Effect of Daily Intake of a Saffron Infusion on Blood Cholesterol Levels
Abstract
1. Introduction
2. Materials and Methods
2.1. Saffron Samples and Reagents
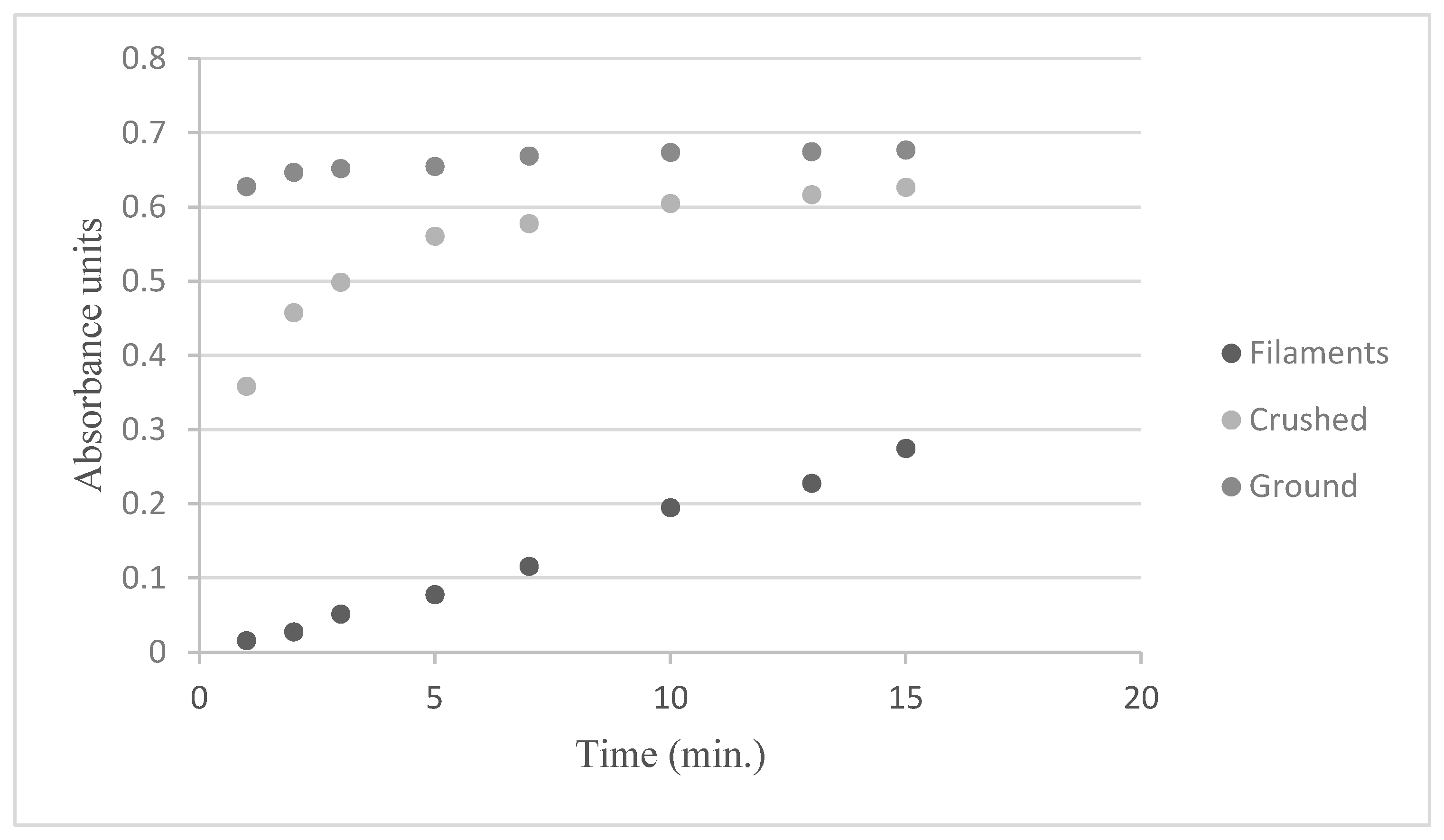
2.2. Preparation of the Saffron and Infusions
2.3. Saffron Dispensing to Volunteers
2.4. Subjects
2.5. Statistical Analysis
3. Results and Discussion
3.1. Preparation of the Saffron and Infusions
3.2. Saffron Dispensing to Patients
3.3. Effect on the Lipid Profile
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| BMI | Body Mass Index |
| CNS | Central Nervous System |
| HDL-C | High Density Lipoproteins Cholesterol |
| LDL-C | Low Density Lipoprotein Cholesterol |
| MS | Multiple Sclerosis |
| PDO | Protected Designation of Origin |
| PTFE | Polytetrafluoroethylene |
| TC | Total Cholesterol |
| TG | Triglycerides |
| WHO | World Health Organization |
References
- Fernández, O.; Fernández, V.E.; Guerrero, M. Esclerosis Múltiple; McGraw-Hill/Interamericana: Madrid, Spain, 2011. [Google Scholar]
- Tettey, P.; Simpson, S., Jr.; Taylor, B.; Blizzard, L.; Ponsonby, A.L.; Dwyer, T.; Kostner, K.; Van Der Mei, I. Adverse lipid profile is not associated with relapse risk in MS: Results from an observational cohort study. J. Neurol. Sci. 2014, 340, 230–232. [Google Scholar] [CrossRef]
- Zhornitsky, S.; McKay, K.A.; Metz, L.M.; Teunissen, C.E.; Rangachari, M. Cholesterol and markers of cholesterol turnover in multiple sclerosis: Relationship with disease outcomes. Mult. Scler. Relat. Disord. 2016, 5, 53–65. [Google Scholar] [CrossRef]
- Mandoj, C.; Renna, R.; Plantone, D.; Sperduti, I.; Cigliana, G.; Conti, L.; Koudriavtseva, T. Anti-annexin antibodies, cholesterol levels and disability in multiple sclerosis. Neurosci. Lett. 2015, 606, 156–160. [Google Scholar] [CrossRef]
- Ulivieri, C.; Baldari, C.T. Statins: From cholesterol-lowering drugs to novel immunomodulators for the treatment of Th17-mediated autoimmune diseases. Pharmacol. Res. 2014, 88, 41–52. [Google Scholar] [CrossRef] [PubMed]
- Ulbricht, C.; Conquer, J.; Costa, D.; Hollands, W.; Iannuzzi, C.; Isaac, R.; Jordan, J.K.; Ledesma, N.; Ostroff, C.; Serrano, J.M.G.; et al. An evidence-based systematic review of saffron (Crocus sativus) by the Natural Standard Research Collaboration. J. Diet. Suppl. 2011, 8, 58–114. [Google Scholar] [CrossRef] [PubMed]
- Akhondzadeh, S.; Shafiee-Sabet, M.; Harirchian, M.H. A 22-week, multicenter, randomized, double-blind controlled trial of Crocus sativus in the treatment of mild-to-moderate Alzheimer’s disease. Psychopharmacology 2010, 207, 637–643. [Google Scholar] [CrossRef] [PubMed]
- Falsini, B.; Piccardi, M.; Minnella, A.; Savastano, C.; Capoluongo, E.; Fadda, A.; Balestrazzi, E.; Maccarone, R.; Bisti, S. Influence of saffron supplementation on retinal flicker sensitivity in early age-related macular degeneration. Invest. Ophthalmol. Vis. Sci. 2010, 51, 6118–6124. [Google Scholar] [CrossRef] [PubMed]
- Kianbakht, S.; Ghazavi, A. Immunomodulatory effects of saffron: A randomized double-blind placebo-controlled clinical trial. Phytother. Res. 2011, 25, 1801–1805. [Google Scholar] [CrossRef] [PubMed]
- Safarinejad, M.R.; Shafiei, N.; Safarinejad, S. A prospective double-blind randomized placebo-controlled study of the effect of saffron (Crocus sativus Linn) on semen parameters and seminal plasma antioxidant capacity in infertile men with idiopathic oligoasthenoteratozoospermia. Phytother. Res. 2011, 25, 8–516. [Google Scholar] [CrossRef]
- Modabbernia, A.; Sohrabi, H.; Nasehi, A.A.; Raisi, F.; Saroukhani, S.; Jamshidi, A.; Tabrizi, M.; Ashrafi, M.; Akhondzadeh, S. Effect of saffron on fluoxetine-induced sexual impairment in men: Randomized double-blind placebo-controlled trial. Psychopharmacology 2012, 223, 381–388. [Google Scholar] [CrossRef]
- Ayatollahi, H.; Javan, A.O.; Khajedaluee, M.; Shahroodian, M.; Hosseinzadeh, H. Effect of Crocus sativus L. (saffron) on coagulation and anticoagulation systems in healthy volunteers. Phytother. Res. 2014, 28, 539–543. [Google Scholar] [CrossRef]
- Kashani, L.; Raisi, F.; Saroukhani, S.; Sohrabi, H.; Modabbernia, A.; Nasehi, A.A.; Jamshidi, A.; Ashrafi, M.; Mansouri, P.; Ghaeli, P.; et al. Saffron for treatment of fluoxetine-induced sexual dysfunction in women: Randomized double-blind placebo-controlled study. Hum. Psychopharmacol. 2013, 28, 54–60. [Google Scholar] [CrossRef] [PubMed]
- Marangoni, D.; Falsini, B.; Piccardi, M.; Ambrosio, L.; Minnella, A.M.; Savastano, M.C.; Bisti, S.; Maccarone, R.; Fadda, A.; Mello, E.; et al. Functional effect of Saffron supplementation and risk genotypes in early age-related macular degeneration: A preliminary report. J. Transl. Med. 2013, 25, 11–228. [Google Scholar] [CrossRef] [PubMed]
- Jabbarpoor Bonyadi, M.H.; Yazdani, S.; Saadat, S. The ocular hypotensive effect of saffron extract in primary open angle glaucoma: A pilot study. BMC Complement. Altern. Med. 2014, 15, 14–399. [Google Scholar] [CrossRef] [PubMed]
- Shahmansouri, N.; Farokhnia, M.; Abbasi, S.H.; Kassaian, S.E.; Tafti, A.A.N.; Gougol, A.; Yekehtaz, H.; Forghani, S.; Mahmoodian, M.; Saroukhani, S.; et al. A randomized, double-blind, clinical trial comparing the efficacy and safety of Crocus sativus L. with fluoxetine for improving mild to moderate depression in post percutaneous coronary intervention patients. J. Affect. Disord. 2014, 155, 216–222. [Google Scholar] [CrossRef]
- Kamalipour, M.; Akhondzadeh, S. Cardiovascular effects of saffron: An evidence-based review. J. Tehran Heart Cent. 2011, 6, 59–61. [Google Scholar]
- Carmona, M.; Zalacain, A.; Sánchez, A.M.; Novella, J.L.; Alonso, G.L. Crocetin esters, picrocrocin and its related compounds present in Crocus sativus stigmas and Gardenia jasminoides fruits. Tentative identification of seven new compounds by LC-ESI-MS. J. Agric. Food Chem. 2006, 54, 973–979. [Google Scholar] [CrossRef]
- He, S.Y.; Qian, Z.Y.; Wen, N.; Tang, F.T.; Xu, G.L.; Zhou, C.H. Influence of Crocetin on experimental atherosclerosis in hyperlipidamic-diet quails. Eur. J. Pharmacol. 2007, 554, 191–195. [Google Scholar] [CrossRef]
- Yoshino, F.; Yoshida, A.; Umigai, N.; Kubo, K.; Masaichi, C.I.L. Crocetin reduces the oxidative stress induced reactive oxygen species in the stroke-prone spontaneously hypertensive rats (SHRSPs) brain. J. Clin. Biochem. Nutr. 2011, 49, 182–187. [Google Scholar] [CrossRef]
- Samarghandian, S.; Azimi-Nezhad, M.; Farkhondeh, T. Crocin attenuate Tumor Necrosis Factor-alpha (TNF-α) and interleukin-6 (IL-6) in streptozotocin-induced diabetic rat aorta. Cytokine 2016, 88, 20–28. [Google Scholar] [CrossRef]
- Asdaq, S.M.; Inamdar, M.N. Potential of Crocus sativus (saffron) and its constituent, crocin, as hypolipidemic and antioxidant in rats. Appl. Biochem. Biotechnol. 2010, 162, 358–372. [Google Scholar] [CrossRef] [PubMed]
- Hänsel, R. Medizinische Empirie und moderne Arzneimittelforschung. Dtsch. Apoth. Zeitag 1977, 117, 1672–1679. [Google Scholar]
- Gainer, J.L. Trans-sodium crocetinate for treating hypoxia/ischemia. Expert Opin. Investig. Drugs 2008, 17, 917–924. [Google Scholar] [CrossRef] [PubMed]
- Zheng, S.; Qian, Z.; Tang, F.; Sheng, L. Suppression of vascular cell adhesion molecule-1 expression by crocetin contributes to attenuation of atherosclerosis in hypercholesterolemic rabbits. Biochem. Pharmacol. 2005, 70, 1192–1199. [Google Scholar] [CrossRef]
- Sheng, L.; Qian, Z.; Zheng, S.; Sun, S. Mechanism of hypolidemic effect of crocin in rats: Crocin inhibits pancreatic lipase. Eur. J. Pharmacol. 2006, 543, 116–122. [Google Scholar] [CrossRef]
- Shirali, S.; Zahra Bathaie, S.; Nakhjavani, M. Effect of crocin on the insulin resistance and lipid profile of streptozotocin-induced diabetic rats. Phytother. Res. 2013, 27, 1042–1047. [Google Scholar] [CrossRef]
- Hosseinzadeh, H.; Nassiri-Asl, M. Acute and subacute toxicity of safranal, a constituent of saffron, in mice and rats. Phytother. Res. 2013, 27, 475–483. [Google Scholar] [CrossRef]
- Shati, A.A.; Alamri, S.A. Role of saffron (Crocus sativus L.) and honey syrup on aluminum-induced hepatotoxicity. Saudi Med. J. 2010, 31, 1106–1113. [Google Scholar]
- Valle Garcia-Rodriguez, M.; Serrano-Diaz, J.; Tarantilis, P.A.; Lopez-Corcoles, H.; Carmona, M.; Alonso, G.L. Determination of saffron quality by high-performance liquid chromatography. J. Agric. Food Chem. 2014, 62, 8068–8074. [Google Scholar] [CrossRef]
- ISO 3632. Saffron (Crocus Sativus, L.). Part 1 (Specification) and Part 2 (Test Methods); International Organization for Standardization: Geneva, Switzerland, 2011. [Google Scholar]
- Moshiri, M.; Vahabzadeh, M.; Hosseinzadeh, H. Clinical Applications of Saffron (Crocus sativus) and Its Constituents: A Review. Drug Res. 2014, 65, 287–295. [Google Scholar] [CrossRef]
- Srivastava, R.; Ahmed, H.; Dixit, R.K.; Dharamveer, S.S. Crocus sativus L.: A comprehensive review. Pharmacogn. Rev. 2010, 4, 200–208. [Google Scholar] [CrossRef] [PubMed]
- Modaghegh, M.H.; Shahabian, M.; Esmaeili, H.A.; Rajbai, O.; Hosseinzadeh, H. Safety evaluation of saffron (Crocus sativus) tablets in healthy volunteers. Phytomedicine 2008, 15, 1032–1037. [Google Scholar] [CrossRef] [PubMed]
- Lage, M.; Cantrel, C.L. Quantification of saffron (Crocus sativus L.) metabolites crocins, picrocrocin and safranal for quality determination of the spice grown under different environmental Moroccan conditions. Sci. Hortic. 2009, 121, 366–373. [Google Scholar] [CrossRef]
- Del Campo, C.P.; Carmona, M.; Maggi, L.; Kanakis, C.D.; Anastasaki, E.G.; Tarantilis, P.A.; Polissiou, M.G.; Alonso, G.L. Effects of mild temperature conditions during dehydration procedures on saffron quality parameters. J. Sci. Food Agric. 2010, 90, 719–725. [Google Scholar] [CrossRef]
- García-Rodríguez, M.V.; López-Córcoles, H.; Alonso, G.L.; Pappas, C.S.; Polissiou, M.G.; Tarantilis, P.A. Comparative evaluation of an ISO 3632 method and an HPLC-DAD method for safranal quantity determination in saffron. Food Chem. 2017, 221, 838–843. [Google Scholar] [CrossRef]
- Sahraian, A.; Jelodar, S.; Javid, Z.; Mowla, A.; Ahmadzadeh, L. Study the effects of saffron on depression and lipid profiles: A double blind comparative study. Asian J. Psychiatr. 2016, 22, 174–176. [Google Scholar] [CrossRef]
- Rahmani, J.; Manzari, N.; Thompson, J.; Clak, C.C.T.; Villanueva, G.; Varkaneh, H.K.; Mirmiran, P. The effect of saffron on weight and lipid profile: A systematic review, meta-analysis, and dose–response of randomized clinical trials. Phytother. Res. 2019, 33, 2244–2255. [Google Scholar] [CrossRef]
- Nichols, M.; Townsend, N.; Luengo-Fernandez, R.; Leal, J.; Gray, A.; Scarborough, P.; Rayner, M. European Cardiovascular Disease Statistics; European Heart Network: Brussels, Belgium, 2012. [Google Scholar]
- Heidenreich, P.A.; Trogdon, J.G.; Khavjou, O.A.; Butler, J.; Dracup, K.; Ezekowitz, M.D.; Finkelstein, E.A.; Hong, Y.; Johnston, S.C.; Khera, A.; et al. Forecasting the future of cardiovascular disease in the United States: A policy statement from the American Heart Association. Circulation. Circulation 2011, 123, 933–944. [Google Scholar] [CrossRef]
- Ezzati, M.; Lopez, A.D.; Rodgers, A.; Murray, C.J.L. Comparative Quantification of Health Risks: Global and Regional Burden of Disease Attributable to Selected Major Risk Factors; World Health Organization: Geneva, Switzerland, 2004. [Google Scholar]
| Route of Administration | n 1 | Pathology | Dose | Days | Reference |
|---|---|---|---|---|---|
| Capsules | 46 | Alzheimer | 30 | 112 | [7] |
| Saffron | 35 | Macular degeneration | 20 | 84 | [8] |
| Tablets | 45 | Immunological disease | 100 | 21 | [9] |
| Saffron | 260 | Infertility | 60 | 182 | [10] |
| Saffron | 36 | Sexual dysfunction | 30 | 28 | [11] |
| Tablets | 60 | Macular degeneration | 43 | 7 | [12] |
| Saffron | 34 | Sexual dysfunction in women | 30 | 42 | [13] |
| Oral supplements | 33 | Macular degeneration | 20 | 84 | [14] |
| Aqueous extracts | 17 | Primary open angle glaucoma | 30 | 28 | [15] |
| Capsules | 40 | Depression | 30 | 42 | [16] |
| Control | Days | A257 | A330 | A440 | Picr | Saf | ΣCrc | Σt-Crc |
|---|---|---|---|---|---|---|---|---|
| 1 | 0 | 84.4 ± 1.2 b | 31.6 ± 0.2 b | 229 ± 6 | 12.3 ± 0.5 | 0.11 ± 0.00 c | 21.2 ± 0.8 a | 18.9 ± 0.5 a |
| 2 | 45 | 87.1 ± 1.1 a | 33.6 ± 0.3 a | 226 ± 5 | 12.9 ± 0.6 | 0.28 ± 0.01 b | 20.8 ± 0.7 a | 17.8 ± 0.4 b |
| 3 | 96 | 66.2 ± 0.9 c | 20.2 ± 0.1 c | 219 ± 4 | 13.0 ± 0.6 | 0.30 ± 0.01 a | 17.9 ± 0.9 b | 17.0 ± 0.6 b |
| Pre Treatment | Post Treatment | p-Value | |
|---|---|---|---|
| TC level | 199.5 ± 27.6 a | 179.5 ± 19.8 b | 0.002 |
| TG level | 124.0 ± 80.0 a | 101.0 ± 51.6 b | 0.002 |
| LDL-C level | 124.86 ± 35.76 | 115.86 ± 27.04 | - |
| HDL-C level | 56.57 ± 12.42 | 60.14 ± 16.34 | - |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bagur, M.J.; Alonso Salinas, G.L.; Jiménez-Monreal, A.M.; Serrano-Heras, G.; Martínez-Tomé., M.; Alonso, G.L. Effect of Daily Intake of a Saffron Infusion on Blood Cholesterol Levels. Appl. Sci. 2020, 10, 4763. https://doi.org/10.3390/app10144763
Bagur MJ, Alonso Salinas GL, Jiménez-Monreal AM, Serrano-Heras G, Martínez-Tomé. M, Alonso GL. Effect of Daily Intake of a Saffron Infusion on Blood Cholesterol Levels. Applied Sciences. 2020; 10(14):4763. https://doi.org/10.3390/app10144763
Chicago/Turabian StyleBagur, M. José, Gonzalo L. Alonso Salinas, Antonia M. Jiménez-Monreal, Gemma Serrano-Heras, Magdalena Martínez-Tomé., and Gonzalo L. Alonso. 2020. "Effect of Daily Intake of a Saffron Infusion on Blood Cholesterol Levels" Applied Sciences 10, no. 14: 4763. https://doi.org/10.3390/app10144763
APA StyleBagur, M. J., Alonso Salinas, G. L., Jiménez-Monreal, A. M., Serrano-Heras, G., Martínez-Tomé., M., & Alonso, G. L. (2020). Effect of Daily Intake of a Saffron Infusion on Blood Cholesterol Levels. Applied Sciences, 10(14), 4763. https://doi.org/10.3390/app10144763







