Current Advances in Plant Growth Promoting Bacteria Alleviating Salt Stress for Sustainable Agriculture
Abstract
1. Introduction
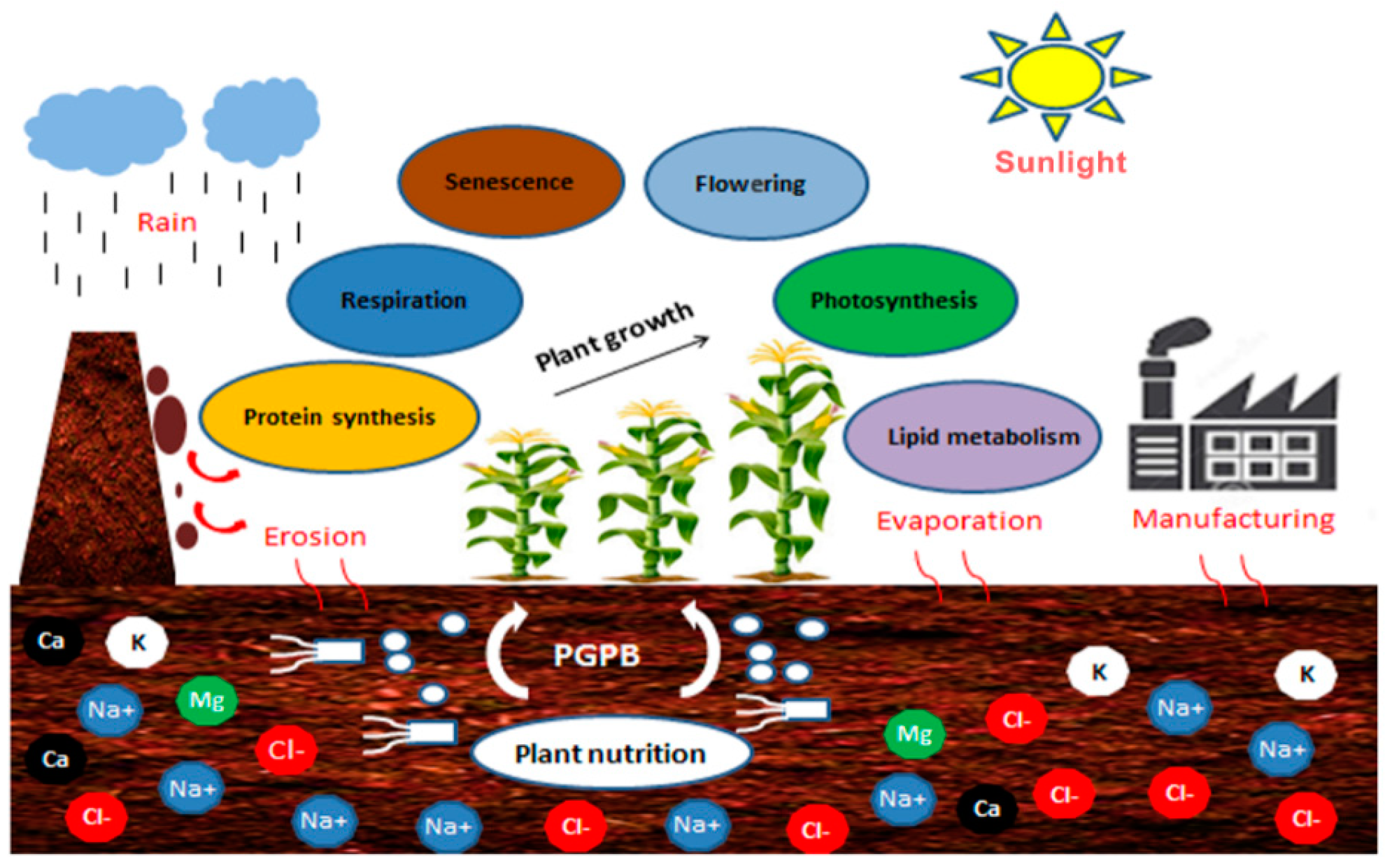
2. Implication of PGPB on Soil Desalinization
3. Impacts of Salinity on Crops
4. Salinity Origin and Its Impacts on Plants
4.1. Seed Germination
4.2. Primary Metabolism
4.2.1. Photosynthesis
4.2.2. Respiration
4.2.3. Senescence
4.2.4. Flowering
4.2.5. Protein Synthesis
4.2.6. Lipid Metabolism
5. Impacts of Salinity on Soil
5.1. Soil Biological Activity
5.2. Soil Microbial Function and Diversity
6. Halophile PGPB for Sustainable Agriculture
6.1. Biofertilization
6.2. Biopesticides
6.3. Bioremediation
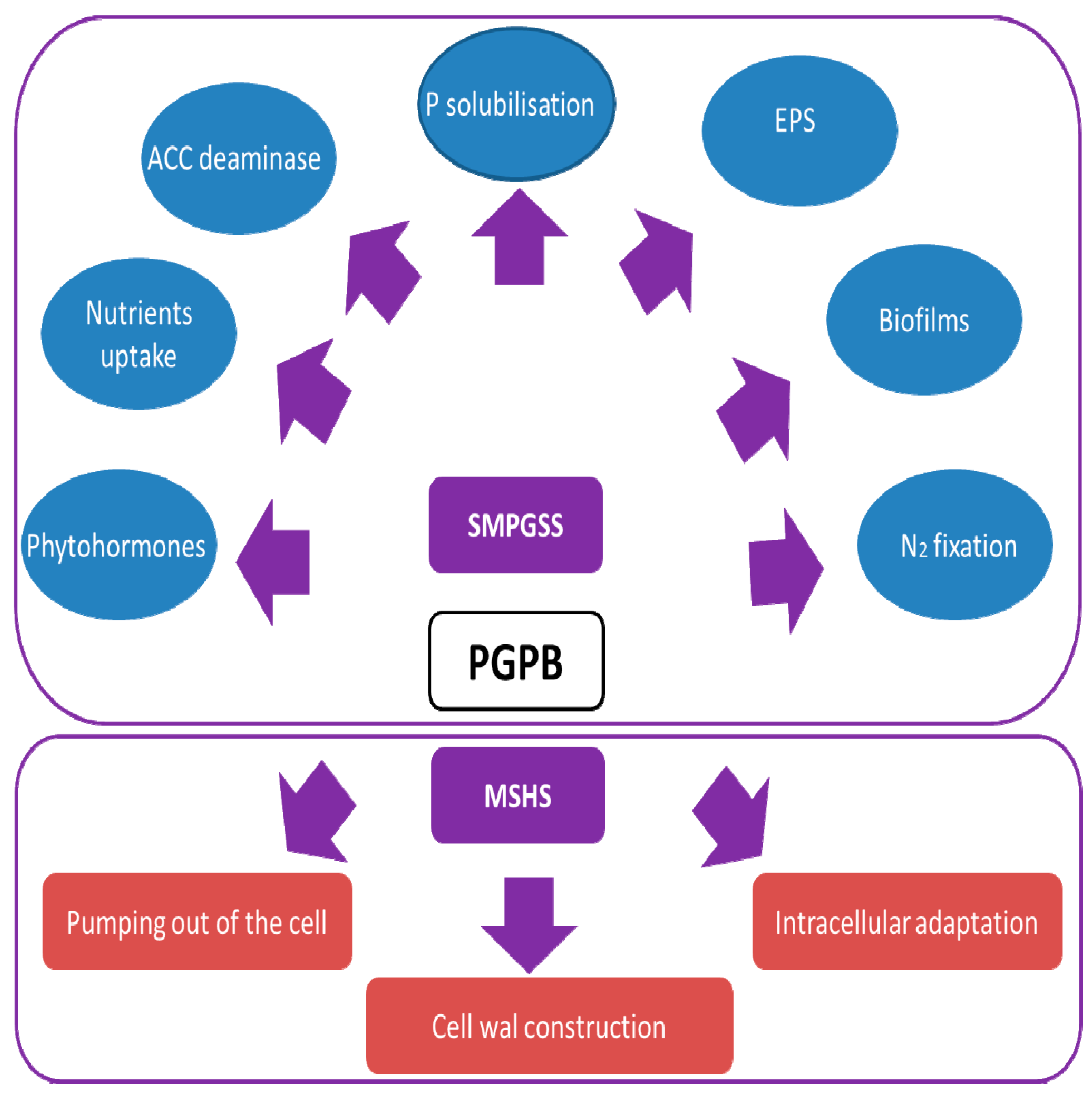
7. Mechanisms of PGPB (Plant Growth Promoting Bacteria) towards Salinity Tolerance
7.1. Direct Mechanisms
7.1.1. Nitrogen Fixation
7.1.2. Phosphate Solubilization
7.1.3. Production of Phytohormones
7.1.4. Exopolysaccharides and Biofilm Formation
7.1.5. Enhancement of Plant Nutrient Uptake
7.1.6. Osmolytes Accumulation
7.2. Indirect Mechanisms
7.2.1. ACC Deaminase
7.2.2. Induced Systemic Resistance
7.2.3. Ethylene
8. PGPB Inoculants under Salinity Can Reduce Chemical Fertilization
9. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Glick, B.R. Plant growth-promoting bacteria: Mechanisms and applications. Scientifica 2012, 2012, 1–15. [Google Scholar] [CrossRef]
- Chandra, D.; Srivastava, R.; Gupta, V.V.; Franco, C.M.; Sharma, A.K. Evaluation of ACC-deaminase-producing rhizobacteria to alleviate water-stress impacts in wheat (Triticum aestivum L.) plants. Can. J. Microbiol. 2019, 65, 387–403. [Google Scholar] [CrossRef] [PubMed]
- Grime, J.P. Evidence for the existence of three primary strategies in plants and its relevance to ecological and evolutionary theory. Am. Nat. 1977, 111, 169–1194. [Google Scholar] [CrossRef]
- Kumar, A.; Verma, J.P. Does plant–microbe interaction confer stress tolerance in plants: A review? Microbiol. Res. 2018, 207, 41–52. [Google Scholar] [CrossRef]
- Machado, R.M.A.; Serralheiro, R.P. Soil salinity: Effect on vegetable crop growth. Management practices to prevent and mitigate soil salinization. Horticulturae 2017, 3, 30. [Google Scholar] [CrossRef]
- Pimentel, D.; Berger, B.; Filiberto, D.; Newton, M.; Wolfe, B.; Karabinakis, E.; Clark, S.; Poon, E.; Abbett, E.; Nandaopal, S. Water Resources: Agricultural and Environmental Issues. BioScience 2004, 54, 909–918. [Google Scholar] [CrossRef]
- Numan, M.; Bashir, S.; Khan, Y.; Mumtaz, R.; Shinwari, Z.K.; Khan, A.L.; Khan, A.; Ahmed, A.H. Plant growth promoting bacteria as an alternative strategy for salt tolerance in plants: A review. Microbiol. Res. 2018, 209, 21–32. [Google Scholar] [CrossRef]
- Yadav, A.N.; Saxena, A.K. Biodiversity and biotechnological applications of halophilic microbes for sustainable agriculture. J. Appl. Biol. Biotechnol. 2018, 6, 48–55. [Google Scholar] [CrossRef]
- Munns, R. Genes and salt tolerance: Bringing them together. New Phytol. 2005, 167, 645–663. [Google Scholar] [CrossRef]
- Jamil, A.; Riaz, S.; Ashraf, M.; Foolad, M.R. Gene expression profiling of plants under salt stress. Crit. Rev. Plant Sci. 2011, 30, 435–458. [Google Scholar] [CrossRef]
- Arora, S. Halotolerant Microbes for Amelioration of Salt-Affected Soils for Sustainable Agriculture. In Phyto-Microbiome in Stress Regulation; Kumar, M., Kumar, V., Prasad, R., Eds.; Springer: Singapore, 2020; pp. 323–343. [Google Scholar]
- Zhang, K.; Shi, Y.; Cui, X.; Yue, P.; Li, K.; Liu, X.; Tripathi, B.M.; Chu, H. Salinity is a key determinant for soil microbial communities in a desert ecosystem. MSystems 2019, 4, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Chu, T.N.; Tran, B.T.H.; Hoang, M.T.T. Plant growth-promoting rhizobacterium Pseudomonas PS01 induces salt tolerance in Arabidopsis thaliana. BMC Res. Notes 2019, 12, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Paul, D.; Lade, H. Plant-growth-promoting rhizobacteria to improve crop growth in saline soils: A review. Agron. Sustain. Dev. 2014, 34, 737–752. [Google Scholar] [CrossRef]
- Vessey, J.K. Plant growth promoting rhizobacteria as biofertilizers. Plant Soil 2003, 255, 571–586. [Google Scholar] [CrossRef]
- Souza, R.D.; Ambrosini, A.; Passaglia, L.M. Plant growth-promoting bacteria as inoculants in agricultural soils. Genet. Mol. Biol. 2015, 38, 401–419. [Google Scholar] [CrossRef]
- Bakka, K.; Challabathula, D. Amelioration of Salt Stress Tolerance in Plants by Plant Growth-Promoting Rhizobacteria: Insights from “Omics” Approaches. In Plant Microbe Symbiosis; Varma, A., Tripathi, S., Prasad, R., Eds.; Springer: Cham, Switzerland, 2020; pp. 303–330. [Google Scholar]
- El-Akhdar, I.; Elsakhawy, T.; Abo-Koura, H.A. Alleviation of Salt Stress on Wheat (Triticum aestivum L.) by Plant Growth Promoting Bacteria strains Bacillus halotolerans MSR-H4 and Lelliottia amnigena MSR-M49. J. Adv. Appl. Microbiol. 2020, 20, 44–58. [Google Scholar] [CrossRef]
- Obledo, E.N.; Barraga’n-Barraga’n, L.B.; Gutie’rrez-Gonza’lez, P.; Ramı’rez-Herna’ndez, B.C.; Ramı’rez, J.J.; Rodrı’guez-Garay, B. Increased photosyntethic efficiency generated by fungal symbiosis in Agave victoria-reginae. Plant Cell Tissue Organ Cult. 2003, 74, 237–241. [Google Scholar] [CrossRef]
- Lynch, J.M. The Rhizosphere; Wiley-Interscience: Chichester, UK, 1990; p. 4. [Google Scholar]
- Schoenborn, L.; Yates, P.S.; Grinton, B.E.; Hugenholtz, P.; Janssen, P.H. Liquid serial dilution is inferior to solid media for isolation of cultures representative of the phylum-level diversity of soil bacteria. Appl. Environ. Environ. Microbiol. 2004, 70, 4363–4366. [Google Scholar] [CrossRef]
- Egamberdieva, D.; Wirth, S.; Bellingrath-Kimura, S.D.; Mishra, J.; Arora, N.K. Salt-Tolerant Plant Growth Promoting Rhizobacteria for Enhancing Crop Productivity of Saline Soils. Front. Microbiol. 2019, 10, 1–18. [Google Scholar] [CrossRef]
- Whipps, J.M. Microbial interactions and biocontrol in the rhizosphere. J. Exp. Bot. 2001, 52, 487–511. [Google Scholar] [CrossRef]
- Ilangumaran, G.; Smith, D.L. Plant Growth Promoting Rhizobacteria in Amelioration of Salinity Stress: A Systems Biology Perspective. Front. Plant Sci. 2017, 8, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Ruppel, S.; Franken, P.; Witzel, K. Properties of the halophyte microbiome and their implications for plant salt tolerance. Funct. Plant Biol. 2013, 40, 940–951. [Google Scholar] [CrossRef] [PubMed]
- Kadmiri, I.M.; Chaouqui, L.; Azaroual, S.E.; Sijilmassi, B.; Yaakoubi, K.; Wahby, I. Phosphate-solubilizing and auxin-producing rhizobacteria promote plant growth under saline conditions. Arab. J. Sci. Eng. 2018, 43, 3403–3415. [Google Scholar] [CrossRef]
- Ismael, R.R.; Ahmad, E.S.; Abdel-Fattah, M.; Omar, M.N.; Azzam, C.R.; Zahran, H.H. Effect of plant growth-promoting rhizobacteria on growth and symbiotic nitrogen fixation of Vicia faba plants under salt stress. Plant Sci. J. 2018, 7, 1–19. [Google Scholar]
- Braud, A.; Jézéquel, K.; Bazot, S.; Lebeau, T. Enhanced phytoextraction of an agricultural Cr-and Pb-contaminated soil by bioaugmentation with siderophore-producing bacteria. Chemosphere 2009, 74, 280–286. [Google Scholar] [CrossRef]
- Kumar, A.; Verma, J.P. The role of microbes to improve crop productivity and soil health. In Ecological Wisdom Inspired Restoration Engineering; Achal, V., Mukherjee, A., Eds.; Springer: Singapore, 2019; pp. 249–265. [Google Scholar]
- Kumar, A.; Singh, S.; Gaurav, A.K.; Srivastava, S.; Verma, J.P. Plant Growth-Promoting Bacteria: Biological Tools for the Mitigation of Salinity Stress in Plants. Front. Microbiol. 2020, 11, 1216. [Google Scholar] [CrossRef]
- Kamran, M.; Parveen, A.; Ahmar, S.; Malik, Z.; Hussain, S.; Chattha, M.S.; Saleem, M.H.; Adil, M.; Heidari, P.; Chen, J.T. An overview of hazardous impacts of soil salinity in crops, tolerance mechanisms, and amelioration through selenium supplementation. Int. J. Mol. Sci. 2020, 21, 148. [Google Scholar] [CrossRef]
- Munns, R.; Gilliham, M. Salinity tolerance of crops—What is the cost? New Phytol. 2015, 208, 668–673. [Google Scholar] [CrossRef]
- Volkmar, K.M.; Hu, Y.; Steppuhn, H. Physiological responses of plants to salinity: A review. Can. J. Plant Sci. 1998, 78, 19–27. [Google Scholar] [CrossRef]
- De Souza Silva, C.M.M.; Fay, E.F. Effect of salinity on soil microorganisms. Soil Health Land Use Manag. 2012, 10, 177–198. [Google Scholar] [CrossRef]
- Zhang, H.X.; Irving, L.J.; McGill, C.; Matthew, C.; Zhou, D.; Kemp, P. The effects of salinity and osmotic stress on barley germination rate: Sodium as an osmotic regulator. Ann. Bot. 2010, 106, 1027–1035. [Google Scholar] [CrossRef] [PubMed]
- Keating, B.; Fisher, M. Comparative tolerance of tropical grain legumes to salinity. Aust. J. Agric. Res. 1985, 36, 373–383. [Google Scholar] [CrossRef]
- Abbaszadeh, F.; Rameeh, V.; Charati, A. Salinity Stress Indices of Seed Yield and Nutrient Compositions in Rapeseed. Int. J. Biol. 2012, 4, 154–162. [Google Scholar] [CrossRef]
- Ghanem, M.E.; van Elteren, J.; Albacete, A.; Quinet, M.; Martínez-Andújar, C.; Kinet, J.M.; Pérez-Alfocea, F.; Lutts, S. Impact of salinity on early reproductive physiology of tomato (Solanum lycopersicum) in relation to a heterogeneous distribution of toxic ions in flower organs. Funct. Plant Biol. 2009, 36, 125–136. [Google Scholar] [CrossRef] [PubMed]
- Hussain, S.; Shaukat, M.; Ashraf, M.; Zhu, C.; Jin, Q.; Zhang, J. Salinity stress in arid and semi-arid climates: Effects and management in field crops. In Climate Change and Agriculture; Hussain, S., Ed.; IntechOpen: London, UK, 2019; pp. 1–26. [Google Scholar]
- Pitman, M.G.; Läuchli, A. Global impact of salinity and agricultural ecosystems. In Salinity: Environment-Plants-Molecules; Läuchli, A., Lüttge, U., Eds.; Springer: Dordrecht, The Netherlands, 2002; pp. 3–20. [Google Scholar]
- Essington, M.E. Soil and Water Chemistry—An Integrative Approach; CRC Press: Boca Raton, FL, USA, 2004; p. 500. [Google Scholar]
- Zhang, H.H.; Xu, N.; Wu, X.; Wang, J.; Ma, S.; Li, X.; Sun, G. Effects of four types of sodium salt stress on plant growth and photosynthetic apparatus in sorghum leaves. J. Plant Interact. 2018, 13, 506–513. [Google Scholar] [CrossRef]
- Negrão, S.; Schmöckel, S.M.; Tester, M. Evaluating physiological responses of plants to salinity stress. Ann. Bot. 2017, 119, 1–11. [Google Scholar] [CrossRef]
- Maggio, A.; Raimondi, G.; Martino, A.; De Pascale, S. Salt stress response in tomato beyond the salinity tolerance threshold. Environ. Exp. Bot. 2007, 59, 276–282. [Google Scholar] [CrossRef]
- Gupta, B.; Huang, B. Mechanism of salinity tolerance in plants: Physiological, biochemical, and molecular characterization. Int. J. Genom. 2014, 2014, 1–18. [Google Scholar] [CrossRef]
- Wu, G.Q.; Jiao, Q.; Shui, Q.Z. Effect of salinity on seed germination, seedling growth, and inorganic and organic solutes accumulation in sunflower (Helianthus annuus L.). Plant Soil Environ. 2015, 61, 220–226. [Google Scholar] [CrossRef]
- Farkhondeh, R.; Nabizadeh, E.; Jalilnezhad, N. Effect of salinity stress on proline content, membrane stability and water relations in two sugar beet cultivars. Int. J. Agric. Sci. 2012, 2, 385–392. [Google Scholar]
- Cassaniti, C.; Romano, D.; Hop, M.E.C.M.; Flowers, T.J. Growing floricultural crops with brackish water. Environ. Exp. Bot. 2013, 92, 165–175. [Google Scholar] [CrossRef]
- Parihar, P.; Singh, S.; Singh, R.; Singh, V.P.; Prasad, S.M. Effect of salinity stress on plants and its tolerance strategies: A review. Environ. Sci. Pollut. Res. 2015, 22, 4056–4075. [Google Scholar] [CrossRef] [PubMed]
- Bensidhoum, L.; Nabti, E.H. Plant Growth-Promoting Bacteria for Improving Crops under Saline Conditions. In Microorganisms in Saline Environments: Strategies and Functions; Giri, B., Varma, A., Eds.; Springer: Cham, Switzerland, 2019; pp. 329–352. [Google Scholar]
- Parida, A.K.; Das, A.B. Salt tolerance and salinity effects on plants: A review. Ecotoxicol. Environ. Saf. 2005, 60, 324–349. [Google Scholar] [CrossRef] [PubMed]
- Parvaiz, A.; Satyawati, S. Salt stress and phyto-biochemical responses of plants—A review. Plant Soil Environ. 2008, 54, 89–99. [Google Scholar] [CrossRef]
- Munns, R.; Husain, S.; Rivelli, A.R.; Richard, A.J.; Condon, A.G.; Megan, P.L.; Evans, S.L.; Schachtman, D.P.; Hare, R.A. Avenues for increasing salt tolerance of crops, and the role of physiologically based selection traits. Plant Soil 2002, 247, 93–105. [Google Scholar] [CrossRef]
- Ashraf, M.; Hasnain, S.; Berge, O.; Mahmood, T. Inoculating wheat seedling with exopolysaccharide-producing bacteria restricts sodium uptake and stimulates plant growth under salt stress. Biol. Fertil. Soils 2004, 40, 152–157. [Google Scholar] [CrossRef]
- Radić, S.; Štefanić, P.P.; Lepeduš, H.; Roje, V.; Pevalek-Kozlina, B. Salt tolerance of Centaurea ragusina L. is associated with efficient osmotic adjustment and increased antioxidative capacity. Environ. Exp. Bot. 2013, 87, 39–48. [Google Scholar] [CrossRef]
- Liu, S.; Hao, H.; Lu, X.; Zhao, X.; Wang, Y.; Zhang, Y.; Xie, Z.; Wang, R. Transcriptome profiling ofgenes involved in induced systemic salt tolerance conferred by Bacillus amyloliquefaciens FZB42 in Arabidopsis thaliana. Sci. Rep. 2017, 7, 1–3. [Google Scholar] [CrossRef]
- Donohue, K. Seeds and seasons: Interpreting germination timing in the field. Seed Sci. Res. 2005, 15, 175–187. [Google Scholar] [CrossRef]
- Živković, S.; Dević, M.; Filipović, B.; Giba, Z.; Grubišić, D. Effect of NaCl on seed germination in some Centaurium Hill. species (Gentianaceae). Arch. Biol. Sci. 2007, 59, 227–231. [Google Scholar] [CrossRef]
- Gulzar, S.; Khan, M.A.; Ungar, I.A. Salt tolerance of a coastal salt marsh grass. Commun. Soil Sci. Plant Anal. 2003, 34, 2595–2605. [Google Scholar] [CrossRef]
- Giménez Luque, E.; Delgado Fernández, I.C.; Gómez Mercado, F. Effect of salinity and temperature on seed germination in Limonium cossonianum. Botany 2013, 91, 12–16. [Google Scholar] [CrossRef]
- Bojović, B.; Đelić, G.; Topuzović, M.; Stanković, M. Effects of NaCl on seed germination in some species from families Brassicaceae and Solanaceae. Kragujev. J. Sci. 2010, 32, 83–87. [Google Scholar]
- Shokohifard, G.; Sakagami, K.; Hamada, R.; Matsumoto, S. Effect of amending materials on growth of radish plant in salinized soil. J. Plant Nutr. 1989, 12, 1195–1214. [Google Scholar] [CrossRef]
- Rahman, M.; Soomro, U.A.; Haq, M.Z.U.; Gul, S.H. Effects of NaCl salinity on wheat (Triticum aestivum L.) cultivars. World J. Agric. Res. 2008, 4, 398–403. [Google Scholar]
- Al-swedi, F.G.; Alshamari, M.; Al_Zaidi, I.H.M.; Rihan, H.Z. Impact of Salinity Stress on Seed Germination in Lettuce (Lactuca Sativa). J. Res. Lepid. 2020, 51, 374–385. [Google Scholar] [CrossRef]
- Bagwasi, G.; Agenbag, G.A.; Swanepoel, P.A. Effect of salinity on the germination of wheat and barley in South Africa. Crop. Forage Turfgrass Manag. 2020. [Google Scholar] [CrossRef]
- Bhatnagar-Mathur, P.; Vadez, V.; Sharma, K.K. Transgenic approaches for abiotic stress tolerance in plants: Retrospect and prospects. Plant Cell Rep. 2008, 27, 411–424. [Google Scholar] [CrossRef]
- Hniličková, H.; Hnilička, F.; Martinkova, J.; Kraus, K. Effects of salt stress on water status, photosynthesis and chlorophyll fluorescence of rocket. Plant Soil Environ. 2017, 63, 362–367. [Google Scholar] [CrossRef]
- Aquino, A.J.S.; Lacerda, C.F.; Bezerra, M.A.; Gomes-Filho, E.; Costa, R.N.T. Crescimento, partição de matéria seca e retenção de Na+ e Cl- em dois genótipos de sorgo irrigados com águas salinas. Rev. Bras. Ciênc. Solo 2007, 31, 961–971. [Google Scholar] [CrossRef]
- Chaves, M.M.; Flexas, J.; Pinheiro, C. Photosynthesis under drought and salt stress: Regulation mechanisms from whole plant to cell. Ann. Bot. 2009, 103, 551–560. [Google Scholar] [CrossRef] [PubMed]
- Qiu, D.L.; Lin, P.; Guo, S.Z. Effects of salinity on leaf characteristics and CO2/H2O exchange of Kandelia candel (L.) Druce seedlings. J. For. Sci. 2007, 53, 13–19. [Google Scholar] [CrossRef]
- Jamil, M.; Rehman, S.; Lee, K.J.; Kim, J.M.; Kim, H.; Rha, E.S. Salinity reduced growth PS2 photochemistry and chlorophyll content in radish. Sci. Agric. 2007, 64, 111–118. [Google Scholar] [CrossRef]
- Desingh, R.; Kanagaraj, G. Effect of salinity stress on photosynthetic enzymes of two sesame (Sesamum indicum L.) Varieties. Plant Arch. 2020, 20, 16–20. [Google Scholar]
- Jacoby, R.P.; Millar, A.H.; Taylor, N.L. Investigating the role of respiration in plant salinity tolerance by analyzing mitochondrial proteomes from wheat and a salinity-tolerant Amphiploid (wheat × Lophopyrum elongatum). J. Proteome Res. 2013, 12, 4807–4829. [Google Scholar] [CrossRef]
- Zapata, P.J.; Serrano, M.; Pretel, M.T.; Amorós, A.; Botella, M.A. Polyamines and ethylene changes during germination of different plant species under salinity. Plant Sci. 2004, 167, 781–788. [Google Scholar] [CrossRef]
- Schwarz, M.; Gale, J. Maintenance respiration and earbon balance of plants at low levels of sodium chloride salinity. J. Exp. Bot. 1981, 32, 933–941. [Google Scholar] [CrossRef]
- Yeo, A.R. Salinity resistance: Physiologies and prices. Physiol. Plant. 1983, 58, 214–222. [Google Scholar] [CrossRef]
- Epron, D.; Toussaint, M.L.; Badot, P.M. Effects of sodium chloride salinity on root growth and respiration in oak seedlings. Ann. For. Sci. 1999, 56, 41–47. [Google Scholar] [CrossRef]
- Moud, A.M.; Maghsoudi, K. Salt stress effects on respiration and growth of germinated seeds of different wheat (Triticum aestivum L.) cultivars. World J. Agric. Sci. 2008, 4, 351–358. [Google Scholar]
- Che-Othman, M.H.; Jacoby, R.P.; Millar, A.H.; Taylor, N.L. Wheat mitochondrial respiration shifts from the tricarboxylic acid cycle to the GABA shunt under salt stress. New Phytol. 2020, 225, 1166–1180. [Google Scholar] [CrossRef] [PubMed]
- Ghanem, M.E.; Albacete, A.; Martínez-Andújar, C.; Acosta, M.; Romero-Aranda, R.; Dodd, I.C.; Lutts, S.; Pérez-Alfocea, F. Hormonal changes during salinity-induced leaf senescence in tomato (Solanum lycopersicum L.). J. Exp. Bot. 2008, 59, 3039–3050. [Google Scholar] [CrossRef] [PubMed]
- Cramer, G.R.; Nowak, R.S. Supplemental manganese improves the relative growth, net assimilation and photosynthetic rates of salt-stressed barley. Physiol. Plant. 1992, 84, 600–605. [Google Scholar] [CrossRef]
- Lim, P.O.; Kim, H.J.; Nam, H.G. Leaf senescence. Annu. Rev. Plant Biol. 2007, 58, 115–136. [Google Scholar] [CrossRef]
- Shabala, S. Salinity and programmed cell death: Unraveling mechanisms for ion specific signaling. J. Exp. Bot. 2009, 60, 709–712. [Google Scholar] [CrossRef]
- Tester, M.; Davenport, R.J. Na+ transport and Na+ tolerance in higher plants. Ann. Bot. 2003, 91, 503–527. [Google Scholar] [CrossRef] [PubMed]
- Lee, R.H.; Wang, C.H.; Huang, L.T.; Chen, S.C.G. Leaf senescence in rice plants: Cloning and characterization of senescence up-regulated genes. J. Exp. Bot. 2001, 52, 1117–1121. [Google Scholar] [CrossRef]
- Li, Y.; Beisson, F.; Pollard, M.; Ohlrogge, J. Oil content of Arabidopsis seeds: The influence of seed anatomy, light and plant-to-plant variation. Phytochemistry 2006, 67, 904–915. [Google Scholar] [CrossRef]
- Yuenyong, W.; Sirikantaramas, S.; Qu, L.J.; Buaboocha, T. Isocitrate lyase plays important roles in plant salt tolerance. BMC Plant Biol. 2019, 19, 1–14. [Google Scholar] [CrossRef]
- Chrysargyris, A.; Tzionis, A.; Xylia, P.; Tzortzakis, N. Effects of salinity on tagetes growth, physiology, and shelf life of edible flowers stored in passive modified atmosphere packaging or treated with ethanol. Front. Plant Sci. 2018, 9, 1–13. [Google Scholar] [CrossRef]
- Zapryanova, N.; Atanassova, B. Effects of salt stress on growth and flowering of ornamental annual species. Biotechnol. Biotechnol. Equip. 2009, 23, 177–179. [Google Scholar] [CrossRef]
- Bybordi, A. Effects of salinity on yield and component characters in canola (Brassica napus L.) cultivars. Not. Sci. Biol. 2010, 2, 81–83. [Google Scholar] [CrossRef][Green Version]
- Mass, E.V.; Poss, J.A. Salt sensitivity of Maintenance respiration was estimated to be about wheat at various growth stages. Irrig. Sci. 1989, 10, 29–40. [Google Scholar]
- Flores, H. Changes in polyamine metabolism in response to abiotic stress. In Biochemistry and Physiology of Polyamines in Plants; Slocum, I.R., Flores, H., Eds.; CRC Press: Boca Raton, FL, USA, 1991; pp. 214–225. [Google Scholar]
- Limami, M.A.; Leach, F.; Brugiere, N.; Aziz, A.; Martin, T.J.; Malmberg, R.L.; Hirel, B.; Tepfer, D. Altered Polyamine Metabolism and Stress-Induced Proline Accumulation in Genetically Manipulated Tobacco. In Proceedings of the Dahlia Greidinger International Symposium on Nutrient Management under Salinity and Water Stress, Haifa, Palestine, 1–4 March 1999. [Google Scholar]
- Salem, N.; Msaada, K.; Dhifi, W.; Limam, F.; Marzouk, B. Effect of salinity on plant growth and biological activities of Carthamus tinctorius L. extracts at two flowering stages. Acta Physiol. Plant 2014, 36, 433–445. [Google Scholar] [CrossRef]
- Singh, N.K.; La Rosa, P.C.; Handa, A.K.; Hasegawa, P.M.; Bressan, R.A. Hormonal regulation of protein synthesis associated with salt tolerance in plant cells. Proc. Natl. Acad. Sci. USA 1987, 84, 739–743. [Google Scholar] [CrossRef]
- Blaha, G.; Stelzl, U.; Spahn, C.M.T.; Agrawal, R.K.; Frank, J.; Nierhaus, K.H. Preparation of functional ribosomal complexes and effect of buffer conditions on tRNA positions observed by cryoelectron microscopy. Methods Enzymol. 2000, 317, 292–309. [Google Scholar] [CrossRef]
- Maaroufi-Dguimi, H.; Debouba, M.; Gaufichon, L.; Clément, G.; Gouia, H.; Hajjaji, A.; Susuki, A. An Arabidopsis mutant disrupted in ASN2 encoding asparagine synthetase 2 exhibits low salt stress tolerance. Plant Physiol. Biochem. 2011, 49, 623–628. [Google Scholar] [CrossRef]
- Silveira, J.A.G.; Melo, A.R.B.; Viégas, R.A.; Oliveira, J.T.A. Salinity-induced effects on nitrogen assimilation related to growth in cowpea plants. Environ. Exp. Bot. 2001, 46, 171–179. [Google Scholar] [CrossRef]
- Qu, C.; Liu, C.; Ze, Y.; Gong, X.; Hong, M.; Wang, L.; Hong, F. Inhibition of nitrogen and photosynthetic carbon assimilation of maize seedlings by exposure to a combination of salt stress and potassium-deficient stress. Biol. Trace Elem. Res. 2011, 144, 1159–1174. [Google Scholar] [CrossRef]
- Queiroz, H.M.; Sodek, L.; Haddad, C.R.B. Effect of salt on the growth and metabolism of Glycine max. Braz. Arch. Biol. Technol. 2012, 55, 809–817. [Google Scholar] [CrossRef][Green Version]
- Arefian, M.; Vessal, S.; Malekzadeh-Shafaroudi, S.; Siddique, K.H.; Bagheri, A. Comparative proteomics and gene expression analyses revealed responsive proteins and mechanisms for salt tolerance in chickpea genotypes. BMC Plant Biol. 2019, 19, 1–26. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Kou, M.; Gao, Z.; Liu, Y.; Xuan, Y.; Tang, Z.; Cao, Q.; Li, Z.; Sun, J. Involvement of phosphatidyl serine and triacylglycerol in the response of sweet potato leaves to salt stress. Front. Plant Sci. 2019, 10, 1–27. [Google Scholar] [CrossRef] [PubMed]
- Bybordi, A. Effects of NaCl salinity levels on lipids and proteins of canola (Brassica napus L.) cultivars. Rom. Agric. Res. 2011, 28, 197–206. [Google Scholar]
- Khalid, A.K.; Shedeed, M.R. Influence of kinetin on growth and biochemical accumulation in Nigella sativa plants grow under salinity stress conditions. Thai J. Agric. Sci. 2014, 47, 195–203. [Google Scholar]
- Mansour, M.M.F.; Salama, K.H.A.; Ali, F.Z.M.; Abou Hadid, A.F. Cell and plant responses to NaCl in Zea mays L. cultivars differing in salt tolerance. Gen. Appl. Plant Physiol. 2005, 31, 29–41. [Google Scholar]
- Elkahoui, S.; Hernández, J.A.; Abdelly, C.; Ghrir, R.; Limam, F. Effects of salt on lipid peroxidation and antioxidant enzyme activities of Catharanthus roseus suspension cells. Plant Sci. 2005, 168, 607–613. [Google Scholar] [CrossRef]
- Watanabe, Y.; Takakuwa, M. Change of lipid composition of Zygosaccharomyces rouxii after transfer to high sodium chloride culture medium. J. Ferment. Technol. 1987, 65, 365–369. [Google Scholar] [CrossRef]
- Sui, N.; Wang, Y.; Liu, S.; Yang, Z.; Wang, F.; Wan, S. Transcriptomic and physiological evidence for the relationship between unsaturated fatty acid and salt stress in peanut. Front. Plant Sci. 2018, 9, 1–12. [Google Scholar] [CrossRef]
- Barros, J.A.; Siqueira, J.A.; Cavalcanti, J.H.; Araújo, W.L.; Avin-Wittenberg, T. Multifaceted Roles of Plant Autophagy in Lipid and Energy Metabolism. Trends Plant Sci. 2020. [Google Scholar] [CrossRef]
- Glass, D.J. US and International Markets for Phytoremediation, 1999–2000; Glass Associates, Inc.: Needham, MA, USA, 1999. [Google Scholar]
- Schirawski, J.; Perlin, M.H. Plant−microbe interaction 2017-the good, the bad and the diverse. Int. J. Mol. Sci. 2018, 19, 1374. [Google Scholar] [CrossRef]
- Frankenberger, W.T.; Bingham, F.T. Influence of salinity on soil enzyme activities. Soil Sci. Soc. Am. J. 1982, 46, 1173–1177. [Google Scholar] [CrossRef]
- Jesus, J.M.; Danko, A.S.; Fiúza, A.; Borges, M.T. Phytoremediation of salt-affected soils: A review of processes, applicability, and the impact of climate change. Environ. Sci. Pollut. Res. 2015, 22, 6511–6525. [Google Scholar] [CrossRef] [PubMed]
- Fazal, A.; Bano, A. Role of plant growth-promoting rhizobacteria (PGPR), biochar, and chemical fertilizer under salinity stress. Commun. Soil Sci. Plant Anal. 2016, 47, 1985–1993. [Google Scholar] [CrossRef]
- Srivastava, P.; Wu, Q.S.; Giri, B. Salinity: An Overview. In Microorganisms in Saline Environments: Strategies and Functions; Giri, B., Varma, A., Eds.; Springer: Cham, Switzerland, 2019; Volume 56, pp. 3–18. [Google Scholar]
- Diacono, M.; Montemurro, F. Effectiveness of organic wastes as fertilizers and amendments in salt-affected soils. Agriculture 2015, 5, 221–230. [Google Scholar] [CrossRef]
- Sadegh-Zadeh, F.; Parichehreh, M.; Jalili, B.; Bahmanyar, M.A. Rehabilitation of calcareous saline-sodic soil by means of biochars and acidified biochars. Land Degrad. Dev. 2018, 29, 3262–3271. [Google Scholar] [CrossRef]
- Saifullah, D.S.; Naeem, A.; Rengel, Z.; Naidu, R. Biochar application for the remediation of salt-affected soils: Challenges and opportunities. Sci. Total Environ. 2018, 625, 320–335. [Google Scholar] [CrossRef]
- Carillo, P.; Annunziata, M.G.; Pontecorvo, G.; Fuggi, A.; Woodrow, P. Salinity stress and salt tolerance. In Abiotic Stress in Plants–Mechanisms and Adaptations; Shanker, A., Venkateswarlu, B., Eds.; InTechOpen: Rijeka, Croatia, 2011; Volume 1, pp. 21–38. [Google Scholar]
- Carmen, B.; Roberto, D. Soil bacteria support and protect plants against abiotic stresses. In Abiotic Stress in Plants: Mechanisms and Adaptations; Shanker, A.K., Venkateswarlu, B., Eds.; InTech: Rijeka, Croatia, 2011; pp. 143–170. [Google Scholar]
- Usman, A.R. Influence of NaCl-induced salinity and Cd toxicity on respiration activity and Cd availability to barley plants in farmyard manure-amended soil. Appl. Environ. Soil Sci. 2015, 2015, 1–8. [Google Scholar] [CrossRef]
- Elmajdoub, B.; Marschner, P. Response of microbial activity and biomass to soil salinity when supplied with glucose and cellulose. J. Soil Sci. Plant Nutr. 2015, 15, 816–832. [Google Scholar] [CrossRef]
- Lodhi, A.; Arshad, M.; Azam, F.; Sajjad, M.H. Changes in mineral and mineralizable N of soil incubated at varying salinity, moisture and temperature regimes. Pak. J. Bot. 2009, 41, 967–980. [Google Scholar]
- Qadir, M.; Schubert, S. Degradation processes and nutrient constraints in sodic soils. Land Degrad. Dev. 2002, 13, 275–294. [Google Scholar] [CrossRef]
- Informativo Capebe-06/12/2010-Umpatrimônio Chamado Solo. Available online: http://www.capebe.org.br/informativo.php?id=355 (accessed on 13 June 2011).
- Egamberdieva, D.; Renella, G.; Wirth, S.; Islam, R. Secondary salinity effects on soil microbial biomass. Biol. Fertil. Soils 2010, 46, 445–449. [Google Scholar] [CrossRef]
- Troxler, T.G.; Ikenaga, M.; Scinto, L.; Boyer, J.N.; Condit, R.; Perez, R.; Gann, G.D.; Childers, D.L. Patterns of Soil bacteria and Canopy Community structure related to tropical peatland development. Wetlands 2012, 32, 769–782. [Google Scholar] [CrossRef]
- Pathak, H.; Rao, D. Carbon and nitrogen mineralization from added organic matter in saline and alkaline soils. Soil Biol. Biochem. 1998, 30, 695–702. [Google Scholar] [CrossRef]
- Muhammad, S.; Müller, T.; Joergensen, R. Decomposition of pea and maize straw in Pakistani soils along a gradient in salinity. Biol. Fertil. Soils 2006, 43, 93–101. [Google Scholar] [CrossRef]
- Zhang, W.W.; Chong, W.A.N.G.; Rui, X.U.E.; Wang, L.J. Effects of salinity on the soil microbial community and soil fertility. J. Integr. Agric. 2019, 18, 1360–1368. [Google Scholar] [CrossRef]
- Ibekwe, A.M.; Ors, S.; Ferreira, J.F.; Liu, X.; Suarez, D.L. Seasonal induced changes in spinach rhizosphere microbial community structure with varying salinity and drought. Sci. Total Environ. 2017, 579, 1485–1495. [Google Scholar] [CrossRef]
- Borneman, J.; Skroch, P.W.; O’Sullivan, K.M.; Palus, J.A.; Rumjanek, N.G.; Jansen, J.L.; Nienhuis, J.; Triplett, E.W. Molecular microbial diversity of an agricultural soil in Wisconsin. Appl. Environ. Microbiol. 1996, 62, 1935–1943. [Google Scholar] [CrossRef]
- Rousk, J.; Elyaagubi, F.K.; Jones, D.L.; Godbold, D.L. Bacterial salt tolerance is unrelated to soil salinity across an arid agro-ecosystem salinity gradient. Soil Biol. Biochem. 2011, 43, 1881–1887. [Google Scholar] [CrossRef]
- Tripathi, S.; Kumari, S.; Chakraborty, A.; Gupta, A.; Chakrabarti, K.; Bandyapadhyay, B.K. Microbial biomass and its activities in salt-affected coastal soils. Biol. Fertil. Soils 2006, 42, 273–277. [Google Scholar] [CrossRef]
- Nabti, E.; Schmid, M.; Hartmann, A. Application of halotolerant bacteria to restore plant growth under salt stress. In Halophile: Biodiversity and Sustainable Exploitation; Maheshwari, D.K., Saraf, M., Eds.; Springer: Cham, Switzerland, 2015; pp. 235–259. [Google Scholar]
- Amato, M.; Ladd, J.N. Application of the ninhydrin-reactive N assay for microbial biomass in acid soils. Soil Biol. Biochem. 1994, 26, 1109–1115. [Google Scholar] [CrossRef]
- Gunde-Cimerman, N.; Zalar, P.; de Hoog, S.; Plemenitas, A. Hypersaline waters in salterns-natural ecological niches for halophilic black yeasts. FEMS Microbiol. Ecol. 2000, 32, 235–240. [Google Scholar] [CrossRef]
- Omar, S.A.; Abdel-Sater, M.A.; Khallil, A.M.; Abdalla, M.H. Growth and enzyme activities of fungi and bacteria in soil salinized with sodium chloride. Folia Microbiol. 1994, 39, 23–28. [Google Scholar] [CrossRef]
- Juniper, S.; Abbott, L.K. Soil salinity delays germination and limits growth of hyphae from propagules of arbuscular mycorrhizal fungi. Mycorrhiza 2006, 16, 371–379. [Google Scholar] [CrossRef] [PubMed]
- Saxena, A.K.; Yadav, A.N.; Rajawat, M.; Kaushik, R.; Kumar, R.; Kumar, M.; Prasanna, R.; Shukla, L. Microbial diversity of extreme regions: An unseen heritage and wealth. Indian J. Plant Genet. Resour. 2016, 29, 246–248. [Google Scholar] [CrossRef]
- Yadav, A.N.; Verma, P.; Kaushik, R.; Dhaliwal, H.S.; Saxena, A.K. Archaea endowed with plant growth promoting attributes. EC Microbiol. 2017, 8, 294–298. [Google Scholar]
- Ramadoss, D.; Lakkineni, V.K.; Bose, P.; Ali, S.; Annapurna, K. Mitigation of salt stress in wheat seedlings by halotolerant bacteria isolated from saline habitats. SpringerPlus 2013, 2, 1–7. [Google Scholar] [CrossRef]
- Yao, L.; Wu, Z.; Zheng, Y.; Kaleem, I.; Li, C. Growth promotion and protection against salt stress by Pseudomonas putida Rs-198 on cotton. Eur. J. Soil Biol. 2010, 46, 49–54. [Google Scholar] [CrossRef]
- Arora, S.; Trivedi, R.; Rao, G.G. Bioremediation of coastal and inland salt affected soils using halophyte plants and halophilic soil microbes. In CSSRI Annual Report 2012–13; CSSRI: Karnal, India, 2013; pp. 94–100. [Google Scholar]
- Grover, M.; Ali, S.Z.; Sandhya, V.; Rasul, A.; Venkateswarlu, B. Role of microorganisms in adaptation of agriculture crops to abiotic stresses. World J. Microbiol. Biotechnol. 2011, 27, 1231–1240. [Google Scholar] [CrossRef]
- Majeed, A.; Muhammad, Z.; Ahmad, H. Plant growth promoting bacteria: Role in soil improvement, abiotic and biotic stress management of crops. Plant Cell Rep. 2018, 37, 1599–1609. [Google Scholar] [CrossRef]
- Islam, S. Microorganisms in the Rhizosphere and their Utilization in Agriculture: A Mini Review. PSM Microbiol. 2018, 3, 105–110. [Google Scholar]
- Shrivastava, P.; Kumar, R. Soil salinity: A serious environmental issue and plant growth promoting bacteria as one of the tools for its alleviation. Saudi J. Biol. Sci. 2015, 22, 123–131. [Google Scholar] [CrossRef] [PubMed]
- Shahzad, R.; Khan, A.L.; Bilal, S.; Waqas, M.; Kang, S.M.; Lee, I.J. Inoculation ofabscisic acid-producing endophytic bacteria enhances salinity stress tolerance in Oryza sativa. Environ. Exp. Bot. 2017, 136, 68–77. [Google Scholar] [CrossRef]
- Zhou, C.; Fi, L.; Xie, Y.; Zhu, L.; Xiao, X.; Ma, Z.; Wang, J. Involvement of abscisic acid in microbe-induced saline-alkaline resistance in plants. Plant Signal. Behav. 2017, 12, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Berendsen, R.L.; Vismans, G.; Yu, K.; Song, Y.; Jonge, R.; Burgman, W.P.; Pieterse, C.M. The disease-induced assemblage of a plant beneficial bacterial consortium. ISME J. 2018, 12, 1496–1507. [Google Scholar] [CrossRef] [PubMed]
- Nadeem, S.M.; Ahmad, M.; Zahir, Z.A.; Javaid, A.; Ashraf, M. The role of mycorrhizae and plant growth promoting rhizobacteria (PGPR) in improving crop productivity under stressful environments. Biotechnol. Adv. 2014, 32, 429–448. [Google Scholar] [CrossRef] [PubMed]
- Egamberdieva, D.; Davranov, K.; Wirth, S.; Hashem, A.; Abd Allah, E.F. Impact of soil salinity on the plant-growth-promoting and biological control abilities of root associated bacteria. Saudi J. Biol. Sci. 2017, 24, 1601–1608. [Google Scholar] [CrossRef]
- Tewari, S.; Arora, N.K. Role of salicylic acid from Pseudomonas aeruginosa PF23EPS+ ingrowth promotion of sunflower in saline soils infested with phytopathogen Macrophomina phaseolina. Environ. Sustain. 2018, 1, 49–59. [Google Scholar] [CrossRef]
- Pacwa-Płociniczak, M.; Płociniczak, T.; Iwan, J.; Żarska, M.; Chorążewski, M.; Dzida, M.; Piotrowska-Seget, Z. Isolation of hydrocarbon-degrading and biosurfactant producing bacteria and assessment their plant growth-promoting traits. J. Environ. Manag. 2016, 168, 175–184. [Google Scholar] [CrossRef]
- Mallick, I.; Bhattacharyya, C.; Mukherji, S.; Dey, D.; Sarkar, S.C.; Mukhopadhyay, U.K.; Ghosh, A. Effective rhizoinoculation and biofilm formation by arsenic immobilizing halophilic plant growth promoting bacteria (PGPB) isolated from mangrove rhizosphere: A step towards arsenic rhizoremediation. Sci. Total Environ. 2018, 610, 1239–1250. [Google Scholar] [CrossRef]
- Kearl, J.; McNary, C.; Lowman, J.S.; Mei, C.; Aanderud, Z.T.; Smith, S.T.; West, J.; Colton, E.; Hamson, M.; Nielsen, B.L. Salt-tolerant halophyte rhizosphere bacteria stimulate growth of alfalfa in salty soil. Front. Microbiol. 2019, 10, 1–11. [Google Scholar] [CrossRef]
- Thavamani, P.; Samkumar, R.A.; Satheesh, V.; Subashchandrabose, S.R.; Ramadass, K.; Naidu, R.; Venkateswarlu, K.; Megharaj, M. Microbes from mined sites: Harnessing their potential for reclamation of derelict mine sites. Environ. Pollut. 2017, 230, 495–505. [Google Scholar] [CrossRef] [PubMed]
- Jaemsaeng, R.; Jantasuriyarat, C.; Thamchaipenet, A. Molecular interaction of1-aminocyclopropane-1-carboxylate deaminase (ACCD)-producing endophytic Streptomyces sp. GMKU 336 towards salt-stress resistance of Oryza sativa L. cv. KDML105. Sci. Rep. 2018, 8, 1–15. [Google Scholar] [CrossRef]
- Hussein, K.A.; Joo, J.H. Plant growth-promoting rhizobacteria improved salinity tolerance of Lactuca sativa and Raphanus sativus. J. Microbiol. Biotechnol. 2018, 28, 938–945. [Google Scholar] [CrossRef] [PubMed]
- Shilev, S.; Sancho, E.D.; Benlloch-Gonzalez, M. Rhizospheric bacteria alleviate salt-produced stress in sunflower. J. Environ. Manag. 2012, 95, S37–S41. [Google Scholar] [CrossRef] [PubMed]
- Karlidag, H.; Esitken, A.; Yildirim, E.; Donmez, M.F.; Turan, M. Effects of plant growth promoting bacteria on yield, growth, leaf water content, membrane permeability, and ionic composition of strawberry under saline conditions. J. Plant Nutr. 2010, 34, 34–45. [Google Scholar] [CrossRef]
- Singh, R.P.; Jha, P.N. Alleviation of salinity-induced damage on wheat plant by an ACC deaminase-producing halophilic bacterium Serratia sp. SL-12 isolated from a salt lake. Symbiosis 2016, 69, 101–111. [Google Scholar] [CrossRef]
- Yasmeen, R.; Siddiqui, Z.S. Physiological responses of crop plants against Trichoderma harzianum in saline environment. Acta Bot. Croat. 2017, 76, 154–162. [Google Scholar] [CrossRef]
- Omar, M.N.A.; Osman, M.E.H.; Kasim, W.A.; Abd El-Daim, I.A. Improvement of Salt Tolerance Mechanisms of Barley Cultivated Under Salt Stress Using Azospirillum brasilense. In Salinity and Water Stress: Improving Crop Efficiency; Ashraf, M., Ozturk, M., Athar, H., Eds.; Springer: Dordrecht, The Netherlands, 2009; Volume 44, pp. 133–147. [Google Scholar]
- Kasim, W.A.; Gaafar, R.M.; Abou-Ali, R.M.; Omar, M.N.; Hewait, H.M. Effect of biofilm forming plant growth promoting rhizobacteria on salinity tolerance in barley. Ann. Agric. Sci. 2016, 61, 217–227. [Google Scholar] [CrossRef]
- Naz, I.; Bano, A.; Ul-Hassan, T. Isolation of phytohormones producing plant growth promoting rhizobacteria from weeds growing in Khewra salt range, Pakistan and their implication in providing salt tolerance to Glycine max L. Afr. J. Biotechnol. 2009, 8, 5762–5768. [Google Scholar] [CrossRef]
- Bal, H.B.; Nayak, L.; Das, S.; Adhya, T.K. Isolation of ACC deaminase producing PGPR from rice rhizosphere and evaluating their plant growth promoting activity under salt stress. Plant Soil 2013, 366, 93–105. [Google Scholar] [CrossRef]
- Sadiki, M.; Rabih, K. Selection of chickpea (Cicer arietinum) for yield and symbiotic nitrogen fixation ability under salt stress. Agronomie 2001, 21, 659–666. [Google Scholar] [CrossRef]
- Jung, J.K.H.; McCouch, S. Getting to the roots of it: Genetic and hormonal control of root architecture. Front. Plant Sci. 2013, 4, 57–60. [Google Scholar] [CrossRef] [PubMed]
- Georgiev, G.I.; Atkins, C.A. Effects of salinity on N2 fixation, nitrogen metabolism and export and diffusive conductance of cowpea root nodules. Symbiosis 1993, 15, 239–255. [Google Scholar]
- Delgado, M.J.; Garrido, J.M.; Ligero, F.; Lluch, C. Nitrogen fixation and carbon metabolism by nodules and bacteroids of pea plants under sodium chloride. Physiol. Plant. 1993, 89, 824–829. [Google Scholar] [CrossRef]
- Hanin, M.; Ebel, C.; Ngom, M.; Laplaze, L.; Masmoudi, K. New insights on plant salt tolerance mechanisms and their potential use for breeding. Front. Plant Sci. 2016, 7, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Bertrand, A.; Prévost, D.; Juge, C.; Chalifour, F.P. Impact of elevated CO2 on carbohydrate and ureide content in soybean inoculated with different strains of Bradyrhizobium japonicum. Botany 2011, 89, 481–490. [Google Scholar] [CrossRef]
- Hafeez, F.Y.; Aslam, Z.; Malik, K.A. Effect of salinity and inoculation on growth, nitrogen fixation and nutrient uptake of Vigna radiata (L.) Wilczek. Plant Soil 1988, 106, 3–8. [Google Scholar] [CrossRef]
- Woldeyohannes, W.H.; Dasilva, M.C.; Gueye, M. Nodulation and nitrogen fixation of Stylosanthes hamata in response to induced drought stress. Arid Land Res. Manag. 2007, 21, 157–163. [Google Scholar] [CrossRef]
- Bala, N.; Sharma, P.K.; Lakshminarayana, K. Nodulation and nitrogen-fixation by salinity-tolerant rhizobia in symbiosis with tree legumes. Agric. Ecosyst. Environ. 1990, 33, 33–46. [Google Scholar] [CrossRef]
- Rabie, G.H.; Aboul-Nasr, M.B.; Al-Humiany, A. Increased salinity tolerance of cowpea plants by dual inoculation of an arbuscular mycorrhizal fungus Glomus clarum and a nitrogen-fixer Azospirillum brasilense. Mycobiology 2005, 33, 51–60. [Google Scholar] [CrossRef]
- Nogales, J.; Campos, R.; BenAbdelkhalek, H.; Olivares, J.; Lluch, C.; Sanjuan, J. Rhizobium tropici genes involved in free-living salt tolerance are required for the establishment of efficient nitrogen-fixing symbiosis with Phaseolus vulgaris. Mol. Plant-Microbe Interact. 2002, 15, 225–232. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.Y.; Roessner, U.; Eickmeier, I.; Genc, Y.; Callahan, D.L.; Shirley, N.; Langridge, P.; Bacic, A. Metabolite profiling reveals distinct changes in carbon and nitrogen metabolism in phosphate-deficient barley plants (Hordeum vulgare L.). Plant Cell Physiol. 2008, 49, 691–703. [Google Scholar] [CrossRef] [PubMed]
- Pradhan, N.; Sukla, L.B. Solubilization of inorganic phosphates by fungi isolated from agriculture soil. Afr. J. Biotechnol. 2006, 5, 850–854. [Google Scholar]
- Nautiyal, C.S. An efficient microbiological growth medium for screening phosphate-solubilizing microorganism. FEMS Microbiol. Lett. 1999, 170, 265–270. [Google Scholar] [CrossRef] [PubMed]
- Zhu, D.; Ouyang, L.; Xu, Z.; Zhang, L. Rhizobacteria of Populus euphratica promoting plant growth against heavy metals. Int. J. Phytoremediat. 2015, 17, 973–980. [Google Scholar] [CrossRef]
- Gupta, N.; Sahoo, D.; Basak, U.C. Evaluation of in vitro solubilization potential of phosphate solubilising Streptomyces isolated from phyllosphere of Heritiera fomes (mangrove). Afr. J. Microbiol. Res. 2010, 4, 136–142. [Google Scholar]
- Rojas-Tapias, D.; Moreno-Galvan, A.; Pardo-Dıaz, S.; Obando, M.; Rivera, D.; Bonilla, R. Effect of inoculation with plant growth-promoting bacteria (PGPB) on amelioration of saline stress in maize (Zea mays). Appl. Soil Ecol. 2012, 61, 264–272. [Google Scholar] [CrossRef]
- Nautiyal, C.S.; Bhadauria, S.; Kumar, P.; Lal, H.; Mondal, R.; Verma, D. Stress induced phosphate solubilization in bacteria isolated from alkaline soils. FEMS Microbiol. Lett. 2000, 182, 291–296. [Google Scholar] [CrossRef]
- Wei, Y.; Zhao, Y.; Shi, M.; Cao, Z.; Lu, Q.; Yang, T.; Fan, Y.; Wei, Z. Effect of organic acids production and bacterial community on the possible mechanism of phosphorus solubilization during composting with enriched phosphate-solubilizing bacteria inoculation. Bioresour. Technol. 2018, 247, 190–199. [Google Scholar] [CrossRef]
- Zaidi, A.; Khan, M.; Ahemad, M.; Oves, M. Plant growth promotion by phosphate solubilizing bacteria. Acta Microbiol. Immunol. Hung. 2009, 56, 263–284. [Google Scholar] [CrossRef]
- Mahajan, S.G.; Nandre, V.S.; Salunkhe, R.C.; Shouche, Y.S.; Kulkarni, M.V. Chemotaxis physiological adaptation of an indigenous abiotic stress tolerant plant growth promoting Pseudomonas stutzeri: Amelioration of salt stress to Cicer arietinum. Biocatal. Agric. Biotechnol. 2020, 27, 1–9. [Google Scholar] [CrossRef]
- Li, X.; Sun, P.; Zhang, Y.; Jin, C.; Guan, C. A novel PGPR strain Kocuria rhizophila Y1 enhances salt stress tolerance in maize by regulating phytohormone levels, nutrient acquisition, redox potential, ion homeostasis, photosynthetic capacity and stress-responsive genes expression. Environ. Exp. Bot. 2020, 174, 1–18. [Google Scholar] [CrossRef]
- Shaterian, J.; Waterer, D.; De-Jong, H.; Tanino, K.K. Differential stress response to NaCl salt application in early and late maturing diploid potato (Solanum sp.) clones. Environ. Exp. Bot. 2005, 54, 202–212. [Google Scholar] [CrossRef]
- Manai, J.; Gouia, H.; Corpas, F.J. Redox and nitric oxide homeostasis are affected in tomato (Solanum lycopersicum) roots under salinity-induced oxidative stress. J. Plant Physiol. 2014, 171, 1028–1035. [Google Scholar] [CrossRef] [PubMed]
- Santner, A.; Estelle, M. Recent advances and emerging trends in plant hormone signaling. Nature 2009, 459, 1071–1078. [Google Scholar] [CrossRef]
- Kazan, K. Diverse roles of jasmonates and ethylene in abiotic stress tolerance. Trends Plant Sci. 2015, 20, 219–229. [Google Scholar] [CrossRef]
- Balloi, A.; Rolli, E.; Marasco, R. The role of microorganisms in bioremediation and phytoremediation of polluted and stressed soils. Agrochimica 2010, 54, 353–369. [Google Scholar]
- Egamberdieva, D.; Jabborova, D.; Hashem, A. Pseudomonas induces salinity tolerance in cotton (Gossypium hirsutum) and resistance to Fusarium root rot through the modulation of indole-3-acetic acid. Saudi J. Biol. Sci. 2015, 22, 773–779. [Google Scholar] [CrossRef]
- Pérez-Alfocea, F.; Albacete, A.; Ghanem, M.E.; Dodd, I.C. Hormonal regulation of source–sink relations to maintain crop productivity under salinity: A case study of root-to-shoot signaling in tomato. Funct. Plant Biol. 2010, 37, 592–603. [Google Scholar] [CrossRef]
- Sharaf, E.F.; Farrag, A.A. Induced resistance in tomato plants by IAA against Fusarium oxysporum lycopersici. Pol. J. Microbiol. 2004, 53, 111–116. [Google Scholar]
- Rupal, K.S.; Raval, V.H.; Saraf, M. Biosynthesis and purification of indole-3-acetic acid by halotolerant rhizobacteria isolated from Little Runn of Kachchh. Biocatal. Agric. Biotechnol. 2020, 23, 1–6. [Google Scholar] [CrossRef]
- Ullah, I.; Khan, A.R.; Park, G.S.; Lim, J.H.; Waqas, M.; Lee, I.J.; Shin, J.H. Analysis of phytohormones and phosphate solubilization in Photorhabdus spp. Food Sci. Biotechnol. 2013, 22, 25–31. [Google Scholar] [CrossRef]
- Gupta, G.; Parihar, S.S.; Ahirwar, N.K.; Snehi, S.K.; Singh, V. Plant growth promoting rhizobacteria (PGPR): Current and future prospects for development of sustainable agriculture. J. Microb. Biochem. Technol. 2015, 7, 96–102. [Google Scholar] [CrossRef]
- Vives-Peris, V.; Gomez-Cadenas, A.; Perez-Clemente, R.M. Salt stress alleviation in citrus plants by plant growth-promoting rhizobacteria Pseudomonas putida and Novosphingobium sp. Plant Cell Rep. 2018, 37, 1557–1569. [Google Scholar] [CrossRef]
- Ghanem, M.E.; Albacete, A.; Smigocki, A.C.; Frébort, I.; Pospíšilová, H.; Martínez-Andújar, C.; Acosta, M.; Sánchez-Bravo, J.; Lutts, S.; Dodd, I.C.; et al. Root-synthesised cytokinins improve shoot growth and fruit yield in salinised tomato (Solanum lycopersicum L.). J. Exp. Bot. 2011, 62, 125–140. [Google Scholar] [CrossRef] [PubMed]
- Wáskiewicz, A.; Gładysz, O.; Goliñski, P. Participation of phytohormones in adaptation to salt stress. In Plant Hormones under Challenging Environmental Factors; Ahammed, G.J., Yu, J.Q., Eds.; Springer: Dordrecht, The Netherlands, 2016; pp. 75–115. [Google Scholar]
- Albacete, A.; Ghanem, M.E.; Martinez-Andujar, C.; Acosta, M.; Sanchez-Bravo, J.; Martinez, V.; Lutts, S.; Dodd, I.C.; Perez-Alfocea, F. Hormonal changes in relation to biomass partitioning and shoot growth impairment in salinized tomato (Solanum lycopersicum L.) plants. J. Exp. Bot. 2008, 59, 4119–4131. [Google Scholar] [CrossRef]
- Kumari, P.; Khanna, V. ACC-deaminase and EPS production by salt tolerant rhizobacteria augment growth in chickpea under salinity stress. Int. J. Bioresour. Stress Manag. 2015, 6, 558–565. [Google Scholar] [CrossRef]
- Poli, A.; Anzelmo, G.; Nicolaus, B. Bacterial Exopolysaccharides from Extreme Marine Habitats: Production, Characterization and Biological Activities. Mar. Drugs 2010, 8, 1779–1802. [Google Scholar] [CrossRef]
- Flemming, H.C.; Wingender, J. Relevance of microbial extracellular polymeric substances (EPSs)-parts I: Structural and ecological aspects. Water Sci. Technol. 2001, 43, 1–8. [Google Scholar] [CrossRef]
- Ojuederie, O.B.; Babalola, O.O. Microbial and Plant-Assisted Bioremediation of Heavy Metal Polluted Environments: A Review. Int. J. Environ. Res. Public Health 2017, 14, 1504. [Google Scholar] [CrossRef]
- Arora, M.; Kaushik, A.; Rani, N.; Kaushik, C.P. Effect of cyanobacterial exopolysaccharides on salt stress alleviation and seed germination. J. Environ. Biol. 2010, 31, 701–704. [Google Scholar]
- Chan, R.; Lam, J.S.; Lam, K.; Costerton, J.W. Influence of culture conditions on expression of the mucoid mode of growth of Pseudomonas aeruginosa. J. Clin. Microbiol. 1984, 19, 8–16. [Google Scholar] [CrossRef] [PubMed]
- Ishii, S.; Koki, J.; Unno, H.; Hori, K. Two Morphological Types of Cell Appendages on a Strongly Adhesive Bacterium, Acinetobacter sp. Strain Tol 5. Appl. Environ. Microbiol. 2004, 70, 5026–5029. [Google Scholar] [CrossRef] [PubMed]
- Qurashi, A.W.; Sabri, A.N. Bacterial exopolysaccharide and biofilm formation stimulate chickpea growth and soil aggregation under salt stress. Braz. J. Microbiol. 2012, 43, 1183–1191. [Google Scholar] [CrossRef] [PubMed]
- Batool, R.; Hasnain, S. Growth stimulatory effects of Enterobacter and Serratia located from biofilms on plant growth and soil aggregation. Biotechnol 2005, 4, 347–353. [Google Scholar] [CrossRef]
- Banerjee, A.; Sarkar, S.; Cuadros-Orellana, S.; Bandopadhyay, R. Exopolysaccharides and Biofilms in Mitigating Salinity Stress: The Biotechnological Potential of Halophilic and Soil-Inhabiting PGPR Microorganisms. In Microorganisms in Saline Environments: Strategies and Functions; Giri, B., Varma, A., Eds.; Springer: Cham, Switzerland, 2019; pp. 303–330. [Google Scholar]
- Fathalla, M.A. Effectiveness of exopolysaccharides and biofilm forming plant growth promoting rhizobacteria on salinity tolerance of faba bean (Vicia faba L.). Afr. J. Microbiol. Res. 2018, 12, 399–404. [Google Scholar] [CrossRef][Green Version]
- Grattan, S.R.; Grieve, C.M. Salinity-mineral nutrient relations in horticultural crops. Sci. Hortic. 1999, 78, 127–157. [Google Scholar] [CrossRef]
- Ahmad, M.T.; Asghar, H.N.; Saleem, M.; Khan, M.Y.; Zahir, Z.A. Synergistic effect of rhizobia and biochar on growth and physiology of maize. Agron. J. 2015, 107, 2327–2334. [Google Scholar] [CrossRef]
- Etesami, H.; Mirseyed Hosseini, H.; Alikhani, H.A. In planta selection of plant growth promoting endophytic bacteria for rice (Oryza sativa L.). J. Soil Sci. Plant Nutr. 2014, 14, 491–503. [Google Scholar] [CrossRef]
- Mukherjee, A.; Gaurav, A.K.; Singh, S.; Chouhan, G.K.; Kumar, A.; Das, S. Role of Potassium (K) Solubilizing Microbes (KSM) in growth and induction of resistance against biotic and abiotic stress in plant: A book review. Clim. Chang. Environ. Sustain. 2019, 7, 212–214. [Google Scholar]
- Vejan, P.; Abdullah, R.; Khadiran, T.; Ismail, S. Role of plant growth promoting rhizobacteria in agricultural sustainability—A review. Molecules 2016, 21, 573. [Google Scholar] [CrossRef] [PubMed]
- Han, H.S.; Lee, K.D. Plant growth-promoting rhizobacteria: Effect on antioxidant status, photosynthesis, mineral uptake and growth of lettuce under soil salinity. Res. J. Agric. Biol. Sci. 2005, 1, 210–215. [Google Scholar]
- Glick, B.R.; Cheng, Z.; Czarny, J.; Duan, J. Promotion of plant growth by ACC deaminase producing soil bacteria. Eur. J. Plant Pathol. 2007, 119, 329–339. [Google Scholar] [CrossRef]
- Adesemoye, A.O.; Egamberdieva, D. Beneficial Effects of Plant Growth-Promoting Rhizobacteria on Improved Crop Production: Prospects for Developing Economies. In Bacteria in Agrobiology: Crop Productivity; Maheshwari, D.K., Saraf, M., Aeron, A., Eds.; Springer: Berlin/Heidelberg, Germany, 2013; pp. 45–63. [Google Scholar]
- Timmusk, S. Mechanism of action of the plant growth promoting Bacterium Paenibacillus polymyxa. In The Comprehensive Summaries of Uppsala Dissertations from the Faculty of Science and Technology 908; Acta Universitatis Upsaliensis: Uppsala, Sweden, 2003; p. 40. [Google Scholar]
- Völker, U.; Engelmann, S.; Maul, B.; Riethdorf, S.; Völker, A.; Schmid, R.; Mach, H.; Hecker, M. Analysis of the induction of general stress proteins of Bacillus subtilis. Microbiology 1994, 140, 741–752. [Google Scholar] [CrossRef] [PubMed]
- Rajendrakumar, C.S.V.; Suryanarayana, T.; Reddy, A.R. DNA helix destabilization by proline and betaine: Possible role in the salinity tolerance process. FEBS Lett. 1997, 410, 201–205. [Google Scholar] [CrossRef]
- Potts, M. Desiccation tolerance of prokaryotes. Microbiol. Rev. 1994, 58, 755–805. [Google Scholar] [CrossRef]
- Breedveld, M.W.; Dijkema, C.; Zevenhuizen, L.; Zehnder, A.J.B. Response of intracellular carbohydrates to a NaCl shock in Rhizobium leguminosarum biovar trifolii TA1 and Rhizobium meliloti SU47. J. Gen. Microbiol. 1993, 139, 3157–3163. [Google Scholar] [CrossRef][Green Version]
- Gouffi, K.; Pica, N.; Pichereau, V.; Blanco, B. Disaccharides as a new class of non-accumulated osmoprotectants for Sinorhizobium meliloti. Appl. Environ. Microbiol. 1999, 65, 1491–1500. [Google Scholar] [CrossRef]
- Flowers, T.J.; Troke, P.F.; Yeo, A.R. The mechanism of salt tolerance in halophytes. Annu. Rev. Plant Physiol. 1977, 28, 89–121. [Google Scholar] [CrossRef]
- Das, P.; Behera, B.K.; Meena, D.K.; Azmi, S.A.; Chatterjee, S.; Meena, K.; Sharma, A.P. Salt stress tolerant genes in halophilic and halotolerant bacteria: Paradigm for salt stress adaptation and osmoprotection. Int. J. Curr. Microbiol. Appl. Sci. 2015, 4, 642–658. [Google Scholar]
- Singh, R.P.; Jha, P.N. The multifarious PGPR Serratia marcescens CDP-13 augments induced systemic resistance and enhanced salinity tolerance of wheat (Triticum aestivum L.). PLoS ONE 2016, 11, e0155026. [Google Scholar] [CrossRef] [PubMed]
- Bartels, D.; Sunkar, R. Drought and salt tolerance in plants. Crit. Rev. Plant Sci. 2005, 24, 23–58. [Google Scholar] [CrossRef]
- Zafar-ul-Hye, M.; Farooq, H.M.; Hussain, M. Bacteria in combination with fertilizers promote root and shoot growth of maize in saline-sodic soil. Braz. J. Microbiol. 2015, 46, 97–102. [Google Scholar] [CrossRef] [PubMed]
- Penrose, D.; Glick, B. Methods for isolating and characterizing ACC deaminase-containing plant growth-promoting rhizobacteria. Physiol. Plant. 2003, 118, 10–15. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, M.; Zahir, Z.A.; Asghar, H.N.; Asghar, M. Inducing salt tolerance in mung bean through coinoculation with rhizobia and plant growth-promoting rhizobacteria containing 1-aminocyclopropane-1-carboxylate deaminase. Can. J. Microbiol. 2011, 57, 578–589. [Google Scholar] [CrossRef] [PubMed]
- Sepúlveda-Caamaño, M.; Gerding, M.; Vargas, M.; Moya-Elizondo, E.; Oyarzúa, P.; Campos, J. Lentil (Lens culinaris L.) growth promoting rhizobacteria and their effect on nodulation in coinoculation with rhizobia. Arch. Agron. Soil Sci. 2018, 64, 244–256. [Google Scholar] [CrossRef]
- Del Carmen Orozco-Mosqueda, M.; Glick, B.R.; Santoyo, G. ACC deaminase in plant growth-promoting bacteria (PGPB): An efficient mechanism to counter salt stress in crops. Microbiol. Res. 2020, 235, 1–10. [Google Scholar] [CrossRef]
- Dobbelaere, S.; Vanderleyden, J.; Okon, Y. Plant growth promoting effects of diazotrophs in the rhizosphere. Crit. Rev. Plant Sci. 2003, 22, 107–149. [Google Scholar] [CrossRef]
- Li, J.; Ovakim, D.H.; Charles, T.C.; Glick, B.R. An ACC deaminase minus mutant of Enterobacter cloacae UW4No longer promotes root elongation. Curr. Microbiol. 2000, 41, 101–105. [Google Scholar] [CrossRef]
- Cheng, Z.; Park, E.; Glick, B.R. 1-Aminocyclopropane-1-carboxylate deaminase from Pseudomonas putida UW4 facilitates the growth of canola in the presence of salt. Can. J. Microbiol. 2007, 53, 912–918. [Google Scholar] [CrossRef]
- Tiwari, G.; Duraivadivel, P.; Sharma, S.; Hariprasad, P. 1- Aminocyclopropane-1-carboxylic acid deaminase producing beneficial rhizobacteria ameliorate the biomass characters of Panicum maximum Jacq. by mitigating drought and salt stress. Sci. Rep. 2018, 8, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Kausar, R.; Shahzad, S. Effect of ACC-deaminase containing rhizobacteria on growth promotion of maize under salinity stress. J. Agric. Soc. Sci. 2006, 2, 216–218. [Google Scholar]
- Yang, J.; Kloepper, J.W.; Ryu, C.M. Rhizosphere bacteria help plants tolerate abiotic stress. Trends Plant Sci. 2010, 14, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Madhaiyan, M.; Poonguzhali, S.; Sa, T.M. Characterization of 1-Aminocyclopropane-1-carboxylate deaminase ACC deaminase containing Methylobacterium oryzae and interactions with auxins and ACC regulation of ethylene in canola (Brassica campestris). Planta 2007, 226, 867–876. [Google Scholar] [CrossRef]
- Shaharoona, B.; Arshad, M.; Zahir, Z.A. Effect of plant growth promoting rhizobacteria containing ACC-deaminase on maize (Zea mays L.) growth under axenic conditions and on nodulation in mung bean (Vigna radiata L.). Lett. Appl. Microbiol. 2006, 42, 155–159. [Google Scholar] [CrossRef]
- Shrivasta, U.P.; Kumar, A. Characterization and optimization of 1-aminocyclopropane-1-carboxylate deaminase (ACCD activity in different rhizospheric PGPR along with Microbacterium sp. strain ECI-12A. Int. J. Appl. Sci. Biotechnol. 2014, 1, 11–15. [Google Scholar] [CrossRef]
- Van Loon, L.C.; Bakker, P.A.; Pieterse, C.M. Systemic resistance induced by rhizosphere bacteria. Annu. Rev. Phytopathol. 1998, 361, 453–483. [Google Scholar] [CrossRef]
- Barriuso, J.; Solano, B.R.; Gutiérrez Mañero, F.J. Protection against pathogen and salt stress by four plant growth-promoting rhizobacteria isolated from Pinus sp. on Arabidopsis thaliana. Phytopathology 2008, 98, 666–672. [Google Scholar] [CrossRef]
- Olanrewaju, O.S.; Glick, B.R.; Babalola, O.O. Mechanisms of action of plant growth promoting bacteria. World J. Microbiol. Biotechnol. 2017, 33, 1–16. [Google Scholar] [CrossRef]
- Singh, R.P.; Jha, P.N. The PGPR Stenotrophomonas maltophilia SBP-9 augments resistance against biotic and abiotic stress in wheat plants. Front. Microbiol. 2017, 8, 1–15. [Google Scholar] [CrossRef]
- Morgan, P.W.; Drew, M.C. Ethylene and plant responses to stress. Physiol. Plant. 1997, 100, 620–630. [Google Scholar] [CrossRef]
- Abeles, F.B.; Morgan, P.W.; Saltveit, M.E., Jr. Ethylene in Plant Biology, 2nd ed.; Academic Press: San Diego, CA, USA, 1992; pp. 56–176. [Google Scholar]
- Selvakumar, G.; Panneerselvam, P.; Ganeshamurthy, A.N. Bacterial mediated alleviation of abiotic stress in crops. In Bacteria in Agrobiology: Stress Management; Maheshwari, D.K., Ed.; Springer: Berlin/Heidelberg, Germany, 2012; pp. 205–224. [Google Scholar]
- Sharma, A.; Vaishnav, A.; Jamali, H.; Srivastava, A.K.; Saxena, A.K.; Srivastava, A.K. Halophilic bacteria: Potential bioinoculants for sustainable agriculture and environment management under salt stress. In Plant-Microbe Interaction: An Approach to Sustainable Agriculture; Choudhary, D., Varma, A., Tuteja, N., Eds.; Springer: Singapore, 2016; pp. 297–325. [Google Scholar]
- Hussain, S.; Huang, J.; Zhu, C.; Zhu, L.; Cao, X.; Hussain, S.; Ashraf, M.; Khaskheli, M.A.; Kong, Y.; Jin, Q.; et al. Pyridoxal 5′-phosphate enhances the growth and morpho-physiological characteristics of rice cultivars by mitigating the ethylene accumulation under salinity stress. Plant Physiol. Biochem. 2020, 154, 782–795. [Google Scholar] [CrossRef] [PubMed]
- Misra, S.; Chauhan, P.S. ACC deaminase-producing rhizosphere competent Bacillus spp. mitigate salt stress and promote Zea mays growth by modulating ethylene metabolism. 3 Biotech 2020, 10, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Gamalero, E.; Glick, B.R. Bacterial modulation of plant ethylene levels. Plant Physiol. 2015, 169, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Timmusk, S.; Behers, L.; Muthoni, J.; Muraya, A.; Aronsson, A.C. Perspectives and challenges of microbial application for crop improvement. Front. Plant Sci. 2017, 8, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Yadav, A.N. Beneficial role of extremophilic microbes for plant health and soil fertility. J. Agric. Sci. Bot. 2017, 1, 30–33. [Google Scholar] [CrossRef]
- Akhtar, S.S.; Andersen, M.N.; Liu, F. Biochar mitigates salinity stress in potato. J. Agron. Crop Sci. 2015, 201, 368–378. [Google Scholar] [CrossRef]
- Amacher, J.K.; Koenig, K.; Kitchen, B. Salinity and Plant Tolerance. Available online: https://extension.usu.edu/files/publications/publication/AG-SO-03.pdf (accessed on 30 April 2020).
- Arora, N.K.; Tewari, S.; Singh, S.; Lal, N.; Maheshwari, D.K. PGPR for protection of plant health under saline conditions. In Bacteria in Agrobiology: Stress Management; Maheshwari, D., Ed.; Springer: Berlin/Heidelberg, Germany, 2012; pp. 239–258. [Google Scholar]
| Crops | PGPB | NaCl (mM) | Beneficial Effects | Reference |
|---|---|---|---|---|
| Lettuce | Azotobacter chroococcum | 50 and 100 | Enhanced the radicle lengths and plumule of germinated seeds | [160] |
| Sunflowers | Pseudomonas fluorescens CECT 378T | 100 | Fresh weight significantly improved by more than 10%; K+/Na+ proportion | [161] |
| Strawberry | Bacilus sp. | 35 | Ameliorated fruit productivity, leaf water amount, ionic constitution, and membrane permeability | [162] |
| Wheat | Serratia sp. SL-12 | 150–200 | Improvement in plant increase, as determined by factors like root/shoot length, dry/fresh weight, and augmentation of photosynthetic pigment | [163] |
| Maize | Trichoderma harzianum Th-6 | 50–150 | Improves water composition and stomatal conductance, elevated pigment amounts, and increases photosynthetic performance | [164] |
| Barley | Azospirillum brasilense NO40 | 250 and 350 | Mitigate photosynthetic pigments in the roots and shoots, photosynthetic effectiveness, transpiration level, stomatal conductance, agglomeration of Mg, P, K, Fe, and Ca | [165] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mokrani, S.; Nabti, E.-h.; Cruz, C. Current Advances in Plant Growth Promoting Bacteria Alleviating Salt Stress for Sustainable Agriculture. Appl. Sci. 2020, 10, 7025. https://doi.org/10.3390/app10207025
Mokrani S, Nabti E-h, Cruz C. Current Advances in Plant Growth Promoting Bacteria Alleviating Salt Stress for Sustainable Agriculture. Applied Sciences. 2020; 10(20):7025. https://doi.org/10.3390/app10207025
Chicago/Turabian StyleMokrani, Slimane, El-hafid Nabti, and Cristina Cruz. 2020. "Current Advances in Plant Growth Promoting Bacteria Alleviating Salt Stress for Sustainable Agriculture" Applied Sciences 10, no. 20: 7025. https://doi.org/10.3390/app10207025
APA StyleMokrani, S., Nabti, E.-h., & Cruz, C. (2020). Current Advances in Plant Growth Promoting Bacteria Alleviating Salt Stress for Sustainable Agriculture. Applied Sciences, 10(20), 7025. https://doi.org/10.3390/app10207025





