Evaluating the Effectiveness of Urban Hedges as Air Pollution Barriers: Importance of Sampling Method, Species Characteristics and Site Location
Abstract
1. Introduction
1.1. Sampling Methods
1.2. Plant Species Characteristics
1.3. Location
- To compare three techniques for their cost-effectiveness and consistency in quantifying pollutant capture by roadside hedge species with different leaf structural properties.
- To assess hedge species differences, and associated leaf structural properties, in their capacity to sequester various forms of roadside pollution (metals and PM).
- To compare the importance of hedge species differences in several traffic intensity scenarios.
2. Materials and Methods
2.1. Study Sites
2.2. Sampling and Analysis Approaches
2.3. Plant Material
2.4. Analysis of the Collected Leaf Samples
2.4.1. GC-MS Determination of Heavy Metals’ Concentrations within Leaf Tissue
2.4.2. Determining the Numbers of Particles on Leaves Using Scanning Electron Microscopy (SEM)
2.4.3. Leaf-Washing Experiments (Gravimetric Method)
2.5. Statistical Analyses
3. Results
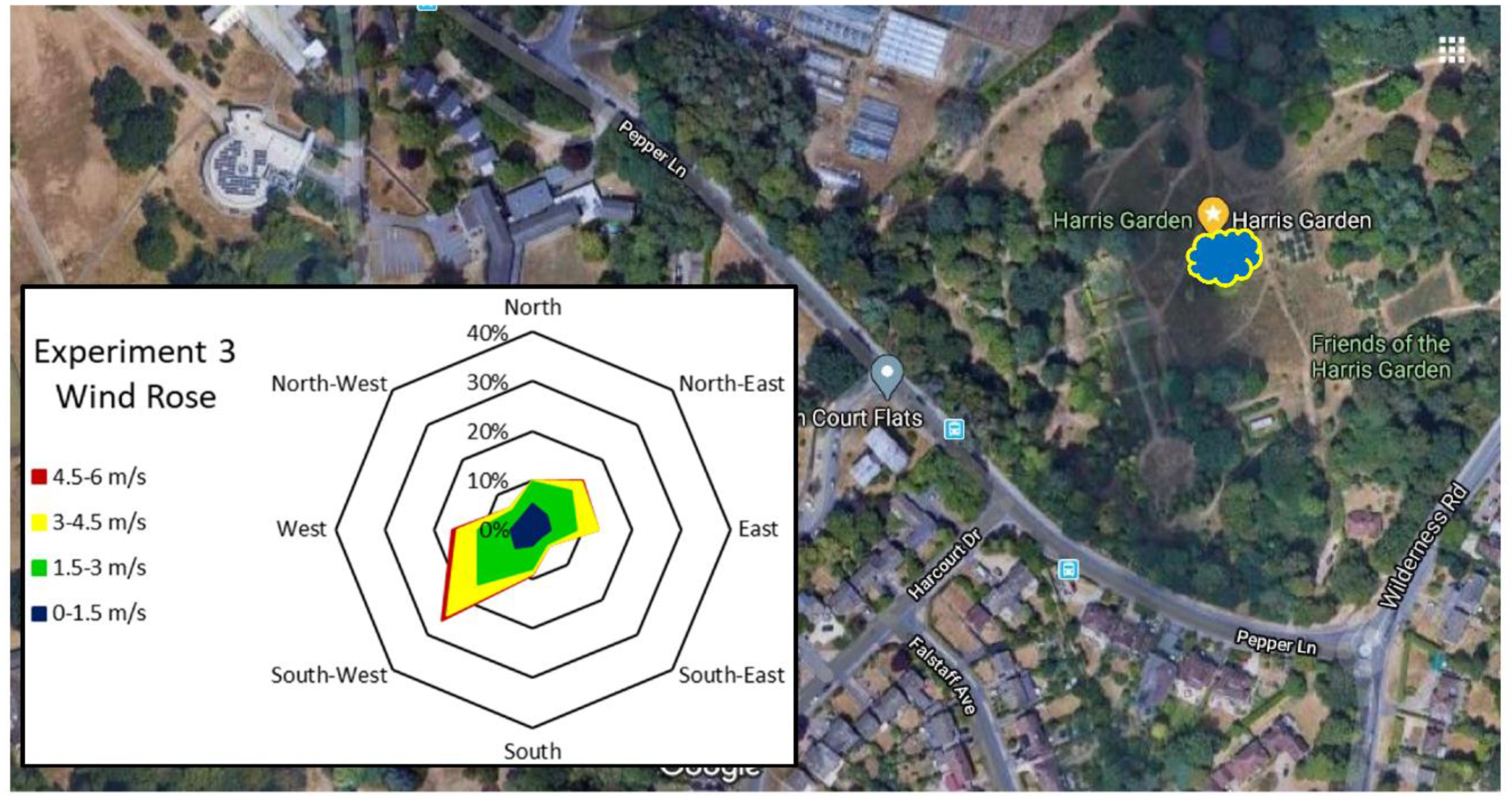
3.1. Weather Data
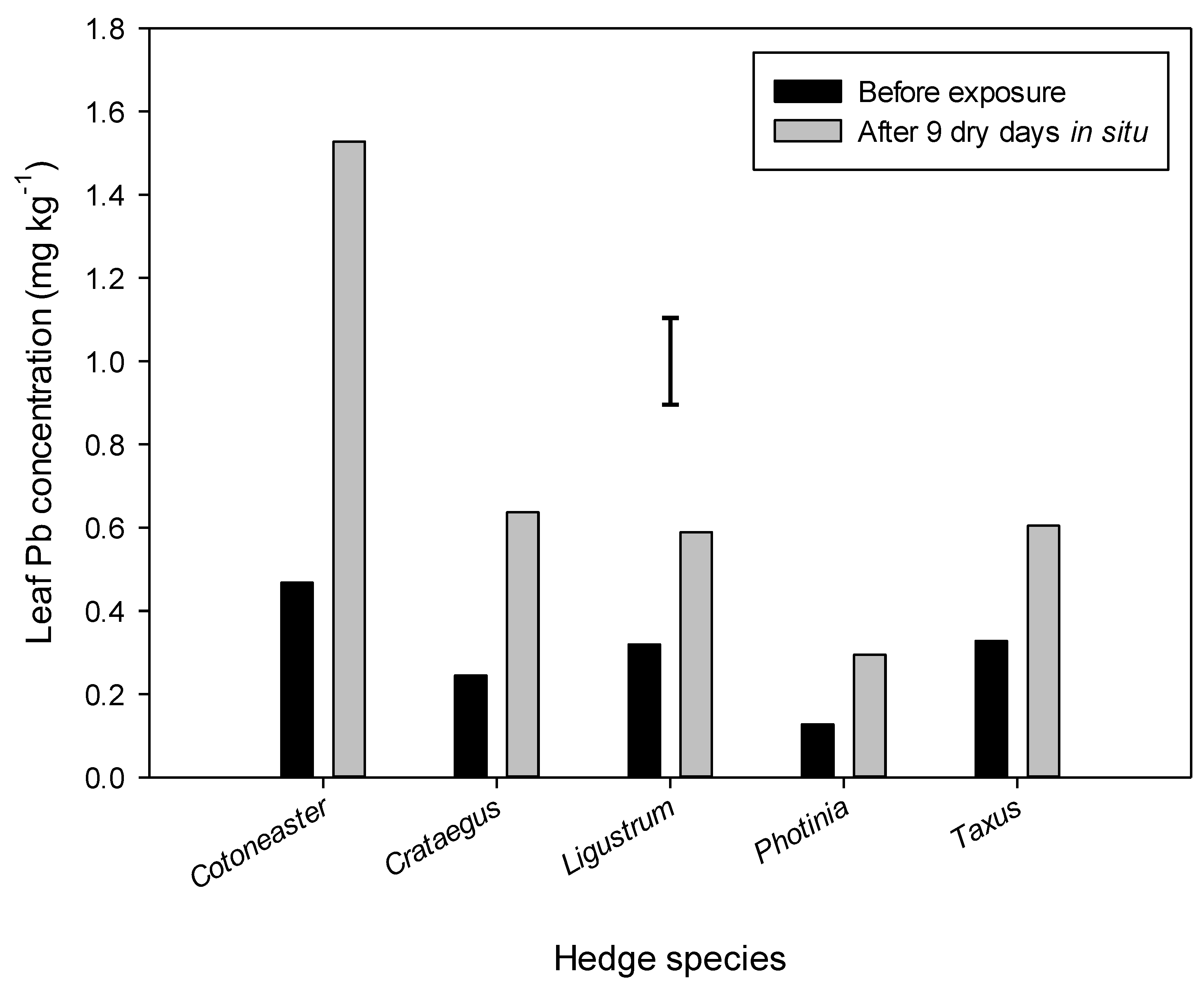
3.2. GC-MS Determination of Heavy Metals’ Concentrations within Leaf Tissue
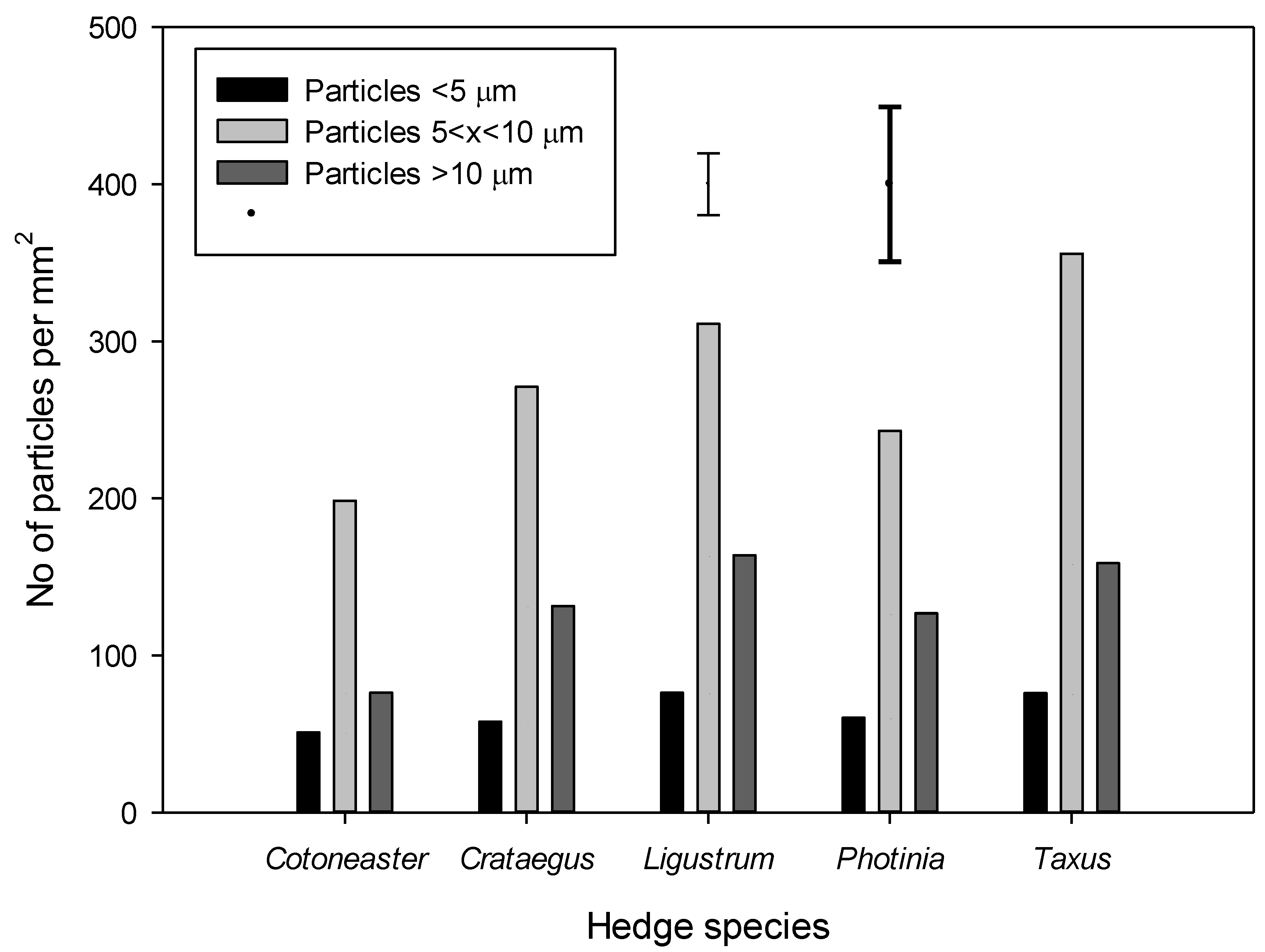
3.3. Determining the Numbers of Particles on Leaves Using Scanning Electron Microscopy (SEM)
3.4. Leaf-Washing Experiments (Gravimetric Method)
4. Discussion
4.1. Comparisons of the Methodologies
4.2. Hedge Species Differences and Links with Leaf Structural Characteristics
4.3. Location
4.4. ‘Bigger Picture’ View
4.5. Limitations of the Study
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Exp. No. | Date | RHmax | RHmin | Tmax | Tmin | Solar Rad (MJ/m2/Day) | Rain Tot (mm) | Ave Wind Spd. (m.s−1) | Median Wind Dir. |
|---|---|---|---|---|---|---|---|---|---|
| Experiment 1 | 02/07/2016 | 92.0 | 45.0 | 18.6 | 9.2 | 24.9 | 2.4 | 2.9 | W |
| 03/07/2016 | 97.0 | 56.0 | 19.9 | 8.5 | 17.3 | 0.0 | 1.6 | SW | |
| 04/07/2016 | 98.0 | 62.0 | 18.7 | 9.3 | 20.2 | 0.0 | 2.3 | SW | |
| 05/07/2016 | 95.0 | 49.0 | 20.1 | 11.6 | 17.5 | 0.0 | 2.2 | NW | |
| 06/07/2016 | 97.0 | 43.0 | 20.2 | 8.2 | 26.8 | 0.0 | 1.2 | W | |
| 07/07/2016 | 85.0 | 60.0 | 21.0 | 13.2 | 20.6 | 0.0 | 2.0 | SW | |
| 08/07/2016 | 92.0 | 57.0 | 21.5 | 14.2 | 13.9 | 0.0 | 3.0 | W | |
| 09/07/2016 | 89.0 | 66.0 | 20.8 | 13.9 | 12.6 | 0.0 | 3.2 | SW | |
| 10/07/2016 | 98.0 | 57.0 | 21.3 | 15.4 | 11.5 | 0.6 | 3.7 | SW | |
| 11/07/2016 | 91.0 | 58.0 | 20.6 | 13.5 | 10.6 | 0.0 | 3.4 | SW | |
| Experiment 2 | 01/10/2016 | 99.0 | 64.0 | 14.9 | 5.7 | 9.1 | 11.0 | 1.1 | S |
| 02/10/2016 | 98.0 | 51.0 | 15.6 | 5.9 | 12.7 | 0.0 | 1.3 | NW | |
| 03/10/2016 | 99.0 | 44.0 | 16.8 | 3.1 | 13.7 | 0.2 | 0.8 | NE | |
| 04/10/2016 | 99.0 | 51.0 | 18.4 | 6.6 | 12.3 | 0.0 | 2.2 | E | |
| 05/10/2016 | 93.0 | 47.0 | 16.3 | 5.9 | 12.2 | 0.0 | 2.7 | E | |
| 06/10/2016 | 94.0 | 48.0 | 14.7 | 6.2 | 6.9 | 0.0 | 1.9 | NE | |
| 07/10/2016 | 95.0 | 74.0 | 14.1 | 10.2 | 2.7 | 0.2 | 1.0 | NE | |
| 08/10/2016 | 95.0 | 71.0 | 15.2 | 10.8 | 4.5 | 0.2 | 1.3 | NW | |
| 09/10/2016 | 96.0 | 55.0 | 15.1 | 7.0 | 7.9 | 0.0 | 1.3 | NE | |
| 10/10/2016 | 99.0 | 52.0 | 13.3 | 3.6 | 11.0 | 0.0 | 1.2 | NW | |
| Experiment 3 | 27/06/2019 | 86.0 | 42.0 | 23.7 | 12.6 | 28.7 | 0.0 | 3.6 | E |
| 28/06/2019 | 88.0 | 48.0 | 23.3 | 12.2 | 28.0 | 0.0 | 3.2 | E | |
| 29/06/2019 | 95.0 | 27.0 | 32.1 | 13.9 | 28.8 | 0.0 | 2.4 | SE | |
| 30/06/2019 | 89.0 | 53.0 | 22.3 | 13.7 | 22.2 | 0.0 | 2.9 | W | |
| 01/07/2019 | 91.0 | 50.0 | 20.0 | 12.2 | 19.3 | 0.0 | 2.7 | NW | |
| 02/07/2019 | 87.0 | 44.0 | 20.0 | 10.1 | 21.8 | 0.0 | 1.7 | NW | |
| 03/07/2019 | 79.0 | 36.0 | 22.5 | 11.3 | 30.3 | 0.0 | 1.7 | NE | |
| 04/07/2019 | 93.0 | 36.0 | 24.9 | 9.0 | 29.1 | 0.0 | 0.9 | S | |
| 05/07/2019 | 90.0 | 40.0 | 24.5 | 13.1 | 26.7 | 0.0 | 1.5 | W | |
| 06/07/2019 | 87.0 | 57.0 | 22.9 | 12.9 | 25.3 | 0.0 | 1.7 | NE |
References
- Pope, C.A.; Dockery, D.W. Health Effects of Fine Particulate Air Pollution: Lines that Connect. J. Air Waste Manag. Assoc. 2006, 56, 709–742. [Google Scholar] [CrossRef]
- Lelieveld, J.; Evans, J.S.; Fnais, M.; Giannadaki, D.; Pozzer, A. The contribution of outdoor air pollution sources to premature mortality on a global scale. Nature 2015, 525, 367–371. [Google Scholar] [CrossRef]
- Maher, B.A.; Moore, C.; Matzka, J. Spatial variation in vehicle-derived metal pollution identified by magnetic and elemental analysis of roadside tree leaves. Atmos. Environ. 2008, 42, 364–373. [Google Scholar] [CrossRef]
- Escobedo, F.J.; Nowak, D.J. Spatial heterogeneity and air pollution removal by an urban forest. Landsc. Urban Plan. 2009, 90, 102–110. [Google Scholar] [CrossRef]
- Grote, R.; Samson, R.; Alonso, R.; Amorim, J.H.; Cariñanos, P.; Churkina, G.; Fares, S.; Thiec, D.L.; Niinemets, Ü.; Mikkelsen, T.N. Functional traits of urban trees: Air pollution mitigation potential. Front. Ecol. Environ. 2016, 14, 543–550. [Google Scholar] [CrossRef]
- Hofman, J.; Bartholomeus, H.; Calders, K.; Van Wittenberghe, S.; Wuyts, K.; Samson, R. On the relation between tree crown morphology and particulate matter deposition on urban tree leaves: A ground-based LiDAR approach. Atmos. Environ. 2014, 99, 130–139. [Google Scholar] [CrossRef]
- Tallis, M.; Taylor, G.; Sinnett, D.; Freer-Smith, P. Estimating the removal of atmospheric particulate pollution by the urban tree canopy of London, under current and future environments. Landsc. Urban Plan. 2011, 103, 129–138. [Google Scholar] [CrossRef]
- Pugh, T.A.M.; MacKenzie, A.R.; Whyatt, J.D.; Hewitt, C.N. Effectiveness of Green Infrastructure for Improvement of Air Quality in Urban Street Canyons. Environ. Sci. Technol. 2012, 46, 7692–7699. [Google Scholar] [CrossRef]
- Abhijith, K.; Kumar, P.; Gallagher, J.; McNabola, A.; Baldauf, R.; Pilla, F.; Broderick, B.; Di Sabatino, S.; Pulvirenti, B. Air pollution abatement performances of green infrastructure in open road and built-up street canyon environments—A review. Atmos. Environ. 2017, 162, 71–86. [Google Scholar] [CrossRef]
- Abhijith, K.V.; Kumar, P. Field investigations for evaluating green infrastructure effects on air quality in open-road conditions. Atmos. Environ. 2019, 201, 132–147. [Google Scholar] [CrossRef]
- Song, Y.; Maher, B.A.; Li, F.; Wang, X.; Sun, X.; Zhang, H. Particulate matter deposited on leaf of five evergreen species in Beijing, China: Source identification and size distribution. Atmos. Environ. 2015, 105, 53–60. [Google Scholar] [CrossRef]
- Weerakkody, U.; Dover, J.W.; Mitchell, P.; Reiling, K. Quantification of the traffic-generated particulate matter capture by plant species in a living wall and evaluation of the important leaf characteristics. Sci. Total Environ. 2018, 635, 1012–1024. [Google Scholar] [CrossRef] [PubMed]
- Weerakkody, U.; Dover, J.W.; Mitchell, P.; Reiling, K. The impact of rainfall in remobilising particulate matter accumulated on leaves of four evergreen species grown on a green screen and a living wall. Urban For. Urban Green. 2018, 35, 21–31. [Google Scholar] [CrossRef]
- Monaci, F.; Moni, F.; Lanciotti, E.; Grechi, D.; Bargagli, R. Biomonitoring of airborne metals in urban environments: New tracers of vehicle emission, in place of lead. Environ. Pollut. 2000, 107, 321–327. [Google Scholar] [CrossRef]
- Blanusa, T.; Fantozzi, F.; Monaci, F.; Bargagli, R. Leaf trapping and retention of particles by holm oak and other common tree species in Mediterranean urban environments. Urban For. Urban Green. 2015, 14, 1095–1101. [Google Scholar] [CrossRef]
- Kumar, R.S.; Rajkumar, P. Characterization of minerals in air dust particles in the state of Tamilnadu, India through FTIR, XRD and SEM analyses. Infrared Phys. Technol. 2014, 67, 30–41. [Google Scholar] [CrossRef]
- Hofman, J.; Wuyts, K.; Van Wittenberghe, S.; Brackx, M.; Samson, R. On the link between biomagnetic monitoring and leaf-deposited dust load of urban trees: Relationships and spatial variability of different particle size fractions. Environ. Pollut. 2014, 189, 63–72. [Google Scholar] [CrossRef]
- Dzierżanowski, K.; Popek, R.; Gawrońska, H.; Sæbø, A.; Gawroński, S.W. Deposition of particulate matter of different size fractions on leaf surfaces and in waxes of urban forest species. Int. J. Phytoremed. 2011, 13, 1037–1046. [Google Scholar] [CrossRef]
- Leonard, R.J.; McArthur, C.; Hochuli, D.F. Particulate matter deposition on roadside plants and the importance of leaf trait combinations. Urban For. Urban Green. 2016, 20, 249–253. [Google Scholar] [CrossRef]
- Castanheiro, A.; Hofman, J.; Nuyts, G.; Joosen, S.; Spassov, S.; Blust, R.; Lenaerts, S.; De Wael, K.; Samson, R. Leaf accumulation of atmospheric dust: Biomagnetic, morphological and elemental evaluation using SEM, ED-XRF and HR-ICP-MS. Atmos. Environ. 2020, 221, 117082. [Google Scholar] [CrossRef]
- Tiwary, A.; Reff, A.; Colls, J.J. Collection of ambient particulate matter by porous vegetation barriers: Sampling and characterization methods. J. Aerosol Sci. 2008, 39, 40–47. [Google Scholar] [CrossRef]
- Sæbø, A.; Popek, R.; Nawrot, B.; Hanslin, H.M.; Gawronska, H.; Gawronski, S.W. Plant species differences in particulate matter accumulation on leaf surfaces. Sci. Total Environ. 2012, 427, 347–354. [Google Scholar] [CrossRef] [PubMed]
- Xie, C.; Kan, L.; Guo, J.; Jin, S.; Li, Z.; Chen, D.; Li, X.; Che, S. A dynamic processes study of PM retention by trees under different wind conditions. Environ. Pollut. 2018, 233, 315–322. [Google Scholar] [CrossRef] [PubMed]
- Blanusa, T.; Garratt, M.; Cathcart-James, M.; Hunt, L.; Cameron, R.W.F. Urban hedges: A review of plant species and cultivars for ecosystem service delivery in north-west Europe. Urban For. Urban Green. 2019, 44, 126391. [Google Scholar] [CrossRef]
- Ottelé, M.; van Bohemen, H.D.; Fraaij, A.L. Quantifying the deposition of particulate matter on climber vegetation on living walls. Ecol. Eng. 2010, 36, 154–162. [Google Scholar] [CrossRef]
- Speak, A.F.; Rothwell, J.J.; Lindley, S.J.; Smith, C.L. Urban particulate pollution reduction by four species of green roof vegetation in a UK city. Atmos. Environ. 2012, 61, 283–293. [Google Scholar] [CrossRef]
- Tiwary, A.; Williams, I.D.; Heidrich, O.; Namdeo, A.; Bandaru, V.; Calfapietra, C. Development of multi-functional streetscape green infrastructure using a performance index approach. Environ. Pollut. 2016, 208, 209–220. [Google Scholar] [CrossRef]
- Weber, F.; Kowarik, I.; Säumel, I. Herbaceous plants as filters: Immobilization of particulates along urban street corridors. Environ. Pollut. 2014, 186, 234–240. [Google Scholar] [CrossRef]
- Beckett, K.P.; Freer-Smith, P.H. Taylor G Particulate pollution capture by urban trees: Effect of species and windspread. Glob. Chang. Biol. 2000, 6, 995–1003. [Google Scholar] [CrossRef]
- Muhammad, S.; Wuyts, K.; Samson, R. Atmospheric net particle accumulation on 96 plant species with contrasting morphological and anatomical leaf characteristics in a common garden experiment. Atmos. Environ. 2019, 202, 328–344. [Google Scholar] [CrossRef]
- Barwise, Y.; Kumar, P. Designing vegetation barriers for urban air pollution abatement: A practical review for appropriate plant species selection. NPJ Clim. Atmos. Sci. 2020, 3, 12. [Google Scholar] [CrossRef]
- Chiam, Z.; Song, X.P.; Lai, H.R.; Tan, H.T.W. Particulate matter mitigation via plants: Understanding complex relationships with leaf traits. Sci. Total Environ. 2019, 688, 398–408. [Google Scholar] [CrossRef] [PubMed]
- Peachey, C.J.; Sinnett, D.; Wilkinson, M.; Morgan, G.W.; Freer-Smith, P.H.; Hutchings, T.R. Deposition and solubility of airborne metals to four plant species grown at varying distances from two heavily trafficked roads in London. Environ. Pollut. 2009, 157, 2291–2299. [Google Scholar] [CrossRef] [PubMed]
- Mori, J.; Hanslin, H.M.; Burchi, G.; Sæbø, A. Particulate matter and element accumulation on coniferous trees at different distances from a highway. Urban For. Urban Green. 2015, 14, 170–177. [Google Scholar] [CrossRef]
- Säumel, I.; Kotsyuk, I.; Hölscher, M.; Lenkereit, C.; Weber, F.; Kowarik, I. How healthy is urban horticulture in high traffic areas? Trace metal concentrations in vegetable crops from plantings within inner city neighbourhoods in Berlin, Germany. Environ. Pollut. 2012, 165, 124–132. [Google Scholar] [CrossRef]
- Antisari, L.V.; Orsini, F.; Marchetti, L.; Vianello, G.; Gianquinto, G. Heavy metal accumulation in vegetables grown in urban gardens. Agron. Sustain. Dev. 2015, 35, 1139–1147. [Google Scholar] [CrossRef]
- Department for Transport, DfT ‘Road Traffic Statistics’. 2018. Available online: https://roadtraffic.dft.gov.uk (accessed on 27 May 2020).
- Fantozzi, F.; Monaci, F.; Blanusa, T.; Bargagli, R. Holm Oak (Quercus ilex L.) canopy as interceptor of airborne trace elements and their accumulation in the litter and topsoil. Environ. Pollut. 2013, 183, 89–95. [Google Scholar] [CrossRef]
- Chen, L.; Xu, Z.; Liu, M.; Huang, Y.; Fan, R.; Su, Y.; Hu, G.; Peng, X.; Peng, X. Lead exposure assessment from study near a lead-acid battery factory in China. Sci. Total Environ. 2012, 429, 191–198. [Google Scholar] [CrossRef]
- Liu, J.; Cao, Z.; Zou, S.; Liu, H.; Hai, X.; Wang, S.; Duan, J.; Xi, B.; Yan, G.; Zhang, S.; et al. An investigation of the leaf retention capacity, efficiency and mechanism for atmospheric particulate matter of five greening tree species in Beijing, China. Sci. Total Environ. 2018, 616, 417–426. [Google Scholar] [CrossRef]
- Chen, L.; Liu, C.; Zhang, L.; Zou, R.; Zhang, Z. Variation in tree species ability to capture and retain airborne fine particulate matter (PM2.5). Sci. Rep. 2017, 7, 1–11. [Google Scholar] [CrossRef]
- Zhang, W.; Zhang, Y.; Gong, J.; Yang, B.; Zhang, Z.; Wang, B.; Zhu, C.; Shi, J.; Yue, K. Comparison of the suitability of plant species for greenbelt construction based on particulate matter capture capacity, air pollution tolerance index, and antioxidant system. Environ. Pollut. 2020, 263, 114615. [Google Scholar] [CrossRef]
- Weerakkody, U.; Dover, J.W.; Mitchell, P.; Reiling, K. Particulate matter pollution capture by leaves of seventeen living wall species with special reference to rail-traffic at a metropolitan station. Urban For. Urban Green. 2017, 27, 173–186. [Google Scholar] [CrossRef]
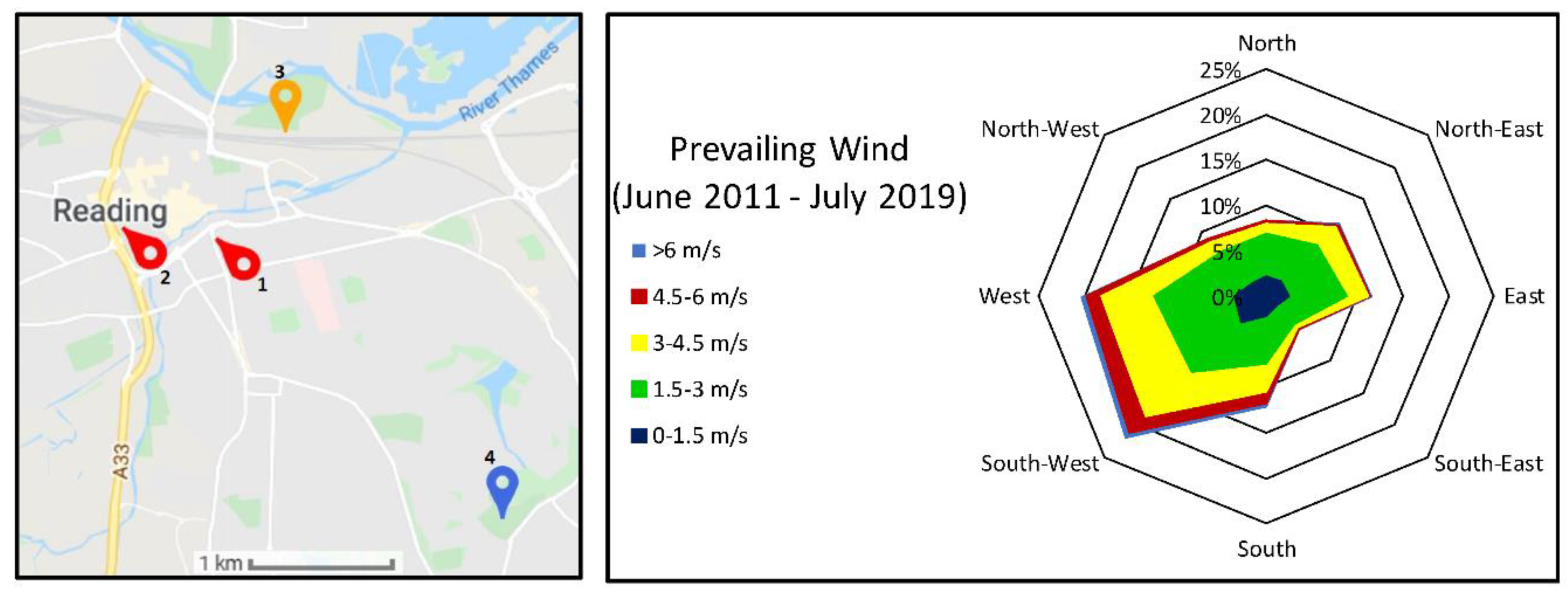
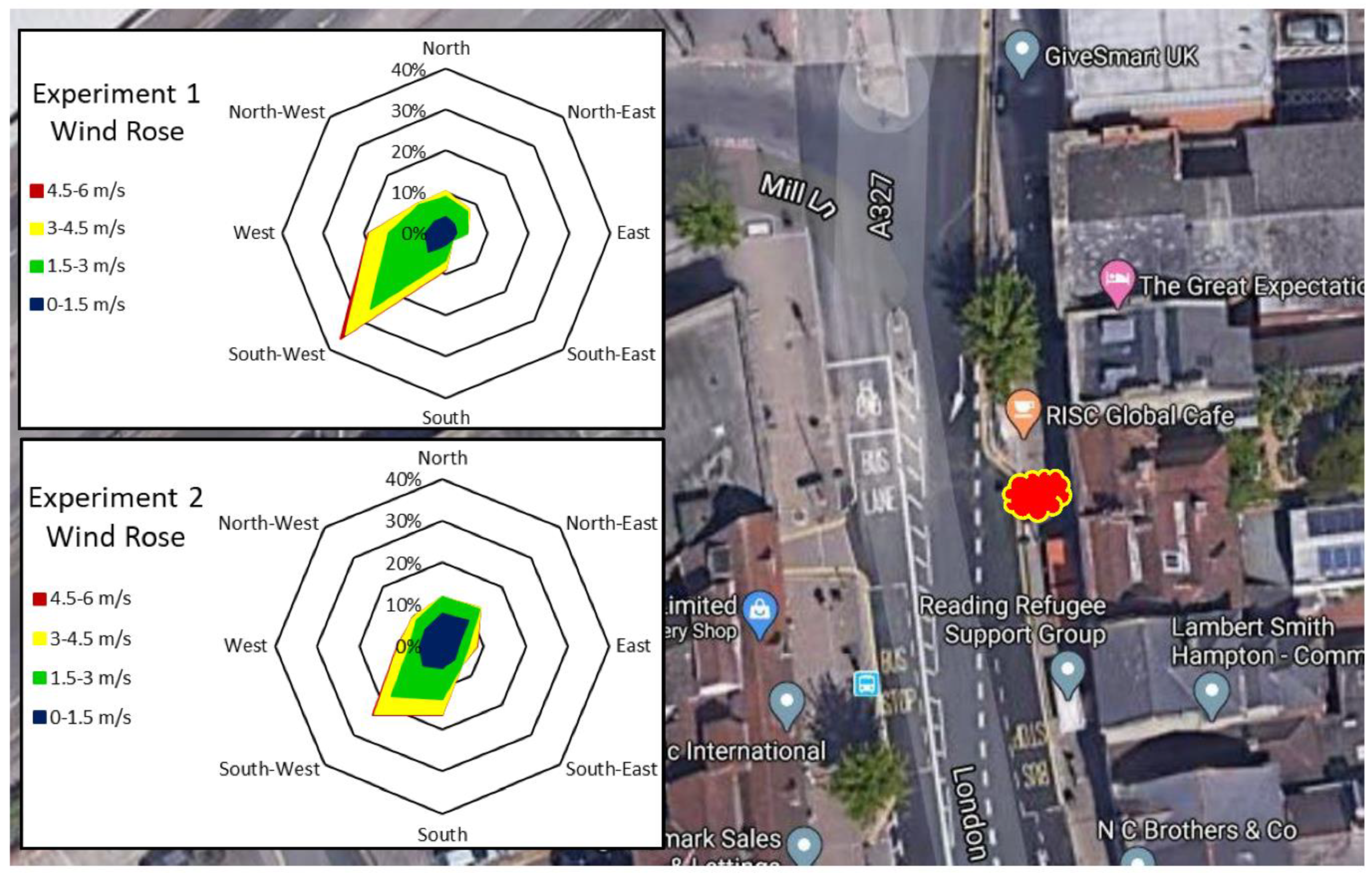
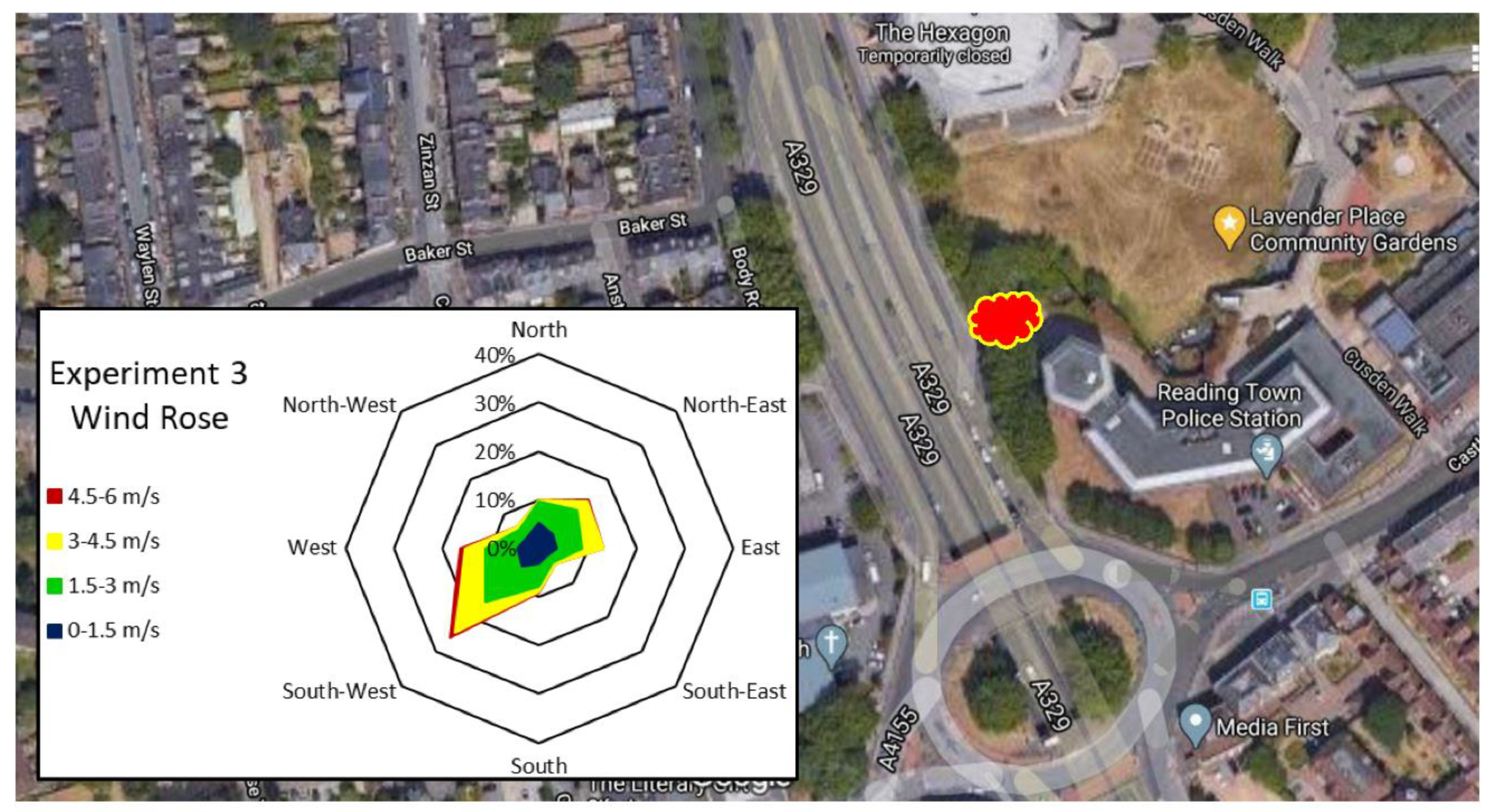
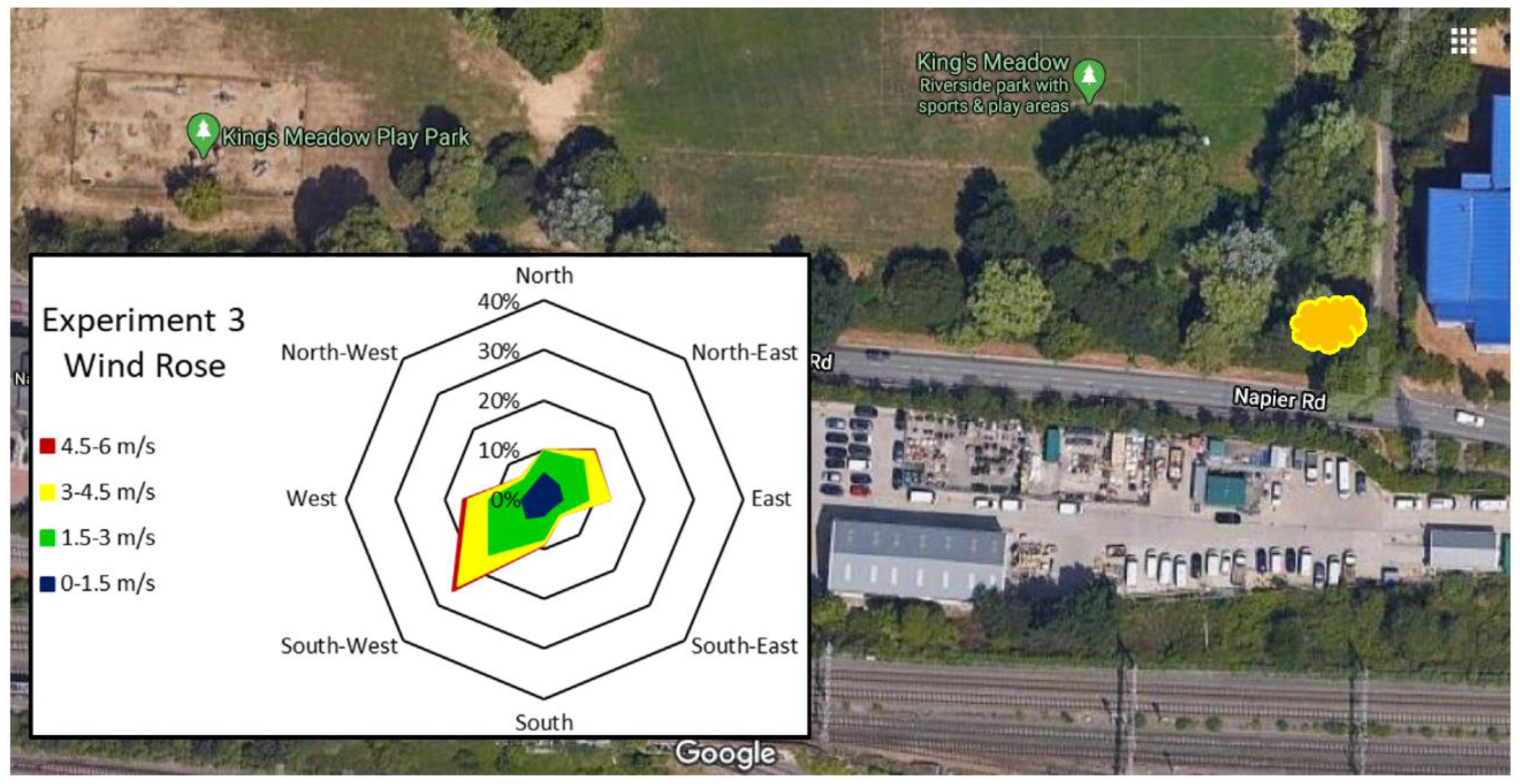
| Experiment 1—Determination of Leaf Heavy Metal Concentrations | Experiment 2—Determination of Particulate Numbers on Leaf Surfaces | Experiment 3—Determination of Particulate Weight in Two Particulate Size Classes, in Three Locations | |
|---|---|---|---|
| Location | Major road—London St, A327, Reading (Figure 2) | Major road—London St, A327, Reading (Figure 2) | Major road—Inner Distribution Road, A329, Reading Minor road—Napier Rd. No road—University of Reading, Harris Gardens (Figure 3, Figure 4 and Figure 5) |
| Arrangement and distance of hedge from the road | Plants in individual containers by the roadside. 1 m | Same as in Experiment 1. 1 m | A full hedgerow in the ground 9 m from major road, 3 m from minor road 100+ m in ‘no road’ scenario |
| Hedge species | Cotoneaster franchetii Crataegus monogyna Ligustrum ovalifolium ‘Aureum’ Photinia x fraseri ‘Red Robin’ Taxus baccata | Same as in Experiment 1 | Acuba japonica Cotoneaster franchetii Crataegus monogyna Thuja plicata |
| Sampling date | 12 July 2016, after 9 rain-free days | 11 October 2016, after 9 rain-free days | 6 June 2019 after 8 rain-free days (Table 4) 8 July 2019, after 12 rain-free days (Table 5) |
| Sampling approach | 4x samples, of each species, approx. 200 cm2 per sample of leaves, front of the hedge | 5x samples, of each species, sample approx. 50 cm2 per sample, front of the hedge. Then 5 leaves in each sample * assessed microscopically, in 2 positions per leaf. | 3–4x samples, of each species, approx. 200 cm2 per sample of leaves, front and back of the hedge |
| Analysis Technique | GC-MS analysis [15] | Scanning electron microscopy—SEM [11] | Leaf washing and sequential filtration/gravimetric method [19,22] |
| Hedge Species | Leaf Image | Leaf Properties |
|---|---|---|
| Acuba japonica |  | Ovate, smooth, large leaves with serrated edges. Evergreen. |
| Cotoneaster franchetii |  | Obovate, small, hairy leaves, particularly abaxially. Evergreen. |
| Crataegus monogyna |  | Deeply lobed, medium sized, smooth surface. Deciduous. |
| Ligustrum ovalifolium ‘Aureum’ |  | Ovate, smooth small to medium sized leaves. Semi-deciduous. |
| Photinia x fraseri ‘Red Robin’ |  | Ovate, smooth, large leaves. Evergreen. |
| Taxus baccata |  | Flat individual needles. Evergreen, conifer. |
| Thuja plicata |  | Complex elongated structure, large scaly leaves. Evergreen, conifer. |
| Hedge Species | Sampling Site | Distance from Road (m) | Hedge Dimensions (m) | |
|---|---|---|---|---|
| Height | Width | |||
| Acuba japonica ‘Crotonifolia | Major rd | 1 | 0.5 | 1 |
| Minor rd | 7 | 1.5 | 2 | |
| No road | >100 | 2 | 2 | |
| Crataegus monogyna | Major rd | 9 | 1.2 | 1.5 |
| Minor rd | 6 | 2 | 1 | |
| No road | >100 | 2 | 2 | |
| Cotoneaster franchetii | Major rd | 9 | 1.2 | 1.5 |
| Minor rd | 6 | 2 | 1.5 | |
| No road | >100 | 2 | 2 | |
| Thuja plicata | Major rd | 9 | 1.6 | 1.5 |
| Hedge Species | Sampling Position | PM, ≥ 8 μm (mg cm−2) ± SEoM | PM, 8-0.2 μm (mg cm−2) ± SEoM | Total PM (mg cm−2) ± SEoM |
|---|---|---|---|---|
| Cotoneaster franchetii | Front | 0.207 ± 0.012 | 0.009 ± 0.001 | 0.216 ± 0.013 |
| Back | 0.143 ± 0.002 | 0.006 ± 0.001 | 0.149 ± 0.002 | |
| Crataegus monogyna | Front | 0.060 ± 0.007 | 0.004 ± 0.001 | 0.063 ± 0.008 |
| Back | 0.059 ± 0.011 | 0.002 ± 0.001 | 0.061 ± 0.012 | |
| Thuja plicata | Front | 0.099 ± 0.012 | 0.007 ± 0.001 | 0.105 ± 0.012 |
| Back | 0.080 ± 0.007 | 0.004 ± 0.001 | 0.085 ± 0.007 | |
| LSD | 0.028 | 0.003 | 0.029 |
| Hedge Species | Location | PM ≥ 8 μm (mg cm−2) ± SEoM | PM 8-0.2 μm (mg cm−2) ± SEoM | Total PM (mg cm−2) ± SEoM |
|---|---|---|---|---|
| Acuba japonica | Major rd | 0.050 ± 0.005 | 0.003 ± 0.000 | 0.053 ± 0.006 |
| Minor rd | 0.020 ± 0.002 | 0.002 ± 0.001 | 0.022 ± 0.002 | |
| No rd | 0.012 ± 0.001 | 0.001 ± 0.001 | 0.013 ± 0.002 | |
| LSD | 0.0112 | 0.002 | 0.0119 | |
| Cotoneaster franchetii | Major rd | 0.193 ± 0.025 | 0.012 ± 0.001 | 0.204 ± 0.026 |
| Minor rd | 0.133 ± 0.011 | 0.009 ± 0.001 | 0.141 ± 0.012 | |
| No rd | 0.103 ± 0.008 | 0.005 ± 0.0003 | 0.108 ± 0.008 | |
| LSD | 0.058 | 0.002 | 0.06 | |
| Crataegus monogyna | Major rd | 0.076 ± 0.005 | 0.004 ± 0.001 | 0.079 ± 0.006 |
| Minor rd | 0.051 ± 0.003 | 0.003 ± 0.000 | 0.054 ± 0.003 | |
| No rd | 0.042 ± 0.009 | 0.003 ± 0.000 | 0.045 ± 0.009 | |
| LSD | 0.022 | 0.002 | 0.023 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Blanuša, T.; Qadir, Z.J.; Kaur, A.; Hadley, J.; Gush, M.B. Evaluating the Effectiveness of Urban Hedges as Air Pollution Barriers: Importance of Sampling Method, Species Characteristics and Site Location. Environments 2020, 7, 81. https://doi.org/10.3390/environments7100081
Blanuša T, Qadir ZJ, Kaur A, Hadley J, Gush MB. Evaluating the Effectiveness of Urban Hedges as Air Pollution Barriers: Importance of Sampling Method, Species Characteristics and Site Location. Environments. 2020; 7(10):81. https://doi.org/10.3390/environments7100081
Chicago/Turabian StyleBlanuša, Tijana, Zeenat Jabeen Qadir, Amanpreet Kaur, James Hadley, and Mark B. Gush. 2020. "Evaluating the Effectiveness of Urban Hedges as Air Pollution Barriers: Importance of Sampling Method, Species Characteristics and Site Location" Environments 7, no. 10: 81. https://doi.org/10.3390/environments7100081
APA StyleBlanuša, T., Qadir, Z. J., Kaur, A., Hadley, J., & Gush, M. B. (2020). Evaluating the Effectiveness of Urban Hedges as Air Pollution Barriers: Importance of Sampling Method, Species Characteristics and Site Location. Environments, 7(10), 81. https://doi.org/10.3390/environments7100081





