Vertebral Malformations in Fish from the Coast of Nayarit, Mexico, and Their Association with Organochlorine and Organophosphate Pesticides
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. Fish Collection and Characterization of Malformations
2.3. Fish Processing
2.4. Preparation of Vertebrae Samples
2.5. Extraction of Organochlorine and Organophosphate Pesticides
2.6. Chromatographic Conditions
2.7. Method Validation
2.8. Quality Control
2.9. Statistical Analysis
3. Results
3.1. Biological Samples and the Presence of Vertebral Malformations
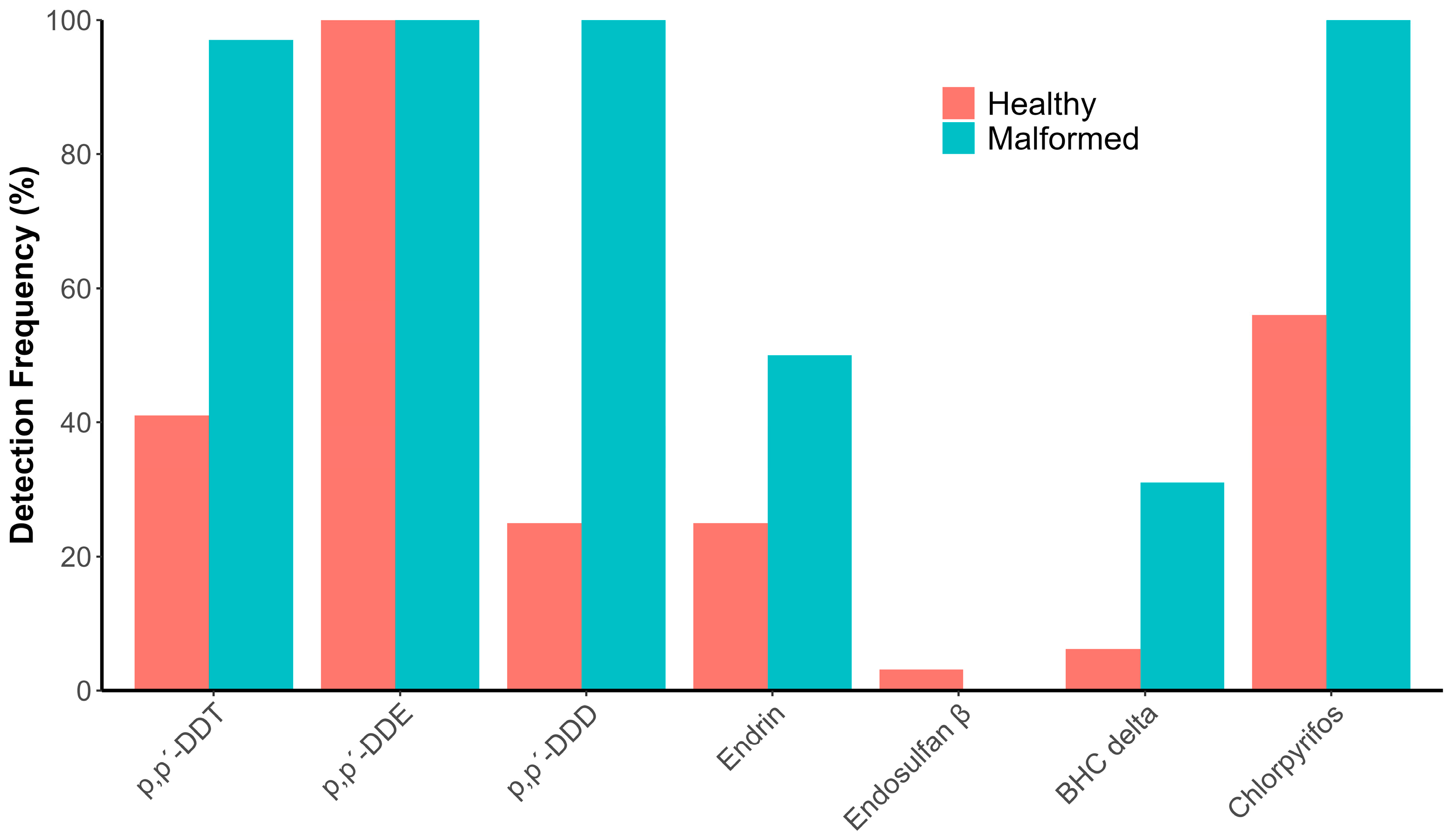
3.2. OCPs and OPPs in the Vertebrae, with and withoutMalformations, of Different Fish Species from the Coast of San Blas, Nayarit, Mexico
3.3. Association Between the Concentration of OCPs and OPPs in Vertebrae of Different Fish Species from the Coast of San Blas, Nayarit, Mexico, and the Presence or Absence of Vertebral Malformations
4. Discussion
4.1. Biological Samples and Presence of Vertebral Malformations
4.2. Detection and Quantification of OCPs and OPPs in Vertebrae, with and Without Malformations, from Different Fish Species of the Coast of San Blas, Nayarit, Mexico
4.3. Association Between the Concentration of Organochlorine and Organophosphate Pesticides in Vertebrae of Different Fish Species from the Coast of San Blas, Nayarit, Mexico, and the Presence or Absence of Vertebral Malformations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Comisión Federal para la Protección Contra Riesgos Sanitarios. Consulta de Registros Sanitarios de Plaguicidas, Nutrientes Vegetales y LMR. Available online: https://siipris03.cofepris.gob.mx/resoluciones/consultas/conwebregplaguicida.asp (accessed on 17 June 2025).
- Fernández-Bremauntz, A.; Yarto-Ramírez, M.; Castro-Díaz, J. Las Sustancias Tóxicas Persistentes en México; Instituto Nacional de Ecología: Tlalpan, Mexico, 2024; Volume 1. [Google Scholar]
- Arellano-Aguilar, O.; Rendón von Osten, J. La Huella de los Plaguicidas en México; Greenpeace: Amsterdam, The Netherlands, 2017. [Google Scholar]
- García Hernández, J.; Leyva Morales, J.B.; Martínez Rodríguez, I.E.; Hernández Ochoa, M.I.; Aldana Madrid, M.L.; Rojas Gracía, A.E.; Betancourt Lozano, M.; Pérez Herrera, N.; Perera Ríos, J.H. Estado actual de la investigación sobre plaguicidas en México. Rev. Int. Contam. Ambient. 2018, 34, 29–60. [Google Scholar] [CrossRef]
- Ponce-Vélez, G.; Botello, A.V. Plaguicidas organoclorados en organismos costeros y marinos de los litorales mexicanos: Una revisión. Rev. Int. Contam. Ambient. 2018, 34, 81–89. [Google Scholar] [CrossRef]
- Mathikere, S.A.; Kundlas, K. Negative impacts of intensive agricultural practices on environment and ecosystem: A review. Int. J. Res. Agron. 2024, 7, 285–289. [Google Scholar] [CrossRef]
- Huang, D.; Gao, L.; Zhu, S.; Qiao, L.; Liu, Y.; Ai, Q.; Xu, C.; Wang, W.; Lu, M.; Zheng, M. Target and non-target analysis of organochlorine pesticides and their transformation products in an agrochemical-contaminated area. Chemosphere 2023, 324, 138314. [Google Scholar] [CrossRef]
- Gao, Z.; Chen, Y.; Qin, Q.; Wang, R.; Dai, Z. Distribution Characteristics and Influencing Factors of Organochlorine Pesticides in Agricultural Soil from Xiamen City. Int. J. Environ. Health Res. Public Health 2023, 20, 1916. [Google Scholar] [CrossRef]
- Wang, H.; Zhang, L.; Yang, F.; Yan, L.; Lin, C.; Shen, C. Characteristics, source analysis, and risk assessment of organochlorine pesticide contamination in nearshore surface sediments of a tropical tourist island. Front. Mar. Sci. 2025, 11, 36–41. [Google Scholar] [CrossRef]
- Walker, C.H. Organic Pollutants: An Ecotoxicological Perspective, 2nd ed.; Routledge: Abingdon, UK, 2008. [Google Scholar]
- Taiwo, A.M. A review of environmental and health effects of organochlorine pesticide residues in Africa. Chemosphere 2019, 220, 1126–1140. [Google Scholar] [CrossRef] [PubMed]
- Zambrano-Soria, M.; Toledo-Ibarra, G.A.; Covantes-Rosales, C.E.; Barajas-Carrillo, V.W.; Rios-Jiménez, I.; Leyva-Morales, J.B.; Navidad-Murrieta, M.S.; Razura-Carmona, F.F.; Girón-Pérez, M.I. Pesticide levels in shrimp on Mexican coasts. Int. J. Environ. Health Res. 2025, 35, 1456–1476. [Google Scholar] [CrossRef] [PubMed]
- Briones-Venegas, A.; Ponce-Vélez, G.; Elías-García, V.G.; Botello, A.V. Organochlorine contaminants in sediments and factors influencing their distribution in the natural marine protected area in the gulf of Mexico. Chemosphere 2023, 339, 139781. [Google Scholar] [CrossRef]
- Bernal-González, K.G.; Covantes-Rosales, C.E.; Camacho-Pérez, M.R.; Mercado-Salgado, U.; Barajas-Carrillo, V.W.; Girón-Pérez, D.A.; Montoya-Hidalgo, A.C.; Díaz-Resendiz, K.J.G.; Barcelos-García, R.G.; Toledo-Ibarra, G.A.; et al. Organophosphate-Pesticide-Mediated Immune Response Modulation in Invertebrates and Vertebrates. Int. J. Mol. Sci. 2023, 24, 5360. [Google Scholar] [CrossRef]
- Carvalho, F.P.; Fowler, S.W.; Gonzalez-Farias, F.; Mee, L.D.; Readman, J.W. Agrochemical residues in the Altata-Ensenada del Pabellon coastal lagoon (Sinaloa, Mexico): A need for integrated coastal zone management. Int. J. Environ. Health Res. 1996, 6, 209–220. [Google Scholar] [CrossRef]
- Carvalho, F.P.; Gonzalez-Farias, F.; Villeneuve, J.P.; Cattini, C.; Hernandez-Garza, M.; Mee, L.D.; Fowler, S.W. Distribution, Fate and Effects of Pesticide Residues in Tropical Coastal Lagoons of Northwestern Mexico. Environ. Technol. 2002, 23, 1257–1270. [Google Scholar] [CrossRef]
- Camacho-Jiménez, L.; González-Ruiz, R.; Yepiz-Plascencia, G. Persistent organic pollutants (POPs) in marine crustaceans: Bioaccumulation, physiological and cellular responses. Mar. Environ. Res. 2023, 192, 106184. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, F.P. Agriculture, pesticides, food security and food safety. Environ. Sci. Policy 2006, 9, 685–692. [Google Scholar] [CrossRef]
- Leyva-Morales, J.B.; Bastidas-Bastidas, P.d.J.; Salvatierra-Stamp, V.d.C.; González-Márquez, L.C.; Davizon, Y.A.; Márquez-Pacheco, H.; Amillano-Cisneros, J.M.; Cruz-Acevedo, E.; Amézquita-López, B.A.; Romo Gómez, C.; et al. Temporal distribution and ecological risk assessment for pesticides in water from the north-central coastal zone of Sinaloa, Mexico. Heliyon 2024, 10, e35207. [Google Scholar] [CrossRef] [PubMed]
- Robledo-Marenco, M.L.; Botello, A.V.; Romero-Banuelos, C.A.; Diaz-Gonzalez, G. Presence of persistent organochlorine pesticides in estuaries of the subtropical Mexican Pacific. Int. J. Environ. Pollut. 2006, 26, 284–294. [Google Scholar] [CrossRef]
- Osuna-López, J.I.; Frías-Espericueta, M.G.; López-López, G.; Zazueta-Padilla, H.M.; Izaguirre-Fierro, G.; Páez-Osuna, F.; Ruiz-Fernández, A.C.; Voltolina, D. Cd, Pb and organochlorine pesticides of Mytella strigata (Pelecypoda: Mytilidae) of six coastal lagoons of NW Mexico. Boletín Investig. Mar. Costeras 2009, 38, 233–239. [Google Scholar]
- Martínez-Villa, G.; Betancourt-Lozano, M.; Aguilar-Zárate, G.; Ruelas-Inzunza, J.; Anislado-Tolentino, V.; Cerdenares Ladrón de Guevara, G.; Ramos Carrillo, S.; González-Medina, G. Contenido de plaguicidas organoclorados en varios peces depredadores de la costa de Oaxaca; evaluación del riesgo de exposición por consumo en la salud humana. In Pacífico Mexicano. Contaminación e Impacto Ambiental: Diagnóstico; Tendencias; Botello, A.V., Páez-Osuna, F., Méndez-Rodríguez, L., Betancourt-Lozano, M., Álvarez-Borrego, S., Lara-Lara, R., Eds.; Epomex Institute-Autonomous University of Campeche: Campeche, México, 2014; pp. 169–208. [Google Scholar]
- Osuna-López, I.; Frías-Espericueta, M.G.; López-López, G.; Izaguirre-Fierro, G.; Zazueta-Padilla, H.; Aguilar-Juárez, M.; Correa-González, E.M.; Bautista-Covarrubias, J.C.; Cervantes-Atondo, J.A.; Sánchez-Osuna, L.; et al. Niveles de concentración de pesticidas organoclorados en moluscos bivalvos del noroeste de México. In Pacífico Mexicano. Contaminación e Impacto Ambiental: Diagnóstico; Tendencias; Botello, A.V., Páez-Osuna, F., Méndez-Rodríguez, L., Betancourt-Lozano, M., Álvarez-Borrego, S., Lara-Lara, R., Eds.; Epomex Institute-Autonomous University of Campeche: Campeche, México, 2014; pp. 33–42. [Google Scholar]
- Aguayo-Quiroz, C.E. Evaluación del riesgo para la salud humana por la ingesta de plaguicidas organoclorados en peces comerciales de la costa de Nayarit, México. Master’s Thesis, Universidad Autónoma de Nayarit: Tepic, México, 2019; Unpublished work. [Google Scholar]
- Akoto, O.; Azuure, A.A.; Adotey, K.D. Pesticide residues in water, sediment and fish from Tono Reservoir and their health risk implications. SpringerPlus 2016, 5, 1849. [Google Scholar] [CrossRef]
- Tiedeken, J.A.; Ramsdell, J.S. DDT exposure of zebrafish embryos enhances seizure susceptibility: Relationship to fetal p,p’-DDE burden and domoic acid exposure of California sea lions. Environ. Health Perspect. 2009, 117, 68–73. [Google Scholar] [CrossRef] [PubMed]
- Ullah, S.; Zorriehzahra, M.J. Ecotoxicology: A Review of Pesticides Induced Toxicity in Fish. Adv. Anim. Vet. Sci. 2015, 3, 40–47. [Google Scholar] [CrossRef]
- Martyniuk, C.J.; Mehinto, A.C.; Denslow, N.D. Organochlorine pesticides: Agrochemicals with potent endocrine-disrupting properties in fish. Mol. Cell. Endocrinol. 2020, 507, 110764. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, A.K.; Sarower, M.G. Potential toxicity of pesticides and its transformation products to aquatic organisms. Khulna Univ. Stud. 2024, 21, 21–32. [Google Scholar] [CrossRef]
- Burch, E.; Hussein, M.A.; Zaki, M.; Kamal, L.T.; Zaki, G.; Shoeib, T.; Dawood, M.; Sewilam, H.; Abdelnaser, A. Assessing the Effects of Pesticides on Aquacultured Fish and Ecosystems: A Comprehensive Environmental Health Review. Fishes 2025, 10, 223. [Google Scholar] [CrossRef]
- Moezzi, S.A.; Ramezani, S.; Rezaei, K.; Khoei, A.J. Mechanisms of Pesticide Toxicity in Fish: Insights into the Ameliorative Role of Plant-Derived Compounds-A Review. Aquac. Nutr. 2025, 2025, 5328773. [Google Scholar] [CrossRef] [PubMed]
- Watson, F.L.; Schmidt, H.; Turman, Z.K.; Hole, N.; Garcia, H.; Gregg, J.; Tilghman, J.; Fradinger, E.A. Organophosphate pesticides induce morphological abnormalities and decrease locomotor activity and heart rate in Danio rerio and Xenopus laevis. Environ. Toxicol. Chem. 2014, 33, 1337–1345. [Google Scholar] [CrossRef] [PubMed]
- Rahman, M.S.; Islam, S.M.M.; Haque, A.; Shahjahan, M. Toxicity of the organophosphate insecticide sumithion to embryo and larvae of zebrafish. Toxicol. Rep. 2020, 7, 317–323. [Google Scholar] [CrossRef]
- Neylon, J.; Fuller, J.N.; van der Poel, C.; Church, J.E.; Dworkin, S. Organophosphate Insecticide Toxicity in Neural Development, Cognition, Behaviour and Degeneration: Insights from Zebrafish. J. Dev. Biol. 2022, 10, 49. [Google Scholar] [CrossRef]
- Erhunmwunse, N.O.; Tongo, I.; Omigie, K. Embryonic toxicity of Imidacloprid: Impact on hatchability, survivability, swimming speed and cardiac function of catfish, Clarias gariepinus. Ecotoxicology 2023, 32, 127–134. [Google Scholar] [CrossRef]
- Dishaw, L.V.; Hunter, D.L.; Padnos, B.; Padilla, S.; Stapleton, H.M. Developmental Exposure to Organophosphate Flame Retardants Elicits Overt Toxicity and Alters Behavior in Early Life Stage Zebrafish (Danio rerio). Toxicol. Sci. 2014, 142, 445–454. [Google Scholar] [CrossRef]
- De La Cruz-Agüero, J.; Perezgómez-Alvarez, L. Lordosis in topsmelt Atherinops affinis (Ayres, 1860) (Teleostei: Atherinopsidae). Rev. Biol. Mar. Oceanogr. 2001, 36, 109–110. [Google Scholar] [CrossRef]
- Rodríguez-Romero, J.; Cárdenas, L.; Pérez-Urbiola, J.C.; Hinohuye-Rivera, R.; Silva-Hernández, M.A. A spinal column malformation in the creolefish Paranthias colonus (Osteichthyes: Serranidae). Rev. Biol. Trop. 2001, 49, 1267–1268. [Google Scholar] [PubMed]
- Comisión Nacional de Acuacultura y Pesca. Anuario Estadístico de Acuacultura y Pesca. Available online: https://nube.conapesca.gob.mx/sites/cona/dgppe/2024/ANUARIO_ESTADISTICO_DE_ACUACULTURA_Y_PESCA_2024.pdf (accessed on 15 June 2024).
- Tirado-León, D.E. Caracterización de Malformaciones Vertebrales en Bagres (Siluriformes: Ariidae) del Litoral de Nayarit, México. Master’s Thesis, Universidad Autónoma de Nayarit, Nayarit, México, 2019. [Google Scholar]
- Instituto Nacional de Estadística y Geografía. Áreas Geográficas de Nayarit. Available online: https://www.inegi.org.mx/app/areasgeograficas/?ag=18 (accessed on 20 May 2024).
- Berlanga-Robles, C.A.; Ruiz-Luna, A. Evaluación de cambios en el paisaje y sus efectos sobre los humedales costeros del sistema estuarino de San Blas, Nayarit (México) por medio de análisis de imágenes Landsat. Cienc. Mar. 2006, 32, 523–538. [Google Scholar] [CrossRef]
- ECURED. San Blas (México). Available online: https://www.ecured.cu/San_Blas_(M%C3%A9xico) (accessed on 9 May 2025).
- Instituto Nacional para el Federalismo y el Desarrollo Municipal. Enciclopedia de los Municipios de México: Nayarit. Available online: http://www.inafed.gob.mx/work/enciclopedia/EMM18nayarit/ (accessed on 10 May 2025).
- Diario Oficial de la Federación. Norma Oficial Mexicana NOM-059-SEMARNAT-2010. Available online: https://www.dof.gob.mx/normasOficiales/4254/semarnat/semarnat.htm (accessed on 10 May 2025).
- Fischer, W.; Krupp, F.; Schnelder, W.; Sommer, C.; Carpenter, K.E.; Niem, V.H. Guía FAO para la Identificación de Especies para los Fines de la Pesca. Pacífico Centro-Oriental; FAO: Rome, Italy, 1995; Volume III. [Google Scholar]
- Nelson, J.S.; Grande, T.C.; Wilson, M.V.H. Fishes of the World, 5th ed.; Wiley: Hoboken, NJ, USA, 2016. [Google Scholar]
- Alexandre, A.P.; Menezes, N.A. Systematics of the family Ariidae (Ostariophysi, Siluriformes), with a redefinition of the genera. Zootaxa 2007, 1416, 1–126. [Google Scholar] [CrossRef]
- Rooker, J.; Zdanowicz, V.; Secor, D. Chemistry of tuna otoliths: Assessment of base composition and postmortem handling effects. Mar. Biol. 2001, 139, 35–43. [Google Scholar] [CrossRef]
- Hashemy-Tonkabony, S.E.; Langaroodi, F.A. Detection and determination of chlorinated pesticide residues in Caspian sea fish by gas-liquid chromatography. Environ. Res. 1976, 12, 275–280. [Google Scholar] [CrossRef]
- Lehotay, S.J. Determination of Pesticide Residues in Foods by Acetonitrile Extraction and Partitioning with Magnesium Sulfate: Collaborative Study. J. AOAC Int. 2019, 90, 485–520. [Google Scholar] [CrossRef]
- Pihlström, T.; Fernández-Alba, A.R.; Ferrer-Amate, C.; Poulsen, M.E.; Lippold, R.; Carrasco-Cabrera, L.; Pelosi, P.; Valverde, A.; Mol, H.; Jezussek, M.; et al. Analytical Quality Control and Method Validation Procedures for Pesticide Residues Analysis in Food and Feed Sante 11312/2021-v2; EURL: Ispra, Italy, 2021. [Google Scholar]
- Leyva-Morales, J.B.; Bastidas-Bastidas, P.J.; Muñiz-Valencia, R.; Ceballos Magaña, S.G.; Ponce-Vélez, G.; Aguilera-Márquez, D.; Grajeda-Cota, P.; Navidad-Murrieta, M.S.; Flores-Munguía, M.E.; Ramírez-Castillo, J.A.; et al. Measurement of organochlorine pesticides in drinking water: Laboratory technical proficiency testing in Mexico. Accredit. Qual. Assur. 2019, 24, 451–461. [Google Scholar] [CrossRef]
- Helsel, D.R. Statistics in Practice. In Statistics for Censored Environmental Data Using Minitab® and R; Wiley: Hoboken, NJ, USA, 2011; p. 325. [Google Scholar]
- Clarke, K.R.; Warwick, R.M. Change in Marine Communities: An Approach to Statistical Analysis and Interpretation, 2nd ed.; Primer-E Ltd.: Plymouth, UK, 2001. [Google Scholar]
- FishBase. Available online: https://www.fishbase.se/search.php (accessed on 11 September 2024).
- R Core Team. R: A Language and Environment for Statistical Computing. Available online: https://www.r-project.org/ (accessed on 20 September 2025).
- González-Díaz, A.A.; Soria-Barreto, M. Lista sistemática preliminar de los peces del Estado de Nayarit, México. BioCiencias 2013, 2, 200–215. [Google Scholar] [CrossRef]
- Marceniuk, A.P.; Betancur-R, R.; Acero, A. A new species of Cathorops (Siluriformes: Ariidae) from Mesoamerica, with redescription or four species from the eastern pacific. Bull. Mar. Sci. 2009, 85, 245–280. [Google Scholar]
- Wakida-Kusunoki, A.T.; Amador-del Ángel, L.E.; Moreno-Miranda, C. Spinal deformities in Amazon sailfin catfish Pterygoplichthys pardalis (Siluriformes: Locariidae), an introduced fish in the Palizada River (Southeastern Mexico). Cybium 2014, 38, 155–157. [Google Scholar]
- Wakida-Kusunoki, A.T.; Amador del Ángel, L.E. Vertebral deformities in hardhead catfish Ariopsis felis (Siluriformes: Ariidae) in the southeastern Mexico. Rev. Biol. Mar. Oceanogr. 2017, 52, 159–162. [Google Scholar] [CrossRef]
- Guzmán-Morales, F.; Ramos-Ávila, A.Y.; Díaz-Santana-Iturrios, M.; Granados-Amores, E.; Granados-Amores, J. Records of malformed sea catfishes (Ariopsis seemanni and Ariopsis guatemalensis) (Actinopterygii: Siluriformes: Ariidae) off San Blas (Mexican Pacific). Acta Ichthyol. Piscat. 2023, 53, 45–49. [Google Scholar] [CrossRef]
- González-Arias, C.A.; Robledo-Marenco, M.D.L.; Medina-Díaz, I.M.; Velázquez-Fernández, J.B.; Girón-Pérez, M.I.; Quintanilla-Vega, B.; Ostrosky-Wegman, P.; Pérez-Herrera, N.E.; Rojas-García, A.E. Patrón de uso y venta de plaguicidas en Nayarit, México. Rev. Int. Contam. Ambient. 2010, 26, 221–228. [Google Scholar]
- Bernal-Hernández, Y.Y. Evaluación de la Exposición a Plaguicidas y Metales Pesados en Ostión (Crassostrea corteziensis) del Estero Boca de Camichín, Nayarit. Master’s Thesis, Universidad Autónoma de Nayarit, Nayarit, México, 2009. [Google Scholar]
- Aranguré Zúñiga, F.J.; Zambrano Cárdenas, R.M.; González Torres, L.; Robledo Marenco, M.L. Evaluación de los niveles de contaminación por residuos de plaguicidas organoclorados en sedimento y peces, de la presa de Aguamilpa, Nayarit, México. Waxapa 2011, 3, 20–35. [Google Scholar]
- Cahu, C.; Zambonino Infante, J.; Takeuchi, T. Nutritional components affecting skeletal development in fish larvae. Aquaculture 2003, 227, 245–258. [Google Scholar] [CrossRef]
- Baeverfjord, G.; Antony Jesu Prabhu, P.; Fjelldal, P.G.; Albrektsen, S.; Hatlen, B.; Denstadli, V.; Ytteborg, E.; Takle, H.; Lock, E.-J.; Berntssen, M.H.G.; et al. Mineral nutrition and bone health in salmonids. Rev. Aquac. 2019, 11, 740–765. [Google Scholar] [CrossRef]
- Sivagurunathan, U.; Dominguez, D.; Tseng, Y.; Zamorano, M.J.; Philip, A.J.P.; Izquierdo, M. Interaction between Dietary Vitamin D3 and Vitamin K3 in Gilthead Seabream Larvae (Sparus aurata) in Relation to Growth and Expression of Bone Development-Related Genes. Aquac. Nutr. 2023, 2023, 3061649. [Google Scholar] [CrossRef] [PubMed]
- Zwart, P.; Nijboer, J.; Schaftenaar, W. Four-Eyed Fish (A. anableps) Fingerlings Demonstrated Multiple Birth Defects Suggestive of Nutritional Deficiencies. J. Pathol. Res. Rev. 2024, 6, 1–7. [Google Scholar] [CrossRef]
- Tseng, Y.; Izquierdo, M.; Sivagurunathan, U.; Philip, A.J.P.; Domínguez, D. Effects of dietary zinc on growth, bone-related genes expression and skeletal anomalies in gilthead seabream (Sparus aurata) larvae. Aquaculture 2025, 599, 742076. [Google Scholar] [CrossRef]
- Tseng, Y.; Dominguez, D.; Sivagurunathan, U.; Zamorano, M.J.; Philip, A.J.P.; Izquierdo, M. Effects of the dietary supplementation of manganese on growth, skeletal development, oxidative stress and bone metabolism related genes in gilthead seabream (Sparus aurata) larvae. Aquaculture 2025, 594, 741439. [Google Scholar] [CrossRef]
- Fisher, S.; Jagadeeswaran, P.; Halpern, M.E. Radiographic analysis of zebrafish skeletal defects. Dev. Biol. 2003, 264, 64–76. [Google Scholar] [CrossRef] [PubMed]
- Aydın, İ.; Alemdağ, M.; Terzi, Y.; Öztürk, R.Ç.; Küçük, E.; Polat, H.; Çoban, D. Ontogeny of vertebral column and fin development, and detection of deformations in hatchery-reared European Flounder (Platichthys flesus) during early life stage. Aquaculture 2024, 582, 740498. [Google Scholar] [CrossRef]
- Kent, M.L.; Watral, V.G.; Whipps, C.M.; Cunningham, M.E.; Criscione, C.D.; Heidel, J.R.; Curtis, L.R.; Spitsbergen, J.; Markle, D.F. A Digenean Metacercaria (Apophallus sp.) and a Myxozoan (Myxobolus sp.) Associated with Vertebral Deformities in Cyprinid Fishes from the Willamette River, Oregon. J. Aquat. Anim. Health 2004, 16, 116–129. [Google Scholar] [CrossRef]
- Ngo, A.L.; Go, J.; Spiers, Z.B.; Jenkins, C. Scoliosis and kyphosis in blue-spotted and marbled flathead fish associated with a Myxobolus acanthogobii-like parasite. J. Vet. Diagn. Investig. 2024, 36, 380–388. [Google Scholar] [CrossRef]
- Harris, M.P.; Henke, K.; Hawkins, M.B.; Witten, P.E. Fish is Fish: The use of experimental model species to reveal causes of skeletal diversity in evolution and disease. J. Appl. Ichthyol. 2014, 30, 616–629. [Google Scholar] [CrossRef] [PubMed]
- Chong, R.S.-M. Chapter 72—Fish genetic malformations. In Aquaculture Pathophysiology; Kibenge, F.S.B., Baldisserotto, B., Chong, R.S.-M., Eds.; Academic Press: Cambridge, MA, USA, 2022; pp. 801–809. [Google Scholar]
- Chen, Y.; Li, H.; Wei, Q.; Liu, G.; Liu, Z.; Wu, X.; Chen, Y.; Yu, Y.; Tu, Q.; Yang, H. Transcriptome sequencing reveals the molecular mechanisms of early malformations in the critically endangered Sichuan taimen (Hucho bleekeri). Comp. Biochem. Physiol. Part D Genom. Proteom. 2025, 56, 101639. [Google Scholar] [CrossRef]
- Pumputis, P.G.; Xu, R.; Gopaul, J.; Panahifar, A.; Erfani, V.; Van Gennip, J.L.M.; Eames, B.F.; Fakhari, N.; Baranger, J.; Lebel, D.E.; et al. Oxidative stress-induced intervertebral disc remodelling and elevated stiffness drive idiopathic scoliosis in preclinical models. Nat. Commun. 2025, 16, 8719. [Google Scholar] [CrossRef]
- Divanach, P.; Boglione, C.; Menu, B.; Koumoundouros, G.; Kentouri, M.; Cataudella, S. Abnormalities in finfish mariculture: An overview of the problem, causes and solutions. Spec. Publ. Eur. Aquac. Soc. 1996, 45–66. [Google Scholar]
- Mo, J.; Chen, Y.; Lai, K.P.; Zhang, Z.; Seemann, F.; Liu, W. Hypoxia to the skeletal systems of teleost: Impacts, mechanisms, and health implications. J. Environ. Manag. 2025, 391, 126530. [Google Scholar] [CrossRef] [PubMed]
- Boglione, C. Skeletal abnormalities. In Climate Change and Non-Infectious Fish Disorders; CABI Digital Library: Davis Davis, CA, USA, 2020; pp. 54–79. [Google Scholar] [CrossRef]
- Georgakopoulou, E.; Katharios, P.; Divanach, P.; Koumoundouros, G. Effect of temperature on the development of skeletal deformities in Gilthead seabream (Sparus aurata Linnaeus, 1758). Aquaculture 2010, 308, 13–19. [Google Scholar] [CrossRef]
- Østervold, M.; Imsland, A.K.D.; Yilmaz, O.; Fjelldal, P.G.; Mangor-Jensen, R.; Siapazis, C.; Mangor-Jensen, A.; Norberg, B. Effect of temperature on growth, expression of growth regulating genes, and deformity development of juvenile European plaice (Pleuronectes platessa). Aquaculture 2025, 594, 741450. [Google Scholar] [CrossRef]
- Pan, L.; Zhang, Y.; Li, Y.; Wen, H.; Liu, X.; Yang, J.; Qi, X.; Zhang, K. Study on the mechanism of skeletal deformity of Lateolabrax maculatus induced by low temperature stress. Comp. Biochem. Physiol. Part D Genom. Proteom. 2025, 56, 101592. [Google Scholar] [CrossRef]
- Rodríguez-Ibarra, L.E.; Abdo-de la Parra, M.I.; Velasco-Blanco, G.; Aguilar-Zárate, G.; Martínez-Brown, J.M. Effect of temperature on the incubation, growth, survival, and presence of skeletal deformities in larvae of the clownfish Amphiprion ocellaris (Pomacentridae) under rearing conditions. Lat. J. Aquat. Res. 2024, 52, 7. [Google Scholar] [CrossRef]
- Mattos, D.d.C.; Cardoso, L.D.; Oliveira, A.T.d.; Screnci-Ribeiro, R.; Mattos, B.O.d.; Aride, P.H.R.; Radael, M.C.; Motta, J.H.d.S.; Vidal, M.V. Effect of temperature on the embryonic and larvae development of discus fish Symphysodon aequifasciatus and time of first feeding. Zygote 2024, 32, 279–284. [Google Scholar] [CrossRef]
- Hassanain, M.A.; Abbas, W.T.; Ibrahim, T.B. Skeletal Ossification Impairment in Nile Tilapia (Oreochromis niloticus) after Exposure to Lead Acetate. Pak. J. Biol. Sci. 2012, 15, 729–735. [Google Scholar] [CrossRef]
- Jezierska, B.; Ługowska, K.; Witeska, M. The effects of heavy metals on embryonic development of fish (a review). Fish Physiol. Biochem. 2009, 35, 625–640. [Google Scholar] [CrossRef]
- Sfakianakis, D.G.; Renieri, E.; Kentouri, M.; Tsatsakis, A.M. Effect of heavy metals on fish larvae deformities: A review. Environ. Res. 2015, 137, 246–255. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; Wang, W.-X. Cadmium impacts on calcium mineralization of zebrafish skeletal development and behavioral impairment. Aquat. Toxicol. 2024, 273, 107033. [Google Scholar] [CrossRef] [PubMed]
- Shahjahan, M.; Taslima, K.; Rahman, M.S.; Al-Emran, M.; Alam, S.I.; Faggio, C. Effects of heavy metals on fish physiology—A review. Chemosphere 2022, 300, 134519. [Google Scholar] [CrossRef] [PubMed]
- Villeneuve, D.L.; Curtis, L.R.; Jenkins, J.J.; Warner, K.E.; Tilton, F.; Kent, M.L.; Watral, V.G.; Cunningham, M.E.; Markle, D.F.; Sethajintanin, D.; et al. Environmental Stresses and Skeletal Deformities in Fish from the Willamette River, Oregon. Environ. Sci. Technol. 2005, 39, 3495–3506. [Google Scholar] [CrossRef]
- Cherr, G.N.; Fairbairn, E.; Whitehead, A. Impacts of Petroleum-Derived Pollutants on Fish Development. Annu. Rev. Anim. Biosci. 2017, 5, 185–203. [Google Scholar] [CrossRef]
- Holden, A.V. Effects of Pesticides on Fish. In Environmental Pollution by Pesticides; Edwards, C.A., Ed.; Springer: Boston, MA, USA, 1973; pp. 213–253. [Google Scholar]
- Bengtsson, B.-e. Biological variables, especially skeletal deformities in fish, for monitoring marine pollution. Philos. Trans. R. Soc. Lond. B Biol. Sci. 1979, 286, 457–464. [Google Scholar] [CrossRef]
- Berillis, P. Factors that can lead to the development of skeletal deformities in fishes: A review. J. Fish. Sci. 2015, 9, 17–23. [Google Scholar]
- Tang, X.; Meng, Y.; Li, H.; Liu, K.; Wang, X.; Zhong, Z.; Ye, J.; Chen, J. Characterization of metalaxyl-induced notochord toxicity based on biochemical and transcriptomics in zebrafish (Danio rerio) model. J. Hazard. Mater. 2025, 486, 136985. [Google Scholar] [CrossRef]
- Fukugami, S.; Yamasaki, M.; Kokushi, E.; Uno, S. Influence of CYP1A and AhR modulation on polycyclic aromatic hydrocarbon-induced developmental defects in Japanese medaka. Aquat. Toxicol. 2025, 280, 107267. [Google Scholar] [CrossRef] [PubMed]
- Sørhus, E.; Sørensen, L.; Grøsvik, B.E.; Le Goff, J.; Incardona, J.P.; Linbo, T.L.; Baldwin, D.H.; Karlsen, Ø.; Nordtug, T.; Hansen, B.H.; et al. Crude oil exposure of early life stages of Atlantic haddock suggests threshold levels for developmental toxicity as low as 0.1 μg total polyaromatic hydrocarbon (TPAH)/L. Mar. Pollut. Bull. 2023, 190, 114843. [Google Scholar] [CrossRef]
- Zhang, T.; Yuan, J.; Guo, Y.; Wang, X.; Li, Q.X.; Zhang, J.; Xie, J.; Miao, W.; Fan, Y. Combined toxicity of trifloxystrobin and fluopyram to zebrafish embryos and the effect on bone development. Aquat. Toxicol. 2024, 268, 106834. [Google Scholar] [CrossRef] [PubMed]
- Johal, M.S.; Sharma, M.L.; Ravneet. Impact of low dose of organophosphate, monocrotophos on the epithelial cells of gills of Cyprinus carpio communis Linn.--SEM study. J. Environ. Biol. 2007, 28, 663–667. [Google Scholar]
- Omitoyin, B.O.; Ajani, E.K.; Adesina, B.T.; Okuagu, C.N.F. Toxicity of Lindane (Gamma Hexachloro-CycloHexane) to Clarias gariepinus (Burchell 1822). World J. Zool. 2006, 1, 57–63. [Google Scholar]
- Singh, N.N.; Srivastava, A.K. Formothion-induced haematological changes in the freshwater Indian catfish Heteropneustes fossilis. J. Ecotoxicol. Environ. Monit. 1994, 4, 137–140. [Google Scholar]
- Singh, N.N.; Das, V.K.; Srivastava, A.K. Formothion and propoxur induced ionic imbalance and skeletal deformity m a catfish, Heteropneustes fossilis. J. Environ. Biol. 1997, 18, 357–363. [Google Scholar]
- Srivastava, P.; Singh, A.; Pandey, A.K. Pesticides toxicity in fishes: Biochemical, physiological and genotoxic aspects. Biochem. Cell. Arch. 2016, 16, 199–218. [Google Scholar]
- Velmurugan, B.; Selvanayagam, M.; Cengiz, E.I.; Unlu, E. The Effects of Monocrotophos to Different Tissues of Freshwater Fish Cirrhinus mrigala. Bull. Environ. Contam. Toxicol. 2007, 78, 450–454. [Google Scholar] [CrossRef]
- Lazhar, M.; Hela, T.; Moncef, B.; Néji, A. Toxicity of three selected pesticides (Alachlor, Atrazine and Diuron) to the marine fish (turbot Psetta maxima). Afr. J. Biotechnol. 2012, 11, 11321–11328. [Google Scholar] [CrossRef]
- Camargo-Buitrago, J.D. Análisis de las Malformaciones del Embrión de Trucha Oncorhynchus Mykiss (Walbaum, 1792) en Condiciones de Laboratorio Bajo los Efectos de Dosis sub Letales de Carbofurano (2,2-Dimethyl-2,3-dihydro-1-benzofuran-7-yl methylcarbamate). Master’s Thesis, Universidad del Bosque, Bogotá, Colombia, 2017. [Google Scholar]
- Samreen; Zhang, X.; Wang, J.; Li, Y.; Li, X.; Zheng, Y.; Arif, M.; Ru, S. Environmental relevant herbicide prometryn induces developmental toxicity in the early life stages of marine medaka (Oryzias melastigma) and its potential mechanism. Aquat. Toxicol. 2022, 243, 106079. [Google Scholar] [CrossRef] [PubMed]




| Species | Kyphosis | Lordosis | Scoliosis |
|---|---|---|---|
| C. raredonae | Central vertebrae (n = 3) Central and precaudal vertebrae (n = 3) | Central vertebrae (n = 6) Central and precaudal vertebrae (n = 1) | Precaudal vertebrae (n = 5) |
| Precaudal vertebrae (n = 4) | - | - | |
| A. guatemalensis | Precaudal vertebrae (n = 1) | Central vertebrae (n = 1) | Central vertebrae (n = 1) |
| - | Central and precaudal Vertebrae (n = 1) | - | |
| - | Precaudal vertebrae (n = 1) | - | |
| A. seemanni | Central and precaudal vertebrae (n = 1) | Central vertebrae (n = 3) | Precaudal vertebrae (n = 1) |
| - | Precaudal vertebrae (n = 2) | - | - |
| N. kessleri | Central vertebrae (n = 2) | Central vertebrae (n = 1) | Central vertebrae (n = 1) |
| B. panamensis | Central vertebrae (n = 1) | Central vertebrae (n = 2) | Precaudal vertebrae (n = 1) |
| - | Precaudal vertebrae (n = 1) | Precaudal vertebrae (n = 1) | - |
| O. altus | Precaudal vertebrae (n = 1) | - | - |
| M. ectenes | Precaudal vertebrae (n = 1) | Precaudal vertebrae (n = 1) | - |
| C. caninus | Precaudal vertebrae (n = 1) | - | - |
| C. viridis | Precaudal vertebrae (n = 1) | - | Precaudal vertebrae (n = 2) |
| - | Central vertebrae (n = 1) | - | - |
| C. chanos | - | Precaudal vertebrae (n = 1) | Precaudal vertebrae (n = 1) |
| L. argentiventris | - | Precaudal vertebrae (n = 1) | - |
| Total | 23 | 20 | 12 |
| Pesticides/Species | p,p’-DDE | p,p’-DDD | p,p’-DDT | Endrin | Endosulfan β | BHC Delta | Chlorpyrifos | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| H | M | H | M | H | M | H | M | H | M | H | M | H | M | |
| C. raredonae | 12 | 12 | 3 | 12 | 7 | 11 | 4 | 8 | - | - | 1 | 6 | 5 | 12 |
| A. guatemalensis | 4 | 4 | 2 | 4 | 3 | 4 | 1 | - | - | - | - | - | 3 | 4 |
| A. seemanni | 3 | 3 | 1 | 3 | 2 | 3 | 2 | 2 | 1 | - | - | 1 | 3 | 3 |
| B. panamensis | 3 | 3 | 2 | 3 | 1 | 3 | - | 2 | - | - | 1 | - | 3 | 3 |
| N. kessleri | 2 | 2 | - | 2 | - | 2 | 1 | 1 | - | - | - | 1 | 2 | 2 |
| O. altus | 1 | 1 | - | 1 | - | 1 | - | - | - | - | - | - | - | 1 |
| M. ectenes | 1 | 1 | - | 1 | - | 1 | - | - | - | - | - | - | 1 | 1 |
| C. caninus | 1 | 1 | - | 1 | - | 1 | - | - | - | - | - | - | - | 1 |
| C. viridis | 2 | 2 | - | 2 | - | 2 | - | 2 | - | - | - | 2 | - | 2 |
| C. chanos | 2 | 2 | - | 2 | - | 2 | - | - | - | - | - | - | - | 2 |
| L. argentiventris | 1 | 1 | - | 1 | - | 1 | - | 1 | - | - | - | - | 1 | 1 |
| Total | 32 | 32 | 8 | 32 | 13 | 31 | 8 | 16 | 1 | 0 | 2 | 10 | 18 | 32 |
| Species/Pesticide | Healthy Fish | Malformed Fish |
|---|---|---|
| Chlorpyrifos | Chlorpyrifos | |
| C. raredonae | 0.030 ± 0.049 | 0.021 ± 0.014 |
| LD-0.163 | <LD-0.034 | |
| (12/4) | (12/10) | |
| A. guatemalensis | 0.053 ± 0.016 | 0.015 ± 0.001 |
| LD-0.070 | <LD-0.016 | |
| (4/1) | (4/2) | |
| A. seemanni | - | 0.032 ± 0.028 |
| <LD-0.041 * | LD-0.058 | |
| (3/2) | (3/1) | |
| B. panamensis | 0.017 ± 0.002 | - |
| LD-0.019 | <LD-0.035 * | |
| (3/1) | (3/2) | |
| N. kessleri | 0.023 ± 0.018 | |
| 0.013–0.039 | NR | |
| (2/0) | ||
| O. altus | - | |
| <LD-0.025 * | NR | |
| (1/0) | ||
| M. ectenes | - | |
| <LD-0.023 * | NR | |
| (1/0) | ||
| C. caninus | - | |
| <LD-0.013 * | NR | |
| (1/0) | ||
| C. viridis | 0.021 ± 0.003 | |
| 0.018–0.023 | NR | |
| (2/0) | ||
| C. chanos | - | |
| <LD-0.035 * | NR | |
| (2/1) | ||
| L. argentiventris | - | |
| <LD-0.026 * | NR | |
| (1/0) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Leyva-Morales, J.B.; Ramos-Ávila, A.Y.; Bastidas-Bastidas, P.d.J.; Granados Amores, J.; Granados Amores, E.; González Ramírez, J.; Salas-Martínez, F.; Acevedo-Sandoval, O.A.; Romo-Gómez, C.; Camacho-López, C.; et al. Vertebral Malformations in Fish from the Coast of Nayarit, Mexico, and Their Association with Organochlorine and Organophosphate Pesticides. Environments 2026, 13, 151. https://doi.org/10.3390/environments13030151
Leyva-Morales JB, Ramos-Ávila AY, Bastidas-Bastidas PdJ, Granados Amores J, Granados Amores E, González Ramírez J, Salas-Martínez F, Acevedo-Sandoval OA, Romo-Gómez C, Camacho-López C, et al. Vertebral Malformations in Fish from the Coast of Nayarit, Mexico, and Their Association with Organochlorine and Organophosphate Pesticides. Environments. 2026; 13(3):151. https://doi.org/10.3390/environments13030151
Chicago/Turabian StyleLeyva-Morales, José Belisario, Angélica Yomira Ramos-Ávila, Pedro de Jesús Bastidas-Bastidas, Jasmin Granados Amores, Esperanza Granados Amores, Javier González Ramírez, Fernando Salas-Martínez, Otilio Arturo Acevedo-Sandoval, Claudia Romo-Gómez, César Camacho-López, and et al. 2026. "Vertebral Malformations in Fish from the Coast of Nayarit, Mexico, and Their Association with Organochlorine and Organophosphate Pesticides" Environments 13, no. 3: 151. https://doi.org/10.3390/environments13030151
APA StyleLeyva-Morales, J. B., Ramos-Ávila, A. Y., Bastidas-Bastidas, P. d. J., Granados Amores, J., Granados Amores, E., González Ramírez, J., Salas-Martínez, F., Acevedo-Sandoval, O. A., Romo-Gómez, C., Camacho-López, C., González-Ramírez, C. A., Leyva-Camacho, L., & Cruz-Acevedo, E. (2026). Vertebral Malformations in Fish from the Coast of Nayarit, Mexico, and Their Association with Organochlorine and Organophosphate Pesticides. Environments, 13(3), 151. https://doi.org/10.3390/environments13030151








