Glass-Ceramic Foams from Alkali-Activated Vitrified Bottom Ash and Waste Glasses
Abstract
1. Introduction
2. Materials and Methods
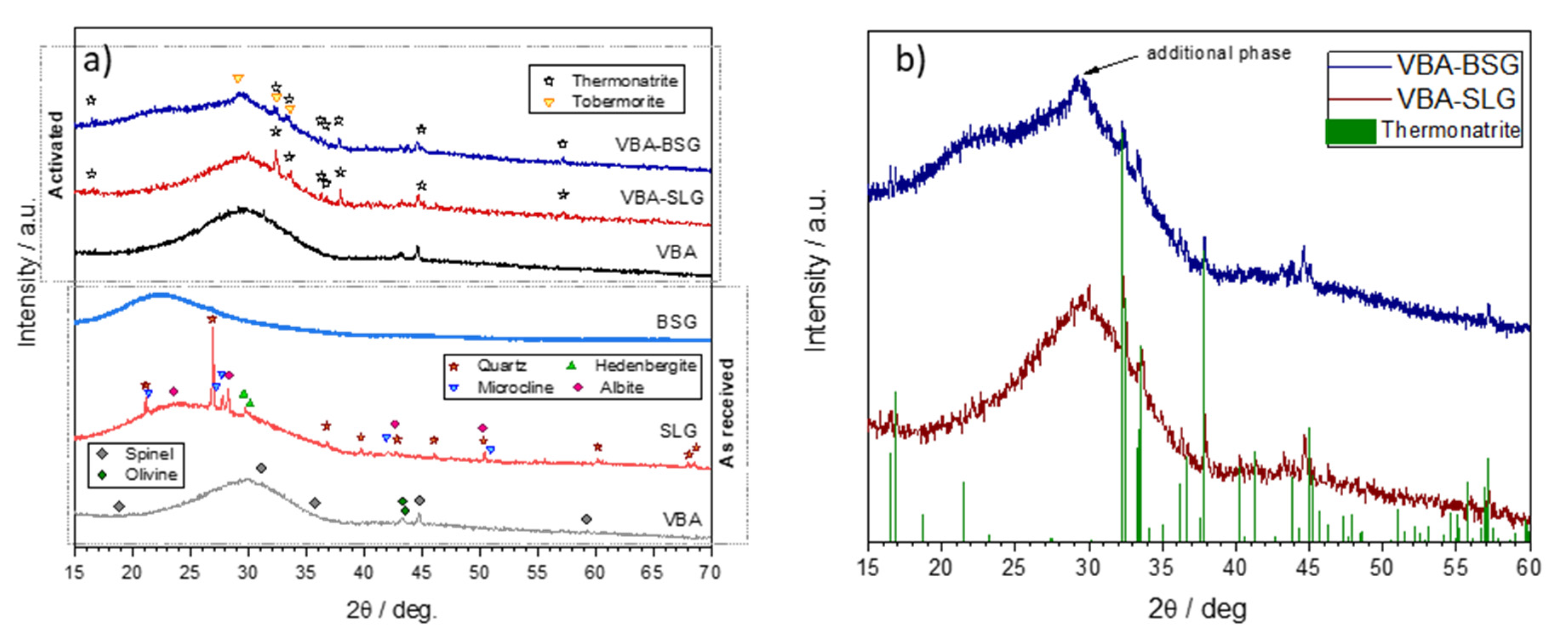
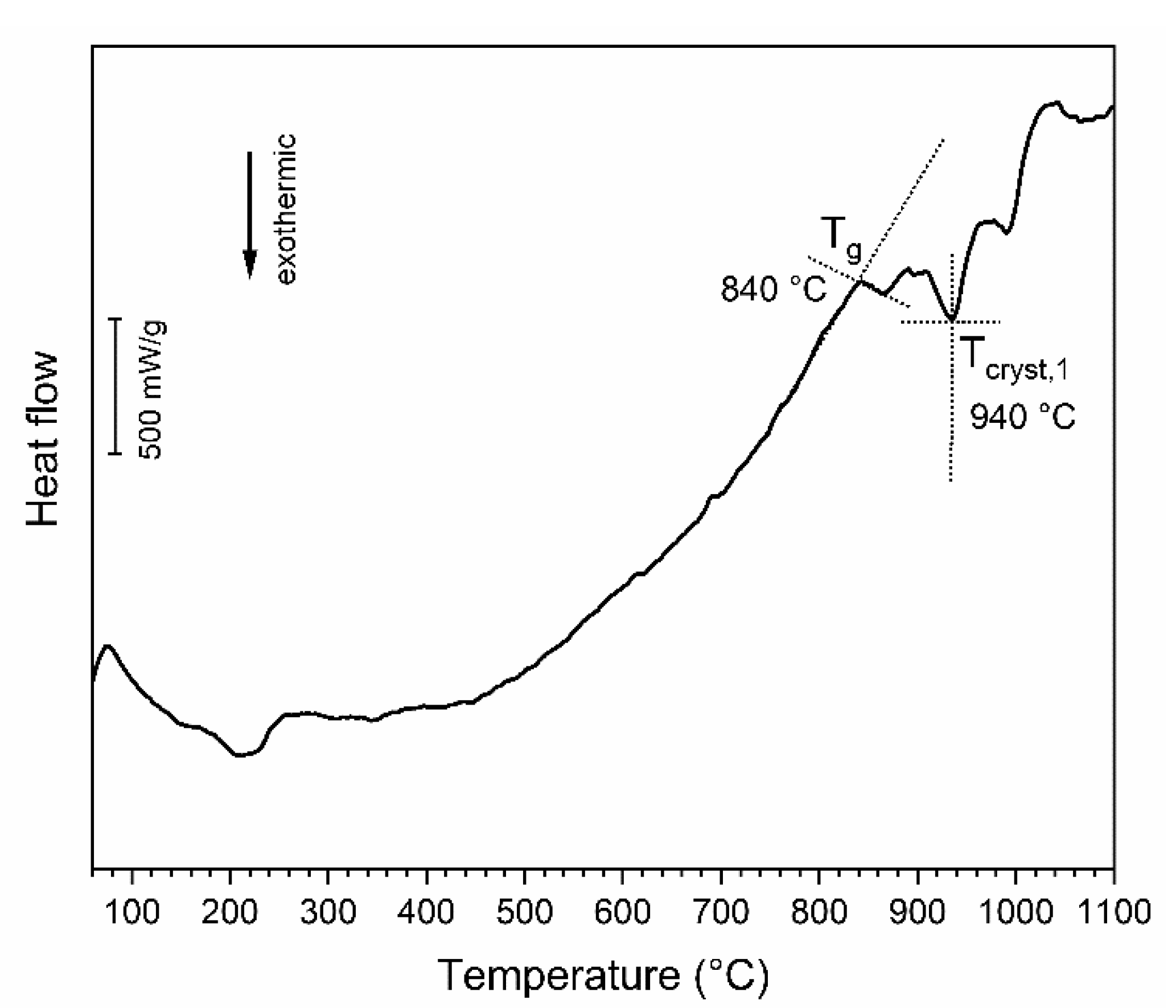
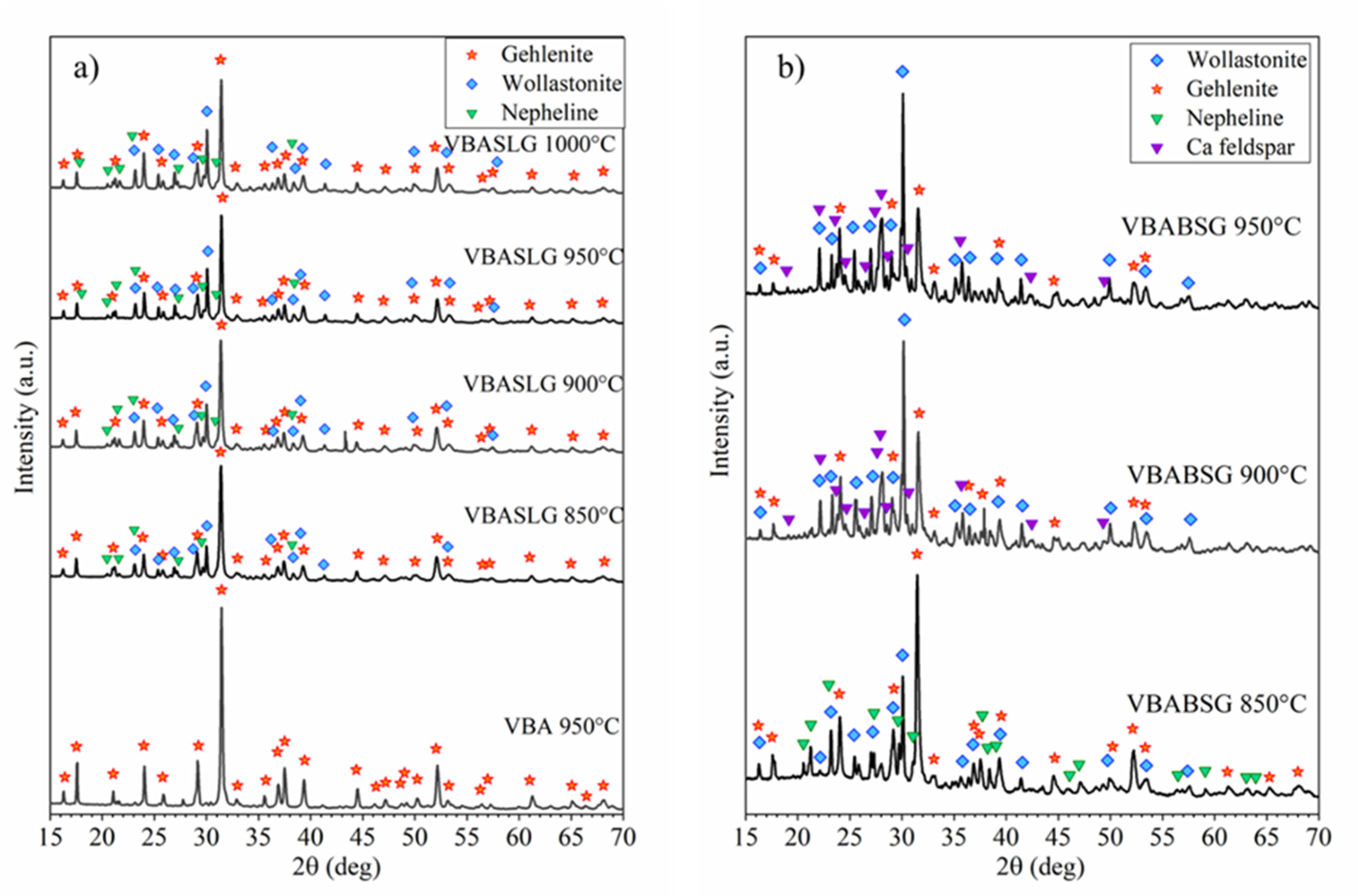
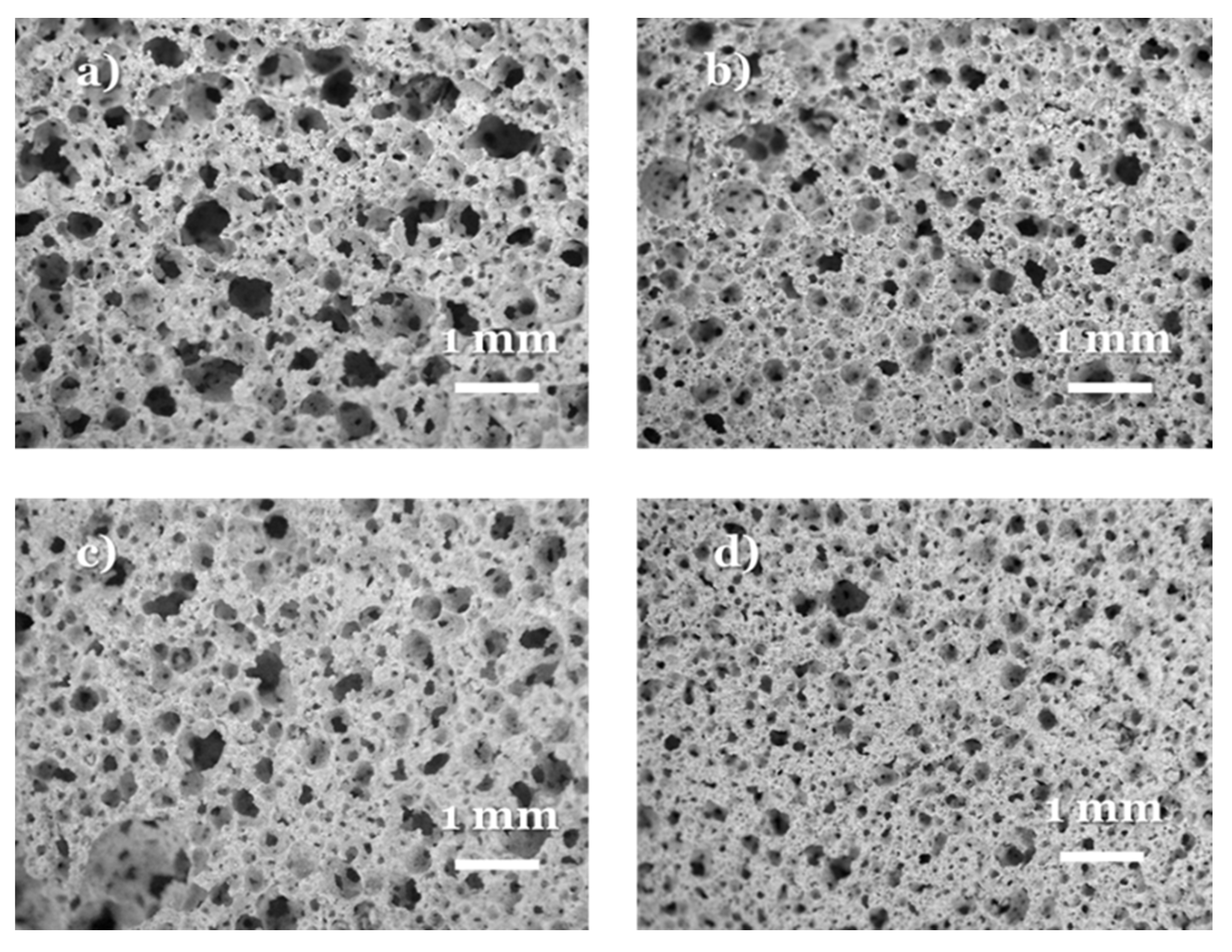
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Silva, R.V.; De Brito, J.; Lye, C.Q.; Dhir, R.K. The role of glass waste in the production of ceramic-based products and other applications: A Review. J. Clean. Prod. 2017, 167, 346–364. [Google Scholar] [CrossRef]
- Hajimohammadi, A.; Ngo, T.; Kashani, A. Glass waste versus sand as aggregates: The characteristics of the evolving geopolymer binders. J. Clean. Prod. 2018, 193, 593–603. [Google Scholar] [CrossRef]
- Lebullenger, R.; Mear, F.O. Glass Recycling. In Springer Handbook of Glass; Musgraves, J.D., Hu, J., Calvez, L., Eds.; Springer: New York, NY, USA, 2019; pp. 1355–1377. [Google Scholar]
- Fernandes, H.R.; Tulyaganov, D.U.; Ferreira, J.M.F. Preparation and characterization of foams from sheet glass and fly ash using carbonates as foaming agents. Ceram. Int. 2009, 35, 229–235. [Google Scholar] [CrossRef]
- Kim, K.; Kim, K.; Hwang, J. Characterization of ceramic tiles containing LCD waste glass. Ceram. Int. 2016, 42, 7626–7631. [Google Scholar] [CrossRef]
- Monich, P.R.; Romero, A.R.; Höllen, D.; Bernardo, E. Porous glass-ceramics from alkali activation and sinter-crystallization of mixtures of waste glass and residues from plasma processing of municipal solid waste. Constr. Build. Mater. 2018, 188, 871–878. [Google Scholar] [CrossRef]
- Hoornweg, D.; Bhada-Tata, P. What a Waste?: A Global Review of Solid Waste Management; The World Bank Group: Washington, DC, USA, 2012. [Google Scholar]
- Morf, L.S.; Brunner, P.H.; Spaun, S. Effect of operating conditions and input variations on the partitioning of metals in a municipal solid waste incinerator. Waste. Manag. Res. 2000, 18, 4–15. [Google Scholar] [CrossRef]
- Huber, F.; Blasenbauer, D.; Aschenbrenner, P.; Fellner, J. Complete determination of the material composition of municipal solid waste incineration bottom ash. Waste. Manag. 2020, 102, 677–685. [Google Scholar] [CrossRef]
- Monich, P.R.; Dogrul, F.; Lucas, H.; Friedrich, B. Strong porous glass-ceramics from alkali-activation and sinter-crystallization of virified MSWI bottom ash. Detritus 2019, 8, 101–108. [Google Scholar]
- Blasenbauer, D.; Huber, F.; Lederer, J.; Quina, M.J.; Blanc-Biscarat, D.; Bogush, A.; Bontempi, E.; Blondeau, J.; Chimenos, J.M.; Dahlbo, H.; et al. Legal situation and current practice of waste incineration bottom ash utilisation in Europe. Waste Manag. 2020, 102, 868–883. [Google Scholar] [CrossRef] [PubMed]
- Andreola, F.; Barbieri, L.; Soares, B.Q.; Karamanov, A.; Schabbach, L.M.; Bernardin, A.M.; Pich, C.T. Toxicological analysis of ceramic building materials—Tiles and glasses—Obtained from post-treated bottom ashes. Waste Manag. 2019, 98, 50–57. [Google Scholar] [CrossRef] [PubMed]
- Baino, F.; Ferraris, M. Production and characterization of ceramic foams derived from vitrified bottom ashes. Mater. Lett. 2019, 236, 281–284. [Google Scholar] [CrossRef]
- Rincon Romero, A.; Salvo, M.; Bernardo, E. Up-cycling of vitrified bottom ash from MSWI into glass-ceramic foams by means of ‘inorganic gel casting ’ and sinter-crystallization. Constr. Build. Mater. 2018, 192, 133–140. [Google Scholar] [CrossRef]
- Machiels, L.; Arnout, L.; Yan, P.; Jones, P.T.; Blanpain, B.; Pontikes, Y. Transforming enhanced landfill mining derived gasification/vitrification glass into low-carbon inorganic polymer binders and building products. J. Sustain. Met. 2017, 3, 405–415. [Google Scholar] [CrossRef]
- Rincón, A.; Giacomello, G.; Pasetto, M.; Bernardo, E. Novel ‘inorganic gel casting’ process for the manufacturing of glass foams. J. Eur. Ceram. Soc. 2017, 37, 2227–2234. [Google Scholar] [CrossRef]
- Silva, R.V.; De Brito, J.; Lynn, C.J.; Dhir, R.K. Environmental impacts of the use of bottom ashes from municipal solid waste incineration: A. Review. Resour. Conserv. Recycl. 2019, 140, 23–35. [Google Scholar] [CrossRef]
- Hruška, B.; Gavenda, T.; Vašíček, F.; Svoboda, R. Raman spectroscopy study of glass corrosion. Vib. Spectrosc. 2020, 109, 103096. [Google Scholar] [CrossRef]
- Yang, S.F.; Chiu, W.T.; Wang, T.M.; Chen, C.T.; Tzeng, C.C. Porous materials produced from incineration ash using thermal plasma technology. Waste. Manag. 2014, 34, 1079–1084. [Google Scholar] [CrossRef]
- Takahashi, S.; Neuville, D.R.; Takebe, H. Thermal properties density and structure of percalcic and peraluminus. J. Non-Cryst. Solids. 2015, 411, 5–12. [Google Scholar] [CrossRef]
- Palou, M.; Majling, J.; Dová, M.; Kozanková, J.; Mojumdar, S.C. Formation and stability of crystallohydrates in the non-equilibrium system during hydration of SAB cements. Ceram. Silik. 2005, 49, 230–236. [Google Scholar]
- Lázár, M.; Čarnogurská, M.; Brestovič, T.; Jasminská, N.; Kmeťová, Ľ.; Kapustová, Ľ.; Jezný, T. High-temperature processing of asbestos-cement roofing material in a plasma reactor. Pol. J. Environ. Stud. 2016, 25, 2027–2033. [Google Scholar] [CrossRef][Green Version]
- Bernardo, E.; Scarinci, G.; Edme, E.; Michon, U.; Planty, N. Fast-sintered gehlenite glass-ceramics from plasma-vitrified municipal solid waste incinerator fly ashes. J. Am. Ceram. Soc. 2009, 92, 528–530. [Google Scholar] [CrossRef]
- Hlavac, J. Fundamentals of Silicate Technology; SNTL: Prague, Czech Republic, 1981; pp. 41–46. [Google Scholar]
- Sugimoto, A.; Nobe, Y.; Yamazaki, T.; Anzai, Y.; Yamagishi, K.; Segawa, Y.; Takei, H. Spectroscopic properties of Cr-doped melilite crystals. Phys. Chem. Miner. 1997, 24, 326–332. [Google Scholar] [CrossRef]
- Zhang, W.; Liu, H. A low cost route for fabrication of wollastonite glass–ceramics directly using soda-lime waste glass by reactive crystallization–sintering. Ceram. Int. 2013, 39, 1943–1949. [Google Scholar] [CrossRef]
- Gibson, L.J.; Ashby, M.F. Cellular Solids, Structure and Properties; Cambridge University Press: Cambridge, UK, 1999. [Google Scholar]
- Merlini, M.; Gemmi, M.; Artioli, G. Thermal expansion and phase transitions in åkermanite and gehlenite. Phys. Chem. Min. 2005, 32, 189–196. [Google Scholar] [CrossRef]
- Bernardo, E.; Bonomo, E.; Dattoli, A. Optimisation of sintered glass-ceramics from an industrial waste glass. Ceram. Int. 2010, 36, 1675–1680. [Google Scholar] [CrossRef]
- Todorovic, J.; Svensson, M.; Herrmann, I.; Ecke, H. Council of the European Union and 2003/33/EC, “Council Decision establishing criteria and procedures for the acceptance of waste at landfills pursuant to Article 16 of and Annex II to Directive 1999/31/EC”. Off. J. Eur. Communities 2003, 8, 27–49. [Google Scholar]



| Vitrified Bottom Ash (VBA) | Soda-Lime Glass (SLG) | Borosilicate Glass (BSG) | |
|---|---|---|---|
| SiO2 | 32.1 | 71.9 | 72 |
| B2O3 | - | - | 12 |
| CaO | 33.5 | 7.5 | 1 |
| Na2O | - | 14.3 | 6 |
| MgO | 4.3 | 4.0 | - |
| Al2O3 | 27.5 | 1.2 | 7 |
| Fe2O3 | 0.3 | - | - |
| K2O | 0.2 | - | 2 |
| TiO2 | 0.6 | - | - |
| Residues | 1.6 | 1.1 | - |
| Glass Additive/T | Porosity Total % | Open Porosity % | Closed Porosity % | Density g cm−3 | Compressive Strength MPa | Bending Strength of the Solid Phase MPa |
|---|---|---|---|---|---|---|
| SLG 900 | 76.9 | 75.2 | 1.7 | 0.68 (±0.03) | 1.7 (±0.4) | 77.1 |
| SLG 950 | 73.5 | 74.9 | 0.1 | 0.70 (±0.01) | 2.0 (±0.2) | 71.4 |
| SLG 1000 | 80.0 | 79.1 | 0.9 | 0.53 (±0.03) | 1.5 (±0.1) | 84.5 |
| BSG 850 | 71.9 | 67.5 | 4.4 | 0.73 (±0.03) | 3.4 (±0.4) | 114.0 |
| BSG 900 | 74.2 | 72.1 | 2.1 | 0.70 (±0.02) | 3.7 (±0.5) | 139.6 |
| BSG 950 | 73.5 | 72.1 | 1.4 | 0.72 (±0.02) | 3.6 (±0.3) | 132.0 |
| c [ppm] | Limit 1 * | Limit 2 ** | VBA AR *** | VBASLG7030 900 °C | VBASLG7030 950 °C | VBASLG7030 1000 °C | VBABSG7030 850 °C | VBABSG7030 900 °C | VBABSG7030 950 °C |
|---|---|---|---|---|---|---|---|---|---|
| As | 0.5 | 2 | <0.0049 | 0.0267 | 0.0132 | <0.0049 | <0.0049 | 0.0171 | 0.0102 |
| Ba | 20 | 100 | 0.0729 | 0.0082 | 0.0128 | 0.0438 | 0.0402 | 0.0105 | 0.0721 |
| Cd | 0.04 | 1 | 0.0003 | <0.0002 | <0.0002 | <0.0002 | <0.0002 | <0.0002 | <0.0002 |
| Cr | 0.5 | 10 | 0.0226 | 0.9596 | 0.1104 | 0.3079 | 0.2602 | 0.0409 | 0.0628 |
| Cu | 2 | 50 | 0.0902 | 0.0022 | 0.0100 | 0.0517 | 0.0620 | 0.0205 | 0.0642 |
| Mo | 0.5 | 10 | <0.0033 | <0.0033 | <0.0033 | <0.0033 | <0.0033 | <0.0033 | <0.0033 |
| Ni | 0.4 | 10 | <0.0014 | <0.0014 | <0.0014 | <0.0014 | <0.0014 | <0.0014 | <0.0014 |
| Pb | 0.5 | 10 | 0.0161 | <0.0047 | <0.0047 | 0.0182 | 0.0079 | <0.0047 | <0.0047 |
| Sb | 0.06 | 0.7 | <0.0099 | <0.0099 | <0.0099 | <0.0099 | <0.0099 | <0.0099 | <0.0099 |
| Se | 0.1 | 0.5 | 0.0525 | 0.0154 | 0.0124 | <0.0122 | <0.0122 | <0.0122 | <0.0122 |
| Zn | 4 | 50 | <0.0203 | <0.0203 | <0.0203 | <0.0203 | <0.0203 | <0.0203 | <0.0203 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hujova, M.; Rabelo Monich, P.; Sedlacek, J.; Hnatko, M.; Kraxner, J.; Galusek, D.; Bernardo, E. Glass-Ceramic Foams from Alkali-Activated Vitrified Bottom Ash and Waste Glasses. Appl. Sci. 2020, 10, 5714. https://doi.org/10.3390/app10165714
Hujova M, Rabelo Monich P, Sedlacek J, Hnatko M, Kraxner J, Galusek D, Bernardo E. Glass-Ceramic Foams from Alkali-Activated Vitrified Bottom Ash and Waste Glasses. Applied Sciences. 2020; 10(16):5714. https://doi.org/10.3390/app10165714
Chicago/Turabian StyleHujova, Miroslava, Patricia Rabelo Monich, Jaroslav Sedlacek, Miroslav Hnatko, Jozef Kraxner, Dusan Galusek, and Enrico Bernardo. 2020. "Glass-Ceramic Foams from Alkali-Activated Vitrified Bottom Ash and Waste Glasses" Applied Sciences 10, no. 16: 5714. https://doi.org/10.3390/app10165714
APA StyleHujova, M., Rabelo Monich, P., Sedlacek, J., Hnatko, M., Kraxner, J., Galusek, D., & Bernardo, E. (2020). Glass-Ceramic Foams from Alkali-Activated Vitrified Bottom Ash and Waste Glasses. Applied Sciences, 10(16), 5714. https://doi.org/10.3390/app10165714







