Long-Term Impact of Mild Traumatic Brain Injuries on Multiple Functional Outcomes and Epigenetics: A Pilot Study with College Students
Abstract
Featured Application
Abstract
1. Introduction
2. Materials and Methods
- (1)
- King–Devick Oculomotor Test—Testing the eye’s ability to ‘track’ or move accurately and efficiently from one point to the next [48]. The test has three sections, which include numbers with guides, numbers without guides, and numbers without guides and with irregular spacing. The subject was timed while reading the numbers left to right, and errors were monitored until the subject completed reading aloud each slide. Initially derived by an optometrist, the test is becoming an increasingly popular interdisciplinary test among professionals working with individuals with mTBI or concussion [48,49].
- (2)
- Binocular Accommodative Facility with +2.00/−2.00 Flipper Lens—Testing the ability of the eye’s ability to accommodate (or, ‘focus’ and ‘make clear’) when simulating near and far distances using a special set of lenses. The visual system’s ability to dynamically focus at different distances (i.e., with close proximity while reading or at the distance when looking toward a street sign) is a visual skill, which may be impacted in mTBI. Inefficiencies may be demonstrated by perceiving double vision upon adjusting focus through the lens, inability to clear a lens (either minus which simulates near focus, or the plus lens which simulates distance focus), or an increased length of time to clear either or both sides.
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- American Congress of Rehabilitation Medicine Definition of mild traumatic brain injury. J. Head Trauma Rehabil. 1993, 8, 86–87. [CrossRef]
- Taylor, C.A.; Bell, J.M.; Breiding, M.J.; Xu, L. Traumatic brain injury–related emergency department visits, hospitalizations, and deaths — United States, 2007 and 2013. MMWR Surveill. Summ. 2017, 66, 1–16. [Google Scholar] [CrossRef]
- Kay, A.; Teasdale, G. Head Injury in the United Kingdom. World J. Surg. 2001, 25, 1210–1220. [Google Scholar] [CrossRef]
- National Center for Injury Prevention and Control. Report to Congress on Mild Traumatic Brain Injury in the United States: Steps to Prevent a Serious Public Health Problem; National Center for Injury Prevention and Control: Atlanta, GA, USA, 2003.
- Teasdale, G.; Jennett, B. Assessment of coma and impaired consciousness: A practical scale. Lancet 1974, 304, 81–84. [Google Scholar] [CrossRef]
- Seabury, S.A.; Gaudette, É.; Goldman, D.P.; Markowitz, A.J.; Brooks, J.; McCrea, M.A.; Okonkwo, D.O.; Manley, G.T.; Adeoye, O.; Badjatia, N.; et al. Assessment of follow-up care after emergency department presentation for mild traumatic brain injury and concussion. JAMA Netw. Open 2018, 1, e180210. [Google Scholar] [CrossRef] [PubMed]
- Katz, D.I.; White, D.K.; Alexander, M.P.; Klein, R.B. Recovery of ambulation after traumatic brain injury. Arch. Phys. Med. Rehabil. 2004, 85, 865–869. [Google Scholar] [CrossRef]
- McCrea, M.A. Mild Traumatic Brain Injury and Postconcussion Syndrome: The New Evidence Base for Diagnosis and Treatment; Oxford University Press: New York, NY, USA, 2008. [Google Scholar]
- Langlois, J.A.; Rutland-Brown, W.; Wald, M.M. The epidemiology and impact of traumatic brain injury. J. Head Trauma Rehabil. 2006, 21, 375–378. [Google Scholar] [CrossRef]
- Bennett, E.R.; Reuter-Rice, K.; Laskowitz, D.T. Genetic influences in traumatic brain injury. In Translational Research in Traumatic Brain Injury; Laskowitz D, G.G., Ed.; CRC Press/Taylor and Francis Group: Boca Raton, FL, USA, 2016; ISBN 9781466584914. [Google Scholar]
- Centers for Disease Control and Prevention CDC grand rounds: Reducing severe traumatic brain injury in the United States. Morb. Mortal. Wkly. Rep. 2013, 62, 549–552.
- Sharp, D.J.; Jenkins, P.O. Concussion is confusing us all. Pract. Neurol. 2015, 15, 172–186. [Google Scholar] [CrossRef]
- Lee, H.; Lee, S.; Salado, L.; Estrada, J.; Danna-dos-Santos, A.; Isla, K.; Lao, J.; Mohapatra, S.; Bernick, C. Long-term impact of mild traumatic brain injuries on cognitive, psychosocial and balance performance and epigenetics. In Proceedings of the 2018 Asian American/Pacific Islander Nurses Association Conference: Local to Global--Future Directions for Research on Health Disparities, Durham, NC, USA, 22–23 September 2018. [Google Scholar]
- Gill, J.; Lee, H.; Barr, T.; Baxter, T.; Heinzelmann, M.; Rak, H.; Mysliwiec, V. Lower health related quality of life in U.S. military personnel is associated with service-related disorders and inflammation. Psychiatry Res. 2014, 216, 116–122. [Google Scholar] [CrossRef]
- Luethcke, C.A.; Bryan, C.J.; Morrow, C.E.; Isler, W.C. Comparison of concussive symptoms, cognitive performance, and psychological symptoms between acute blast-versus nonblast-induced mild traumatic brain injury. J. Int. Neuropsychol. Soc. 2011, 17, 36–45. [Google Scholar] [CrossRef] [PubMed]
- Giza, C.C.; Hovda, D.A. The new neurometabolic cascade of concussion. Neurosurgery 2014, 75, S24–S33. [Google Scholar] [CrossRef] [PubMed]
- McInnes, K.; Friesen, C.L.; MacKenzie, D.E.; Westwood, D.A.; Boe, S.G. Mild traumatic brain injury (mTBI) and chronic cognitive impairment: A scoping review. PLoS ONE 2017, 12, e0174847. [Google Scholar] [CrossRef] [PubMed]
- Tremblay, S.; de Beaumont, L.; Lassonde, M.; Théoret, H. Evidence for the specificity of intracortical inhibitory dysfunction in asymptomatic concussed athletes. J. Neurotrauma 2011, 28, 493–502. [Google Scholar] [CrossRef]
- Ciuffreda, K.J.; Kapoor, N.; Rutner, D.; Suchoff, I.B.; Han, M.E.; Craig, S. Occurrence of oculomotor dysfunctions in acquired brain injury: A retrospective analysis. Optom. J. Am. Optom. Assoc. 2007, 78, 155–161. [Google Scholar] [CrossRef]
- Thiagarajan, P.; Ciuffreda, K.J.; Ludlam, D.P. Vergence dysfunction in mild traumatic brain injury (mTBI): A review. Ophthalmic Physiol. Opt. 2011, 31, 456–468. [Google Scholar] [CrossRef]
- Valovich McLeod, T.C.; Hale, T.D. Vestibular and balance issues following sport-related concussion. Brain Inj. 2015, 29, 175–184. [Google Scholar] [CrossRef]
- Lee, H.; Merchant-Borna, K.; Wang, D.; Bogner, V.; van Griensven, M.; Gill, J.; Bazarian, J.J. Genome-wide changes in peripheral gene expression following sports-related concussion. J. Neurotrauma 2016, 33, 1576–1585. [Google Scholar]
- Jung, M.; Pfeifer, G.P. Aging and DNA methylation. BMC Biol. 2015, 13, 7. [Google Scholar] [CrossRef]
- Barter, J.D.; Foster, T.C. Aging in the brain: New roles of epigenetics in cognitive decline. Neuroscience 2018, 24, 516–525. [Google Scholar] [CrossRef]
- Chouliaras, L.; Pishva, E.; Haapakoski, R.; Zsoldos, E.; Mahmood, A.; Filippini, N.; Burrage, J.; Mill, J.; Kivimäki, M.; Lunnon, K.; et al. Peripheral DNA methylation, cognitive decline and brain aging: Pilot findings from the Whitehall II imaging study. Epigenomics 2018, 10, 585–595. [Google Scholar] [CrossRef] [PubMed]
- Villela, D.; Ramalho, R.F.; Silva, A.R.T.; Brentani, H.; Suemoto, C.K.; Pasqualucci, C.A.; Grinberg, L.T.; Krepischi, A.C.V.; Rosenberg, C. Differential DNA methylation of microRNA genes in temporal cortex from Alzheimer’s disease individuals. Neural Plast. 2016, 2016. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Mut, J.V.; Heyn, H.; Vidal, E.; Moran, S.; Sayols, S.; Delgado-Morales, R.; Schultz, M.D.; Ansoleaga, B.; Garcia-Esparcia, P.; Pons-Espinal, M.; et al. Human DNA methylomes of neurodegenerative diseases show common epigenomic patterns. Transl. Psychiatry 2016, 6, e718. [Google Scholar] [CrossRef] [PubMed]
- McCrea, M.; Meier, T.; Huber, D.; Ptito, A.; Bigler, E.; Debert, C.T.; Manley, G.; Menon, D.; Chen, J.-K.; Wall, R.; et al. Role of advanced neuroimaging, fluid biomarkers and genetic testing in the assessment of sport-related concussion: A systematic review. Br. J. Sports Med. 2017, 51, 919–929. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention. Report to Congress on Traumatic Brain Injury in the United States: Epidemiology and Rehabilitation; Centers for Disease Control and Prevention: Atlanta, GA, USA, 2015.
- Berger, R.P. The use of serum biomarkers to predict outcome after traumatic brain injury in adults and children. J. Head Trauma Rehabil. 2006, 21, 315–333. [Google Scholar] [CrossRef]
- Tremblay, S.; Henry, L.C.; Bedetti, C.; Larson-Dupuis, C.; Gagnon, J.F.; Evans, A.C.; Théoret, H.; Lassonde, M.; De Beaumont, L. Diffuse white matter tract abnormalities in clinically normal ageing retired athletes with a history of sports-related concussions. Brain 2014, 137, 2997–3011. [Google Scholar] [CrossRef]
- Zetterberg, H.; Blennow, K. Fluid biomarkers for mild traumatic brain injury and related conditions. Nat. Rev. Neurol. 2016, 12. [Google Scholar] [CrossRef]
- Tator, C.H. Concussions and their consequences: Current diagnosis, management and prevention. CMAJ 2013, 185, 975–979. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention. Traumatic Brain Injury & Concussion: Signs and Symptoms. Available online: https://www.cdc.gov/traumaticbraininjury/symptoms.html (accessed on 27 September 2018).
- Dams-O’Connor, K.; Cuthbert, J.P.; Whyte, J.; Corrigan, J.D.; Faul, M.; Harrison-Felix, C. Traumatic brain injury among older adults at level I and II trauma centers. J. Neurotrauma 2013, 30, 2001–2013. [Google Scholar] [CrossRef] [PubMed]
- McQuistion, K.; Zens, T.; Jung, H.S.; Beems, M.; Leverson, G.; Liepert, A.; Scarborough, J.; Agarwal, S. Insurance status and race affect treatment and outcome of traumatic brain injury. J. Surg. Res. 2016, 205, 261–271. [Google Scholar] [CrossRef]
- Peskind, E.R.; Brody, D.; Cernak, I.; McKee, A.; Ruff, R.L. Military- and sports-related mild traumatic brain injury: Clinical presentation, management, and long-term consequences. J. Clin. Psychiatry 2013, 74, 180–188. [Google Scholar] [CrossRef] [PubMed]
- Iverson, G.L.; Lange, R.T. Mild traumatic brain injury. In The Little Black Book of Neuropsychology; Springer US: Boston, MA, USA, 2011; pp. 697–719. [Google Scholar]
- Glang, A.; Ettel, D.; Todis, B.; Gordon, W.A.; Oswald, J.M.; Vaughn, S.L.; Connors, S.H.; Brown, M. Services and supports for students with traumatic brain injury: Survey of state educational agencies. Exceptionality 2015, 23, 211–224. [Google Scholar] [CrossRef]
- King, N.S.; Crawford, S.; Wenden, F.J.; Moss, N.E.; Wade, D.T. The Rivermead Post Concussion Symptoms Questionnaire: A measure of symptoms commonly experienced after head injury and its reliability. J. Neurol. 1995, 242, 587–592. [Google Scholar] [CrossRef] [PubMed]
- Soble, J.R.; Silva, M.A.; Vanderploeg, R.D.; Curtiss, G.; Belanger, H.G.; Donnell, A.J.; Scott, S.G. Normative data for the neurobehavioral symptom inventory (NSI) and post-concussion symptom profiles among TBI, PTSD, and nonclinical samples. Clin. Neuropsychol. 2014, 28, 614–632. [Google Scholar] [CrossRef] [PubMed]
- Cicerone, K.D.; Kalmar, K. Persistent postconcussion syndrome: The structure of subjective complaints after mild traumatic brain injury. J. Head Trauma Rehabil. 1995, 10, 1–17. [Google Scholar] [CrossRef]
- Belanger, H.G.; Silva, M.A.; Donnell, A.J.; McKenzie-Hartman, T.; Lamberty, G.J.; Vanderploeg, R.D. Utility of the Neurobehavioral Symptom Inventory as an outcome measure: A VA TBI Model Systems Study. J. Head Trauma Rehabil. 2017, 32, 46–54. [Google Scholar] [CrossRef] [PubMed]
- Weintraub, S.; Dikmen, S.S.; Heaton, R.K.; Tulsky, D.S.; Zelazo, P.D.; Bauer, P.J.; Carlozzi, N.E.; Slotkin, J.; Blitz, D.; Wallner-Allen, K.; et al. Cognition assessment using the NIH Toolbox. Neurology 2013, 80, S54–S64. [Google Scholar] [CrossRef]
- PROMIS Network PROMIS: Dynamic Tools to Measure Health Outcomes from the Patient Perspective. Available online: http://www.nihpromis.org (accessed on 1 July 2019).
- Degani, A.M.; Santos, M.M.; Leonard, C.T.; Rau, T.F.; Patel, S.A.; Mohapatra, S.; Danna-dos-Santos, A. The effects of mild traumatic brain injury on postural control. Brain Injury 2017, 31, 49–56. [Google Scholar] [CrossRef]
- Santos, M. Changes in Postural Sway Behavior across the Life Span. Master’s Thesis, University of Montana, Missoula, MT, USA, 2017. [Google Scholar]
- Galetta, K.M.; Brandes, L.E.; Maki, K.; Dziemianowicz, M.S.; Laudano, E.; Allen, M.; Lawler, K.; Sennett, B.; Wiebe, D.; Devick, S.; et al. The King–Devick test and sports-related concussion: Study of a rapid visual screening tool in a collegiate cohort. J. Neurol. Sci. 2011, 309, 34–39. [Google Scholar] [CrossRef]
- Ventura, R.; Balcer, L.; Galetta, S. The Concussion Toolbox: The role of vision in the assessment of concussion. Semin. Neurol. 2015, 35, 599–606. [Google Scholar] [CrossRef]
- Dikmen, S.; Machamer, J.; Temkin, N. Mild traumatic brain injury: Longitudinal study of cognition, functional status, and post-traumatic symptoms. J. Neurotrauma 2017, 34, 1524–1530. [Google Scholar] [CrossRef]
- Wammes, J.D.; Good, T.J.; Fernandes, M.A. Autobiographical and episodic memory deficits in mild traumatic brain injury. Brain Cogn. 2017, 111, 112–126. [Google Scholar] [CrossRef] [PubMed]
- Bernick, C.; Banks, S.J.; Shin, W.; Obuchowski, N.; Butler, S.; Noback, M.; Phillips, M.; Lowe, M.; Jones, S.; Modic, M. Repeated head trauma is associated with smaller thalamic volumes and slower processing speed: The professional fighters’ brain health study. Br. J. Sports Med. 2015, 49, 1007–1011. [Google Scholar] [CrossRef] [PubMed]
- McCarthy, M.T.; Kosofsky, B.E. Clinical features and biomarkers of concussion and mild traumatic brain injury in pediatric patients. Ann. N. Y. Acad. Sci. 2015, 1345, 89–98. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, K.A.; Edmed, S.L.; Allan, A.C.; Karlsson, L.J.E.; Smith, S.S. Characterizing self-reported sleep disturbance after mild traumatic brain injury. J. Neurotrauma 2015, 32, 474–486. [Google Scholar] [CrossRef]
- Childers, C.; Hux, K. Invisible injuries: The experiences of college students with histories of mild traumatic brain injury. J. Postsecond. Educ. Disabil. 2016, 29, 389–405. [Google Scholar]
- Covassin, T.; Elbin, R.J.; Beidler, E.; LaFevor, M.; Kontos, A.P. A review of psychological issues that may be associated with a sport-related concussion in youth and collegiate athletes. Sport. Exerc. Perform. Psychol. 2017, 6, 220–229. [Google Scholar] [CrossRef]
- Thastum, M.M.; Rask, C.U.; Naess-Schmidt, E.T.; Jensen, J.S.; Frederiksen, O.V.; Tuborgh, A.; Svendsen, S.W.; Nielsen, J.F.; Schröder, A. Design of an early intervention for persistent post-concussion symptoms in adolescents and young adults: A feasibility study. NeuroRehabilitation 2018, 43, 155–167. [Google Scholar] [CrossRef] [PubMed]
- Hux, K.; Bush, E.; Zickefoose, S.; Holmberg, M.; Henderson, A.; Simanek, G. Exploring the study skills and accommodations used by college student survivors of traumatic brain injury. Brain Inj. 2010, 24, 13–26. [Google Scholar] [CrossRef] [PubMed]
- Carulli, L.A.; Olney, M.F.; Degeneffe, C.E.; Conrad, G. Social engagement among community college students with traumatic brain injury. J. Rehabil. 2018, 84, 29–38. [Google Scholar]
- Kennedy, M.R.T.; Krause, M.O.; Turkstra, L.S. An electronic survey about college experiences after traumatic brain injury. NeuroRehabilitation 2008, 23, 511–520. [Google Scholar] [CrossRef] [PubMed]
- Bryson, C.N.; Cramer, R.J.; Schmidt, A.T. Traumatic brain injury and lifetime suicidality: Applying the interpersonal-psychological theory perspective. Death Stud. 2017, 41, 399–405. [Google Scholar] [CrossRef] [PubMed]
- Shaw, N.A. The neurophysiology of concussion. Prog. Neurobiol. 2002, 67, 281–344. [Google Scholar] [CrossRef]
- Guskiewicz, K.M. Balance assessment in the management of sport-related concussion. Clin. Sports Med. 2011, 30, 89–102. [Google Scholar] [CrossRef] [PubMed]
- Ingersoll, C.D.; Armstrong, C.W. The effects of closed-head injury on postural sway. Med. Sci. Sports Exerc. 1992, 24, 739–743. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Lee, S.; Salado, L.; Estrada, J.; White, J.; Muthukumar, V.; Lee, S.-P.; Mohapatra, S. Proof-of-concept testing of a real-time mHealth measure to estimate postural control during walking: A potential application for mild traumatic brain injuries. Asian / Pac. Isl. Nurs. J. 2019, 3. [Google Scholar] [CrossRef][Green Version]
- U.S. Food and Drug Administration. Product Classification: Brain Injury Adjunctive Interpretive Oculomotor Assessment Aid. Available online: https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfpcd/classification.cfm?id=3820 (accessed on 10 August 2019).
- Voelker, R. Eye-Tracking Test Approved to Help Diagnose Concussion. JAMA 2019, 321, 638. [Google Scholar] [CrossRef]
- Thompson, C.; Davies, P.; Herrmann, L.; Summers, M.; Potter, S. Approaches to establishing validated cut-off scores on the Rivermead post-concussion symptoms questionnaire (RPQ). In Proceedings of the International Brain Injury Association’s Eleventh World Congress on Brain Injury; Taylor & Francis: Hague, The Netherlands, 2016. [Google Scholar]
- Vos, L.; Whiteneck, G.G.; Ngan, E.; Leon Novelo, L.; Harik, L.M.; Sherer, M. Comparison of the Neurobehavioral Symptom Inventory and the Rivermead Postconcussion Symptoms Questionnaire. Brain Injury 2019, 33, 1165–1172. [Google Scholar] [CrossRef]
- Montenigro, P.H.; Alosco, M.L.; Martin, B.M.; Daneshvar, D.H.; Mez, J.; Chaisson, C.E.; Nowinski, C.J.; Au, R.; McKee, A.C.; Cantu, R.C.; et al. Cumulative head impact exposure predicts later-life depression, apathy, executive dysfunction, and cognitive impairment in former high school and college football players. J. Neurotrauma 2017, 34, 328–340. [Google Scholar] [CrossRef]
- Tsankova, N.; Renthal, W.; Kumar, A.; Nestler, E.J. Epigenetic regulation in psychiatric disorders. Nat. Rev. Neurosci. 2007, 8, 355–367. [Google Scholar] [CrossRef]
- Lee, H.; Gill, J.; Barr, T.; Yun, S.; Kim, H. Primer in genetics and genomics, article 2—Advancing nursing research with genomic approaches. Biol. Res. Nurs. 2017, 19, 229–239. [Google Scholar] [CrossRef] [PubMed]
- Wen, K.; Miliç, J.; El-Khodor, B.; Dhana, K.; Nano, J.; Pulido, T.; Kraja, B.; Zaciragic, A.; Bramer, W.M.; Troup, J.; et al. The role of DNA methylation and histone modifications in neurodegenerative diseases: A systematic review. PLoS ONE 2016, 11, e0167201. [Google Scholar] [CrossRef] [PubMed]
- Bellizzi, D.; D’Aquila, P.; Montesanto, A.; Corsonello, A.; Mari, V.; Mazzei, B.; Lattanzio, F.; Passarino, G. Global DNA methylation in old subjects is correlated with frailty. Age (Omaha) 2012, 34, 169–179. [Google Scholar] [CrossRef] [PubMed]
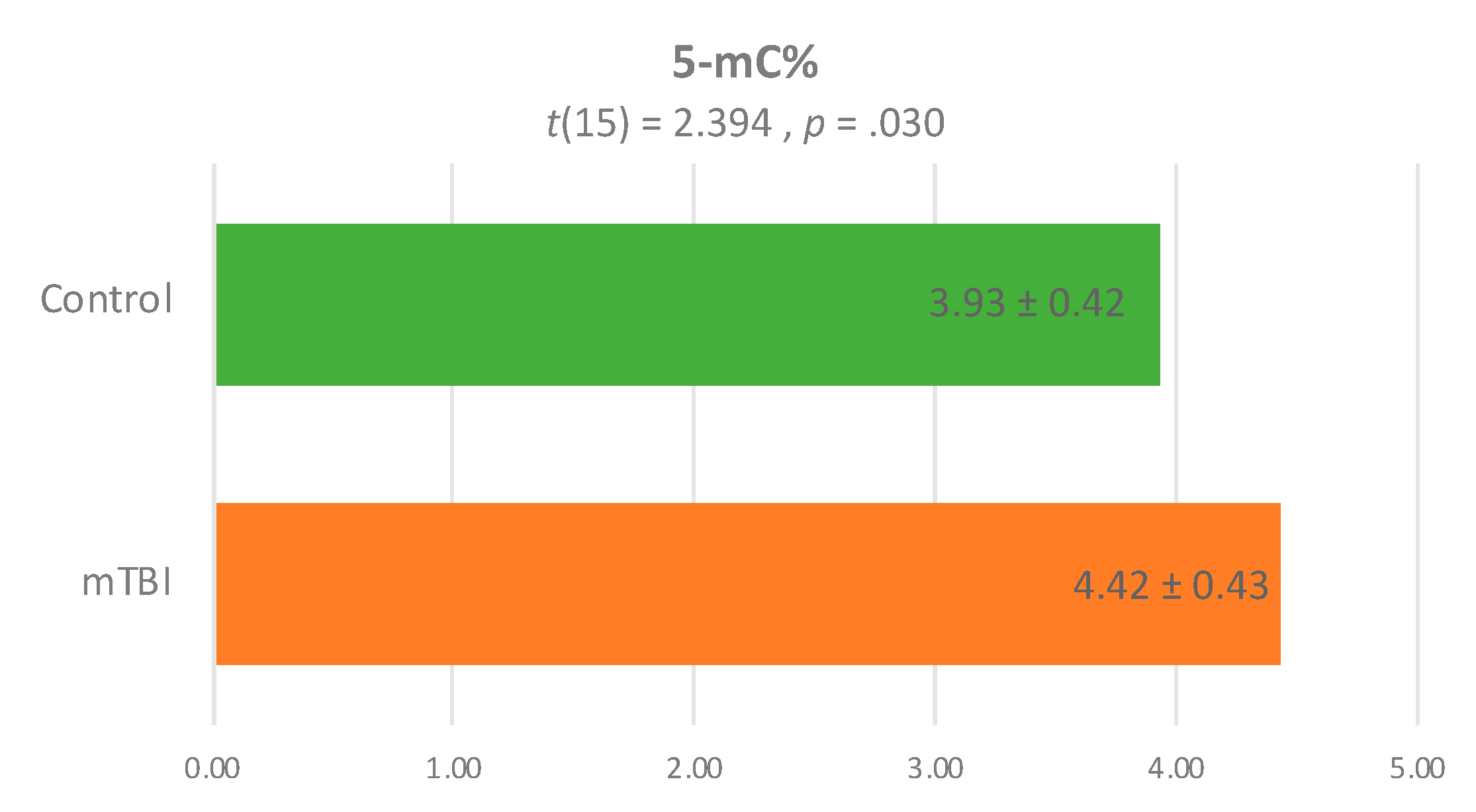
| Characteristic | Overall (n = 25) | mTBI (n = 11) | Control (n = 14) | x2 or t | p |
|---|---|---|---|---|---|
| Age | 24.96 ± 5.96 | 28.73 ± 5.92 | 22.00 ± 4.13 | 3.348 | 0.003 |
| Gender | |||||
| Males | 13 (52) | 8 (72.7) | 5 (35.7) | 3.381 | 0.066 |
| Females | 12 (48) | 3 (27.3) | 9 (64.3) | ||
| Weight (kg) | 70.85 ± 16.77 | 75.67 ± 14.97 | 67.07 ± 17.66 | 1.129 | 0.210 |
| Height (cm) | 169.06 ± 11.81 | 173.87 ± 10.62 | 165.28 ± 11.64 | 1.903 | 0.070 |
| Ethnicity/Race | |||||
| Hispanic | 4 (16.0) | 2 (18.2) | 2 (14.3) | 0.263 | 0.992 |
| White | 14 (56.0) | 6 (54.5) | 8 (57.1) | ||
| Black | 2 (8.0) | 1 (9.1) | 1 (7.1) | ||
| Asian | 3 (12.0) | 1 (9.1) | 2 (14.3) | ||
| Other | 2 (8.0) | 1 (9.1) | 1 (7.1) | ||
| Handedness | |||||
| Right | 22 (88.0) | 9 (81.8) | 13 (92.9) | 0.711 | 0.399 |
| Left | 3 (12.0) | 2 (18.2) | 1 (7.1) | ||
| Education | |||||
| In college | 22 (88.0) | 10 (90.9) | 12 (85.7) | 0.834 | 0.659 |
| In graduate school | 3 (12.0) | 1 (9.1) | 2 (14.3) | ||
| Marital Status * | |||||
| Single | 21 (84.0) | 9 (81.8) | 12 (92.6) | 1.376 | 0.503 |
| Married | 3 (12.0) | 2 (18.2) | 1 (7.7) | ||
| Employment Status | |||||
| Yes | 16 (64.0) | 5 (45.5) | 10 (71.4) | 0.762 | 0.383 |
| No | 9 (36.0) | 6 (54.5) | 4 (28.6) | ||
| Income * | |||||
| Low (≤ $50K) | 13 (52.0) | 6 (54.2) | 7 (58.3) | 1.703 | 0.427 |
| Middle ($50K < ≤ $100K) | 6 (24.0) | 2 (18.2) | 4 (33.3) | ||
| High ($100K <) | 4 (16.0) | 3 (27.3) | 1 (8.3) |
| Functional Variables | Overall (n = 25) | mTBI (n = 11) | Control (n = 14) | t | p |
|---|---|---|---|---|---|
| Cognitive Function by NIH Toolbox (age corrected): | |||||
| Dimension Change Card Sort Test (DCCS) | 98.17 ± 18.83 | 97.20 ± 15.85 | 98.86 ± 21.26 | −0.208 | 0.837 |
| Flanker Inhibitory Control and Attention Test | 83.67 ± 14.64 | 82.10 ± 15.99 | 84.79 ± 14.11 | −0.435 | 0.668 |
| Picture Sequence Memory Test | 108.67 ± 17.99 | 99.00 ± 17.19 | 115.57 ± 15.64 | −2.457 | 0.022 |
| List Sorting Working Memory Test | 103.92 ± 12.25 | 103.40 ± 11.94 | 104.29 ± 12.90 | −0.171 | 0.866 |
| Picture Vocabulary Test | 110.08 ± 10.97 | 113.60 ± 7.59 | 107.57 ± 12.51 | 1.3511 | 0.190 |
| Pattern Comparison Processing Speed Test | 102.75 ± 22.52 | 107.70 ± 20.30 | 99.21 ± 24.08 | 0.906 | 0.375 |
| Fluid Cognition Composite Score | 99.08 ± 15.73 | 96.70 ± 18.89 | 100.79 ± 13.54 | −0.619 | 0.542 |
| Early Childhood Composite Score | 99.96 ± 14.25 | 96.80 ± 15.33 | 102.21 ± 13.53 | −0.915 | 0.370 |
| Psychological Function by PROMIS: | |||||
| Depression | 50.73 ± 7.26 | 54.04 ± 7.57 | 48.37 ± 6.26 | 2.005 | 0.057 |
| Anxiety | 52.15 ± 8.15 | 56.55 ± 9.77 | 49.00 ± 5.09 | 2.472 | 0.022 |
| Sleep Disturbance | 50.24 ± 11.4 | 56.34 ± 11.82 | 45.89 ± 9.17 | 2.637 | 0.015 |
| Visual Function by | |||||
| King–Devick Test: | |||||
| Number of misses | 0.04 ± 0.20 | 0.00 ± 0.00 | 0.07 ± 0.27 | −0.840 | 0.410 |
| Total time (s) | 50.73 ± 22.11 | 57.66 ± 31.68 | 45.29 ± 7.64 | 1.417 | 0.170 |
| Accommodation Test: | |||||
| Number of tests per min | 10.96 ± 3.55 | 10.59 ± 3.00 | 11.25 ± 4.01 | −0.453 | 0.654 |
| Body Balance Performance by a force platform: | |||||
| Area (cm2) | 1.96 ± 1.73 | 2.23 ± 2.16 | 1.76 ± 1.39 | 0.652 | 0.521 |
| AmplitudeAP (cm2) | 1.71 ± 1.12 | 2.09 ± 1.41 | 1.44 ± 0.81 | 1.428 | 0.167 |
| AmplitudeML (cm2) | 2.62 ± 1.09 | 2.32 ± 0.77 | 2.84 ± 1.24 | −1.149 | 0.263 |
| MeanVelocityAP (cm s−1) | 3.10 ± 1.34 | 3.49 ± 1.55 | 2.82 ± 1.17 | 1.222 | 0.068 |
| MeanVelocityML (cm s−1) | 5.31 ± 1.72 | 5.16 ± 2.10 | 5.41 ± 1.47 | −0.339 | 0.738 |
| F80AP (Hz) | 0.45 ± 0.43 | 0.52 ± 0.64 | 0.40 ± 0.19 | 0.709 | 0.486 |
| F80ML (Hz) | 0.26 ± 0.10 | 0.27 ± 0.08 | 0.25 ± 0.11 | 0.437 | 0.666 |
| SEntAP | 0.56 ± 0.19 | 0.53 ± 0.14 | 0.59 ± 0.22 | −0.724 | 0.477 |
| SEntML | 0.52 ± 0.13 | 0.54 ± 0.12 | 0.51 ± 0.14 | 0.575 | 0.571 |
| CrossSEnt | 1.86 ± 1.24 | 1.56 ± 1.06 | 2.07 ± 1.35 | −0.993 | 0.332 |
| Overall post-mTBI symptoms by | |||||
| RPQ Total score | 11.71 ± 14.28 | 24.40 ± 13.83 | 2.64 ± 3.95 | 5.617 | 0.000 |
| NSI Total score | 13.57 ± 14.43 | 24.80 ± 15.02 | 4.92 ± 5.16 | 4.469 | 0.000 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, H.; Lee, S.; Black, I.; Salado, L.; Estrada, J.; Isla, K. Long-Term Impact of Mild Traumatic Brain Injuries on Multiple Functional Outcomes and Epigenetics: A Pilot Study with College Students. Appl. Sci. 2020, 10, 4131. https://doi.org/10.3390/app10124131
Lee H, Lee S, Black I, Salado L, Estrada J, Isla K. Long-Term Impact of Mild Traumatic Brain Injuries on Multiple Functional Outcomes and Epigenetics: A Pilot Study with College Students. Applied Sciences. 2020; 10(12):4131. https://doi.org/10.3390/app10124131
Chicago/Turabian StyleLee, Hyunhwa, Sungchul Lee, Ipuna Black, Laura Salado, Jonica Estrada, and Katrina Isla. 2020. "Long-Term Impact of Mild Traumatic Brain Injuries on Multiple Functional Outcomes and Epigenetics: A Pilot Study with College Students" Applied Sciences 10, no. 12: 4131. https://doi.org/10.3390/app10124131
APA StyleLee, H., Lee, S., Black, I., Salado, L., Estrada, J., & Isla, K. (2020). Long-Term Impact of Mild Traumatic Brain Injuries on Multiple Functional Outcomes and Epigenetics: A Pilot Study with College Students. Applied Sciences, 10(12), 4131. https://doi.org/10.3390/app10124131





