Influence of Chloride Ions on Electrochemical Corrosion Behavior of Dual-Phase Steel over Conventional Rebar in Pore Solution
Abstract
1. Introduction
2. Materials and Methods
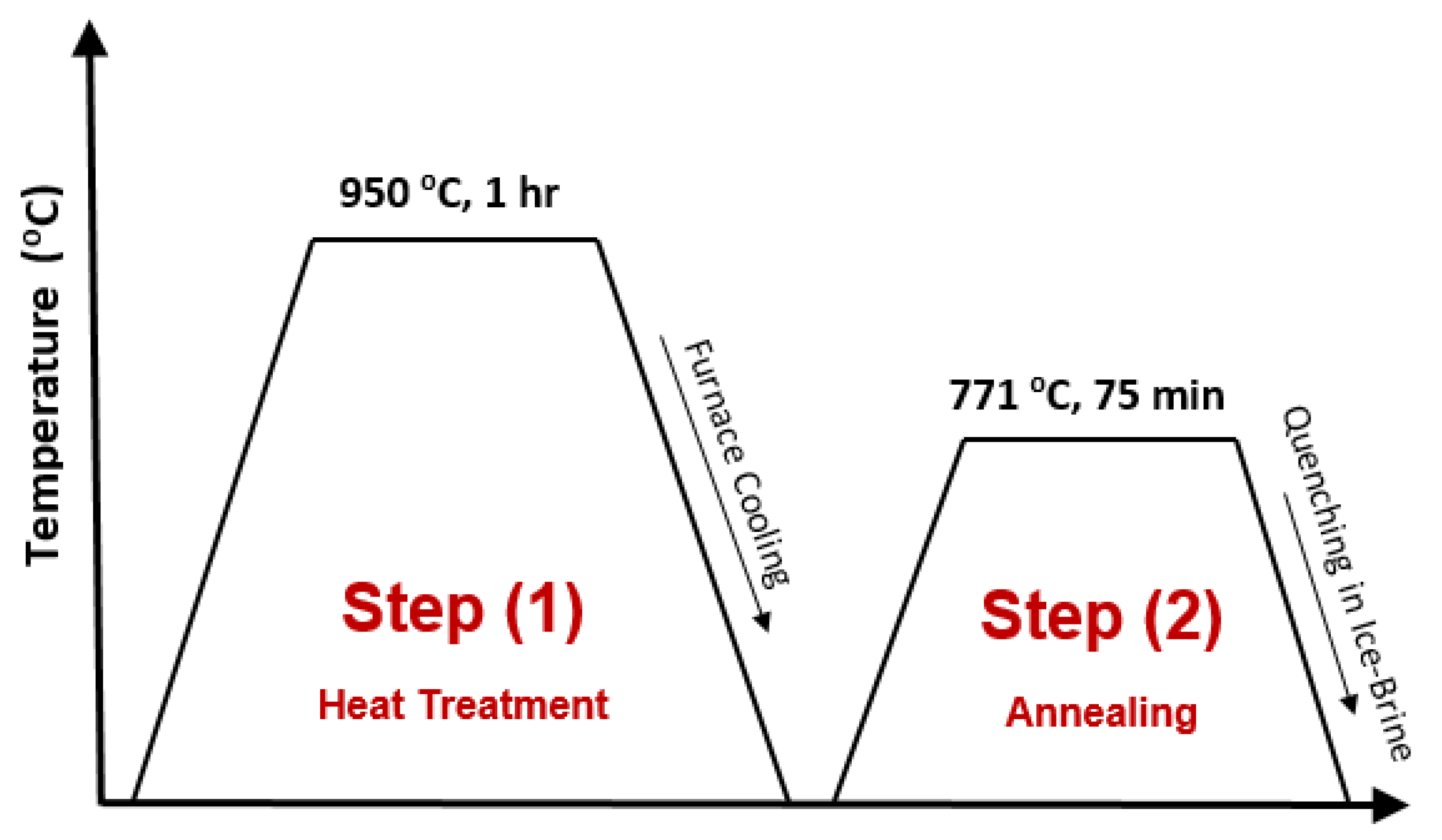
2.1. Heat Treatment
2.2. Metallography
2.3. Hardness
2.4. Volume Fraction
2.5. Corrosion
3. Results and Discussion
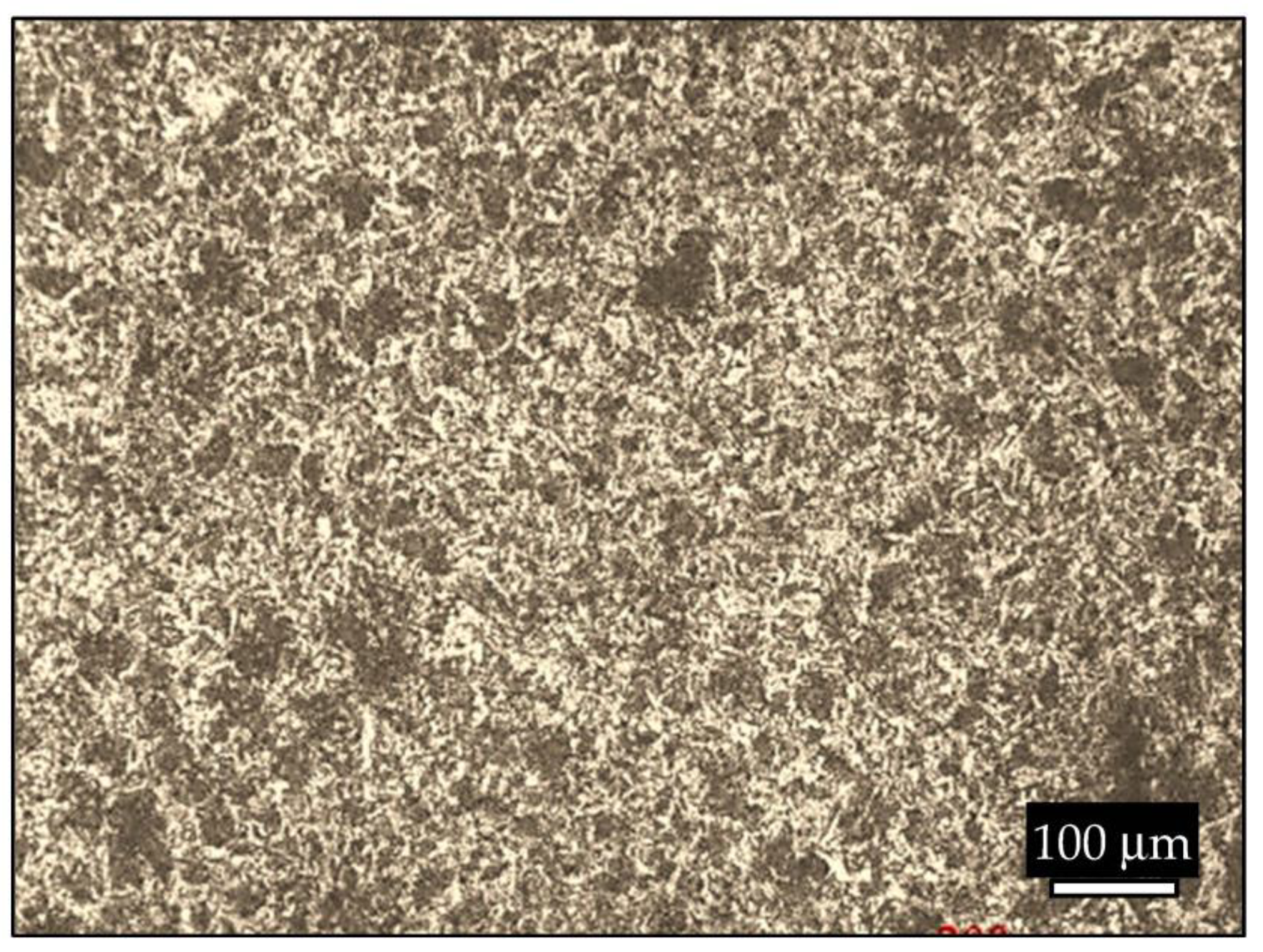
3.1. Microstructure
3.2. Hardness
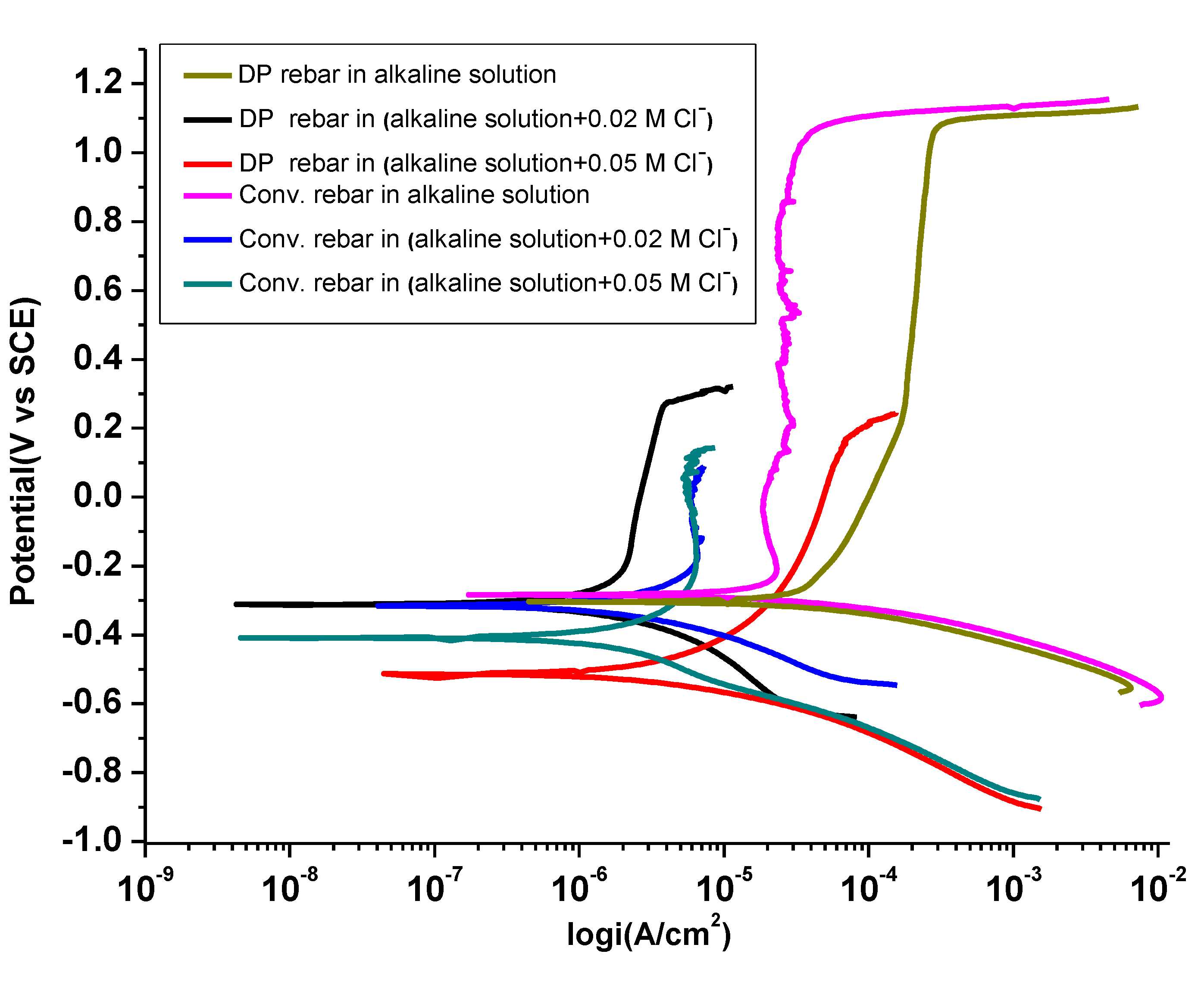
3.3. Potentiodynamic Polarization
3.3.1. Influence of Cl− ions on Passivity in Alkaline Solution
3.3.2. Oxide or Passive Layer Breakdown Mechanism and Pit Morphology
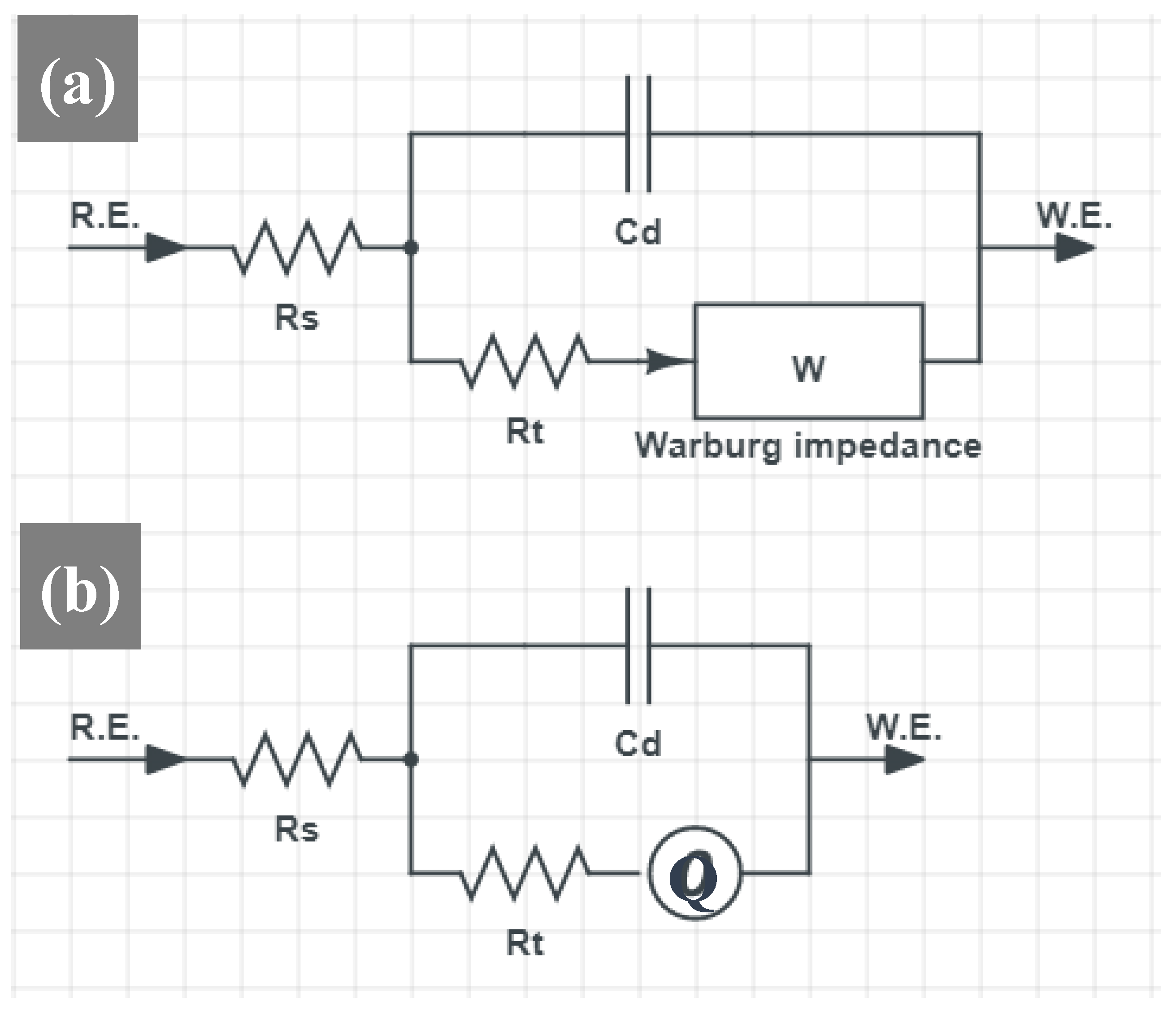
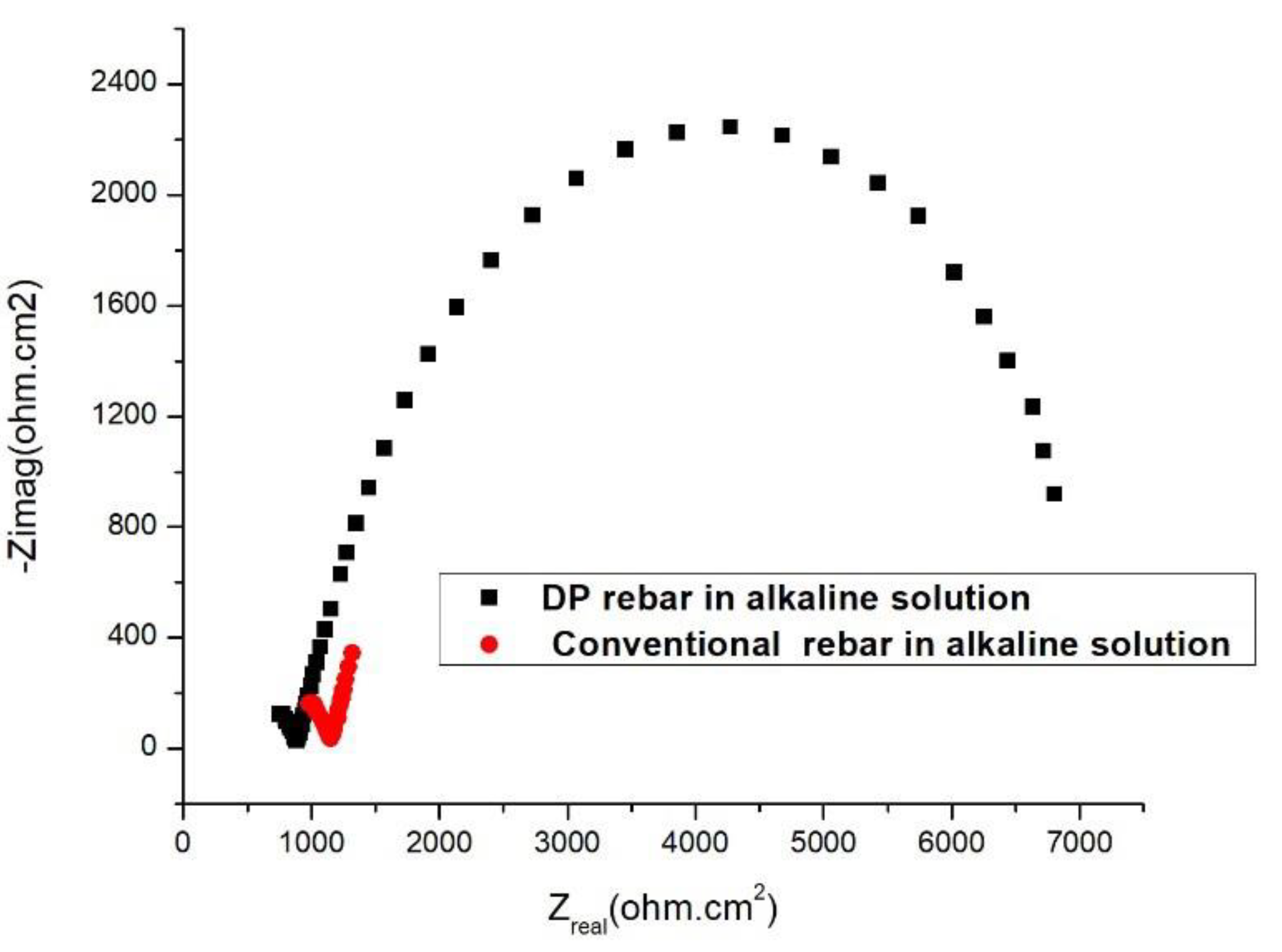
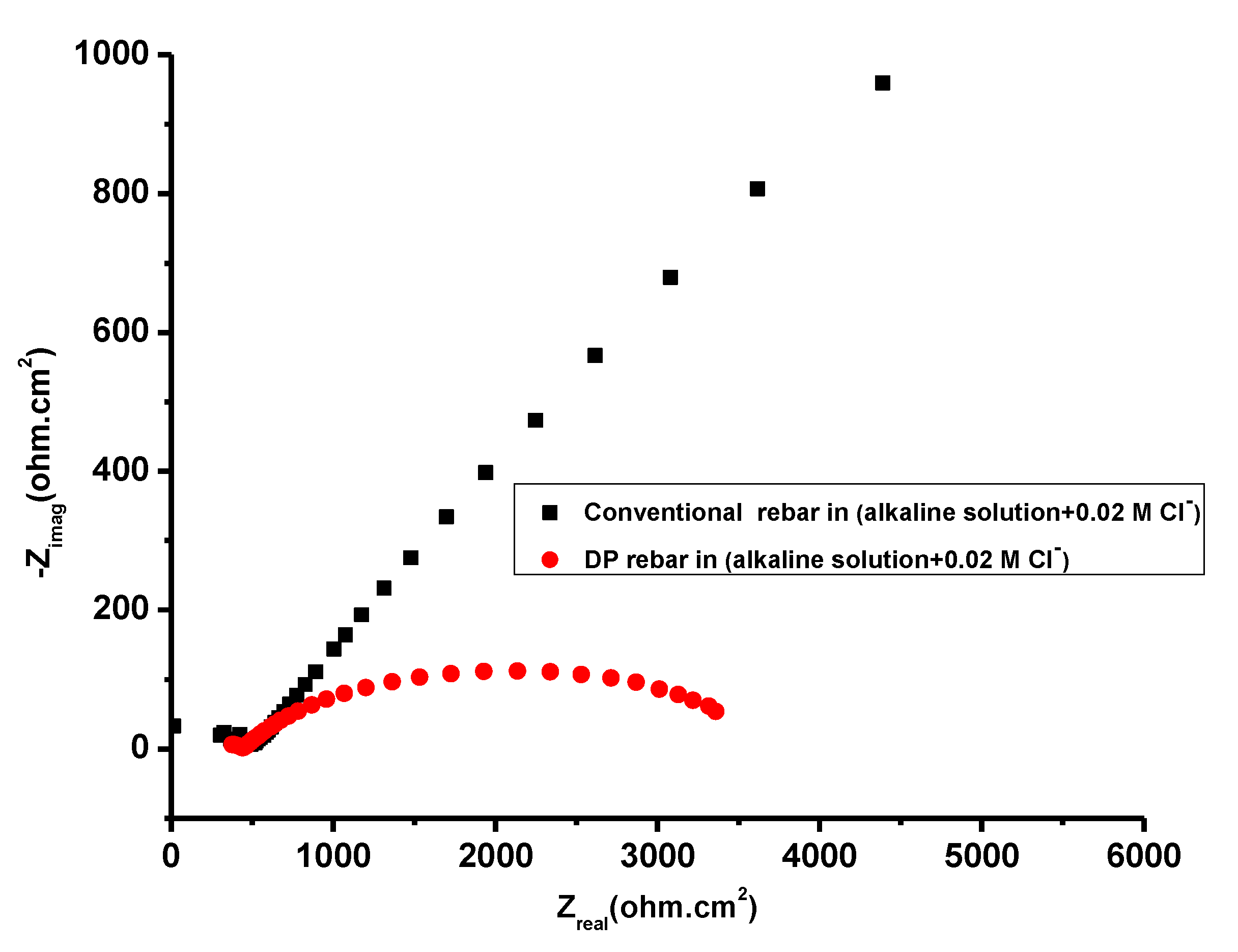
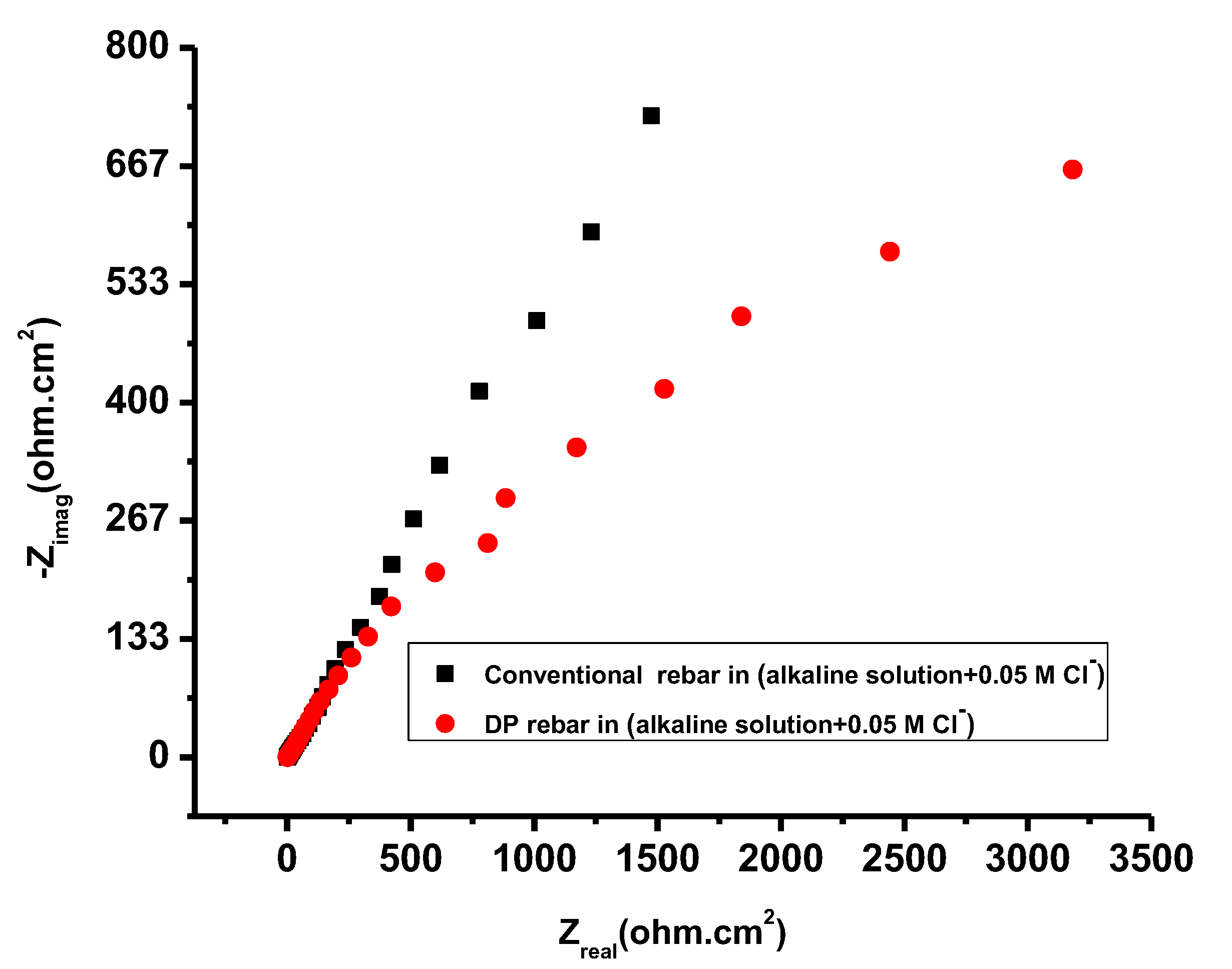
3.4. Electrochemical Impedance Spectroscopy
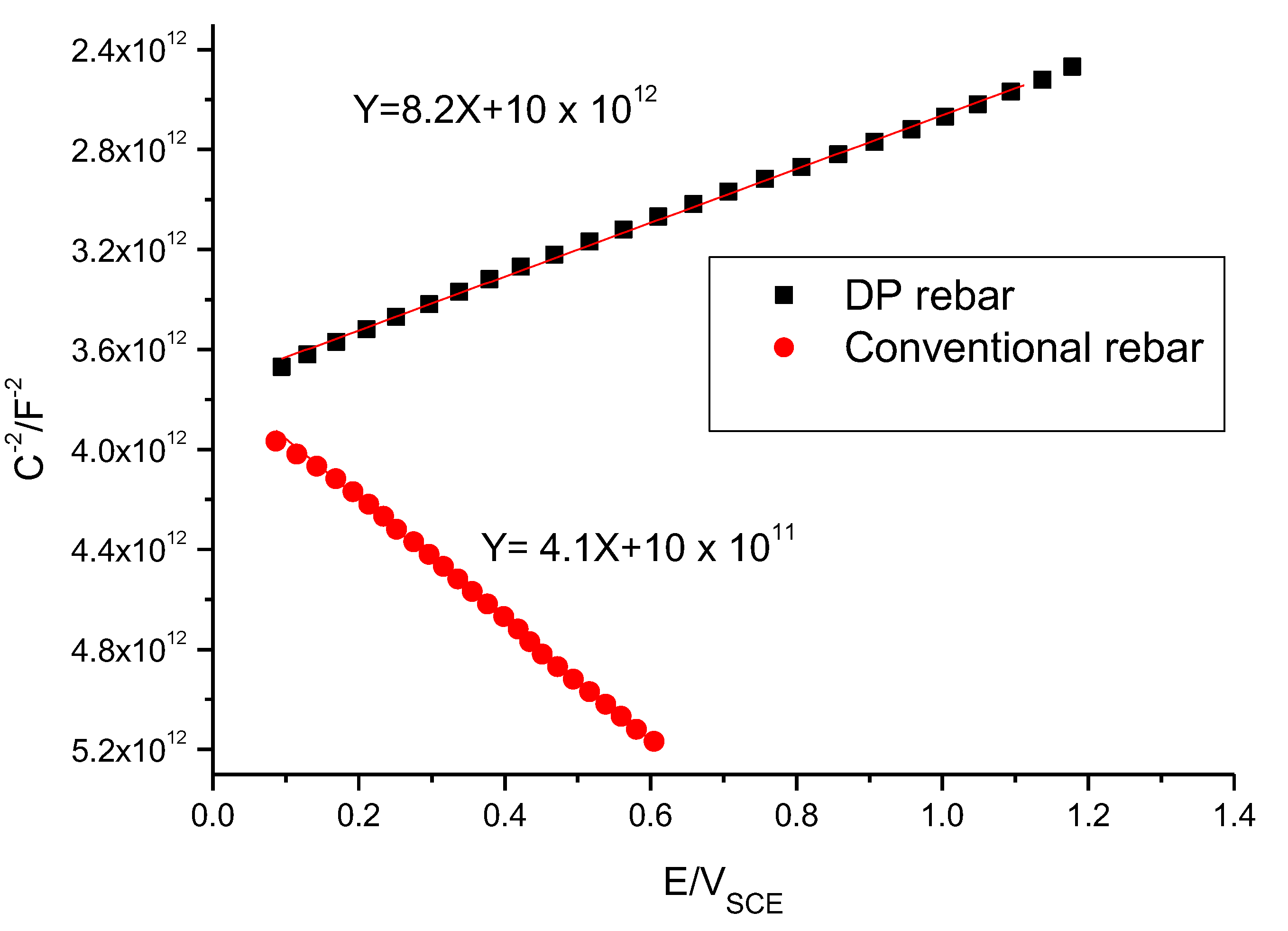
3.5. Capacitance
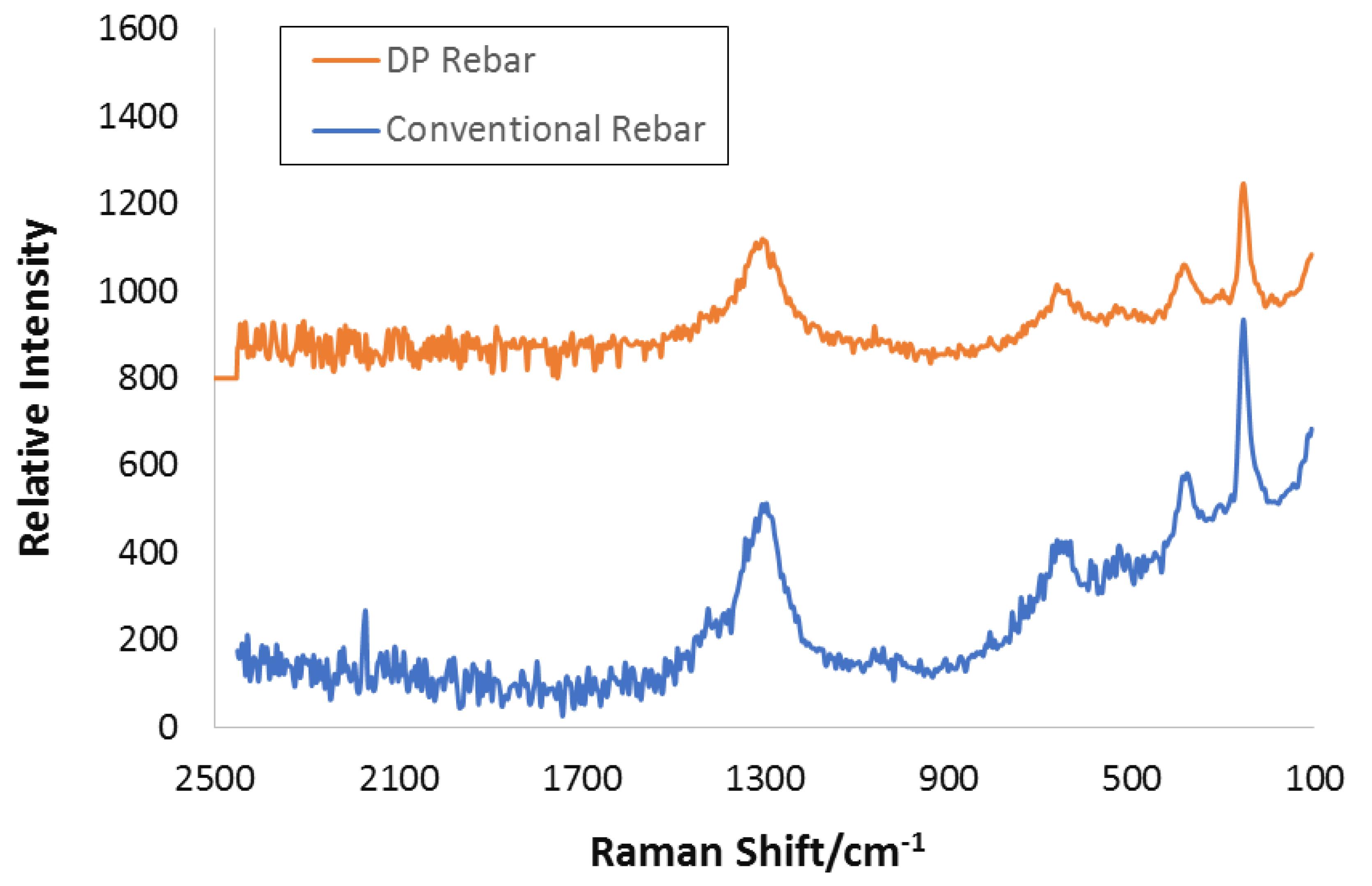
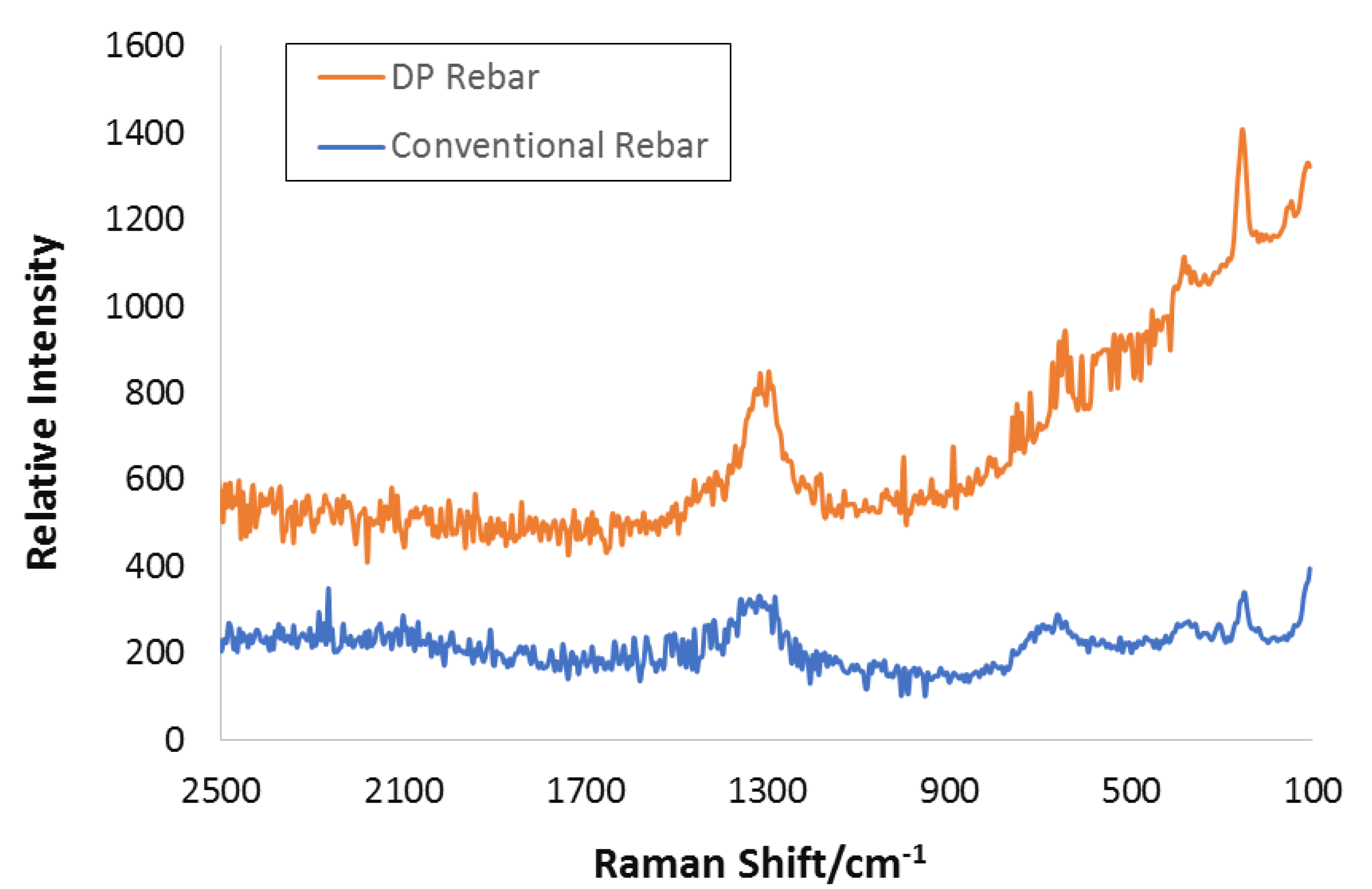
3.6. Raman Spectroscopy
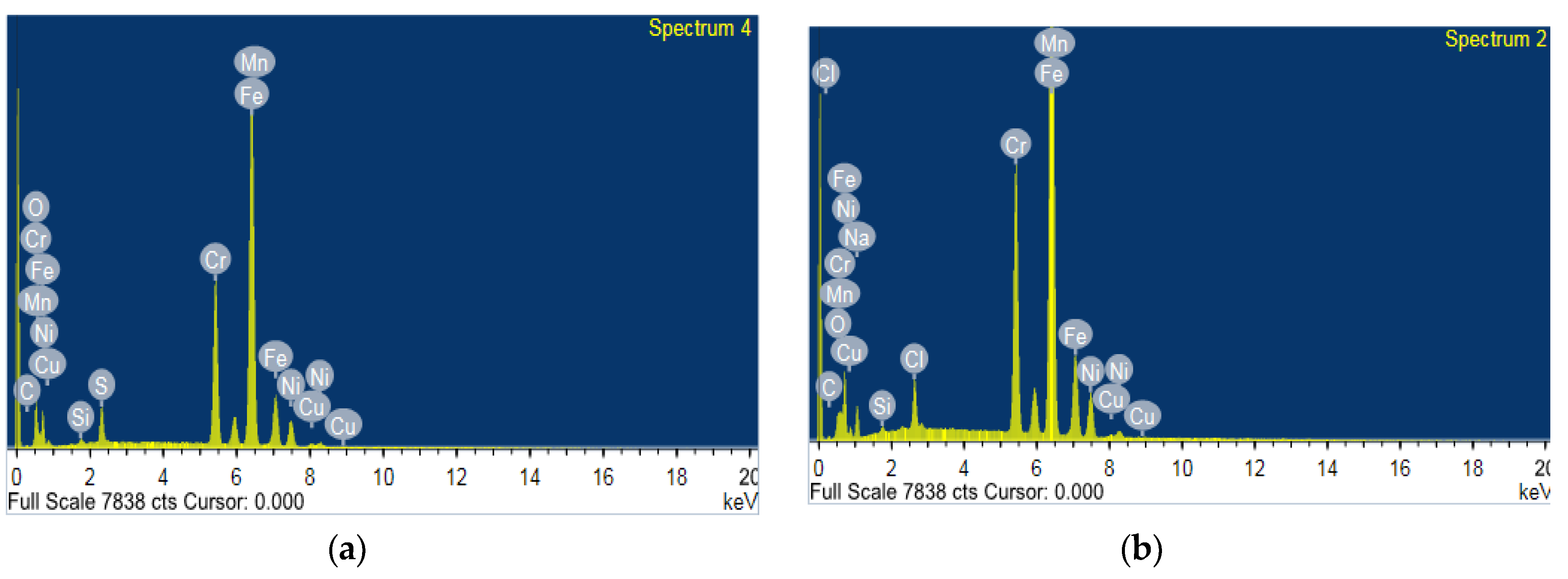
3.7. Scanning Electron Microscopy
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Alonso, C.; Andrade, C.; Castellote, M.; Castro, P. Chloride threshold values to depassivate reinforcing bars embedded in a standardized OPC mortar. Cem. Concr. Res. 2000, 30, 1047–1055. [Google Scholar] [CrossRef]
- Kayyali, O.A.; Haque, M.N. The C1−/OH− ratio in chloride-contaminated concrete—A most important criterion. Mag. Concr. Res. 1995, 47, 235–242. [Google Scholar] [CrossRef]
- Raupach, M. Chloride-induced macrocell corrosion of steel in concrete—Theoretical background and practical consequences. Constr. Build. Mater. 1996, 10, 329–338. [Google Scholar] [CrossRef]
- Thomas, M. Chloride thresholds in marine concrete. Cem. Concr. Res. 1996, 26, 513–519. [Google Scholar] [CrossRef]
- Melchers, R.E.; Li, C.Q. Reinforcement corrosion initiation and activation times in concrete structures exposed to severe marine environments. Cem. Concr. Res. 2009, 39, 1068–1076. [Google Scholar] [CrossRef]
- Lambert, P.; Page, C.L.; Vassie, P.R.W. Investigations of reinforcement corrosion. 2. Electrochemical monitoring of steel in chloride-contaminated concrete. Mater. Struct. 1991, 24, 351–358. [Google Scholar] [CrossRef]
- Gouda, V.K.; Shater, M.A. ChemInform Abstract: Corrosion inhibition of reinforcing steel by using hydrazine hydrate. Chem. Informationsd. 1975, 6. [Google Scholar] [CrossRef]
- Bouteiller, V.; Cremona, C.; Baroghel-Bouny, V.; Maloula, A. Corrosion initiation of reinforced concretes based on Portland or GGBS cements: Chloride contents and electrochemical characterizations versus time. Cem. Concr. Res. 2012, 42, 1456–1467. [Google Scholar] [CrossRef]
- Hansson, C.M.; Sorensen, B. Threshold concentration of chloride in concrete for the initiation of reinforcement corrosion. ASTM Spec. Tech. Publ. 1990, 19, 3–16. [Google Scholar]
- Mangat, P.S.; Molloy, B.T. Factors influencing chloride-induced corrosion of reinforcement in concrete. Mater. Struct. 1992, 25, 404–411. [Google Scholar] [CrossRef]
- Almusallam, A.A. Effect of degree of corrosion on the properties of reinforcing steel bars. Constr. Build. Mater. 2001, 15, 361–368. [Google Scholar] [CrossRef]
- Goñi, S.; Andrade, C. Synthetic concrete pore solution chemistry and rebar corrosion rate in the presence of chlorides. Cem. Concr. Res. 1990, 20, 525–539. [Google Scholar] [CrossRef]
- Hansson, C.M. Corrosion of Reinforcing Bars in Concrete. ACI J. Proc. 1965, 62. [Google Scholar] [CrossRef]
- Glass, G.K.; Buenfeld, N.R. The influence of chloride binding on the chloride induced corrosion risk in reinforced concrete. Corros. Sci. 2000, 42, 329–344. [Google Scholar] [CrossRef]
- Andrade, C.; Page, C.L. Pore solution chemistry and corrosion in hydrated cement systems containing chloride salts: A study of cation specific effects. Br. Corros. J. 1986, 21, 49–54. [Google Scholar] [CrossRef]
- Li, L.; Sagüés, A.A. Effect of chloride concentration on the pitting and repassivation potentials of reinforcing steel in alkaline solutions. In Proceedings of the NACE—International Corrosion Conference Series, San Antonio, Texas, USA, 25–30 April 1999; p. 19. [Google Scholar]
- Alonso, C.; Castellote, M.; Andrade, C. Chloride threshold dependence of pitting potential of reinforcements. Electrochim. Acta 2002, 47, 3469–3481. [Google Scholar] [CrossRef]
- Bertolini, L.; Bolzoni, F.; Pastore, T.; Pedeferri, P. Behaviour of stainless steel in simulated concrete pore solution. Br. Corros. J. 1996, 31, 218–222. [Google Scholar] [CrossRef]
- Schießl, P.; Raupach, M. Laboratory Studies and Calculations on the Influence of Crack Width on Chloride-Induced Corrosion of Steel in Concrete. ACI Mater. J. 1997, 94. [Google Scholar] [CrossRef]
- Andrade, C.; Maribona, I.R.; Feliu, S.; González, J.A.; Feliu, S. The effect of macrocells between active and passive areas of steel reinforcements. Corros. Sci. 1992, 33, 237–249. [Google Scholar] [CrossRef]
- Andrade, C.; Merino, P.; Nóvoa, X.R.; Pérez, M.C.; Soler, L. Passivation of Reinforcing Steel in Concrete. Mater. Sci. Forum 1995, 192, 891–898. [Google Scholar] [CrossRef]
- Moser, R.D.; Singh, P.M.; Kahn, L.F.; Kurtis, K.E. Chloride-induced corrosion resistance of high-strength stainless steels in simulated alkaline and carbonated concrete pore solutions. Corros. Sci. 2012, 57, 241–253. [Google Scholar] [CrossRef]
- Li, C.Q.; Yang, S.T. Prediction of Concrete Crack Width under Combined Reinforcement Corrosion and Applied Load. J. Eng. Mech. 2011, 137, 722–731. [Google Scholar] [CrossRef]
- Blunt, J.; Jen, G.; Ostertag, C.P. Enhancing corrosion resistance of reinforced concrete structures with hybrid fiber reinforced concrete. Corros. Sci. 2015, 92, 182–191. [Google Scholar] [CrossRef]
- Dieter, G.E. Mechanical Metallurgy; McGraw-Hill: New York, NY, USA, 1961. [Google Scholar]
- Galán, J.; Samek, L.; Verleysen, P.; Verbeken, K.; Houbaert, Y. Advanced high strength steels for automotive industry. Rev. Metal. 2012, 48, 118–131. [Google Scholar] [CrossRef]
- Mintz, B. Hot dip galvanising of transformation induced plasticity and other intercritically annealed steels. Int. Mater. Rev. 2001, 46, 169–197. [Google Scholar] [CrossRef]
- Bleck, W.; Phiu-On, K. Microalloying of cold-formable multi phase steel grades. Mater. Sci. Forum 2005, 500, 97–114. [Google Scholar] [CrossRef]
- Ball, A.; Chauhan, Y.; Schaffer, G.B. Microstructure, phase equilibria, and transformations in corrosion resistant dual phase steel designated 3CR12. Mater. Sci. Technol. 1987, 3, 189–196. [Google Scholar] [CrossRef]
- Verdeja, J.I.; Pero-Sanz, J.A.; Asensio, J. Multiphase Steels: Structure-Mechanical Properties Relationships in the Cold Rolled and Continuous Annealed Condition. Mater. Sci. Forum 2005, 500–501, 429–436. [Google Scholar] [CrossRef]
- Byers, H.G.; Langdon, S.C. Passivity of Metals. J. Am. Chem. Soc. 1914, 36, 2004–2011. [Google Scholar] [CrossRef]
- Bode, R.; Meurer, M.; Schaumann, T.W.; Warnecke, W. Selection and use of coated advanced high-strength steels for automotive applications. Rev. Métallurgie 2004, 101, 551–558. [Google Scholar] [CrossRef]
- Zackay, V.; Parker, E.R.; Fahr, D.; Busch, R. The enhancement of ductility in high-strength steels. Trans. ASM 1967, 60, 252–259. [Google Scholar]
- Parsons, R. Atlas of electrochemical equilibria in aqueous solutions. J. Electroanal. Chem. Interfacial Electrochem. 1967, 13, 471. [Google Scholar] [CrossRef]
- Galvele, J.R. Effect of Molybdenum on the Pitting Potential of High Purity 18% Cr Ferritic Stainless Steels. J. Electrochem. Soc. 1978, 125, 1204. [Google Scholar] [CrossRef]
- Park, S.A.; Kim, S.H.; Yoo, Y.H.; Kim, J.G. Effect of chloride ions on the corrosion behavior of low-alloy steel containing copper and antimony in sulfuric acid solution. Met. Mater. Int. 2015, 21, 470–478. [Google Scholar] [CrossRef]
- Mao, X.; Liu, X.; Revie, R.W. Pitting Corrosion of Pipeline Steel in Dilute Bicarbonate Solution with Chloride Ions. Corrosion 1994, 50, 651–657. [Google Scholar] [CrossRef]
- El-Moneim, A.; Gebert, A.; Uhlemann, M.; Gutfleisch, O.; Schultz, L. The influence of Co and Ga additions on the corrosion behavior of nanocrystalline NdFeB magnets. Corros. Sci. 2002, 44, 1857–1874. [Google Scholar] [CrossRef]
- González-Cortés, A. Electrochemical Impedance Spectroscopy. In Agricultural and Food Electroanalysis; John Wiley & Sons, Ltd.: Chichester, UK, 2015; pp. 381–419. ISBN 9781118684030. [Google Scholar]
- Orazem, M.E.; Tribollet, B. Electrochemical Impedance Spectroscopy; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2008; ISBN 9780470381588. [Google Scholar]
- Cicek, V. Corrosion Engineering; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2014; ISBN 9781118720837. [Google Scholar]
- Leidheiser, H. Electrochemistry at semiconductor and oxidized metal electrodes. J. Colloid Interface Sci. 1982, 88, 310. [Google Scholar] [CrossRef]
- Sato, N. Electrochemistry at Metal and Semiconductor Electrodes; Elsevier: Amsterdam, The Netherlands, 1998; ISBN 9780444828064. [Google Scholar]
- John, N.; George, S. Raman Spectroscopy. In Spectroscopic Methods for Nanomaterials Characterization; Elsevier: Amsterdam, The Netherlands, 2017; pp. 95–127. ISBN 9780323461467. [Google Scholar]
- Baek, W.C.; Kang, T.; Sohn, H.J.; Kho, Y.T. In situ surface enhanced Raman spectroscopic study on the effect of dissolved oxygen on the corrosion film on low carbon steel in 0.01 M NaCl solution. Electrochim. Acta 2001, 46, 2321–2325. [Google Scholar] [CrossRef]
- Ohtsuka, T.; Kubo, K.; Sato, N. Raman Spectroscopy of Thin Corrosion Films on Iron at 100 to 150 C in Air. Corrosion 1986, 42, 476–481. [Google Scholar] [CrossRef]
- Otto, A.; Bruckbauer, A.; Chen, Y. On the chloride activation in SERS and single molecule SERS. J. Mol. Struct. 2003, 661, 501–514. [Google Scholar] [CrossRef]
- Li, Y.S.; Li, J.C.; Cheng, Y.W. Surface-enhanced raman spectra of dyes and organic acids in silver solutions: Chloride ion effect. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2003, 56A, 2067. [Google Scholar]
- Kumar, V. Protection of Steel Reinforcement for Concrete—A Review. Corros. Rev. 1998, 16. [Google Scholar] [CrossRef]
- Koleva, D.A.; de Wit, J.H.W.; van Breugel, K.; Lodhi, Z.F.; van Westing, E. Investigation of Corrosion and Cathodic Protection in Reinforced Concrete. J. Electrochem. Soc. 2007, 154, 52. [Google Scholar] [CrossRef]
- Ryan, M.P.; Williams, D.E.; Chater, R.J.; Hutton, B.M.; McPhail, D.S. Why stainless steel corrodes. Nature 2002, 415, 770–774. [Google Scholar] [CrossRef] [PubMed]
- Ke, R. Initiation of Corrosion Pits at Inclusions on 304 Stainless Steel. J. Electrochem. Soc. 1995, 142, 4056. [Google Scholar] [CrossRef]
- Punckt, C. Sudden Onset of Pitting Corrosion on Stainless Steel as a Critical Phenomenon. Science 2004, 305, 1133–1136. [Google Scholar] [CrossRef] [PubMed]



| (a) | (b) | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| C | Mn | Si | S | P | V | Mo | Nb | C | Mn | Si | S | P |
| 0.20 | 0.70 | 0.172 | 0.027 | 0.020 | <0.02 | <0.02 | <0.01 | 0.10 | 0.90 | 0.30 | 0.010 | 0.02 |
| Material | Solution | icorr (A/cm2) µA | Ecorr (mv) (V vs. SCE) | βc (V/decade) | βa (V/decade) | Ru (ohm cm2) × 10−6 | Rp (ohm cm2) | Cdl × 10−4 (F cm2) | X2, Chi-Squared × 10−2 |
|---|---|---|---|---|---|---|---|---|---|
| DP steel | Alkaline | 12 | −294 | 0.151 | 0.140 | 132 | 7000 | 7.24 | 3.142 |
| Rebar | 25 | −300 | 0.159 | 0.148 | 154 | 6100 | 8.45 | 3.876 | |
| DP steel | Alkaline +0.02 M Cl− | 80 | −304 | 0.168 | 0.156 | 160 | 3600 | 9.26 | 3.768 |
| Rebar | 104 | −310 | 0.170 | 0.162 | 172 | 300 | 10.89 | 4.181 | |
| DP steel | Alkaline +0.05M Cl− | 102 | −400 | 0.180 | 0.172 | 183 | 2800 | 11.98 | 4.251 |
| Rebar | 143 | −550 | 0.192 | 0.180 | 190 | 2260 | 14.10 | 4.166 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abdo, H.S.; Seikh, A.H.; Mohammed, J.A.; Luqman, M.; Ragab, S.A.; Almotairy, S.M. Influence of Chloride Ions on Electrochemical Corrosion Behavior of Dual-Phase Steel over Conventional Rebar in Pore Solution. Appl. Sci. 2020, 10, 4568. https://doi.org/10.3390/app10134568
Abdo HS, Seikh AH, Mohammed JA, Luqman M, Ragab SA, Almotairy SM. Influence of Chloride Ions on Electrochemical Corrosion Behavior of Dual-Phase Steel over Conventional Rebar in Pore Solution. Applied Sciences. 2020; 10(13):4568. https://doi.org/10.3390/app10134568
Chicago/Turabian StyleAbdo, Hany S., Asiful H. Seikh, Jabair A. Mohammed, Monis Luqman, Sameh A. Ragab, and Saud M. Almotairy. 2020. "Influence of Chloride Ions on Electrochemical Corrosion Behavior of Dual-Phase Steel over Conventional Rebar in Pore Solution" Applied Sciences 10, no. 13: 4568. https://doi.org/10.3390/app10134568
APA StyleAbdo, H. S., Seikh, A. H., Mohammed, J. A., Luqman, M., Ragab, S. A., & Almotairy, S. M. (2020). Influence of Chloride Ions on Electrochemical Corrosion Behavior of Dual-Phase Steel over Conventional Rebar in Pore Solution. Applied Sciences, 10(13), 4568. https://doi.org/10.3390/app10134568






