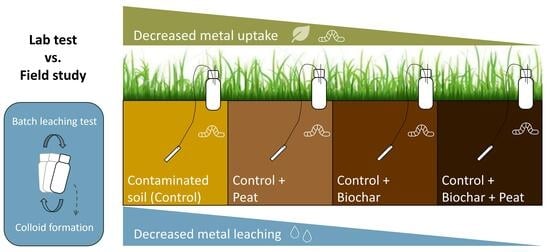
Amendment of Contaminated Soils with Biochar and Peat: Effects on Metal Solubility and Uptake in Grass and Earthworms in a Field Trial
Abstract
1. Introduction
2. Materials and Methods
2.1. Field Trial
| Soil Without Peat Addition | Soil with 1.5% Peat | Soil with 3% Peat | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Treatment | BC0P0 | BC3P0 | BC6P0 | BC0P1.5 | BC3P1.5 | BC6P1.5 | BC0P3 | BC3P3 | BC6P3 |
| TOC (%) * | 2.2 (0.10) | 4.7 (0.96) | 6.8 (0.83) | 3.4 (0.56) | 4.8 (0.12) | 6.7 (0.44) | 4.4 (0.48) | 5.6 (0.48) | 8.0 (0.09) |
| TC (%) * | 3.9 (0.11) | 6.1 (0.77) | 7.7 (0.44) | 4.3 (0.49) | 5.9 (0.69) | 7.7 (0.62) | 4.9 (0.35) | 6.5 (0.13) | 7.7 (0.09) |
| CEC (meq/100 g) * | 9.0 (0.20) | 9.6 (0.28) | 9.7 (0.33) | 10.4 (0.32) | 10.9 (0.32) | 10.6 (0.11) | 12.0 (0.32) | 11.8 (0.16) | 12.3 (0.07) |
| Soil pH in water * | 7.7 (0.14) | 7.7 (0.02) | 7.7 (0.04) | 7.7 (0.10) | 7.6 (0.12) | 8.5 (0.05) | 7.8 (0.21) | 8.3 (0.02) | 8.1 (0.08) |
| Ca (mg/kgdw) | 1630 (30.6) | 1690 (51.3) | 1663 (63.6) | 1883 (61.7) | 1890 (60.0) | 1810 (24.5) | 2160 (60.3) | 2050 (35.1) | 2063 (13.3) |
| K (mg/kgdw) | 84 (3.42) | 172 (7.02) | 205 (1.45) | 81 (3.27) | 162 (3.06) | 196 (2.52) | 86 (8.58) | 159 (3.28) | 203 (5.51) |
| Mg (mg/kgdw) | 53 (2.86) | 75 (1.70) | 91 (1.69) | 77 (1.24) | 106 (4.83) | 118 (1.15) | 103 (1.76) | 129 (3.53) | 159 (7.37) |
| Ba (mg/kgdw) | 297 (8.82) | 273 (3.33) | 287 (3.33) | 300 (20.82) | 270 (5.77) | 277 (12.02) | 293 (16.67) | 273 (3.33) | 273 (18.56) |
| Pb (mg/kgdw) | 473 (71.3) | 373 (13.3) | 513 (135.3) | 410 (32.2) | 433 (93.5) | 337 (29.6) | 503 (33.8) | 347 (38.4) | 327 (23.3) |
| Cu (mg/kgdw) | 237 (36.7) | 197 (16.7) | 167 (12.0) | 203 (12.0) | 180 (10.0) | 163 (3.33) | 207 (17.6) | 177 (14.5) | 197 (27.3) |
| Hg (mg/kgdw) | 1.7 (0.03) | 1.5 (0.06) | 1.6 (0.06) | 1.5 (0.06) | 1.5 (0.15) | 1.4 (0.03) | 1.5 (0.25) | 1.3 (0) | 1.3 (0) |
| Zn (mg/kgdw) | 417 (34.8) | 350 (15.3) | 340 (0) | 343 (13.3) | 403 (29.6) | 327 (8.82) | 337 (6.67) | 330 (10.0) | 340 (11.59) |
| Cd (mg/kgdw) | 0.42 (0.07) | 0.33 (0.02) | 0.37 (0.02) | 0.32 (0.02) | 0.42 (0.06) | 0.32 (0.02) | 0.32 (0.01) | 0.35 (0.01) | 0.35 (0.01) |
| Ni (mg/kgdw) | 13 (0.33) | 13 (0.67) | 13 (0.33) | 12 (0.33) | 12 (0) | 12 (0) | 13 (0.33) | 12 (0) | 12 (0.33) |
| As (mg/kgdw) | 8.3 (0.21) | 7.6 (0.06) | 7.5 (0.17) | 7.9 (0.20) | 7.6 (0.23) | 7.7 (0.46) | 7.6 (0.31) | 7.2 (0.23) | 7.1 (0.13) |
| Cr (mg/kgdw) | 16.3 (0.67) | 14.7 (0.33) | 15.0 (0.00) | 14.3 (0.33) | 14.3 (0.33) | 14.3 (0.33) | 15.3 (0.33) | 15.0 (0.00) | 15.3 (0.33) |
| Mo (mg/kgdw) | <2.1 | <2.2 | <2.5 | <2.2 | <2.2 | <2.3 | <2.4 | <3.2 | <2.5 |
| V (mg/kgdw) | 21 (0.33) | 20 (0.58) | 19 (0.33) | 20 (0.58) | 19 (0.33) | 19 (0.33) | 20 (0.33) | 19 (0.33) | 19 (0.58) |
2.2. Sampling and Analysis of Soil
2.3. Sampling and Analysis of In Situ Soil Solution
2.4. Sampling and Analysis of Grass
2.5. Metal Uptake in Earth Worms
2.6. Standardized Batch Test
2.7. Data Analyses
3. Results and Discussion
3.1. Biochar and Peat Effects on Metal Concentrations in In Situ Soil Solutions
3.2. Treatment Effects on Uptake of Metal(oid)s by Ryegrass and Earthworms
3.3. Using Standardized Batch Leaching Test to Evaluate Solubility of Metal(oid)s in Biochar- and Peat-Amended Soil
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Beesley, L.; Moreno-Jiménez, E.; Gomez-Eyles, J.L.; Harris, E.; Robinson, B.; Sizmur, T. A Review of Biochars’ Potential Role in the Remediation, Revegetation and Restoration of Contaminated Soils. Environ. Pollut. 2011, 159, 3269–3282. [Google Scholar] [CrossRef]
- Hilber, I.; Bastos, A.C.; Loureiro, S.; Soja, G.; Marsz, A.; Cornelissen, G.; Bucheli, T.D. The Different Faces of Biochar: Contamination Risk versus Remediation Tool. J. Environ. Eng. Landsc. Manag. 2017, 25, 86–104. [Google Scholar] [CrossRef]
- Ahmad, M.; Rajapaksha, A.U.; Lim, J.E.; Zhang, M.; Bolan, N.; Mohan, D.; Vithanage, M.; Lee, S.S.; Ok, Y.S. Biochar as a Sorbent for Contaminant Management in Soil and Water: A Review. Chemosphere 2014, 99, 19–33. [Google Scholar] [CrossRef] [PubMed]
- Pecha, B.; Hills, K.M.; Garcia-Pérez, M.; Hunt, J.; Miles, T.R.; Wilson, K.; Amonette, J.E. Biochar Production. In Biomass to Biochar: Maximizing the Carbon Value; Center for Sustaining Agriculture and Natural Resources: Wenatchee, WA, USA, 2022; Chapter 11. [Google Scholar]
- Ungureanu, N.; Vlăduț, N.V.; Biriș, S.Ș.; Gheorghiță, N.E.; Ionescu, M. Bio-mass Pyrolysis Pathways for Renewable Energy and Sustainable Resource Recovery: A Critical Review of Processes, Parameters, and Product Valorization. Sustainability 2025, 17, 7806. [Google Scholar] [CrossRef]
- Luo, L.; Wang, J.; Lv, J.; Liu, Z.; Sun, T.; Yang, Y.; Zhu, Y.G. Carbon Sequestration Strategies in Soil Using Biochar: Advances, Challenges, and Opportunities. Environ. Sci. Technol. 2023, 57, 11357–11372. [Google Scholar] [CrossRef]
- Papageorgiou, A.; Azzi, E.S.; Enell, A.; Sundberg, C. Biochar Produced from Wood Waste for Soil Remediation in Sweden: Carbon Sequestration and Other Environmental Impacts. Sci. Total Environ. 2021, 776, 145953. [Google Scholar] [CrossRef]
- O’Connor, D.; Peng, T.; Zhang, J.; Tsang, D.C.W.; Alessi, D.S.; Shen, Z.; Bolan, N.S.; Hou, D. Biochar Application for the Remediation of Heavy Metal Polluted Land: A Review of in Situ Field Trials. Sci. Total Environ. 2018, 619–620, 815–826. [Google Scholar] [CrossRef] [PubMed]
- Guo, M.; Song, W.; Tian, J. Biochar-Facilitated Soil Remediation: Mechanisms and Efficacy Variations. Front. Environ. Sci. 2020, 8, 521512. [Google Scholar] [CrossRef]
- Qiu, M.; Liu, L.; Ling, Q.; Cai, Y.; Yu, S.; Wang, S.; Fu, D.; Hu, B.; Wang, X. Biochar for the Removal of Contaminants from Soil and Water: A Review. Biochar 2022, 4, 19. [Google Scholar] [CrossRef]
- Long, X.X.; Yu, Z.N.; Liu, S.W.; Gao, T.; Qiu, R.L. A Systematic Review of Biochar Aging and the Potential Eco-Environmental Risk in Heavy Metal Contaminated Soil. J. Hazard. Mater. 2024, 472, 134345. [Google Scholar] [CrossRef]
- Majewska, M.; Hanaka, A. Biochar in the Bioremediation of Metal-Contaminated Soils. Agronomy 2025, 15, 273. [Google Scholar] [CrossRef]
- Bandara, T.; Franks, A.; Xu, J.; Bolan, N.; Wang, H.; Tang, C. Chemical and Biological Immobilization Mechanisms of Potentially Toxic Elements in Biochar-Amended Soils. Crit. Rev. Environ. Sci. Technol. 2020, 50, 903–978. [Google Scholar] [CrossRef]
- Chen, D.; Liu, X.; Bian, R.; Cheng, K.; Zhang, X.; Zheng, J.; Joseph, S.; Crowley, D.; Pan, G.; Li, L. Effects of Biochar on Availability and Plant Uptake of Heavy Metals—A Meta-Analysis. J. Environ. Manag. 2018, 222, 76–85. [Google Scholar] [CrossRef]
- Peng, X.; Deng, Y.; Peng, Y.; Yue, K. Effects of Biochar Addition on Toxic Element Concentrations in Plants: A Meta-Analysis. Sci. Total Environ. 2018, 616–617, 970–977. [Google Scholar] [CrossRef] [PubMed]
- Choppala, G.; Bolan, N.; Kunhikrishnan, A.; Bush, R. Differential Effect of Biochar upon Reduction-Induced Mobility and Bioavailability of Arsenate and Chromate. Chemosphere 2016, 144, 374–381. [Google Scholar] [CrossRef]
- Wang, H.T.; Ding, J.; Chi, Q.Q.; Li, G.; Pu, Q.; Xiao, Z.F.; Xue, X.M. The Effect of Biochar on Soil-Plant-Earthworm-Bacteria System in Metal(Loid) Contaminated Soil. Environ. Pollut. 2020, 263, 114610. [Google Scholar] [CrossRef]
- Xiao, R.; Liu, X.; Ali, A.; Chen, A.; Zhang, M.; Li, R.; Chang, H.; Zhang, Z. Bioremediation of Cd-Spiked Soil Using Earthworms (Eisenia Fetida): Enhancement with Biochar and Bacillus Megatherium Application. Chemosphere 2021, 264, 128517. [Google Scholar] [CrossRef]
- Gomez-Eyles, J.L.; Sizmur, T.; Collins, C.D.; Hodson, M.E. Effects of Biochar and the Earthworm Eisenia Fetida on the Bioavailability of Polycyclic Aromatic Hydrocarbons and Potentially Toxic Elements. Environ. Pollut. 2011, 159, 616–622. [Google Scholar] [CrossRef]
- ISO 21268-2:2019; Soil Quality—Leaching Procedures for Subsequent Chemical and Ecotoxicological Testing of Soil and Soil-like Materials Part 2: Batch Test Using a Liquid to Solid Ratio of 10 l/Kg Dry Matter. International Organization for Standardization: Geneva, Switzerland, 2019.
- Tiberg, C.; Smolders, E.; Fröberg, M.; Gustafsson, J.P.; Kleja, D.B. Combining a Standardized Batch Test with the Biotic Ligand Model to Predict Copper and Zinc Ecotoxicity in Soils. Environ. Toxicol. Chem. 2022, 41, 1540–1554. [Google Scholar] [CrossRef]
- Degryse, F.; Broos, K.; Smolders, E.; Merckx, E. Soil Solution Concentration of Cd and Zn Can Be Predicted with a CaCl2 Soil Extract. Eur. J. Soil Sci. 2003, 54, 149–157. [Google Scholar] [CrossRef]
- Beesley, L.; Moreno-Jiménez, E.; Gomez-Eyles, J.L. Effects of Biochar and Greenwaste Compost Amendments on Mobility, Bioavailability and Toxicity of Inorganic and Organic Contaminants in a Multi-Element Polluted Soil. Environ. Pollut. 2010, 158, 2282–2287. [Google Scholar] [CrossRef]
- Rijk, I.; Ekblad, A.; Dahlin, A.S.; Enell, A.; Larsson, M.; Leroy, P.; Kleja, D.B.; Tiberg, C.; Hallin, S.; Jones, C. Biochar and Peat Amendments Affect Nitrogen Retention, Microbial Capacity and Nitrogen Cycling Microbial Communities in a Metal and Polycyclic Aromatic Hydrocarbon Contaminated Urban Soil. Sci. Total Environ. 2024, 936, 173454. [Google Scholar] [CrossRef]
- Lian, F.; Xing, B. Black Carbon (Biochar) in Water/Soil Environments: Molecular Structure, Sorption, Stability, and Potential Risk. Environ. Sci. Technol. 2017, 51, 13517–13532. [Google Scholar] [CrossRef]
- Oleszczuk, P.; Zielińska, A.; Cornelissen, G. Stabilization of Sewage Sludge by Different Biochars towards Reducing Freely Dissolved Polycyclic Aromatic Hydrocarbons (PAHs) Content. Bioresour. Technol. 2014, 156, 139–145. [Google Scholar] [CrossRef] [PubMed]
- Kupryianchyk, D.; Hale, S.; Zimmerman, A.R.; Harvey, O.; Rutherford, D.; Abiven, S.; Knicker, H.; Schmidt, H.P.; Rumpel, C.; Cornelissen, G. Sorption of Hydrophobic Organic Compounds to a Diverse Suite of Carbonaceous Materials with Emphasis on Biochar. Chemosphere 2016, 144, 879–887. [Google Scholar] [CrossRef]
- Antonangelo, J.A.; Culman, S.; Zhang, H. Comparative Analysis and Prediction of Cation Exchange Capacity via Summation: Influence of Biochar Type and Nutrient Ratios. Front. Soil Sci. 2024, 4, 1371777. [Google Scholar] [CrossRef]
- EBC European Biochar Certificate. Guidelines for a Sustainable Production of Biochar; EBC European Biochar Certificate: Frick, Switzerland, 2024. [Google Scholar]
- Andriesse, J.P. Nature and Management of Tropical Peat Soils. FAO Soils Bull. 1988, 59, 48. [Google Scholar]
- ISO 11885:2007; Water Quality—Determination of Selected Elements by Inductively CCoupled Plasma Optical Emission Spectrometry (ICP-OES). International Organization for Standardization: Geneva, Switzerland, 2019.
- ISO 17294-2:2016; Water Quality—Application of Inductively Coupled Plasma Mass Spectrometry (ICP-MS), Part 2: Determination of Selected Elements Including Uranium Isotopes. International Organization for Standardization: Geneva, Switzerland, 2019.
- Swedish EPA. Tabell över Generella Riktvärden för Förorenad Mark (Table of Generic Guideline Values for Contaminated Soils in Sweden), Version 2.3. Available online: https://www.naturvardsverket.se/4a3dbd/globalassets/vagledning/fororenade-omraden/riktvarden/generella-riktvarden-for-fororenad-mark-2025.pdf (accessed on 16 July 2024).
- Mccoy, E.L. Commercial Amendments for Sand-Based Root Zones: Review and Interpretation. Horttechnology 2013, 23, 803–813. [Google Scholar] [CrossRef]
- Siregar, M.A.; Ma’as, A.; Nurudin, M. Determination of Cation Exchange Capacity and Analysis of Cation Availability in Hemic and Sapric Peat with Different Preparation and Extraction Methods. Ilmu Pertan. (Agric. Sci.) 2021, 6, 47–53. [Google Scholar] [CrossRef]
- ISO/IEC 17025:2017; General Requirements for the Competence of Testing and Calibration Laboratories. International Organization for Standardization: Geneva, Switzerland, 2017.
- ISO 17852:2008; Water Quality—Determination of Mercury—Method Using Atomic Fluorescence Spectrometry. International Organization for Standardization: Geneva, Switzerland, 2019.
- ISO 10304-1:2009; Water Quality—Determination of Dissolved Anions by Liquid Chromatography of Ions—Part 1: Determination of Bromide, Chloride, Fluoride, Nitrate, Nitrite, Phosphate and Sulfate. International Organization for Standardization: Geneva, Switzerland, 2019.
- ISO 6878:2004; Water Quality—Determination of Phosphorus—Ammonium Molybdate Spectrometric Method. International Organization for Standardization: Geneva, Switzerland, 2019.
- ISO 11268-2:2012; Soil Quality—Effects of Pollutants on Earthworms, Part 2: Determination of Effects on Reproduction of Eisenia fetida/Eisenia andrei and Other Earthworm Species. International Organization for Standardization: Geneva, Switzerland, 2019.
- Gustafsson, J.P. Visual MINTEQ, Version 3.1. Stockholm, Sweden, 2013. Available online: https://vminteq.com/ (accessed on 13 November 2025).
- Gustafsson, J.P. Modeling the Acid-Base Properties and Metal Complexation of Humic Substances with the Stockholm Humic Model. J. Colloid Interface Sci. 2001, 244, 102–112. [Google Scholar] [CrossRef]
- Essington, M.E. Soil and Water Chemistry—An Integrative Approach; CRC Press: Boca Raton, FL, USA, 2004; ISBN 0-8493-1258-2. [Google Scholar]
- Singh, B.P.; Cowie, A.L.; Smernik, R.J. Biochar Carbon Stability in a Clayey Soil as a Function of Feedstock and Pyrolysis Temperature. Environ. Sci. Technol. 2012, 46, 11770–11778. [Google Scholar] [CrossRef]
- Feng, Z.; Fan, Z.; Song, H.; Li, K.; Lu, H.; Liu, Y.; Cheng, F. Biochar Induced Changes of Soil Dissolved Organic Matter: The Release and Adsorption of Dissolved Organic Matter by Biochar and Soil. Sci. Total Environ. 2021, 783, 147091. [Google Scholar] [CrossRef]
- Lehmann, J.; Liang, B.; Solomon, D.; Lerotic, M.; Luizão, F.; Kinyangi, J.; Schäfer, T.; Wirick, S.; Jacobsen, C. Near-Edge X-Ray Absorption Fine Structure (NEXAFS) Spectroscopy for Mapping Nano-Scale Distribution of Organic Carbon Forms in Soil: Application to Black Carbon Particles. Glob. Biogeochem. Cycles 2005, 19, GB1013. [Google Scholar] [CrossRef]
- Pietikäinen, J.; Kiikkilä, O.; Fritze, H. Charcoal as a Habitat for Microbes and Its Effect on the Microbial Community of the Underlying Humus. Oikos 2000, 89, 231–242. [Google Scholar] [CrossRef]
- Karlsson, T.; Persson, P.; Skyllberg, U. Complexation of Copper(II) in Organic Soils and in Dissolved Organic Matter—EXAFS Evidence for Chelate Ring Structures. Environ. Sci. Technol. 2006, 40, 2623–2628. [Google Scholar] [CrossRef]
- Skyllberg, U.; Bloom, P.R.; Qian, J.; Lin, C.M.; Bleam, W.F. Complexation of Mercury(II) in Soil Organic Matter: EXAFS Evidence for Linear Two-Coordination with Reduced Sulfur Groups. Environ. Sci. Technol. 2006, 40, 4174–4180. [Google Scholar] [CrossRef]
- Chaudhuri, S.; Sigmund, G.; Bone, S.E.; Kumar, N.; Hofmann, T. Mercury Removal from Contaminated Water by Wood-Based Biochar Depends on Natural Organic Matter and Ionic Composition. Environ. Sci. Technol. 2022, 56, 11354–11362. [Google Scholar] [CrossRef] [PubMed]
- Thomas, E.; Borchard, N.; Sarmiento, C.; Atkinson, R.; Ladd, B. Key Factors Determining Biochar Sorption Capacity for Metal Contaminants: A Literature Synthesis. Biochar 2020, 2, 151–163. [Google Scholar] [CrossRef]
- Das, S.K. Adsorption and Desorption Capacity of Different Metals Influenced by Biomass Derived Biochar. Environ. Syst. Res. 2024, 13, 5. [Google Scholar] [CrossRef]
- Zaccone, C.; Plaza, C.; Ciavatta, C.; Miano, T.M.; Shotyk, W. Advances in the Determination of Humification Degree in Peat Since Achard (1786): Applications in Geochemical and Paleoenvironmental Studies. Earth Sci. Rev. 2018, 185, 163–178. [Google Scholar] [CrossRef]
- Strawn, D.G.; Sparks, D.L. Effects of Soil Organic Matter on the Kinetics and Mechanisms of Pb(II) Sorption and Desorption in Soil. Soil Sci. Soc. Am. J. 2000, 64, 144–156. [Google Scholar] [CrossRef]
- Liu, Q.; Luo, J.; Tang, J.; Chen, Z.; Chen, Z.; Lin, Q. Remediation of Cadmium and Lead Contaminated Soils Using Fe-OM Based Materials. Chemosphere 2022, 307, 135853. [Google Scholar] [CrossRef] [PubMed]
- Du, H.; Huang, Q.; Lei, M.; Tie, B. Sorption of Pb(II) by Nanosized Ferrihydrite Organo-Mineral Composites Formed by Adsorption versus Coprecipitation. ACS Earth Space Chem. 2018, 2, 556–564. [Google Scholar] [CrossRef]
- Cappuyns, V. Barium (Ba) Leaching from Soils and Certified Reference Materials. Appl. Geochem. 2018, 88, 68–84. [Google Scholar] [CrossRef]
- Younis, S.A.; El-Salamony, R.A.; Tsang, Y.F.; Kim, K.H. Use of Rice Straw-Based Biochar for Batch Sorption of Barium/Strontium from Saline Water: Protection against Scale Formation in Petroleum/Desalination Industries. J. Clean. Prod. 2020, 250, 119442. [Google Scholar] [CrossRef]
- Anderson, M.A.; Malotky, D.T. The Adsorption of Protolyzable Anions on Hydrous Oxides at the Isoelectric PH. J. Colloid Interface Sci. 1979, 72, 413–427. [Google Scholar] [CrossRef]
- Zama, E.F.; Reid, B.J.; Arp, H.P.H.; Sun, G.X.; Yuan, H.Y.; Zhu, Y.G. Advances in Research on the Use of Biochar in Soil for Remediation: A Review. J. Soils Sediments 2018, 18, 2433–2450. [Google Scholar] [CrossRef]
- Gustafsson, J.P.; Persson, I.; Oromieh, A.G.; Van Schaik, J.W.J.; Sjöstedt, C.; Kleja, D.B. Chromium(III) Complexation to Natural Organic Matter: Mechanisms and Modeling. Environ. Sci. Technol. 2014, 48, 1753–1761. [Google Scholar] [CrossRef] [PubMed]
- Jardine, P.M.; Fendorf, S.E.; Mayes, M.A.; Larsen, I.L.; Brooks, S.C.; Bailey, W.B. Fate and Transport of Hexavalent Chromium in Undisturbed Heterogeneous Soil. Environ. Sci. Technol. 1999, 33, 2939–2944. [Google Scholar] [CrossRef]
- Kuperman, R.G.; Heckai, R.T.C.; Simini, M.; PHillips, C.T.; Speicher, J.A.; Barclift, D.J. Toxicity Benchmarks for Antimony, Barium, and Beryllium Determined Using Reproduction Endpoints for Folsomia candida, Eisenia fetida, and Enchytraeus cryptus. Environ. Toxicol. Chem. 2006, 25, 754–762. [Google Scholar] [CrossRef]
- Lamb, D.T.; Matanitobua, V.P.; Palanisami, T.; Megharaj, M.; Naidu, R. Bioavailability of Barium to Plants and Invertebrates in Soils Contaminated by Barite. Environ. Sci. Technol. 2013, 47, 4670–4676. [Google Scholar] [CrossRef]
- Löv, Å.; Larsbo, M.; Sjöstedt, C.; Cornelis, G.; Gustafsson, J.P.; Kleja, D.B. Evaluating the Ability of Standardised Leaching Tests to Predict Metal(Loid) Leaching from Intact Soil Columns Using Size-Based Elemental Fractionation. Chemosphere 2019, 222, 453–460. [Google Scholar] [CrossRef]
- Löv, Å.; Cornelis, G.; Larsbo, M.; Persson, I.; Sjöstedt, C.; Gustafsson, J.P.; Boye, K.; Kleja, D.B. Particle- and Colloid-Facilitated Pb Transport in Four Historically Contaminated Soils—Speciation and Effect of Irrigation Intensity. Appl. Geochem. 2018, 96, 327–338. [Google Scholar] [CrossRef]
- Sjöstedt, C.; Löv, A.; Olivecrona, Z.; Boye, K.; Kleja, D.B. Improved Geochemical Modeling of Lead Solubility in Contaminated Soils by Considering Colloidal Fractions and Solid Phase EXAFS Speciation. Appl. Geochem. 2018, 92, 110–120. [Google Scholar] [CrossRef]
- Benjamin, M.M.; Leckie, J.O. Multiple-Site Adsorption of Cd, Cu, Zn, and Pb on Amorphous Iron Oxyhydroxide. J. Colloid Interface Sci. 1981, 79, 209–221. [Google Scholar] [CrossRef]
- Triffeau, C.; Lützenkirchen, J.; Behra, P. Hg Adsorption to HFO and Quarts. J. Colloid Interface Sci. 1995, 172, 82–93. [Google Scholar]
- Kleja, D.B.; Nakata, S.; Persson, I.; Gustafsson, J.P. Silver(I) Binding Properties of Organic Soil Materials Are Different from Those of Isolated Humic Substances. Environ. Sci. Technol. 2016, 50, 7453–7460. [Google Scholar] [CrossRef] [PubMed]





| Biochar | Peat | |
|---|---|---|
| Moisture (%) | 23.7 | |
| TOC (%) | 86.1 | 49.6 |
| pH | 8.6 | 5.9 * |
| BET m2/g ** | 204.5 | |
| Ash content (550 °C) % (w/w) | 9.8 | |
| H/C ratio (molar) | 0.07 | |
| O/C ratio (molar) | 0.04 | |
| CEC (meq/100 g) *** | 15.8 (4.2) | 96.7 (3.3) |
| DOC (mg/L) **** | <0.5 mg/L | |
| Humification degree (von Post) | 4–5 |
| Soil Solution | Batch Eluate | Grass | Earthworm | |||||
|---|---|---|---|---|---|---|---|---|
| BC | P | BC | P | BC | P | BC | P | |
| pH | n.s. | n.s. | n.s. | n.s. | ||||
| DOC | − | n.s. | − | + | ||||
| Fe | − | n.s. | − | + | ||||
| Al | n.s. | n.s. | − | + | ||||
| Ba | + | n.s. | − | n.s. | − | − | ||
| Cd | − | n.s. | − | + | − | n.s. | n.s. | n.s. |
| Cu | − | n.s. | − | + | − | − | − | − |
| Hg | − | − | − | + | ||||
| Ni | − | n.s | − | + | − | − | − | − |
| Pb | n.s | n.s | − | + | n.s. | − | − | − |
| Zn | − | n.s | − | + | − | − | − | − |
| As | + | − | − | + | − | n.s. | − | + |
| Cr-tot | + | − | n.s. | −/+ | n.s. | n.s. | n.s. | n.s. |
| Cr(VI) | + | − | ||||||
| Mo | + | − | n.s. | − | + | + | n.s. | n.s. |
| V | + | − | − | + | − | − | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tiberg, C.; Kleja, D.B.; Sjöstedt, C.; Fröberg, M.; Rijk, I.; Dahlin, A.S.; Larsson, M.; Ekblad, A.; Sjöberg, V.; Enell, A. Amendment of Contaminated Soils with Biochar and Peat: Effects on Metal Solubility and Uptake in Grass and Earthworms in a Field Trial. Environments 2025, 12, 447. https://doi.org/10.3390/environments12110447
Tiberg C, Kleja DB, Sjöstedt C, Fröberg M, Rijk I, Dahlin AS, Larsson M, Ekblad A, Sjöberg V, Enell A. Amendment of Contaminated Soils with Biochar and Peat: Effects on Metal Solubility and Uptake in Grass and Earthworms in a Field Trial. Environments. 2025; 12(11):447. https://doi.org/10.3390/environments12110447
Chicago/Turabian StyleTiberg, Charlotta, Dan B. Kleja, Carin Sjöstedt, Mats Fröberg, Ingrid Rijk, A. Sigrun Dahlin, Maria Larsson, Alf Ekblad, Viktor Sjöberg, and Anja Enell. 2025. "Amendment of Contaminated Soils with Biochar and Peat: Effects on Metal Solubility and Uptake in Grass and Earthworms in a Field Trial" Environments 12, no. 11: 447. https://doi.org/10.3390/environments12110447
APA StyleTiberg, C., Kleja, D. B., Sjöstedt, C., Fröberg, M., Rijk, I., Dahlin, A. S., Larsson, M., Ekblad, A., Sjöberg, V., & Enell, A. (2025). Amendment of Contaminated Soils with Biochar and Peat: Effects on Metal Solubility and Uptake in Grass and Earthworms in a Field Trial. Environments, 12(11), 447. https://doi.org/10.3390/environments12110447