1. Introduction
In the last decade, the replacement of televisions (TVs) with Cathode Ray Tube (CRT) glass by flat-panel displays caused a drastic increase of Waste Electrical and Electronic Equipment (WEEE) characterized by a high lead and barium content. Discarded computer monitors and television sets are identified as hazardous materials due to the high content of lead in their cathode ray tubes with the role of protection from X-ray radiation [
1]. In biological systems, indeed, heavy metals can induce multiple organ damage. They are able to interact with several organelles and components of cells (e.g., membrane, mitochondrial, enzymes, DNA, and nuclear proteins), leading to DNA damage and conformational changes which cause cell cycle modulation, carcinogenesis, and/or apoptosis [
2]. Today, the disposal of WEEE glass has become a serious environmental issue. A high amount of obsolete electronics including TV and CRT monitors are in storage because appropriate electronic waste (e-waste) management and remediation technologies are insufficient. In this context, there is an increasing urgency to develop new applications for recovered WEEE glass in agreement with the European Community (EC) Directives 2002/96/EC regarding the recyclability; 2003/108/EC on WEEE management; and 2002/95/EC on the restriction of the use of certain hazardous substances in electrical and electronic equipment (RoHS). Due to the amorphous nature of glass, in this paper authors propose the use of WEEE glass in geopolymer preparation, and show a deepened role played by glass in the interaction with geopolymeric gel. The geopolymeric matrix chosen for this research is based on commercial metakaolin and a low-cost alkali source. These mixtures consolidate at room temperature, offering a chemically and mechanically stable paste to which different types of solid or liquid wastes can be added [
3,
4].
The glass used in the formulation of geopolymers was obtained from cathode ray tube glass coming from funnel and panel parts of the equipment. As those are heavy metal-containing materials, the potential toxicity of the obtained geopolymers was evaluated by investigating the geopolymer metal release ability. In addition, in order to assess whether the amount of released metals could pose a risk to human health, a cytotoxicity assay was carried out. In the literature, no specific studies on the cytotoxicity of geopolymers containing waste were found; authors are aware of papers published only on cement for dental application [
5,
6], glasses [
7], materials for shore protection [
8], and traditional building materials [
9]. Moreover, some studies [
10,
11] are reported which evaluate the potential application of geopolymers in the biomedical field. However, their clinical use is hindered by Al release. The cytotoxicity of Al, as well as that of other metal ions, is due to the ability to form complexes within the cells, close to DNA, which can catalyse DNA oxidation and thus lead to the DNA damage. However, metals ion toxicity is concentration-dependent [
12]. Therefore, to reduce Al release, the use of raw materials with ratio of Si/Al = 31 was successfully proposed [
10,
11].
Other authors have already studied the role of different kind of glass cullet in geopolymers, such as glass containers coming from urban collection [
13], bottle glass [
14], fluorescent lamps [
15], liquid crystal display glass [
16], glasses derived from the vitrification of municipal residues [
17], and waste from demolitions [
18], demonstrating the feasibility of using recycled glass in geopolymers. To the authors’ knowledge, no papers focused on CRT glasses were found. Concerning geopolymers, the innovative aspect of these kind of aluminosilicate materials is related to the possibility of hardening at room temperature without high treatment temperature and consequently reduced CO
2 emissions, representing an eco-friendly innovative alternative to cement both for new products and for the immobilization of wastes. For geopolymers preparation, low-cost materials can be used, including waste materials that are not currently reused in other industrial sectors but are abundant and urgently need to be disposed, such as incinerator bottom ash, ladle slag, red mud, electroplating sludge, mine tailings, etc. [
19,
20,
21,
22]. In this preliminary research, the authors decided to use metakaolin as a reference material.
2. Materials and Methods
2.1. Materials Used
The metakaolin used in the synthesis of geopolymer specimens, as the principal source of aluminosilicate, was produced by the calcination of kaolinite at 700 °C for 4 h and was then ground and sieved <75 µm. A solutions mixture of sodium hydroxide and sodium silicate with a volume ratio of 1:1 was used as an alkaline solution. The sodium hydroxide solution was prepared by dissolving pellets (99.6 wt %, Carlo Erba, Italy) in distilled water to reach 8 M concentration. The sodium silicate solution used was a viscous liquid produced for the cement industry (Ingessil, Verona, Italy) with molar SiO2/Na2O = 3 and weight ratio = 2.89 (26.45 wt % of SiO2 and 9.14 wt % of Na2O), as well as a density of 1.38 g/cm3 and 39.44 Baume scales.
WEEE glasses were used for this research, in particular a glass rich in Ba and Sr, named VBa, and funnel glass rich in Pb, named VFNL. These glasses come from the dismantling plant of TV and personal computer (PC) kinescopes.
The as-received glass was dry-ground in a fast laboratory ball mill and sieved through 125- and 45-µm filters in order to evaluate the effect of grain size. Geopolymers were prepared with a composition of 60 wt % metakaolin and 40 wt % of different kinds of glass; samples were named GVBa and GVFNL. The formulation of mixtures was designed to obtain Si/Al and Na/Al ratios of 2.16 and 1.01, respectively. The paste was poured into a plastic mold and the curing step was conducted at room temperature for 7, 14, and 28 days. This last curing time was investigated for samples containing glass with a fine grain size only.
2.2. Microstructural Analysis
Microstructure observations were conducted by environmental scanning electron microscope (ESEM) (ESEM-Quanta200-FEI, Eindhoven, The Netherlands) equipped with energy dispersive spectroscopy (EDS) to evaluate both the formation of geopolymeric amorphous phase and the presence of unreacted particles of waste glass in the hardened samples at different curing times. Before SEM analysis, the sample was coated with an Au-Pd sputtered layer.
2.3. pH and Conductivity Analysis
pH and conductivity measurements, over a period of 24 h in stirring water and with a solid/liquid ratio of 1/10, were performed in order to indirectly evaluate ions release.
2.4. Chemical Analysis of Glasses and Eluates
The chemical analysis of glasses was performed by Inductively coupled plasma atomic emission spectroscopy (ICP-AES Varian Liberty 200). The sample was dissolved in HNO3 solution.
The metal content of all samples was investigated by microwave-assisted wet digestion using a 3:1 mixture of HNO3 and HCl. The determination was made using an inductively coupled plasma-emission spectrometer (ICPE-9000 Shimadzu, Tokyo, Japan).
2.5. Leaching Test
The ability of leaching heavy metals of all samples was carried out according to the European standard EN 12457 “Characterization of waste-leaching-compliance test for leaching of granular waste materials and sludge”. Samples, crushed and sieved to the particle size under 2 mm, were placed in bidistilled water with a liquid volume (L)/solid weight (kg) ratio of 10:1 and maintained for 24 h. After the extraction and filtration of the leachates, heavy metal ions concentrations were determined by ICPE (ICPE-9000 Shimadzu, Tokyo, Japan). Before ICPE analysis, the samples were acidified with HNO3 to pH = 2.
2.6. Cell Culture
NIH-3T3 (National Institutes of Health—3 day transfer, inoculum 3 × 105 cells) murine fibroblast cell line was purchased from ATCC (American Type Culture Collection). The cell line was grown in RPMI (Roswell Park Memorial Institute) 1640 high glucose medium supplemented with 10% fetal bovine serum, 50.0 U/mL penicillin, and 100.0 μg/mL streptomycin, at 37 °C in a humidified atmosphere containing 5% CO2.
2.7. Cytotoxicity Assessment
Cytotoxicity evaluation was performed using the MTT (3-(4,5-dimethyl-2-thiazolyl)-2,5-diphenyl-2H-tetrazolium bromide) cell viability assay on the murine fibroblast NIH-3T3 cell line. The yellow tetrazolium MTT is reduced by metabolically active cells, in part by the action of dehydrogenase enzymes, to generate reducing equivalents such as the reduced form of the nicotinamide adenine dinucleotide (NAD + hydrogen or NADH) and the Nicotinamide adenine dinucleotide phosphate (NADPH). The resulting intracellular purple formazan is in a direct ratio to the number of living cells. The cells were maintained at 37 °C under 5% CO2 and 95% air in a humidified incubator and were grown in Dulbecco’s Modified Eagle Medium supplemented with 10% (v/v) fetal bovine serum (FBS) and 1% Pen-strep. Cells were seeded into 24-well plates at a density of 1.0 × 105 cells/well. After 24 h of incubation, cells were treated with the powders of each synthesized material (1.0 and 2.0 mg) for 48 h. Afterwards the medium was removed, the cells were washed three times using PBS (Phosphate Buffered Saline), and then treated with MTT solution in a fresh medium (500 μL; 0.50 mg/mL) and incubated for 2 h at 37 °C in 5% CO2. The quantitative evaluation of the formazan amount was performed by a fluorescence and absorbance reader (VictorIII Perkin Elmer, Waltham, MA, USA) measuring the UV absorbance at 570 nm. Cell viability was expressed as a percentage of the mitochondrial redox activity of the cells treated with the samples compared to an untreated control, considered as 100% cell viability. The results were expressed as the mean value of three determinations ± SD.
3. Results and Discussion
The chemical analysis (
Table 1) and the ability of metals leaching (
Table 2) of all samples were evaluated by ICP analysis. The results of glass chemical analysis (
Table 1) confirmed that the VFNL was rich in Pb and alkaline metals (Na
2O + K
2O about 15 wt %), with lower amounts of alkaline-earth metals (CaO + MgO + SrO + BaO around 7 wt %) and traces of Zn and Sb. On the other hand, VBa was rich in Ba and Sr (for a total of about 19 wt %), and alkaline metals (Na
2O + K
2O about 15 wt %), with very low amounts of alkaline earth oxides CaO and MgO and traces of Zn, Pb, and Sb.
In order to evaluate the effect of grain size, the grain size distribution curves are reported for Pb glass ground and sieved <45 µm and <125 µm. From the curves shown in
Figure 1, it appears evident that glass sieved <45 µm shows a higher fraction <10 µm (26% in volume) with respect to glass sieved <125 µm with a corresponding fraction of 7% in volume. Glass <45 µm has 50% of particles less than 19 µm and 99% under 82 µm. Glass <125 µm has 50% of particles less than 82 µm and 99% under 214 µm. For both types of glass, a fraction larger with respect to the sieve meshes is present.
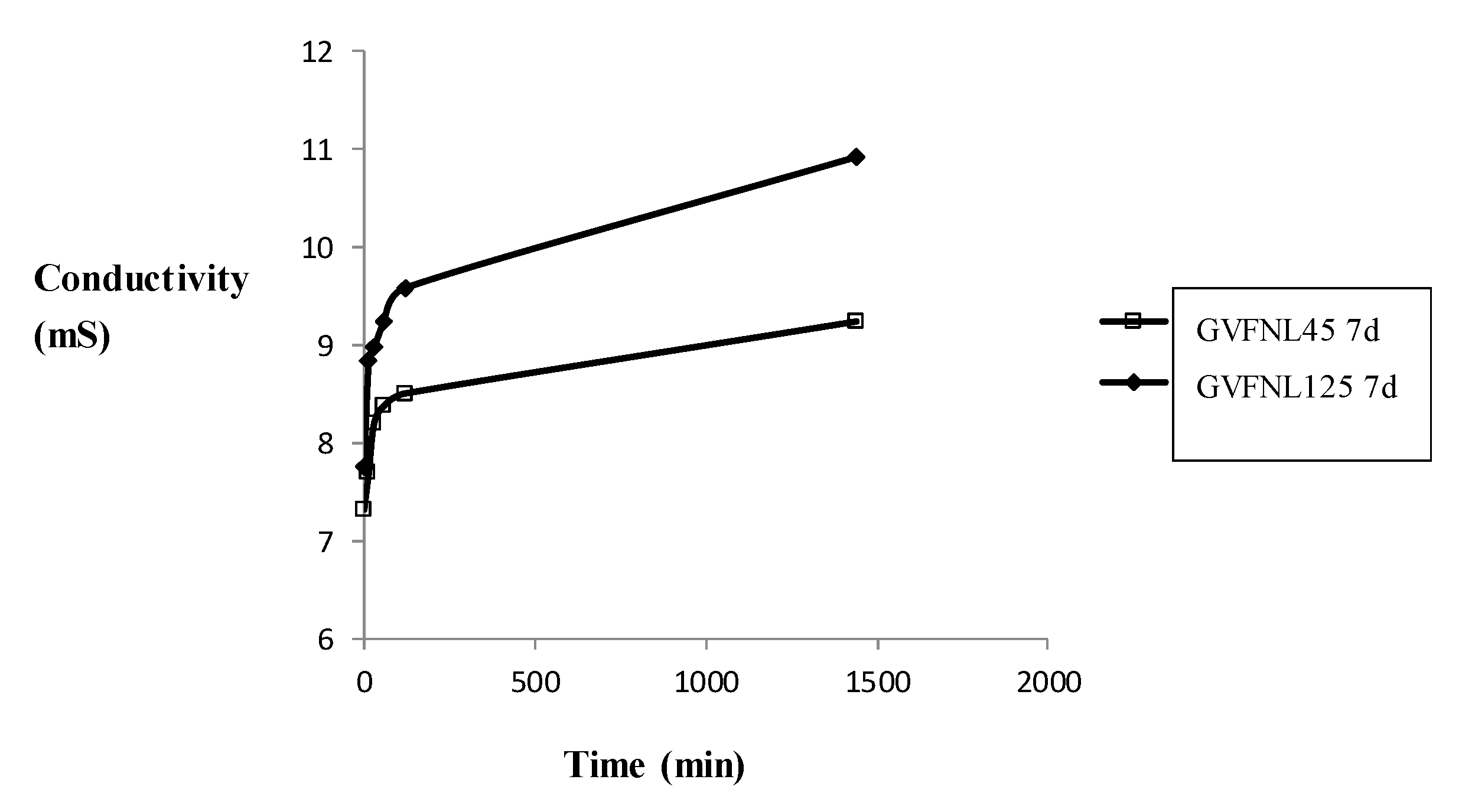
Geopolymeric samples were subjected to pH and conductivity tests in order to evaluate the occurrence of the geopolymerization process as a function of curing time. From
Figure 2, the decrease of pH corresponding to the increase of curing time is evident. This decrease is imputable to the decrease of free ions in the network, corresponding to a higher degree of geopolymerization. For glass containing Ba, the results are confirmed. Analyzing the pH trend for geopolymers containing glass with a large grain size (125 µm), the results are similar, with a decrease of pH value occurring as a function of aging time. This confirms the influence of curing time on the geopolymer properties. The different behavior of large and fine grain size glass is also evidenced by the conductivity values, which show lower values for samples containing a fine grain size, corresponding to a lower amount of ions in the eluates (
Figure 3). These results are confirmed by the microstructural analysis of the samples in
Figure 4.
In
Figure 4a,b, SEM micrographs of geopolymers containing VFNL glass with the two different grain sizes are shown after a curing time of 14 days. It can be observed that the microstructure of the sample with the larger grain size is coarser if compared to the sample with the fine grain size. In both samples, unreacted particles of glass are still evident, probably due to the scarce reactivity of the glassy network. While 45 µm particles do not hinder the geopolymerization process, as the matrix is better consolidated, the presence of larger particles leads to a geopolymer with a heterogeneous microstructure.
Figure 4c highlights that, notwithstanding the presence of unreacted particles, the formed geopolymeric gel is homogeneous. On the contrary,
Figure 4d shows, in a more detailed way, the heterogeneity of the material obtained using glass with a particle size <125 µm, it appears as a compacted powder more than a consolidated material.
The results of the leaching test (
Table 2), obtained on stable samples aged 28 days, prove that the waste glass containing geopolymers, in particular GVFNL, releases a lower amount of Al than pure geopolymer (GP). This can be explained by the increase of the silica content due to the addition of glass. In the literature [
10], indeed, it is reported that geopolymer with a high Si/Al content releases a lower content of Al.
Concerning the release of heavy metals from geopolymers by comparing the law limits for landfill for not dangerous wastes, it appears evident that Zn- and Ba-leaching species are lower than the law limits, while Sb is slightly higher. Similar results have been previously reported [
23,
24,
25,
26,
27]. The behavior of Sb is due to the fact that this element can form oxyanions, and the higher leachability of these oxyanions is related to the alkaline conditions typical of the geopolymer, and to the elevated sodium levels with respect to calcium. Oxyanions form much more soluble compounds with sodium than with calcium [
24], for example:
Oxyanions have a negative oxidation state; they cannot balance the negative charge of the geopolymer matrix, and thus are not strongly immobilized in the matrix. The immobilization of oxyanions containing antimony in geopolymer GVBa, and less relevantly in GVFNL, might occur by physical entrapment rather than chemical entrapment due to the low porosity of geopolymers. In conclusion, concerning antimony, the geopolymeric matrix has a good stabilizing efficiency.
The Pb concentration in the leachate from the geopolymers is higher than the law limit; however, considering the high amount contained in the VFNL glass, the % efficiency of immobilization is still very high. This behavior is related to the reactivity of WEEE glass within the geopolymeric matrix, which favors Pb mobility; for such a reason, the amount of WEEE glass within NaOH-metakaolin geopolymer needs to be checked carefully.
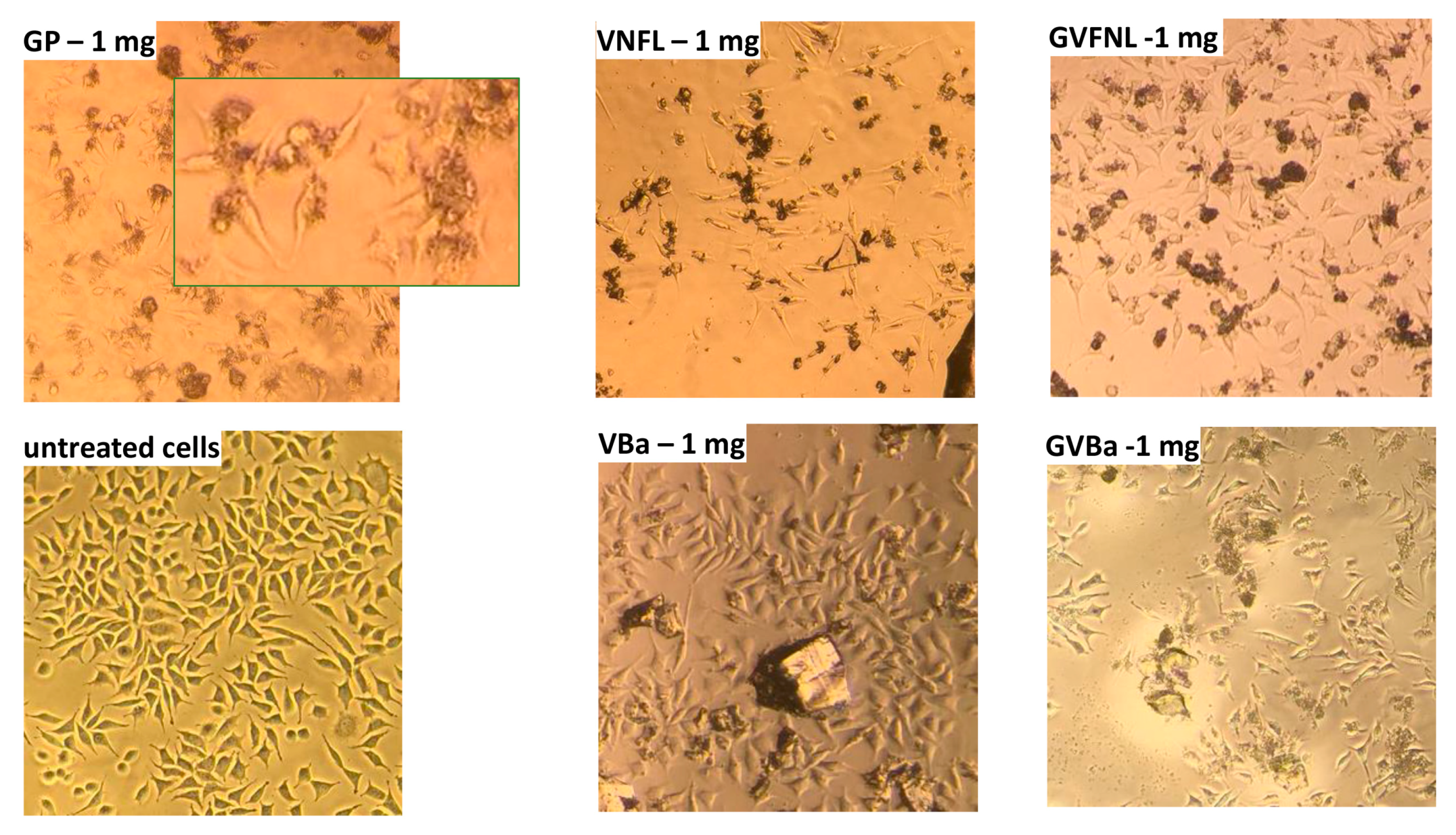
In order to assess whether the amount of released metals could pose a risk to human health, cytotoxicity assays were carried out. In particular, to evaluate the influence of the material powders on morphology and cell proliferation, the NIH-3T3 murine fibroblast cell line was grown in the presence of the investigated materials for 48 h. A slight change in the treated NIH-3T3 cells morphology was observed compared to the untreated control (
Figure 5), which suggests that the cells are suffering.
Moreover, after 48 h of exposure to the materials, an MTT assay was carried out to evaluate material cytotoxicity. Mitochondrial toxicity is one of the well-known mechanisms of cytotoxicity because of the central metabolic role of mitochondria in energy homeostasis, oxidative stress reduction, and apoptosis induction. In order to assess the influence of the materials on mitochondria, an MTT cytotoxicity assay was performed. The quantitative measurement of extracellular reduction of the yellow-colored, water-soluble tetrazolium dye to insoluble formazan crystals allowed us to state that all materials affect the viability of the tested NIH-3T3 cells.
Figure 6 shows that the presence of pure geopolymer in the culture medium leads to a decrease of cell viability. Its toxicity was due to the high amount of Al released, as shown by the leaching test (
Table 2). A decrease of the cell viability was also recorded after exposure to pure VNFL, which is ascribable to the Pb release. A decrease of about 20% cell viability was instead observed after the exposure to pure VBa glass. This observation allows us to state that VBa is bioinert, according to Reference [
28]. When the waste glasses were incorporated into the geopolymer matrix, a modulation of Al and Pb release occurred, which explains the different toxicity of the materials. The exposure to GVFNL led to a decrease of cell viability higher than that observed when the cells were exposed to the pure geopolymer. According to the leaching test, indeed, although GVFNL releases a lower Al amount than VNFL, it releases also a higher amount of Pb, which causes a more extensive toxic effect. However, although GVFNL releases a high amount of Pb and Al, the viability of the cells exposed to GVNFL powder was slightly higher than that observed when the cells were grown in contact with pure VNFL. This result can be explained by the higher SiO
2 amount in GVFNL compared to the pure glass. It is known, indeed, that silica-based materials are bioactive and biocompatible and, thus, they are widely used as biomaterials, mainly in orthopedic and dental fields. Those materials are able to stimulate osteogenesis via both their dissolution and degradation products. Soluble silica ions activate and stimulate osteoprogenitor cells at the implant site, promoting bone tissue growth [
29,
30].
Geopolymer containing VBa (GVBa), instead, causes a different cell response. The exposure to GVBa does not affect cell proliferation as well as pure VBa. Moreover, GVBa shows also lower toxicity than the pure geopolymer, because the addition of glass to the geopolymeric matrix causes the increase of the Si/Al ratio in GVBa compared to pure GP, which leads to a decrease in the Al release [
10].
The cell viability, moreover, depends on the concentration of the materials in the culture medium. In fact, when the cells are exposed to 2 mg of samples, all materials show higher toxicity. The cell viability as a function of the sample chemical composition follows the same described trend. Only the cell response to the GVBa exposure has a different trend. The cells exposed to 1 mg of VBa have the same viability of those exposed to 1 mg of GVBa. On the contrary, the viability of the cells exposed to 2 mg of GVBa decreased compared to that of the cells exposed to 2 mg of VBa. This observation suggests that an increase of the released metal concentrations occurs and that the reached concentrations match the toxic value. In particular, this effect can be ascribable to the increase of the Al concentration. In the literature [
31], using the MTT assay, it was proved that a liver-derived cell line showed a decrease of mitochondrial function when exposed to doses higher than 50 µg/mL of Al nanoparticles, whereas no significant effects were observed at lower doses. However, cell viability observed after exposure to 2 mg of GVBa was higher than that observed when the cells were treated with pure GP and GVFNL, as well as when 1 mg was used.