Abstract
Thraustochytrids have gained increasing relevance over the last decades, due to their fast growth and outstanding capacity to accumulate polyunsaturated fatty acids (PUFAs), particularly docosahexaenoic acid (DHA). In this context, the present work aimed to optimize the growth performance and DHA yields by improving the culture medium of Aurantiochytrium sp. AF0043. Accordingly, two distinct culture media were optimized: (i) an inorganic optimized medium (IOM), containing only monosodium glutamate and glucose as nitrogen and carbon sources, respectively; and (ii) an organic and sustainable waste-based optimized medium (WOM), containing corn steep powder and glycerol, added in fed-batch mode, as nitrogen and carbon sources, respectively. Overall, the lab-scale optimization allowed to increase the biomass yield 1.5-fold and enhance DHA content 1.7-fold using IOM. Moreover, WOM enabled a 2-fold increase in biomass yield and a significant improvement in lipid contents, from 22.78% to 31.14%. However, DHA content was enhanced almost 3-fold, from an initial content of 10.12% to 29.66% of total fatty acids contained in the biomass. Therefore, these results strongly suggest, not only that the production pipeline was significantly improved but also confirmed the potential use of Aurantiochytrium sp. AF0043 as a source of DHA.
1. Introduction
Thraustochytrids are unicellular eukaryotic protists belonging to the superphylum Heterokonta (also known as Stramenopiles) and more specifically to the heterotrophic phylum Bigyra and the class Labyrinthulea [1]. Thraustochytrids are associated with a wide range of biotechnological applications, which triggered their industrial production for different high-value markets [2,3,4]. From a biotechnological point of view, two main features are responsible for the increasing interest in this family of microorganisms. They display high growth performance, reaching very high cell concentrations in a few days, and exhibit the striking ability to produce and accumulate docosahexaenoic acid (DHA) [4,5,6]. In contrast with the majority of microorganisms that produce saturated fatty acids (FA) as energy storage lipids, thraustochytrids synthesize long chain-polyunsaturated fatty acids (LC-PUFAs), like DHA, as energy storage, by an alternative pathway catalyzed by the polyketide synthase (PKS) enzymatic complex [5,7]. Usually, besides DHA, they also produce docosapentaenoic acid (DPA), another typical LC-PUFA in thraustochytrids [4,5].
DHA belongs to a class of LC-PUFA often called “omega-3 fatty acids”, which are in high demand due to the many known benefits in nutrition. DHA is also widely known for its fundamental role in preventing cardiovascular diseases, being essential for brain and eye development (particularly in children), learning capacities, as well as for the prevention of neurodegenerative conditions, arthritis, asthma and skin diseases [7,8,9,10,11,12,13,14,15,16,17,18,19,20]. In addition, DHA plays a key role in membrane fluidity, cell signaling, cell interactions and in the development of the immune system and inflammatory responses [21,22,23].
Although DHA is well established in the food, feed, nutraceutical, and dietary supplement markets, it has been sold mainly in the form of fish oil, of which 70% is currently used for aquafeeds [3,5]. Nevertheless, several sustainability problems are strongly related to fish oil, namely overfishing and chemical products used in the extraction of this oil [4,7,14,17,20,24]. In addition, fish oil is associated with an unpleasant smell, taste, low DHA amount, and possible presence of harmful contaminants, due to ocean pollution and bioaccumulation of heavy metals, dioxins and polychlorinated biphenyls [7,14,16,17,20,24,25]. Moreover, the increasing need for food and the spread of aquaculture worldwide will also require higher amounts of these oils for aquafeed. In addition, the growing demand for DHA from a non-animal origin created a gap in the market, which could be fulfilled by DHA of microbial origin, with particular relevance in the vegan market.
In this context, the present work reports the optimization of lab-scale culture media of Aurantiochytrium sp. AF0043 for the purpose of maximizing biomass, lipid and DHA yields. In addition, the impact of the different conditions on the biochemical composition, in particular on the proximate composition and FAME profile, was evaluated.
2. Materials and Methods
2.1. Thraustochytrid Strain
The thraustochytrid strain used in this work, Aurantiochytrium sp. AF0043, was obtained from Allmicroalgae’s culture collection. The strain was cryopreserved under liquid nitrogen and an aliquot was taken when necessary.
2.2. Experimental Trials
All laboratory experiments were performed with a final volume of 50 mL, using 250-mL Erlenmeyer flasks with caps of cotton and gaze (unless otherwise stated). After inoculation, the Erlenmeyer flasks were placed in an orbital incubator (ArgoLab® shaker SKI 4, Carpi, Italy), at 28 °C and a mixing speed of 150 rpm. All experiments were carried out in triplicate.
In all trials, samples were taken daily and the growth, pH, and glucose concentration were monitored. At the end of each assay, samples were centrifuged at 4500× g for 15 min (Hermle® Z300 centrifuge, Gosheim, Germany) and the pellet was frozen at −20 °C for biochemical analysis.
2.2.1. Effect of Basal Culture Medium and Trace Metals Solution
A standard heterotrophic medium, MB10, was used as a starting point for the optimization of the culture medium. In the first trial, this basal medium was compared to MT1, a culture medium that was designed in accordance with the information available in the literature [2,6,10,11,14,17,20,26,27,28,29,30,31,32,33,34]. The MT1 medium was tested using two trace metals solutions: (i) TM1, the trace metals solution of MB10; and (ii) TM2, formulated based on previous results obtained in the literature [6,8,10,14,16,17,20,24,26,27,32,33,34,35].
MB10 was composed of 9.0 g/L K2HPO4, 8.7 g/L NaH2PO4.H2O, 5.0 g/L MgSO4.7H2O, 1.2 g/L citric acid, 6.3 g/L ammonium sulfate, 0.3 g/L CaCl2, 2.0 g/L (monosodium) glutamate and 15 g/L NaCl, supplemented with 20 mL/L of TM1 and 1.5 mL of vitamins solution (VIT).
MT1 was formulated to contain 4 g/L K2HPO4, 2 g/L MgSO4.7H2O, 0.8 g/L of ammonium sulfate, 0.1 g/L CaCl2, 20 g/L (monosodium) glutamate, 0.5 g/L of KCl, 10 g/L NaSO4 and 4 g/L of yeast extract and was further supplemented with 20 mL/L of TM1 or TM2 and 1.5 mL of VIT1.
The composition of TM1 was as follows: 0.1 g/L CuSO4.5H2O, 3.3 g/L H3BO3, 14 g/L ZnSO4.7H2O, 8.1 g/L MnSO4.H2O, 0.39 g/L Na2MoO4.2H2O, 0.13 g/L NiCl2.6H2O, 33 g/L monohydrated citric acid and 1.1 g/L FeSO4.7H2O. TM2 was formulated to contain 0.6 g/L CuSO4.5H2O, 0.8 g/L ZnSO4.7H2O, 0.86 g/L MnCl2.4H2O, 0.01 g/L Na2MoO4.2H2O, 0.06 g/L NiSO4.6H2O, 0.29 g/L FeSO4.7H2O, 0.01 g/L CoCl2.6H2O and 6 g/L of disodium EDTA.
VIT1 was composed of 1 g/L thiamine-HCl (vitamin B1), 0.015 g/L d-biotin (vitamin B7), 0.012 g/L cyanocobalamin (vitamin B12), 0.030 g/L pantothenate (vitamin B5) and 0.060 g/L p-aminobenzoic acid (PABA).
The carbon source was a glucose stock solution at 50% w/v, which was given in order to obtain a final concentration of 50 g/L of glucose in the culture’s medium in all treatments.
Henceforth, MT1 supplemented with TM1 and VIT1 and glucose at 50 g/L was the medium used in the following assays unless stated otherwise.
2.2.2. C Source Optimization
For the optimization of the carbon source, two carbon sources were tested individually and combined at different concentrations with the whole MT1 medium, using the following treatments: (i) 50 g/L glucose, (ii) 100 g/L glucose, (iii) 50 g/L glycerol, (iv) 100 g/L glycerol and, (v) 25 g/L glucose + 25 g/L glycerol.
2.2.3. N Source Optimization
For nitrogen optimization, each N source of the original MT1 medium, namely yeast extract, glutamate and ammonium sulphate, was tested individually at a concentration of 150 mM N, using MT1 medium without any N source. The whole MT1 medium containing the three N sources was used as control.
Thereafter, a second assay, considering waste-based alternative N sources, was performed. Accordingly, cheese whey, corn steep powder and an organic liquid medium made from fertilizers and agricultural wastes (MNBIO) were tested at a concentration of 150 mM N, using MT1 medium without N. The whole MT1 medium was used as control. All treatments were supplemented with TM1 and VIT1 and 50 g/L glucose.
2.2.4. Non-Optimized vs. Optimized Conditions
Finally, in order to validate the results obtained, a final trial was conducted in baffled Erlenmeyer flasks, comparing the initial culture medium (MB10) with two culture media formulated according to the results previously obtained, namely: (i) an inorganic optimized medium (IOM), composed of MT1 containing only glutamate as N source and 200 g/L glucose as C source; and (ii) an organic waste-based optimized medium (WOM), with MT1 containing corn steep powder as N source and 50 g/L glycerol as C source (fed-batch). The conditions tested in this assay are summarized below, in Table 1.

Table 1.
Conditions of the final assay comparing the initial and optimized conditions with strain AF0043.
2.3. Growth Assessment
Cultures were analyzed daily by optical density measurement at 600 nm (OD600), in a Zuzi® spectrophotometer (Navarra, Spain) and microscopic observations (Zeiss AX10®, Oberkochen, Germany). To determine biomass dry weight (DW), samples were filtered through 0.45-µm cellulose filters (VWR, Amadora, Portugal), and washed with the same volume of an ammonium formate solution (61 g/L). Finally, they were dried at 120 °C in a Radwag® MA50.R moisture analyzer (Radom, Poland). The DW was calculated as represented in Equation (1).
A correlation between DW and OD was obtained particularly for this strain as it is represented by Equation (2):
Biomass productivity (P) was obtained by Equation (3) and specific growth rate (µ) by Equation (4), where DWf and DWi corresponded to the final and initial dry weights taken at the time points tf and ti, respectively:
The maximum specific growth rate (µmax) was calculated using Equation (4), but solely for the time interval of the exponential phase, i.e., considering the biomass concentration at the beginning and end of the exponential phase.
2.4. pH Control
pH was measured with pH meter 110 (VWR, Amadora, Portugal) and pH stripes (Labbox, Barcelona, Spain). Cultures were maintained at a pH of about 6.5 and adjusted when necessary with 2M HCl or 2M NaOH, sterilized by 0.2-µm vacuum filtration (VWR, Amadora, Portugal).
2.5. Determination of Glucose Concentration
For glucose quantification, samples were centrifuged for 10 min at 2000 g (VWR® MiniStar Silverline microcentrifuge, Amadora, Portugal). The supernatant was diluted using 0.9% NaCl (w/v) to measure glucose’s concentration in a YSI 2950D Biochemistry Analyzer (Xylem Inc., Yellow Springs, OH, USA).
2.6. Biochemical Composition
All frozen samples were freeze-dried in a Telstar LyoAlfa 15 (Barcarena, Portugal) and stored in a desiccator, prior to all biochemical analysis.
2.6.1. Proximate Composition
Determination of Total Lipid Content
The total lipid content was determined according to a modified Bligh and Dyer [36] method, reported by Pereira et al. [37]. Briefly, 10 mg of dried biomass was extracted with a mixture of MeOH:CHCl3:H2O (2:2:1) and homogenized with an Ultra-Turrax disperser (IKA, Staufen, Germany), for 2 min. Thereafter, the mixture was centrifuged at 2500× g, for 10 min, to separate phases, and the chloroform phase was transferred to pre-weighed tubes and dried overnight (60 °C). The final weight of the tubes was determined, which allowed one to estimate the percentage of lipids by gravimetry.
Determination of Ash Content
Ash content was determined by burning biomass samples for 8 h, at 550 °C, in a muffle furnace (J. P. Selecta, Sel horn R9-L, Barcelona, Spain). The final weight was determined, and ash content was calculated by the initial and final weight difference.
Determination of Protein and Carbohydrate Contents
In accordance with the procedure provided by the manufacturer, C, H and N elemental analysis was carried out in a Vario EL®, Elementar Analyser System (GmbH, Hanau, Germany). The protein content was then estimated by multiplying the nitrogen content, as determined by the elemental analyzer, by a 6.25 factor [38].
The carbohydrate content of each sample was estimated by the difference of the sum of ash, lipid and protein contents.
2.6.2. Fatty Acid Profile
The fatty acid profile was determined according to a modified Lepage and Roy [39] protocol, reported by Pereira et al. [40]. Biomass samples were homogenized with an IKA Ultra-Turrax T10B disperser for 2 min in a solution of methanol and acetyl chloride (20:1, v/v). Thereafter, the reaction vessels were placed in a bath for 60 min at 90 °C. Following the derivatization procedure, the organic phase was sequentially extracted four times with n-hexane. In order to dry the extracts, anhydrous sodium sulphate was added and later filtered with a 0.2-µm filter (Whatman® Puradisc, PTFE). Extracts were then evaporated with a gentle stream of nitrogen and the residue was resuspended in GC grade hexane and stored at −20 °C.
Samples were analyzed by GC-MS (Bruker SCION 456 TQ GC/MS, Austin, TX, USA), using a ZB-5MS and helium as the carrier gas. Samples were injected at 300 °C and submitted to a temperature cycle: A first temperature increase starting at 60 °C, with an increment of 30 °C per min until 120 °C, a second heating phase that increased 5 °C per min until 250 °C, and a final gradient of 20 °C ascent per min until 300 °C were reached. Supelco® 37 FAME mix (Sigma-Aldrich, Sintra, Portugal) was used as standard.
2.7. Statistical Analysis
Statistical analysis was performed by analysis of variance (ANOVA), followed by post-hoc Tukey-HSD test using R software (version 3.6.1). All experiments were performed with three biological replicates, and results were expressed as mean ± standard deviation.
3. Results
3.1. Effect of Different Medium and Trace Metals Composition on Growth
The optimization of the culture medium started with a standard growth medium (MB10) that was tested against MT1 medium using two distinct trace metal solutions (TM1 and TM2; Figure 1).
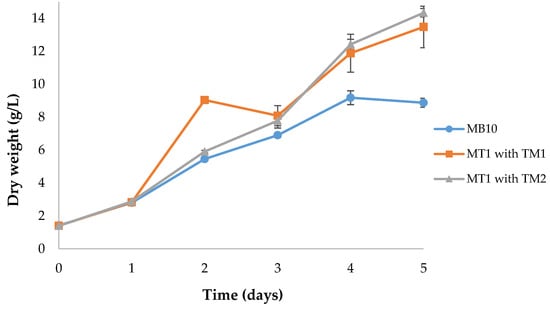
Figure 1.
Batch growth of Aurantiochytrium sp. AF0043, in MB10 and MT1, supplemented with two distinct trace metal solutions (TM1 and TM2). Error bars represent the standard deviation between three biological replicates.
The results obtained show that cultures grown in the MB10 medium reached a maximum biomass concentration of 9.18 g/L, which is significantly lower than the growth verified in MT1 media supplemented with both trace metal solutions (p < 0.05; Figure 1). MT1 supplemented with TM1 and TM2 did not show any significant differences in biomass concentrations among them (p > 0.05), reaching 13.47 and 14.33 g/L, respectively. Accordingly, the MT1 medium allowed a 1.6-fold increase in the biomass productivity from 1.49 to 2.42–2.59 g/L/day, and the specific growth rate was improved from 0.37 to 0.45–0.47 day−1.
3.2. Carbon Source Optimization
In order to evaluate the effect of different carbon sources and their concentration, glycerol was tested and compared to glucose, individually and in combination as a carbon source to grow Aurantiochytrium sp. (Figure 2)
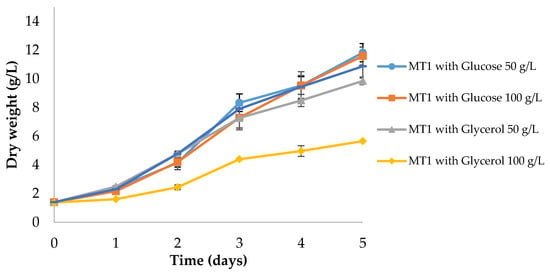
Figure 2.
Batch growth of Aurantiochytrium sp. AF0043 strain in MT1 culture medium with different carbon sources and concentrations: 50 g/L glucose, 100 g/L glucose, 50 g/L glycerol, 100 g/L glycerol and 25 g/L glucose + 25 g/L glycerol. Error bars represent the standard deviation of three biological replicates.
All treatments displayed similar growth performances, reaching a final DW around 10.88–11.83 g/L at day 5 (p > 0.05; Figure 2). The exceptions were the cultures grown with 50 and 100 g/L of glycerol, which attained, respectively, 9.86 and 5.66 g/L of DW, which indicates that increasing glycerol concentrations inhibit the growth of this strain. Overall, biomass productivities between 1.89 and 2.08 g/L/day and growth rates between 0.39–0.43 day−1 were obtained.
Glucose was not fully consumed by day 5 in any of the conditions (data not shown). However, glucose was consumed at a much faster rate when glucose was the sole carbon source; conversely, when a mix of carbon sources was employed (glucose and glycerol; Figure 2), glucose remained almost constant throughout the growth period, indicating a preference of Aurantiochytrium sp. for glycerol over glucose.
3.3. N Source Optimization
MT1 medium has three N sources in its composition: glutamate, ammonium sulphate and yeast extract. Since glutamate and yeast extract are industrially expensive and the latter provides higher contamination risks, the possibility of excluding it from the medium was assessed (Figure 3A). In addition, alternative waste-based organic N sources were tested (Figure 3B).
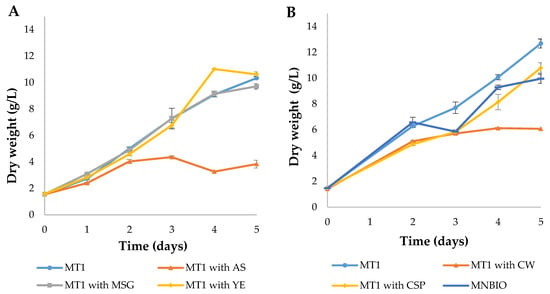
Figure 3.
Batch growth of Aurantiochytrium sp. AF0043 strain, with different N sources, at a normalized N concentration of 150 mM. (A) MT1 with ammonium sulfate (AS), monosodium glutamate (MSG) and yeast extract (YE). (B) MT1 with cheese whey (CW), corn steep powder (CSP) and an organic liquid medium made from fertilizers and agricultural wastes (MNBIO). Error bars represent the standard deviation between three biological replicates.
In the first trial of N optimization, cultures grown in MT1 containing either yeast extract or glutamate reached similar productivities (between 1.63–1.81 g/L/d), maximum DW (between 9.71–11.02 g/L) and growth rates (0.37–0.38 day−1) compared to those obtained with the whole MT1 medium (p > 0.05). On the other hand, MT1 medium containing only ammonium sulphate led to significantly lower growth performance of Aurantiochytrium sp., as compared to those observed when cells were grown on other N sources (p < 0.05). This difference is probably related to the abrupt pH decrease associated with ammonium sulphate (data not shown).
Regarding the alternative N sources tested (Figure 3B), all treatments displayed significant differences among each other (p < 0.05). The highest growth performance of Aurantiochytrium sp. cultures was obtained with the control medium (MT1), which achieved a maximum concentration of 12.68 g/L DW after 5 days. Cells grown on corn steep powder attained the second-highest concentration, 10.79 g/L of DW, whereas MNBIO was the third most productive, reaching a final DW of 9.95 g/L. Cultures grown in cheese whey displayed the lowest growth performance that only reached 6.12 g/L of biomass DW.
3.4. Non-Optimized vs. Optimized Conditions
In order to validate the optimization carried out in the previous trials, MB10 medium with ammonium sulphate and glutamate and glucose was compared to: (i) an inorganic optimized medium (IOM), MT1 containing only glutamate and glucose; and (ii) an organic and waste-based optimized medium (WOM), with MT1 containing corn steep powder and glycerol (fed-batch; Figure 4).
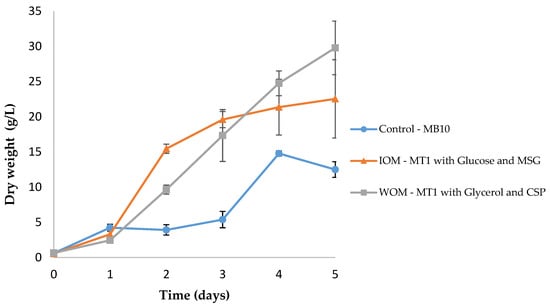
Figure 4.
Batch growth of Aurantiochytrium sp. AF0043, with MB10 medium (control), inorganic optimized medium (IOM), and waste-based optimized medium (WOM). Error bars represent the standard deviation between three biological replicates.
The results obtained revealed that the starting medium, MB10, led to the lowest maximum concentration (14.78 g/L), biomass productivity (3.54 g/L/day) and growth rate (0.60 day−1) of Aurantiochytrium sp. (p < 0.05). The optimized culture media, IOM and WOM, improved respectively 1.5 and 2-fold the growth performance of Aurantiochytrium sp. in terms of maximum concentration when compared to the starting medium. In addition, the biomass productivity increased 0.7 and 1.6-fold and the growth rate 1.2 and 1.7-fold, respectively. Interestingly, the cheapest and most sustainable medium (WOM) reached a maximum concentration of 29.78 g/L of DW.
3.5. Biochemical Composition of Biomass
In this trial, the quality of the biomass obtained was compared regarding proximate composition (Table 2) and FAME profile (Table 3), in order to identify the effect of the optimization on the biochemical composition of Aurantiochytrium sp.

Table 2.
Proximate composition of Aurantiochytrium sp. AF0043 biomass strain cultures, grown in MB10 medium (control), inorganic optimized medium (IOM), and waste-based optimized medium (WOM). Values are the mean and corresponding standard deviation of three biological replicates. Different letters indicate significant differences between biomass grown in different media.

Table 3.
Fatty acid methyl esters profile of Aurantiochytrium sp. AF0043 cultures, grown in MB10 medium (control), inorganic optimized medium (IOM), and waste-based optimized medium (WOM). Values are given as the mean ± standard deviation percentage of the total FAME detected.
The biomass grown with standard growth medium (MB10) presented the highest protein content (24% of DW), followed by the WOM (21% of DW), while IOM led to the lowest protein content (14% of DW) (p < 0.05). Regarding total lipids, Aurantiochytrium sp. grown in WOM presented the highest values, 31% of DW, compared to 23 and 15% of DW in MB10 and IOM, respectively (p < 0.05). The carbohydrate content was significantly higher in cultures grown in IOM (59% of DW) (p < 0.05), than in MB10 (28% of DW) and WOM (33% of DW). MB10 promoted the highest ash content in Aurantiochytrium sp. cultures, 26% of DW (p < 0.05), while IOM- and WOM-grown cells presented similar ash values of 12 and 15% of DW, respectively.
The FAME profile (Table 3) of Aurantiochytrium sp. was mainly composed of myristic (C14:0), palmitic (C16:0), docosapentaenoic (C22:5 n-3, DPA) and docosahexaenoic (C22:6 n-3, DHA) acids. Other FAs common in this species were also detected in minor abundances, such as pentadecanoic (C15:0), heptadecanoic (C17:0) and stearic (C18:0) acids.
When the different media are compared among each other, it is clear that MB10 led to a higher amount of saturated fatty acids (SFA) in the biomass of Aurantiochytrium sp., 87.65%, mainly hexadecanoic acid (C16:0) and a lower amount of DHA, which accounted for 10.12% of total FA. On the other hand, both IOM and WOM led to a steep increase in the contents of DHA and to lower contents of SFA in the final biomass. Overall, the optimization allowed a significant improvement in the DHA content of produced biomass, from 10.12% to 17.05% and 29.66% with IOM and WOM, respectively.
4. Discussion
The optimization of the culture medium is one of the most important parameters to obtain concentrated cultures with high DHA amount of Aurantiochytrium sp., and the lab-scale optimization carried out in this work allowed significant improvements that can be key to industrial cultivation.
Aurantiochytrium sp. was first cultivated in MB10, as it is the stock medium used in-house for heterotrophic cultivation of other microorganisms. Nevertheless, in this strain, the preliminary growth using this medium was unsatisfactory, given its low growth rates and biomass productivity. As MB10 contains NaCl, the presence of chloride ions in the medium was detected. This is undesired, because such ions are corrosive, affecting the operation of industrial fermentation vessels. Thus, to prevent this, the inclusion of Cl must be avoided in the industrial medium formulation [30,34]. However, most thraustochytrids require sea salts for growing, which are essential for lipid synthesis and osmotic adjustment [26,30,34]. Therefore, to adjust the medium to the species under study and bearing in mind the aim of maintaining its industrial feasibility, an extensive literature review was performed, focusing on the macronutrients, trace metals, vitamins and N sources of the culture medium. This search resulted in the MT1 medium, which was formulated taking into account the best growth performance and DHA accumulation. In MT1, Na2SO4 is the largest contributor to the medium salinity, eliminating chlorine ions from the medium without compromising the organism’s growth or DHA production. In fact, Chen et al. [30] found out that the replacement of sea salt (2% w/v) by Na2SO4 (1% w/v) improved the maximum concentration of biomass from 35 to around 39 g/L, while DHA yield (7.95 g/L) increased from 40% to 44% of the total lipids in the thraustochytrid Schizochytrium sp., corroborating the feasibility of NaCl replacement. Similarly, the results obtained in the first trial revealed that MT1 supplemented with TM1 outperformed MB10 and was further used for the whole optimization process. After these significant improvements in the formulation of macronutrients and trace metals, the next step was to optimize the carbon source and its concentration.
In the present study, Aurantiochytrium sp. was grown using glucose and glycerol independently and in combination as the C-source. Glucose is the most used substrate in heterotrophic cultivation, since it displays, generally, higher growth rates and biomass yields in most heterotrophic microorganisms, including thraustochytrids [25]. Nevertheless, alternative carbon sources, such as glycerol, must be pursued particularly from waste streams, with the goal of reducing production costs [25]. Glycerol is a carbon source of excellence that has received attention in the biotechnology field due to its abundance as a cheap by-product of biodiesel industries [25,35,41]. Moreover, glycerol used as a feedstock in heterotrophic growth maintains the food-grade status of the produced biomass and sub-products [4,41]. Thraustochytrids, such as Aurantiochytrium, have been reported as being able to utilize glycerol as a carbon source, obtaining comparable and even higher biomass and DHA yields [35,41]. Nevertheless, when 100 g/L of glycerol was tested, growth was inhibited. In fact, inhibition at glycerol concentrations above 50 g/L was already reported for Aurantiochytrium limacinum [42].
The same philosophy was used to evaluate the impact of different N sources in the growth of Aurantiochytrium sp. In the first N optimization trial, three traditionally used N sources present in MT1 were evaluated independently, namely yeast extract, ammonium sulphate and glutamate. Results showed that both yeast extract and glutamate displayed similar growth results to the whole MT1 medium. These results unveiled the larger contribution of glutamate for growth and the possibility of using it as sole N source. This is a highly interesting finding because that eliminates the need of adding yeast extract, reducing medium costs. Accordingly, glutamate is often used as N source in thraustochytrid-based DHA production, since it accelerates growth and promotes DHA accumulation [2,7,11]. Interestingly, Wu et al. [21] attained slightly higher biomass, lipid and DHA content with glutamate, rather than with yeast extract. In a similar experiment, Ren et al. [10] reported glutamate as being able to promote growth up to higher biomass concentration; however, a right balance should be found, as N depletion is a known trigger of lipid accumulation, and excess N might be detrimental to DHA production [2,4,7].
Regarding alternative and more sustainable N sources, corn steep powder displayed a greater potential for the improved growth of Aurantiochytrium sp., when compared to cheese whey and MNBIO. High fructose corn syrup or corn steep liquor/powder (CSP) is a co-product of corn wet milling process, a well-reported agro-waste often used as a sugar substitute, due to its low cost and high solubility [43]. Apart from its elevated fermentable sugar content, it contains between 35%–45% of protein, being both an N and C source. Interestingly, Yokochi et al. [44] obtained a Schizochytrium limacinum biomass yield of 15 g/L DW using yeast extract and CSP, but a higher lipid content and twice as much DHA yield (1.7 g/L) was achieved with CSP, rather than with yeast extract. In this assay, the cultures using CSP as N source reached around 12 g/L DW. Bailey et al. [34] also recommended 2–10 g/L CSP in fed-batch mode, in order to obtain high-density cultures and high DHA productivities. On the other hand, Chen et al. [32] reported the lowest Aurantiochytrium sp. productivity and maximum biomass concentration with CSP, when compared to yeast extract, peptone and tryptone. Therefore, CSP is an interesting N source for thraustochytrid-based DHA fermentation, but its utilization and effectiveness seem to be strain-dependent.
To validate the whole optimization process, an inorganic and a waste-based optimized culture media, IOM and WOM, respectively, were established and validated in a final trial. IOM exhibited a much higher growth rate from the first to the second day (1.45 day−1) than the other conditions. This behavior might be related to N exhaustion. On the other hand, WOM led to 31.14% lipid content, a 1.4-fold increase compared to the initial conditions. It is worth noting that CSP and glycerol seem to be suitable alternatives to replace the commonly used glutamate and glucose, since they not only allowed a lipid yield increase, but also enhanced DHA production. Yu et al. [43] also reported high-fructose corn syrup as a possible option for Aurantiochytrium sp. DHA fermentation, reaching final concentration values of up to 78.5 g/L DW and 51 g/L lipid yields, with a DHA content of 46.4%.
Comparing the Aurantiochytrium strain studied here as a DHA feedstock with other thraustochytrids described in the literature is a complex proposition. Indeed, the genus Schizochytrium was amended leading to the introduction of two more genera, Aurantiochytrium and Oblongichytrium [45]. This resulted in the distribution of preexisting commercial strains among the three genera, based on their disparate phylogenetics, morphological features, as well as fatty acid and carotenoid profiles [8,45]. Currently, Schizochytrium sp. is often chosen for industrial production of DHA, since it has been far more studied and optimized, resulting in higher biomass productivities than Aurantiochytrium and other strains [20,33,34]. However, Aurantiochytrium sp. has the potential of accumulating higher amounts of DHA (up to 80%) and further optimization is thus possible in the near future, even with the use of growth media that can be labelled as “organic”. In addition, Aurantiochytrium is able to produce other high-value pigments, such as astaxanthin and β-carotene, which can further increase the commercial value of the biomass produced [8,45]. Taken together, the genus Aurantiochytrium will thus be the target of further research to yield further optimization concerning their cultivation in low-cost “organic” media.
Concerning the FAME profiles obtained in this study, they are in accordance with those previously described in the literature for Aurantiochytrium sp., with particularly high contents of myristic, palmitic, DPA and DHA [11,42,46,47] (Table 3). Interestingly, in the final validation trial, although WOM and IOM led to similar growth performances, higher biomass lipid and DHA contents were obtained in WOM. Similarly, Chang et al. [35] obtained around 55 g/L DW and total FAME around 33% with 40 g/L glucose or glycerol and posterior feeding, while DHA concentration was higher with glycerol, 48.4% of total lipids, than with glucose (43.2%).
5. Conclusions
The lab-scale optimization of Aurantiochytrium sp. AF0043 culture medium was successfully achieved and led to the establishment of two culture media, IOM and WOM. IOM allowed the biomass yield to increase 1.5-fold and enhance DHA content 1.7-fold. Furthermore, an alternative waste-based medium (WOM) doubled the biomass yield, achieving a maximum of 29.78 g DW/L and further enhancing the lipid and DHA contents to 31.14% and 29.66%, respectively. This optimization process provided significant improvements regarding biomass, lipid and DHA yields and unveiled great potential for the scale-up of Aurantiochytrium sp. AF0043 to pilot and industrial scales, particularly if the cost cuts provided by the use of wastes as feedstocks are taken into account.
Author Contributions
M.T. performed the experiments. H.P., M.C., J.V. and J.S. designed the experiments. M.T., M.C., A.B. and H.P. wrote the manuscript. A.M., A.B., M.S., B.C. and J.T.S. contributed to the interpretation of results, statistical analysis and preparation of figures and tables. J.S. and J.V. contributed with funding. All authors have read and agreed to the published version of the manuscript.
Funding
This work has received funding under the project AlgaValor, from the Portugal 2020 program (grant agreement n° POCI-01-0247-FEDER-035234; LISBOA-01-0247-FEDER-035234; ALG-01-0247-FEDER-035234) and Portuguese national funds from FCT—Foundation for Science and Technology, through project UIDB/04326/2020.
Acknowledgments
The authors would like to acknowledge all members of Allmicroalgae Natural Products S.A. for their kind support and help throughout this work.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Ruggiero, M.A.; Gordon, D.P.; Orrell, T.M.; Bailly, N.; Bourgoin, T.; Brusca, R.C.; Cavalier-Smith, T.; Guiry, M.; Kirk, P.M. A higher level classification of all living organisms. PLoS ONE 2015, 10, 54. [Google Scholar]
- Jakobsen, A.N.; Aasen, I.M.; Josefsen, K.D.; Strøm, A.R. Accumulation of docosahexaenoic acid-rich lipid in thraustochytrid Aurantiochytrium sp. strain T66: Effects of N and P starvation and O2 limitation. Appl. Microbiol. Biotechnol. 2008, 80, 297. [Google Scholar] [CrossRef] [PubMed]
- Leyland, B.; Leu, S.; Boussiba, S. Are thraustochytrids algae? Fungal Biol. 2017, 121, 835–840. [Google Scholar] [CrossRef] [PubMed]
- Marchan, L.F.; Chang, K.J.L.; Nichols, P.D.; Mitchell, W.J.; Polglase, J.L.; Gutierrez, T. Taxonomy, ecology and biotechnological applications of thraustochytrids: A review. Biotechnol. Adv. 2018, 36, 26–46. [Google Scholar] [CrossRef]
- Aasen, I.M.; Ertesvag, H.; Heggeset, T.M.B.; Liu, B.; Brautaset, T.; Vadstein, O.; Ellingsen, T.E. Thraustochytrids as production organisms for docosahexaenoic acid (DHA), squalene, and carotenoids. Appl. Microbiol. Biotechnol. 2016, 100, 4309–4321. [Google Scholar] [CrossRef]
- Guo, D.S.; Ji, X.J.; Ren, L.J.; Li, G.L.; Yin, F.W.; Huang, H. Development of a real-time bioprocess monitoring method for docosahexaenoic acid production by Schizochytrium sp. Bioresour. Technol. 2016, 216, 422–427. [Google Scholar] [CrossRef]
- Gupta, A.; Barrow, C.J.; Puri, M. Omega-3 biotechnology: Thraustochytrids as a novel source of omega-3 oils. Biotechnol. Adv. 2012, 30, 1733–1745. [Google Scholar] [CrossRef]
- Ren, L.J.; Ji, X.J.; Huang, H.; Qu, L.A.; Feng, Y.; Tong, Q.Q.; Ouyang, P.K. Development of a stepwise aeration control strategy for efficient docosahexaenoic acid production by Schizochytrium sp. Appl. Microbiol. Biotechnol. 2010, 87, 1649–1656. [Google Scholar] [CrossRef]
- Richardson, A.J.; Burton, J.R.; Sewell, R.P.; Spreckelsen, T.F.; Montgomery, P. Docosahexaenoic acid for reading, cognition and behavior in children aged 7–9 years: A randomized, controlled trial (The DOLAB Study). PLoS ONE 2012, 7, 14. [Google Scholar] [CrossRef]
- Ren, L.J.; Sun, L.N.; Zhuang, X.Y.; Qu, L.; Ji, X.J.; Huang, H. Regulation of docosahexaenoic acid production by Schizochytrium sp.: Effect of nitrogen addition. Bioprocess Biosyst. Eng. 2014, 37, 865–872. [Google Scholar] [CrossRef]
- Ling, X.P.; Guo, J.; Liu, X.T.; Zhang, X.; Wang, N.; Lu, Y.H.; Ng, I.S. Impact of carbon and nitrogen feeding strategy on high production of biomass and docosahexaenoic acid (DHA) by Schizochytrium sp. LU310. Bioresour. Technol. 2015, 184, 139–147. [Google Scholar] [CrossRef] [PubMed]
- Allaire, J.; Harris, W.S.; Vors, C.; Charest, A.; Marin, J.; Jackson, K.H.; Tchernofd, A.; Couturea, P.; Lamarchea, B. Supplementation with high-dose docosahexaenoic acid increases the Omega-3 Index more than high-dose eicosapentaenoic acid. Prostaglandins Leukot. Essent. Fatty Acids 2017, 120, 8–14. [Google Scholar] [CrossRef] [PubMed]
- Hadley, K.B.; Bauer, J.; Milgram, N.W. The oil-rich alga Schizochytrium sp. as a dietary source of docosahexaenoic acid improves shape discrimination learning associated with visual processing in a canine model of senescence. Prostaglandins Leukot. Essent. Fatty Acids 2017, 118, 10–18. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ren, L.J.; Sun, X.M.; Ji, X.J.; Chen, S.L.; Guo, D.S.; Huang, H. Enhancement of docosahexaenoic acid synthesis by manipulation of antioxidant capacity and prevention of oxidative damage in Schizochytrium sp. Bioresour. Technol. 2017, 223, 141–148. [Google Scholar] [CrossRef] [PubMed]
- Chang, J.P.C.; Su, K.P.; Mondelli, V.; Pariante, C.M. Omega-3 polyunsaturated fatty acids in youths with attention deficit hyperactivity disorder: A systematic review and meta- analysis of clinical trials and biological studies. Neuropsychopharmacology 2018, 43, 534–545. [Google Scholar] [CrossRef]
- Hoang, L.A.T.; Nguyen, H.C.; Le, T.T.; Hoang, T.H.Q.; Pham, V.N.; Hoang, M.H.T.; Ngo, H.T.T.; Hong, D.D. Different fermentation strategies by Schizochytrium mangrovei strain pq6 to produce feedstock for exploitation of squalene and omega-3 fatty acids. J. Phycol. 2018, 54, 550–556. [Google Scholar] [CrossRef]
- Guo, D.S.; Ji, X.J.; Ren, L.J.; Li, G.L.; Sun, X.M.; Chen, K.Q.; Gao, S.; Huang, H. Development of a scale-up strategy for fermentative production of docosahexaenoic acid by Schizochytrium sp. Chem. Eng. Sci. 2018, 176, 600–608. [Google Scholar] [CrossRef]
- Da Silva, P.S.; Mediano, M.F.F.; da Silva, G.M.S.; de Brito, P.D.; Cardoso, C.S.D.; de Almeida, C.F.; Sangenis, L.H.C.; Pinheiro, R.O.; Hasslocher-Moreno, A.M.; Brasil, P.E.A.A.; et al. Omega-3 supplementation on inflammatory markers in patients with chronic Chagas cardiomyopathy: A randomized clinical study. Nutr. J. 2017, 16, 9. [Google Scholar] [CrossRef][Green Version]
- Sparkes, C.; Gibson, R.; Sinclair, A.; Else, P.L.; Meyer, B.J. Effect of low dose docosahexaenoic acid-rich fish oil on plasma lipids and lipoproteins in pre-menopausal women: A dose-response randomized placebo-controlled trial. Nutrients 2018, 10, 1460. [Google Scholar] [CrossRef]
- Sun, X.M.; Ren, L.J.; Bi, Z.Q.; Ji, X.J.; Zhao, Q.Y.; Jiang, L.; Huang, H. Development of a cooperative two-factor adaptive-evolution method to enhance lipid production and prevent lipid peroxidation in Schizochytrium sp. Biotechnol. Biofuels 2018, 11, 16. [Google Scholar] [CrossRef]
- Wu, S.T.; Yu, S.T.; Lin, L.P. Effect of culture conditions on docosahexaenoic acid production by Schizochytrium sp. S31. Process Biochem. 2005, 40, 3103–3108. [Google Scholar] [CrossRef]
- Allaire, J.; Couture, P.; Leclerc, M.; Charest, A.; Marin, J.; Lepine, M.C.; Talbot, D.; Tchernof, A.; Lamarche, B. A randomized, crossover, head-to-head comparison of eicosapentaenoic acid and docosahexaenoic acid supplementation to reduce inflammation markers in men and women: The Comparing EPA to DHA (ComparED) Study. Am. J. Clin. Nutr. 2016, 104, 280–287. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C. Omega-3 fatty acids and inflammatory processes: From molecules to man. Biochem. Soc. Trans. 2017, 45, 1105–1115. [Google Scholar] [CrossRef] [PubMed]
- Qu, L.; Ren, L.J.; Huang, H. Scale-up of docosahexaenoic acid production in fed-batch fermentation by Schizochytrium sp based on volumetric oxygen-transfer coefficient. Biochem. Eng. J. 2013, 77, 82–87. [Google Scholar] [CrossRef]
- Li, J.; Liu, R.J.; Chang, G.F.; Li, X.Y.; Chang, M.; Liu, Y.F.; Jin, Q.; Wang, X. A strategy for the highly efficient production of docosahexaenoic acid by Aurantiochytrium limacinum SR21 using glucose and glycerol as the mixed carbon sources. Bioresour. Technol. 2015, 177, 51–57. [Google Scholar] [CrossRef]
- Yin, F.W.; Guo, D.S.; Ren, L.J.; Ji, X.J.; Huang, H. Development of a method for the valorization of fermentation wastewater and algal-residue extract in docosahexaenoic acid production by Schizochytrium sp. Bioresour. Technol. 2018, 266, 482–487. [Google Scholar] [CrossRef]
- Ganuza, E.; Anderson, A.J.; Ratledge, C. High-cell-density cultivation of Schizochytrium sp. in an ammonium/pH-auxostat fed-batch system. Biotechnol. Lett. 2008, 30, 1559–1564. [Google Scholar] [CrossRef]
- Wang, K.; Sun, T.; Cui, J.Y.; Liu, L.S.; Bi, Y.Q.; Pei, G.S.; Chen, L.; Zhang, W. Screening of chemical modulators for lipid accumulation in Schizochytrium sp. S31. Bioresour. Technol. 2018, 260, 124–129. [Google Scholar] [CrossRef]
- Chi, Z.Y.; Pyle, D.; Wen, Z.Y.; Frear, C.; Chen, S.L. A laboratory study of producing docosahexaenoic acid from biodiesel-waste glycerol by microalgal fermentation. Process Biochem. 2007, 42, 1537–1545. [Google Scholar] [CrossRef]
- Chen, W.; Zhou, P.P.; Zhu, Y.M.; Xie, C.; Ma, L.; Wang, X.P.; Bao, Z.; Yu, L. Improvement in the docosahexaenoic acid production of Schizochytrium sp. S056 by replacement of sea salt. Bioprocess Biosyst. Eng. 2016, 39, 315–321. [Google Scholar] [CrossRef]
- Patil, K.P.; Gogate, P.R. Improved synthesis of docosahexaenoic acid (DHA) using Schizochytrium limacinum SR21 and sustainable media. Chem. Eng. J. 2015, 268, 187–196. [Google Scholar] [CrossRef]
- Chen, G.Q.; Fan, K.W.; Lu, F.P.; Li, Q.A.; Aki, T.; Chen, F.; Jiang, Y. Optimization of nitrogen source for enhanced production of squalene from thraustochytrid Aurantiochytrium sp. New Biotechnol. 2010, 27, 382–389. [Google Scholar] [CrossRef] [PubMed]
- Guo, D.S.; Ji, X.J.; Ren, L.J.; Yin, F.W.; Sun, X.M.; Huang, H.; Zhen, G. Development of a multi-stage continuous fermentation strategy for docosahexaenoic acid production by Schizochytrium sp. Bioresour. Technol. 2018, 269, 32–39. [Google Scholar] [CrossRef] [PubMed]
- Bailey, R.B.; DiMasi, D.; Hansen, J.M.; Mirrasoul, P.J.; Ruecker, C.M.; Veeder, G.T., III; Kaneko, T.; Barclay, W.R. Enhanced Production of Lipids Containing Polyenoic Fatty Acid by Very High Density Cultures of Eukaryotic Microbes in Fermentors. U.S. Patent No. 7,732,170, 7 February 2008. [Google Scholar]
- Chang, G.F.; Gao, N.S.; Tian, G.W.; Wu, Q.H.; Chang, M.; Wang, X.G. Improvement of docosahexaenoic acid production on glycerol by Schizochytrium sp. S31 with constantly high oxygen transfer coefficient. Bioresour. Technol. 2013, 142, 400–406. [Google Scholar] [CrossRef]
- Bligh, E.G.; Dyer, W.J. A rapid method of total lipid extractionand purification. Can. J. Biochem. Physiol. 1959, 37, 911–917. [Google Scholar] [CrossRef]
- Pereira, H.; Barreira, L.; Mozes, A.; Florindo, C.; Polo, C.; Duarte, C.V.; Custódio, L.; Varela, J. Microplatebased high throughput screening procedure for the isolation of lipid-rich marine microalgae. Biotechnol. Biofuels 2011, 4, 12. [Google Scholar] [CrossRef]
- Merrill, A.L.; Watt, B.K. Energy value of foods: Basis and derivation. Agriculture Handbook; US Department of Agriculture, Forest Service: Washington, DC, USA, 1973; p. 74.
- Lepage, G.; Roy, C.C. Improved recovery of fatty-acid through direct transesterification without prior extraction or purification. J. Lipid Res. 1984, 25, 1391–1396. [Google Scholar]
- Pereira, H.; Barreira, L.; Figueiredo, F.; Custodio, L.; Vizetto-Duarte, C.; Polo, C.; Resek, E.; Engelen, A.; Varela, J. Polyunsaturated fatty acids of marine macroalgae: Potential for nutritional and pharmaceutical applications. Mar. Drugs 2012, 10, 1920–1935. [Google Scholar] [CrossRef]
- Bindea, M.; Rusu, B.; Rusu, A.; Trif, M.; Leopold, L.F.; Dulf, F.; Vodnar, D.C. Valorification of crude glycerol for pure fractions of docosahexaenoic acid and β-carotene production by using Schizochytrium limacinum and Blakeslea trispora. Microb. Cell Factories 2018, 17, 97. [Google Scholar] [CrossRef]
- Huang, T.Y.; Lu, W.C.; Chu, I.M. A fermentation strategy for producing docosahexaenoic acid in Aurantiochytrium limacinum SR21 and increasing C22:6 proportions in total fatty acid. Bioresour. Technol. 2012, 123, 8–14. [Google Scholar] [CrossRef]
- Yu, X.J.; Yu, Z.Q.; Liu, Y.L.; Sun, J.; Zheng, J.Y.; Wang, Z. Utilization of high-fructose corn syrup for biomass production containing high levels of docosahexaenoic acid by a newly isolated Aurantiochytrium sp. YLH70. Appl. Biochem. Biotechnol. 2015, 177, 1229–1240. [Google Scholar] [CrossRef] [PubMed]
- Yokochi, T.; Honda, D.; Higashihara, T.; Nakahara, T. Optimization of docosahexaenoic acid production by Schizochytrium limacinum SR21. Appl. Microbiol. Biotechnol. 1998, 49, 72–76. [Google Scholar] [CrossRef]
- Yokoyama, R.; Honda, D. Taxonomic rearrangement of the genus Schizochytrium sensu lato based on morphology, chemotaxonomic characteristics, and 18S rRNA gene phylogeny (Thraustochytriaceae, Labyrinthulomycetes): Emendation for Schizochytrium and erection of Aurantiochytrium and Oblongichytrium gen. nov. Mycoscience 2007, 48, 199–211. [Google Scholar]
- Taoka, Y.; Nagano, N.; Okita, Y.; Izumida, H.; Sugimoto, S.; Hayashi, M. Influences of culture temperature on the growth, lipid content and fatty acid composition of Aurantiochytrium sp. strain mh0186. Mar. Biotechnol. 2009, 11, 368–374. [Google Scholar] [CrossRef]
- Nakazawa, A.; Matsuura, H.; Kose, R.; Ito, K.; Ueda, M.; Honda, D.; Inouyea, I.; Kayaa, K.; Watanabe, M.M. Optimization of biomass and fatty acid production by Aurantiochytrium sp. strain 4W-1b. Innov. Res. Algal Biomass 2012, 15, 27–33. [Google Scholar] [CrossRef][Green Version]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).