Performance of Reverse Osmosis Membranes in the Treatment of Flue-Gas Desulfurization (FGD) Wastewaters
Abstract
1. Introduction
2. Materials and Methods
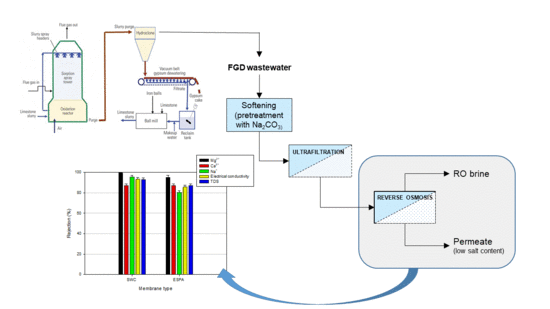
2.1. Softening and Pre-Treatment Processes of FGD Wastewaters
2.2. Treatment of Permeate UF by RO: Experimental Set-Up and Membranes
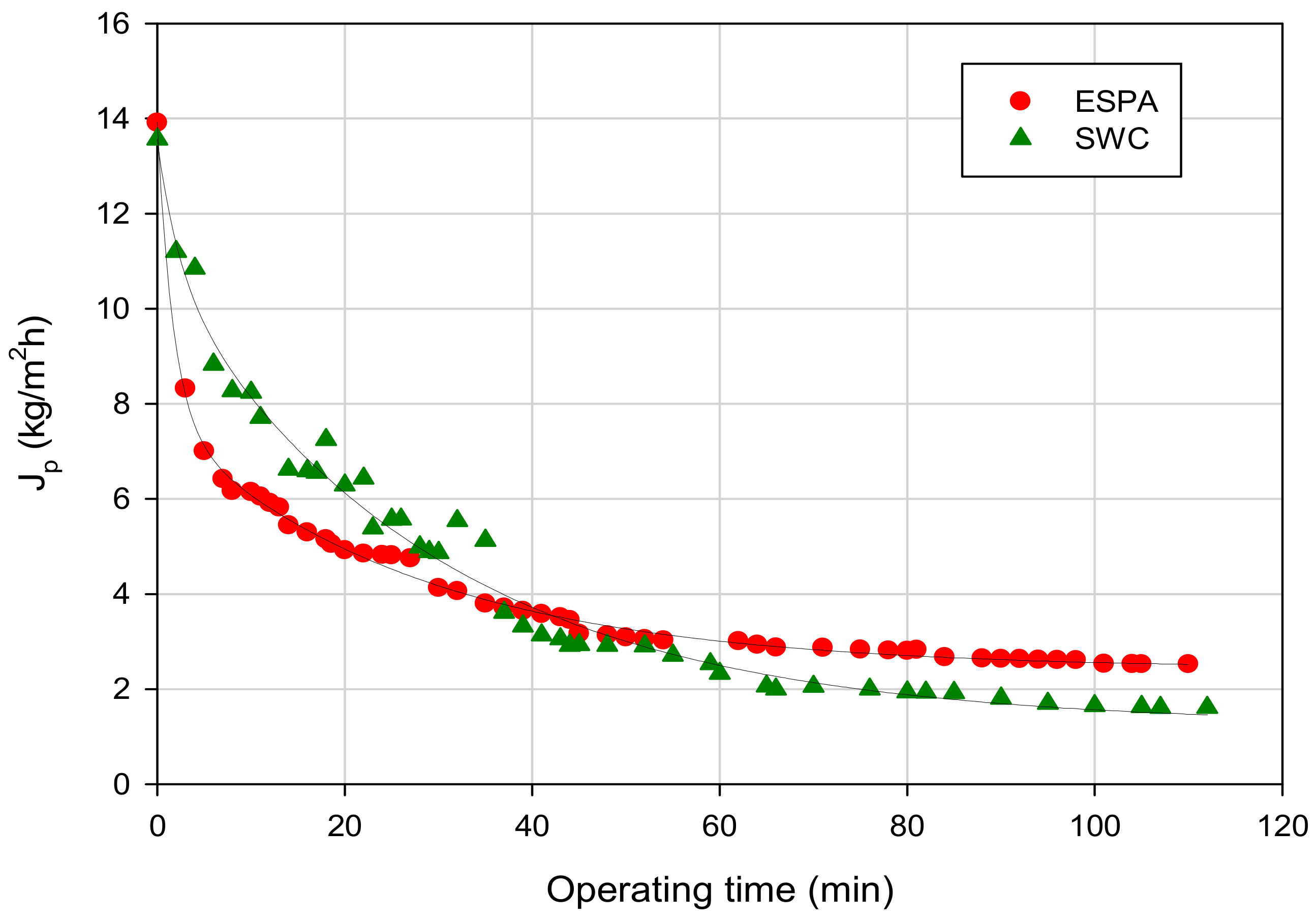
2.3. Permeate Fluxes, Hydraulic Permeability, Fouling Index and Cleaning Efficiency
2.4. Analytical Methods
3. Results and Discussion
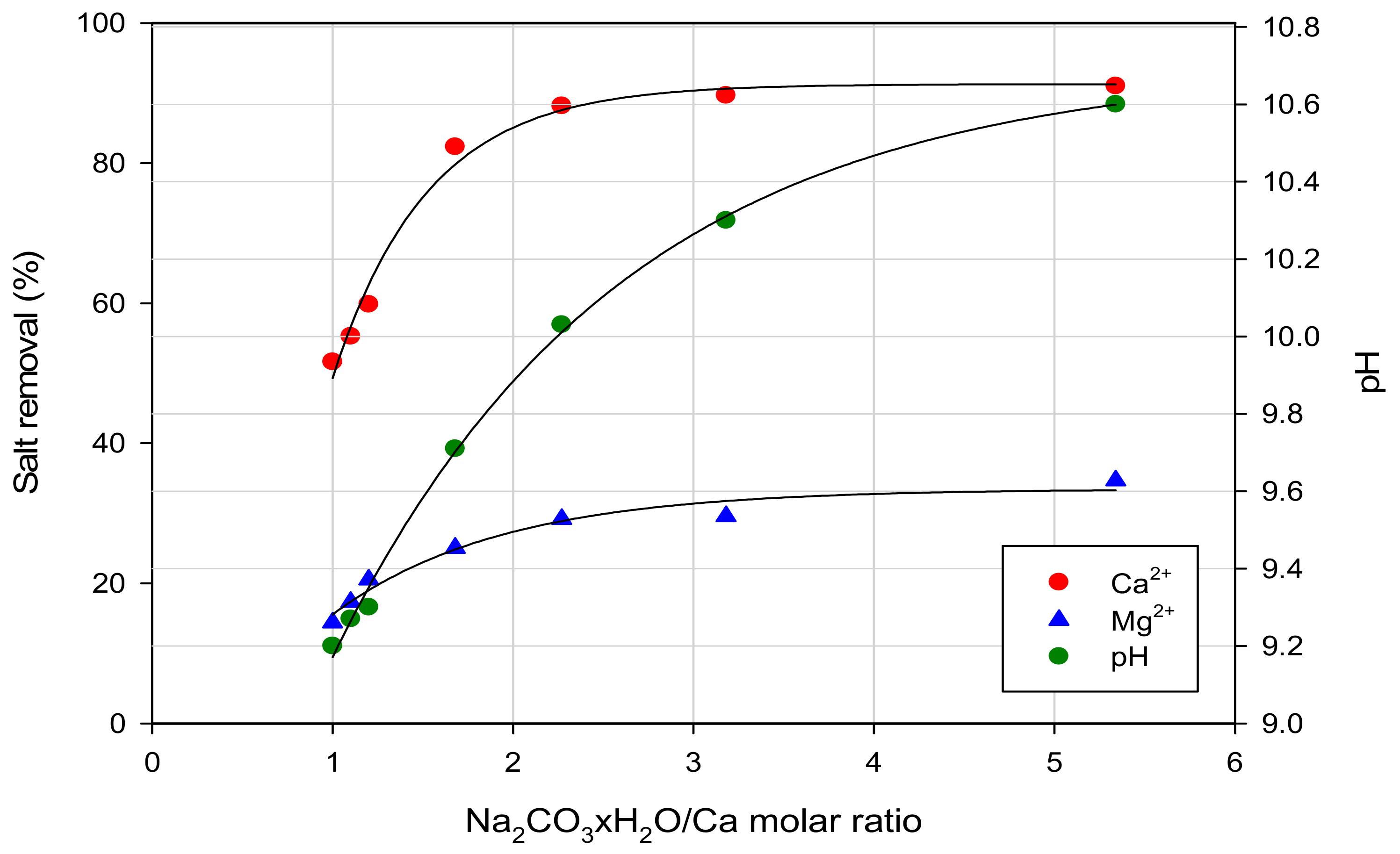
3.1. Pre-Treatment Processes
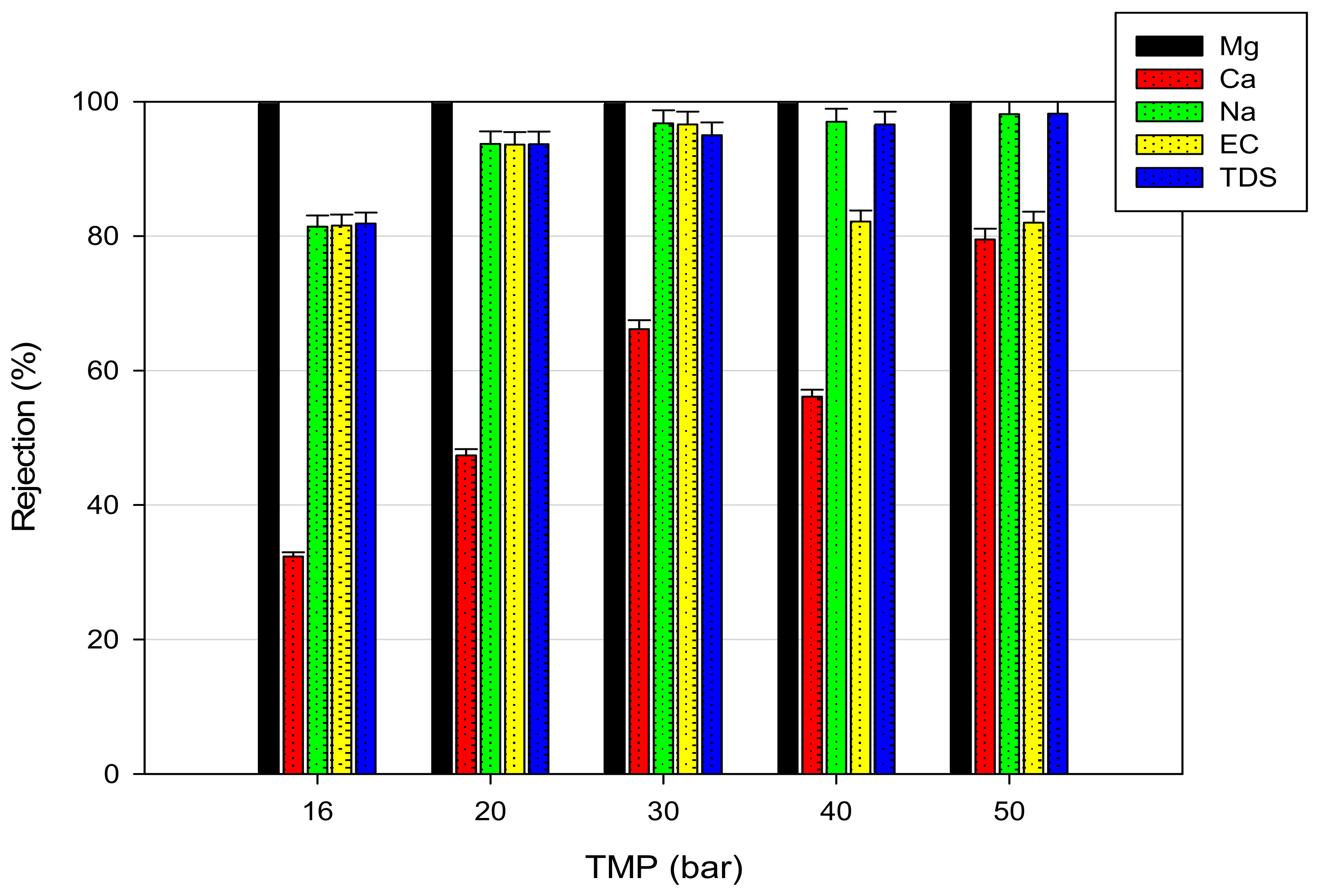
3.2. RO of Pre-Treated FGD Wastewaters
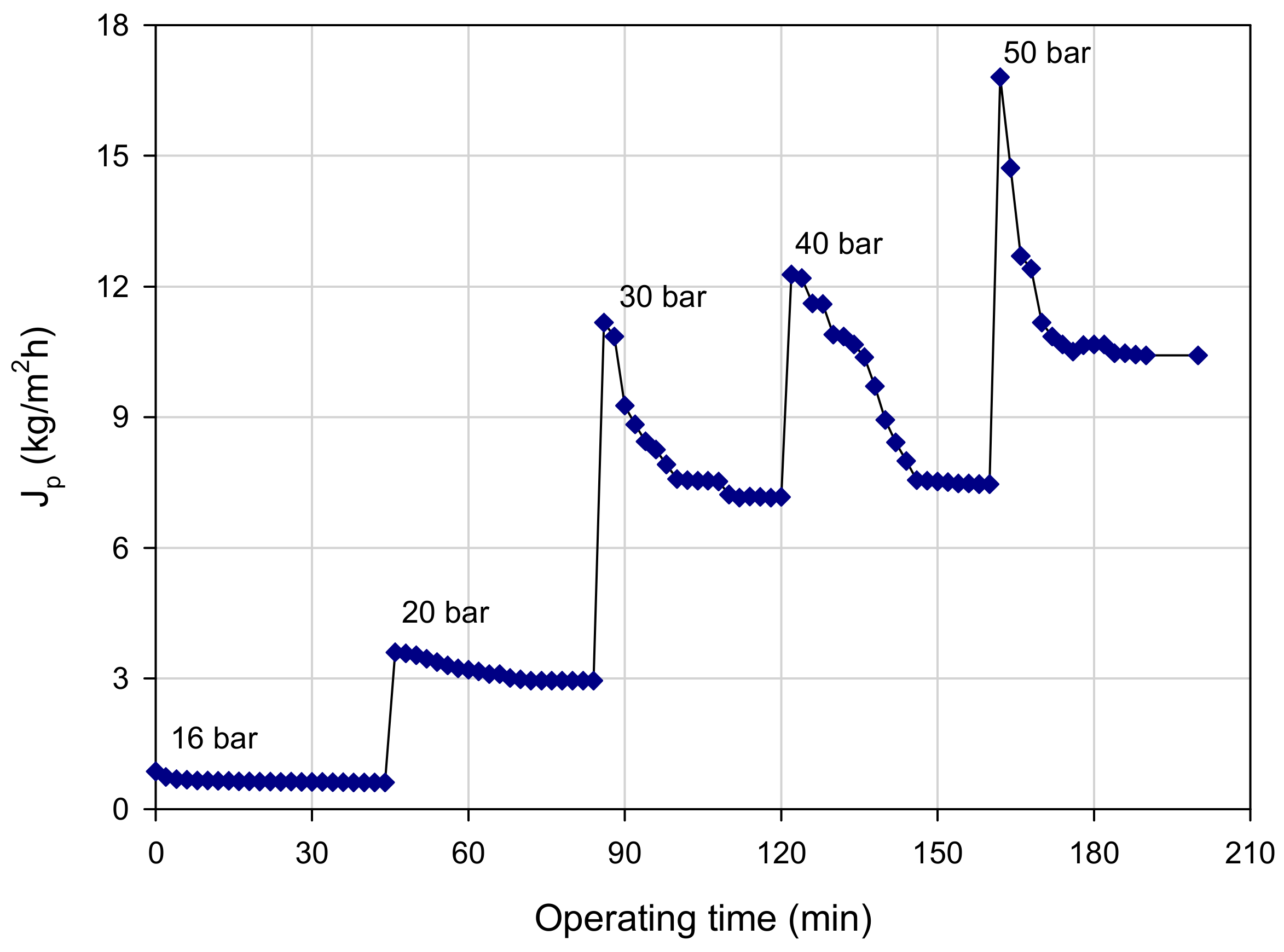
3.3. Experiments in Total Recycle Mode
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- USEPA. Steam Electric Power Generating Point Source Category: Final Detailed Study Report. EPA 821-R-09-08; 2009. Available online: https://www.epa.gov/sites/production/files/2015-06/documents/steam-electric_detailed_study_report_2009.pdf (accessed on 26 April 2018).
- Pickett, T.; Sonstegard, J.; Bonkoski, B. Using biology to treat selenium. Power Eng. 2008, 110, 140–145. [Google Scholar]
- Wei, C.; He, W.J.; Wei, L.; Ma, J.; Li, C.Y. The performance and microbial communities of biodegradation-electron transfer with sulfur metabolism integrated process for flue gas desulfurization wastewater treatment. Bioprocess. Biosyst. Eng. 2017, 40, 1543–1553. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Lin, X.J.; Wang, J.T.; Jiang, F.; Wei, L.; Chen, G.H.; Hao, X.D. Effects of lead and mercury on sulfate-reducing bacterial activity in a biological process for flue gas desulfurization wastewater treatment. Sci. Rep. 2016, 6, 30455. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.B.; Ni, W.M.; Guan, B.H. Application of chitosan as flocculant for coprecipitation of Mn(II) and suspended solids from dual-alkali FGD regenerating process. J. Hazard. Mater. 2008, 152, 757–764. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.H.; Peddi, P.K.; Tang, C.L.; Zeng, H.; Teng, X.J. Hybrid zero-valent iron process for removing heavy metals and nitrate from flue-gas-desulfurization wastewater. Sep. Purif. Technol. 2013, 118, 690–698. [Google Scholar] [CrossRef]
- Sundberg-Jones, S.E.; Hassan, S.M. Macrophyte sorption and bioconcentration of elements in a pilot constructed wetland for flue gas desulfurization wastewater treatment. Water Air Soil Pollut. 2007, 183, 187–200. [Google Scholar] [CrossRef]
- Ma, S.C.; Chai, J.; Chen, G.D.; Yu, W.J.; Zhu, S.J. Research on desulfurization wastewater evaporation: Present and future perspectives. Renew. Sustain. Energy Rev. 2016, 58, 1143–1151. [Google Scholar]
- Yu, J.; Lu, J.; Kang, Y. Removal of sulfate from wet FGD wastewater by co-precipitation with calcium hydroxide and sodium aluminate. Water Sci. Technol. 2018, 77, 1336–1345. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.N.; Ye, X.K.; He, K.; Chen, Y.C.; Hu, Y.Y. Simultaneous removal of Ni(II) and fluoride from a real flue gas desulfurization wastewater by electrocoagulation using Fe/C/Al electrode. J. Water Reuse Desalin. 2017, 7, 288–297. [Google Scholar] [CrossRef]
- López, J.; Coello, M.D.; Quiroga, J.M. Comparative studies of reverse osmosis membranes for wastewater reclamation. Desalination 2006, 191, 137–147. [Google Scholar] [CrossRef]
- Van der Bruggen, B.; Vandecasteele, C. Removal of pollutants from surface water and groundwater by nanofiltration: Overview of possible applications in the drinking water industry. Environ. Pollut. 2003, 122, 435–445. [Google Scholar] [CrossRef]
- Malaeb, L.; Ayoub, G.M. Reverse osmosis technology for water treatment: State of the art review. Desalination 2011, 267, 1–8. [Google Scholar] [CrossRef]
- Liu, C.; Farooq, K.; Doll, B.; Venkatadri, R. Economical and reliable mercury reduction in refinery and power plant discharge wastewater with robust microfiltration membrane technology. Desalin. Water Treat. 2013, 51, 4980–4986. [Google Scholar] [CrossRef]
- Jia, F.; Wang, J. Treatment of flue gas desulfurization wastewater with near-zero liquid discharge by nanofiltration-membrane distillation process. Sep. Sci. Technol. 2018, 53, 146–153. [Google Scholar] [CrossRef]
- Li, Q.; Song, J.; Yu, H.; Li, Z.K.; Pan, X.H.; Yang, B. Investigating the microstructures and surface features of seawater RO membranes and the dependencies of fouling resistance performances. Desalination 2014, 352, 109–117. [Google Scholar] [CrossRef]
- Dražević, E.; Košutić, K.; Fingler, S.; Drevenkar, V. Removal of pesticides from the water and their adsorption on the reverse osmosis membranes of defined porous structure. Desalin. Water Treat. 2011, 30, 161–170. [Google Scholar] [CrossRef]
- Lee, S.; Lee, E.; Elimelech, M.; Hong, S. Membrane characterization by dynamic hysteresis: Measurements, mechanisms, and implications for membrane fouling. J. Membr. Sci. 2011, 366, 17–24. [Google Scholar] [CrossRef]
- Ishida, K.P.; Bold, R.; Phipps, D.W. Identification and Evaluation of Unique Chemicals for Optimum Membrane Compatibility and Improved Cleaning Efficiency; Technical Report April; Orange County Water District: Fountain Valley, CA, USA, 2005. [Google Scholar]
- Ayoub, G.M.; Zayyar, R.M.; Al-Hindi, M. Precipitation softening: A pre-treatment process for seawater desalination. Environ. Sci. Pollut. Res. 2014, 21, 2876–2887. [Google Scholar] [CrossRef] [PubMed]
- Zheng, L.; Wang, X.; Wang, X. Reuse of reverse osmosis concentrate in textile and dyeing industry by combined process of persulfate oxidation and lime-soda softening. J. Clean. Prod. 2015, 108, 525–533. [Google Scholar] [CrossRef]
- Teixeira, A.R.S.; Santos, J.L.C.; Crespo, J.C. Sustainable membrane-based process for valorization of cork boiling wastewaters. Sep. Purif. Technol. 2009, 66, 35–44. [Google Scholar] [CrossRef]
- Vourch, M.; Balannec, B.; Chaufer, B.; Dorange, G. Treatment of dairy industry wastewater by reverse osmosis for water reuse. Desalination 2008, 219, 190–202. [Google Scholar] [CrossRef]
- Dolar, D.; Košutić, K.; Strmecky, T. Hybrid processes for treatment of landfill leachate: Coagulation/UF/NF-RO and adsorption/UF/NF-RO. Sep. Purif. Technol. 2016, 168, 39–46. [Google Scholar] [CrossRef]
- Tang, C.Y.; Fu, Q.S.; Robertson, A.P.; Criddle, C.; Leckie, J.O. Use of reverse osmosis to remove perfluorooctane sulfonate (PFOS) from semiconductor wastewater. Environ. Sci. Technol. 2006, 40, 7343–7349. [Google Scholar] [CrossRef] [PubMed]
- Jiang, S.; Li, Y.; Ladewig, B.P. A review of reverse osmosis membrane fouling and control strategies. Sci. Total Environ. 2017, 595, 567–583. [Google Scholar] [CrossRef] [PubMed]
- Sert, G.; Bunani, S.; Yörükoğlu, E.; Kabay, N.; Egemen, Ö.; Arda, M.; Yüksel, M. Performances of some NF and RO membranes for desalination of MBR treated wastewater. J. Water Proc. Eng. 2017, 16, 193–198. [Google Scholar] [CrossRef]
- Mohammadi, T.; Moghadam, M.K.; Madaeni, S.S. Hydrodynamic factors affecting flux and fouling during reverse osmosis of seawater. Desalination 2002, 151, 239–245. [Google Scholar] [CrossRef]
- Mondal, S.; Wickramasinghe, S.R. Produced water treatment by nanofiltration and reverse osmosis membranes. J. Membr. Sci. 2008, 322, 162–170. [Google Scholar] [CrossRef]
- Liu, M.; Lü, Z.; Chen, Z.; Yu, S.; Gao, C. Comparison of reverse osmosis and nanofiltration membranes in the treatment of biologically treated textile effluent for water reuse. Desalination 2011, 281, 372–378. [Google Scholar] [CrossRef]
- Goh, P.S.; Lau, W.J.; Othman, M.H.D.; Ismail, A.F. Membrane fouling in desalination and its mitigation strategies. Desalination 2018, 425, 130–155. [Google Scholar] [CrossRef]
- Nataraj, S.K.; Hosamani, K.M.; Aminabhavi, T.M. Nanofiltration and reverse osmosis thin film composite membrane module for the removal of dye and salts from the simulated mixtures. Desalination 2009, 249, 12–17. [Google Scholar] [CrossRef]

| Membrane Type | SWC-2540 | ESPA-2540 |
|---|---|---|
| Manufacturer | Hydranautics | Hydranautics |
| Membrane material | Composite polyamide | Composite polyamide |
| Salt rejection (%) | 99.4 (minimum 99) | 99.4 (minimum 98) |
| pH operating range | 2–11 | 2–10 |
| Max. operating temperature (°C) | 45 | 45 |
| Max. operating pressure (bar) | 69 | 21 |
| Membrane surface area (m2) | 2.34 | 2.34 |
| Water permeability (kg/m2hbar) | 1.77 a | 5.90 a |
| Contact angle (°) | 58.9 ± 1.2 b | 34.0 ± 2.00 c |
| Zeta potential (mV) | −21.2 at pH 7 d | −26.0 at pH 7.0 e |
| Sample | Ca2+ (ppm) | Mg2+ (ppm) | Na+ (g/L) | EC (mS/cm) | TDS (g/L) | TC (mg/L) | TOC (mg/L) | IC (mg/L) |
|---|---|---|---|---|---|---|---|---|
| FGD wastewater | 384.4 ± 4.8 | 289.4 ± 2.6 | 7.2 ± 0.6 | 33.6 ± 2.1 | 16.9 ± 0.6 | - | - | - |
| After softening | 83.8 ± 0.5 | 174.7 ± 1.2 | 6.4 ± 0.8 | 25.08 ± 1.6 | 12.46 ± 1.8 | 90.96 ± 0.91 | 90.12 ± 0.90 | 0.57 ± 0.05 |
| After ultrafiltration | 82.0 ± 1.6 | 171.8 ± 3.4 | 6.3 ± 0.12 | 25.8 ± 0.5 | 12.5 ± 0.2 | 82.98 ± 0.83 | 33.80 ± 0.34 | 49.18 ± 0.49 |
| Parameter | Membrane Type | |
|---|---|---|
| SWC-2540 | ESPA-2540 | |
| Wp0 (kg/m2hbar) | 1.7 | 5.9 |
| Wp1 (kg/m2hbar) | 1.1 | 4.2 |
| Wp2 (kg/m2hbar) | 1.6 | 5.2 |
| Wp3 (kg/m2hbar) | 1.7 | 5.4 |
| Fouling index (FI) (%) | 35.3 | 28.8 |
| Cleaning efficiency (CE) (%) | 100.0 | 92.0 |
| Membrane Type | Sample | Ca2+ (ppm) | Mg2+ (ppm) | Na+ (g/L) | EC (mS/cm) | TDS (g/L) |
|---|---|---|---|---|---|---|
| SWC-2540 | Feed | 83.5 ± 1.7 | 178.4 ± 3.5 | 6.4 ± 0.1 | 26.1 ± 0.52 | 12.6 ± 0.25 |
| Permeate | 11.0 ± 0.2 | n.d. | 0.29 ± 0.01 | 1.76 ± 0.03 | 0.87 ± 0.01 | |
| Retentate | 159.2 ± 3.4 | 362.2 ± 7.8 | 12.52 ± 0.26 | 49.3 ± 1.0 | 24.6 ± 0.5 | |
| ESPA-2540 | Feed | 52.1 ± 1.03 | 196.2 ± 3.9 | 6.4 ± 0.1 | 25.1 ± 0.5 | 13.9 ± 0.3 |
| Permeate | 6.75 ± 0.13 | 6.8 ± 0.13 | 1.25 ± 0.02 | 3.6 ± 0.2 | 1.8 ± 0.04 | |
| Retentate | 98.6 ± 2.2 | 383.6 ± 7.8 | 11.5 ± 0.25 | 45.6 ± 2.3 | 22.8 ± 0.7 |
| Parameter | Feed | Permeate | Retentate | Balance | ||
|---|---|---|---|---|---|---|
| Volume | 18 L | 9 L | 50.0% | 9 L | 50.0% | 100.0% |
| Ca2+ | 1503 mg | 99.0 mg | 6.5% | 1432.8 mg | 95.3% | 101.8% |
| Mg2+ | 3211.2 mg | 0 mg | 0.0% | 3259.8 g | 101.5% | 101.5% |
| Na+ | 115.2 mg | 2.6 mg | 2.2% | 112.7 g | 97.8% | 100.0% |
| Parameter | Feed | Permeate | Retentate | Balance | ||
|---|---|---|---|---|---|---|
| Volume | 18 L | 9 L | 50.0% | 9 L | 50.0% | 100.0% |
| Ca2+ | 937.8 mg | 60.7 mg | 6.5% | 887.4 mg | 94.6% | 101.1% |
| Mg2+ | 3531.6 mg | 61.2 mg | 1.7% | 3452.4 g | 97.7% | 99.4% |
| Na+ | 115.2 mg | 11.2 mg | 9.7% | 103.5 g | 89.9% | 99.6% |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Conidi, C.; Macedonio, F.; Argurio, P.; Cassano, A.; Drioli, E. Performance of Reverse Osmosis Membranes in the Treatment of Flue-Gas Desulfurization (FGD) Wastewaters. Environments 2018, 5, 71. https://doi.org/10.3390/environments5060071
Conidi C, Macedonio F, Argurio P, Cassano A, Drioli E. Performance of Reverse Osmosis Membranes in the Treatment of Flue-Gas Desulfurization (FGD) Wastewaters. Environments. 2018; 5(6):71. https://doi.org/10.3390/environments5060071
Chicago/Turabian StyleConidi, Carmela, Francesca Macedonio, Pietro Argurio, Alfredo Cassano, and Enrico Drioli. 2018. "Performance of Reverse Osmosis Membranes in the Treatment of Flue-Gas Desulfurization (FGD) Wastewaters" Environments 5, no. 6: 71. https://doi.org/10.3390/environments5060071
APA StyleConidi, C., Macedonio, F., Argurio, P., Cassano, A., & Drioli, E. (2018). Performance of Reverse Osmosis Membranes in the Treatment of Flue-Gas Desulfurization (FGD) Wastewaters. Environments, 5(6), 71. https://doi.org/10.3390/environments5060071