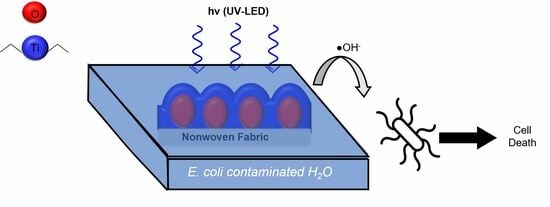
TiO2-Coated Meltblown Nonwoven Fabrics Prepared via Atomic Layer Deposition for the Inactivation of E. coli as a Model Photocatalytic Drinking Water Treatment System
Abstract
1. Introduction
2. Materials and Methods
2.1. Bacterial Stock Preparation
2.2. Preparation of Meltblown Nonwoven Fabrics
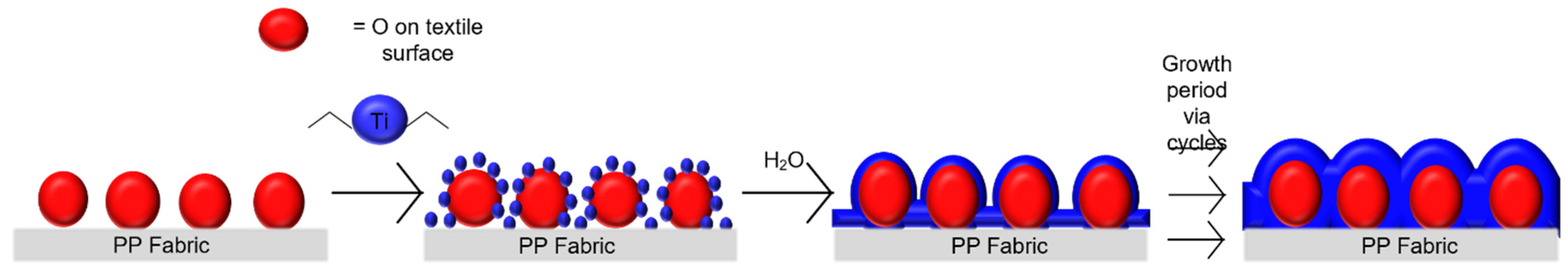
2.3. Synthesis of TiO2 Films
2.4. Photocatalytic Studies
2.4.1. Ultraviolet Light-Emitting Diode Irradiation
2.4.2. Experimental Conditions
2.4.3. Inactivation Kinetics
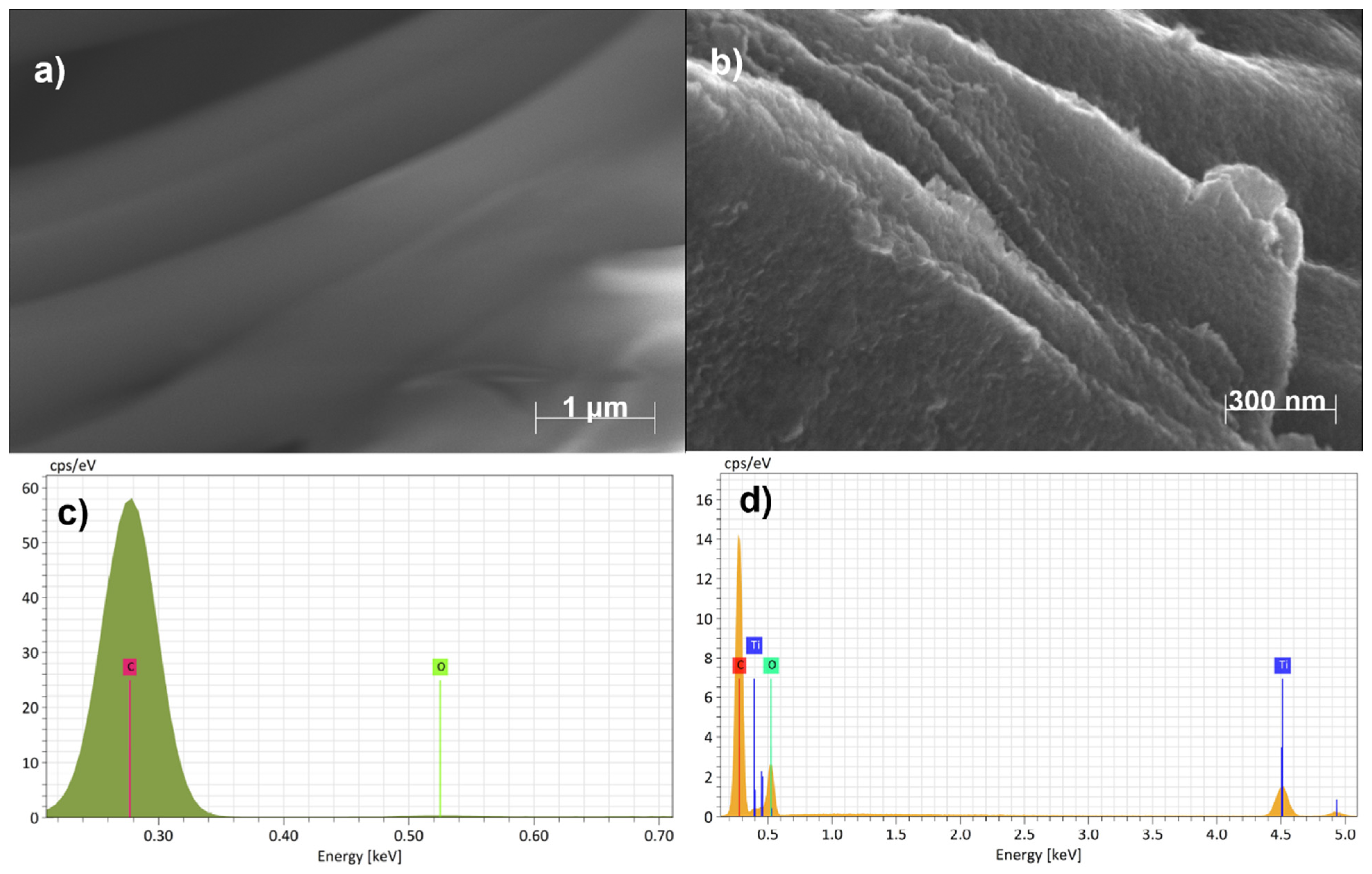
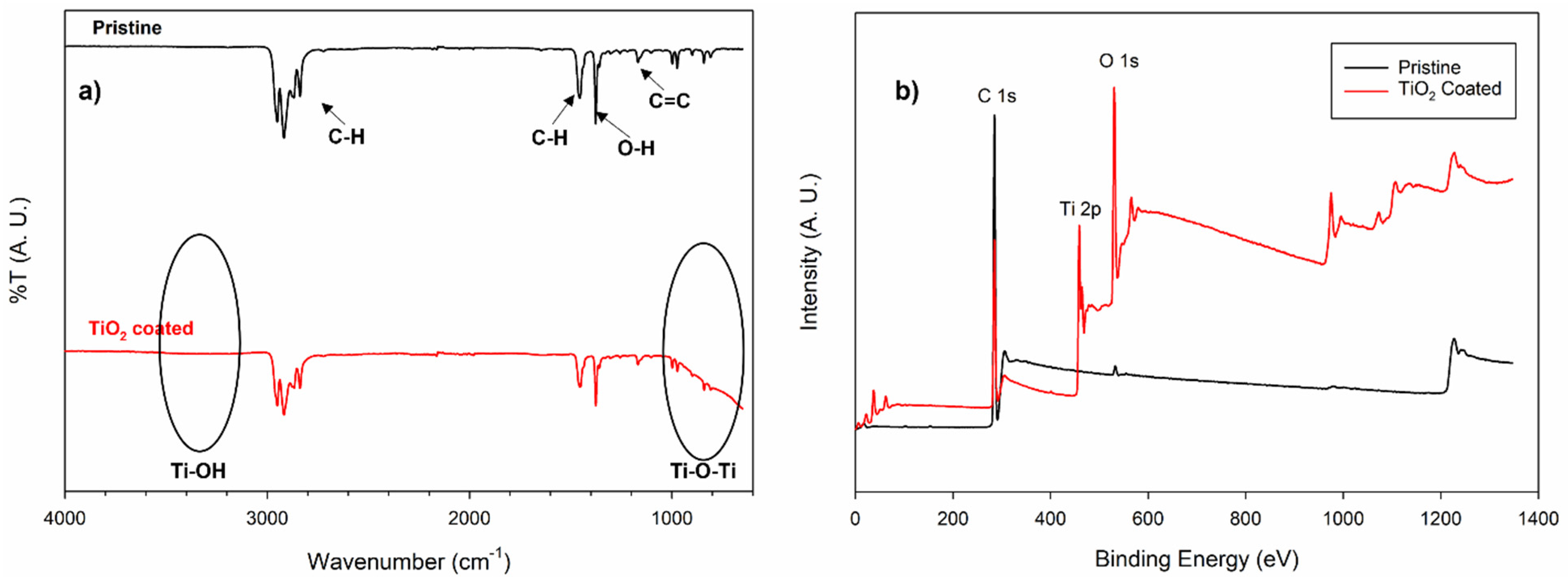
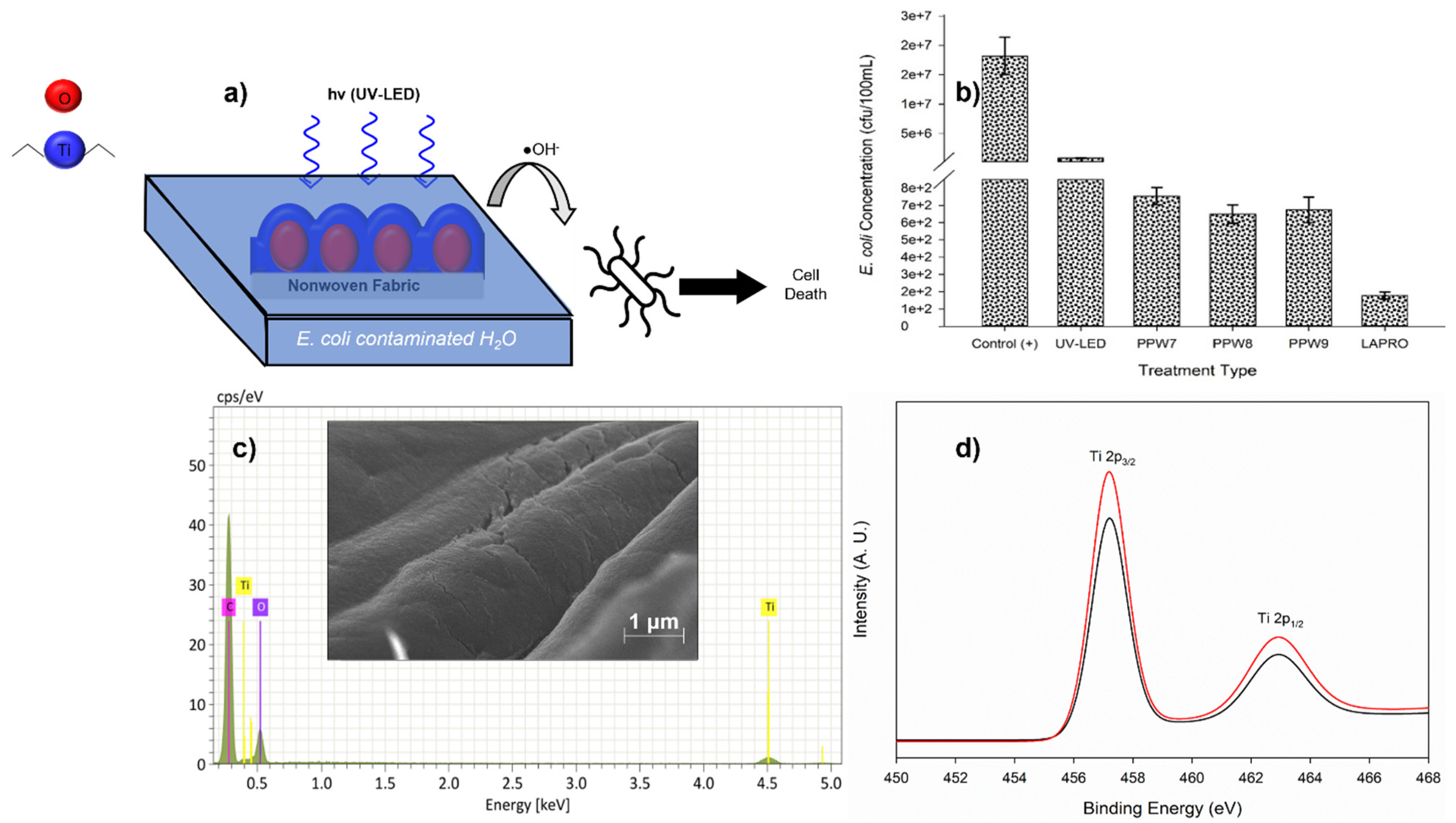
3. Results and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Odonkor, S.T.; Ampofo, J.K. Escherichia coli as an Indicator of Bacteriological Quality of Water: An Overview. Microbiol. Res. 2013, 4, e2. [Google Scholar] [CrossRef]
- Vietro, N.D.; Tursi, A.; Beneduci, A.; Chidichimo, F.; Milella, A.; Fracassi, F.; Chatzisymeon, E.; Chidichimo, G. Photocatalytic Inactivation of Escherichia coli Bacteria in Water Using Low Pressure Plasma Deposited TiO2 Cellulose Fabric. Photochem. Photobiol. Sci. 2019, 18, 2248–2258. [Google Scholar] [CrossRef] [PubMed]
- Park, J.; Kettleson, E.; An, W.-J.; Tang, Y.J.; Biswas, P. Inactivation of E. coli in Water Using Photocatalytic, Nanostructured Films Synthesized by Aerosol Routes. Catalysts 2013, 3, 247–260. [Google Scholar] [CrossRef]
- Ajiboye, T.O.; Babalola, S.O.; Onwudiwe, D.C. Photocatalytic Inactivation as a Method of Elimination of E. coli from Drinking Water. Appl. Sci. 2021, 11, 1313. [Google Scholar] [CrossRef]
- Khani, M.; Amin, N.A.S.; Hosseini, S.N.; Heidarrezaei, M. Kinetics Study of the Photocatalytic Inactivation of Escherichia coli. IJNBM 2016, 6, 139. [Google Scholar] [CrossRef]
- Ribeiro, M.A.; Cruz, J.M.; Montagnolli, R.N.; Bidoia, E.D.; Lopes, P.R.M. Photocatalytic and Photoelectrochemical Inactivation of Escherichia coli and Staphylococcus aureus. Water Supply 2014, 15, 107–113. [Google Scholar] [CrossRef]
- Dunlop, P.S.M.; Ciavola, M.; Rizzo, L.; McDowell, D.A.; Byrne, J.A. Effect of Photocatalysis on the Transfer of Antibiotic Resistance Genes in Urban Wastewater. Catal. Today 2015, 240, 55–60. [Google Scholar] [CrossRef]
- Friedman, N.D.; Temkin, E.; Carmeli, Y. The Negative Impact of Antibiotic Resistance. Clin. Microbiol. Infect. 2016, 22, 416–422. [Google Scholar] [CrossRef]
- Benabbou, A.K.; Derriche, Z.; Felix, C.; Lejeune, P.; Guillard, C. Photocatalytic Inactivation of Escherischia Coli: Effect of Concentration of TiO2 and Microorganism, Nature, and Intensity of UV Irradiation. Appl. Catal. B Environ. 2007, 76, 257–263. [Google Scholar] [CrossRef]
- Aragon, A.G.; Kierulf-Vieira, W.; Łęcki, T.; Zarębska, K.; Widera-Kalinowska, J.; Skompska, M. Synthesis and Application of N-Doped TiO2/CdS/Poly(1,8-Diaminocarbazole) Composite for Photocatalytic Degradation of 4-Chlorophenol under Visible Light. Electrochim. Acta 2019, 314, 73–80. [Google Scholar] [CrossRef]
- Badhe, R.A.; Ansari, A.; Garje, S.S. Study of Optical Properties of TiO2 Nanoparticles and CdS@TiO2 Nanocomposites and Their Use for Photocatalytic Degradation of Rhodamine B under Natural Light Irradiation. Bull. Mater. Sci. 2021, 44, 11. [Google Scholar] [CrossRef]
- Madkour, M.; Allam, O.G.; Abdel Nazeer, A.; Amin, M.O.; Al-Hetlani, E. CeO2-Based Nanoheterostructures with p–n and n–n Heterojunction Arrangements for Enhancing the Solar-Driven Photodegradation of Rhodamine 6G Dye. J. Mater. Sci. Mater. Electron. 2019, 30, 10857–10866. [Google Scholar] [CrossRef]
- He, Z.; Sun, C.; Yang, S.; Ding, Y.; He, H.; Wang, Z. Photocatalytic Degradation of Rhodamine B by Bi2WO6 with Electron Accepting Agent under Microwave Irradiation: Mechanism and Pathway. J. Hazard. Mater. 2009, 162, 1477–1486. [Google Scholar] [CrossRef]
- Olagunju, M.O.; Zahran, E.M.; Reed, J.M.; Zeynaloo, E.; Shukla, D.; Cohn, J.L.; Surnar, B.; Dhar, S.; Bachas, L.G.; Knecht, M.R. Halide Effects in BiVO4/BiOX Heterostructures Decorated with Pd Nanoparticles for Photocatalytic Degradation of Rhodamine B as a Model Organic Pollutant. ACS Appl. Nano Mater. 2021, 4, 3262–3272. [Google Scholar] [CrossRef]
- Bessekhouad, Y.; Chaoui, N.; Trzpit, M.; Ghazzal, N.; Robert, D.; Weber, J.V. UV–Vis versus Visible Degradation of Acid Orange II in a Coupled CdS/TiO2 Semiconductors Suspension. J. Photochem. Photobiol. A Chem. 2006, 183, 218–224. [Google Scholar] [CrossRef]
- Choi, H.; Stathatos, E.; Dionysiou, D.D. Photocatalytic TiO2 Films and Membranes for the Development of Efficient Wastewater Treatment and Reuse Systems. Desalination 2007, 202, 199–206. [Google Scholar] [CrossRef]
- Baghdadi, A.M.; Saddiq, A.A.; Aissa, A.; Algamal, Y.; Khalil, N.M. Structural Refinement and Antimicrobial Activity of Aluminum Oxide Nanoparticles. J. Ceram. Soc. Jpn. 2022, 130, 257–263. [Google Scholar] [CrossRef]
- Akyildiz, H.I.; Diler, S.; Islam, S. Evaluation of TiO2 and ZnO Atomic Layer Deposition Coated Polyamide 66 Fabrics for Photocatalytic Activity and Antibacterial Applications. J. Vac. Sci. Technol. A 2021, 39, 022405. [Google Scholar] [CrossRef]
- Pham, K.; Pelisset, S.; Kinnunen, N.; Karvinen, P.; Hakala, T.K.; Saarinen, J.J. Controlled Photocatalytic Activity of TiO2 Inverse Opal Structures with Atomic Layer Deposited (ALD) Metal Oxide Thin Films. Mater. Chem. Phys. 2022, 277, 125533. [Google Scholar] [CrossRef]
- Islam, S.; Akyildiz, H.I. Atomic Layer Deposition of TiO2 Thin Films on Glass Fibers for Enhanced Photocatalytic Activity. J. Mater. Sci. Mater. Electron. 2022, 33, 18002–18013. [Google Scholar] [CrossRef]
- Li, L.; Yu, P.; Li, Y.; Wu, X.; Li, W.; Cheng, X. A Facile Approach to Fabricating Antibacterial Textile with High Efficiency and Compact Process. Adv. Mater. Interfaces 2021, 8, 2101197. [Google Scholar] [CrossRef]
- Popescu, M.C.; Ungureanu, C.; Buse, E.; Nastase, F.; Tucureanu, V.; Suchea, M.; Draga, S.; Popescu, M.A. Antibacterial Efficiency of Cellulose-Based Fibers Covered with ZnO and Al2O3 by Atomic Layer Deposition. Appl. Surf. Sci. 2019, 481, 1287–1298. [Google Scholar] [CrossRef]
- Wang, Z.; Zhang, L.; Liu, Z.; Sang, L.; Yang, L.; Chen, Q. The Antibacterial Polyamide 6-ZnO Hierarchical Nanofibers Fabricated by Atomic Layer Deposition and Hydrothermal Growth. Nanoscale Res. Lett. 2017, 12, 421. [Google Scholar] [CrossRef] [PubMed]
- Nam, T.; Kim, J.M.; Kim, M.K.; Kim, H.; Kim, W.H. Low-Temperature Atomic Layer Deposition of TiO2, Al2O3, and ZnO Thin Films. J. Korean Phys. Soc. 2011, 59, 452–457. [Google Scholar] [CrossRef]
- Aghaee, M.; Maydannik, P.S.; Johansson, P.; Kuusipalo, J.; Creatore, M.; Homola, T.; Cameron, D.C. Low Temperature Temporal and Spatial Atomic Layer Deposition of TiO2 Films. J. Vac. Sci. Technol. A 2015, 33, 041512. [Google Scholar] [CrossRef]
- Wu, P.; Imlay, J.A.; Shang, J.K. Mechanism of Escherichia coli Inactivation on Palladium-Modified Nitrogen-Doped Titanium Dioxide. Biomaterials 2010, 31, 7526–7533. [Google Scholar] [CrossRef]
| Fabric Type | Catalyst | External Light | Log Inactivation |
|---|---|---|---|
| PPW7 | TiO2 | UV-LED | 4.40 ± 0.043 |
| PPW8 | TiO2 | UV-LED | 4.40 ± 0.032 |
| PPW9 | TiO2 | UV-LED | 4.40 ± 0.041 |
| Laprotex™ | TiO2 | UV-LED | 5.00 ± 0.031 |
| - | - | UV-LED | 1.35 ± 0.0422 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aragon, A.G.; Cárdenas Sánchez, J.A.; Zimeri, C.; Shim, E.; Fang, X.; Young, K.R.L. TiO2-Coated Meltblown Nonwoven Fabrics Prepared via Atomic Layer Deposition for the Inactivation of E. coli as a Model Photocatalytic Drinking Water Treatment System. Environments 2024, 11, 92. https://doi.org/10.3390/environments11050092
Aragon AG, Cárdenas Sánchez JA, Zimeri C, Shim E, Fang X, Young KRL. TiO2-Coated Meltblown Nonwoven Fabrics Prepared via Atomic Layer Deposition for the Inactivation of E. coli as a Model Photocatalytic Drinking Water Treatment System. Environments. 2024; 11(5):92. https://doi.org/10.3390/environments11050092
Chicago/Turabian StyleAragon, Alexander G., Jaime A. Cárdenas Sánchez, Carlos Zimeri, Eunkyoung Shim, Xiaomeng Fang, and Kyana R. L. Young. 2024. "TiO2-Coated Meltblown Nonwoven Fabrics Prepared via Atomic Layer Deposition for the Inactivation of E. coli as a Model Photocatalytic Drinking Water Treatment System" Environments 11, no. 5: 92. https://doi.org/10.3390/environments11050092
APA StyleAragon, A. G., Cárdenas Sánchez, J. A., Zimeri, C., Shim, E., Fang, X., & Young, K. R. L. (2024). TiO2-Coated Meltblown Nonwoven Fabrics Prepared via Atomic Layer Deposition for the Inactivation of E. coli as a Model Photocatalytic Drinking Water Treatment System. Environments, 11(5), 92. https://doi.org/10.3390/environments11050092