Assessment of Diatom Assemblages in Close Proximity to Mining Activities in Nunavik, Northern Quebec (Canada)
Abstract
:1. Introduction
2. Materials and Methods
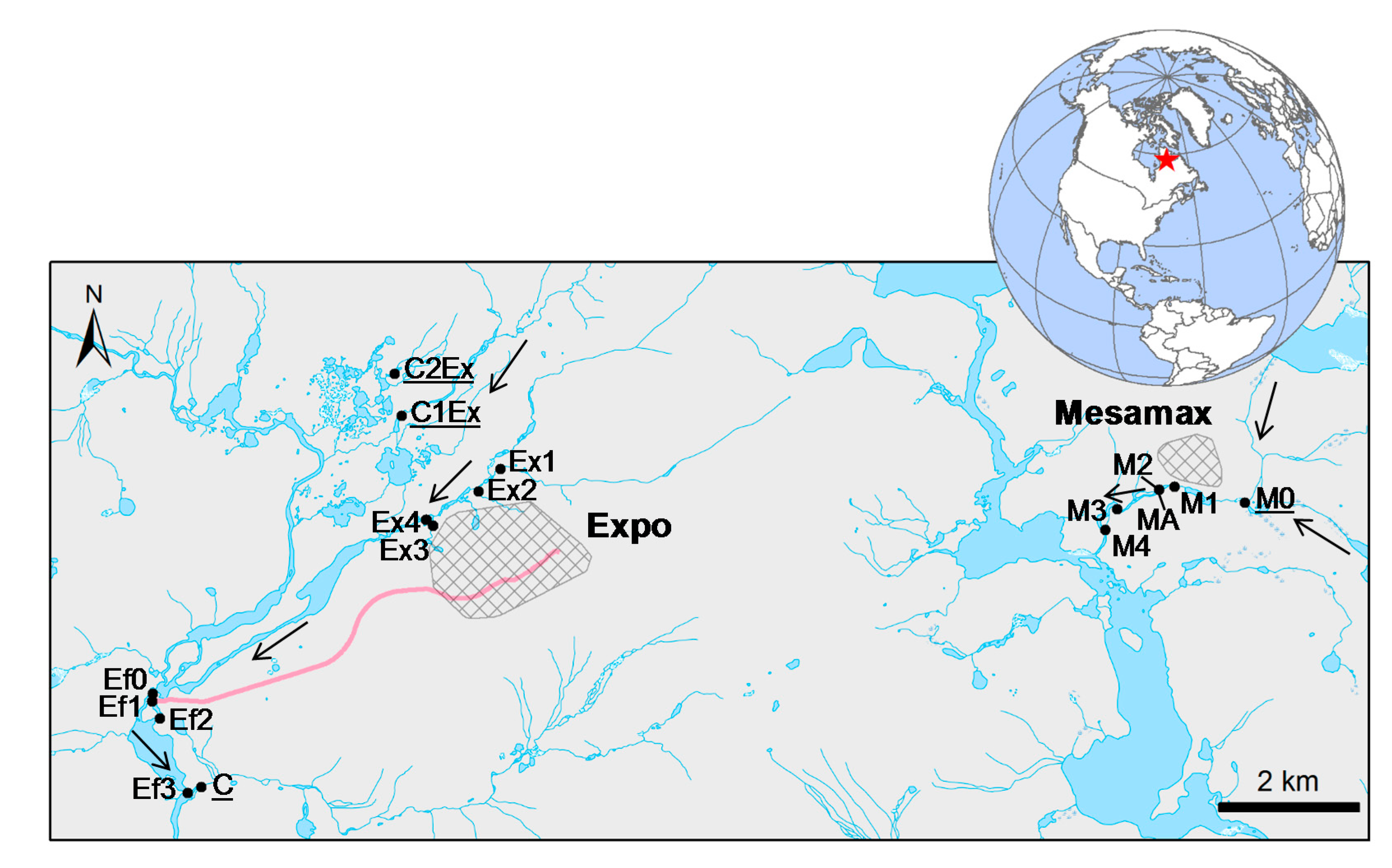
2.1. Study Area
2.2. Water and Biofilm Collection
2.3. Water Chemistry and Diatom Assemblage Analyses
3. Results and Discussion
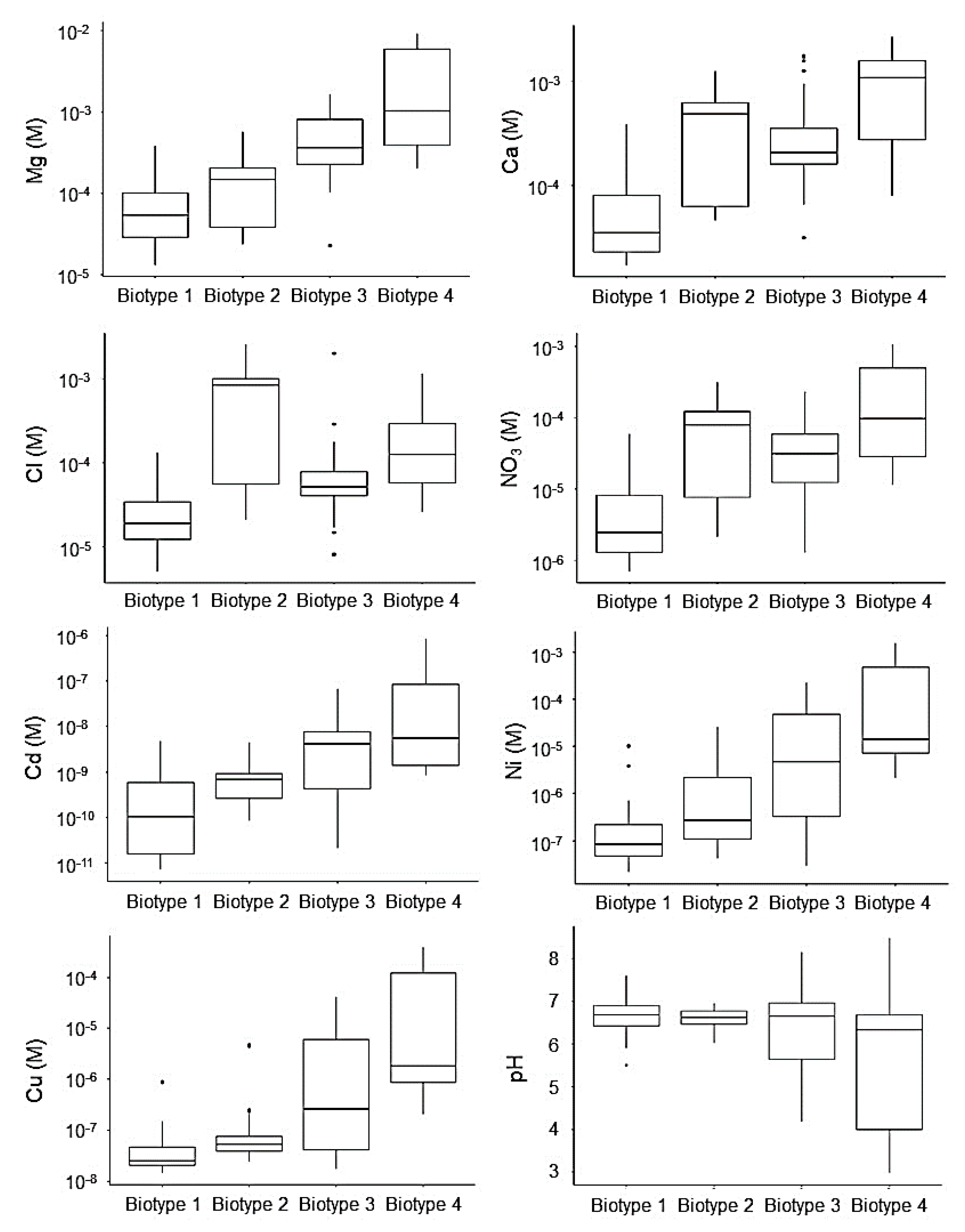
3.1. Water Chemistry Values
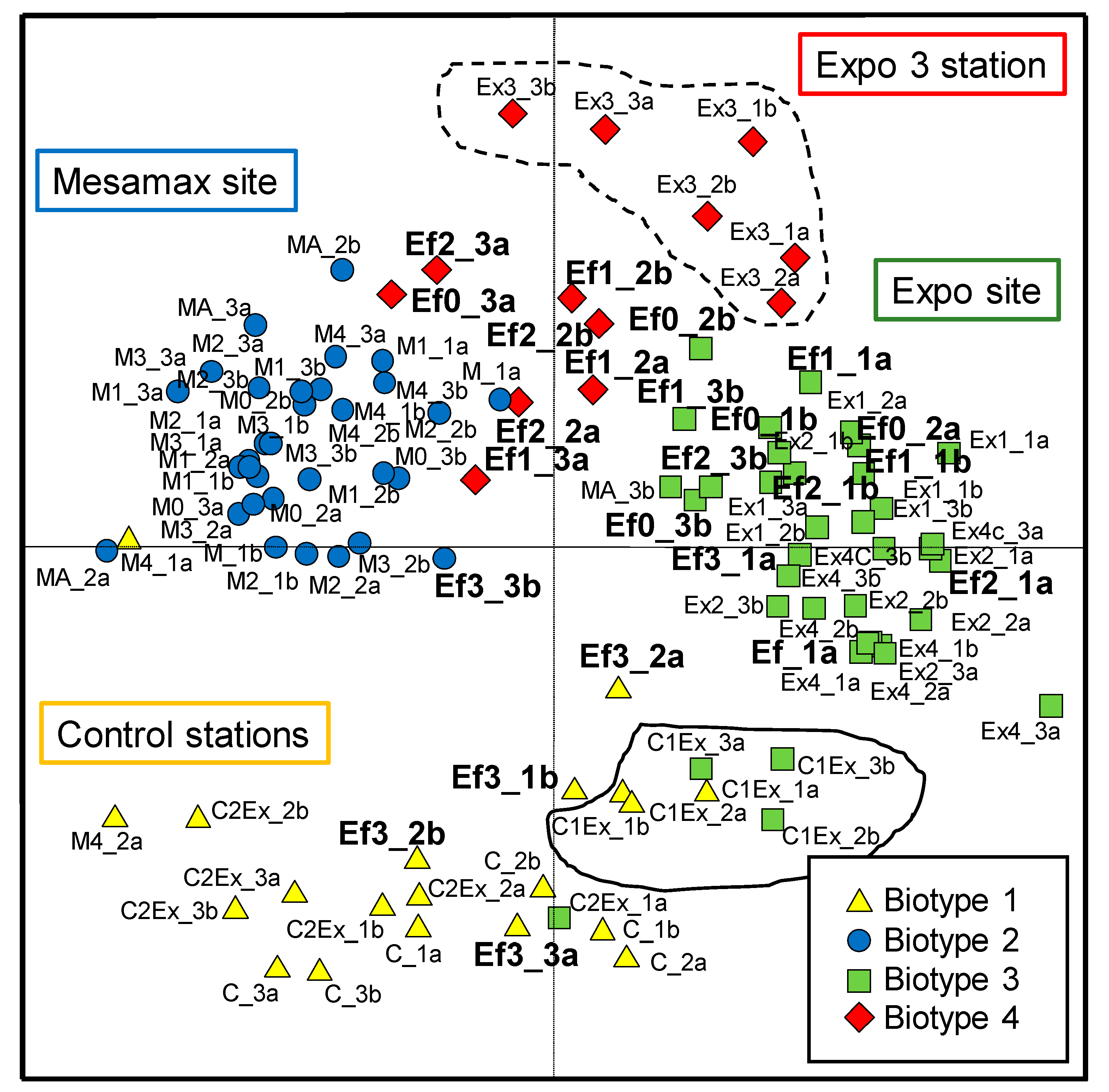
3.2. Diatom Assemblages and Biotypes
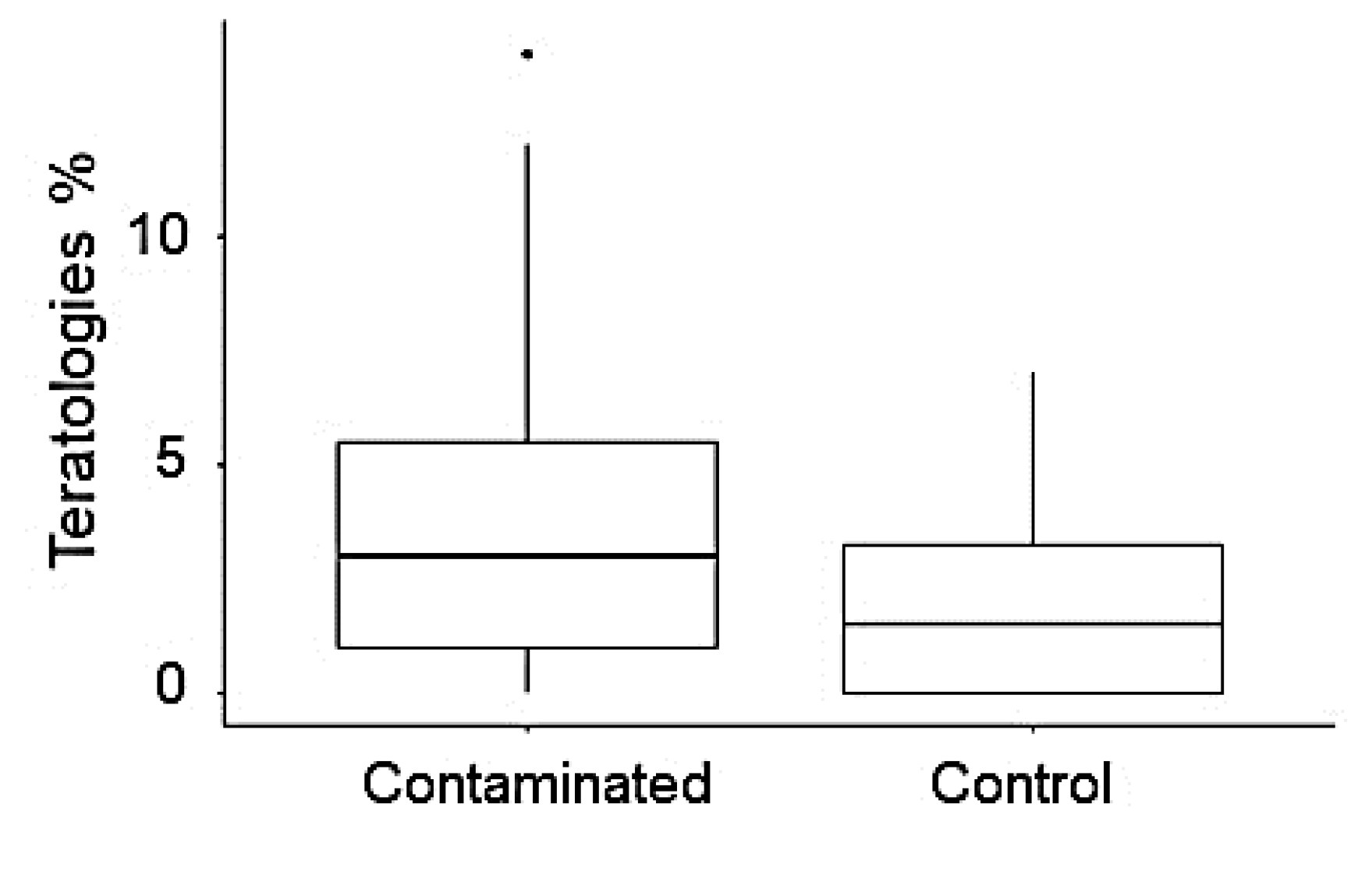
3.3. Diatom Assemblages versus Water Chemistry, Tolerant Taxa and Teratologies
3.4. Diatom-Based Monitoring North of the 49th Parallel
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wah Chu, K.; Chow, K.L. Synergistic toxicity of multiple heavy metals is revealed by a biological assay using a nematode and its transgenic derivative. Aquat. Toxicol. 2002, 61, 53–64. [Google Scholar] [CrossRef]
- Prygiel, J.; Coste, M. Mise au point de l’Indice Biologique Diatomée, un indice diatomique pratique applicable au réseau hydrographique français. Eau Ind. Nuis. 1998, 211, 40–45. [Google Scholar]
- Lavoie, I.; Campeau, S.; Grenier, M.; Dillon, P.J. A diatom-based index for the biological assessment of eastern Canadian rivers: An application of correspondence analysis (CA). Can. J. Fish. Aquat. Sci. 2006, 63, 1793–1811. [Google Scholar] [CrossRef]
- Lavoie, I.; Campeau, S.; Zugic-Drakulic, N.; Winter, J.G.; Fortin, C. Using diatoms to monitor stream biological integrity in Eastern Canada: An overview of 10 years of index development and ongoing challenges. Sci. Total Environ. 2014, 475, 187–200. [Google Scholar] [CrossRef] [PubMed]
- Morin, S.; Cordonier, A.; Lavoie, I.; Arini, A.; Blanco, S.; Duong, T.T.; Tornés, E.; Bonet, B.; Corcoll, N.; Faggiano, L.; et al. Consistency in diatom response to metal-contaminated environments. In Handbook of Environmental Chemistry; Guasch, H., Ginebreda, A., Geiszinger, A., Eds.; Springer: Heidelberg, Germany, 2012; Volume 19, pp. 117–146. [Google Scholar]
- Lavoie, I.; Hamilton, P.B.; Morin, S.; Kim Tiam, S.; Kahlert, M.; Gonçalves, S.; Falasco, E.; Fortin, C.; Gontero, B.; Heudre, D.; et al. Diatom teratologies as biomarkers of contamination: Are all deformities ecologically meaningful? Ecol. Indic. 2017, 82, 539–550. [Google Scholar] [CrossRef]
- Debenest, T.; Silvestre, J.; Coste, M.; Delmas, F.; Pinelli, E. Herbicide effects on freshwater benthic diatoms: Induction of nucleus alterations and silica cell wall abnormalities. Aquat. Toxicol. 2008, 88, 88–94. [Google Scholar] [CrossRef] [Green Version]
- Bottin, M.; Giraudel, J.-L.; Lek, S.; Tison-Rosebery, J. diatSOM: A R-package for diatom biotypology using self-organizing maps. Diatom Res. 2014, 29, 5–9. [Google Scholar] [CrossRef]
- Ter Braak, C.J.F.; Smilauer, P. CANOCO Reference Manual and CanoDraw for Windows User’s Guide: Software for Canonical Community Ordination (Version 4.5); Biometris: Wageningen, The Netherlands, 2002. [Google Scholar]
- Team, R.C. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2015. [Google Scholar]
- Leguay, S.; Lavoie, I.; Levy, J.L.; Fortin, C. Using biofilms for monitoring metal contamination in lotic ecosystems: The protective effects of hardness and pH on metal bioaccumulation. Environ. Toxicol. Chem. 2016, 35, 1489–1501. [Google Scholar] [CrossRef] [PubMed]
- Cantonati, M.; Angeli, N.; Virtanen, L.; Wojtal, A.Z.; Gabriell, J.; Falasco, E.; Lavoie, I.; Morin, S.; Marchetoo, A.; Fortin, C.; et al. Achnanthidium minutissimum (Bacillariophyta) valve deformities as indicators of metal enrichment in diverse widely-distributed freshwater habitats. Sci. Total Environ. 2014, 75, 201–215. [Google Scholar] [CrossRef]
- Antoniades, D.; Douglas, M.S.V. Characterization of High Arctic stream diatom assemblages from Cornwallis Island, Nunavut, Canada. Can. J. Bot. 2002, 80, 50–58. [Google Scholar] [CrossRef]
- Michelutti, N.; Holtham, A.J.; Douglas, M.S.V.; Smol, J.P. Periphytic diatom assemblages from ultra-oligotrophic and UV transparent lakes and ponds on Victoria Island and comparisons with other diatom surveys in the Canadian Arctic. J. Phycol. 2003, 39, 465–480. [Google Scholar] [CrossRef]
- Van De Vijver, B.; Van Kerckvoorde, A.; Beyens, L. Freshwater and terrestrial moss diatom assemblages of the Cambridge Bay area, Victoria Island (Nunavut, Canada). Nova Hedwigia. 2003, 76, 225–243. [Google Scholar] [CrossRef]
- Van Dam, H.; Merten, S.A.; Sinkeldam, J. A coded checklist and ecological indicator values of freshwater diatoms from the Netherlands. Neth. J. Aquat. Ecol. 1994, 28, 117–133. [Google Scholar]
- Keatley, B.E.; Douglas, M.S.V.; Smol, J.P. Evaluating the influence of environmental and spatial variables on diatom species distributions from Melville Island (Canadian High Arctic) lakes and ponds. Botany 2008, 86, 76–90. [Google Scholar] [CrossRef]
- Cattaneo, A.; Couillard, Y.; Wunsam, S.; Courcelles, M. Diatom taxonomic and morphological changes as indicators of metal pollution and recovery in Lac Dufault (Québec, Canada). J. Paleolimnol. 2004, 32, 163–175. [Google Scholar] [CrossRef]
- Morin, S.; Coste, M.; Hamilton, P.B. Scanning electron microscopy observations of deformities in small pennate diatoms exposed to high cadmium concentrations. J. Phycol. 2008, 44, 1512–1518. [Google Scholar] [CrossRef] [PubMed]
- Lavoie, I.; Lavoie, M.; Fortin, C. A mine of information: Benthic algal communities as biomonitors of metal contamination from abandoned tailings. Sci. Total Environ. 2012, 425, 231–241. [Google Scholar] [CrossRef]
- Lavoie, I.; Morin, S.; Laderriere, V.; Fortin, C. Freshwater diatoms as indicators of combined long-term mining and urban stressors in Junction creek (Ontario, Canada). Environments 2018, 5, 30. [Google Scholar] [CrossRef]
- Di Toro, D.M.; Allen, H.E.; Bergman, H.L.; Meyer, J.S.; Paquin, P.R.; Santore, R.C. Biotic ligand model of the acute toxicity of metals. 1. Technical basis. Environ. Toxicol. Chem. 2001, 20, 2383–2396. [Google Scholar] [CrossRef]
- Fortin, C.; Denison, F.H.; Garnier-Laplace, J. Metal-phytoplankton interactions: Modeling the effect of competing ions (H+, Ca2+, and Mg2+) on uranium uptake. Environ. Toxicol. Chem. 2007, 26, 242–248. [Google Scholar] [CrossRef]
- Tudesque, L.; Grenouillet, G.; Gevrey, M.; Khazraie, K.; Brosse, S. Influence of small-scale gold mining on French Guiana streams: Are diatom assemblages valid disturbance sensors? Ecol. Indic. 2012, 14, 100–106. [Google Scholar] [CrossRef]
- Luís, A.T.; Teixeira, P.; Alexander, A.C.; Almeida, S.F.P.; da Silva, E.F.; Culp, J.M. Benthic diatom communities in streams from zinc mining areas in continental (Canada) and Mediterranean climates (Portugal). Water Qual. Res. J. Can. 2013, 48, 180–191. [Google Scholar] [CrossRef] [Green Version]
- Luís, A.T.; Teixeira, P.; Almeida, S.F.P.; Matos, J.X.; da Silva, E.F. Environmental impact of mining activities in the Lousal area (Portugal): Chemical and diatom characterization of metal-contaminated stream sediments and surface water of Corona stream. Sci. Total Environ. 2011, 409, 4312–4325. [Google Scholar] [CrossRef] [PubMed]
- Zalack, J.T.; Smucker, N.; Vis, M. Development of a diatom index of biotic integrity for acid mine drainage impacted streams. Ecol. Indic. 2010, 10, 287–295. [Google Scholar] [CrossRef]
- Morin, S.; Duong, T.T.; Dabrin, A.; Coynel, A.; Herlory, O.; Baudrimont, M.; Delmas, F.; Durrieu, G.; Schäfer, J.; Winterton, P.; et al. Long term survey of heavy metal pollution, biofilm contamination and diatom community structure in the Riou-Mort watershed, South West France. Environ. Pollut. 2008, 151, 532–542. [Google Scholar] [CrossRef] [PubMed]
| Stations | pH | Ni | Cu | Cd | Mg | Ca | Cl | NO3 | SO4 | COD | PO4 | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| µM | µM | nM | mM | mM | µM | µM | mM | mg/L | nM | |||||||||||||
| EX1 | 6.9 | (0.3) | 0.29 | (0.05) | 0.04 | (0.04) | 0.47 | (0.51) | 0.17 | (0.05) | 0.15 | (0.05) | 65 | (38) | 13.0 | (6.0) | 0.23 | (0.10) | 0.64 | (0.17) | 50 | (47) |
| EX2 | 4.9 | (0.6) | 56 | (26) | 6.1 | (4.7) | 16 | (17) | 1.18 | (0.24) | 0.30 | (0.07) | 51.0 | (9.7) | 59 | (14) | 1.67 | (0.34) | 0.78 | (0.35) | 96 | (58) |
| EX3 | 4.3 | (1.4) | 680 | (480) | 170 | (140) | 210 | (290) | 5.7 | (2.3) | 1.52 | (0.71) | 440 | (340) | 540 | (330) | 9.7 | (5.4) | 1.03 | (0.29) | 170 | (120) |
| EX4 | 6.3 | (0.9) | 42 | (66) | 10 | (13) | 15 | (24) | 0.55 | (0.40) | 0.23 | (0.10) | 77 | (48) | 38 | (26) | 0.78 | (0.55) | 0.83 | (0.23) | 61 | (38) |
| EX4C | 4.1 | (0.7) | 400 | (260) | 61 | (51) | 33 | (32) | 3.6 | (2.3) | 1.00 | (0.55) | 180 | (89) | 390 | (280) | 6.3 | (4.0) | 1.05 | (0.33) | 160 | (100) |
| EF0 | 6.6 | (0.7) | 19 | (24) | 3.4 | (4.7) | 3.8 | (3.0) | 0.35 | (0.14) | 0.19 | (0.07) | 40 | (11) | 22 | (13) | 0.53 | (0.28) | 1.16 | (0.21) | 82 | (35) |
| EF1 | 6.9 | (1.0) | 16 | (18) | 2.3 | (3.3) | 6.8 | (8.3) | 0.90 | (0.46) | 0.93 | (0.61) | 160 | (100) | 180 | (190) | 4.7 | (4.7) | 1.00 | (0.48) | 64 | (52) |
| EF2 | 6.6 | (0.5) | 12 | (11) | 1.9 | (2.1) | 3.3 | (2.2) | 0.68 | (0.22) | 0.72 | (0.27) | 72 | (17) | 53 | (31) | 1.47 | (0.64) | 1.22 | (0.19) | 64 | (44) |
| EF3 | 6.7 | (0.4) | 6.6 | (9.4) | 1.0 | (1.7) | 1.7 | (1.7) | 0.25 | (0.17) | 0.25 | (0.22) | 39 | (19) | 22 | (20) | 0.51 | (0.49) | 2.03 | (0.97) | 73 | (53) |
| M0* | 6.5 | (0.2) | 0.16 | (0.11) | 0.04 | (0.01) | 0.62 | (0.26) | 0.09 | (0.07) | 0.38 | (0.40) | 780 | (930) | 87 | (110) | 0.05 | (0.03) | 1.07 | (0.31) | 40 | (15) |
| M1 | 6.5 | (0.2) | 0.21 | (0.16) | 0.04 | (0.01) | 0.43 | (0.31) | 0.08 | (0.06) | 0.34 | (0.30) | 680 | (720) | 85 | (84) | 0.05 | (0.02) | 1.14 | (0.33) | 55 | (11) |
| M2 | 6.6 | (0.2) | 0.24 | (0.20) | 0.06 | (0.05) | 0.43 | (0.32) | 0.08 | (0.06) | 0.33 | (0.30) | 670 | (720) | 70 | (82) | 0.05 | (0.02) | 1.12 | (0.35) | 54 | (18) |
| M3 | 6.7 | (0.2) | 2.0 | (1.6) | 0.09 | (0.05) | 1.5 | (1.6) | 0.17 | (0.13) | 0.46 | (0.39) | 620 | (620) | 85 | (72) | 0.46 | (0.41) | 1.12 | (0.30) | 51 | (13) |
| M4 | 6.7 | (0.2) | 2.6 | (2.1) | 0.10 | (0.07) | 1.6 | (1.7) | 0.19 | (0.12) | 0.48 | (0.37) | 590 | (590) | 80 | (67) | 0.48 | (0.39) | 1.21 | (0.45) | 58.2 | (9.5) |
| MA | 6.7 | (0.2) | 0.86 | (0.71) | 0.07 | (0.06) | 0.54 | (0.27) | 0.24 | (0.18) | 0.74 | (0.32) | 1100 | (630) | 157 | (59) | 0.7 | (1.1) | 1.15 | (0.46) | 71 | (29) |
| C * | 6.8 | (0.6) | 0.09 | (0.07) | 0.03 | (0.02) | 0.08 | (0.17) | 0.06 | (0.02) | 0.03 | (0.01) | 21 | (123) | 2.5 | (2.6) | 0.01 | (0.00) | 2.62 | (0.91) | 60 | (26) |
| C1EX * | 6.6 | (0.4) | 0.11 | (0.03) | 0.03 | (0.02) | 0.21 | (0.26) | 0.22 | (0.11) | 0.12 | (0.05) | 18.8 | (8.5) | 6.2 | (3.8) | 0.31 | (0.15) | 1.23 | (0.35) | 44 | (21) |
| C2EX * | 6.3 | (0.4) | 0.04 | (0.01) | 0.02 | (0.01) | 0.07 | (0.10) | 0.02 | (0.01) | 0.03 | (0.01) | 17.0 | (9.9) | 3.2 | (2.1) | 0.02 | (0.01) | 1.63 | (0.53) | 71 | (22) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lavoie, I.; Morin, S.; Laderriere, V.; Paris, L.-E.; Fortin, C. Assessment of Diatom Assemblages in Close Proximity to Mining Activities in Nunavik, Northern Quebec (Canada). Environments 2019, 6, 74. https://doi.org/10.3390/environments6060074
Lavoie I, Morin S, Laderriere V, Paris L-E, Fortin C. Assessment of Diatom Assemblages in Close Proximity to Mining Activities in Nunavik, Northern Quebec (Canada). Environments. 2019; 6(6):74. https://doi.org/10.3390/environments6060074
Chicago/Turabian StyleLavoie, Isabelle, Soizic Morin, Vincent Laderriere, Louise-Emmanuelle Paris, and Claude Fortin. 2019. "Assessment of Diatom Assemblages in Close Proximity to Mining Activities in Nunavik, Northern Quebec (Canada)" Environments 6, no. 6: 74. https://doi.org/10.3390/environments6060074
APA StyleLavoie, I., Morin, S., Laderriere, V., Paris, L.-E., & Fortin, C. (2019). Assessment of Diatom Assemblages in Close Proximity to Mining Activities in Nunavik, Northern Quebec (Canada). Environments, 6(6), 74. https://doi.org/10.3390/environments6060074







