Optimisation of Growth of Raphidocelis subcapitata Immobilised for Biofuel Production: Influence of Alginate and CaCl2 Concentrations on Growth
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Microalga and Culture Medium
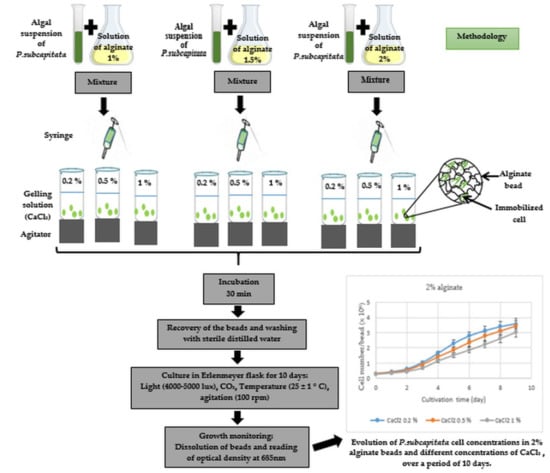
2.3. Algae Immobilization
2.4. Culture Conditions
2.5. Growth Monitoring
2.6. Calculation of Growth Rate and Generation Time
3. Results and Discussion
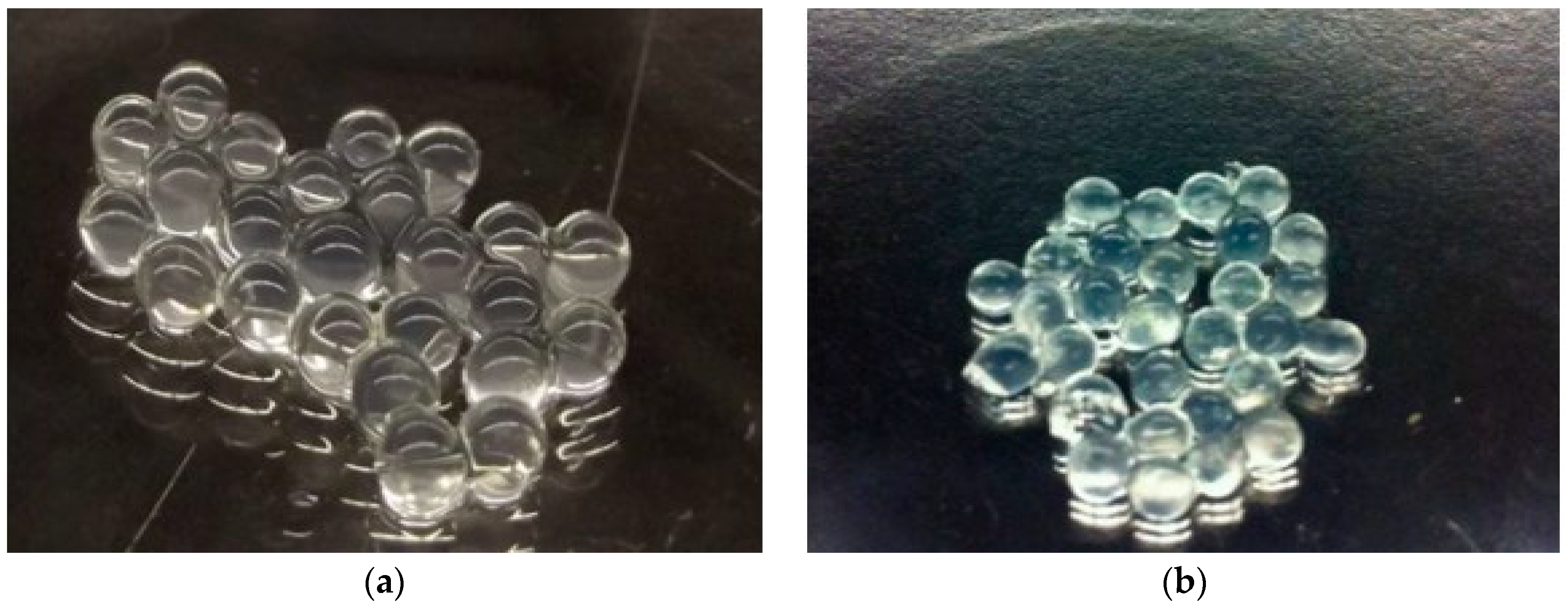
3.1. Beads Analysis
3.2. Effects of Alginate and CaCl2 Concentrations on R. subcapitata Growth
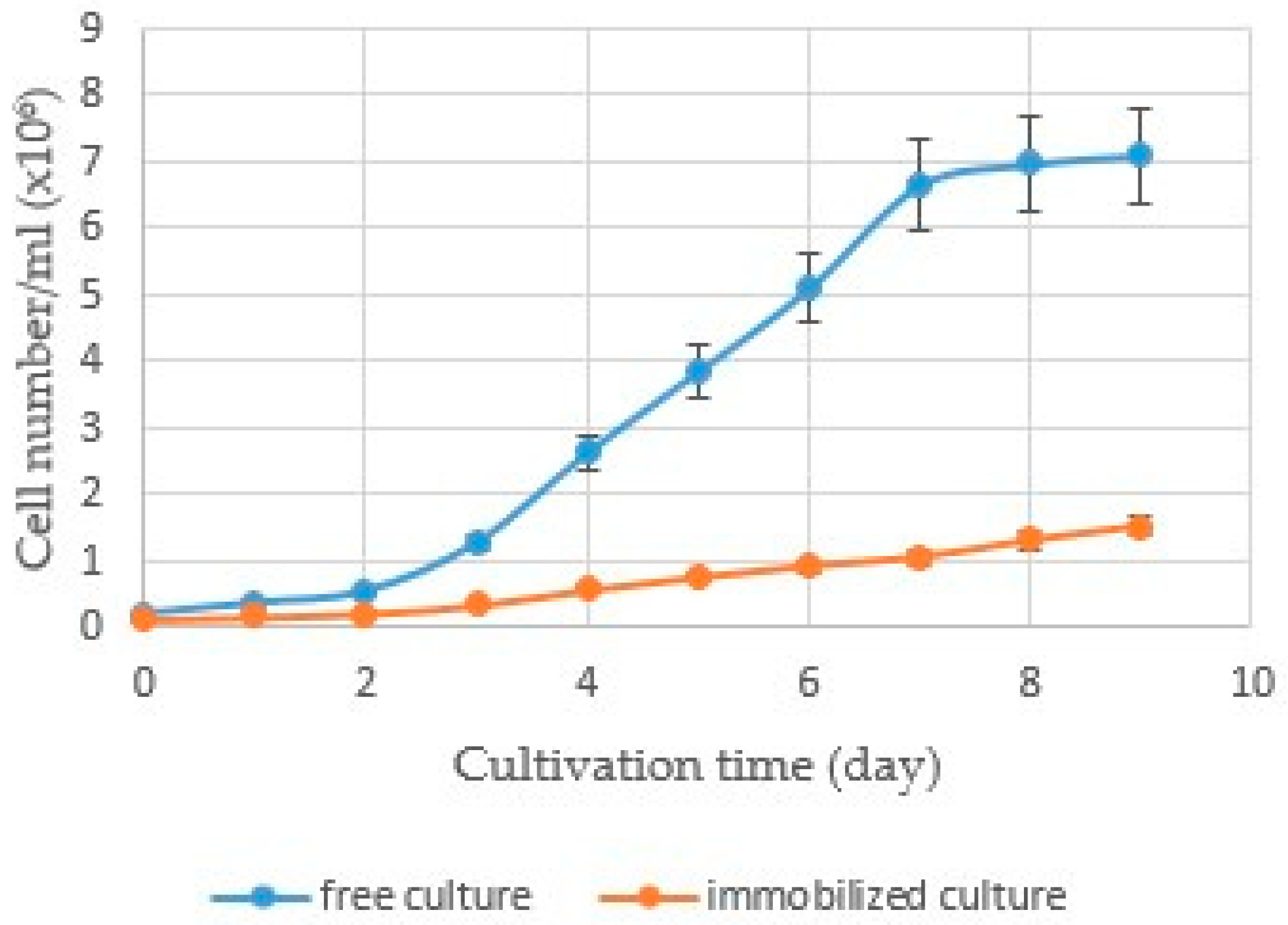
3.3. Comparison of Free and Immobilized R. subcapitata Growth in Alginate Beads
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Labriet, M.; Waaub, J.-P.; Prades, J.A. Stratégies de gestion des gaz à effet de serre au Québec: Grandes lignes et enseignements d’une recherche interdisciplinaire rapport de recherche. Nat. Sci. Soc. 2000, 8, 68–75. [Google Scholar] [CrossRef]
- Petit, M. L’humanité face à l’effet de serre additionnel qu’elle crée. C. R. Acad. Sci. Ser. IIA Earth Planet. Sci. 2001, 333, 775–786. [Google Scholar] [CrossRef]
- André, J.-C. Sur la crédibilité des conséquences de l’effet de serre. C. R. Geosci. 2003, 335, 503–507. [Google Scholar] [CrossRef]
- Tissot, B. Quel avenir pour les combustibles fossiles? Les avancées scientifiques et technologiques permettront-elles la poursuite d’un développement soutenable avec les énergies carbonées? C. R. Acad. Sci. Ser. IIA Earth Planet. Sci. 2001, 333, 787–796. [Google Scholar] [CrossRef]
- Lorius, C. Effet de serre: Les lacunes du savoir et de la perception. C. R. Geosci. 2003, 335, 545–549. [Google Scholar] [CrossRef]
- Gavrilescu, M.; Chisti, Y. Biotechnology—A sustainable alternative for chemical industry. Biotechnol. Adv. 2005, 23, 471–499. [Google Scholar] [CrossRef] [PubMed]
- Chisti, Y. Biodiesel from microalgae. Biotechnol. Adv. 2007, 25, 294–306. [Google Scholar] [CrossRef] [PubMed]
- Khan, S.A.; Rashmi; Hussain, M.Z.; Prasad, S.; Banerjee, U.C. Prospects of biodiesel production from microalgae in India. Renew. Sustain. Energy Rev. 2009, 13, 2361–2372. [Google Scholar] [CrossRef]
- Brennan, L.; Owende, P. Biofuels from microalgae—A review of technologies for production, processing, and extractions of biofuels and co-products. Renew. Sustain. Energy Rev. 2010, 14, 557–577. [Google Scholar] [CrossRef]
- Lam, M.K.; Lee, K.T. Microalgae biofuels: A critical review of issues, problems and the way forward. Biotechnol. Adv. 2012, 30, 673–690. [Google Scholar] [CrossRef] [PubMed]
- Cantin, I. La Production de Biodiesel à Partir des Microalgues Ayant un Métabolisme Hétérotrophe; Centre Universitaire de Formation en Environnement Université de Sherbrooke: Québec, QC, Canada, 2010. [Google Scholar]
- Kadam, K.L. Environmental implications of power generation via coal-microalgae cofiring. Energy 2002, 27, 905–922. [Google Scholar] [CrossRef]
- Doré-Deschênes, F. Utilisation des Microalgues Comme Source D’énergie Durable; Université de Sherbrooke: Québec, QC, Canada, 2009. [Google Scholar]
- Molino, A.; Larocca, V.; Chianese, S.; Musmarra, D. Biofuels Production by Biomass Gasification: A Review. Energies 2018, 11, 811. [Google Scholar] [CrossRef]
- Sialve, B.; Bernet, N.; Bernard, O. Anaerobic digestion of microalgae as a necessary step to make microalgal biodiesel sustainable. Biotechnol. Adv. 2009, 27, 409–416. [Google Scholar] [CrossRef] [PubMed]
- Carver, S.M.; Hulatt, C.J.; Thomas, D.N.; Tuovinen, O.H. Thermophilic, anaerobic co-digestion of microalgal biomass and cellulose for H2 production. Biodegradation 2011, 22, 805–814. [Google Scholar] [CrossRef] [PubMed]
- Mussgnug, H.; Klassen, V.; Schlüter, A.; Kruse, O. Microalgae as substrates for fermentative biogas production in a combined biorefinery concept. J. Biotechnol. 2010, 150, 51–56. [Google Scholar] [CrossRef] [PubMed]
- Scott, S.A.; Davey, M.P.; Dennis, J.S.; Horst, I.; Howe, C.J.; Lea-Smith, D.J.; Smith, A.G. Biodiesel from algae: Challenges and prospects. Curr. Opin. Biotechnol. 2010, 21, 277–286. [Google Scholar] [CrossRef] [PubMed]
- Pienkos, P.T.; Darzins, A. The promise and challenges of microalgal-derived biofuels. Biofuels Bioprod. Biorefin. 2009, 3, 431–440. [Google Scholar] [CrossRef]
- Lee, J.-Y.; Yoo, C.; Jun, S.-Y.; Ahn, C.-Y.; Oh, H.-M. Comparison of several methods for effective lipid extraction from microalgae. Bioresour. Technol. 2010, 101, S75–S77. [Google Scholar] [CrossRef] [PubMed]
- Suali, E.; Sarbatly, R. Conversion of microalgae to biofuel. Renew. Sustain. Energy Rev. 2012, 16, 4316–4342. [Google Scholar] [CrossRef]
- Sander, K.; Murthy, G.S. Life cycle analysis of algae biodiesel. Int. J. Life Cycle Assess. 2010, 15, 704–714. [Google Scholar] [CrossRef]
- Moreira, S.M.; Moreira-Santos, M.; Guilhermino, L.; Ribeiro, R. Immobilization of the marine microalga Phaeodactylum tricornutum in alginate for in situ experiments: Bead stability and suitability. Enzyme Microb. Technol. 2006, 38, 135–141. [Google Scholar] [CrossRef]
- Cao, Y.R.; Liu, Z.; Cheng, G.L.; Jing, X.B.; Xu, H. Exploring single and multi-metal biosorption by immobilized spent Tricholoma lobayense using multi-step response surface methodology. Chem. Eng. J. 2010, 164, 183–195. [Google Scholar] [CrossRef]
- Moreno-Garrido, I. Microalgae immobilization: Current techniques and uses. Bioresour. Technol. 2008, 99, 3949–3964. [Google Scholar] [CrossRef] [PubMed]
- Dulieu, C.; Poncelet, D.; Neufeld, R.J. Encapsulation and immobilization techniques. Cell Encapsul. Technol. Ther. 1999, 1, 1–17. [Google Scholar]
- Draget, K.I.; Steinsvåg, K.; Onsøyen, E.; Smidsrød, O. Na- and K-alginate; effect on Ca2+-gelation. Carbohydr. Polym. 1998, 35, 1–6. [Google Scholar] [CrossRef]
- Ruiz-Marin, A.; Mendoza-Espinosa, L.G. Ammonia removal and biomass characteristics of alginate-immobilized Scenedesmus obliquus cultures treating real wastewater. Fresenius Environ. Bull. 2008, 17, 1236–1241. [Google Scholar]
- Lopez, B.R.; Hernandez, J.-P.; Bashan, Y.; de-Bashan, L.E. Immobilization of microalgae cells in alginate facilitates isolation of DNA and RNA. J. Microbiol. Methods 2017, 135, 96–104. [Google Scholar] [CrossRef] [PubMed]
- Moreno-Garrido, I.; Campana, O.; Lubián, L.M.; Blasco, J. Calcium alginate immobilized marine microalgae: Experiments on growth and short-term heavy metal accumulation. Mar. Pollut. Bull. 2005, 51, 823–829. [Google Scholar] [CrossRef] [PubMed]
- Antal, T.K.; Kukarskikh, G.P.; Volgusheva, A.A.; Krendeleva, T.E.; Tyystjärvi, E.; Rubin, A.B. Hydrogen photoproduction by immobilized S-deprived Chlamydomonas reinhardtii: Effect of light intensity and spectrum, and initial medium pH. Algal Res. 2016, 17, 38–45. [Google Scholar] [CrossRef]
- Amsden, B. Solute diffusion in hydrogels: An examination of the retardation effect. Polym. Gels Netw. 1998, 6, 13–43. [Google Scholar] [CrossRef]
- Patil, V.; Källqvist, T.; Olsen, E.; Vogt, G.; Gislerød, H.R. Fatty acid composition of 12 microalgae for possible use in aquaculture feed. Aquac. Int. 2007, 15, 1–9. [Google Scholar] [CrossRef]
- Gonçalves, A.L.; Pires, J.C.; Simões, M. Lipid production of Chlorella vulgaris and Pseudokirchneriella subcapitata. Int. J. Energy Environ. Eng. 2013, 4, 14. [Google Scholar] [CrossRef]
- Pires, J.C.M.; Gonçalves, A.L.; Martins, M.C.M.; Alvim-Ferraz, M.; Simões, M. Effect of light supply on CO2 capture from atmosphere by Chlorella vulgaris and Pseudokirchneriella subcapitata. Mitig. Adapt. Strateg. Glob. Chang. 2014, 19, 1109–1117. [Google Scholar] [CrossRef]
- Polona, S.; Marija, B.; Anamarija, Z.; Odon, P.; Ales, M. Shape optimization and characterization of polysaccharide beads prepared by ionotropic gelation. J. Microencapsul. 2007, 25, 90–105. [Google Scholar]
- Panouillé, M.; Larreta-Garde, V. Gelation behaviour of gelatin and alginate mixtures. Food Hydrocoll. 2009, 23, 1074–1080. [Google Scholar] [CrossRef]
- Khromova, Y.L. The effect of chlorides on alginate gelation in the presence of calcium sulfate. Colloid J. 2006, 68, 115–119. [Google Scholar] [CrossRef]
- Série de la Protection de L’environnement. Méthode D’essai Biologique: Essai D’inhibition de la Croissance d’une Algue d’eau Douce. Centre des Sciences et Technologies Environnementales; Environnement et Changement Climatique Canada: Ottawa, ON, Canada, 2007.
- Santos, M.M.D.; Moreno-Garrido, I.; Gonçalves, F.; Soares, A.M.; Ribeiro, R. An in situ bioassay for estuarine environments using the microalga Phaeodactylum tricornutum. Environ. Toxicol. Chem. 2002, 21, 567–574. [Google Scholar] [CrossRef] [PubMed]
- Voo, W.-P.; Lee, B.-B.; Idris, A.; Islam, A.; Tey, B.-T.; Chan, E.-S. Production of ultra-high concentration calcium alginate beads with prolonged dissolution profile. RSC Adv. 2015, 5, 36687–36695. [Google Scholar] [CrossRef]
- Richmond, A. Handbook of Microalgal Culture: Biotechnology and Applied Phycology; John Wiley & Sons: Hoboken, NJ, USA, 2008. [Google Scholar]
- Boyaval, P.; Lebrun, A.; Goulet, J. Etude de l’immobilisation de Lactobacillus helveticus dans des billes d’alginate de calcium. Le Lait 1985, 65, 185–199. [Google Scholar] [CrossRef]
- Prevost, H.; Divies, C.; Rousseau, E. Continuous yoghurt production with Lactobacillus bulgaricus and Streptococcus thermophilus entrapped in Ca-alginate. Biotechnol. Lett. 1985, 7, 247–252. [Google Scholar] [CrossRef]
- Prevost, H.; Divies, C. Fresh fermented cheese production with continuous pre-fermented milk by a mixed culture of mesophilic lactic streptococci entrapped in Ca-Al ginate. Biotechnol. Lett. 1987, 9, 789–794. [Google Scholar] [CrossRef]
- Zhang, L.J.; Ying, G.G.; Chen, F.; Zhao, J.L.; Wang, L.; Fang, Y.X. Development and application of whole-sediment toxicity test using immobilized freshwater microalgae Pseudokirchneriella subcapitata. Environ. Toxicol. Chem. 2012, 31, 377–386. [Google Scholar] [CrossRef] [PubMed]



| Alginate% (w/v) | CaCl2% (w/v) | ||
|---|---|---|---|
| 0.2 | 0.5 | 1 | |
| 1 | 3.0 ± 0.2 | 2.5 ± 0.1 | 2.0 ± 0.1 |
| 1.5 | 3.5 ± 0.2 | 3.0 ± 0.1 | 2.5 ± 0.1 |
| 2 | 3.8 ± 0.2 | 3.5 ± 0.2 | 3.0 ± 0.1 |
| 2.5 | 3.8 ± 0.2 | 3.8 ± 0.2 | 3.5 ± 0.2 |
| 3 | - | - | - |
| Alginate% (w/v) | CaCl2% (w/v) | ||
|---|---|---|---|
| 0.2 | 0.5 | 1 | |
| 1 | 0.83 ± 0.08 | 1.21 ± 0.12 | 1.48 ± 0.35 |
| 1.5 | 3.92 ± 0.39 | 2.74±0.27 | 2.62 ± 0.26 |
| 2 | 3.58 ± 0.35 | 3.46 ± 0.34 | 3.04 ± 0.3 |
| Alginate% (w/v) | CaCl2% (w/v) | ||
|---|---|---|---|
| 0.2 | 0.5 | 1 | |
| 1 | 1.54 ± 0.15 | 1.28 ± 0.12 | 1.0 ± 0.1 |
| 1.5 | 0.8 ± 0.08 | 0.34±0.03 | 0.21 ± 0.02 |
| 2 | 0.27±0.02 | 0.16 ± 0.01 | 0.12 ± 0.01 |
| CaCl2% (w/v) | Alginate% (w/v) | |||||
|---|---|---|---|---|---|---|
| 1 | 1.5 | 2 | ||||
| µmax | G | µmax | G | µmax | G | |
| 0.2 | 0.53 ± 0.05 | 1.30 ± 0.13 | 0.27 ± 0.02 | 2.55 ± 0.25 | 0.25 ± 0.02 | 2.76 ± 0.27 |
| 0.5 | 0.49 ± 0.04 | 1.40 ± 0.14 | 0.22 ± 0.02 | 3.13 ± 0.31 | 0.22 ± 0.02 | 3.13 ± 0.31 |
| 1 | 0.17 ± 0.01 | 4.05 ± 0.40 | 0.19 ± 0.02 | 3.63 ± 0.36 | 0.20 ± 0.02 | 3.13 ± 0.31 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Benasla, A.; Hausler, R. Optimisation of Growth of Raphidocelis subcapitata Immobilised for Biofuel Production: Influence of Alginate and CaCl2 Concentrations on Growth. Environments 2018, 5, 60. https://doi.org/10.3390/environments5050060
Benasla A, Hausler R. Optimisation of Growth of Raphidocelis subcapitata Immobilised for Biofuel Production: Influence of Alginate and CaCl2 Concentrations on Growth. Environments. 2018; 5(5):60. https://doi.org/10.3390/environments5050060
Chicago/Turabian StyleBenasla, Amel, and Robert Hausler. 2018. "Optimisation of Growth of Raphidocelis subcapitata Immobilised for Biofuel Production: Influence of Alginate and CaCl2 Concentrations on Growth" Environments 5, no. 5: 60. https://doi.org/10.3390/environments5050060
APA StyleBenasla, A., & Hausler, R. (2018). Optimisation of Growth of Raphidocelis subcapitata Immobilised for Biofuel Production: Influence of Alginate and CaCl2 Concentrations on Growth. Environments, 5(5), 60. https://doi.org/10.3390/environments5050060