Combined Effects of Trace Metals and Light on Photosynthetic Microorganisms in Aquatic Environment
Abstract
1. Introduction
2. Light Radiation from Environmental Factor to Stressor
3. Light as a Modifier of the Ambient Medium Characteristics
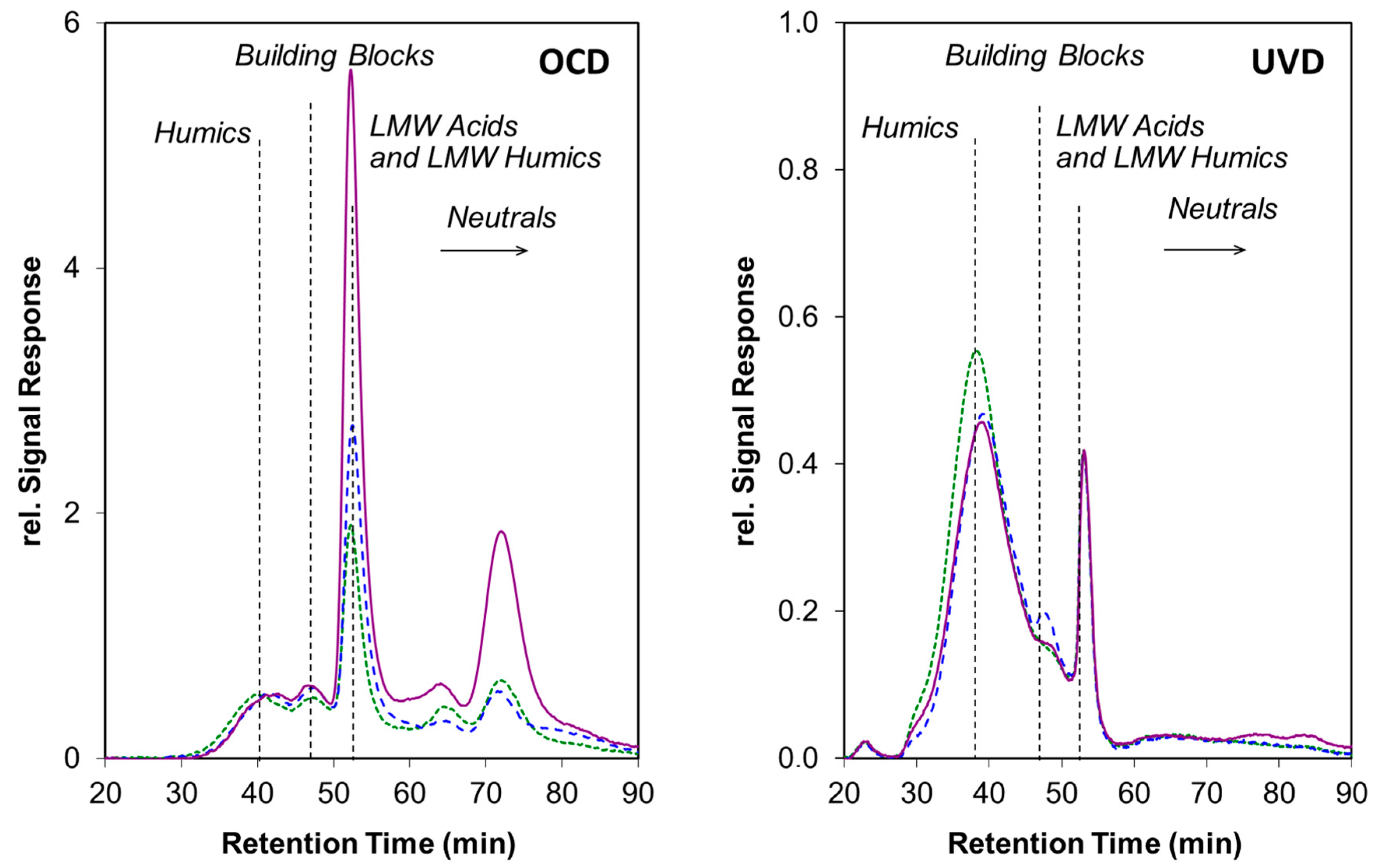
3.1. Photo-Transformations of Dissolved Organic Matter
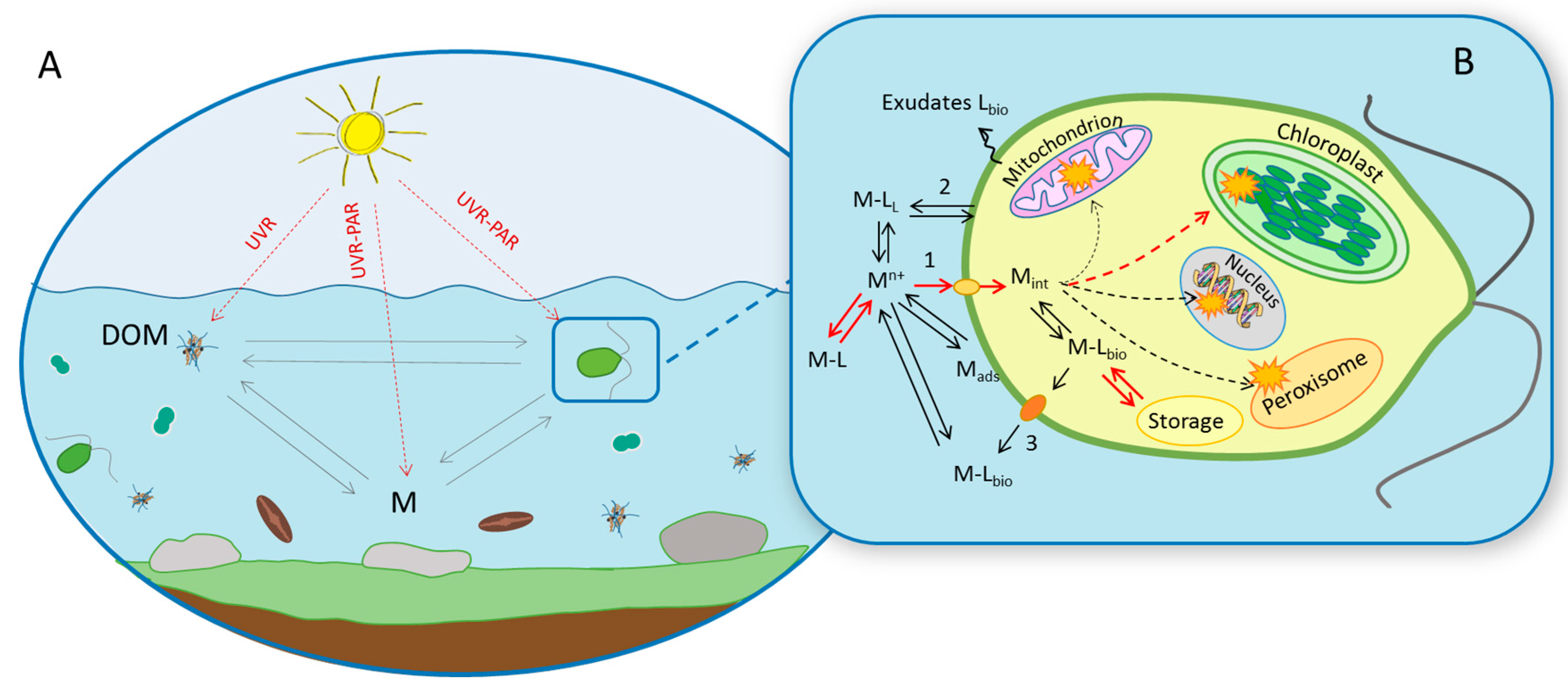
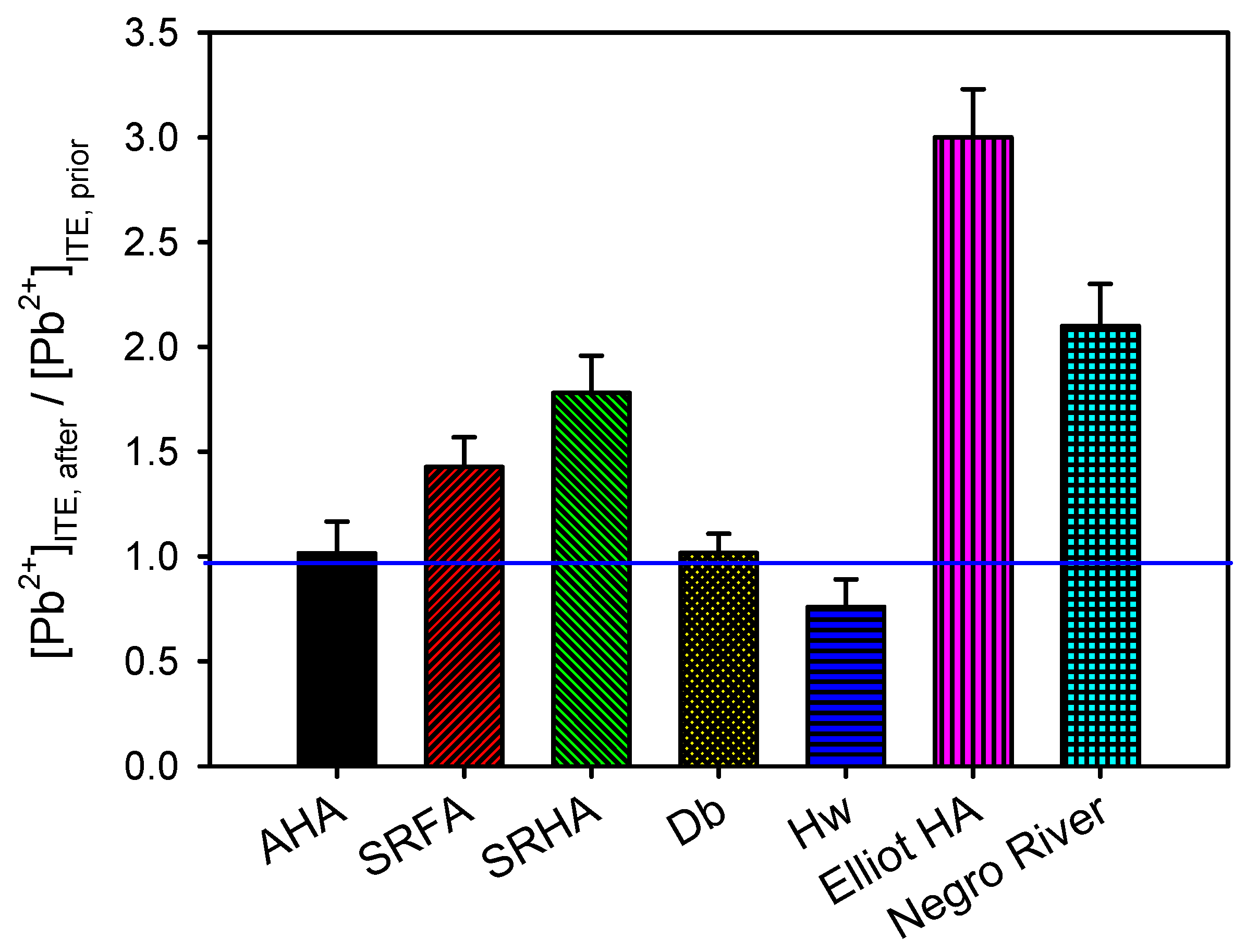
3.2. Consequences for Trace Metal Speciation
3.3. Consequences for Trace Metal Bioavailability
3.4. Light-Induced Enhancement of the Oxidative Activity in Ambient Medium
4. Light as a Modifier of the Trace Metal Effect to Photosynthetic Microorganisms
4.1. Toxicokinetics Aspects
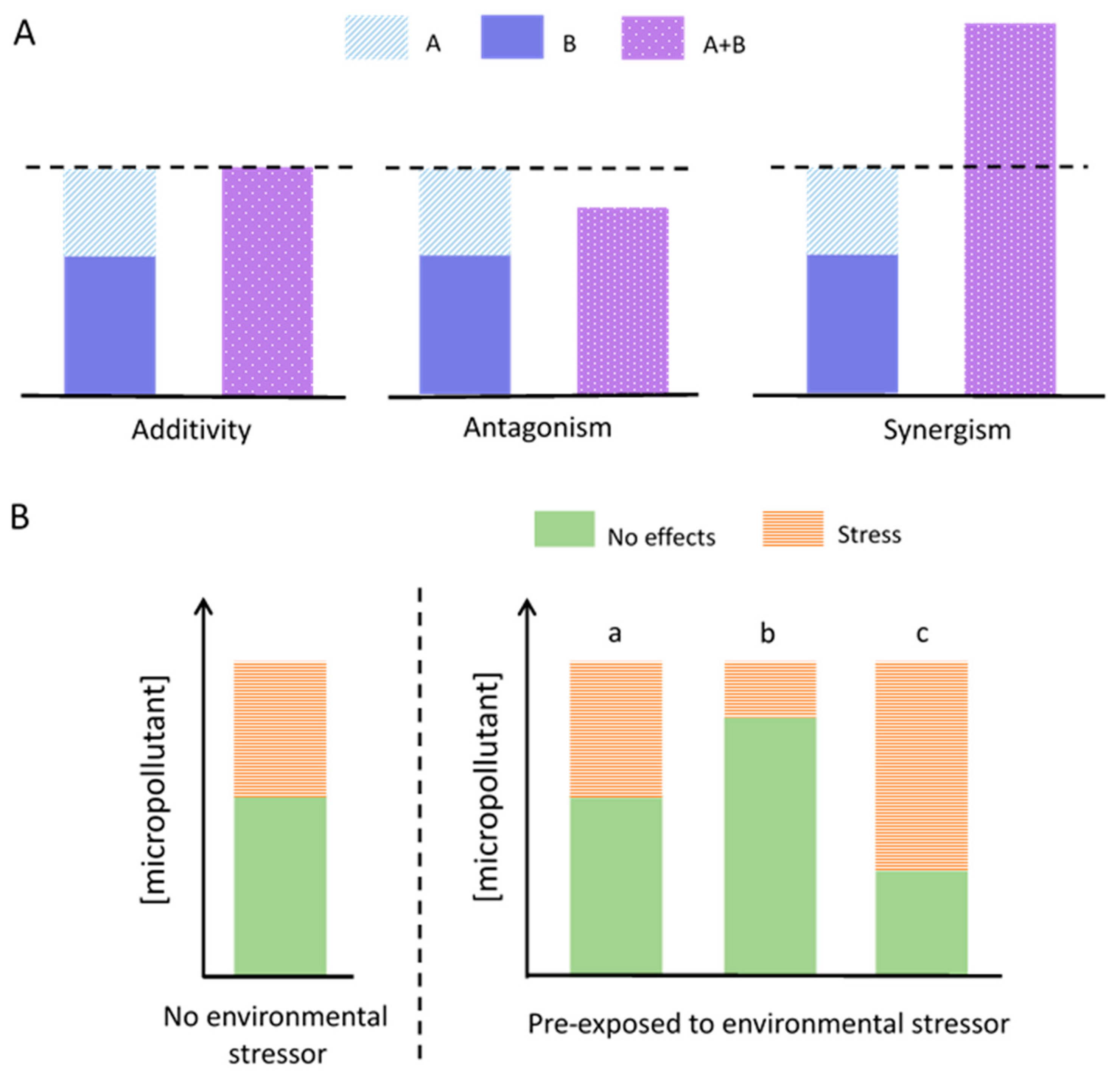
4.2. Mode-of-Action and Multistressor Interactions
4.3. Toxicodynamics Aspects
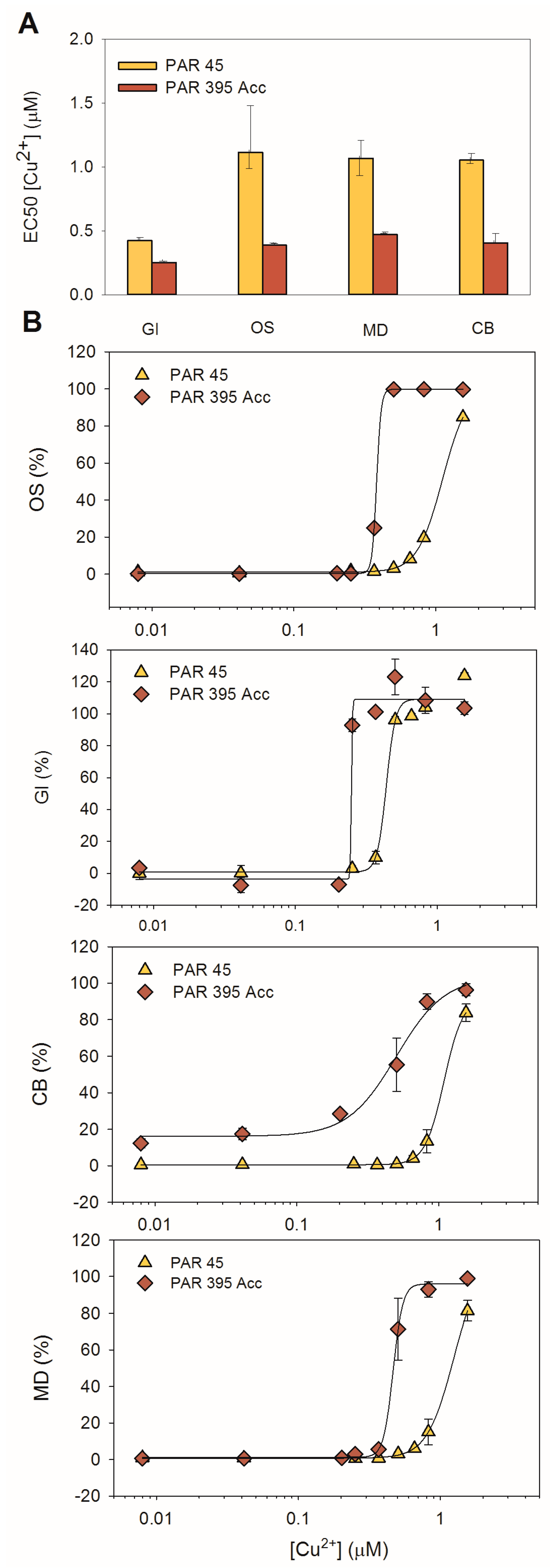
4.4. Role of the Light Pre-Exposure History and Acclimation
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Artigas, J.; Arts, G.; Babut, M.; Caracciolo, A.B.; Charles, S.; Chaumot, A.; Combourieu, B.; Dahllof, I.; Despreaux, D.; Ferrari, B.; et al. Towards a renewed research agenda in ecotoxicology. Environ. Pollut. 2012, 160, 201–206. [Google Scholar] [CrossRef] [PubMed]
- Heugens, E.H.W.; Hendriks, A.J.; Dekker, T.; Straalen, N.M.V.; Admiraal, W. A review of the effects of multiple stressors on aquatic organisms and analysis of uncertainty factors for use in risk assessment. Crit. Rev. Toxicol. 2002, 31, 247–284. [Google Scholar] [CrossRef]
- Holmstrup, M.; Bindesbol, A.M.; Oostingh, G.J.; Duschl, A.; Scheil, V.; Kohler, H.R.; Loureiro, S.; Soares, A.M.; Ferreira, A.L.; Kienle, C.; et al. Interactions between effects of environmental chemicals and natural stressors: A review. Sci. Total Environ. 2010, 408, 3746–3762. [Google Scholar] [CrossRef] [PubMed]
- Fischer, B.B.; Pomati, F.; Eggen, R.I. The toxicity of chemical pollutants in dynamic natural systems: The challenge of integrating environmental factors and biological complexity. Sci. Total Environ. 2013, 449, 253–259. [Google Scholar] [CrossRef] [PubMed]
- Segner, H.; Schmitt-Jansen, M.; Sabater, S. Assessing the impact of multiple stressors on aquatic biota: The receptor’s side matters. Environ. Sci. Technol. 2014, 48, 7690–7696. [Google Scholar] [CrossRef] [PubMed]
- Laskowski, R.; Bednarska, A.J.; Kramarz, P.E.; Loureiro, S.; Scheil, V.; Kudlek, J.; Holmstrup, M. Interactions between toxic chemicals and natural environmental factors a meta-analysis and case studies. Sci. Total Environ. 2010, 408, 3763–3774. [Google Scholar] [CrossRef] [PubMed]
- Korkaric, M.; Behra, R.; Fischer, B.B.; Junghans, M.; Eggen, R.I. Multiple stressor effects in chlamydomonas reinhardtii toward understanding mechanisms of interaction between effects of ultraviolet radiation and chemical pollutants. Aquat. Toxicol. 2015, 162, 18–28. [Google Scholar] [CrossRef] [PubMed]
- Clements, W.H.; Rohr, J.R. Community responses to contaminants: Using basic ecological principles to predict ecotoxicological effects. Environ. Toxicol. Chem. 2009, 28, 1789–1800. [Google Scholar] [CrossRef] [PubMed]
- Van Straalen, N.M. Ecotoxicology becomes stress ecology. Environ. Sci. Technol. 2003, 37, 324–330. [Google Scholar] [CrossRef]
- Lyons, T.W.; Reinhard, C.T.; Planavsky, N.J. The rise of oxygen in earth’s early ocean and atmosphere. Nature 2014, 506, 307–315. [Google Scholar] [CrossRef] [PubMed]
- Tercier-Waeber, M.-L.; Stoll, S.; Slaveykova, V.I. Trace metal behavior in surface waters: Emphasis on dynamic speciation, sorption processes and bioavailability. Arch. Sci. 2012, 65, 1–24. [Google Scholar]
- Templeton, D.M.; Ariese, F.; Cornelis, R.; Danielsson, L.-G.; Muntau, H.; van Leeuwen, H.P.; Lobinski, R. IUPAC guidelines for terms related to speciation of trace elements. Pure Appl. Chem. 2000, 72, 1453–1470. [Google Scholar]
- Campbell, P.G.C. Interactions between trace metals and aquatic organisms: A critique of the free-ion activity model. Met. Speciat. Bioavailab. 1994, 2, 45–102. [Google Scholar]
- Wilkinson, K.J.; Buffle, J. Critical evaluation of physicochemical parameters and processes for modelling the biological uptake of trace metals in environmental (aquatic) systems. Physicochem. Kinet. Trans. Biointerfaces 2004, 9, 447–533. [Google Scholar]
- Buffle, J.; Wilkinson, K.J.; van Leeuwen, H.P. Chemodynamics and bioavailability in natural waters. Environ. Sci. Technol. 2009, 43, 7170–7174. [Google Scholar] [CrossRef] [PubMed]
- Worms, I.; Simon, D.F.; Hassler, C.S.; Wilkinson, K.J. Bioavailability of trace metals to aquatic microorganisms: Importance of chemical, biological and physical processes on biouptake. Biochimie 2006, 88, 1721–1731. [Google Scholar] [CrossRef] [PubMed]
- Lavoie, M.; Le Faucheur, S.; Fortin, C.; Campbell, P.G. Cadmium detoxification strategies in two phytoplankton species: Metal binding by newly synthesized thiolated peptides and metal sequestration in granules. Aquat. Toxicol. 2009, 92, 65–75. [Google Scholar] [CrossRef] [PubMed]
- Slaveykova, V.I.; Guignard, C.; Eybe, T.; Migeon, H.N.; Hoffmann, L. Dynamic nanosims ion imaging of unicellular freshwater algae exposed to copper. Anal. Bioanal. Chem. 2009, 393, 583–589. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Wakao, S.; Fischer, B.B.; Niyogi, K.K. Sensing and responding to excess light. Annu. Rev. Plant Biol. 2009, 60, 239–260. [Google Scholar] [CrossRef] [PubMed]
- Hader, D.P.; Williamson, C.E.; Wangberg, S.A.; Rautio, M.; Rose, K.C.; Gao, K.; Helbling, E.W.; Sinha, R.P.; Worrest, R. Effects of UV radiation on aquatic ecosystems and interactions with other environmental factors. Photochem. Photobiol. Sci. 2015, 14, 108–126. [Google Scholar] [CrossRef] [PubMed]
- He, Y.-Y.; Hader, D.-P. Reactive oxygen species and UV-B: Effect on cyanobacteria. Photochem. Photobiol. Sci. 2002, 1, 729–736. [Google Scholar] [CrossRef] [PubMed]
- Sinha, R.P.; Häder, D.-P. UV-induced DNA damage and repair: A review. Photochem. Photobiol. Sci. 2002, 1, 225–236. [Google Scholar] [CrossRef] [PubMed]
- Warwick, F.V.; Roy, S. Solar ultraviolet-b radiation and aquatic primary production: Damage, protection, and recovery. Environ. Rev. 1993, 1, 1–12. [Google Scholar]
- Cheloni, G.; Slaveykova, V. Photo-oxidative stress in green algae and cyanobacteria. React. Oxyg. Species 2018, 5, 126–133. [Google Scholar] [CrossRef]
- Singh, S.P.; Hader, D.P.; Sinha, R.P. Cyanobacteria and ultraviolet radiation (UVR) stress: Mitigation strategies. Ageing Res. Rev. 2010, 9, 79–90. [Google Scholar] [CrossRef] [PubMed]
- Sinha, R.P.; Ambasht, N.K.; Sinha, J.P.; Klisch, M.; Häder, D.-P. UV-B-induced synthesis of mycosporine-like amino acids in three strains of nodularia (cyanobacteria). J. Photochem. Photobiol. B 2003, 71, 51–58. [Google Scholar] [CrossRef] [PubMed]
- Laurion, I.; Lami, A.; Sommaruga, R. Distribution of mycosporine-like amino acids and photoprotective carotenoids among freshwater phytoplankton assemblages. Aquat. Microb. Ecol. 2002, 26, 283–294. [Google Scholar] [CrossRef]
- Norval, M.; Cullen, A.P.; de Gruijl, F.R.; Longstreth, J.; Takizawa, Y.; Lucas, R.M.; Noonan, F.P.; van der Leun, J.C. The effects on human health from stratospheric ozone depletion and its interactions with limate change. Photochem. Photobiol. Sci. 2007, 6, 232–251. [Google Scholar] [CrossRef] [PubMed]
- Zepp, R.G.; Erickson, D.J., III; Paul, N.D.; Sulzberger, B. Interactive effects of solar uv and climate change on biogeochemical cycles. Photochem. Photobiol. Sci. 2007, 6, 286–300. [Google Scholar] [CrossRef] [PubMed]
- Sulzberger, B.; Durisch-Kaiser, E. Chemical characterization of dissolved organic matter (DOM): A prerequisite for understanding UV-induced changes of DOM absorption properties and bioavailability. Aquat. Sci. 2009, 71, 104–126. [Google Scholar] [CrossRef]
- Gonsior, M.; Peake, B.M.; Cooper, W.T.; Podgorski, D.; Andrilli, J.; Cooper, W.J. Photochemically induced changes in dissolved organic matter identified by ultrahigh resolution fourier transform ion cyclotron resonance mass spectrometry. Environ. Sci. Technol. 2009, 43, 698–703. [Google Scholar] [CrossRef] [PubMed]
- Porcal, P.; Amirbahman, A.; Kopacek, J.; Novak, F.; Norton, S.A. Photochemical release of humic and fulvic acid-bound metals from simulated soil and streamwater. J. Environl. Monit. 2009, 11, 1064–1071. [Google Scholar] [CrossRef] [PubMed]
- Zepp, R.G.; Callaghan, T.V.; Erickson, D.J., III. Interactive effects of ozone depletion and climate change on biogeochemical cycles. Photochem. Photobiol. Sci. 2003, 2, 51–61. [Google Scholar] [CrossRef] [PubMed]
- Brooks, M.L.; Meyer, J.S.; McKnight, D.M. Photooxidation of wetland and riverine dissolved organic matter: Altered copper complexation and organic composition. Hydrobiologia 2007, 579, 95–113. [Google Scholar] [CrossRef]
- Moran, M.A.; Sheldon, W.M.; Zepp, R.G. Carbon loss and optical property changes dring long-term photochemical and biological degradation of estuarine dissolved organic matter. Limnol. Oceangr. 2000, 45, 1254–1264. [Google Scholar] [CrossRef]
- Del Vecchio, R.; Blough, N.V. Photobleaching of chromophoric dissolved organic matter in natural waters: Kinetics and modeling. Mar. Chem. 2002, 78, 231–253. [Google Scholar] [CrossRef]
- Obernosterer, I.; Benner, R. Competition between biological and photochemical processes in the mineralization of dissolved organic carbon. Limnol. Oceangr. 2004, 2004, 117–124. [Google Scholar] [CrossRef]
- Mayer, L.M.; Schick, L.L.; Skorko, K.; Boss, E. Photdissolution of particulate organic matter from sediments. Limnol. Oceangr. 2006, 51, 1064–1071. [Google Scholar] [CrossRef]
- Zhang, Y.; Liu, M.; Qin, B.; Feng, S. Photochemical degradation of chromophoric-dissolved organic matter exposed to simulated UV-B and natural solar radiation. Hydrobiologia 2009, 627, 159–168. [Google Scholar] [CrossRef]
- Dalzell, B.J.; Minor, E.C.; Mopper, K.M. Photodegradation of estuarine dissolved organic matter: A multi-method assessment of DOM transformation. Org. Geochem. 2009, 40, 243–257. [Google Scholar] [CrossRef]
- Spierings, J.; Worms, I.A.M.; Mieville, P.; Slaveykova, V.I. Effect of humic substance photoalteration on lead bioavailability to freshwater microalgae. Environ. Sci. Technol. 2011, 45, 345–3458. [Google Scholar] [CrossRef] [PubMed]
- Worms, I.A.; Adenmatten, D.; Mieville, P.; Traber, J.; Slaveykova, V.I. Photo-transformation of pedogenic humic acid and consequences for Cd(ii), Cu(ii) and Pb(ii) speciation and bioavailability to green microalga. Chemosphere 2015, 138, 908–915. [Google Scholar] [CrossRef] [PubMed]
- Miller, W.L.; Moran, M.A.; Sheldon, W.M.; Zepp, R.G.; Opsahl, S. Determination of apparent quantum yeald spectra for the formation of biologically labile photoproducts. Limnol. Oceangr. 2002, 47, 343–352. [Google Scholar] [CrossRef]
- Obernosterer, I.; Sempere, R.; Herndl, G.J. Ultraviolet radiation induces reversal of the bioavailability of dom to marine bacterioplankton. Aquat. Microb. Ecol. 2001, 24, 61–68. [Google Scholar] [CrossRef]
- Xu, H.; Jiang, H. UV-induced photochemical heterogeneity of dissolved and attached organic matter associated with cyanobacterial blooms in a eutrophic freshwater lake. Water Res. 2013, 47, 6506–6515. [Google Scholar] [CrossRef] [PubMed]
- Du, Y.X.; Zhang, Y.Y.; Chen, F.Z.; Chang, Y.G.; Liu, Z.W. Photochemical reactivities of dissolved organic matter (DOM) in a sub-alpine lake revealed by EEM-PARAFAC: An insight into the fate of allochthonous DOM in alpine lakes affected by climate change. Sci. Total Environ. 2016, 568, 216–225. [Google Scholar] [CrossRef] [PubMed]
- Zhu, W.-Z.; Yang, G.-P.; Zhang, H.-H. Photochemical behavior of dissolved and colloidal organic matter in estuarine and oceanic waters. Sci. Total Environ. 2017, 607–608, 214–224. [Google Scholar] [CrossRef] [PubMed]
- Shammi, M.; Pan, X.; Mostofa, K.M.G.; Zhang, D.; Liu, C.-Q. Photo-flocculation of microbial mat extracellular polymeric substances and their transformation into transparent exopolymer particles: Chemical and spectroscopic evidences. Sci. Rep. 2017, 7, 9074. [Google Scholar] [CrossRef] [PubMed]
- Song, W.; Zhao, C.; Mu, S.; Pan, X.; Zhang, D.; Al-Misned, F.A.; Mortuza, M.G. Effects of irradiation and pH on fluorescence properties and flocculation of extracellular polymeric substances from the cyanobacterium Chroococcus minutus. Coll. Surf. B Biointerfaces 2015, 128, 115–118. [Google Scholar] [CrossRef] [PubMed]
- Tonietto, A.E.; Lombardi, A.T.; Vieira, A.A.H. The effects of solar irradiation on copper speciation and organic complexation. J. Braz. Chem. Soc. 2011, 22, 1695–1700. [Google Scholar] [CrossRef]
- Vidali, R.; Remoundaki, E.; Tsezos, M. Humic acids copper binding following their photochemical alteration by simulated solar light. Aquat. Geochem. 2010, 16, 207–218. [Google Scholar] [CrossRef]
- Shank, G.C.; Whitehead, R.F.; Smith, M.L.; Skrabal, S.A.; Kieber, R.J. Photodegradation of strong copper complexing ligands in organic-rich estuarine waters. Limnol. Oceangr. 2006, 51, 884–892. [Google Scholar] [CrossRef]
- Sander, S.; Kim, J.P.; Anderson, B.; Hinter, K.A. Effect of UVB irradiation on Cu2+-binding organic ligands and Cu2+ speciation in alpine lake water of new zealand. Environ. Chem. 2005, 2, 56–62. [Google Scholar] [CrossRef]
- Lindim, C.; Mota, A.M.; Goncalves, M.L.S. Influence of UV-B irradiation in lead speciation from an estuarine sample. Water Res. 2000, 34, 3325. [Google Scholar] [CrossRef]
- Neron, R.; Auclair, J.C.; Fortin, C. Rate of Cd2+ release from dissolved fulvic acid and natural dissolved organic carbon as a function of UVB dose. Environ. Chem. 2006, 3, 433–438. [Google Scholar] [CrossRef]
- Worms, I.A.M.; Szigeti, Z.A.G.; Dubascoux, S.; Lespes, G.; Traber, J.; Sigg, L.; Slaveykova, V.I. Colloidal organic matter from wastewater treatment plant effluents: Characterization and role in metal distribution. Water Res. 2010, 44, 340–350. [Google Scholar] [CrossRef] [PubMed]
- Worms, I.A.M.; Traber, J.; Kistler, D.; Sigg, L.; Slaveykova, V.I. Uptake of Cd(ii) and Pb(ii) by microalgae in presence of colloidal organic matter from wastewater treatment plant effluents. Environ. Pollut. 2010, 158, 369–374. [Google Scholar] [CrossRef] [PubMed]
- Alasonati, E.; Slaveykova, V.I.; Gallard, H.; Croué, J.-P.; Benedetti, M.F. Characterization of the colloidal organic matter from the amazonian basin by asymmetrical flow field-flow fractionation and size exclusion chromatography. Water Res. 2010, 44, 223–231. [Google Scholar] [CrossRef] [PubMed]
- Batley, G.E.; Apte, S.C.; Stauber, J.L. Speciation and bioavailability of trace metals in water: Progress since 1982. Aust. J. Chem. 2004, 57, 903–919. [Google Scholar] [CrossRef]
- Sigg, L.; Black, F.; Buffle, J.; Cao, J.; Cleven, R.; Davison, W.; Galceran, J.; Gunkel, P.; Kalis, E.; Kistler, D.; et al. Comparison of analytical techniques for dynamic trace metal speciation in natural freshwaters. Environ. Sci. Technol. 2006, 40, 1934–1941. [Google Scholar] [CrossRef] [PubMed]
- Van Leeuwen, H.P.; Buffle, J. Chemodynamics of aquatic metal complexes: From small ligands to colloids. Environ. Sci. Technol. 2009, 43, 7175–7183. [Google Scholar] [CrossRef] [PubMed]
- Tercier-Waeber, M.-L.; Hezard, T.; Masson, M.; Schäfer, J.R. In situ monitoring of the diurnal cycling of dynamic metal species in a stream under contrasting photobenthic biofilm activity and hydrological conditions. Environ. Sci. Technol. 2009, 43, 7237–7244. [Google Scholar] [CrossRef] [PubMed]
- Winch, S.; Ridal, J.; Lean, D. Increased metal bioavailability following alteration of freshwater dissolved organic carbon by ultraviolet B radiation exposure. Environ. Toxicol. 2002, 17, 267–274. [Google Scholar] [CrossRef] [PubMed]
- Winch, S.; Lean, D. Comparison of changes in metal toxicity following exposure of water with high dissolved organic carbon content to solar, UV-B and UV-A radiation. Photochem. Photobiol. 2005, 81, 1469–1480. [Google Scholar] [CrossRef] [PubMed]
- Brooks, M.L.; Meyer, J.S.; Boese, C.J. Toxicity of copper to larval pimephales promelas in the presence of photodegraded natural dissolved organic matter. Can. J. Fish. Aquat. Sci. 2007, 64, 391–401. [Google Scholar] [CrossRef]
- Helbling, E.W.; Zagarese, H.; Kieber, D.J.; Peake, B.M.; Scully, N.M. Reactive oxygen species in aquatic ecosystems. UV effects in aquatic organisms and ecosystems. R. Soc. Chem. 2003, 1, 251–288. [Google Scholar]
- Passardi, F.; Cosio, C.; Penel, C.; Dunand, C. Peroxidases have more functions than a Swiss army knife. Plant. Cell Rep. 2005, 24, 255–265. [Google Scholar] [CrossRef] [PubMed]
- Kieber, R.J.; Skrabal, S.A.; Smith, C.; Willey, J.D. Redox speciation of copper in rainwater: Temporal variability and atmospheric deposition. Environ. Sci. Technol. 2004, 38, 3587–3594. [Google Scholar] [CrossRef] [PubMed]
- Deguillaume, L.; Leriche, M.; Desboeufs, K.; Mailhot, G.; George, C.; Chaumerliac, N. Transition metals in atmospheric liquid phases: Sources, reactivity, and sensitive parameters. Chem. Rev. 2005, 105, 3388–3431. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Garg, S.; Waite, T.D. Light-mediated reactive oxygen species generation and iron redox transformations in the presence of exudate from the cyanobacterium Microcystis aeruginosa. Environ. Sci. Technol. 2017, 51, 8384–8395. [Google Scholar] [CrossRef] [PubMed]
- Johannsson, O.E.; Smith, D.S.; Sadauskas-Henrique, H.; Cimprich, G.; Wood, C.M.; Val, A.L. Photo-oxidation processes, properties of doc, reactive oxygen species (ROS), and their potential impacts on native biota and carbon cycling in the Rio Negro (Amazonia, Brazil). Hydrobiologia 2017, 789, 7–29. [Google Scholar] [CrossRef]
- Zeng, J.; Wang, W.X. Temperature and irradiance influences on cadmium and zinc uptake and toxicity in a freshwater cyanobacterium, Microcystis aeruginosa. J. Hazard. Mater. 2011, 190, 922–929. [Google Scholar] [CrossRef] [PubMed]
- Xu, K.; Juneau, P. Different physiological and photosynthetic responses of three cyanobacterial strains to light and zinc. Aquat. Toxicol. 2016, 170, 251–258. [Google Scholar] [CrossRef] [PubMed]
- Xu, K.; Li, Z.-K.; Qiu, B.-S.; Juneau, P. Different responses to high light stress of toxic and non-toxic Microcystis aeruginosa acclimated under two light intensities and zinc concentrations. Toxicol. Environ. Chem. 2013, 95, 1145–1156. [Google Scholar] [CrossRef]
- Cheloni, G.; Cosio, C.; Slaveykova, V.I. Antagonistic and synergistic effects of light irradiation on the effects of copper on Chlamydomonas reinhardtii. Aquat. Toxicol. 2014, 155, 275–282. [Google Scholar] [CrossRef] [PubMed]
- Rai, L.C.; Tyagi, B.; Mallick, N.; Rai, P.K. Interactive effects of UV-B and copper on photosynthetic activity of the cyanobacterium Anabaena doliolum. Environ. Exp. Bot. 1995, 35, 177–185. [Google Scholar] [CrossRef]
- Manson, A.Z.; Jenkins, K.D. Metal detoxification in aquatic organisms. In Metal Speciation and Bioavailability in Aquatic Systems; Tessier, A., Turner, D.R., Eds.; Wiley: Hoboken, NJ, USA, 1992. [Google Scholar]
- Barón, M.; Arellano, J.B.; López Gorgé, J. Copper and photosystem II: A controversial relationship. Physiol. Plant. 1995, 94, 174–180. [Google Scholar] [CrossRef]
- Fernandes, J.C.; Henriques, F.S. Biochemical, physiological, and structural effects of excess copper in plants. Bot. Rev. 1991, 57, 246–273. [Google Scholar] [CrossRef]
- Juneau, A.; El Berdey, P.; Popovic, R. Pam fluorometry in the determination of the sensitivity of Chlorella vulgaris, Selenastrum capricornutum, and Chlamydomonas reinhardtii to copper. Arch. Environ. Contam. Toxicol. 2002, 42, 155–164. [Google Scholar] [CrossRef] [PubMed]
- Perales-Vela, H.V.; Gonzalez-Moreno, S.; Montes-Horcasitas, C.; Canizares-Villanueva, R.O. Growth, photosynthetic and respiratory responses to sub-lethal copper concentrations in Scenedesmus incrassatulus (chlorophyceae). Chemosphere 2007, 67, 2274–2281. [Google Scholar] [CrossRef] [PubMed]
- Mallick, N.; Mohn, F. Use of chlorophyll fluorescence in metal-stress research: A case study with the green microalga Scenedesmus. Ecotoxicol. Environ. Saf. 2003, 55, 64–69. [Google Scholar] [CrossRef]
- Franklin, N.M.; Stauber, J.L.; Lim, R.P. Development of flow cytometry-based algal bioassays for assessing toxicity of copper in natural waters. Environ. Toxicol. Chem. 2001, 20, 160–170. [Google Scholar] [CrossRef] [PubMed]
- Hadjoudja, S.; Vignoles, C.; Deluchat, V.; Lenain, J.F.; Le Jeune, A.H.; Baudu, M. Short term copper toxicity on Microcystis aeruginosa and Chlorella vulgaris using flow cytometry. Aquat. Toxicol. 2009, 94, 255–264. [Google Scholar] [CrossRef] [PubMed]
- Jamers, A.; Blust, R.; De Coen, W.; Griffin, J.L.; Jones, O.A. Copper toxicity in the microalga Chlamydomonas reinhardtii: An integrated approach. Biometals 2013, 26, 731–740. [Google Scholar] [CrossRef] [PubMed]
- Cirulis, J.T.; Scott, J.A.; Ross, G.M. Management of oxidative stress by microalgae. Can. J. Physiol. Pharmacol. 2013, 91, 15–21. [Google Scholar] [CrossRef] [PubMed]
- Pinto, E.; Sigaud-Kunter, T.C.S.; Leitao, M.A.S.; Okamoto, O.K.; Morse, D.; Colepicolo, P. Heavy metal-induced oxidative stress in algae. J. Phycol. 2003, 39, 1008–1018. [Google Scholar] [CrossRef]
- Von Moos, N.; Slaveykova, V.I. Oxidative stress induced by inorganic nanoparticles in bacteria and aquatic microalgae—State of the art and knowledge gaps. Nanotoxicology 2014, 8, 605–630. [Google Scholar] [CrossRef] [PubMed]
- Tripathi, B.N.; Mehta, S.K.; Amar, A.; Gaur, J.P. Oxidative stress in Scenedesmus sp. During short- and long-term exposure to Cu and Zn. Chemosphere 2006, 62, 538–544. [Google Scholar] [CrossRef] [PubMed]
- Stoiber, T.L.; Shafer, M.M.; Karner Perkins, D.A.; Hemming, J.D.C.; Armstrong, E.D. Analysis of glutathione endpoints for measuring copper stress in Chlamydomonas reinhardtii. Environ. Toxicol. Chem. 2007, 26, 1563–1571. [Google Scholar] [CrossRef] [PubMed]
- Stoiber, T.L.; Shafer, M.M.; Armstrong, D.E. Induction of reactive oxygen species in Chlamydomonas reinhardtii in response to contrasting trace metal exposures. Environ. Toxicol. 2013, 28, 516–523. [Google Scholar] [CrossRef] [PubMed]
- Szivák, I.; Behra, R.; Sigg, L. Metal-induced reactive oxygen species production in Chlamydomonas reinhardtii (chlorophyceae). J. Phycol. 2009, 45, 427–435. [Google Scholar] [CrossRef] [PubMed]
- Latifi, A.; Ruiz, M.; Zhang, C.C. Oxidative stress in cyanobacteria. FEMS Microbiol. Rev. 2009, 33, 258–278. [Google Scholar] [CrossRef] [PubMed]
- Witman, G.B. Chlamydomonas phototaxis. Trends Cell Biol. 1993, 3, 403–408. [Google Scholar] [CrossRef]
- Krause, G.H. Photoinhibition of photosynthesis. An evaluation of damaging and protective mechanisms. Physiol. Plant. 1988, 74, 566–574. [Google Scholar] [CrossRef]
- Hooper, M.J.; Ankley, G.T.; Cristol, D.A.; Maryoung, L.A.; Noyes, P.D.; Pinkerton, K.E. Interactions between chemical and climate stressors: A role for mechanistic toxicology in assessing climate change risks. Environ. Toxicol. Chem. 2013, 32, 32–48. [Google Scholar] [CrossRef] [PubMed]
- West, L.J.A.; Li, K.; Greenberg, B.M.; Mierle, G.; Smith, R.E.H. Combined effects of copper and ultraviolet radiation on a microscopic green alga in natural soft lake waters of varying dissolved organic carbon content. Aquat. Toxicol. 2003, 64, 39–52. [Google Scholar] [CrossRef]
- Prasad, S.M.; Zeeshan, M. UV-B radiation and cadmium induced changes in growth, photosynthesis, and antioxidant enzymes of cyanobacterium Plectonema boryanum. Biol. Plant. 2005, 49, 229–236. [Google Scholar] [CrossRef]
- Singh, R.; Srivastava, P.K.; Singh, V.P.; Dubey, G.; Prasad, S.M. Light intensity determines the extent of mercury toxicity in the cyanobacterium Nostoc muscorum. Acta Physiol. Plant. 2011, 34, 1119–1131. [Google Scholar] [CrossRef]
- Singh, P.K.; Rai, S.; Pandey, S.; Agrawal, C.; Shrivastava, A.K.; Kumar, S.; Rai, L.C. Cadmium and UV-B induced changes in proteome and some biochemical attributes of Anabaena sp. Pcc7120. Phykos 2012, 42, 39–50. [Google Scholar]
- Lee, W.M.; An, Y.J. Effects of zinc oxide and titanium dioxide nanoparticles on green algae under visible, UVA, and UVB irradiations: No evidence of enhanced algal toxicity under UV pre-irradiation. Chemosphere 2013, 91, 536–544. [Google Scholar] [CrossRef] [PubMed]
- Navarro, E.; Robinson, C.T.; Behra, R. Increased tolerance to ultraviolet radiation (UVR) and cotolerance to cadmium in UVR-acclimatized freshwater periphyton. Limnol. Oceanogr. 2008, 53, 1149–1158. [Google Scholar] [CrossRef]
- Corcoll, N.; Bonet, B.; Leira, M.; Montuelle, B.; Tlili, A.; Guasch, H. Light history influences the response of fluvial biofilms to Zn exposure. J. Phycol. 2012, 48, 1411–1423. [Google Scholar] [CrossRef] [PubMed]
- Hader, D.P.; Kumar, H.D.; Smith, R.C.; Worrest, R.C. Effects of solar UV radiation on aquatic ecosystems and interactions with climate change. Photochem. Photobiol. Sci. 2007, 6, 267–285. [Google Scholar] [CrossRef] [PubMed]
- Navarro, E.; Muniz, S.; Korkaric, M.; Wagner, B.; de Caceres, M.; Behra, R. Ultraviolet radiation dose calculation for algal suspensions using UVA and UVB extinction coefficients. J. Photochem. Photobiol. B 2014, 132, 94–101. [Google Scholar] [CrossRef] [PubMed]
- Harris, C.A.; Scott, A.P.; Johnson, A.C.; Panter, G.H.; Sheahan, D.; Roberts, M.; Sumpter, J.P. Principles of sound ecotoxicology. Environ. Sci. Technol. 2014, 48, 3100–3111. [Google Scholar] [CrossRef] [PubMed]
| Light | Metal | Organisms | Test Conditions | Effect | Reference |
|---|---|---|---|---|---|
| PAR | Zn Cd | M. aeruginosa | Light (18, 32, 55 µmol photons m−2 s−1) and temperature (15, 24, 30 °C) acclimated cultures exposed to Zn (4.4 × 10−11 M to 1.4 × 10−7 M) and Cd (5.3 × 10−11 M to 3.3 × 10−7 M) for 4 h and 72 h | ↑ | [72] |
| PAR | Hg | Nostoc muscorum | 72 h exposure to Hg under different levels of visible light (10, 70, 130 µmol photons m−2 s−1) | ↑ | [98] |
| PAR | Zn | M. aeruginosa CPCC299 M. aeruginosa CPCC632 | Light (50 and 500 µmol photons m−2 s−1) and Zn (7.8 × 10−7 M and 7.8 × 10−6 M) acclimated cultures exposed to 2000 µmol photons m−2 s−1 for 20 min | ↑ | [74] |
| PAR | Zn | M. aeruginosa CPCC299 M. aeruginosa CPCC632 Synechocystis sp. | Light (50 and 300 µmol photons m−2 s−1) acclimated cultures exposed for 4 h 30 min to Zn (0.78, 7.8 and 39 µM) | ↑ M = Sy | [73] |
| PAR | Zn | Periphytic communities | Light acclimated periphyton (25, 100, 500 µmol photons m−2 s−1) exposed for 6 h to 10, 25, 50, 100, 250, and 500 µmol photons m−2 s−1 and to Zn 1.5 mg L−1 | (LL) ↑↑ (ML,HL) ↑ | [103] |
| PAR, UVA, UVB | Cu | C. reinhardtii | Cultures exposed to light (artificial light: PAR 13.3 W m−2, UVAR 0.29 W m−2 and UVBR 0.08 W m−2; solar simulated light: PAR 230 W m−2, UVAR 9.75 W m−2 and UVBR 0.73 W m−2; solar simulated light with enhanced UVB: PAR 232.2 W m−2, UVAR 10.01 W m−2 and UVBR 2.01 W m−2) and Cu2+ 0.06, 0.6 and 6 µM | (HL) A (UVB) S | [75] |
| UVB | Cd | Plectonema boryanum | Cultures pre-incubated for 2 h with Cd (2 and 8 µM) and exposed to 20 µmol m−2 s−1 and 0.4 W m−2 UVB (0.4 Wm−2) for 30 and 90 min | S | [98] |
| UVB | Cd | Anabaena sp. | Combined exposure to 10 µM Cd and 30 min UVB (9.6 kJ m−2) | A | [100] |
| UVA+ UVB | Cd | C. reinhadtii | 48 h exposure to 100 ± 10 µmol m−2 s−1 with UVR (13 Wm−2 UVA; 0.5 Wm−2 UVB) or without UVR. | S | [7] |
| UVA+ UVB | Cd | Periphytic communities | After 38, 52, and 66 days acclimation to high UVR (686 ± 40 µmol photons s−1 m−2 PAR, 10.88 ± 1.9 mW cm−2 UVA, and 1.03 ± 0.11 mW cm−2 UVB) and low UVR (633 ± 31 µmol photons s−1 m−2 PAR, 3.61 ± 0.48 mWcm−2 UVA, 0.07 ± 0.007 mW cm−2 UVB) periphyton were exposed to 2, 10, 20, 40, and 60 µM Cd for 2 h | Co-T | [102] |
| UV-B | Cu | A. doliolum | Combined exposure to UV-B (12.9 mW m −2 nm −1) and Cu (0.3 and 0.5 mg L−1) for 12 and 25 min | S | [76] |
| PAR UVA UVB | Cu | Pseudokirchneriella subcapitata | 7 days exposure to copper in high DOM or low DOM natural water under PAR; PAR + UVA, PAR + UVA + UVB. 14:10 light dark cycle | = | [97] |
| UVA UVB | Pb Cu Ni Cd | P. subcapitata | Natural fresh water pre irradiated with UVA (16.14 W m−2) and UVB (3.4 W m−2) for up to 20 days and then used to perform toxicity tests | ↑ | [64] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cheloni, G.; Slaveykova, V.I. Combined Effects of Trace Metals and Light on Photosynthetic Microorganisms in Aquatic Environment. Environments 2018, 5, 81. https://doi.org/10.3390/environments5070081
Cheloni G, Slaveykova VI. Combined Effects of Trace Metals and Light on Photosynthetic Microorganisms in Aquatic Environment. Environments. 2018; 5(7):81. https://doi.org/10.3390/environments5070081
Chicago/Turabian StyleCheloni, Giulia, and Vera I. Slaveykova. 2018. "Combined Effects of Trace Metals and Light on Photosynthetic Microorganisms in Aquatic Environment" Environments 5, no. 7: 81. https://doi.org/10.3390/environments5070081
APA StyleCheloni, G., & Slaveykova, V. I. (2018). Combined Effects of Trace Metals and Light on Photosynthetic Microorganisms in Aquatic Environment. Environments, 5(7), 81. https://doi.org/10.3390/environments5070081





