Plant-Wide Models for Optimizing the Operation and Maintenance of BTEX-Contaminated Wastewater Treatment and Reuse
Abstract
1. Introduction
2. Materials and Methods
2.1. Mathematical Model Development
2.1.1. Biodegradation
2.1.2. Gas–Liquid Transfer
2.1.3. Adsorption on Granular Activated Carbon
2.2. Model Configuration
3. Results and Discussion
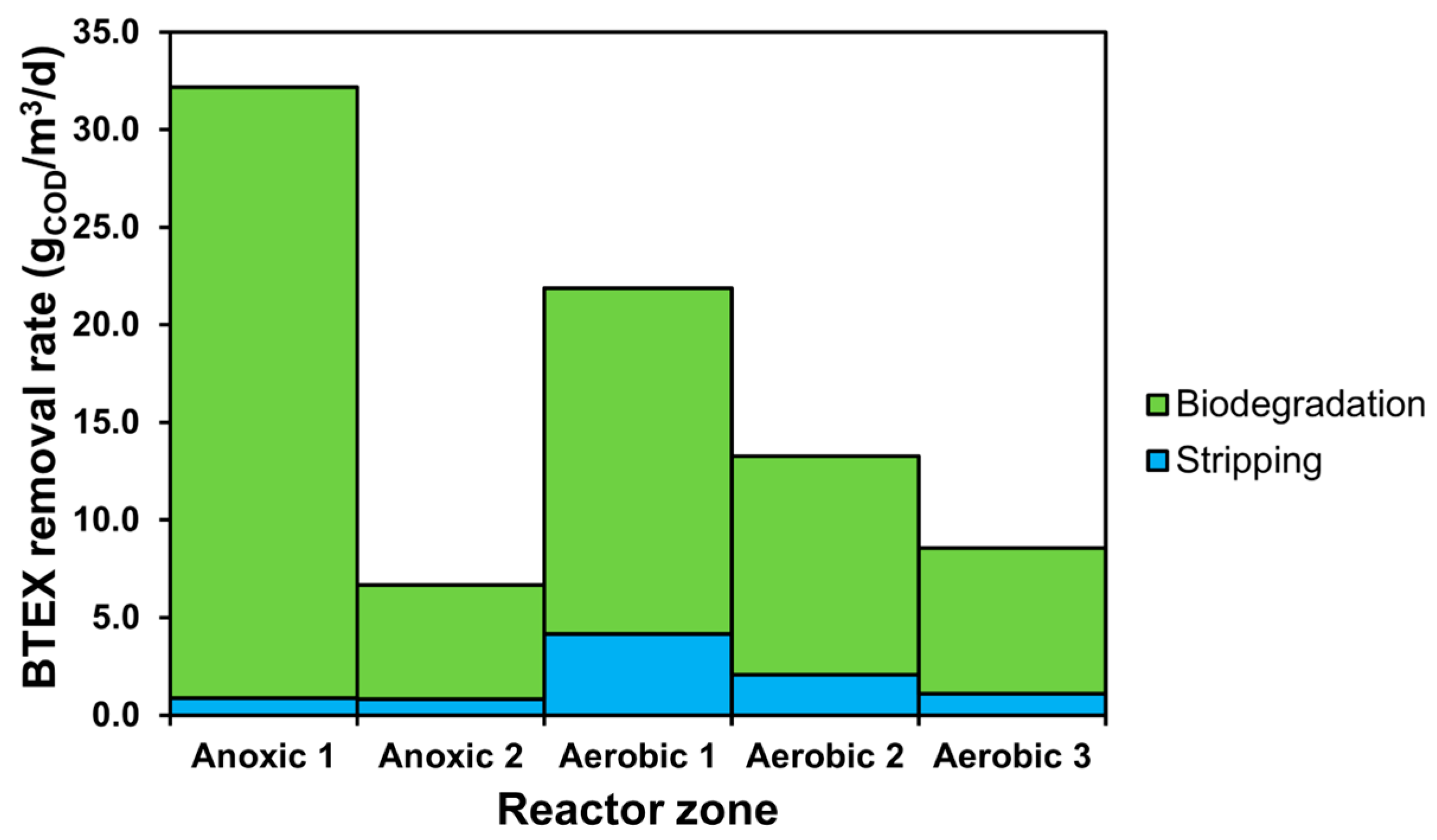
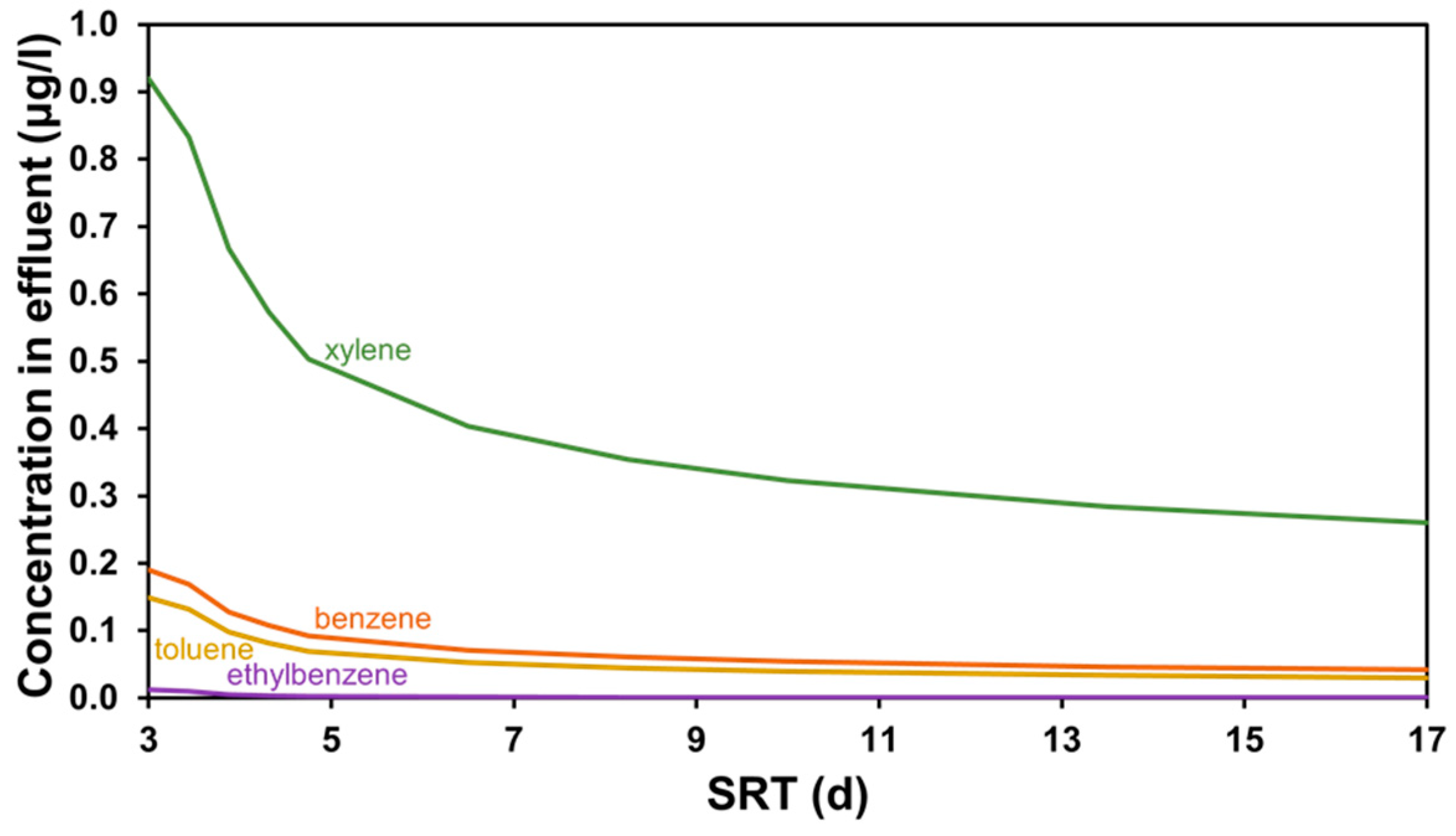
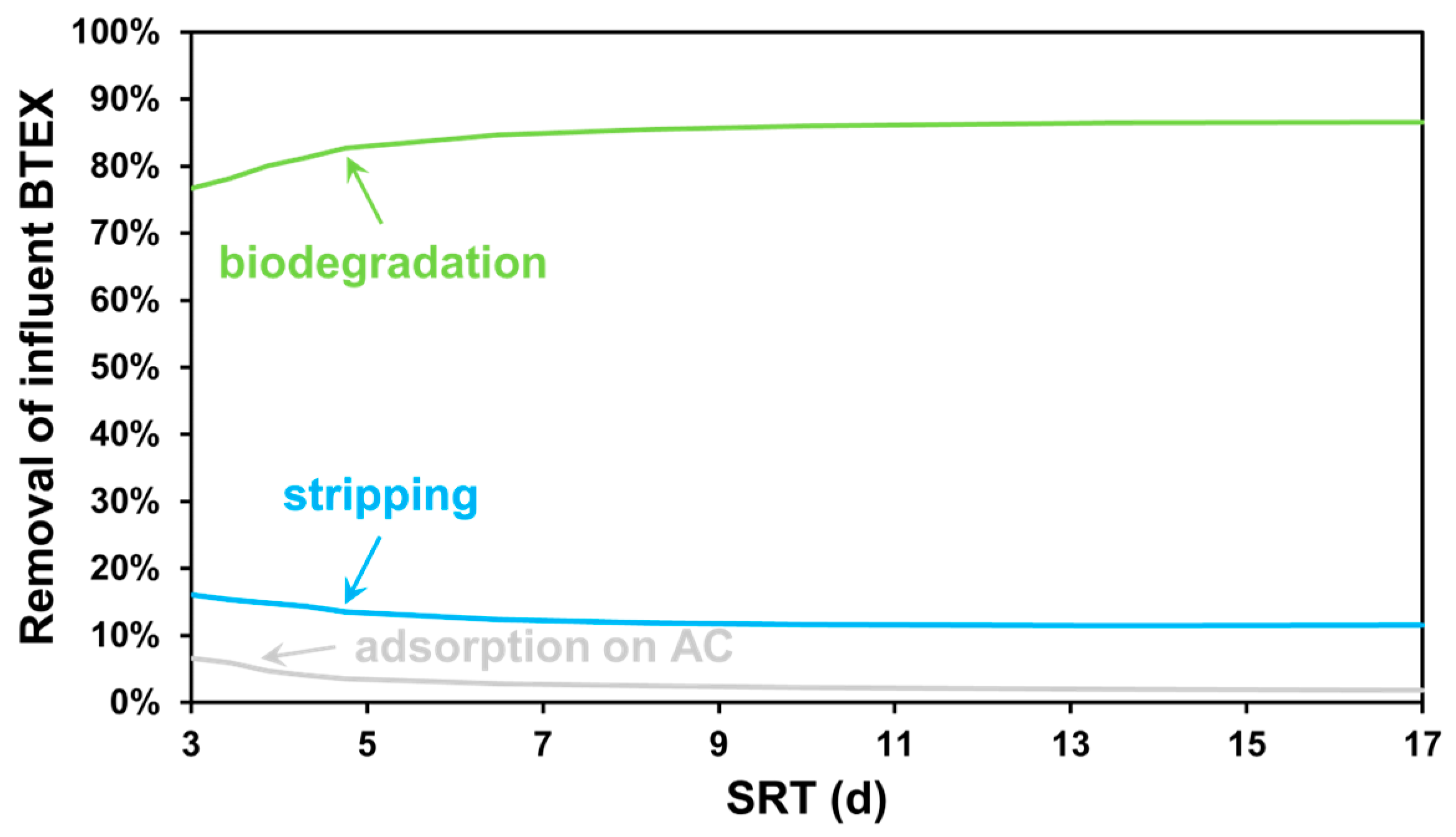
3.1. SRT-Based Scenarios
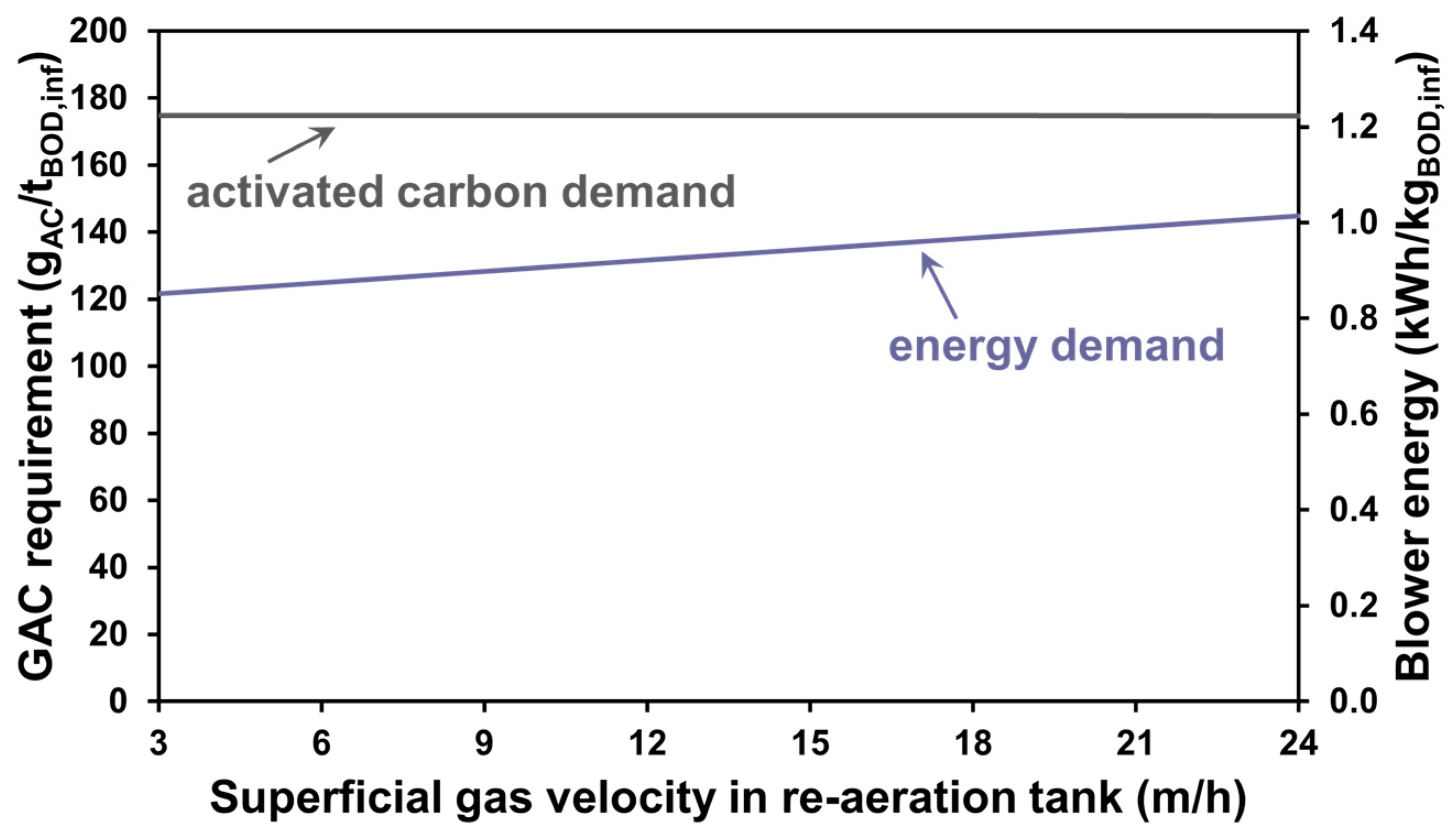
3.2. The Effect of Aeration Intensity
3.3. GAC Operational Strategies
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Nomenclature
| α | alpha (wastewater/clean water) correction factor for mass transfer coefficient |
| abub | specific contact area between the gas bubble surface and liquid phase [m2 m−3] |
| asur | specific contact area between the surface gas and liquid phase [m2 m−3] |
| Adiff,sp | area per diffuser [m2] |
| Ar | liquid surface [m2] |
| β | beta (wastewater/clean water) correction factor for the saturation concentration |
| BTC | TOC breakthrough capacity (in concentration unit) [gC m−3] |
| BTCm | TOC adsorption capacity (at breakpoint, in mass fraction unit) [gC gAC−1] |
| Cmid | midpoint concentration of breakthrough curve, with asymmetry correction [gC m−3] |
| Cmid,symm | midpoint concentration of curve, without asymmetry correction [gC m−3] |
| coefflead,h,diff | leading coefficient in a diffuser submergence correction term [m−1] |
| coefflin,h,diff | linear coefficient in a diffuser submergence correction term [m−1] |
| dbub | bubble Sauter mean diameter [m] |
| ddiff | diffuser density [m2 m−2] |
| Di,25 | diffusion coefficient of gas state variable i in water [m2 d−1] |
| divd,diff | divisor value in a diffuser density correction term [m2 m−2] |
| ε | gas hold-up [m3gas m−3] |
| EQC,ad,total | carbon equivalent for all adsorbed components on a GAC bed [g C m−3] |
| expSSOTE | exponent in SSOTE correlation [d m−3gas] |
| F | diffuser fouling factor |
| Fac | replaced activated carbon mass flow [g d−1] |
| fcover | covered fraction of the reactor surface |
| FGi | mass flow of gas phase state variable i [g d−1] |
| fh,sat,eff | effective saturation depth fraction |
| fkL,i | fraction in the liquid side for the mass transfer of gas state variable i |
| FLi | mass flow of liquid phase state variable i [g d−1] |
| fwave | waviness factor |
| Gi | concentration of gas phase state variable i in off-gas, per liquid volume [g m−3] |
| Gi,air,inp | concentration of gas phase state variable i in the air input [%V V−1] |
| Gi,atm | concentration of gas phase state variable i in the atmosphere [%V V−1] |
| Gi,percent | concentration of gas phase state variable i in off-gas, percentage [%V V−1] |
| hdiff | diffuser submergence [m] |
| hdiff,floor | diffuser height from floor [m] |
| Henryi,dt | temperature dependency factor for Henry coefficient of gas i [K] |
| Henryi,SATP | Henry coefficient of gas i, standard (SATP) temperature (25 °C) [mol m−3 Pa−1] |
| hr | reactor depth [m] |
| hsat,eff | effective saturation depth [m] |
| hsea | elevation above sea level [m] |
| iC,i | equivalent mass of soluble organic state variable i per unit mass of carbon [g gC−1] |
| kL,i,bub,st,cw | liquid-side mass transfer coefficient for gas bubbles, standard conditions [m d−1] |
| kL,i,sur,st,cw | liquid-side mass transfer coefficient for liquid surface, standard conditions [m d−1] |
| kLai,bub | volumetric mass transfer coefficient for gas bubbles, field conditions [d−1] |
| kLai,bub,st,cw | volumetric mass transfer coefficient for gas bubbles, standard conditions [d−1] |
| kLai,sur | volumetric mass transfer coefficient for liquid surface, field conditions [d−1] |
| kLai,sur,st,cw | volumetric mass transfer coefficient for liquid surface, standard conditions [d−1] |
| Lair | temperature lapse rate for air pressure calculation [K m−1] |
| Li | concentration of liquid phase state variable i [g m−3] |
| Li,ad | adsorbed soluble organic state variable i mass per bed volume [g m−3] |
| Mac,cycle | mass of activated carbon filled per cycle [g] |
| magnmid,asymm | magnitude of the breakthrough curve midpoint asymmetry correction term |
| MMair | molar mass of air [g mol−1] |
| MMEQ,i | equivalent molar mass of gas phase state variable i [g mol−1] |
| ndiff | number of diffusers |
| ngas,bub | molar quantity of gas bubbles per unit liquid volume [mol m−3] |
| Nrepl | activated carbon bed replacement cycle frequency [d−1] |
| pair | air pressure at field elevation [Pa] |
| pgas | gas phase pressure [Pa] |
| pNTP | pressure at standard (NTP) conditions (101,325 Pa) [Pa] |
| powd,diff | power value in a diffuser density correction term |
| powh,diff | power value in a diffuser submergence correction term |
| powmid,asymm | power of the breakthrough curve midpoint asymmetry correction term |
| ppartial,i,bub | partial pressure of gas state variable i in the gas phase [Pa] |
| ppartial,i,bub,st | partial pressure of gas state variable i in the gas phase, standard conditions [Pa] |
| ppartial,i,sur | partial pressure of gas state variable i in the atmosphere [Pa] |
| ppartial,i,sur,st | partial pressure of gas state variable i in the atmosphere, standard conditions [Pa] |
| pst,h,sat,eff | pressure at standard conditions and effective saturation depth [Pa] |
| pv,T | saturated vapor pressure of water at temperature T [Pa] |
| θ | Arrhenius temperature correction factor for the mass transfer coefficient |
| Q | volumetric flow of wastewater [m3 d−1] |
| Qair,NTP | air flow at standard (NTP) conditions [m3gas d−1] |
| Qair,NTP,sp | air flow per diffuser at standard (NTP) conditions [m3gas d−1] |
| Qgas,transfer,NTP | gas transfer flow at standard (NTP) conditions [m3gas d−1] |
| Qgas,outp,NTP | off-gas flow at standard (NTP) conditions [m3gas d−1] |
| ρac | apparent density of granular activated carbon [gAC m−3] |
| rateFi | mass rate of state variable i [g d−1] |
| ratei | reaction rate for the state variable [g m−3 d−1] |
| RemGAC,i | removal ratio of soluble organic state variable i by granular activated carbon |
| rj | process rate regarding process j (from Gujer matrix) [g m−3 d−1] |
| Si,bub,sat | saturation concentration at the gas bubble interface [g m−3] |
| Si,bub,sat,st,cw | saturation concentration at the gas bubble interface, standard conditions [g m−3] |
| Si,sur,sat | saturation concentration at the atmospheric interface [g m−3] |
| Si,sur,sat,st,cw | saturation concentration at the atmospheric interface, standard conditions [g m−3] |
| slbreak | slope of the breakthrough curve [m3 gC−1] |
| SO2 | dissolved oxygen concentration [gO2 m−3] |
| SOTRbub | standard oxygen transfer rate from bubbles [g d−1] |
| SSOTE | specific standard oxygen transfer efficiency [% m−1] |
| SSOTE0 | intercept in SSOTE correlation [% m−1] |
| SSOTEasym | asymptote in SSOTE correlation [% m−1] |
| T | liquid temperature [°C] |
| Tair,K | field air temperature [K] |
| TK | liquid temperature in an SI unit [K] |
| TNTP,K | temperature at standard (NTP) conditions (20 °C) [K] |
| trepl | duration of activated carbon bed replacement [d] |
| TSATP,K | temperature at standard (SATP) conditions (25 °C) [K] |
| Vac | activated carbon bed volume [m3] |
| Vgas | gas phase volume [m3gas] |
| Vgas,NTP | gas phase volume at standard (NTP) conditions [m3gas] |
| vj,i | stoichiometric coefficient of state variable i in process j |
| Vr | reactive volume [m3] |
Appendix A. Gujer Matrix Development
| Symbol | Process Name |
|---|---|
| 1 | OHO growth on VFAs, O2 |
| 2 | OHO growth on VFAs, NOx |
| 3 | OHO growth on benzene, O2 |
| 4 | OHO growth on benzene, NOx |
| 5 | OHO growth on toluene, O2 |
| 6 | OHO growth on toluene, NOx |
| 7 | OHO growth on ethylbenzene, O2 |
| 8 | OHO growth on ethylbenzene, NOx |
| 9 | OHO growth on xylene, O2 |
| 10 | OHO growth on xylene, NOx |
| 11 | OHO growth on SB, O2 |
| 12 | OHO growth on SB, NOx |
| 13 | SB fermentation with high VFA (OHO growth, anaerobic) |
| 14 | SB fermentation with low VFA (OHO growth, anaerobic) |
| 15 | Benzene fermentation with low VFA (OHO growth, anaerobic) |
| 16 | Toluene fermentation with low VFA (OHO growth, anaerobic) |
| 17 | Ethylbenzene fermentation with low VFA (OHO growth, anaerobic) |
| 18 | Xylene fermentation with low VFA (OHO growth, anaerobic) |
| 19 | OHO decay |
| 20 | NITO growth |
| 21 | NITO decay |
| 22 | AMETO growth |
| 23 | AMETO decay |
| 24 | HMETO growth |
| 25 | HMETO decay |
| 26 | XB hydrolysis |
| 27 | XB anaerobic hydrolysis (fermentation) |
| 28 | SN,B ammonification |
| 29 | NOx assimilative reduction |
| 30 | FeP precipitation |
| 31 | FeP redissolution |
| 32 | AlP precipitation |
| 33 | AlP redissolution |
| 34 | Elimination of surfactants |
| 35 | Methane gas transfer—bubbles |
| 36 | Hydrogen gas transfer—bubbles |
| 37 | Oxygen gas transfer—bubbles |
| 38 | Nitrogen gas transfer—bubbles |
| 39 | Benzene gas transfer—bubbles |
| 40 | Toluene gas transfer—bubbles |
| 41 | Ethylbenzene gas transfer—bubbles |
| 42 | Xylene gas transfer—bubbles |
| 43 | Methane gas transfer—surface |
| 44 | Hydrogen gas transfer—surface |
| 45 | Oxygen gas transfer—surface |
| 46 | Nitrogen gas transfer—surface |
| 47 | Benzene gas transfer—surface |
| 48 | Toluene gas transfer—surface |
| 49 | Ethylbenzene gas transfer—surface |
| 50 | Xylene gas transfer—surface |
| SBENE | STENE | SEBENE | SXENE | SB | XB | SU | XU | XE | XOHO | |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 1 | |||||||||
| 2 | 1 | |||||||||
| 3 | −1/YOHO,BTEX,ox | 1 | ||||||||
| 4 | −1/YOHO,BTEX,anox | 1 | ||||||||
| 5 | −1/YOHO,BTEX,ox | 1 | ||||||||
| 6 | −1/YOHO,BTEX,anox | 1 | ||||||||
| 7 | −1/YOHO,BTEX,ox | 1 | ||||||||
| 8 | −1/YOHO,BTEX,anox | 1 | ||||||||
| 9 | −1/YOHO,BTEX,ox | 1 | ||||||||
| 10 | −1/YOHO,BTEX,anox | 1 | ||||||||
| 11 | −1/YOHO,SB,ox | 1 | ||||||||
| 12 | −1/YOHO,SB,anox | 1 | ||||||||
| 13 | −1/YOHO,SB,ana | 1 | ||||||||
| 14 | −1/YOHO,SB,ana | 1 | ||||||||
| 15 | −1/YOHO,BTEX,ana | 1 | ||||||||
| 16 | −1/YOHO,BTEX,ana | 1 | ||||||||
| 17 | −1/YOHO,BTEX,ana | 1 | ||||||||
| 18 | −1/YOHO,BTEX,ana | 1 | ||||||||
| 19 | 1 − fE | fE | −1 | |||||||
| 21 | 1 − fE | fE | ||||||||
| 23 | 1 − fE | fE | ||||||||
| 25 | 1 − fE | fE | ||||||||
| 26 | 1 | −1 | ||||||||
| 27 | 1 − fH2 | −1 | ||||||||
| 29 | −EEQNO3 × XOHO/XBIO,kin | |||||||||
| 39 | 1 | |||||||||
| 40 | 1 | |||||||||
| 41 | 1 | |||||||||
| 42 | 1 | |||||||||
| 47 | 1 | |||||||||
| 48 | 1 | |||||||||
| 49 | 1 | |||||||||
| 50 | 1 |
| SVFA | |
|---|---|
| 1 | −1/YOHO,VFA,ox |
| 2 | −1/YOHO,VFA,anox |
| 13 | (1 − YOHO,SB,ana − YOHO,H2,ana,high)/YOHO,SB,ana |
| 14 | (1 − YOHO,SB,ana − YOHO,H2,ana,low)/YOHO,SB,ana |
| 15 | (1 − YOHO,SB,ana − YOHO,H2,ana,low)/YOHO,SB,ana |
| 16 | (1 − YOHO,SB,ana − YOHO,H2,ana,low)/YOHO,SB,ana |
| 17 | (1 − YOHO,SB,ana − YOHO,H2,ana,low)/YOHO,SB,ana |
| 18 | (1 − YOHO,SB,ana − YOHO,H2,ana,low)/YOHO,SB,ana |
| 22 | −1/YAMETO |
| XNITO | XAMETO | XHMETO | |
|---|---|---|---|
| 20 | 1 | ||
| 21 | −1 | ||
| 22 | 1 | ||
| 23 | −1 | ||
| 24 | 1 | ||
| 25 | −1 | ||
| 29 | −EEQNO3 × XNITO/XBIO,kin | −EEQNO3 × XAMETO/XBIO,kin | −EEQNO3 × XHMETO/XBIO,kin |
| SNHx | SNOx | SN2 | |
|---|---|---|---|
| 1 | −iN,BIO | ||
| 2 | −iN,BIO | −(1 − YOHO,VFA,anox)/(EEQN2,NO3 × YOHO,VFA,anox) | (1 − YOHO,VFA,anox)/(EEQN2,NO3 × YOHO,VFA,anox) |
| 3 | −iN,BIO | ||
| 4 | −iN,BIO | −(1 − YOHO,BTEX,anox)/(EEQN2,NO3 × YOHO,BTEX,anox) | (1 − YOHO,BTEX,anox)/(EEQN2,NO3 × YOHO,BTEX,anox) |
| 5 | −iN,BIO | ||
| 6 | −iN,BIO | −(1 − YOHO,BTEX,anox)/(EEQN2,NO3 × YOHO,BTEX,anox) | (1 − YOHO,BTEX,anox)/(EEQN2,NO3 × YOHO,BTEX,anox) |
| 7 | −iN,BIO | ||
| 8 | −iN,BIO | −(1 − YOHO,BTEX,anox)/(EEQN2,NO3 × YOHO,BTEX,anox) | (1 − YOHO,BTEX,anox)/(EEQN2,NO3 × YOHO,BTEX,anox) |
| 9 | −iN,BIO | ||
| 10 | −iN,BIO | −(1 − YOHO,BTEX,anox)/(EEQN2,NO3 × YOHO,BTEX,anox) | (1 − YOHO,BTEX,anox)/(EEQN2,NO3 × YOHO,BTEX,anox) |
| 11 | −iN,BIO | ||
| 12 | −iN,BIO | −(1 − YOHO,SB,anox)/(EEQN2,NO3 × YOHO,SB,anox) | (1 − YOHO,SB,anox)/(EEQN2,NO3 × YOHO,SB,anox) |
| 13 | −iN,BIO | ||
| 14 | −iN,BIO | ||
| 15 | −iN,BIO | ||
| 16 | −iN,BIO | ||
| 17 | −iN,BIO | ||
| 18 | −iN,BIO | ||
| 19 | −fE × (iN,XE − iN,BIO) | ||
| 20 | −1/YNITO − iN,BIO | 1/YNITO | |
| 21 | −fE × (iN,XE − iN,BIO) | ||
| 22 | −iN,BIO | ||
| 23 | −fE × (iN,XE − iN,BIO) | ||
| 24 | −iN,BIO | ||
| 25 | −fE × (iN,XE − iN,BIO) | ||
| 28 | 1 | ||
| 29 | 1 + EEQNO3 × iN,BIO | −1 | |
| 38 | 1 | ||
| 46 | 1 |
| SN,B | XN,B | SPO4 | XP,B | SO2 | SCH4 | SH2 | |
|---|---|---|---|---|---|---|---|
| 1 | −iP,BIO | −(1 − YOHO,VFA,ox)/YOHO,VFA,ox | |||||
| 2 | −iP,BIO | ||||||
| 3 | −iP,BIO | −(1 − YOHO,BTEX,ox)/YOHO,BTEX,ox | |||||
| 4 | −iP,BIO | ||||||
| 5 | −iP,BIO | −(1 − YOHO,BTEX,ox)/YOHO,BTEX,ox | |||||
| 6 | −iP,BIO | ||||||
| 7 | −iP,BIO | −(1 − YOHO,BTEX,ox)/YOHO,BTEX,ox | |||||
| 8 | −iP,BIO | ||||||
| 9 | −iP,BIO | −(1 − YOHO,BTEX,ox)/YOHO,BTEX,ox | |||||
| 10 | −iP,BIO | ||||||
| 11 | −iP,BIO | −(1 − YOHO,SB,ox)/YOHO,SB,ox | |||||
| 12 | −iP,BIO | ||||||
| 13 | −iP,BIO | YOHO,H2,ana,high/YOHO,SB,ana | |||||
| 14 | −iP,BIO | YOHO,H2,ana,low/YOHO,SB,ana | |||||
| 15 | −iP,BIO | YOHO,H2,ana,low/YOHO,BTEX,ana | |||||
| 16 | −iP,BIO | YOHO,H2,ana,low/YOHO,BTEX,ana | |||||
| 17 | −iP,BIO | YOHO,H2,ana,low/YOHO,BTEX,ana | |||||
| 18 | −iP,BIO | YOHO,H2,ana,low/YOHO,BTEX,ana | |||||
| 19 | (1 − fE) × iN,BIO | (1 − fE) × iP,BIO | |||||
| 20 | −iP,BIO | −(EEQNO3 − YNITO)/YNITO | |||||
| 21 | (1 − fE) × iN,BIO | (1 − fE) × iP,BIO | |||||
| 22 | −iP,BIO | (1 − YAMETO)/YAMETO | |||||
| 23 | (1 − fE) × iN,BIO | (1 − fE) × iP,BIO | |||||
| 24 | −iP,BIO | (1 − YHMETO)/YHMETO | −1/YHMETO | ||||
| 25 | (1 − fE) × iN,BIO | (1 − fE) × iP,BIO | |||||
| 26 | XN,B/XB | −XN,B/XB | XP,B/XB | −XP,B/XB | |||
| 27 | XN,B/XB | −XN,B/XB | XP,B/XB | −XP,B/XB | fH2 | ||
| 28 | −1 | ||||||
| 29 | EEQNO3 × iP,BIO | ||||||
| 30 | −fP,Fe | ||||||
| 31 | fP,Fe | ||||||
| 32 | −fP,Al | ||||||
| 33 | fP,Al | ||||||
| 35 | 1 | ||||||
| 36 | 1 | ||||||
| 37 | 1 | ||||||
| 43 | 1 | ||||||
| 44 | 1 | ||||||
| 45 | 1 |
| SALK | XFeOH | XFeP | XAlOH | XAlP | |
|---|---|---|---|---|---|
| 1 | (−iN,BIO × CHNHx − iP,BIO × CHPO4) | ||||
| 2 | (−(1 − YOHO,VFA,anox)/(EEQN2,NO3 × YOHO,VFA,anox) × CHNO3 − iN,BIO × CHNHx − iP,BIO × CHPO4) | ||||
| 3 | (−iN,BIO × CHNHx − iP,BIO × CHPO4) | ||||
| 4 | (−(1 − YOHO,BTEX,anox)/(EEQN2,NO3 × YOHO,BTEX,anox) × CHNO3 − iN,BIO × CHNHx − iP,BIO × CHPO4) | ||||
| 5 | (−iN,BIO × CHNHx − iP,BIO × CHPO4) | ||||
| 6 | (−(1 − YOHO,BTEX,anox)/(EEQN2,NO3 × YOHO,BTEX,anox) × CHNO3 − iN,BIO × CHNHx − iP,BIO × CHPO4) | ||||
| 7 | (−iN,BIO × CHNHx − iP,BIO × CHPO4) | ||||
| 8 | (−(1 − YOHO,BTEX,anox)/(EEQN2,NO3 × YOHO,BTEX,anox) × CHNO3 − iN,BIO × CHNHx − iP,BIO × CHPO4) | ||||
| 9 | (−iN,BIO × CHNHx − iP,BIO × CHPO4) | ||||
| 10 | (−(1 − YOHO,BTEX,anox)/(EEQN2,NO3 × YOHO,BTEX,anox) × CHNO3 − iN,BIO × CHNHx − iP,BIO × CHPO4) | ||||
| 11 | (−iN,BIO × CHNHx − iP,BIO × CHPO4) | ||||
| 12 | (−(1 − YOHO,SB,anox)/(EEQN2,NO3 × YOHO,SB,anox) × CHNO3 − iN,BIO × CHNHx − iP,BIO × CHPO4) | ||||
| 13 | (−iN,BIO × CHNHx − iP,BIO × CHPO4) | ||||
| 14 | (−iN,BIO × CHNHx − iP,BIO × CHPO4) | ||||
| 15 | (−iN,BIO × CHNHx − iP,BIO × CHPO4) | ||||
| 16 | (−iN,BIO × CHNHx − iP,BIO × CHPO4) | ||||
| 17 | (−iN,BIO × CHNHx − iP,BIO × CHPO4) | ||||
| 18 | (−iN,BIO × CHNHx − iP,BIO × CHPO4) | ||||
| 19 | −fE × (iN,XE − iN,BIO) × CHNHx | ||||
| 20 | ((−1/YNITO − iN,BIO) × CHNHx + 1/YNITO × CHNO3 − iP,BIO × CHPO4) | ||||
| 21 | −fE × (iN,XE − iN,BIO) × CHNHx | ||||
| 22 | (−iN,BIO × CHNHx − iP,BIO × CHPO4) | ||||
| 23 | −fE × (iN,XE − iN,BIO) × CHNHx | ||||
| 24 | (−iN,BIO × CHNHx − iP,BIO × CHPO4) | ||||
| 25 | −fE × (iN,XE − iN,BIO) × CHNHx | ||||
| 26 | XP,B/XB × CHPO4 | ||||
| 27 | XP,B/XB × CHPO4 | ||||
| 28 | CHNHx | ||||
| 29 | ((1 + EEQNO3 × iN,BIO) × CHNHx − CHNO3 + EEQNO3 × iP,BIO × CHPO4) | ||||
| 30 | −fP,Fe × CHPO4 | −1 | 1 | ||
| 31 | fP,Fe × CHPO4 | 1 | −1 | ||
| 32 | −fP,Al × CHPO4 | −1 | 1 | ||
| 33 | fP,Al × CHPO4 | 1 | −1 |
| SALPHA | GCH4 | GH2 | GO2 | GN2 | GBENE | GTENE | GEBENE | GXENE | |
|---|---|---|---|---|---|---|---|---|---|
| 34 | 1 | ||||||||
| 35 | −1 | ||||||||
| 36 | −1 | ||||||||
| 37 | −1 | ||||||||
| 38 | −1 | ||||||||
| 39 | −1 | ||||||||
| 40 | −1 | ||||||||
| 41 | −1 | ||||||||
| 42 | −1 |
| Rate | |
|---|---|
| 1 | µOHO,T × XOHO × MsatSVFA,KVFA × MsatSO2,KO2,OHO × MsatSNHx,KNHx,BIO × MsatSPO4,KPO4,BIO × MsatSALK,KALK |
| 2 | µOHO,T × XOHO × ηOHO,anox × MsatSVFA,KVFA × MsatSNOx,KNOx,OHO × MinhSO2,KO2,OHO × MsatSNHx,KNHx,BIO × MsatSPO4,KPO4,BIO × MsatSALK,KALK |
| 3 | µOHO,BENE,T × XOHO × MsatSBENE,KBENE × MsatSO2,KO2,OHO × MsatSNHx,KNHx,BIO × MsatSPO4,KPO4,BIO × MsatSALK,KALK |
| 4 | µOHO,BENE,T × XOHO × ηOHO,anox × MsatSBENE,KBENE × MsatSNOx,KNOx,OHO × MinhSO2,KO2,OHO × MsatSNHx,KNHx,BIO × MsatSPO4,KPO4,BIO × MsatSALK,KALK |
| 5 | µOHO,TENE,T × XOHO × MsatSTENE,KTENE × MsatSO2,KO2,OHO × MsatSNHx,KNHx,BIO × MsatSPO4,KPO4,BIO × MsatSALK,KALK |
| 6 | µOHO,TENE,T × XOHO × ηOHO,anox × MsatSTENE,KTENE × MsatSNOx,KNOx,OHO × MinhSO2,KO2,OHO × MsatSNHx,KNHx,BIO × MsatSPO4,KPO4,BIO × MsatSALK,KALK |
| 7 | µOHO,EBENE,T × XOHO × MsatSEBENE,KEBENE × MsatSO2,KO2,OHO × MsatSNHx,KNHx,BIO × MsatSPO4,KPO4,BIO × MsatSALK,KALK |
| 8 | µOHO,EBENE,T × XOHO × ηOHO,anox × MsatSEBENE,KEBENE × MsatSNOx,KNOx,OHO × MinhSO2,KO2,OHO × MsatSNHx,KNHx,BIO × MsatSPO4,KPO4,BIO × MsatSALK,KALK |
| 9 | µOHO,XENE,T × XOHO × MsatSXENE,KXENE × MsatSO2,KO2,OHO × MsatSNHx,KNHx,BIO × MsatSPO4,KPO4,BIO × MsatSALK,KALK |
| 10 | µOHO,XENE,T × XOHO × ηOHO,anox × MsatSXENE,KXENE × MsatSNOx,KNOx,OHO × MinhSO2,KO2,OHO × MsatSNHx,KNHx,BIO × MsatSPO4,KPO4,BIO × MsatSALK,KALK |
| 11 | µOHO,T × MsatSB,KSB × MinhSVFA,KVFA × XOHO × MsatSO2,KO2,OHO × MsatSNHx,KNHx,BIO × MsatSPO4,KPO4,BIO × MsatSALK,KALK |
| 12 | µOHO,T × ηOHO,anox × MsatSB,KSB × MinhSVFA,KVFA × XOHO × MsatSNOx,KNOx,OHO × MinhSO2,KO2,OHO × MsatSNHx,KNHx,BIO × MsatSPO4,KPO4,BIO |
| 13 | µFERM,OHO,T × XOHO × LogsatSVFA,KVFA,FERM × MsatSB,KSB,ana × MinhSO2,KO2,OHO × MinhSNOx,KNOx,OHO × MsatSNHx,KNHx,BIO × MsatSPO4,KPO4,BIO × MsatSALK,KALK |
| 14 | µFERM,OHO,T × XOHO × LoginhSVFA,KVFA,FERM × MsatSB,KSB,ana × MinhSO2,KO2,OHO × MinhSNOx,KNOx,OHO × MsatSNHx,KNHx,BIO × MsatSPO4,KPO4,BIO × MsatSALK,KALK |
| 15 | µFERM,OHO,BENE,T × XOHO × LoginhSVFA,KVFA,FERM × MsatSBENE,KBENE,ana × MinhSO2,KO2,OHO × MinhSNOx,KNOx,OHO × MsatSNHx,KNHx,BIO × MsatSPO4,KPO4,BIO × MsatSALK,KALK |
| 16 | µFERM,OHO,TENE,T × XOHO × LoginhSVFA,KVFA,FERM × MsatSTENE,KTENE,ana × MinhSO2,KO2,OHO × MinhSNOx,KNOx,OHO × MsatSNHx,KNHx,BIO × MsatSPO4,KPO4,BIO × MsatSALK,KALK |
| 17 | µFERM,OHO,EBENE,T × XOHO × LoginhSVFA,KVFA,FERM × MsatSEBENE,KEBENE,ana × MinhSO2,KO2,OHO × MinhSNOx,KNOx,OHO × MsatSNHx,KNHx,BIO × MsatSPO4,KPO4,BIO × MsatSALK,KALK |
| 18 | µFERM,OHO,XENE,T × XOHO × LoginhSVFA,KVFA,FERM × MsatSXENE,KXENE,ana × MinhSO2,KO2,OHO × MinhSNOx,KNOx,OHO × MsatSNHx,KNHx,BIO × MsatSPO4,KPO4,BIO × MsatSALK,KALK |
| 19 | bOHO,T × XOHO × (MsatSO2,KO2,OHO + ηb,anox × MsatSNOx,KNOx,OHO × MinhSO2,KO2,OHO + ηb,ana × MinhSNOx,KNOx,OHO × MinhSO2,KO2,OHO) |
| 20 | µNITO,T × MsatSNHx,KNHx,NITO × XNITO × MsatSO2,KO2,NITO × MsatSPO4,KPO4,BIO × MsatSALK,KALK |
| 21 | bNITO,T × XNITO × (MsatSO2,KO2,NITO + ηb,anox × MsatSNOx,KNOx,NITO × MinhSO2,KO2,NITO + ηb,ana × MinhSNOx,KNOx,NITO × MinhSO2,KO2,NITO + mtox,ana) |
| 22 | µAMETO,T × HsatSVFA,AMETO × XAMETO × MinhSO2,KiO2,AMETO × MinhSNOx,KNOx,AMETO × MsatSNHx,KNHx,BIO × MsatSPO4,KPO4,BIO × MsatSALK,KALK |
| 23 | bAMETO,T × XAMETO × (MsatSO2,KiO2,AMETO + ηb,anox × MsatSNOx,KNOx,AMETO × MinhSO2,KiO2,AMETO + ηb,ana × MinhSNOx,KNOx,AMETO × MinhSO2,KiO2,AMETO) |
| 24 | µHMETO,T × MsatSH2,KH2,HMETO × XHMETO × MinhSO2,KiO2,HMETO × MinhSNOx,KNOx,HMETO × MsatSNHx,KNHx,BIO × MsatSPO4,KPO4,BIO × MsatSALK,KALK |
| 25 | bHMETO,T × XHMETO × (MsatSO2,KiO2,HMETO + ηb,anox × MsatSNOx,KNOx,HMETO × MinhSO2,KiO2,HMETO + ηb,ana × MinhSNOx,KNOx,HMETO × MinhSO2,KiO2,HMETO) |
| 26 | qHYD,T × XBIO,kin × MRsatXB,XBIO,kin,KHYD × (MsatSO2,KO2,OHO + ηb,anox × MsatSNOx,KNOx,OHO × MinhSO2,KO2,OHO) × MsatSALK,KALK |
| 27 | qHYD,T × XBIO,kin × MRsatXB,XBIO,kin,KHYD × ηb,ana × MinhSNOx,KNOx,OHO × MinhSO2,KO2,OHO × MsatSALK,KALK |
| 28 | qAMMON,T × SN,B × XBIO,kin |
| 29 | qASSIM,T × MsatSNOx,KNOx,ASSIM × MinhSNHx,KiNHx,ASSIM × XBIO,kin |
| 30 | qFeOH,PREC,Me × SPO4 × XFeOH |
| 31 | qFeOH,DISSOL,Me × XFeP × MsatSALK,KALK |
| 32 | qAlOH,PREC,Me × SPO4 × XAlOH |
| 33 | qAlOH,DISSOL,Me × XAlP × MsatSALK,KALK |
| 34 | qALPHA,O2 × XVSS × dampALPHA × (SALPHA,sat — SALPHA) |
| 35 | kLaGCH4,bub × (SGCH4,bub,sat — SCH4) |
| 36 | kLaGH2,bub × (SGH2,bub,sat — SH2) |
| 37 | kLaGO2,bub × (SGO2,bub,sat — SO2) |
| 38 | kLaGN2,bub × (SGN2,bub,sat — SN2) |
| 39 | kLaGBENE,bub × (SGBENE,bub,sat — SBENE) |
| 40 | kLaGTENE,bub × (SGTENE,bub,sat — STENE) |
| 41 | kLaGEBENE,bub × (SGEBENE,bub,sat — SEBENE) |
| 42 | kLaGXENE,bub × (SGXENE,bub,sat — SXENE) |
| 43 | kLaGCH4,sur × (SGCH4,sur,sat — SCH4) |
| 44 | kLaGH2,sur × (SGH2,sur,sat — SH2) |
| 45 | kLaGO2,sur × (SGO2,sur,sat — SO2) |
| 46 | kLaGN2,sur × (SGN2,sur,sat — SN2) |
| 47 | kLaGBENE,sur × (SGBENE,sur,sat — SBENE) |
| 48 | kLaGTENE,sur × (SGTENE,sur,sat — STENE) |
| 49 | kLaGEBENE,sur × (SGEBENE,sur,sat — SEBENE) |
| 50 | kLaGXENE,sur × (SGXENE,sur,sat — SXENE) |
| Symbol | Name | Expression |
|---|---|---|
| Msat(var; k) | Monod saturation | var/(k + var) |
| Minh(var; k) | Monod inhibition | k/(k + var) |
| MRsat(s;x;k) | Monod ratio saturation | (s/x)/(s/x + k) |
| Logsat(var; halfval; slope) | Logistic saturation | 1/(1 + Exp((halfval − var) × slope)) |
| Loginh(var; halfval; slope) | Logistic inhibition | 1/(1 + Exp((var − halfval) × slope)) |
| Hsat(var; halfval; halfinh) | Haldane equation | var/(halfval + var + (var2/halfinh)) |
Appendix B. BTEX Kinetic and Stoichiometric Model Parameters
| Ordinary Heterotrophic Organism Kinetics (OHO) | |||
|---|---|---|---|
| Symbol | Name | Value | Unit |
| µOHO,BENE | Maximum specific growth rate of OHOs on benzene | 0.006 | d−1 |
| µOHO,TENE | Maximum specific growth rate of OHOs on toluene | 0.014 | d−1 |
| µOHO,EBENE | Maximum specific growth rate of OHOs on ethylbenzene | 0.014 | d−1 |
| µOHO,XENE | Maximum specific growth rate of OHOs on xylene | 0.010 | d−1 |
| µFERM,OHO,BENE | Fermentation growth rate of OHOs on benzene | 0.0030 | d−1 |
| µFERM,OHO,TENE | Fermentation growth rate of OHOs on toluene | 0.0042 | d−1 |
| µFERM,OHO,EBENE | Fermentation growth rate of OHOs on ethylbenzene | 0.0035 | d−1 |
| µFERM,OHO,XENE | Fermentation growth rate of OHOs on xylene | 0.0050 | d−1 |
| KBENE | Half-saturation of benzene for OHOs | 6.8 | gCOD m−3 |
| KTENE | Half-saturation of toluene for OHOs | 14.8 | gCOD m−3 |
| KEBENE | Half-saturation of ethylbenzene for OHOs | 3.8 | gCOD m−3 |
| KXENE | Half-saturation of xylene for OHOs | 17.6 | gCOD m−3 |
| KBENE,ana | Half-saturation of benzene in fermentation by OHOs | 238 | gCOD m−3 |
| KTENE,ana | Half-saturation of toluene in fermentation by OHOs | 310 | gCOD m−3 |
| KEBENE,ana | Half-saturation of ethylbenzene in fermentation by OHOs | 67 | gCOD m−3 |
| KXENE,ana | Half-saturation of xylene in fermentation by OHOs | 615 | gCOD m−3 |
| Stoichiometric yields | |||
| Symbol | Name | Value | Unit |
| YOHO,BTEX,ox | Yield of OHOs on BTEX under aerobic conditions | 0.55 | g XOHO g SBTEX−1 |
| YOHO,BTEX,anox | Yield of OHOs on BTEX under anoxic conditions | 0.35 | g XOHO g SBTEX−1 |
| YOHO,BTEX,ana | Yield of OHOs on BTEX under anaerobic conditions | 0.10 | g XOHO g SBTEX−1 |
Appendix C. Gas Transfer, Aeration and BTEX Model Parameters
| Henry Coefficients | |||
|---|---|---|---|
| Symbol | Name | Value | Unit |
| HenryBENE,25 | Henry coefficient for benzene at 25 °C | 1.70 × 10−3 | mol m−3 Pa−1 |
| HenryBENE,dt | Henry’s law temperature dependency factor of benzene | 4150 | K |
| HenryTENE,25 | Henry coefficient for toluene at 25 °C | 1.50 × 10−3 | mol m−3 Pa−1 |
| HenryTENE,dt | Henry’s law temperature dependency factor of toluene | 4150 | K |
| HenryEBENE,25 | Henry coefficient for ethylbenzene at 25 °C | 1.27 × 10−3 | mol m−3 Pa−1 |
| HenryEBENE,dt | Henry’s law temperature dependency factor of ethylbenzene | 5100 | K |
| HenryXENE,25 | Henry coefficient for xylene at 25 °C | 1.56 × 10−3 | mol m−3 Pa−1 |
| HenryXENE,dt | Henry’s law temperature dependency factor of xylene | 4083 | K |
| Diffusion coefficients | |||
| Symbol | Name | Value | Unit |
| DBENE,25 | Diffusion coefficient of benzene in water at 25 °C | 9.13 × 10−5 | m2 d−1 |
| DTENE,25 | Diffusion coefficient of toluene in water at 25 °C | 7.89 × 10−5 | m2 d−1 |
| DEBENE,25 | Diffusion coefficient of ethylbenzene in water at 25 °C | 7.27 × 10−5 | m2 d−1 |
| DXENE,25 | Diffusion coefficient of xylene in water at 25 °C | 7.08 × 10−5 | m2 d−1 |
| Oxygen transfer efficiency correlation parameters | |||
| Symbol | Name | Value | Unit |
| SSOTE0 | Intercept in SSOTE correlation | 7.77 | % m−1 |
| expSSOTE | Exponent (absolute value) in SSOTE correlation | 0.01041 | d m−3gas |
| SSOTEasym | Asymptote in SSOTE correlation | 5.75 | % m−1 |
| divd,diff | Divisor value in a diffuser density correction term | 0.1173 | m2 m−2 |
| powd,diff | Power value in a diffuser density correction term | 0.1329 | |
| coefflead,h,diff | Leading coefficient in a diffuser submergence correction term | 0.011 | m−1 |
| powh,diff | Power value in a diffuser submergence correction term | 1.6031 | |
| coefflin,h,diff | Linear coefficient in a diffuser submergence correction term | −0.0229 | m−1 |
| Specific molecular masses | |||
| Symbol | Name | Value | Unit |
| MMEQ,GBENE | Equivalent molar mass of benzene | 239.97 | gCOD mol−1 |
| MMEQ,GTENE | Equivalent molar mass of toluene | 287.96 | gCOD mol−1 |
| MMEQ,GEBENE | Equivalent molar mass of ethylbenzene | 335.95 | gCOD mol−1 |
| MMEQ,GXENE | Equivalent molar mass of xylene | 335.95 | gCOD mol−1 |
Appendix D. GAC Model Parameters
| State Variable Equivalent Mass Ratios to Carbon | |||
|---|---|---|---|
| Symbol | Name | Value | Unit |
| iC,VFA | COD-to-carbon-mass ratio of VFA | 5.33 | gCOD gC−1 |
| iC,BENE | COD-to-carbon-mass ratio of benzene | 19.98 | gCOD gC−1 |
| iC,TENE | COD-to-carbon-mass ratio of toluene | 23.98 | gCOD gC−1 |
| iC,EBENE | COD-to-carbon-mass ratio of ethylbenzene | 27.97 | gCOD gC−1 |
| iC,XENE | COD-to-carbon-mass ratio of xylene | 27.97 | gCOD gC−1 |
| iC,SB | COD-to-carbon-mass ratio of readily biodegradable substrate | 3.20 | gCOD gC−1 |
| iC,SU | COD-to-carbon-mass ratio of soluble unbiodegradable organics | 2.80 | gCOD gC−1 |
| iC,SN,B | Nitrogen-to-carbon-mass ratio of soluble biodegradable organic N | 1.17 | gN gC−1 |
| Breakthrough curve parameters | |||
| Symbol | Name | Value | Unit |
| fbreak | Breakpoint fraction | 0.05 | |
| slbreak | Breakthrough curve slope | 0.00015 | m3 g−1 |
| powmid,asymm | Power of the midpoint asymmetry correction term | 20.00 | |
| magnmid,asymm | Magnitude of the midpoint asymmetry correction term | 0.50 | |
References
- Scheierling, S.M.; Bartone, C.; Mara, D.D.; Drechsel, P. Improving Wastewater Use in Agriculture: An Emerging Priority; World Bank Policy Research Working Paper Series 5412; The World Bank: Washington, DC, USA, 2010. [Google Scholar] [CrossRef]
- Scheierling, S.M.; Bartone, C.R.; Mara, D.D.; Drechsel, P. Towards an agenda for improving wastewater use in agriculture. Water Int. 2011, 36, 420–440. [Google Scholar] [CrossRef]
- Ofori, S.; Puškáčová, A.; Růžičková, I.; Wanner, J. Treated wastewater reuse for irrigation: Pros and cons. Sci. Total Environ. 2021, 760, 144026. [Google Scholar] [CrossRef] [PubMed]
- Pedrero, F.; Kalavrouziotis, I.; Alarcón, J.J.; Koukoulakis, P.; Asano, T. Use of treated municipal wastewater in irrigated agriculture—Review of some practices in Spain and Greece. Agric. Water Manag. 2010, 97, 1233–1241. [Google Scholar] [CrossRef]
- Jaramillo, M.F.; Restrepo, I. Wastewater Reuse in Agriculture: A Review about Its Limitations and Benefits. Sustainability 2017, 9, 1734. [Google Scholar] [CrossRef]
- Khalil, S.; Kakar, M.K. Agricultural use of untreated urban wastewater in Pakistan. Asian J. Agric. Rural Dev. 2011, 1, 21–26. [Google Scholar]
- Sheikholeslami, Z.; Kebria, D.Y.; Qaderi, F. Nanoparticle for degradation of BTEX in produced water; an experimental procedure. J. Mol. Liq. 2018, 264, 476–482. [Google Scholar] [CrossRef]
- Dehghani, M.; Abbasi, A.; Taherzadeh, Z.; Dehghani, S. Exposure assessment of wastewater treatment plant employees to BTEX: A biological monitoring approach. Sci. Rep. 2022, 12, 21433. [Google Scholar] [CrossRef]
- Davidson, C.J.; Hannigan, J.H.; Bowen, S.E. Effects of inhaled combined Benzene, Toluene, Ethylbenzene, and Xylenes (BTEX): Toward an environmental exposure model. Environ. Toxicol. Pharmacol. 2021, 81, 103518. [Google Scholar] [CrossRef]
- Dhivakar, V.; Rajan, T. BTEX compounds removal from wastewater by using UV&UV/H2O2 process. Int. J. Recent Eng. Sci. 2018, 5, 22–25. [Google Scholar] [CrossRef]
- Elmobarak, W.F.; Hameed, B.H.; Almomani, F.; Abdullah, A.Z. A Review on the Treatment of Petroleum Refinery Wastewater Using Advanced Oxidation Processes. Catalysts 2021, 11, 782. [Google Scholar] [CrossRef]
- Mello, J.M.M.; Brandao, H.L.; Valerio, A.; de Souza, A.A.U.; de Oliveira, D.; da Silva, A. Biodegradation of BTEX compounds from petrochemical wastewater: Kinetic and toxicity. J. Water Process Eng. 2019, 32, 100914. [Google Scholar] [CrossRef]
- Gusmão, V.R.; Martins, T.H.; Chinalia, F.A.; Sakamoto, I.K.; HenriqueThiemann, O.; Varesche, M.B.A. BTEX and ethanol removal in horizontal-flow anaerobic immobilized biomass reactor, under denitrifying condition. Process Biochem. 2006, 41, 1391–1400. [Google Scholar] [CrossRef]
- Kuyukina, M.S.; Krivoruchko, A.V.; Ivshina, I.B. Advanced Bioreactor Treatments of Hydrocarbon-Containing Wastewater. Appl. Sci. 2020, 10, 831. [Google Scholar] [CrossRef]
- Trusek-Holownia, A.; Noworyta, A. Advanced treatment of wastewater with BTEX. Desalination Water Treat. 2012, 50, 440–445. [Google Scholar] [CrossRef]
- Takáčová, A.; Smolinská, M.; Semerád, M.; Matúš, P. Degradation of BTEX by microalgae Parachlorella kessleri. Pet. Coal 2015, 57, 2. [Google Scholar]
- Anjum, H.; Johari, K.; Gnanasundaram, N.; Ganesapillai, M.; Arunagiri, A.; Regupathi, I.; Thanabalan, M. A review on adsorptive removal of oil pollutants (BTEX) from wastewater using carbon nanotubes. J. Mol. Liq. 2019, 277, 1005–1025. [Google Scholar] [CrossRef]
- El-Naas, M.H.; Acio, J.A.; El Telib, A.E. Aerobic biodegradation of BTEX: Progresses and prospects. J. Environ. Chem. Eng. 2014, 2, 1104–1122. [Google Scholar] [CrossRef]
- Carvajal, A.; Akmirza, I.; Navia, D.; Pérez, R.; Muñoz, R.; Lebrero, R. Anoxic denitrification of BTEX: Biodegradation kinetics and pollutant interactions. J. Environ. Manag. 2018, 214, 125–136. [Google Scholar] [CrossRef] [PubMed]
- Kasi, M.; Wadhawan, T.; McEvoy, J.; Padmanabhan, G.; Khan, E. Effect of Carbon Source during Enrichment on BTEX Degradation by Anaerobic Mixed Bacterial Cultures. Biodegradation 2013, 24, 279–293. [Google Scholar] [CrossRef] [PubMed]
- de Nardi, I.R.; Zaiat, M.; Foresti, E. Kinetics of BTEX degradation in a packed-bed anaerobic reactor. Biodegradation 2007, 18, 83. [Google Scholar] [CrossRef]
- Lee, K.C.; Rittmann, B.E.; Shi, J.; McAvoy, D. Advanced steady-state model for the fate of hydrophobic and volatile compounds in activated sludge. Water Environ. Res. 1998, 70, 1118–1131. [Google Scholar] [CrossRef]
- Pomiès, M.; Wisniewski, C.; Choubert, J.M.; Coquery, M. Modelling of micropollutant removal in biological wastewater treatments: A review. Sci. Total Environ. 2012, 443, 733–748. [Google Scholar] [CrossRef]
- Orhon, D.; Çokgör, E.U. COD fractionation in wastewater characterization—The state of the art. J. Chem. Technol. Biotechnol. 1997, 68, 283–293. [Google Scholar] [CrossRef]
- Patry, G.G.; Takács, I. Settling of flocculent suspensions in secondary clarifiers. Water Res. 1992, 26, 473–479. [Google Scholar] [CrossRef]
- Henze, M.; Gujer, W.; Mino, T.; Matsuo, T.; Wentzel, M.C.; Marais, G.V.R.; Van Loosdrecht, M.C. Activated sludge model no. 2d, ASM2d. Water Sci. Technol. 1999, 39, 165–182. [Google Scholar] [CrossRef]
- Arnell, M.; Ahlström, M.; Wärff, C.; Saagi, R.; Jeppsson, U. Plant-Wide Modelling and Analysis of WWTP Temperature Dynamics for Sustainable Heat Recovery from Wastewater. Water Sci. Technol. 2021, 84, 1023–1036. [Google Scholar] [CrossRef]
- Rieger, L.; Gillot, S.; Langergraber, G.; Ohtsuki, T.; Shaw, A.; Takács, I. Guidelines for Using Activated Sludge Models; IWA Publishing: London, UK, 2012; ISBN 978-1-84339-174-6. [Google Scholar]
- Henze, M.; Grady, L., Jr.; Gujer, W.; Marais, G.; Matsuo, T. Activated Sludge Model No 1; IAWPRC Publishing: London, UK, 1987. [Google Scholar]
- Dynamita. Sumo22 User Manual; Dynamita SARL: Sigale, France, 2022. [Google Scholar]
- Gazsó, Z.; Házi, F.; Kenyeres, I.; Váci, L. Full-Scale Wastewater Treatment Plant Simulation for Real-Time Optimization. Water Pract. Technol. 2017, 12, 848–856. [Google Scholar] [CrossRef]
- Herrmann-Heber, R.; Reinecke, S.F.; Hampel, U. Dynamic Aeration for Improved Oxygen Mass Transfer in the Wastewater Treatment Process. Chem. Eng. J. 2019, 386, 122068. [Google Scholar] [CrossRef]
- U.S. Environmental Protection Agency. Design Manual: Fine Pore Aeration Systems; EPA/625/1-89/023; U.S. Environmental Protection Agency: Cincinnati, OH, USA, 1989. [Google Scholar]
- Sincero, A.P.; Sincero, G.A. Physical-Chemical Treatment of Water and Wastewater, 1st ed.; CRC Press: Boca Raton, FL, USA, 2002; ISBN 978-1-58716-124-7. [Google Scholar]
- Stenstrom, M.K.; Leu, S.-Y.; Jiang, P. Theory to Practice: Oxygen Transfer and the New ASCE Standard. Proc. Water Environ. Fed. 2006, 7, 4838–4852. [Google Scholar] [CrossRef]
- DelSontro, T.; McGinnis, D.; Wehrli, B.; Ostrovsky, I. Size Does Matter: Importance of Large Bubbles and Small-Scale Hot Spots for Methane Transport. Environ. Sci. Technol. 2015, 49, 1268–1276. [Google Scholar] [CrossRef]
- Jensen, M.B.; Kofoed, M.V.W.; Fischer, K.; Voigt, N.V.; Agneessens, L.M.; Batstone, D.J.; Ottosen, L.D.M. Venturi-Type Injection System as a Potential H2 Mass Transfer Technology for Full-Scale in Situ Biomethanation. Appl. Energy 2018, 222, 840–846. [Google Scholar] [CrossRef]
- Roberts, P.V.; Munz, C.; Dändliker, P. Modeling Volatile Organic Solute Removal by Surface and Bubble Aeration. J. Water Pollut. Control Fed. 1984, 56, 157–163. [Google Scholar]
- Khalil, A.; Rosso, D.; DeGroot, C.T. Effects of Flow Velocity and Bubble Size Distribution on Oxygen Mass Transfer in Bubble Column Reactors—A Critical Evaluation of the Computational Fluid Dynamics-Population Balance Model. Water Environ. Res. 2021, 93, 2274–2297. [Google Scholar] [CrossRef] [PubMed]
- Hayduk, W.; Laudie, H. Prediction of Diffusion Coefficients for Nonelectrolytes in Dilute Aqueous Solutions. AIChE J. 1974, 20, 611–615. [Google Scholar] [CrossRef]
- Yaws, C.L. Handbook of Transport Property Data: Viscosity, Thermal Conductivity, and Diffusion Coefficients of Liquids and Gases; Library of Physico-Chemical Property Data; Gulf Publishing Company: Houston, TX, USA, 1995; ISBN 978-0-88415-392-4. [Google Scholar]
- New Jersey Department of Environmental Protection. Chemical Properties for Calculation of Impact to Ground Water Soil Remediation Standards; New Jersey Department of Environmental Protection: Trenton, NJ, USA, 2008. [Google Scholar] [CrossRef]
- Batstone, D.; Flores-Alsina, X. Generalised Physicochemical Model (PCM) for Wastewater Processes; IWA Publishing: London, UK, 2022; ISBN 978-1-78040-982-5. [Google Scholar]
- Bencsik, D.; Takács, I.; Rosso, D. Dynamic Alpha Factors: Prediction in Time and Evolution along Reactors. Water Res. 2022, 216, 118339. [Google Scholar] [CrossRef]
- U.S. Environmental Protection Agency. Summary Report: Fine Pore (Fine Bubble) Aeration Systems; EPA/625/8-85/010; U.S. Environmental Protection Agency: Cincinnati, OH, USA, 1989. [Google Scholar]
- Jiang, L.-M.; Chen, L.; Zhou, Z.; Sun, D.; Li, Y.; Zhang, M.; Liu, Y.; Du, S.; Chen, G.; Yao, J. Fouling Characterization and Aeration Performance Recovery of Fine-Pore Diffusers Operated for 10 Years in a Full-Scale Wastewater Treatment Plant. Bioresour. Technol. 2020, 307, 123197. [Google Scholar] [CrossRef]
- Bencsik, D.; Wadhawan, T.; Takács, I.; Bott, C.; Rosso, D. Improved Aeration Modelling Using Innovative Concepts for Prediction of Key Factors in Oxygen Transfer. In Proceedings of the Innovations in Process Engineering 2021, Virtual, 9–10 and 15–16 June 2022; Water Environment Federation: Alexandria, VA, USA, 2021. [Google Scholar]
- Morgan, P.F.; Bewtra, J.K. Air diffuser efficiencies. J. Water Pollut. Control Fed. 1960, 32, 1047–1059. [Google Scholar]
- Eckenfelder, W.W. Factors affecting the aeration efficiency of sewage and industrial wastes. Sew. Ind. Wastes 1959, 31, 60–70. [Google Scholar]
- Wagner, M.R.; Pöpel, H.J. Oxygen transfer and aeration efficiency—Influence of diffuser submergence, diffuser density, and blower type. Water Sci. Technol. 1998, 38, 1–6. [Google Scholar] [CrossRef]
- Pöpel, H.J.; Wagner, M.R. Modelling of Oxygen Transfer in Deep Diffused-Aeration Tanks and Comparison with Full-Scale Plant Data. Water Sci. Technol. 1994, 30, 71–80. [Google Scholar] [CrossRef]
- Rosso, D.; Iranpour, R.; Stenstrom, M.K. Fifteen Years of Offgas Transfer Efficiency Measurements on Fine-Pore Aerators: Key Role of Sludge Age and Normalized Air Flux. Water Environ. Res. 2005, 77, 266–273. [Google Scholar] [CrossRef] [PubMed]
- Plósz, B.G.; Jobbágy, A.; Grady, C.P.L. Factors Influencing Deterioration of Denitrification by Oxygen Entering an Anoxic Reactor through the Surface. Water Res. 2003, 37, 853–863. [Google Scholar] [CrossRef] [PubMed]
- Sander, R. Compilation of Henry’s Law Constants (Version 4.0) for Water as Solvent. Atmos. Chem. Phys. 2015, 15, 4399–4981. [Google Scholar] [CrossRef]
- Mackay, D.; Shiu, W.Y. A Critical Review of Henry’s Law Constants for Chemicals of Environmental Interest. J. Phys. Chem. Ref. Data 1981, 10, 1175–1199. [Google Scholar] [CrossRef]
- Staudinger, J.; Roberts, P.V. A Critical Review of Henry’s Law Constants for Environmental Applications. Crit. Rev. Environ. Sci. Technol. 1996, 26, 205–297. [Google Scholar] [CrossRef]
- Staudinger, J.; Roberts, P.V. A Critical Compilation of Henry’s Law Constant Temperature Dependence Relations for Organic Compounds in Dilute Aqueous Solutions. Chemosphere 2001, 44, 561–576. [Google Scholar] [CrossRef] [PubMed]
- Water Environment Federation. Design of Municipal Wastewater Treatment Plants MOP 8, 5th ed.; McGraw-Hill Education: Alexandria, VA, USA, 2009; ISBN 978-0-07-166358-8. [Google Scholar]
- Cecen, F.; Aktas, Ö. Activated Carbon for Water and Wastewater Treatment: Integration of Adsorption and Biological Treatment; WILEY-VCH: Weinheim, Germany, 2011; ISBN 978-3-527-63945-8. [Google Scholar]
- Benedek, P.; Major, V.; Takács, I. Mathematical Model Suggested for a Carbon-Activated Sludge System. Water Res. 1985, 19, 407–413. [Google Scholar] [CrossRef]
- Sun, Y.-X.; Yang, Z.; Ye, T.; Shi, N.; Tian, Y. Evaluation of the Treatment of Reverse Osmosis Concentrates from Municipal Wastewater Reclamation by Coagulation and Granular Activated Carbon Adsorption. Environ. Sci. Pollut. Res. 2016, 23, 13543–13553. [Google Scholar] [CrossRef] [PubMed]
- Vahala, R.; Rintala, J.; Järvinen, A. Removal of TOC by Two-Step GAC Filtration in Cold Humic Waters. In Proceedings of the NOM Workshop, Poitiers, France, 18 September 1996. [Google Scholar]
- Alex, J.; Benedetti, L.; Copp, J.; Gernaey, K.; Jeppsson, U.; Nopens, I.; Pons, M.; Rieger, L.; Rosen, C.; Steyer, J.-P. Benchmark Simulation Model No. 1 (BSM1); Report by the IWA Taskgroup on Benchmarking of Control Strategies for WWTPs; Lund University: Lund, Sweden, 2008. [Google Scholar]
- Mrowiec, B. Effect of BTX on Biological Treatment of Sewage. Environ. Prot. Eng. 2009, 35, 197–206. [Google Scholar]
- Smith, R.C.; Elger, S.O.; Mleziva, S. Implementation of solids retention time (SRT) control in wastewater treatment. Xylem Anal. 2015, 20, 1–6. [Google Scholar]
- Benstoem, F.; Nahrstedt, A.; Boehler, M.; Knopp, G.; Montag, D.; Siegrist, H.; Pinnekamp, J. Performance of granular activated carbon to remove micropollutants from municipal wastewater—A meta-analysis of pilot-and large-scale studies. Chemosphere 2017, 185, 105–118. [Google Scholar] [CrossRef] [PubMed]
- Zha, Y.; Wang, Y.; Liu, S.; Liu, S.; Yang, Y.; Jiang, H.; Zhang, Y.; Qi, L.; Wang, H. Adsorption characteristics of organics in the effluent of ultra-short SRT wastewater treatment by single-walled, multi-walled, and graphitized multi-walled carbon nanotubes. Sci. Rep. 2018, 8, 17245. [Google Scholar] [CrossRef] [PubMed]
- Costa, A.S.; Romão, L.P.C.; Araújo, B.R.; Lucas, S.C.O.; Maciel, S.T.A.; Wisniewski Jr, A.; Alexandre, M.D.R. Environmental strategies to remove volatile aromatic fractions (BTEX) from petroleum industry wastewater using biomass. Bioresour. Technol. 2012, 105, 31–39. [Google Scholar] [CrossRef] [PubMed]
- Torfs, E.; Nicolaï, N.; Daneshgar, S.; Copp, J.B.; Haimi, H.; Ikumi, D.; Johnson, B.; Plosz, B.B.; Snowling, S.; Townley, L.R.; et al. The Transition of WRRF Models to Digital Twin Applications. Water Sci. Technol. 2022, 85, 2840–2853. [Google Scholar] [CrossRef]
- Zietzschmann, F.; Stützer, C.; Jekel, M. Granular activated carbon adsorption of organic micro-pollutants in drinking water and treated wastewater–aligning breakthrough curves and capacities. Water Res. 2016, 92, 180–187. [Google Scholar] [CrossRef]


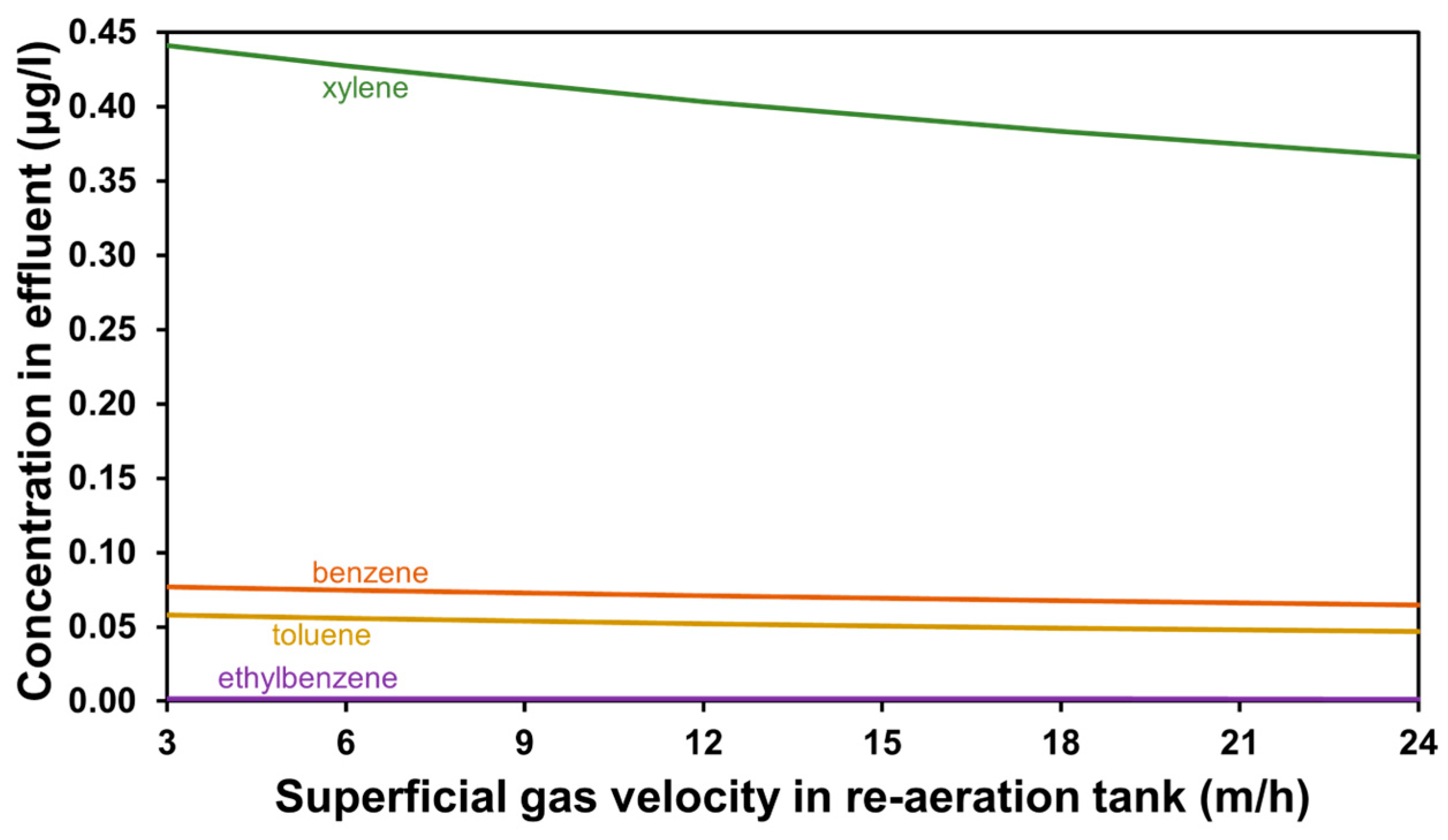
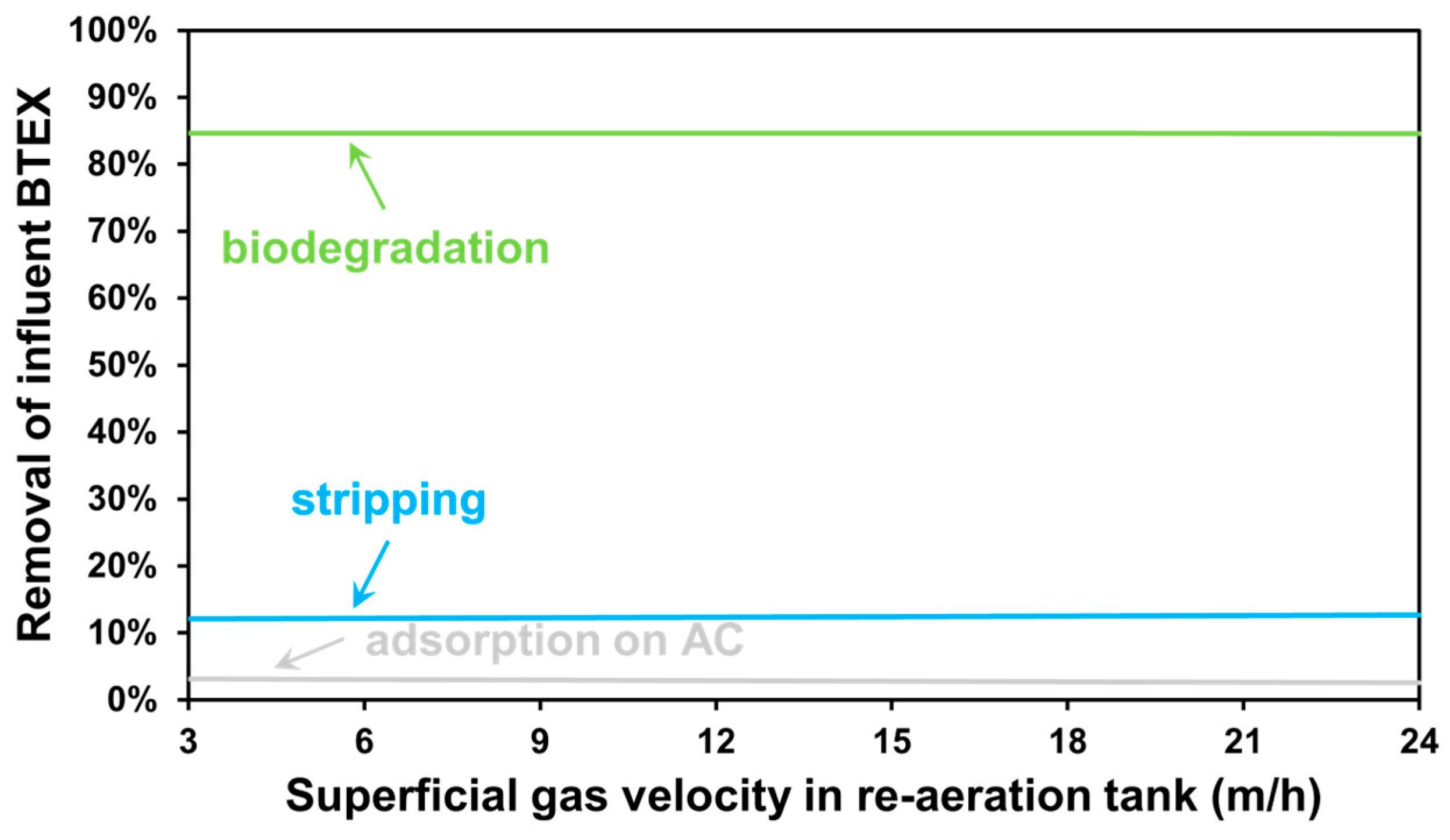

| Parameter | Value | Unit |
|---|---|---|
| Influent properties | ||
| Flow | 18,446 | m3 d−1 |
| COD | 360 | gCOD m−3 |
| Filtered COD | 144 | gCOD m−3 |
| TOC | 114 | gC m−3 |
| TKN | 47 | gN m−3 |
| NH4-N | 30 | gN m−3 |
| Tank dimensions | ||
| Anoxic 1 zone volume | 1000 | m3 |
| Anoxic 2 zone volume | 1000 | m3 |
| Aerobic 1 zone volume | 1333 | m3 |
| Aerobic 2 zone volume | 1333 | m3 |
| Aerobic 3 zone volume | 1333 | m3 |
| Clarifier surface area | 1500 | m2 |
| Clarifier depth | 4 | m |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bencsik, D.; Wadhawan, T.; Házi, F.; Karches, T. Plant-Wide Models for Optimizing the Operation and Maintenance of BTEX-Contaminated Wastewater Treatment and Reuse. Environments 2024, 11, 88. https://doi.org/10.3390/environments11050088
Bencsik D, Wadhawan T, Házi F, Karches T. Plant-Wide Models for Optimizing the Operation and Maintenance of BTEX-Contaminated Wastewater Treatment and Reuse. Environments. 2024; 11(5):88. https://doi.org/10.3390/environments11050088
Chicago/Turabian StyleBencsik, Dániel, Tanush Wadhawan, Ferenc Házi, and Tamás Karches. 2024. "Plant-Wide Models for Optimizing the Operation and Maintenance of BTEX-Contaminated Wastewater Treatment and Reuse" Environments 11, no. 5: 88. https://doi.org/10.3390/environments11050088
APA StyleBencsik, D., Wadhawan, T., Házi, F., & Karches, T. (2024). Plant-Wide Models for Optimizing the Operation and Maintenance of BTEX-Contaminated Wastewater Treatment and Reuse. Environments, 11(5), 88. https://doi.org/10.3390/environments11050088








