasserflora von Mitteleuropa 19/1; Ettl, H., Gärtner, G., Heynig, H., Mollenhauer, D., Eds.; Gustav Fischer: Jena, Germany, 1998; p. 548. [Google Scholar]
- Komárek, J.; Anagnostidis, K. Cyanoprokaryota. 2. Oscillatoriales. In Süsswasserflora von Mitteleuropa 19/2; Büdel, B., Krienitz, L., Gärtner, G., Schagerl, M., Eds.; Elsevier/Spektrum: Heidelberg, Germany, 2005; p. 759. [Google Scholar]
- Komárek, J. Cyanoprokaryota. 3. Heterocytous genera. In Süswasserflora von Mitteleuropa/Freshwater flora of Central Europe; Büdel, B., Gärtner, G., Krienitz, L., Schagerl, M., Eds.; Springer/Spektrum: Berlin/Heidelberg, Germany, 2013; p. 1130. [Google Scholar]
- Kiviranta, J.; Sivonen, K.; Niemelä, S.I.; Huovinen, K. Detection of toxicity of cyanobacteria by Artemia salina bioassay. Environ. Toxicol. Water Qual. 1991, 6, 423–436. [Google Scholar] [CrossRef]
- Tokodi, N.; Drobac, D.; Lazić, G.; Petrović, T.; Lujić, J.; Marinović, Z.; Palanački Malešević, T.; Meriluoto, J.; Svirčev, Z. Screening of cyanobacterial cultures originating from different environments for cyanotoxicity and cyanotoxins. Toxicon 2018, 154, 1–6. [Google Scholar] [CrossRef]
- Mackinney, G. Absorption of light by chlorophyll solutions. J. Biol. Chem. 1941, 140, 315–322. [Google Scholar]
- APHA. Standard Methods for the Examination of Waste and Wastewater; American Public Health Association: Washington, DC, USA, 1992. [Google Scholar]
- Dubois, M.; Gilles, K.A.; Hamilton, J.K.; Rebers, P.A.; Smith, F. Colorimetric method for determination of sugars and related substances. Anal. Chem. 1956, 38, 350–356. [Google Scholar] [CrossRef]
- Masuko, T.; Minami, A.; Iwasaki, N.; Majima, T.; Nishimura, S.I.; Lee, Y.C. Carbohydrate analysis by a phenol-sulfuric acid method in a microplate format. Anal. Biochem. 2005, 339, 69–72. [Google Scholar] [CrossRef]
- Garcia-Pichel, F.; Castenholz, R.W. Characterization and biological implications of scytonemin, a cyanobacterial sheath pigment. J. Phycol. 1991, 27, 395–409. [Google Scholar] [CrossRef]
- Garcia-Pichel, F.; Sherry, N.D.; Castenholz, R.W. Evidence for an ultraviolet sunscreen role of the extracellular pigment scytonemin in the terrestrial cyanobacterium. Chlorogloeopsis. Photochem. Photobiol. 1992, 56, 17–23. [Google Scholar] [CrossRef]
- Svirčev, Z.; Krstić, S.; Miladinov-Mikov, M.; Vidović, M. Freshwater cyanobacterial blooms and primary liver cancer epidemiological studies in Serbia. J. Environ. Sci. Health C 2009, 27, 36–55. [Google Scholar] [CrossRef]
- Svirčev, Z.; Lujić, J.; Marinović, Z.; Drobac, D.; Tokodi, N.; Stojiljković, B.; Meriluoto, J. Toxicopathology induced by microcystins and nodularin: A histopathological review. J. Environ. Sci. Health C 2015, 33, 125–167. [Google Scholar] [CrossRef] [PubMed]
- Svirčev, Z.; Obradović, V.; Codd, G.A.; Marjanović, P.; Spoof, L.; Drobac, D.; Tokodi, N.; Petković, A.; Nenin, T.; Simeunović, J.; et al. Massive fish mortality and Cylindrospermopsis raciborskii bloom in Aleksandrovac Lake. Ecotoxicology 2016, 25, 1353–1363. [Google Scholar] [CrossRef] [PubMed]
- Metcalf, J.S.; Codd, G.A. Cyanotoxins. In Ecology of Cyanobacteria II Their Diversity in Space and Time; Whitton, B.A., Ed.; Springer: Dordrecht, The Netherlands, 2012; pp. 651–675. [Google Scholar]
- Svirčev, Z.; Baltić, V.; Gantar, M.; Juković, M.; Stojanović, D.; Baltić, M. Molecular aspects of microcystin-induced hepatotoxicity and hepatocarcinogenesis. J. Environ. Sci. Health C 2010, 28, 39–59. [Google Scholar] [CrossRef] [PubMed]
- Svirčev, Z.; Drobac, D.; Tokodi, N.; Vidović, M.; Simeunović, J.; Miladinov-Mikov, M.; Baltić, V. Epidemiology of primary liver cancer in Serbia and possible connection with cyanobacterial blooms. J. Environ. Sci. Health C 2013, 31, 181–200. [Google Scholar] [CrossRef] [PubMed]
- Svirčev, Z.; Drobac, D.; Tokodi, N.; Lužanin, Z.; Munjas, A.M.; Nikolin, B.; Vuleta, D.; Meriluoto, J. Epidemiology of cancers and in Serbia and possible connection with cyanobacterial blooms. J. Environ. Sci. Health C 2014, 32, 319–337. [Google Scholar] [CrossRef] [PubMed]
- Žegura, B.; Štraser, A.; Filipič, M. Genotoxicity and potential carcinogenicity of cyanobacterial toxins-a review. Mutat. Res. 2011, 727, 16–41. [Google Scholar] [CrossRef]
- Bláha, L.; Babica, P.; Maršálek, B. Toxins produced in cyanobacterial water blooms-toxicity and risks. Interdisc. Toxicol. 2009, 2, 36–41. [Google Scholar] [CrossRef]
- Marsalek, B.; Bláha, L. Methods for detection and quantification of cyanobacterial toxins-a review. Algol. Stud. 2000, 99, 1–22. [Google Scholar]
- Yu, H.; Jia, S.; Dai, Y. Accumulation of Exopolysaccharides in Liquid Suspension Culture of Nostoc flagelliforme Cells. Appl. Bichem. Biotechnol. 2010, 160, 552–560. [Google Scholar] [CrossRef]
- Aboim, J.B.; De Oliveira, D.T.; De Mescouto, V.A.; Reis, A.S.; Rocha Filho, G.N.; Santos, A.V.; Xavier, L.P.; Santos, A.S.; Gonçalves, E.C.; Nascimento, L.A.S. Optimization of light intensity and NaNO3 concentration in Amazon Cyanobacteria cultivation to produce biodiesel. Molecules 2019, 24, 2326. [Google Scholar] [CrossRef]
- Chaneva, G.; Furnadzhieva, S.; Minkova, K.; Lukavsky, J. Effect of light and temperature on the cyanobacterium Arthronema africanum a prospective phycobiliprotein producing strain. J. Appl. Phycol. 2007, 19, 537–544. [Google Scholar] [CrossRef]
- Kovač, D.; Babić, O.; Milovanović, I.; Mišan, A.; Simeunović, J. The production of biomass and phycobiliprotein pigments in filamentous cyanobacteria: The impact of light and carbon sources. Appl. Biochem. Microbiol. 2017, 53, 539–545. [Google Scholar] [CrossRef]
- Billi, D.; Grilli Caiola, M. Effects of nitrogen limitations and starvation Chroococcidiopsis sp. (Chroococcales). New Phytol. 1996, 133, 563–571. [Google Scholar] [CrossRef]
- Boison, G.; Mergel, A.; Jolkver, H.; Bothe, H. Bacterial life and dinitrogen fixation at Gypsum Rock. Appl. Environ. Microbiol. 2004, 70, 7070–7077. [Google Scholar] [CrossRef] [PubMed]
- Pereira, S.; Zille, A.; Micheletti, E.; Moradas-Ferreira, P.; De Philippis, R.; Tamagnini, P. Complexityof cyanobacterial exopolysaccharides: Composition, structures, inducing factors and putative genes involved in their biosynthesis and assembly. FEMS Microbiol. Rev. 2009, 33, 917–941. [Google Scholar] [CrossRef] [PubMed]
- Moreno, J.; Vargas, M.A.; Olivares, H.; Rivas, J.; Guerrero, M.G. Exopolysaccharide production by the cyanobacterium Anabaena sp. ATCC 33047 in batch and continuous culture. J. Biotechnol. 1998, 60, 175–182. [Google Scholar] [CrossRef]
- Su, C.; Chi, Z.; Lu, W. Optimization of medium and cultivation conditions for enhanced exopolysaccharide yield by marine Cyanothece sp. 113. Chin. J. Oceanol. Limnol. 2007, 25, 411–417. [Google Scholar] [CrossRef]
- Richert, L.; Golubic, S.; Le Gu´ede‘s, R.; Ratiskol, J.; Payri, C.; Guezennec, J. Characterization of exopolysaccharides produced by cyanobacteria isolated from Polynesian microbial mats. Curr. Microbiol. 2005, 51, 379–384. [Google Scholar] [CrossRef]
- Nicolaus, B.; Panico, A.; Lama, L.; Romano, I.; Manca, M.C.; De Giulio, A.; Gambacorta, A. Chemical composition and production of exopolysaccharides from representative members of heterocystous and non-heterocystous cyanobacteria. Phytochemistry 1999, 52, 639–647. [Google Scholar] [CrossRef]
- Trabelsi, L.; Ouada, H.B.; Bacha, H.; Ghoul, M. Combined effect of temperature and light intensity on growth and extracellular polymeric substance production by the cyanobacterium Arthrospira platensis. J. Appl. Phycol. 2009, 21, 405–412. [Google Scholar] [CrossRef]
- Mota, R.; Guimarães, R.; Büttel, Z.; Rossi, F.; Colica, G.; Silva, C.J.; Santos, C.; Gales, L.; Zille, A.; De Philippis, R.; et al. Production and characterization of extracellular carbohydrate polymer from Cyanothece sp. CCY 0110. Carbohyd. Polym. 2013, 92, 1408–1415. [Google Scholar] [CrossRef] [PubMed]
- Whistler, R.; Daniel, R. Function of polysaccharides in foods. In Food Additives; Branen, A.L., Davidson, P.M., Salminen, S., Eds.; Marcel Dekker: New York, NY, USA, 1990. [Google Scholar]
- Lan, S.; Wu, L.; Zhang, D.; Hu, C. Successional stages of biological soil crusts and their microstructure variability in Shapotou region (China). Environ. Earth Sci. 2012, 65, 77–88. [Google Scholar] [CrossRef]
- Zaady, E.; Offer, Z.Y. Biogenic soil crusts and soil depth: A long-term case study from the Central Negev desert highland. Sedimentology 2010, 57, 351–358. [Google Scholar] [CrossRef]
- Lange, O.L. Photosynthesis of soil crust biota is dependent on environmental factors. In Biological Soil Crusts: Structure, Function and Management; Belnap, J., Lange, O.L., Eds.; Springer: Berlin, Germany, 2003; pp. 217–240. [Google Scholar]
- Verrecchia, E.; Yair, A.; Kidron, G.J.; Verrecchia, K. Physical properties of the psammophile cryptogamic crust and their consequences to the water regime of sandy soils, north-western Negev Desert, Israel. J. Arid Environ. 1995, 29, 427–437. [Google Scholar] [CrossRef]
- Zhang, Y.M.; Wang, H.L.; Wang, X.Q.; Yang, W.K.; Zhang, D.Y. The microstructure of microbiotic crust and its influence on wind erosion for a sandy soil surface in the Gurbantunggut Desert of Northwestern China. Geoderma 2006, 132, 441–449. [Google Scholar] [CrossRef]
- Dulić, T.; Meriluoto, J.; Palanački Malešević, T.; Gajić, V.; Važić, T.; Tokodi, N.; Obreht, I.; Kostić, B.; Kosijer, P.; Khormali, F.; et al. Cyanobacterial diversity and toxicity of biocrusts from the Caspian Lowland loess deposits, North Iran. Quat. Int. 2017, 429, 74–85. [Google Scholar] [CrossRef]
- Zhao, Y.; Bowker, M.A.; Zhang, Y.; Zaady, E. Enhanced Recovery of Biological Soil Crusts after Disturbance. In Biological Soil Crusts: An Organising Principle in Drylands; Weber, B., Büdel, B., Belnap, J., Eds.; Springer International Publishing: Cham, Switzerland, 2016; pp. 498–523. [Google Scholar]
- Cox, P.A.; Richer, R.; Metcalf, J.S.; Banack, S.A.; Codd, G.A.; Bradley, W.G. Cyanobacteria and BMAA exposure from desert dust: A possible link to sporadic ALS among Gulf War veterans. Amyotroph. Lateral. Scle. 2009, 10, 109–117. [Google Scholar] [CrossRef]
- Metcalf, J.S.; Richer, R.; Cox, P.A.; Codd, G.A. Cyanotoxins in desert environments may present a risk to human health. Sci. Total Environ. 2012, 421–422, 118–123. [Google Scholar] [CrossRef]
- Metcalf, J.S.; Banack, S.A.; Richer, R.; Cox, P.A. Neurotoxic amino acids and their isomers in desert environments. J. Arid Environ. 2015, 112, 140–144. [Google Scholar] [CrossRef]
- Richer, R.; Banack, S.A.; Metcalf, J.S.; Cox, P.A. The persistence of cyanobacterial toxins in desert soils. J. Arid Environ. 2015, 112, 134–139. [Google Scholar] [CrossRef]
- Hu, C.; Liu, Y.; Song, L.; Zhang, D. Effect of desert soil algae on the stabilization of fine sands. J. Appl. Phycol. 2002, 14, 281–292. [Google Scholar] [CrossRef]
- Giraldo-Silva, A.; Nelson, C.; Barger, N.N.; Garcia-Pichel, F. Nursing bio-crusts: Isolation, cultivation, and fitness test of indigenous cyanobacteria. Restor. Ecol. 2019, 27, 793–803. [Google Scholar] [CrossRef]
- Mugnai, G.; Rossi, F.; Chamizo, S.; Adessi, A.; De Philippis, R. The role of grain size and inoculum amount on biocrust formation by Leptolyngbya ohadii. Catena 2020, 184, 104248. [Google Scholar] [CrossRef]
- Chamizo, S.; Adessi, A.; Mugnai, G.; Simiani, A.; De Philippis, R. Soil type and cyanobacteria species influence the macromolecular and chemical characteristics of the polysaccharidic matrix in induced biocrusts. Microb. Ecol. 2019, 78, 482–493. [Google Scholar] [CrossRef]
- Bu, C.; Wu, S.; Yang, Y.; Zheng, M. Identification of factors influencing the restoration of Cyanobacteria-dominated biological soil crusts. PLoS ONE 2014, 9, e90049. [Google Scholar] [CrossRef]
- Rossi, F.; Hua, L.; Liu, Y.; De Philippis, R. Cyanobacterial inoculation (cyanobacterisation): Perspectives for the development of a standardized multifunctional technology for soil fertilization and desertification reversal. Earth-Sci. Rev. 2017, 171, 28–43. [Google Scholar] [CrossRef]
- Chamizo, S.; Mugnai, G.; Rossi, F.; Certini, G.; De Philippis, R. Cyanobacteria inoculation improves soil stability and fertility on different textured soils: Gaining insights for applicability in soil restoration. Front. Environ. Sci. 2018, 6. [Google Scholar] [CrossRef]
- West, N.E. Structure and function of microphytic soil crusts in wildland ecosystems of arid to semi-arid regions. Adv. Ecol. Res. 1990, 20, 179–223. [Google Scholar]
- Malam Issa, O.; Défarge, C.; Trichet, J.; Valentin, C.; Rajot, J.L. Microbiotic soil crusts in the Sahel of Western Niger and their influence on soil porosity and water dynamics. Catena 2009, 77, 48–55. [Google Scholar] [CrossRef]
- Bailey, D.; Mazurak, A.P.; Rosowski, J.R. Aggregation of soil particles by algae. J. Phycol. 1973, 9, 99–101. [Google Scholar] [CrossRef]





| Strain | Cyanobacteria | Medium | 7-Week-Old Cultures | 16-Week-Old Cultures | ||
|---|---|---|---|---|---|---|
| Artemia salina Mortality (%) | Artemia salina Mortality (%) | |||||
| 24 h | 48 h | 24 h | 48 h | |||
| L1 | Chroococcidiopsis sp. | N+ | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 6.6 ± 0.6 |
| N− | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 6.8 ± 1.3 | ||
| L2 | Nostoc sp. | N+ | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 4.5 ± 3.9 |
| N− | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 5.9 ± 5.6 | ||
| L4 | Tolypothrix sp. | N+ | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 10.2 ± 3.4 |
| N− | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 16.1 ± 4.7 | ||
| L5 | Nostoc sp. | N+ | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 7.2 ± 2.6 |
| N− | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 8.5 ± 3.5 | ||
| L6 | Nostoc sp. | N+ | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 2.1 ± 3.6 |
| N− | 0.0 ± 0.0 | 2.4 ± 4.1 | 6.4 ± 0.2 | 8.5 ± 3.5 | ||
| PCC 7806 | Microcystis sp. | N+/− | 87.7 ± 5.5 | 96.0 ± 3.5 | 93.8 ± 6.3 | 97.9 ± 3.6 |
| Strain | Med. | 1st Week | 2nd Week | 3rd Week | 4th Week | 5th Week | 6th Week | 7th Week |
|---|---|---|---|---|---|---|---|---|
| L1 | N+ | 6.2 ± 1.0 | 18.1 ± 1.0 | 23.7 ± 1.7 | 31.6 ± 1.0 | 39.5 ± 1.9 | 50.8 ± 4.5 | 63.8 ± 2.6 |
| N− | 5.7 ± 1.0 | 18.6 ± 1.7 | 23.7 ± 1.7 | 34.4 ± 2.6 | 19.2 ± 1.0 | 14.1 ± 1.0 | 7.9 ± 1.0 | |
| L2 | N+ | 16.4 ± 1.0 | 50.3 ± 2.6 | 55.9 ± 2.9 | 69.4 ± 4.5 | 89.8 ± 11.9 | 112.9 ± 1.9 | 139.5 ± 4.9 |
| N− | 14.7 ± 1.0 | 40.1 ± 2.6 | 44.0 ± 2.9 | 51.9 ± 2.6 | 58.2 ± 5.2 | 77.3 ± 5.2 | 80.7 ± 2.6 | |
| L4 | N+ | 19.2 ± 1.0 | 32.7 ± 2.6 | 45.2 ± 3.9 | 71.1 ± 5.1 | 94.3 ± 6.8 | 119.1 ± 10.3 | 130.4 ± 2.9 |
| N− | 7.9 ± 1.0 | 23.2 ± 2.0 | 27.7 ± 2.0 | 39.5 ± 2.6 | 54.8 ± 4.9 | 77.3 ± 3.5 | 89.2 ± 6.0 | |
| L5 | N+ | 39.0 ± 1.7 | 138.9 ± 3.4 | 247.3 ± 19.5 | 287.9 ± 1.7 | 358.5 ± 2.6 | 394.6 ± 29.4 | 501.4 ± 25.6 |
| N− | 18.1 ± 1.0 | 37.3 ± 1.7 | 47.4 ± 1.7 | 58.7 ± 1.9 | 84.1 ± 2.6 | 83.6 ± 2.6 | 76.8 ± 6.9 | |
| L6 | N+ | 7.3 ± 1.0 | 14.1 ± 1.0 | 14.7 ± 1.0 | 20.3 ± 1.7 | 22.0 ± 1.7 | 24.3 ± 1.9 | 27.7 ± 2.6 |
| N− | 7.9 ± 1.0 | 19.8 ± 1.0 | 21.5 ± 2.0 | 27.1 ± 1.7 | 43.5 ± 3.5 | 57.6 ± 3.4 | 48.6 ± 1.0 |
| Strain | Med. | Total Polysaccharide (mg/L) | RPS (mg/L) | RPS Share (%) | Total Polysaccharide Yield (mg/g Biomass) | RPS Yield (mg/g Biomass) |
|---|---|---|---|---|---|---|
| L1 | N+ | 3.1 ± 0.3 | 1.4 ± 0.0 | 45.6 | 48.4 | 22.1 |
| N− | 1.6 ± 0.1 | 0.6 ± 0.0 | 39.3 | 196.6 | 77.4 | |
| L2 | N+ | 5.4 ± 0.3 | 1.9 ± 0.1 | 35.4 | 38.7 | 13.7 |
| N− | 3.3 ± 0.2 | 0.7 ± 0.0 | 20.9 | 41.4 | 8.7 | |
| L4 | N+ | 7.1 ± 0.6 | 2.7 ± 0.1 | 37.9 | 54.5 | 20.7 |
| N− | 1.6 ± 0.2 | 0.5 ± 0.0 | 29.3 | 18.2 | 5.3 | |
| L5 | N+ | 2.3 ± 0.2 | 1 ± 0.1 | 42.6 | 4.7 | 2.0 |
| N− | 2.9 ± 0.3 | 1.1 ± 0.0 | 37.6 | 37.1 | 13.9 | |
| L6 | N+ | 4.0 ± 0.3 | 1.7 ± 0.1 | 41.9 | 145.6 | 61.0 |
| N− | 3.7 ± 0.2 | 1.6 ± 0.1 | 44.4 | 75.9 | 33.7 |
| Parameter | Variability | SS | DF | MS | F-Value | p-Value |
|---|---|---|---|---|---|---|
| Biomass (mg/L) | Strain | 255081.4 | 4 | 63770.4 | 806.4 | <0.000001 |
| Medium composition | 93918.8 | 1 | 93918.8 | 1187.7 | <0.000001 | |
| Strain and medium composition | 189539.9 | 4 | 47384.2 | 599.2 | <0.000001 | |
| Error | 1581.5 | 20 | 79.1 | - | - | |
| RPS (mg/L) | Strain | 2.2 | 4 | 0.6 | 88.9 | <0.000001 |
| Medium composition | 5.3 | 1 | 5.3 | 853.1 | <0.000001 | |
| Strain and medium composition | 5.3 | 4 | 1.3 | 211.6 | <0.000001 | |
| Error | 0.1 | 20 | 0.0 | - | - | |
| Total polysaccharide (mg/L) | Strain | 23.1 | 4 | 5.8 | 61.0 | <0.000001 |
| Medium composition | 23.8 | 1 | 23.8 | 251.5 | <0.000001 | |
| Strain and medium composition | 31.8 | 4 | 7.9 | 84.0 | <0.000001 | |
| Error | 1.9 | 20 | 0.1 | - | - | |
| Viscosity (mPa·s) | Strain | 540.3 | 4 | 135.1 | 109.4 | <0.000001 |
| Medium composition | 0.4 | 1 | 0.4 | 0.3 | 0.573595 | |
| Strain and medium composition | 52.3 | 4 | 13.1 | 10.6 | 0.000090 | |
| Error | 24.7 | 20 | 1.2 | - | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Palanački Malešević, T.; Dulić, T.; Obreht, I.; Trivunović, Z.; Marković, R.; Kostić, B.; Važić, T.; Meriluoto, J.; Svirčev, Z. Cyanobacterial Potential for Restoration of Loess Surfaces through Artificially Induced Biocrusts. Appl. Sci. 2021, 11, 66. https://doi.org/10.3390/app11010066
Palanački Malešević T, Dulić T, Obreht I, Trivunović Z, Marković R, Kostić B, Važić T, Meriluoto J, Svirčev Z. Cyanobacterial Potential for Restoration of Loess Surfaces through Artificially Induced Biocrusts. Applied Sciences. 2021; 11(1):66. https://doi.org/10.3390/app11010066
Chicago/Turabian StylePalanački Malešević, Tamara, Tamara Dulić, Igor Obreht, Zorana Trivunović, Rastko Marković, Bojan Kostić, Tamara Važić, Jussi Meriluoto, and Zorica Svirčev. 2021. "Cyanobacterial Potential for Restoration of Loess Surfaces through Artificially Induced Biocrusts" Applied Sciences 11, no. 1: 66. https://doi.org/10.3390/app11010066
APA StylePalanački Malešević, T., Dulić, T., Obreht, I., Trivunović, Z., Marković, R., Kostić, B., Važić, T., Meriluoto, J., & Svirčev, Z. (2021). Cyanobacterial Potential for Restoration of Loess Surfaces through Artificially Induced Biocrusts. Applied Sciences, 11(1), 66. https://doi.org/10.3390/app11010066







