Genotype May Influence Bacterial Diversity in Bark and Bud of Vitis vinifera Cultivars Grown under the Same Environment
Abstract
1. Introduction
2. Materials and Methods
2.1. Vine Cultivars and Sampling
2.2. DNA Extraction
2.3. High-Throughput Sequencing and Statistical Analysis
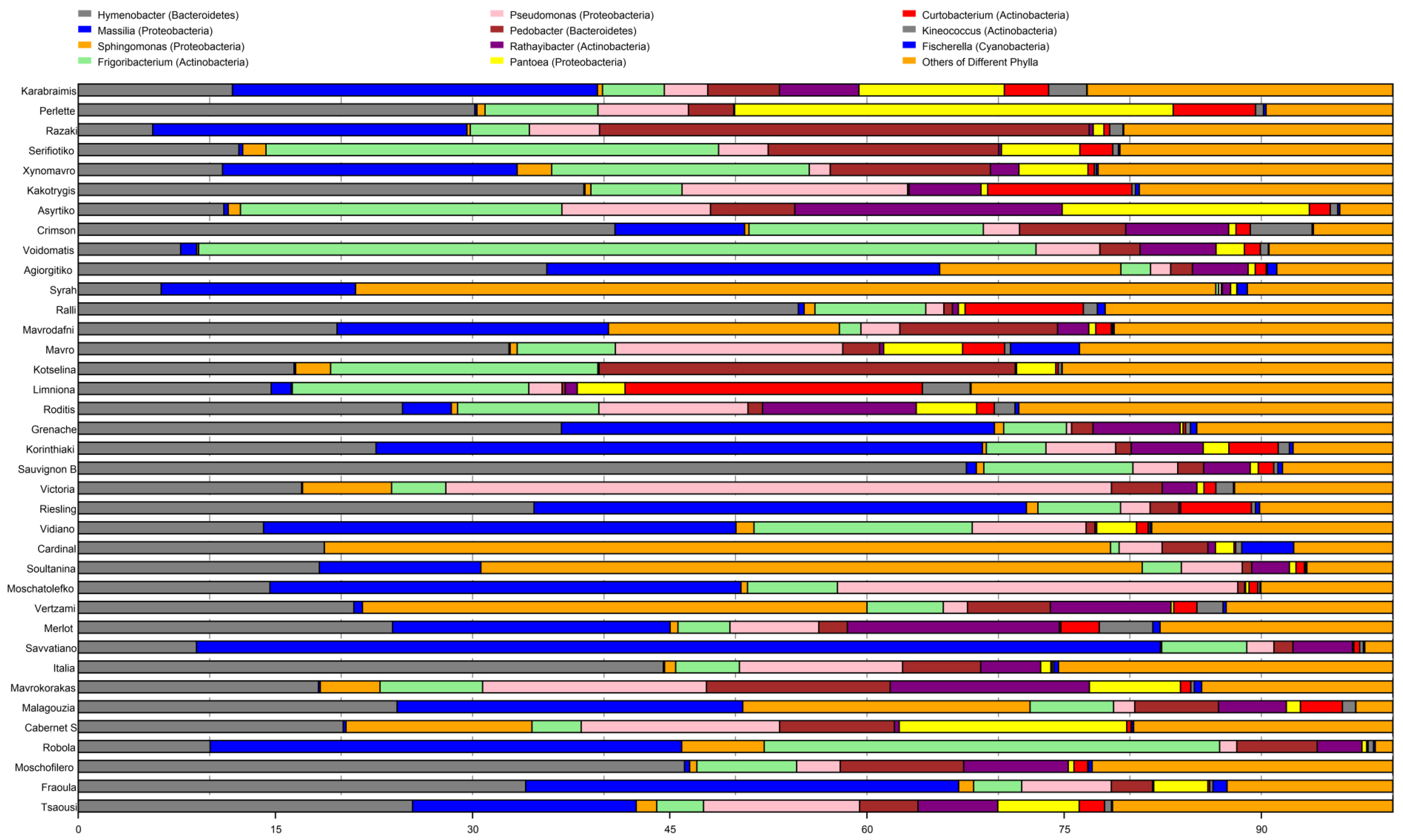
3. Results
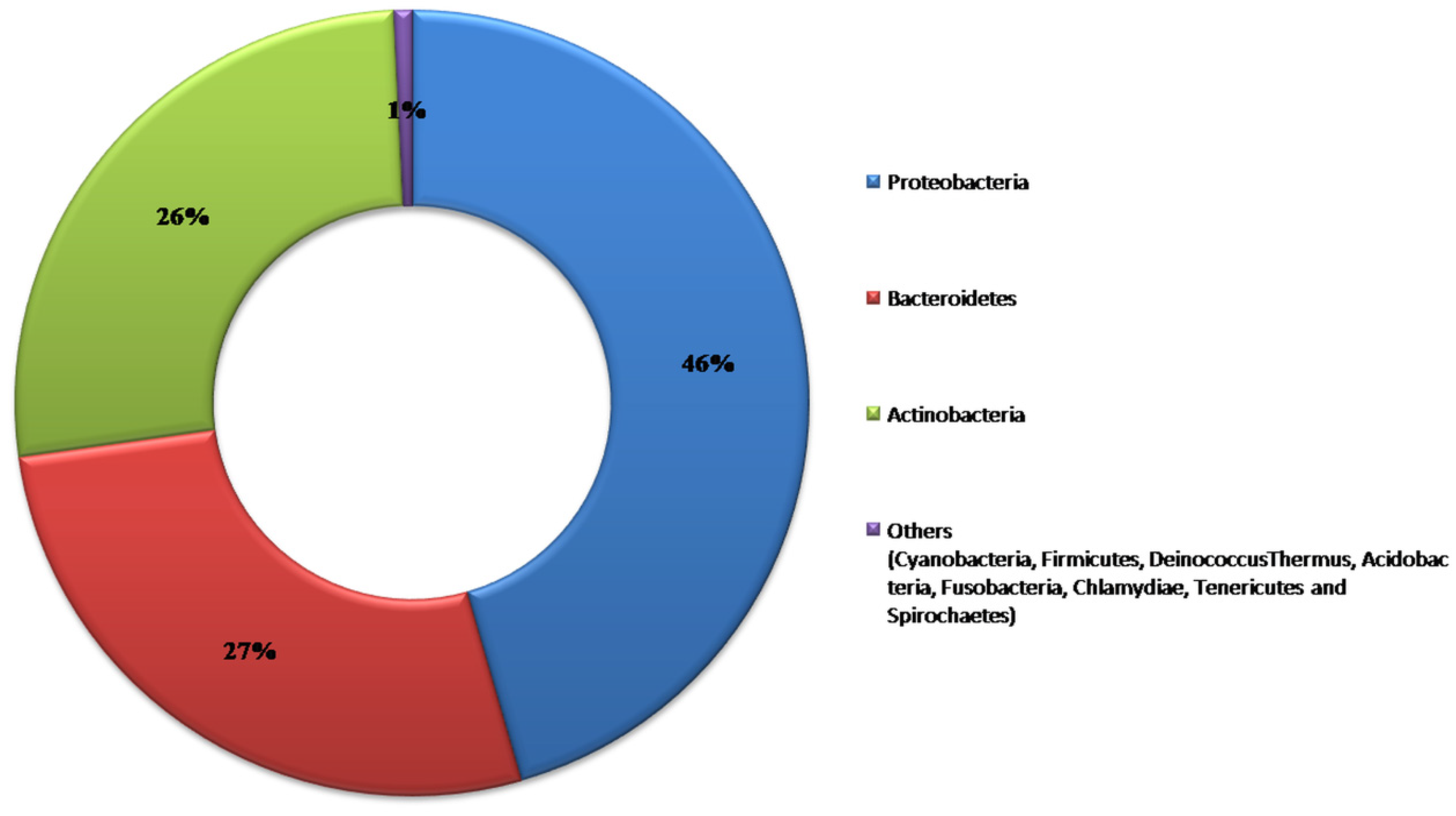
3.1. Sequence Analysis
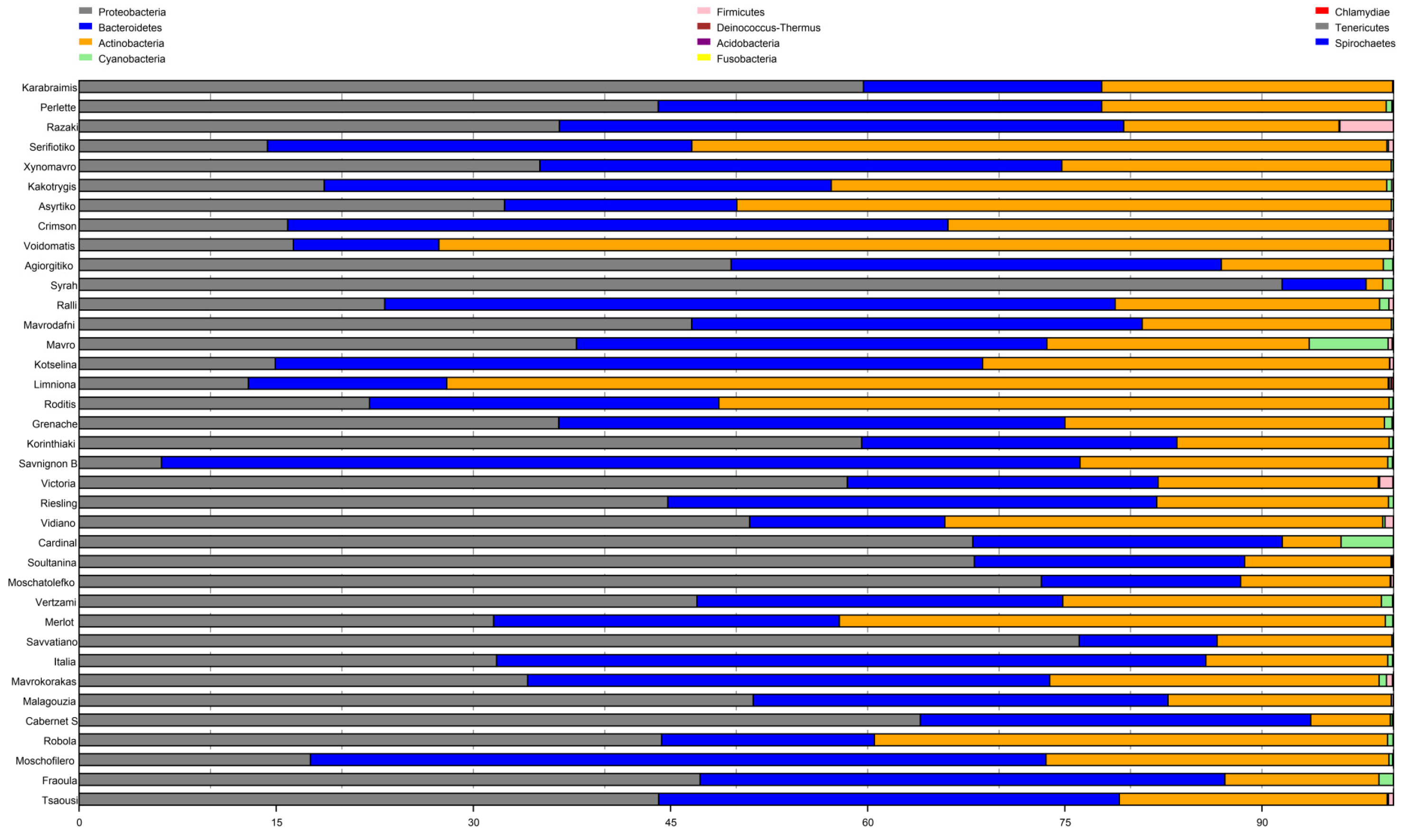
3.2. Microbial Diversity among Cultivars
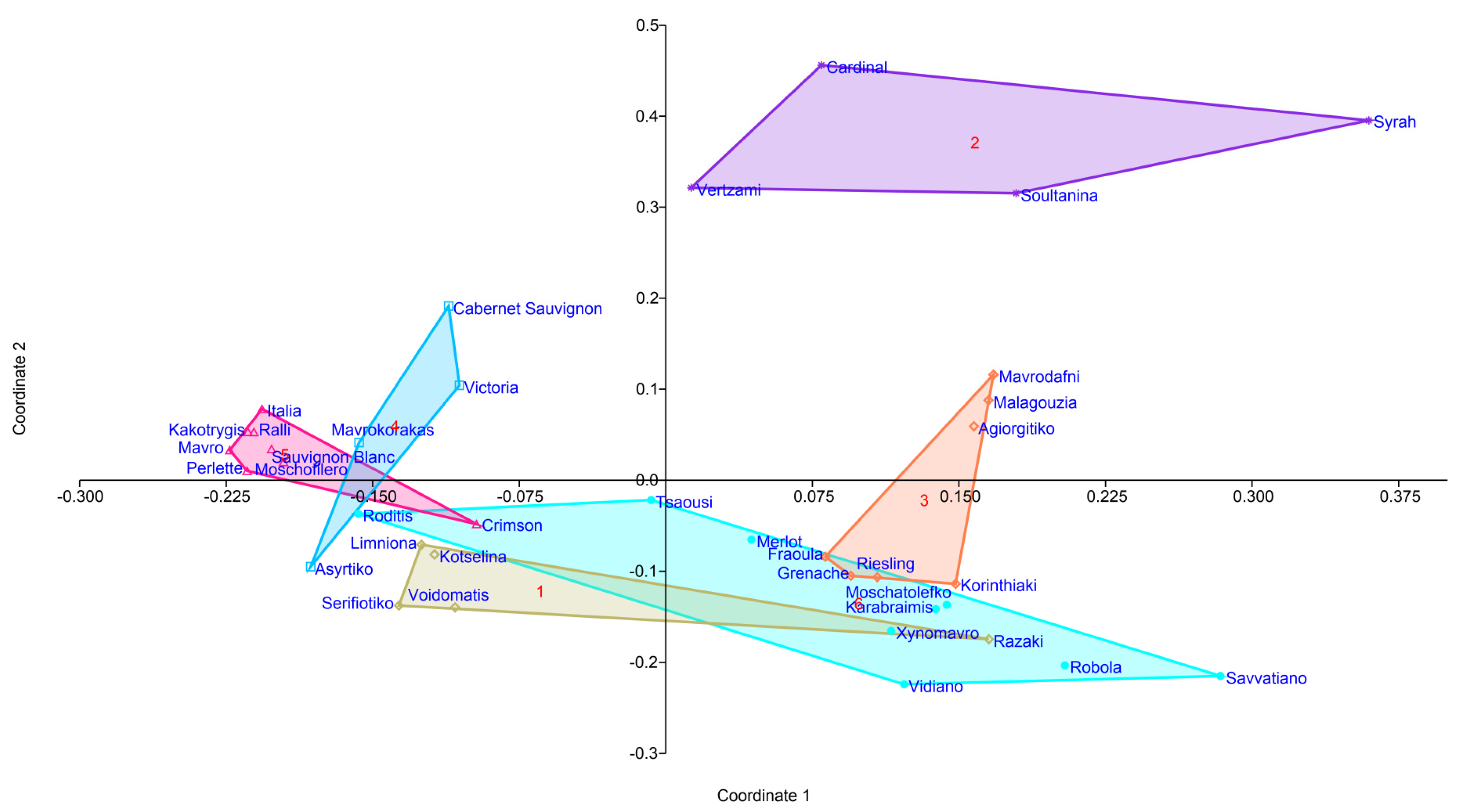
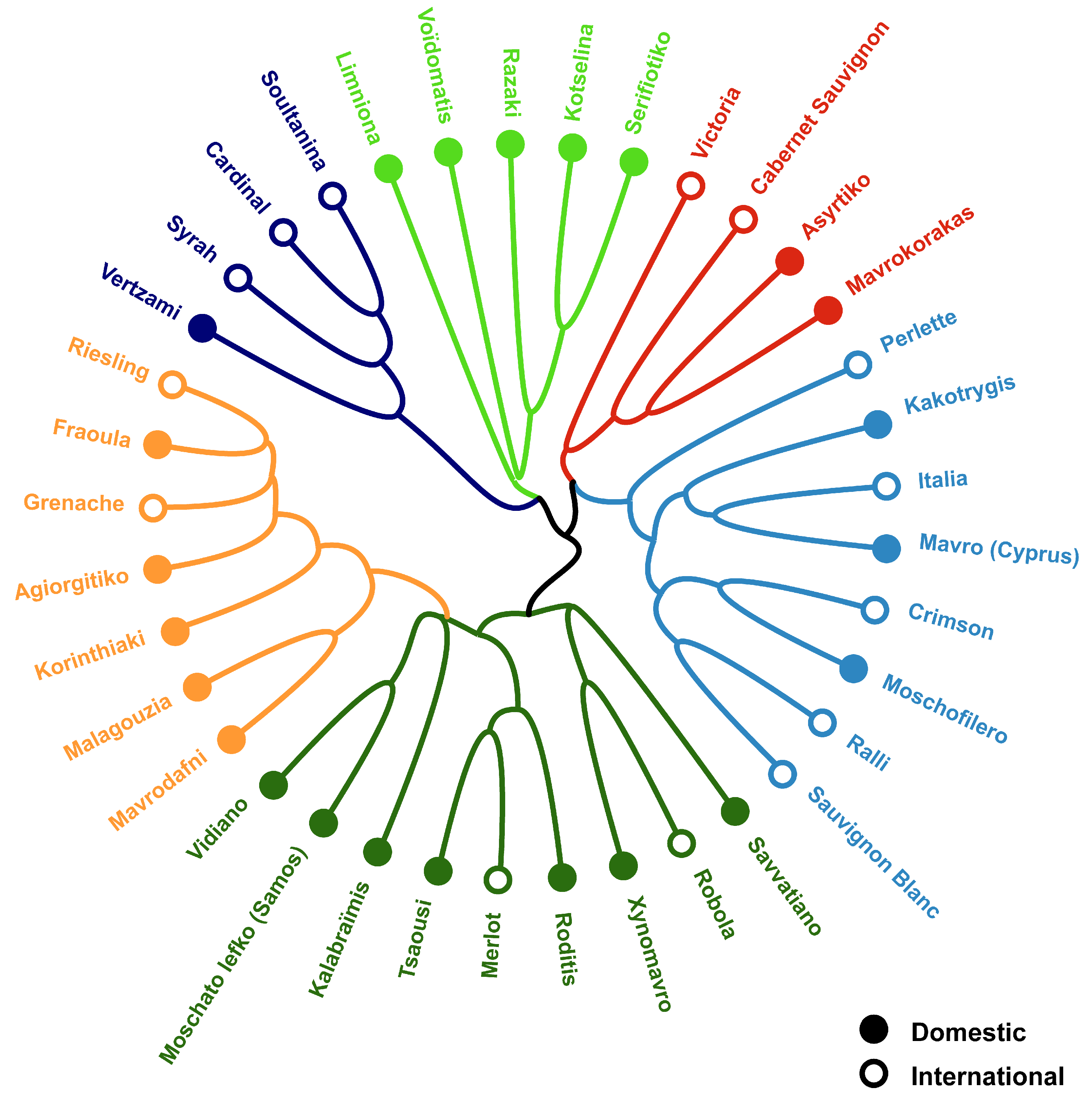
3.3. Cultivar Genotype May Influence Bacterial Community Structure
3.4. Assessment of Vine Bacteria of Economic Importance
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- OIV. International Organisation of Vine and Wine 2019 Statistical Report on World Vitiviniculture; International Organisation of Vine and Wine Intergovernmental Organisation: Paris, France, 2019; Volume 23. [Google Scholar]
- Yang, C.H.; Crowley, D.E.; Borneman, J.; Keen, N.T. Microbial phyllosphere populations are more complex than previously realized. Proc. Natl. Acad. Sci. USA 2001, 98, 3889–3894. [Google Scholar] [CrossRef] [PubMed]
- Müller, T.; Ruppel, S. Progress in cultivation-independent phyllosphere microbiology. FEMS Microbiol. Ecol. 2014, 87, 2–17. [Google Scholar] [CrossRef]
- Singh, P.; Santoni, S.; This, P.; Péros, J.-P. Genotype-Environment Interaction Shapes the Microbial Assemblage in Grapevine’s Phyllosphere and Carposphere: An NGS Approach. Microorganisms 2018, 6, 96. [Google Scholar] [CrossRef] [PubMed]
- Renouf, V.; Claisse, O.; Lonvaud-Funel, A. Inventory and monitoring of wine microbial consortia. Appl. Microbiol. Biotechnol. 2007, 75, 149–164. [Google Scholar] [CrossRef]
- Martins, G.; Lauga, B.; Miot-Sertier, C.; Mercier, A.; Lonvaud, A.; Soulas, M.L.; Soulas, G.; Masneuf-Pomarède, I. Characterization of Epiphytic Bacterial Communities from Grapes, Leaves, Bark and Soil of Grapevine Plants Grown, and Their Relations. PLoS ONE 2013, 8, e073013. [Google Scholar] [CrossRef] [PubMed]
- Vitulo, N.; Lemos, W.J.F.; Calgaro, M.; Confalone, M.; Felis, G.E.; Zapparoli, G.; Nardi, T. Bark and grape microbiome of Vitis vinifera: Influence of geographic patterns and agronomic management on bacterial diversity. Front. Microbiol. 2019, 10, 3203. [Google Scholar] [CrossRef] [PubMed]
- Swiegers, J.H.; Bartowsky, E.J.; Henschke, P.A.; Pretorius, I.S. Yeast and bacterial modulation of wine aroma and flavour. Aust. J. Grape Wine Res. 2005, 11, 139–173. [Google Scholar] [CrossRef]
- Grangeteau, C.; Roullier-Gall, C.; Rousseaux, S.; Gougeon, R.D.; Schmitt-Kopplin, P.; Alexandre, H.; Guilloux-Benatier, M. Wine microbiology is driven by vineyard and winery anthropogenic factors. Microb. Biotechnol. 2017, 10, 354–370. [Google Scholar] [CrossRef]
- Oliveira, M.; Arenas, M.; Lage, O.; Cunha, M.; Amorim, M.I. Epiphytic fungal community in Vitis vinifera of the Portuguese wine regions. Lett. Appl. Microbiol. 2018, 66, 93–102. [Google Scholar] [CrossRef]
- Wei, Y.J.; Wu, Y.; Yan, Y.Z.; Zou, W.; Xue, J.; Ma, W.R.; Wang, W.; Tian, G.; Wang, L.Y. High-throughput sequencing of microbial community diversity in soil, grapes, leaves, grape juice and wine of grapevine from China. PLoS ONE 2018, 13, e0193097. [Google Scholar] [CrossRef]
- Gamalero, E.; Bona, E.; Novello, G.; Boatti, L.; Mignone, F.; Massa, N.; Cesaro, P.; Berta, G.; Lingua, G. Discovering the bacteriome of Vitis vinifera cv. Pinot Noir in a conventionally managed vineyard. Sci. Rep. 2020, 10, 6453. [Google Scholar] [CrossRef] [PubMed]
- Barata, A.; Malfeito-Ferreira, M.; Loureiro, V. The microbial ecology of wine grape berries. Int. J. Food Microbiol. 2012, 153, 243–259. [Google Scholar] [CrossRef]
- Setati, M.E.; Jacobson, D.; Andong, U.C.; Bauer, F. The Vineyard Yeast Microbiome, a Mixed Model Microbial Map. PLoS ONE 2012, 7, e52609. [Google Scholar] [CrossRef] [PubMed]
- Martins, G.; Miot-Sertier, C.; Lauga, B.; Claisse, O.; Lonvaud-Funel, A.; Soulas, G.; Masneuf-Pomarède, I. Grape berry bacterial microbiota: Impact of the ripening process and the farming system. Int. J. Food Microbiol. 2012, 158, 93–100. [Google Scholar] [CrossRef] [PubMed]
- Bokulich, N.A.; Collins, T.; Masarweh, C.; Allen, G.; Heymann, H.; Ebeler, S.E.; Mills, D.A. Fermentation Behavior Suggest Microbial Contribution to Regional. mBio 2016, 7, 1–12. [Google Scholar] [CrossRef] [PubMed]
- del Carmen Portillo, M.; Mas, A. Analysis of microbial diversity and dynamics during wine fermentation of Grenache grape variety by high-throughput barcoding sequencing. LWT Food Sci. Technol. 2016, 72, 317–321. [Google Scholar] [CrossRef]
- Salvetti, E.; Campanaro, S.; Campedelli, I.; Fracchetti, F.; Gobbi, A.; Tornielli, G.B.; Torriani, S.; Felis, G.E. Whole-metagenome-sequencing-based community profiles of Vitis vinifera L. cv. Corvina berries withered in two post-harvest conditions. Front. Microbiol. 2016, 7, 937. [Google Scholar] [CrossRef]
- Cortesi, P.; Bisiach, M.; Ricciolini, M.; Gadoury, D.M. Cleistothecia of Uncinula necator—An additional source of inoculum in Italian vineyards. Plant Dis. 1997, 81, 922–926. [Google Scholar] [CrossRef]
- Grove, G.G. Perennation of Uncinula necator in vineyards of Eastern Washington. Plant Dis. 2004, 88, 242–247. [Google Scholar] [CrossRef]
- Behar, A.; Jurkevitch, E.; Yuval, B. Bringing back the fruit into fruit fly-bacteria interactions. Mol. Ecol. 2008, 17, 1375–1386. [Google Scholar] [CrossRef]
- Munkvold, G.P. Efficacy of Natural Epiphytes and Colonizers of Grapevine Pruning Wounds for Biological Control of Eutypa Dieback. Phytopathology 1993, 83, 624. [Google Scholar] [CrossRef]
- Komatsu, T.; Kondo, N. Winter habitat of Xylophilus ampelinus, the cause of bacterial blight of grapevine, in Japan. J. Gen. Plant Pathol. 2015, 81, 237–242. [Google Scholar] [CrossRef]
- Hall, S.J.; Dry, I.B.; Blanchard, C.L.; Whitelaw-Weckert, M.A. Phylogenetic relationships of Pseudomonas syringae pv. Syringae isolates associated with bacterial inflorescence rot in Grapevine. Plant Dis. 2016, 100, 607–616. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Bultreys, A.; Kaluzna, M. Bacterial cankers caused by Pseudomonas syringae on stone fruit species with special emphasis on the pathovars syringae and morsprunorum race 1 and race 2. J. Plant Pathol. 2010, 92. [Google Scholar] [CrossRef]
- Pinto, C.; Pinho, D.; Sousa, S.; Pinheiro, M.; Egas, C.; Gomes, A.C. Unravelling the diversity of grapevine microbiome. PLoS ONE 2014, 9, e85622. [Google Scholar] [CrossRef]
- Bukin, Y.S.; Galachyants, Y.P.; Morozov, I.V.; Bukin, S.V.; Zakharenko, A.S.; Zemskaya, T.I. The effect of 16s rRNA region choice on bacterial community metabarcoding results. Sci. Data 2019, 6, 190007. [Google Scholar] [CrossRef]
- Meyer, F.; Paarmann, D.; D’Souza, M.; Olson, R.; Glass, E.M.; Kubal, M.; Paczian, T.; Rodriguez, A.; Stevens, R.; Wilke, A.; et al. The metagenomics RAST server—A public resource for the automatic phylogenetic and functional analysis of metagenomes. BMC Bioinform. 2008, 9, 386. [Google Scholar] [CrossRef]
- Hammer, Ø.; Harper, D.; Ryan, P. PAST: Paleontological statistics software package for education education and data analysis. Palaeontol. Electron. 2001, 4, 1–9. [Google Scholar]
- Heberle, H.; Meirelles, V.G.; da Silva, F.R.; Telles, G.P.; Minghim, R. InteractiVenn: A web-based tool for the analysis of sets through Venn diagrams. BMC Bioinform. 2015, 16. [Google Scholar] [CrossRef]
- Siegwald, L.; Touzet, H.; Lemoine, Y.; Hot, D.; Audebert, C.; Caboche, S. Assessment of common and emerging bioinformatics pipelines for targeted metagenomics. PLoS ONE 2017, 12, e0169563. [Google Scholar] [CrossRef]
- Campisano, A.; Antonielli, L.; Pancher, M.; Yousaf, S.; Pindo, M.; Pertot, I. Bacterial endophytic communities in the grapevine depend on pest management. PLoS ONE 2014, 9, e112763. [Google Scholar] [CrossRef]
- Marzano, M.; Fosso, B.; Manzari, C.; Grieco, F.; Intranuovo, M.; Cozzi, G.; Mulè, G.; Scioscia, G.; Valiente, G.; Tullo, A.; et al. Complexity and dynamics of the winemaking bacterial communities in berries, musts, and wines from apulian grape cultivars through time and space. PLoS ONE 2016, 11, e0157383. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Chen, X.; Zhong, Q.; Huang, Z.; Bai, Z. Relations among epiphytic microbial communities from soil, leaves and grapes of the grapevine. Front. Life Sci. 2017, 10, 73–83. [Google Scholar] [CrossRef]
- Novello, G.; Gamalero, E.; Bona, E.; Boatti, L.; Mignone, F.; Massa, N.; Cesaro, P.; Lingua, G.; Berta, G. The rhizosphere bacterial microbiota of Vitis vinifera cv. Pinot Noir in an integrated pest management vineyard. Front. Microbiol. 2017, 8, 1528. [Google Scholar] [CrossRef] [PubMed]
- Mezzasalma, V.; Sandionigi, A.; Bruni, I.; Bruno, A.; Lovicu, G.; Casiraghi, M.; Labra, M. Grape microbiome as a reliable and persistent signature of field origin and environmental conditions in Cannonau wine production. PLoS ONE 2017, 12, e0184615. [Google Scholar] [CrossRef]
- Mezzasalma, V.; Sandionigi, A.; Guzzetti, L.; Galimberti, A.; Grando, M.S.; Tardaguila, J.; Labra, M. Geographical and cultivar features differentiate grape microbiota in Northern Italy and Spain vineyards. Front. Microbiol. 2018, 9, 946. [Google Scholar] [CrossRef]
- Liu, D.; Zhang, P.; Chen, D.; Howell, K. From the Vineyard to the Winery: How Microbial Ecology Drives Regional Distinctiveness of Wine. Front. Microbiol. 2019, 10, 2679. [Google Scholar] [CrossRef]
- Chou, M.Y.; Vanden Heuvel, J.; Bell, T.H.; Panke-Buisse, K.; Kao-Kniffin, J. Vineyard under-vine floor management alters soil microbial composition, while the fruit microbiome shows no corresponding shifts. Sci. Rep. 2018, 8, 11039. [Google Scholar] [CrossRef]
- Canfora, L.; Vendramin, E.; Felici, B.; Tarricone, L.; Florio, A.; Benedetti, A. Vineyard microbiome variations during different fertilisation practices revealed by 16s rRNA gene sequencing. Appl. Soil Ecol. 2018, 125, 71–80. [Google Scholar] [CrossRef]
- Gupta, V.V.S.R.; Bramley, R.G.V.; Greenfield, P.; Yu, J.; Herderich, M.J. Vineyard soil microbiome composition related to rotundone concentration in Australian cool climate “peppery” Shiraz grapes. Front. Microbiol. 2019, 10, 1607. [Google Scholar] [CrossRef]
- Campanaro, S.; Treu, L.; Vendramin, V.; Bovo, B.; Giacomini, A.; Corich, V. Metagenomic analysis of the microbial community in fermented grape marc reveals that Lactobacillus fabifermentans is one of the dominant species: Insights into its genome structure. Appl. Microbiol. Biotechnol. 2014, 98, 6015–6037. [Google Scholar] [CrossRef] [PubMed]
- Deyett, E.; Rolshausen, P.E. Endophytic microbial assemblage in grapevine. FEMS Microbiol. Ecol. 2020, 96, fiaa053. [Google Scholar] [CrossRef] [PubMed]
- Gobbi, A.; Kyrkou, I.; Filippi, E.; Ellegaard-Jensen, L.; Hansen, L.H. Seasonal epiphytic microbial dynamics on grapevine leaves under biocontrol and copper fungicide treatments. Sci. Rep. 2020, 10, 681. [Google Scholar] [CrossRef] [PubMed]
- Pinto, C.; Pinho, D.; Cardoso, R.; Custódio, V.; Fernandes, J.; Sousa, S.; Pinheiro, M.; Egas, C.; Gomes, A.C. Wine fermentation microbiome: A landscape from different Portuguese wine appellations. Front. Microbiol. 2015, 6. [Google Scholar] [CrossRef]
- Bokulich, N.A.; Joseph, C.M.L.; Allen, G.; Benson, A.K.; Mills, D.A. Next-generation sequencing reveals significant bacterial diversity of botrytized wine. PLoS ONE 2012, 7, e36357. [Google Scholar] [CrossRef]
- Zarraonaindia, I.; Gilbert, J.A. Understanding grapevine-microbiome interactions: Implications for viticulture industry. Microb. Cell 2015, 2, 171–173. [Google Scholar] [CrossRef]
- Alonso, A.; De Celis, M.; Ruiz, J.; Vicente, J.; Navascués, E.; Acedo, A.; Ortiz-Álvarez, R.; Belda, I.; Santos, A.; Gómez-Flechoso, M.Á.; et al. Looking at the origin: Some insights into the general and fermentative microbiota of vineyard soils. Fermentation 2019, 5, 78. [Google Scholar] [CrossRef]
- Burns, K.N.; Bokulich, N.A.; Cantu, D.; Greenhut, R.F.; Kluepfel, D.A.; O’Geen, A.T.; Strauss, S.L.; Steenwerth, K.L. Vineyard soil bacterial diversity and composition revealed by 16S rRNA genes: Differentiation by vineyard management. Soil Biol. Biochem. 2016, 103, 337–348. [Google Scholar] [CrossRef]
- Hall, M.E.; O’Bryon, I.; Wilcox, W.F.; Osier, M.V.; Cadle-Davidson, L. The epiphytic microbiota of sour rot-affected grapes differs minimally from that of healthy grapes, indicating causal organisms are already present on healthy berries. PLoS ONE 2019, 14, e0211378. [Google Scholar] [CrossRef]
- Schriefer, A.E.; Cliften, P.F.; Hibberd, M.C.; Sawyer, C.; Brown-Kennerly, V.; Burcea, L.; Klotz, E.; Crosby, S.D.; Gordon, J.I.; Head, R.D. A multi-amplicon 16S rRNA sequencing and analysis method for improved taxonomic profiling of bacterial communities. J. Microbiol. Methods 2018, 154, 6–13. [Google Scholar] [CrossRef]
- Bulgarelli, D.; Schlaeppi, K.; Spaepen, S.; Van Themaat, E.V.L.; Schulze-Lefert, P. Structure and functions of the bacterial microbiota of plants. Annu. Rev. Plant Biol. 2013, 64, 807–838. [Google Scholar] [CrossRef]
- Kim, M.J.; Jeon, C.W.; Cho, G.; Kim, D.R.; Kwack, Y.B.; Kwak, Y.S. Comparison of microbial community structure in kiwifruit pollens. Plant Pathol. J. 2018, 34, 143–149. [Google Scholar] [CrossRef] [PubMed]
- Lauber, C.L.; Hamady, M.; Knight, R.; Fierer, N. Pyrosequencing-based assessment of soil pH as a predictor of soil bacterial community structure at the continental scale. Appl. Environ. Microbiol. 2009, 75, 5111–5120. [Google Scholar] [CrossRef]
- Philippot, L.; Raaijmakers, J.M.; Lemanceau, P.; Van Der Putten, W.H. Going back to the roots: The microbial ecology of the rhizosphere. Nat. Rev. Microbiol. 2013, 11, 789–799. [Google Scholar] [CrossRef] [PubMed]
- Leveau, J.H.J.; Tech, J.J. Grapevine microbiomics: Bacterial diversity on grape leaves and berries revealed by high-throughput sequence analysis of 16S rRNA amplicons. Acta Hortic. 2011, 905, 31–42. [Google Scholar] [CrossRef]
- Miura, T.; Sánchez, R.; Castañeda, L.E.; Godoy, K.; Barbosa, O. Is microbial terroir related to geographic distance between vineyards? Environ. Microbiol. Rep. 2017, 9, 742–749. [Google Scholar] [CrossRef] [PubMed]
- Hendgen, M.; Hoppe, B.; Döring, J.; Friedel, M.; Kauer, R.; Frisch, M.; Dahl, A.; Kellner, H. Effects of different management regimes on microbial biodiversity in vineyard soils. Sci. Rep. 2018, 8, 9393. [Google Scholar] [CrossRef] [PubMed]
- Morgan, H.H.; du Toit, M.; Setati, M.E. The grapevine and wine microbiome: Insights from high-throughput amplicon sequencing. Front. Microbiol. 2017, 8. [Google Scholar] [CrossRef]
- Stefanini, I.; Cavalieri, D. Metagenomic approaches to investigate the contribution of the vineyard environment to the quality of wine fermentation: Potentials and difficulties. Front. Microbiol. 2018, 9, 991. [Google Scholar] [CrossRef]
- Kearsey, M. The principles of QTL analysis (a minimal mathematics approach). J. Exp. Bot. 1998, 49, 1619–1623. [Google Scholar] [CrossRef]
- Compant, S.; Samad, A.; Faist, H.; Sessitsch, A. A review on the plant microbiome: Ecology, functions, and emerging trends in microbial application. J. Adv. Res. 2019, 19, 29–37. [Google Scholar] [CrossRef] [PubMed]
- Bokulich, N.A.; Thorngate, J.H.; Richardson, P.M.; Mills, D.A. Microbial biogeography of wine grapes is conditioned by cultivar, vintage, and climate. Proc. Natl. Acad. Sci. USA 2014, 111, 139–148. [Google Scholar] [CrossRef] [PubMed]
- Morrison-Whittle, P.; Lee, S.A.; Goddard, M.R. Fungal communities are differentially affected by conventional and biodynamic agricultural management approaches in vineyard ecosystems. Agric. Ecosyst. Environ. 2017, 246, 306–313. [Google Scholar] [CrossRef]
- Drumonde-Neves, J.; Franco-Duarte, R.; Vieira, E.; Mendes, I.; Lima, T.; Schuller, D.; Pais, C. Differentiation of Saccharomyces cerevisiae populations from vineyards of the Azores Archipelago: Geography vs. Ecology. Food Microbiol. 2018, 74, 151–162. [Google Scholar] [CrossRef]
- Adesemoye, A.O.; Obini, M.; Ugoji, E.O. Comparison of plant growth-promotion with Pseudomonas aeruginosa and Bacillus subtilis in three vegetables. Braz. J. Microbiol. 2008, 39, 423–426. [Google Scholar] [CrossRef]
- Ahemad, M.; Kibret, M. Mechanisms and applications of plant growth promoting rhizobacteria: Current perspective. J. King Saud Univ. Sci. 2014, 26, 1–20. [Google Scholar] [CrossRef]
- Sarris, P.F.; Trantas, E.A.; Mpalantinaki, E.; Ververidis, F.; Goumas, D.E. Pseudomonas viridiflava, a multi host plant pathogen with significant genetic variation at the molecular level. PLoS ONE 2012, 7, e036090. [Google Scholar] [CrossRef]
- Bartoli, C.; Berge, O.; Monteil, C.L.; Guilbaud, C.; Balestra, G.M.; Varvaro, L.; Jones, C.; Dangl, J.L.; Baltrus, D.A.; Sands, D.C.; et al. The Pseudomonas viridiflava phylogroups in the P.syringae species complex are characterized by genetic variability and phenotypic plasticity of pathogenicity-related traits. Environ. Microbiol. 2014, 16, 2301–2315. [Google Scholar] [CrossRef]
- Rozpdek, P.; Domka, A.; Turnau, K. Chapter 29 Mycorrhizal Fungi and Accompanying Microorganisms in Improving Phytoremediation Techniques. In The Fungal Community: Its Organization and Role in the Ecosystem; CRC Press: Boca Raton, FL, USA, 2017; ISBN 9781498706650. [Google Scholar]
- Bassas-Galia, M.; Nogales, B.; Arias, S.; Rohde, M.; Timmis, K.N.; Molinari, G. Plant original Massilia isolates producing polyhydroxybutyrate, including one exhibiting high yields from glycerol. J. Appl. Microbiol. 2012, 112, 443–454. [Google Scholar] [CrossRef]
- Rastogi, G.; Sbodio, A.; Tech, J.J.; Suslow, T.V.; Coaker, G.L.; Leveau, J.H.J. Leaf microbiota in an agroecosystem: Spatiotemporal variation in bacterial community composition on field-grown lettuce. ISME J. 2012, 6, 1812–1822. [Google Scholar] [CrossRef]
- Yashiro, E.; McManus, P.S. Effect of streptomycin treatment on bacterial community structure in the apple phyllosphere. PLoS ONE 2012, 7, e37131. [Google Scholar] [CrossRef] [PubMed]
- Innerebner, G.; Knief, C.; Vorholt, J.A. Protection of Arabidopsis thaliana against leaf-pathogenic Pseudomonas syringae by Sphingomonas strains in a controlled model system. Appl. Environ. Microbiol. 2011, 77, 3202–3210. [Google Scholar] [CrossRef] [PubMed]
- Qin, C.; Tao, J.; Liu, T.; Liu, Y.; Xiao, N.; Li, T.; Gu, Y.; Yin, H.; Meng, D. Responses of phyllosphere microbiota and plant health to application of two different biocontrol agents. AMB Express 2019, 9. [Google Scholar] [CrossRef]
- Buonaurio, R.; Stravato, V.M.; Kosako, Y.; Fujiwara, N.; Naka, T.; Kobayashi, K.; Cappelli, C.; Yabuuchi, E. Sphingomonas melonis sp. nov., a novel pathogen that causes brown spots on yellow Spanish melon fruits. Int. J. Syst. Evol. Microbiol. 2002, 52, 2081–2087. [Google Scholar] [CrossRef] [PubMed]
- Lim, J.A.; Lee, D.H.; Kim, B.Y.; Heu, S. Draft genome sequence of Pantoea agglomerans R190, a producer of antibiotics against phytopathogens and foodborne pathogens. J. Biotechnol. 2014, 188, 7–8. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Feng, L.; Fan, X.; Jiang, J.; Zheng, X.-b.; Sun, H.; Chonghuai, L. Genome-wide assessment of population structure, linkage disequilibrium and resistant QTLs in Chinese wild grapevine. Sci. Hortic. 2017, 215, 59–64. [Google Scholar] [CrossRef]
- Morrison-Whittle, P.; Goddard, M.R. Quantifying the relative roles of selective and neutral processes in defining eukaryotic microbial communities. ISME J. 2015, 9, 2003–2011. [Google Scholar] [CrossRef]
- Keller, M. Phenology and Growth Cycle; Academic Press: Cambridge, MA, USA, 2015; ISBN 9780124199873. [Google Scholar]
| Classification | Cultivar | Main Purpose of Use | Fruit Color |
|---|---|---|---|
| Greek | Agiorgitiko | Wine | B |
| Assyrtiko | Wine | W | |
| Fraoula | Table grapes | R | |
| Kakotrygis | Wine | R | |
| Karabraimis | Wine | Β | |
| Korinthiaki | Wine, raisins | B | |
| Kotselina | Wine | B | |
| Limniona | Wine | Β | |
| Mavro (from Cyprus) | Wine | B | |
| Mavrodafni | Wine | Β | |
| Mavrokorakas | Wine | B | |
| Moschofilero | Wine | B | |
| Moschatolefko | Wine | W | |
| Razaki | Table grapes | W | |
| Robola | Wine | W | |
| Roditis | Wine | R | |
| Savvatiano | Wine | W | |
| Serifiotiko | Wine | B | |
| Tsaousi | Wine | W | |
| Voidomatis | Wine | B | |
| Vertzami | Wine | B | |
| Vidiano | Wine | W | |
| Xinomavro | Wine | B | |
| Malagouzia | Wine | W | |
| International | Cabernet Sauvignon | Wine | B |
| Cardinal | Table grapes | B | |
| Crimson | Table grapes | R | |
| Grenache | Wine | B | |
| Italia | Table grapes | W | |
| Merlot | Wine | B | |
| Perlette | Table grapes | W | |
| Ralli | Table grapes | R | |
| Riesling | Wine | W | |
| Sauvignon Blanc | Wine | W | |
| Soultanina | Table grapes, raisins | W | |
| Syrah | Wine | B | |
| Victoria | Table grapes | W |
| Cultivar | Good’s Coverage | OTUs Excluding Singletons | Chao-1 | OTUs Including Singletons | Shannon-H | Simpson-1D | Reads Obtained |
|---|---|---|---|---|---|---|---|
| Limniona | 0.997 | 164 | 422.1 | 266 | 2.85 | 0.891 | 31,399 |
| Voidomatis | 0.997 | 109 | 489.3 | 230 | 1.80 | 0.587 | 38,102 |
| Victoria | 0.992 | 113 | 399.0 | 217 | 2.71 | 0.866 | 12,826 |
| Vertzami | 0.996 | 101 | 453.6 | 207 | 2.38 | 0.808 | 23,965 |
| Karabraimis | 0.996 | 105 | 364.4 | 197 | 2.85 | 0.886 | 24,956 |
| Crimson | 0.995 | 96 | 471.0 | 196 | 2.44 | 0.837 | 19,410 |
| Tsaousi | 0.992 | 96 | 357.9 | 194 | 2.92 | 0.908 | 11,580 |
| Xinomavro | 0.996 | 100 | 424.6 | 192 | 2.71 | 0.884 | 24,394 |
| Malagouzia | 0.997 | 86 | 451.0 | 191 | 2.20 | 0.828 | 31,896 |
| Serifiotiko | 0.988 | 82 | 353.4 | 178 | 2.61 | 0.843 | 7839 |
| Mavrokorakas | 0.983 | 79 | 373.3 | 175 | 3.05 | 0.920 | 5798 |
| Vidiano | 0.983 | 77 | 310.0 | 165 | 2.38 | 0.814 | 5271 |
| Mavrodafni | 0.996 | 84 | 334.2 | 163 | 2.64 | 0.887 | 19,820 |
| Roditis | 0.994 | 88 | 290.6 | 162 | 2.74 | 0.887 | 11,964 |
| Merlot | 0.993 | 80 | 341.0 | 161 | 2.56 | 0.872 | 11,869 |
| Cardinal | 0.997 | 81 | 352.6 | 160 | 1.76 | 0.620 | 25,021 |
| Razaki | 0.997 | 82 | 303.1 | 157 | 2.21 | 0.794 | 22,208 |
| Italia | 0.994 | 68 | 403.5 | 148 | 2.31 | 0.805 | 14,163 |
| Kotselina | 0.995 | 81 | 274.6 | 148 | 2.74 | 0.898 | 13,264 |
| Soultanina | 0.997 | 67 | 434.3 | 147 | 1.85 | 0.704 | 27,181 |
| Korinthiaki | 0.994 | 62 | 543.7 | 147 | 2.11 | 0.748 | 14,552 |
| Moschatolefko | 0.992 | 64 | 379.4 | 147 | 2.28 | 0.823 | 9944 |
| Ralli | 0.989 | 60 | 379.2 | 142 | 2.03 | 0.712 | 7139 |
| Savvatiano | 0.999 | 72 | 246.5 | 137 | 1.23 | 0.452 | 44,328 |
| Kakotrygis | 0.990 | 53 | 284.8 | 125 | 2.37 | 0.847 | 7182 |
| Assyrtiko | 0.992 | 60 | 255.1 | 120 | 2.42 | 0.852 | 7637 |
| Fraoula | 0.995 | 71 | 188.2 | 119 | 2.34 | 0.802 | 9883 |
| Perlette | 0.988 | 49 | 292.2 | 117 | 2.27 | 0.829 | 5587 |
| Agiorgitiko | 0.993 | 48 | 321.0 | 113 | 2.05 | 0.786 | 9735 |
| Sauvignon Blanc | 0.990 | 49 | 239.0 | 106 | 1.87 | 0.665 | 5480 |
| Riesling | 0.991 | 44 | 410.0 | 105 | 2.11 | 0.783 | 6821 |
| Moschofilero | 0.993 | 50 | 217.8 | 103 | 2.37 | 0.821 | 7154 |
| Mavro | 0.977 | 51 | 191.4 | 103 | 2.82 | 0.889 | 2301 |
| Syrah | 0.998 | 45 | 193.8 | 102 | 1.21 | 0.537 | 30,409 |
| Robola | 0.997 | 49 | 204.1 | 96 | 1.77 | 0.738 | 15,095 |
| Grenache | 0.987 | 37 | 215.3 | 90 | 2.06 | 0.791 | 4158 |
| Cabernet Sauvignon | 0.993 | 52 | 183.1 | 88 | 2.55 | 0.884 | 5188 |
| Cultivar | # of Unique Genera Detected | Unique Genera |
|---|---|---|
| Moschofilero, Robola, Cabernet Sauvignon, Moschatolefko, Riesling, Sauvignon Blanc, Korinthiaki, Mavrodafni, Grenache, Agiorgitiko, Kakotrygis | Zero | --- |
| Tsaousi | One | Rhodobacter |
| Italia | One | Myroides |
| Savvatiano | One | Zymomonas |
| Mavro | One | Pediococcus |
| Ralli | One | Oceanobacillus |
| Serifiotiko | One | Gluconobacter |
| Perlette | One | Pigmentiphaga |
| Vertzami | One | Buchnera |
| Asyrtiko | One | Janibacter |
| Fraoula | Two | Verminephrobacter, Pseudochrobactrum |
| Soultanina | Two | Spiroplasma, Sodalis |
| Cardinal | Two | Cellulophaga, Azotobacter |
| Vidiano | Two | Symploca, Enterococcus |
| Kotselina | Two | Taylorella, Megamonas |
| Syrah | Two | Ralstonia, Methylophilus |
| Crimson | Two | Tetragenococcus, Aerococcus |
| Razaki | Two | Nonomuraea, Brochothrix |
| Karabraimis | Two | Pectobacterium, Mobiluncus |
| Malagouzia | Three | Ureibacillus, Oxalobacter, Acidithiobacillus |
| Mavrokorakas | Three | Geobacillus, CandidatusSolibacter, CandidatusPhytoplasma |
| Roditis | Three | Saccharothrix, Rhodothalassium, Renibacterium |
| Voidomatis | Three | Serinicoccus, Lysinibacillus, Bradyrhizobium |
| Xinomavro | Four | Promicromonospora, Microcystis, Herminiimonas, CandidatusAmoebophilus |
| Victoria | Eight | Williamsia, Treponema, Terrimonas, Porphyromonas, Granulicatella, Gemella, Fusobacterium, Abiotrophia |
| Limniona | Thirteen | Tetrathiobacter, Streptoalloteichus, Streptacidiphilus, Pseudonocardia, Planomicrobium, Nesterenkonia, Lechevalieria, Halorhodospira, Gordonia, Deinococcus, Candidatus, Cardinium, Brachybacterium, Anabaenopsis |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Awad, M.; Giannopoulos, G.; Mylona, P.V.; Polidoros, A.N. Genotype May Influence Bacterial Diversity in Bark and Bud of Vitis vinifera Cultivars Grown under the Same Environment. Appl. Sci. 2020, 10, 8405. https://doi.org/10.3390/app10238405
Awad M, Giannopoulos G, Mylona PV, Polidoros AN. Genotype May Influence Bacterial Diversity in Bark and Bud of Vitis vinifera Cultivars Grown under the Same Environment. Applied Sciences. 2020; 10(23):8405. https://doi.org/10.3390/app10238405
Chicago/Turabian StyleAwad, Murad, Georgios Giannopoulos, Photini V. Mylona, and Alexios N. Polidoros. 2020. "Genotype May Influence Bacterial Diversity in Bark and Bud of Vitis vinifera Cultivars Grown under the Same Environment" Applied Sciences 10, no. 23: 8405. https://doi.org/10.3390/app10238405
APA StyleAwad, M., Giannopoulos, G., Mylona, P. V., & Polidoros, A. N. (2020). Genotype May Influence Bacterial Diversity in Bark and Bud of Vitis vinifera Cultivars Grown under the Same Environment. Applied Sciences, 10(23), 8405. https://doi.org/10.3390/app10238405









