The Effects of a Marathon Effort on Psychomotor Performance and Catecholamine Concentration in Runners over 50 Years of Age
Abstract
1. Introduction
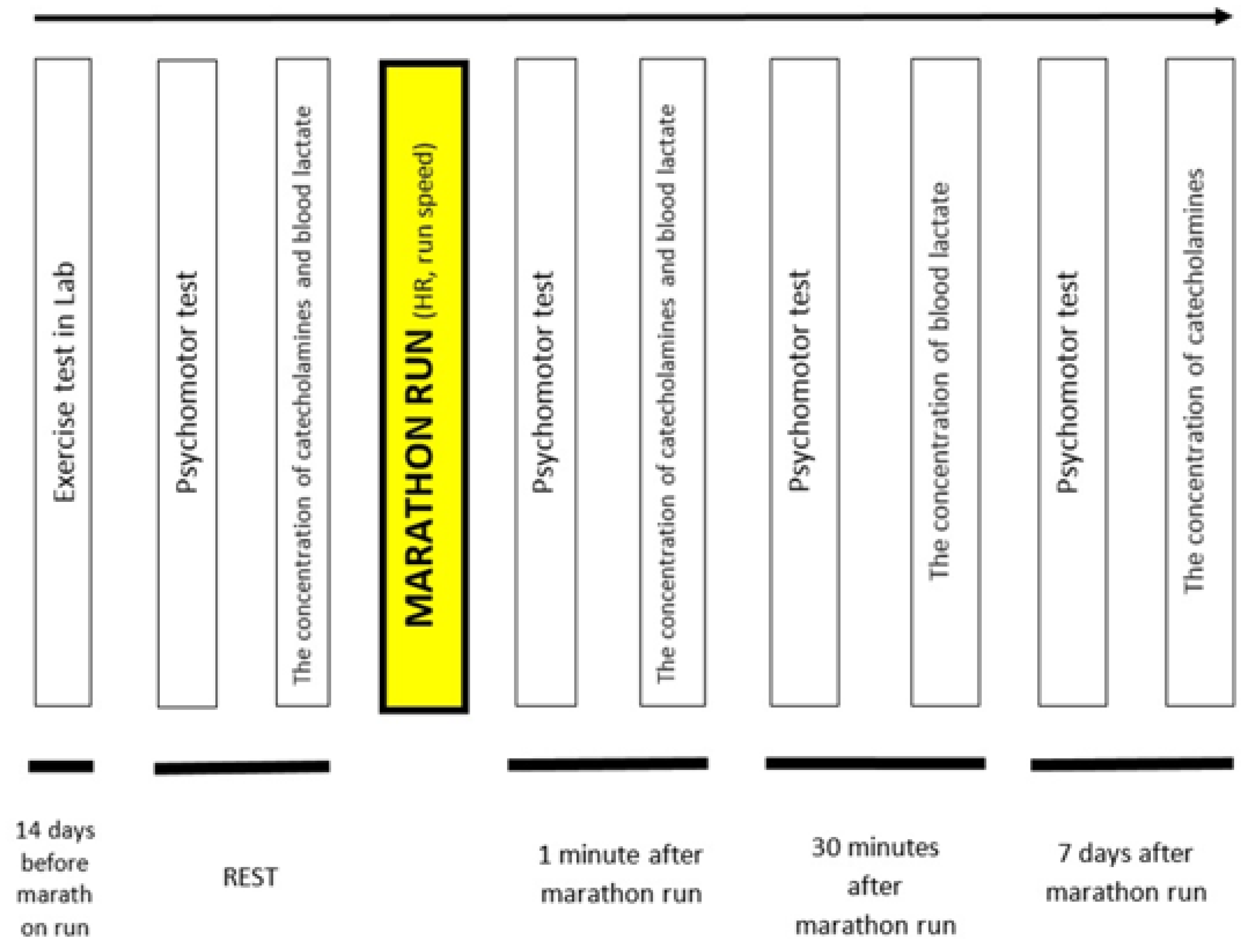
2. Materials and Methods
2.1. Participants
2.2. Weather Conditions and Route Profile during the Marathon Race
2.3. The Indoor Treadmill Test
2.4. Psychomotor Test—Choice Reaction Time
2.5. Heart Rate and Running Speed during the Marathon Race
2.6. Catecholamine levels
2.7. Lactate Concentration Measurements
2.8. Statistical Analysis
3. Results
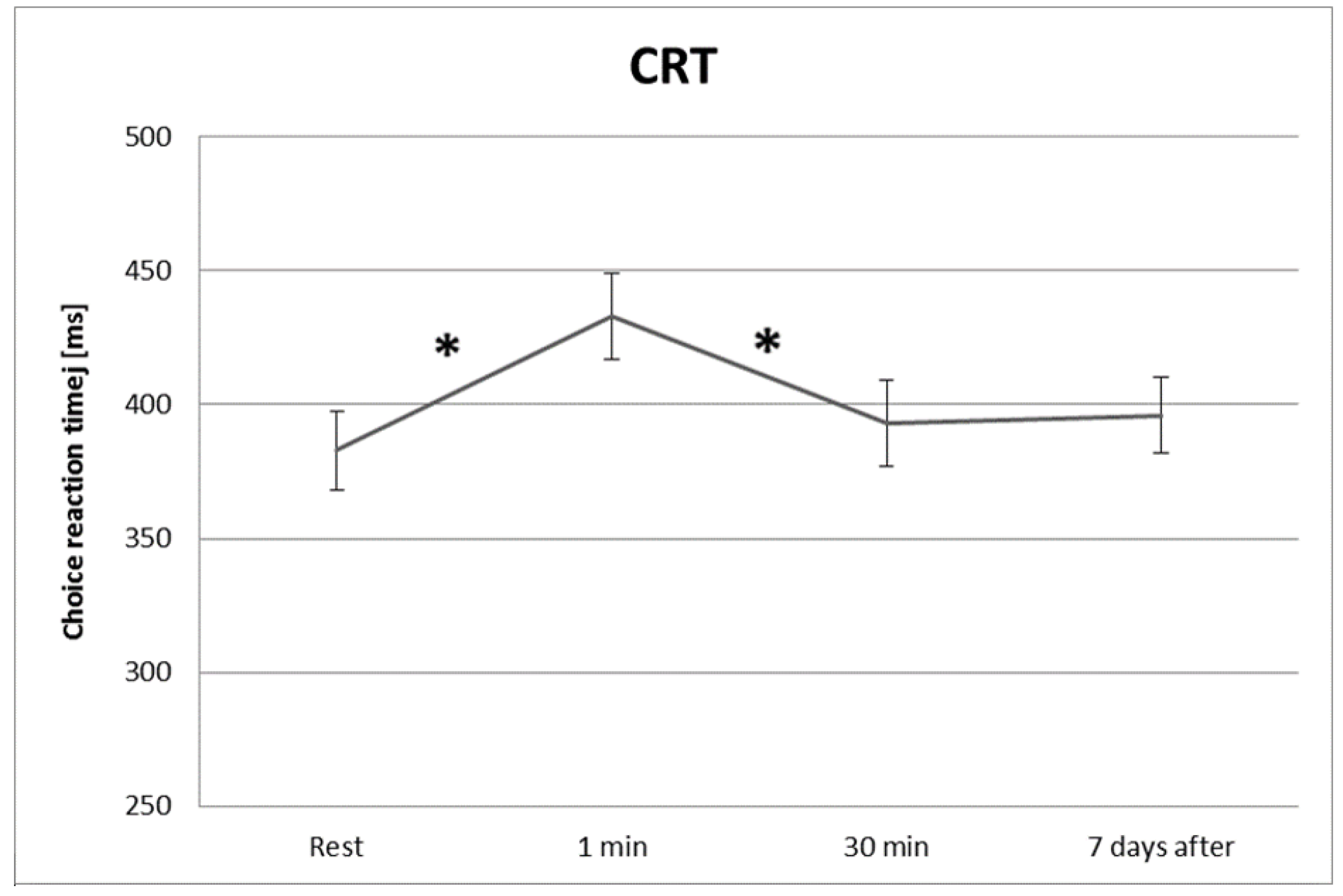
3.1. Psychomotor Test—Choice Reaction Time
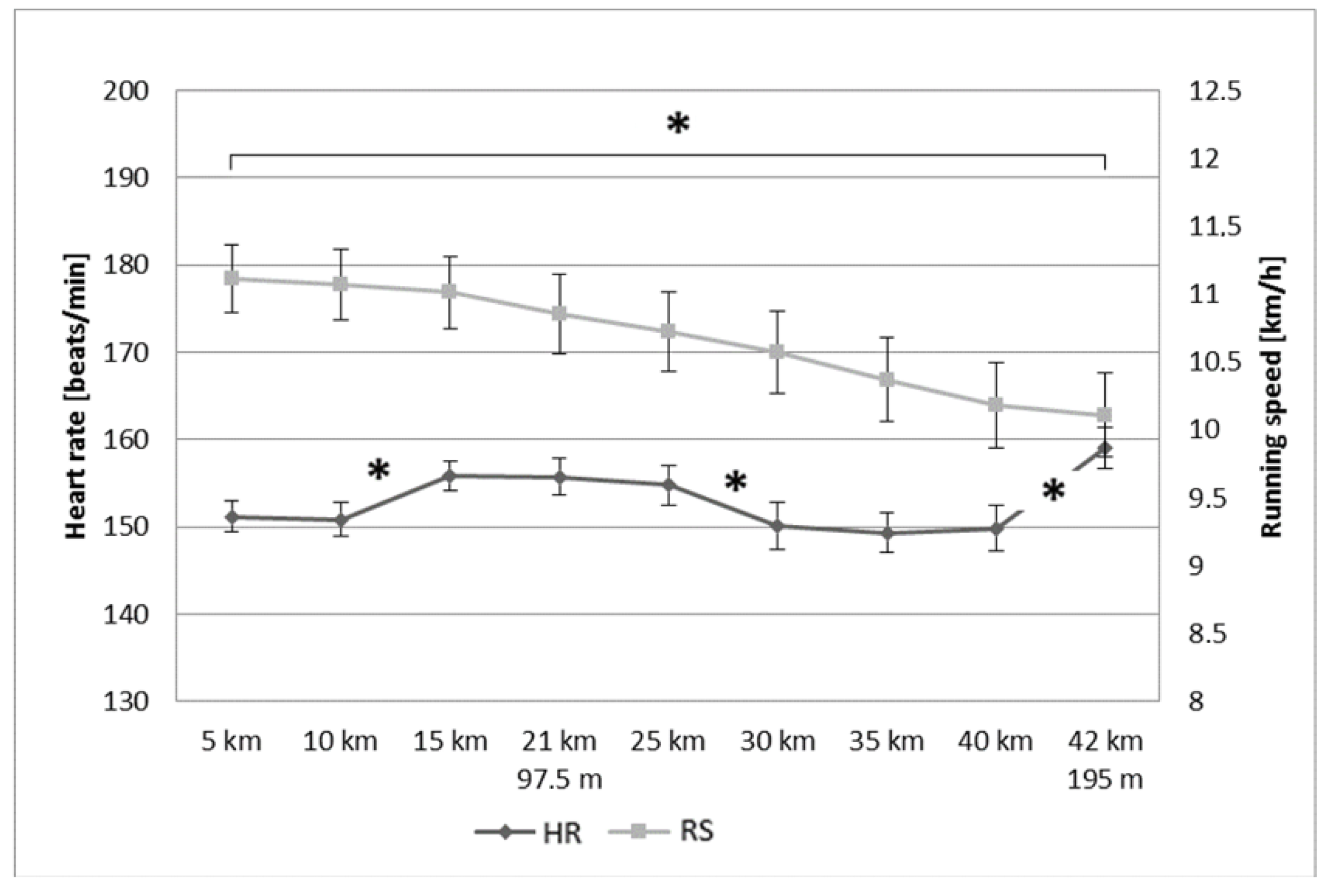
3.2. Heart Rate and Running Speed during the Marathon Race
3.3. Catecholamine and Lactate Levels
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Lepers, R.; Cattagni, T. Do older athletes reach limits in their performance during marathon running? Age 2012, 34, 773–781. [Google Scholar] [CrossRef]
- Leyk, D.; Rüther, T.; Wunderlich, M.; Sievert, A.; Essfeld, D.; Witzki, A.; Erley, O.; Küchmeister, G.; Piekarski, C.; Löllgen, H. Physical performance in middle age and old age: Good news for our sedentary and aging society. Dtsch. Arztebl. Int. 2010, 107, 809–816. [Google Scholar] [PubMed]
- Quinn, T.J.; Manley, M.J.; Aziz, J.; Padham, J.L.; MacKenzie, A.M. Aging and factors related to running economy. J. Strength Cond. Res. 2011, 25, 2971–2979. [Google Scholar] [CrossRef] [PubMed]
- Wiswell, R.A.; Jaque, S.V.; Marcell, T.J.; Hawkins, S.A.; Tarpenning, K.M.; Constantino, N.; Hyslop, D.M. Maximal aerobic power, lactate threshold, and running performance in master athletes. Med. Sci. Sports Exerc. 2000, 32, 1165–1170. [Google Scholar] [CrossRef] [PubMed]
- Daniels, J.; Daniels, N. Running economy of elite male and elite female runners. Med. Sci. Sports Exerc. 1992, 24, 483–489. [Google Scholar] [CrossRef] [PubMed]
- Gordon, D.; Wightman, S.; Basevitch, I.; Johnstone, J.; Espejo-Sanchez, C.; Beckford, C.; Boal, M.; Scruton, A.; Ferrandino, M.; Merzbach, V. Physiological and training characteristics of recreational marathon runners. Open Access J. Sports Med. 2017, 24, 231–241. [Google Scholar] [CrossRef]
- Nybo, L.; Secher, N.H. Cerebral perturbations provoked by prolonged exercise. Prog. Neurobiol. 2004, 72, 223–261. [Google Scholar] [CrossRef]
- Secher, N.H.; Seifert, T.; Van Lieshout, J.J. Cerebral blood flow and metabolism during exercise: Implications for fatigue. J. Appl. Physiol. 2008, 104, 306–314. [Google Scholar] [CrossRef]
- Sagnol, M.; Claustre, J.; Cottet-Emard, J.M.; Pequignot, J.M.; Fellmann, N.; Coudert, J.; Peyrin, L. Plasma free and sulphated catecholamines after ultra-long exercise and recovery. Eur. J. Appl. Physiol. Occup. Physiol. 1990, 60, 91–97. [Google Scholar] [CrossRef]
- Hodgetts, V.; Coppack, S.W.; Frayn, K.N.; Hockaday, T.D. Factors controlling fat mobilization from human subcutaneous adipose tissue during exercise. J. Appl. Physiol. 1991, 71, 445–451. [Google Scholar] [CrossRef]
- Chmura, J.; Krysztofiak, H.; Ziemba, A.W.; Nazar, K.; Kaciuba-Uscilko, H. Psychomotor performance during prolonged exercise above and below the blood lactate threshold. Eur. J. Appl. Physiol. Occup. Physiol. 1998, 77, 77–80. [Google Scholar] [CrossRef] [PubMed]
- Chmura, J.; Nazar, K.; Kaciuba-Uscilko, H. Choice reaction time during graded exercise in relation to blood lactate and plasma catecholamine thresholds. Int. J. Sports Med. 1994, 15, 172–176. [Google Scholar] [CrossRef] [PubMed]
- Chmura, J.; Nazar, K. Parallel changes in the onset of blood lactate accumulation (OBLA) and threshold of psychomotor performance deterioration during incremental exercise after training in athletes. Int. J. Psychophysiol. 2010, 75, 287–290. [Google Scholar] [CrossRef] [PubMed]
- Fery, Y.A.; Ferry, A.; Vom Hofe, A.; Rieu, M. Effect of physical exhaustion on cognitive functioning. Percept. Mot. Skills 1997, 84, 291–298. [Google Scholar] [CrossRef] [PubMed]
- Hogervorst, E.; Riedel, W.; Jeukendrup, A.; Jolles, J. Cognitive performance after strenuous physical exercise. Percept. Mot. Skills 1996, 83, 479–488. [Google Scholar] [CrossRef]
- Lambourne, K.; Tomporowski, P. The effect of exercise-induced arousal on cognitive task performance: A meta-regression analysis. Brain Res. 2010, 1341, 12–24. [Google Scholar] [CrossRef] [PubMed]
- Audiffren, M.; Tomporowski, P.D.; Zagrodnik, J. Acute aerobic exercise and information processing: Energizing motor processes during a choice reaction time task. Acta Psychol. 2008, 129, 410–419. [Google Scholar] [CrossRef] [PubMed]
- Grego, F.; Vallier, J.M.; Collardeau, M.; Bermon, S.; Ferrari, P.; Candito, M.; Bayer, P.; Magnié, M.N.; Brisswalter, J. Effects of long duration exercise on cognitive function, blood glucose, and counterregulatory hormones in male cyclists. Neurosci. Lett. 2004, 364, 76–80. [Google Scholar] [CrossRef]
- Der, G.; Deary, I.J. Age and sex differences in reaction time in adulthood: Results from the United Kingdom Health and Lifestyle Survey. Psychol. Aging 2006, 21, 62–73. [Google Scholar] [CrossRef]
- Chodzko-Zajko, W.J. Normal aging and human physiology. Semin. Speech Lang. 1997, 18, 95–104, quiz 104–105. [Google Scholar] [CrossRef]
- Chodzko-Zajko, W.J.; Moore, K.A. Physical fitness and cognitive functioning in aging. Exerc. Sport Sci. Rev. 1994, 22, 195–220. [Google Scholar] [CrossRef] [PubMed]
- Tseng, C.N.; Gau, B.S.; Lou, M.F. The effectiveness of exercise on improving cognitive function in older people: A systematic review. J. Nurs. Res. 2011, 19, 119–131. [Google Scholar] [CrossRef] [PubMed]
- van Boxtel, M.P.; Paas, F.G.; Houx, P.J.; Adam, J.J.; Teeken, J.C.; Jolles, J. Aerobic capacity and cognitive performance in a cross-sectional aging study. Med. Sci. Sports Exerc. 1997, 29, 1357–1365. [Google Scholar] [CrossRef] [PubMed]
- Rogers, R.L.; Meyer, J.S.; Mortel, K.F. After reaching retirement age physical activity sustains cerebral perfusion and cognition. J. Am. Geriatr. Soc. 1990, 38, 123–128. [Google Scholar] [CrossRef]
- Brisswalter, J.; Collardeau, M.; Rene, A. Effects of acute physical exercise characteristics on cognitive performance. Sports Med. 2002, 32, 555–566. [Google Scholar] [CrossRef]
- Tomporowski, P.D. Effects of acute bouts of exercise on cognition. Acta Psychol. 2003, 112, 297–324. [Google Scholar] [CrossRef]
- Zavorsky, G.S.; Tomko, K.A.; Smoliga, J.M. Declines in marathon performance: Sex differences in elite and recreational athletes. PLoS ONE 2017, 12, e0172121. [Google Scholar] [CrossRef]
- Bruce, R.A. Multi-stage treadmill test of maximal and sub maximal exercise. In Exercise Testing and Training of Apparently Health Individuals: A Handbook for Physicians; American Heart Association: Dallas, TX, USA, 1972; pp. 32–34. [Google Scholar]
- Wasserman, K.; Whipp, B.J.; Koyl, S.N.; Beaver, W.L. Anaerobic threshold and respiratory gas exchange during exercise. J. Appl. Physiol. 1973, 35, 236–243. [Google Scholar] [CrossRef]
- McMorris, T.; Keen, P. Effect of exercise on simple reaction times of recreational athletes. Percept. Mot. Skills 1994, 78, 123–130. [Google Scholar] [CrossRef]
- McMorris, T.; Sproule, J.; Draper, S.; Child, R. Performance of a psychomotor skill following rest, exercise at the plasma epinephrine threshold and maximal intensity exercise. Percept. Mot. Skills 2000, 91, 553–562. [Google Scholar] [CrossRef]
- Joyner, M.J.; Coyle, E.F. Endurance exercise performance: The physiology of champions. J Physiol. 2008, 586, 35–44. [Google Scholar] [CrossRef] [PubMed]
- Loftin, M.; Sothern, M.; Koss, C.; Tuuri, G.; Vanvrancken, C.; Kontos, A.; Bonis, M. Energy expenditure and influence of physiologic factors during marathon running. J. Strength Cond. Res. 2007, 21, 1188–1191. [Google Scholar] [PubMed]
- Allen, D.G.; Lamb, G.D.; Westerblad, H. Skeletal muscle fatigue: Cellular mechanisms. Physiol. Rev. 2008, 88, 287–332. [Google Scholar] [CrossRef] [PubMed]
- Cairns, S.P. Lactic acid and exercise performance: Culprit or friend? Sports Med. 2006, 36, 279–291. [Google Scholar] [CrossRef] [PubMed]
- Collardeau, M.; Brisswalter, J.; Audiffren, M. Effects of a prolonged run on simple reaction time of well trained runners. Percept. Mot. Skills 2001, 93, 679–689. [Google Scholar] [CrossRef]
- Tomporowski, P.D.; Beasman, K.; Ganio, M.S.; Cureton, K. Effects of dehydration and fluid ingestion on cognition. Int. J. Sports Med. 2007, 28, 891–896. [Google Scholar] [CrossRef]
- Cian, C.; Barraud, P.A.; Melin, B.; Raphel, C. Effects of fluid ingestion on cognitive function after heat stress or exercise-induced dehydration. Int. J. Psychophysiol. 2001, 42, 243–251. [Google Scholar] [CrossRef]
- Cheuvront, S.N.; Haymes, E.M. Thermoregulation and marathon running: Biological and environmental influences. Sports Med. 2001, 31, 743–762. [Google Scholar] [CrossRef]
- Smith, J.E.; Garbutt, G.; Lopes, P.; Pedoe, D.T. Effects of prolonged strenuous exercise (marathon running) on biochemical and haematological markers used in the investigation of patients in the emergency department. Br. J. Sports Med. 2004, 38, 292–294. [Google Scholar] [CrossRef]
- Zouhal, H.; Jacob, C.; Delamarche, P.; Gratas-Delamarche, A. Catecholamines and the effects of exercise, training and gender. Sports Med. 2008, 38, 401–423. [Google Scholar] [CrossRef]
- Kyrolainen, H.; Pullinen, T.; Candau, R.; Avela, J.; Huttunen, P.; Komi, P.V. Effects of marathon running on running economy and kinematics. Eur. J. Appl. Physiol. 2000, 82, 297–304. [Google Scholar] [CrossRef] [PubMed]
- Maron, M.B.; Horvath, S.M.; Wilkerson, J.E. Blood biochemical alterations during recovery from competitive marathon running. Eur. J. Appl. Physiol. Occup. Physiol. 1977, 36, 231–238. [Google Scholar] [CrossRef] [PubMed]
- Christou, D.D.; Seals, D.R. Decreased maximal heart rate with aging is related to reduced {beta}-adrenergic responsiveness but is largely explained by a reduction in intrinsic heart rate. J. Appl. Physiol. 2008, 105, 24–29. [Google Scholar] [CrossRef]
- Mazzeo, R.S. Aging, immune function, and exercise: Hormonal regulation. Int. J. Sports Med. 2000, 21 (Suppl. 1), S10–S13. [Google Scholar] [CrossRef]
- Seals, D.R.; Esler, M.D. Human ageing and the sympathoadrenal system. J. Physiol. 2000, 528 Pt 3, 407–417. [Google Scholar] [CrossRef]
- de Leeuw, A.W.; Meerhoff, L.A.; Knobbe, A. Effects of Pacing Properties on Performance in Long-Distance Running. Big Data 2018. [Google Scholar] [CrossRef]
- Díaz, J.J.; Fernández-Ozcorta, E.J.; Santos-Concejero, J. The influence of pacing strategy on marathon world records. Eur. J. Sport Sci. 2018, 18, 781–786. [Google Scholar] [CrossRef]
- Nikolaidis, P.T.; Knechtle, B. Effect of age and performance on pacing of marathon runners. Open Access J. Sports Med. 2017, 8, 171–180. [Google Scholar] [CrossRef]
- Stones, M.J.; Hartin, A. Aging and Half-Ironman Performance. Exp. Aging Res. 2017, 43, 178–191. [Google Scholar] [CrossRef]
| VO2max (mL·kg−1·min 1) | HRmax (Beats·min−1) | HRAT (Beats·min−1) | HRAT (% HRmax) | VE Max (L·min−1) |
|---|---|---|---|---|
| mean ± SE | ||||
| 51 ± 2 | 170 ± 2 | 153 ± 2 | 90 ± 1 | 144 ± 4 |
| Catecholamine | Rest (R) | After the Marathon | Statistical Significance (p < 0.05) | |
|---|---|---|---|---|
| 1st Min (AM) | 7th Day (A7) | |||
| mean ± SE | ||||
| Adrenaline (nmol·L−1) | 0.25 ± 0.02 | 0.89 ± 0.10 | 0.24 ± 0.03 | R vs. AM |
| AM vs. A7 | ||||
| Noradrenaline (nmol·L−1) | 1.89 ± 0.13 | 6.33 ± 0.32 | 1.78 ± 0.20 | R vs. AM |
| AM vs. A7 | ||||
| Lactate (mmol·L−1) | Statistical Significance (p < 0.05) | ||
|---|---|---|---|
| Rest (Baseline) | After the Marathon (Post-Exercise) | ||
| (LAR) | 1 Min (LA 1′) | 30 Min (LA 30′) | |
| mean ± SE | |||
| 1.67 ± 0.05 | 3.83 ± 0.35 | 2.77 ± 0.16 | LAR vs. LA 1′ |
| LA 1′ vs. LA 30′ | |||
| LAR vs. LA 30′ | |||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chmura, J.; Chmura, P.; Konefał, M.; Batra, A.; Mroczek, D.; Kosowski, M.; Młynarska, K.; Andrzejewski, M.; Rokita, A.; Ponikowski, P. The Effects of a Marathon Effort on Psychomotor Performance and Catecholamine Concentration in Runners over 50 Years of Age. Appl. Sci. 2020, 10, 2067. https://doi.org/10.3390/app10062067
Chmura J, Chmura P, Konefał M, Batra A, Mroczek D, Kosowski M, Młynarska K, Andrzejewski M, Rokita A, Ponikowski P. The Effects of a Marathon Effort on Psychomotor Performance and Catecholamine Concentration in Runners over 50 Years of Age. Applied Sciences. 2020; 10(6):2067. https://doi.org/10.3390/app10062067
Chicago/Turabian StyleChmura, Jan, Paweł Chmura, Marek Konefał, Amit Batra, Dariusz Mroczek, Michał Kosowski, Katarzyna Młynarska, Marcin Andrzejewski, Andrzej Rokita, and Piotr Ponikowski. 2020. "The Effects of a Marathon Effort on Psychomotor Performance and Catecholamine Concentration in Runners over 50 Years of Age" Applied Sciences 10, no. 6: 2067. https://doi.org/10.3390/app10062067
APA StyleChmura, J., Chmura, P., Konefał, M., Batra, A., Mroczek, D., Kosowski, M., Młynarska, K., Andrzejewski, M., Rokita, A., & Ponikowski, P. (2020). The Effects of a Marathon Effort on Psychomotor Performance and Catecholamine Concentration in Runners over 50 Years of Age. Applied Sciences, 10(6), 2067. https://doi.org/10.3390/app10062067








