Evaluation of Microbial Load, Formation of Odorous Metabolites and Lipid Stability during Wet Preservation of Nannochloropsis gaditana Concentrates
Abstract
1. Introduction
2. Materials and Methods
2.1. Algae Growth and Harvesting
2.2. Algae Storage
2.3. Gas Analysis
2.4. Short Chain Fatty Acid Analysis
2.5. Microbial Analysis
2.6. Lipid Content
2.7. Free Fatty Acid Level
2.8. Chlorophyll Content
2.9. Statistics
3. Results
3.1. Storage Experiment 1—Microbiological and Metabolic Analysis
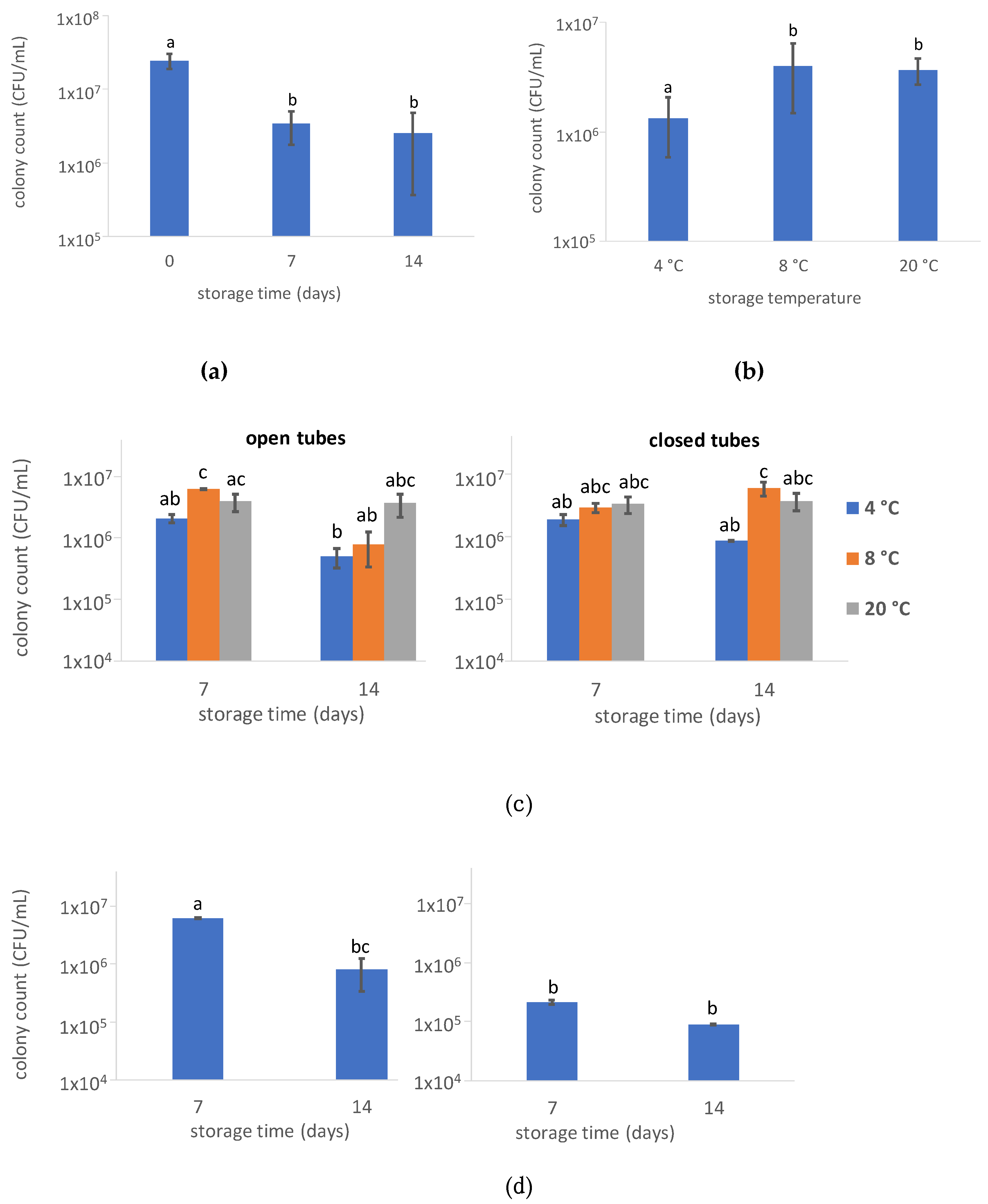
3.1.1. Total Microbial Count
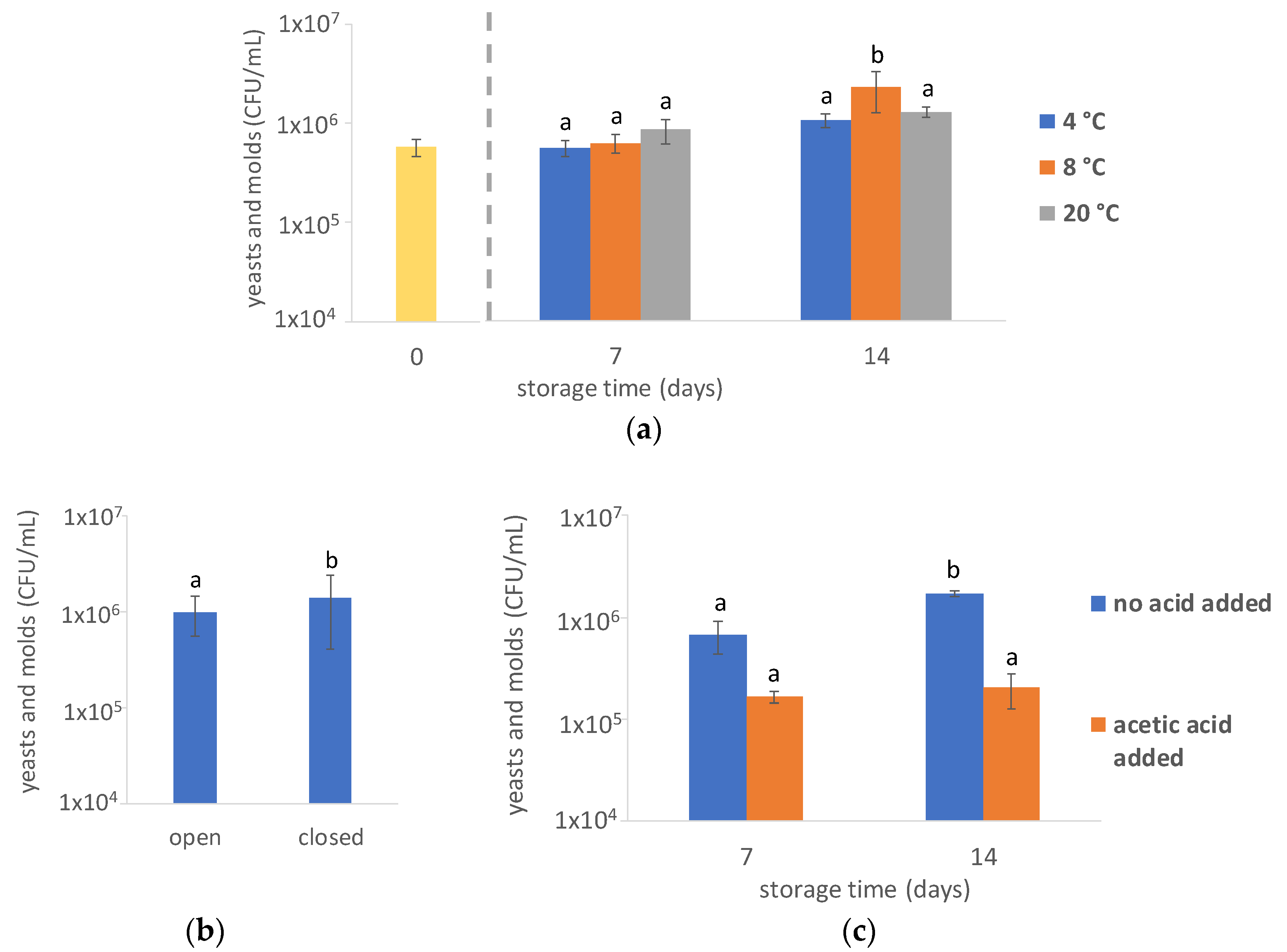
3.1.2. Yeasts and Molds
3.1.3. Headspace Analysis in Closed Storage Tubes
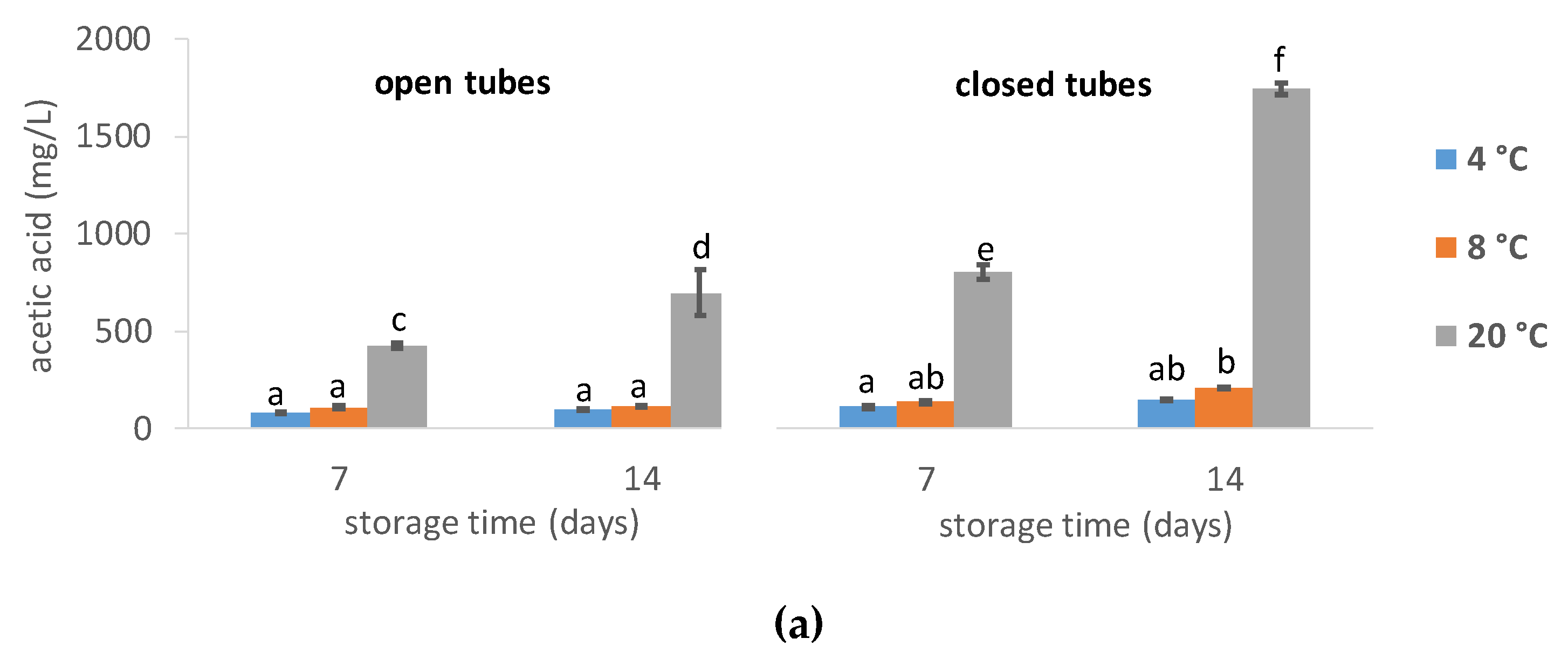
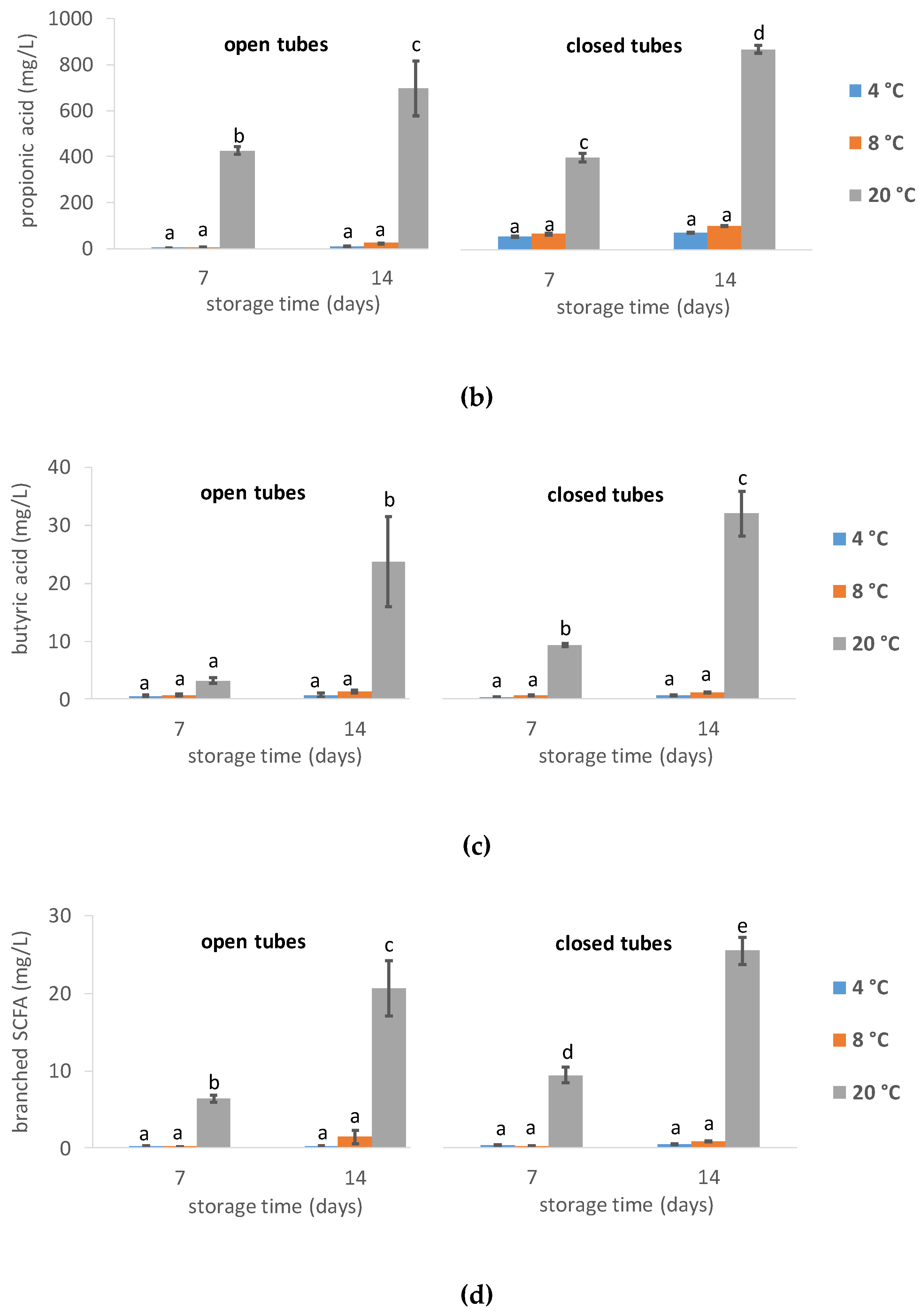
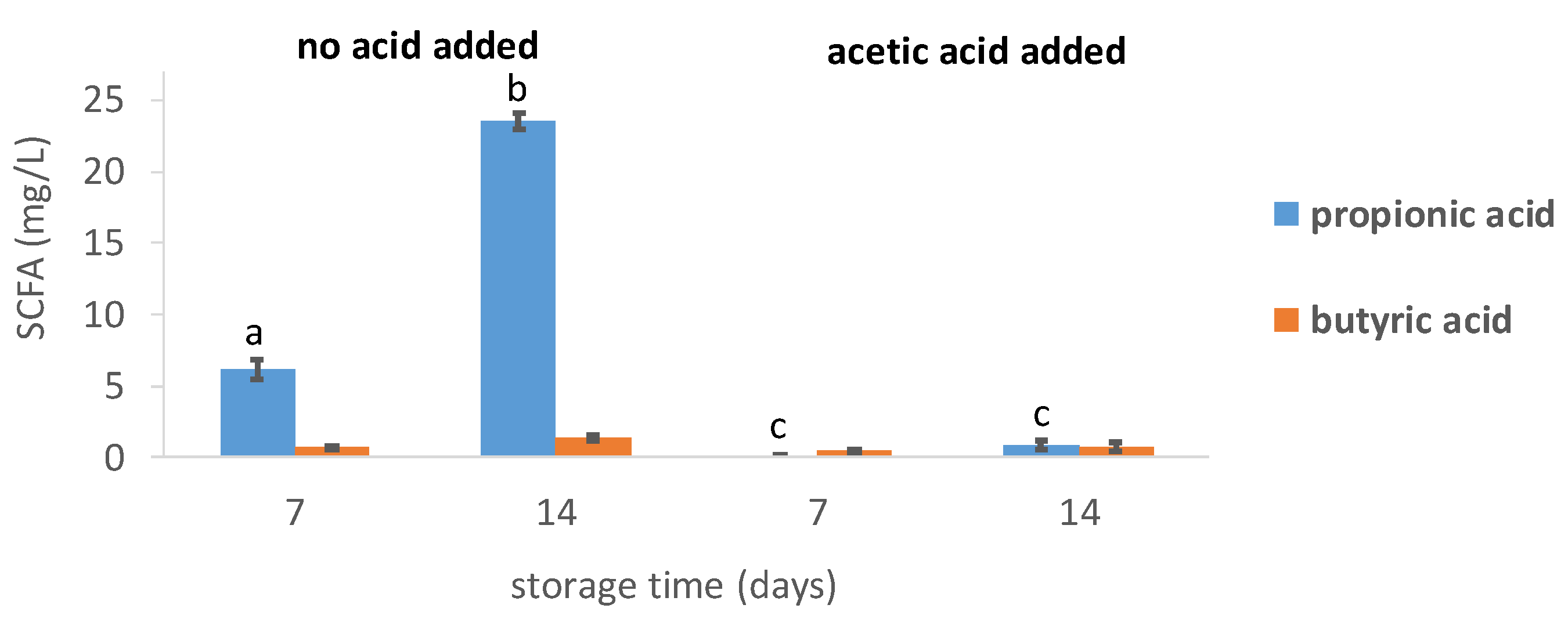
3.1.4. Short-Chain Fatty Acids Levels
3.2. Storage Experiment 2—Lipid and Chlorophyll Analysis
4. Discussion
4.1. Changing O2 Levels Set the Scene
4.2. Effect of Storage Temperature
4.3. Open Tube vs. Closed Tube Storage
4.4. Acetic Acid Addition
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ma, X.N.; Chen, T.P.; Yang, B.; Liu, J.; Chen, F. Lipid production from Nannochloropsis. Mar. Drugs 2016, 14, 61. [Google Scholar] [CrossRef]
- Ma, Y.; Wang, Z.; Yu, C.; Yin, Y.; Zhou, G. Evaluation of the potential of 9 Nannochloropsis strains for biodiesel production. Bioresour. Technol. 2014, 167, 503–509. [Google Scholar] [CrossRef]
- Maffei, G.; Bracciale, M.P.; Broggi, A.; Zuorro, A.; Santarelli, M.L.; Lavecchia, R. Effect of an enzymatic treatment with cellulase and mannanase on the structural properties of Nannochloropsis microalgae. Bioresour. Technol. 2018, 249, 592–598. [Google Scholar] [CrossRef] [PubMed]
- Hulatt, C.J.; Wijffels, R.H.; Bolla, S.; Kiron, V. Production of fatty acids and protein by nannochloropsis in flat-plate photobioreactors. PLoS ONE 2017, 12, e0170440. [Google Scholar] [CrossRef] [PubMed]
- Harris, W.S.; Kris-Etherton, P.M.; Harris, K.A. Intakes of long-chain omega-3 fatty acid associated with reduced risk for death from coronary heart disease in healthy adults. Curr. Atheroscler. Rep. 2008, 10, 503–509. [Google Scholar] [CrossRef] [PubMed]
- Markou, G.; Wang, L.; Ye, J.; Unc, A. Using agro-industrial wastes for the cultivation of microalgae and duckweeds: Contamination risks and biomass safety concerns. Biotechnol. Adv. 2018, 36, 1238–1254. [Google Scholar] [CrossRef]
- IPCC. Special Report on Climate Change, Desertification, Land Degradation, Sustainable Land Management, Food Security, and Greenhouse Gas Fluxes in Terrestrial Ecosystems; IPCC: Geneva, Switzerland, 2019. [Google Scholar]
- Chua, E.T.; Schenk, P.M. A biorefinery for Nannochloropsis: Induction, harvesting, and extraction of EPA-rich oil and high-value protein. Bioresour. Technol. 2017, 244, 1416–1424. [Google Scholar] [CrossRef]
- Mathimani, T.; Pugazhendhi, A. Utilization of algae for biofuel, bio-products and bio-remediation. Biocatal. Agric. Biotechnol. 2019, 17, 326–330. [Google Scholar] [CrossRef]
- Zhang, R.; Parniakov, O.; Grimi, N.; Lebovka, N.; Marchal, L.; Vorobiev, E. Emerging techniques for cell disruption and extraction of valuable bio-molecules of microalgae Nannochloropsis sp. Bioprocess Biosyst. Eng. 2019, 42, 173–186. [Google Scholar] [CrossRef]
- Soomro, R.R.; Ndikubwimana, T.; Zeng, X.; Lu, Y.; Lin, L.; Danquah, M.K. Development of a two-stage microalgae dewatering process—A life cycle assessment approach. Front. Plant Sci. 2016, 7, 1–12. [Google Scholar] [CrossRef]
- Milledge, J.J.; Heaven, S. A review of the harvesting of micro-algae for biofuel production. Rev. Environ. Sci. Biotechnol. 2013, 12, 165–178. [Google Scholar] [CrossRef]
- Ryckebosch, E.; Muylaert, K.; Eeckhout, M.; Ruyssen, T.; Foubert, I. Influence of drying and storage on lipid and carotenoid stability of the microalga Phaeodactylum tricornutum. J. Agric. Food Chem. 2011, 59, 11063–11069. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, F.; Li, Y.; Fanning, K.; Netzel, M.; Schenk, P.M. Effect of drying, storage temperature and air exposure on astaxanthin stability from Haematococcus pluvialis. Food Res. Int. 2015, 74, 231–236. [Google Scholar] [CrossRef] [PubMed]
- Ge, H.; Li, J.; Chang, Z.; Chen, P.; Shen, M.; Zhao, F. Effect of microalgae with semicontinuous harvesting on water quality and zootechnical performance of white shrimp reared in the zero water exchange system. Aquac. Eng. 2016, 72–73, 70–76. [Google Scholar] [CrossRef]
- Spolaore, P.; Joannis-Cassan, C.; Duran, E.; Isambert, A. Commercial applications of microalgae. J. Biosci. Bioeng. 2006, 101, 87–96. [Google Scholar] [CrossRef]
- Balduyck, L.; Stock, T.; Bijttebier, S.; Bruneel, C.; Jacobs, G.; Voorspoels, S.; Muylaert, K.; Foubert, I. Integrity of the microalgal cell plays a major role in the lipolytic stability during wet storage. Algal Res. 2017, 25, 516–524. [Google Scholar] [CrossRef]
- Sales, R.; Mélo, R.C.S.; de Moraes, R.M.; da Silva, R.C.S.; Cavalli, R.O.; do Amaral Ferraz Navarro, D.M.; de Souza Santos, L.P. Production and use of a flocculated paste of Nannochloropsis oculata for rearing newborn seahorse Hippocampus reidi. Algal Res. 2016, 17, 142–149. [Google Scholar] [CrossRef]
- Welladsen, H.; Kent, M.; Mangott, A.; Li, Y. Shelf-life assessment of microalgae concentrates: Effect of cold preservation on microalgal nutrition profiles. Aquaculture 2014, 430, 241–247. [Google Scholar] [CrossRef]
- Napan, K.; Christianson, T.; Voie, K.; Quinn, J.C. Quantitative assessment of microalgae biomass and lipid stability post-cultivation. Front. Energy Res. 2015, 3, 1–6. [Google Scholar] [CrossRef]
- Camacho-Rodríguez, J.; Cerón-García, M.C.; Macías-Sánchez, M.D.; Fernández-Sevilla, J.M.; López-Rosales, L.; Molina-Grima, E. Long-term preservation of concentrated Nannochloropsis gaditana cultures for use in aquaculture. J. Appl. Phycol. 2016, 28, 299–312. [Google Scholar] [CrossRef]
- Refsgaard, H.H.F.; Brockhoff, P.M.B.; Jensen, B. Free polyunsaturated fatty acids cause taste deterioration of salmon during frozen storage. J. Agric. Food Chem. 2000, 48, 3280–3285. [Google Scholar] [CrossRef] [PubMed]
- Ganesan, B.; Brothersen, C.; McMahon, D.J. Fortification of foods with omega-3 polyunsaturated fatty acids. Crit. Rev. Food Sci. Nutr. 2014, 54, 98–114. [Google Scholar] [CrossRef] [PubMed]
- Gheysen, L.; Bernaerts, T.; Bruneel, C.; Goiris, K.; Van Durme, J.; Van Loey, A.; De Cooman, L.; Foubert, I. Impact of processing on n-3 LC-PUFA in model systems enriched with microalgae. Food Chem. 2018, 268, 441–450. [Google Scholar] [CrossRef] [PubMed]
- Van Durme, J.; Goiris, K.; De Winne, A.; De Cooman, L.; Muylaert, K. Evaluation of the volatile composition and sensory properties of five species of microalgae. J. Agric. Food Chem. 2013, 61, 10881–10890. [Google Scholar] [CrossRef] [PubMed]
- Wendt, L.M.; Kinchin, C.; Wahlen, B.D.; Davis, R.; Dempster, T.A.; Gerken, H. Assessing the stability and techno-economic implications for wet storage of harvested microalgae to manage seasonal variability. Biotechnol. Biofuels 2019, 12, 80. [Google Scholar] [CrossRef]
- Belitz, H.-D.; Grosch, W.; Schiberle, P. Food Chemistry, 3rd ed.; Springer: Berlin/Heidelberg, Germany, 2009; ISBN 978-3-540-69933-0. [Google Scholar]
- Ponis, E.; Parisi, G.; Chini Zittelli, G.; Lavista, F.; Robert, R.; Tredici, M.R. Pavlova lutheri: Production, preservation and use as food for Crassostrea gigas larvae. Aquaculture 2008, 282, 97–103. [Google Scholar] [CrossRef]
- ISO/TC 34/SC 9 Microbiology ISO 4833-1:2013—Microbiology of the Food Chain—Horizontal Method for the Enumeration of Microorganisms—Part 1: Colony Count at 30 Degrees C by the Pour Plate Technique. Available online: https://www.iso.org/standard/53728.html (accessed on 1 September 2019).
- Ryckebosch, E.; Muylaert, K.; Foubert, I. Optimization of an analytical procedure for extraction of lipids from microalgae. J. Am. Oil. Chem. Soc. 2012, 89, 189–198. [Google Scholar] [CrossRef]
- Kangani, C.O.; Kelley, D.E.; De Lany, J.P. New method for GC/FID and GC–C-IRMS analysis of plasma free fatty acid concentration and isotopic enrichment. J. Chromatogr. B 2008, 873, 95–101. [Google Scholar] [CrossRef]
- Lichtenthaler, H.K.; Buschmann, C. Chlorophylls. In Handbook of Food Analytical Chemistry; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2005; pp. 153–199. [Google Scholar]
- Ritchie, R.J. Consistent sets of spectrophotometric chlorophyll equations for acetone, methanol and ethanol solvents. Photosynth. Res. 2006, 89, 27–41. [Google Scholar] [CrossRef]
- Halim, R.; Hill, D.R.A.; Hanssen, E.; Webley, P.A.; Blackburn, S.; Grossman, A.R.; Posten, C.; Martin, G.J.O. Towards sustainable microalgal biomass processing: Anaerobic induction of autolytic cell-wall self-ingestion in lipid-rich Nannochloropsis slurries. Green Chem. 2019, 21, 2967–2982. [Google Scholar] [CrossRef]
- Madigan, M.T.; Martinko, J.M. Brock Biology of Microorganisms, 11th ed.; Pearson Prentice Hall: Upper Saddle River, NJ, USA, 2006; ISBN 0-13-144329-1. [Google Scholar]
- Catalanotti, C.; Yang, W.; Posewitz, M.C.; Grossman, A.R. Fermentation metabolism and its evolution in algae. Front. Plant Sci. 2013, 4, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Klassen, V.; Blifernez-Klassen, O.; Wibberg, D.; Winkler, A.; Kalinowski, J.; Posten, C.; Kruse, O. Highly efficient methane generation from untreated microalgae biomass. Biotechnol. Biofuels 2017, 10, 186. [Google Scholar] [CrossRef] [PubMed]
- Jinkerson, R.E.; Radakovits, R.; Posewitz, M.C. Genomic insights from the oleaginous model alga Nannochloropsis gaditana. Bioengineered 2013, 4, 37–43. [Google Scholar] [CrossRef] [PubMed]
- Leffingwell, J.C.; Leffingwell, D. GRAS flavor chemicals detection thresholds. Perfum. Flavorist 1991, 16, 1–13. [Google Scholar]
- Ara, K.; Hama, M.; Akiba, S.; Koike, K.; Okisaka, K.; Hagura, T.; Kamiya, T.; Tomita, F. Foot odor due to microbial metabolism and its control. Can. J. Microbiol. 2006, 52, 357–364. [Google Scholar] [CrossRef] [PubMed]
- Smith, E.; Macfarlane, G. Dissimilatory amino acid metabolism in human colonic bacteria. Anaerobe 1997, 3, 327–337. [Google Scholar] [CrossRef]
- Roberts, K.P.; Heaven, S.; Banks, C.J. Comparative testing of energy yields from micro-algal biomass cultures processed via anaerobic digestion. Renew. Energy 2016, 87, 744–753. [Google Scholar] [CrossRef]
- Kadota, H.; Ishida, Y. Production of volatile sulfur compounds by microorganisms. Annu. Rev. Microbiol. 1972, 26, 127–138. [Google Scholar] [CrossRef]
- Achyuthan, K.; Harper, J.; Manginell, R.; Moorman, M. Volatile metabolites emission by In Vivo microalgae—An overlooked opportunity? Metabolites 2017, 7, 39. [Google Scholar] [CrossRef]
- Watson, S.B.; Jüttner, F. Malodorous volatile organic sulfur compounds: Sources, sinks and significance in inland waters. Crit. Rev. Microbiol. 2017, 43, 210–237. [Google Scholar] [CrossRef]
- Ilić, Z.S.; Milenković, L.; Šunić, L.; Fallik, E. Effect of coloured shade-nets on plant leaf parameters and tomato fruit quality. J. Sci. Food Agric. 2015, 95, 2660–2667. [Google Scholar] [CrossRef] [PubMed]
- Wendt, L.M.; Wahlen, B.D.; Li, C.; Kachurin, G.; Ogden, K.L.; Murphy, J.A. Evaluation of a high-moisture stabilization strategy for harvested microalgae blended with herbaceous biomass: Part I—Storage performance. Algal Res. 2017, 25, 567–575. [Google Scholar] [CrossRef]
- Zuo, Z.; Zhu, Y.; Bai, Y.; Wang, Y. Acetic acid-induced programmed cell death and release of volatile organic compounds in Chlamydomonas reinhardtii. Plant Physiol. Biochem. 2012, 51, 175–184. [Google Scholar] [CrossRef]
- Scholz, M.J.; Weiss, T.L.; Jinkerson, R.E.; Jing, J.; Roth, R.; Goodenough, U.; Posewitz, M.C.; Gerken, H.G. Ultrastructure and composition of the Nannochloropsis gaditana cell wall. Eukaryot. Cell 2014, 13, 1450–1464. [Google Scholar] [CrossRef]

| Storage Condition | Storage Time (Days) | Lipid (% dm) | FFA (% Lipid) | Chlorophyll (mg/g dm) |
|---|---|---|---|---|
| t0 | 0 | 17.3 ± 0.2 | 2.3 ± 0.4 | 19.7 ± 0.8 |
| 4 °C, open | 7 | 17.2 ± 0.1 | 2.0 ± 0.2 | 23.2 ± 2.7 |
| 17 | 18.0 ± 0.3 | 1.8 ± 0.1 | 24.5 ± 3.6 | |
| 4 °C, closed | 7 | 17.2 ± 0.1 | 1.9 ± 0.1 | 17.2 ± 2.5 |
| 17 | 16.7 ± 1.0 | 1.9 ± 0.2 | 19.1 ± 2.3 | |
| 8 °C, open | 7 | 18.2 ± 0.2 | 1.5 ± 0.1 | 27.0 ± 2.0 |
| 17 | 19.0 ± 0.3 | 2.1 ± 0.1 | 29.1 ± 1.7 | |
| 8 °C, closed | 7 | 16.6 ± 0.3 | 1.8 ± 0.1 | 20.6 ± 0.3 |
| 17 | 17.0 ± 0.2 | 2.3 ± 0.1 | 19.4 ± 0.6 | |
| 8 °C open + acetic acid | 7 | 18.0 ± 0.4 | 2.9 ± 0.2 | 25.8 ± 1.4 |
| 17 | 17.2 ± 0.1 | 21.6 ± 3.7 | 10.5 ± 0.6 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Verspreet, J.; Kreps, S.; Bastiaens, L. Evaluation of Microbial Load, Formation of Odorous Metabolites and Lipid Stability during Wet Preservation of Nannochloropsis gaditana Concentrates. Appl. Sci. 2020, 10, 3419. https://doi.org/10.3390/app10103419
Verspreet J, Kreps S, Bastiaens L. Evaluation of Microbial Load, Formation of Odorous Metabolites and Lipid Stability during Wet Preservation of Nannochloropsis gaditana Concentrates. Applied Sciences. 2020; 10(10):3419. https://doi.org/10.3390/app10103419
Chicago/Turabian StyleVerspreet, Joran, Sabine Kreps, and Leen Bastiaens. 2020. "Evaluation of Microbial Load, Formation of Odorous Metabolites and Lipid Stability during Wet Preservation of Nannochloropsis gaditana Concentrates" Applied Sciences 10, no. 10: 3419. https://doi.org/10.3390/app10103419
APA StyleVerspreet, J., Kreps, S., & Bastiaens, L. (2020). Evaluation of Microbial Load, Formation of Odorous Metabolites and Lipid Stability during Wet Preservation of Nannochloropsis gaditana Concentrates. Applied Sciences, 10(10), 3419. https://doi.org/10.3390/app10103419





