Assessment of Dispersed Oil Sorption in Oily Wastewater onto Hydrophobized/Oleophilized Autoclaved Aerated Concrete (AAC) Grains
Abstract
1. Introduction
2. Materials and Methods
2.1. AAC Grains and Sands
2.2. Hydrophobic Agents and Coating
2.3. Dispersed Oil
2.4. Batch Sorption Test
2.5. Analysis
3. Results and Discussions
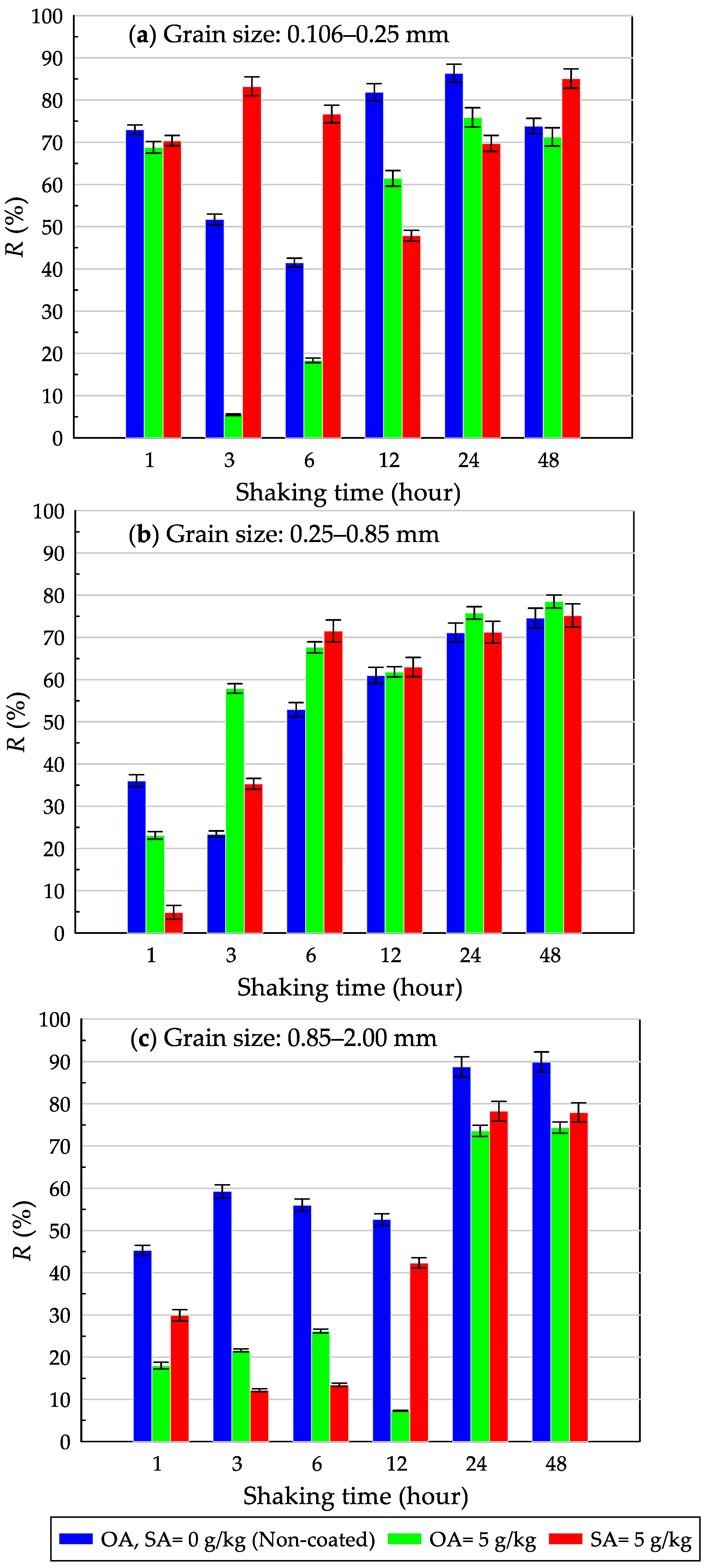
3.1. Effects of Shaking Time on Removal of Dispersed Oil in Water
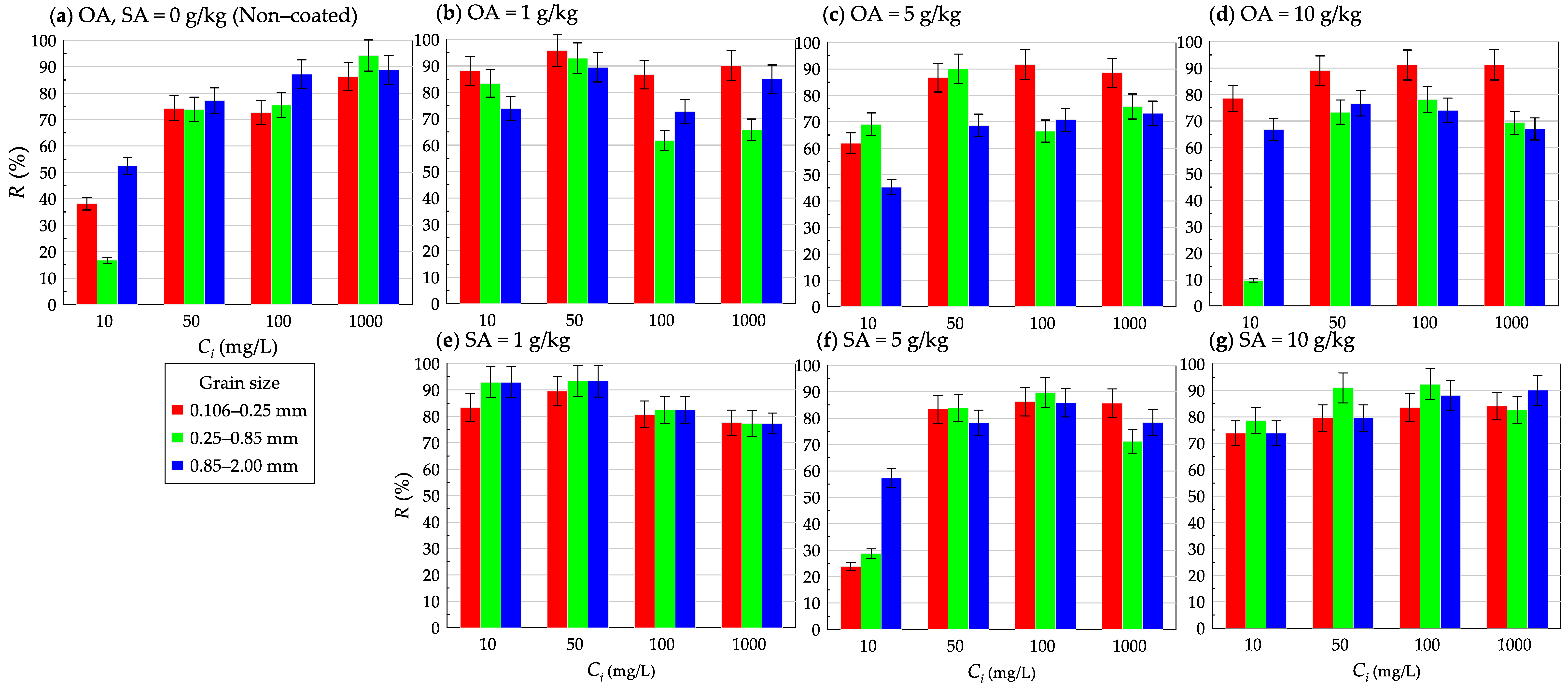
3.2. Effect of Initial Dispersed Oil Concentration on Removal Percent of Dispersed Oil
3.3. Application of Adsorption Isotherm Models to Characterize the Dispersed Oil Sorption onto AAC Grains
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AAC | Autoclaved aerated concrete |
| Ce | Equilibrium concentration of dispersed oil in water |
| Ci | Initial concentration of dispersed oil in water |
| HA | Hydrophobic agents |
| OA | Oleic acid |
| qe | Oil sorption amount at equilibrium (mg/g) |
| R | Removal percentage (%) of dispersed oil in water |
| SA | Stearic acid |
Appendix A
| Act Name | Parameter | Environmental Standards (mg/L) | Analysis Method | |||
|---|---|---|---|---|---|---|
| Environmental standards: Surface water, groundwater, seawater, and others | ||||||
| QCVN 08–MT:2015/BTNMT [7] | Total oil and grease | Domestic use | Irrigation | TCVN 7875: 2008 5 [8] SMEWW 5520B: 2012 [9] | ||
| A1 1 | A2 1 | B1 1 | B2 1 | |||
| 0.3 | 0.5 | 1.0 | 1.0 | |||
| QCVN 10–MT:2015/BTNMT [10] | Total mineral oil and grease | Area of aquaculture and conservation | Beach and water sport area | Other areas | TCVN 7875: 2008 5 [8] SMEWW 5520B: 2012 [9] SMEWW 5520C: 2012 [11] | |
| 0.5 | 0.5 | 0.5 | ||||
| QCVN 38:2011/BTNMT [12] | Total mineral oil and grease | 0.05 | ||||
| Emission limits: Domestic and industrial wastewater | ||||||
| QCVN 14:2008/BTNMT [13] | Animal and vegetable oil and grease | A 2 | B 2 | EPA Method 1664: 2010 [14] | ||
| 10 | 20 | |||||
| QCVN 40:2011/BTNMT [15] | Total mineral oil and grease | A 2 | B 2 | TCVN 7875: 2008 5 [8] EPA Method 1664: 2010 [14] | ||
| 5 | 10 | |||||
| Emission limits: Specific wastewater from industries and service sectors | ||||||
| QCVN 14:2008/BTNMT [13] | Animal and vegetable oil and grease | A 3 | B 3 | EPA Method 1664: 2010 5 [14] | ||
| 10 | 20 | |||||
| QCVN 29:2010/BTNMT [16] | Mineral oil (Total hydrocarbon) | A 4 | B 4 | TCVN 7875: 2008 5 [8] | ||
| Warehouse | Car wash | Stations | ||||
| 5 | 15 | 18 | 30 | |||
| QCVN 52:2013/BTNMT [17] | Total mineral oil | A 3 | B 3 | TCVN 7875: 2008 5 [8] SMEWW 5520C: 2012 [11] | ||
| 5 | 10 | |||||
| QCVN 11–MT:2015/BTNMT [18] | Total animal and vegetable oil | A 3 | B 3 | TCVN 7875: 2008 5 [8] | ||
| 10 | 20 | SMEWW 5520C: 2012 [11] | ||||
| Location | Province/ Prefecture or City | Year | Oil and Grease (mg/L) | Remarks | Reference |
|---|---|---|---|---|---|
| Surface water | |||||
| Cau River | Thai Nguyen/Thai Nguyen | 2004, 2005 | 0.02–0.16 | 6 monitoring points | MONRE, WB and DANIDA (2006) [64] |
| Dong Nai River | Hoa An/Cao Bang | 2000–2006 | 0.025–0.029 | ||
| Saigon River | Binh Phuoc/Binh Duong, etc. | 2000–2006 | 0.025–0.12 | ||
| Saigon–Nha Be River | Thu Dau Mot/Binh Duong, etc. | N/D | 0.0–0.3 | 5 monitoring points | IGES (2007) [65] |
| Ha Thanh River | Quy Nhon/Binh Dinh | 2016 | 27–250 | Ha Thanh Bridge, Canal, Channel | Binh Dinh PPC (2016) [66] |
| Ditch | Quy Nhon/Binh Dinh | 2019 | 15 | An upstream ditch of Bau Sen Lake | Nguyen et al. (2020) [67] |
| Coastal water | |||||
| Red and Mekong River | 1996–2001 | >0.05–2.4 | MONRE (2003) [4] | ||
| Coastal zone (1) | Rach Gia/Kien Giang: | 1997–2015 | 0.12–0.79 | Rach Gia monitoring station | Le and Pham (2017) [68] |
| Coastal zone (2) | Nha Trang/Khanh Hoa | 2013–2017 | 0.35–0.55 | Nha Trang monitoring station | Pham (2018) [69] |
| Coastal zone (3) | Khanh Hoa/Trung binh, etc. | 2010–2018 | 0.007–0.65 | 4 monitoring stations | Pham (2019) [70] |
| Coastal zones | Hue/Thua Thien Hue, etc. | 2011–2015 | 0.012–1.3 | Total of 16 monitoring stations | MONRE (2015) [71] |
| Domestic wastewater | |||||
| In and Out of WWTP | Quy Nhon/Binh Dinh | 2016 | 0.8–160 | Nearby Dam, Nhon Binh, Bau Luc | Binh Dinh PPC (2016) [66] |
| In and Out of WTF | Hai Ba Trung/Hanoi | 2016 | 2.0–14.6 | WTF for condominium in Hanoi | JICA (2019) [72] |
| Industrial wastewater | |||||
| Landfill leachate | Tuy Phuoc/Binh Dinh | 2016 | 3.79 × 104 | Long My waste landfill | Binh Dinh PPC (2016) [66] |
| Wastewater from construction site | Quy Nhon/Binh Dinh | 2016 | 200 | Cleaning and washing of equipment and materials at a bridge construction site | |
| Location | Year | Type and Sources | Oil Pollution Loads/Oil Spills | Descriptions and Other Remarks | Reference |
|---|---|---|---|---|---|
| East sea | 1995–2002 | Oil spills | Approx. 7.72 × 105 tons/year | Oil leaks from crude oil exploitation. A total of 9.2 × 104 tons is estimated to flow into the coastal and marine environment. Total financial losses caused by a major oil spill in 2001 were estimated at 250 billion VND (17 million USD), while costs for cleaning up polluted waters and beaches reached 60 billion VND (4 million USD). | ADB (2006) [73]; MONRE, WB and DANIDA (2003) [4] |
| Dong Nai River basin | 2004 | Domestic wastewater: HCMC 5 provinces | 3.45 × 104 kg/day 90–4.1 × 103 kg/day | MONRE, WB and DANIDA (2006) [64] | |
| Cau River basin | 2005 | Domestic wastewater from 6 provinces | 66 tons/day | ||
| Cau Bay River basin | 2013 | 48 enterprises at Long Bien District, Hanoi | 6.34 kg/day | JICA (2013) [74] | |
| HCMC | 2000 | Industrial wastewater | Major industrial contributors are oil refining, chemical, and food processing industries (20–30% of the total industrial wastewater discharges into river systems in Vietnam from HCMC) | ADB (2000) [75] |
| Location | Year | Type and Sources | Oil Pollution Loads/Oil Spills | Descriptions and Other Remarks | Reference |
|---|---|---|---|---|---|
| Craft village | 2002 | Tra Co cassava processing village Bao Loc silk village | (BOD5 kg/day, COD kg/day) 218–379, 145–544 | Pollution loads of SS, TN, TP, CN andNH4+ | MONRE, WB and DANIDA (2006) [64] |
| Whole country | 2003 | (Volume of wastewater) Total | (m3/year) 896 × 106 | ADB, GEF, UNEP (2006) [73]; WWF (2018) [76] | |
| Whole country: Industrial zones | 2005 | (Load of pollutants) HCMC, Dong Nai, Binh Duong, BR–VT | (BOD5 kg/day, COD kg/day) 2.24 × 102–1.28 × 104, 1.06 × 103–4.68 × 104 | Pollution loads of TSS, TN, TP, and wastewater volume | MONRE, WB and DANIDA (2006) [64] |
| Hanoi | 2005 | (Wastewater discharge from industrial sectors) Chemical factories, Textile and dyeing, Foodstuff companies, Mechanical factories | (m3/day) 3.73 × 103–2.65 × 104 | MONRE, WB and DANIDA (2006) [64] | |
| Food processing villages | 2005 | (Wastewater discharge) Phu Do rice noodle, Vu Hai rice noodle, Ninh Hong rice noodle, Tan Do wine, etc. | (BOD5 tons/year, COD tons/year) 10–53, 15–1.3 × 104 | MONRE (2008) [24] | |
| Red River Delta | 2006 | (Wastewater discharge) Food processing, husbandry, slaughtering Weaving, dyeing, leather processing, etc. | (COD tons/year) ~1.5 × 104 ~1.5 × 103 | MONRE (2008) [24] | |
| Nhue–Day River | 2006 | (Proportion of wastewater discharge) Domestic, Industrial, Farming and husbandry, etc. | 4–56% | MONRE, WB and DANIDA (2006) [64] | |
| Hanoi, Hai Phong HCMC | 2009 | (Wastewater discharge) Domestic wastewater, Industrial wastewater, etc. | (BOD5 tons/day) 0.3–214 | Nguyen et al. (2020) [67] | |
| Hanoi, Hai Phong Da Nang, HCMC | 2010 | Share of pollution load on the sum of domestic and industrial wastewater. Domestic wastewater and Industrial wastewater | 15–85% | 1 | Do and Nguyen (2014) [77] |
| Hanoi, Hai Phong Da Nang, HCMC | 2010 2019 | Wastewater discharge into canals from agricultural activities Domestic wastewater discharge rate and amount | (BOD5 tons/day, COD tons/day) 17–193, 26–305 | Pollution loads of SS, TSS are also given. | VEA (2012) [78] |
| Whole country | 2019 | Domestic wastewater discharge rate and amount. Upper: Urban area Lower: Rural area | 150 L/capita/day (4.66 × 106 m3/day) 80 L/capital/day (4.85 × 106 m3/day) | Blackwater 93–94%, Graywater 6–7% (Hanoi, Hai Phong) | JICA (2019) [72] |
References
- Walker, A.H. Chapter 1—Oil Spills and Risk Perceptions. In Oil Spill Science and Technology, 2nd ed.; Fingas, M., Ed.; Gulf Professional Publishing: Boston, MA, USA, 2017; pp. 1–70. [Google Scholar]
- Huynh, B.Q.; Kwong, L.H.; Kiang, M.V.; Chin, E.T.; Mohareb, A.M.; Jumaan, A.O.; Basu, S.; Geldsetzer, P.; Karaki, F.M.; Re hkopf, D.H. Public health impacts of an imminent Red Sea oil spill. Nat. Sustain. 2021, 4, 1084–1091. [Google Scholar] [CrossRef] [PubMed]
- Murawski, S.A.; Ainsworth, C.H.; Gilbert, S.; Hollander, D.J.; Paris, C.B.; Schlüter, M.; Wetzel, D.L. Introduction to the Volume. In Scenarios and Responses to Future Deep Oil Spills: Fighting the Next War; Murawski, S.A., Ainsworth, C.H., Gilbert, S., Hollander, D.J., Paris, C.B., Schlüter, M., Wetzel, D.L., Eds.; Springer International Publishing: Berlin/Heidelberg, Germany, 2020; pp. 4–15. [Google Scholar]
- Ministry of Natural Resources Environment Vietnam (MoNRE); World Bank; DANIDA. Vietnam Environment Monitor 2003: Water; World Bank: Hanoi, Vietnam, 2003.
- Asia Pro Eco Programme. The Risks and Consequences of Oil Pollution in Vietnam and Guidelines on Bioremediation of Oil Spills; European Commission: Bremen, Germany, 2006. [Google Scholar]
- Institute for Global Environmental Strategies (IGES). Water Resources Management in Ho Chi Minh City. In Sustainable Groundwater Management in Asian Cities: A Final Report of Research on Sustainable Water Management Policy; Institute for Global Environmental Strategies: Hayama, Japan, 2006; p. 97. [Google Scholar]
- QCVN 08-MT:2015/BTNMT National Technical Regulation on Surface Water Quality. 2015. Available online: https://cem.gov.vn/storage/documents/5d6f3ecb26484qcvn-08-mt2015btnmt.pdf (accessed on 25 January 2023).
- TCVN 7875: 2008 Water—Determination of Oil and Grease—Partition-Infrared Method. 2008. Available online: https://vanbanphapluat.co/data/2017/08/289065_tcvn7875-2008.pdf (accessed on 5 January 2023).
- American Water Works Association; Water Environment Federation. 5520B Oil and Grease by Partition-Gravimetric Method; The partition-gravimetric method is recommended for liquid samples. In Standard Methods for the Examination of Water and Wastewater; American Public Health Association: Washington, DC, USA, 2012. [Google Scholar]
- QCVN 10-MT:2015/BTNMT National Technical Regulation on Marinewater Quality-2015. 2015. Available online: http://vea.gov.vn/Quy%20chun%20Vit%20Nam/QCVN%2010-MT-2015-BTNMT.pdf (accessed on 5 January 2023).
- American Water Works Association; Water Environment Federation. 5520C Oil and Grease by Partition-Infrared Method. In Standard Methods for the Examination of Water and Wastewater; American Public Health Association: Washington, DC, USA, 2012. [Google Scholar]
- QCVN 38:2011/BTNMT National Technical Regulation on Surface Water Quality for Protection of Aquatic Lifes. 2011. Available online: https://faolex.fao.org/docs/pdf/vie205179.pdf (accessed on 10 January 2023).
- QCVN 14: 2008/BTNMT National Technical Regulation on Domestic Wastewater. 2008. Available online: http://vea.gov.vn/Quy%20chun%20Vit%20Nam/QCVN%2014-2008-BTNMT.pdf (accessed on 10 January 2023).
- Environmental Protection Agency (US EPA). Method 1664, Revision B: n-Hexane Extractable Material (HEM; Oil and Grease) and Silica Gel Treated n-Hexane Extractable Material (SGT-HEM; Non-Polar Material) by Extraction and Gravimetry; Environmental Protection Agency (US EPA): Washington, DC, USA, 2010. [Google Scholar]
- QCVN 40:2011/BTNMT National Technical Regulation on Industrial Wastewater. 2011. Available online: https://emas.tdtu.edu.vn/sites/emas/files/EMAS/Văn%20bản%20pháp%20luật/qcvn-40-nước-thải-cn.pdf (accessed on 11 January 2023).
- QCVN 29:2010/BTNMT National Technical Regulation On the Effluent of Petroleum Terminal and Stations. 2010. Available online: http://vea.gov.vn/Quy%20chun%20Vit%20Nam/QCVN%2029-2010-BTNMT.pdf (accessed on 3 January 2023).
- QCVN 52:2013/BTNMT National Technical Regulation on Wastewater of Steel Industry. 2013. Available online: https://circabc.europa.eu/sd/a/c1026ee2-028a-4f2b-8a26-18667b751727/National%20Technical%20Regulation%20on%20Wastewater%20of%20Steel%20Industry.pdf (accessed on 10 January 2023).
- QCVN 11-MT:2015/BTNMT National Technical Regulation on the Effluent of Aquatic Products Processing Industry. 2015. Available online: http://www.gree-vn.com/pdf/QCVN-11-MT-2015-BTNMT-nuoc-thai-che-bien-thuy-san.pdf (accessed on 4 January 2023).
- Wang, B.; Liang, W.; Guo, Z.; Liu, W. Biomimetic super-lyophobic and super-lyophilic materials applied for oil/water separa tion: A new strategy beyond nature. Chem. Soc. Rev. 2015, 44, 336–361. [Google Scholar] [CrossRef] [PubMed]
- Yong, J.; Huo, J.; Chen, F.; Yang, Q.; Hou, X. Oil/water separation based on natural materials with super-wettability: Recent advances. Phys. Chem. Chem. Phys. 2018, 20, 25140–25163. [Google Scholar] [CrossRef] [PubMed]
- Hassanshahian, M.; Emtiazi, G.; Caruso, G.; Cappello, S. Bioremediation (bioaugmentation/biostimulation) trials of oil polluted seawater: A mesocosm simulation study. Mar. Environ. Res. 2014, 95, 28–38. [Google Scholar] [CrossRef]
- Cheng, Y.; Wang, L.; Faustorilla, V.; Megharaj, M.; Naidu, R.; Chen, Z. Integrated electrochemical treatment systems for facil itating the bioremediation of oil spill contaminated soil. Chemosphere 2017, 175, 294–299. [Google Scholar] [CrossRef]
- Pintor, A.M.A.; Vilar, V.J.P.; Botelho, C.M.S.; Boaventura, R.A.R. Oil and grease removal from wastewaters: Sorption treatment as an alternative to state-of-the-art technologies. A critical review. Chem. Eng. J. 2016, 297, 229–255. [Google Scholar] [CrossRef]
- Ministry of Natural Resources Environment Vietnam (MoNRE). National State of Environment 2008 Vietnam Craft Village Environment; Ministry of Natural Resources Environment Vietnam: Hanoi, Vietnam, 2008.
- Waterworks Bureau City of Kawasaki Japan. Oil and Public Sewage Systems; Waterworks Bureau City of Kawasaki Japan: Kawasaki, Japan, 2020. [Google Scholar]
- You, Z.; Xu, H.; Sun, Y.; Zhang, S.; Zhang, L. Effective treatment of emulsified oil wastewater by the coagulation-flotation process. RSC Adv. 2018, 8, 40639–40646. [Google Scholar] [CrossRef]
- Medeiros, A.D.L.M.d.; Silva Junior, C.J.G.d.; Amorim, J.D.P.d.; Durval, I.J.B.; Costa, A.F.d.S.; Sarubbo, L.A. Oily WastewaterTreatment: Methods, Challenges, and Trends. Processes 2022, 10, 743. [Google Scholar] [CrossRef]
- Stewart, M.; Arnold, K. Chapter 1—Crude Oil Treating Systems. In Emulsions and Oil Treating Equipment; Stewart, M., Arnold, K., Eds.; Gulf Professional Publishing: Burlington, VT, USA, 2009; pp. 1–80. [Google Scholar]
- Oliveira, L.; Saleem, J.; Bazargan, A.; Duarte, J.; McKay, G.; Meili, L. Sorption as a rapidly response for oil spill accidents: A material and mechanistic approach. J. Hazard. Mater. 2021, 407, 124842. [Google Scholar] [CrossRef]
- Torres, C.E.I.; Quezada, T.E.S.; Kharissova, O.V.; Kharisov, B.I.; de la Fuente, M.I.G. Carbon-based aerogels and xerogels: Synthesis, properties, oil sorption capacities, and DFT simulations. J. Environ. Chem. Eng. 2021, 9, 104886. [Google Scholar] [CrossRef]
- Al-Majed, A.A.; Adebayo, A.R.; Hossain, M.E. A sustainable approach to controlling oil spills. J. Environ. Manag. 2012, 113, 213–227. [Google Scholar] [CrossRef]
- El Gheriany, I.A.; El Saqa, F.A.; El Razek Amer, A.A.; Hussein, M. Oil spill sorption capacity of raw and thermally modified orange peel waste. Alex. Eng. J. 2020, 59, 925–932. [Google Scholar] [CrossRef]
- Akihiro, M.; Muhammad, J.Z.; Takeshi, S.; Thanh, D.H.T.; Thanh, H.P.; Viet, N.T.T.; Ken, K. Oil/water separation techniques using hydrophobized/oleophilized grains: A review of recent studies. GEOMATE J. 2021, 20, 28–34. [Google Scholar]
- Gong, Z.; Alef, K.; Wilke, B.M.; Li, P. Activated carbon adsorption of PAHs from vegetable oil used in soil remediation. J. Hazard. Mater. 2007, 143, 372–378. [Google Scholar] [CrossRef]
- Liu, L.; Wang, L.; Song, W.; Yang, L.; Yin, L.; Xia, S.; Wang, H.; Strong, P.J.; Song, Z. Crude oil removal from aqueous solution using raw and carbonized Xanthoceras sorbifolia shells. Environ. Sci. Pollut. Res. Int. 2018, 25, 29325–29334. [Google Scholar] [CrossRef]
- Jain, S.N.; Garud, V.B.; Dawange, S.D.; Sonawane, D.D.; Shaikh, E.R. Sesame (Sesamum indicum) oil cake—Industrial waste biomass for sequestration of Basic Blue 26 from aqueous media. Biomass Convers. Biorefinery 2020, 12, 3783–3793. [Google Scholar] [CrossRef]
- Van, T.N.; Ton, K.T.; Thanh, H.D.T.; Viet, N.T.T.; Hoang, G.N.; Tien, D.N.; Yugo, I.; Tomonori, I.; Ken, K. Current status of construction and demolition waste management in vietnam: Challenges and opportunities. GEOMATE J. 2018, 15, 23–29. [Google Scholar]
- Giang, N.H.; Tan, N.H.; Quang, N.T.; Hien, N.X.; Kawamoto, K. Sustainable management and technologies for recycled con struction demolition waste in Vietnam. IOP Conf. Ser. Mater. Sci. Eng. 2020, 869, 032040. [Google Scholar] [CrossRef]
- Nguyen, H.G.; Nguyen, D.T.; Nghiem, H.T.; Tran, V.C.; Kato, A.; Matsuno, A.; Isobe, Y.; Kawasaki, M.; Kawamoto, K. Current Management Condition and Waste Composition Characteristics of Construction and Demolition Waste Landfills in Hanoi of Vietnam. Sustainability 2021, 13, 10148. [Google Scholar] [CrossRef]
- Alexanderson, J. Relations between structure and mechanical properties of autoclaved aerated concrete. Cem. Concr. Res. 1979, 9, 507–514. [Google Scholar] [CrossRef]
- Nguyen Trong, L.; Asamoto, S.; Matsui, K. Sorption isotherm and length change behavior of autoclaved aerated concrete. Cem. Concr. Compos. 2018, 94, 136–144. [Google Scholar] [CrossRef]
- Akihiro, M.; Shin, I.; Lam, N.T.; Tien, D.N.; Tuan, N.V.; Giang, N.H.; Ken, K. Comparison of macropore structures and networks of autoclaved aerated concrete blocks using micro-focus X-ray computed tomography. GEOMATE J. 2020, 19, 160–165. [Google Scholar]
- Aroni, S. Autoclaved Aerated Concrete—Properties, Testing and Design; CRC Press: Boca Raton, FL, USA, 2014. [Google Scholar]
- Tada, S. Microstructural approach to properties of moist cellular concrete. In Autoclaved Aerated Concrete-Moisture and Properties; Elsevier Science Ltd.: Amsterdam, The Netherlands, 1983. [Google Scholar]
- Wittmann, F.H. Autoclaved Aerated Concrete Moisture and Properties; Elsevier Scientific Publishing Co.: New York, NK, USA, 1983. [Google Scholar]
- Kumara, G.M.P.; Kawamoto, K.; Saito, T.; Hamamoto, S.; Asamoto, S. Evaluation of Autoclaved Aerated Concrete Fines for Removal of Cd(II) and Pb(II) from Wastewater. J. Environ. Eng. 2019, 145, 04019078. [Google Scholar] [CrossRef]
- Kumara, G.M.P.; Kawamoto, K. Steel Slag and Autoclaved Aerated Concrete Grains as Low-Cost Adsorbents to Remove Cd2+ and Pb2+ in Wastewater: Effects of Mixing Proportions of Grains and Liquid-to-Solid Ratio. Sustainability 2021, 13, 10321. [Google Scholar] [CrossRef]
- Dang, H.T.T.; Le, C.Q.; Tran, S.H.; Kawamoto, K.; Tran, N.T.V.; Huyen, P.T. Application of Autoclaved Aerated Concrete Grains Coated with Stearic Acid for Removal of Oil from Water. Water Air Soil Pollut. 2022, 233, 530. [Google Scholar] [CrossRef]
- Thai, H.N.; Kawamoto, K.; Nguyen, H.G.; Sakaki, T.; Komatsu, T.; Moldrup, P. Measurements and Modeling of Thermal Con ductivity of Recycled Aggregates from Concrete, Clay Brick, and Their Mixtures with Autoclaved Aerated Concrete Grains. Sustainability 2022, 14, 2417. [Google Scholar] [CrossRef]
- JIS Z 8801-1: 2019; Test Sieves—Part 1: Test Sieves of Metal Wire Cloth. Japanese Standards Association: Tokyo, Japan, 2019.
- JWWA A 103-1: 2006-2; Filter Sand for Water Supply. Japan Water Works Association: Tokyo, Japan, 2006; p. 18.
- Subedi, S.; Kawamoto, K.; Jayarathna, L.; Vithanage, M.; Moldrup, P.; Wollesen de Jonge, L.; Komatsu, T. Characterizing Time-Dependent Contact Angles for Sands Hydrophobized with Oleic and Stearic Acids. Vadose Zone J. 2012, 11, vzj2011-0055. [Google Scholar] [CrossRef]
- Wijewardana, N.S.; Kawamoto, K.; Moldrup, P.; Komatsu, T.; Kurukulasuriya, L.C.; Priyankara, N.H. Characterization of wa ter repellency for hydrophobized grains with different geometries and sizes. Environ. Earth Sci. 2015, 74, 5525–5539. [Google Scholar] [CrossRef]
- Leelamanie, D.A.L.; Karube, J.; Yoshida, A. Characterizing water repellency indices: Contact angle and water drop penetration time of hydrophobized sand. Soil Sci. Plant Nutr. 2008, 54, 179–187. [Google Scholar] [CrossRef]
- Karatza, Z.; Buckman, J.; Medero, G.M.; Beckett, C.T.S. Evolution of meniscus structures in hydrophobic granular systems. J. Hydrol. 2021, 603, 126954. [Google Scholar] [CrossRef]
- Matsuno, A.; Kawamoto, K. Hydrophobicity/Oleophilicity of Autoclaved Aerated Concrete (AAC) Grains Coated with Oleic and Stearic Acids for Application as Oil/Water Separating Filtration and Adsorbent Materials in Vietnam. Environments 2022, 9, 101. [Google Scholar] [CrossRef]
- The Chemical Daily. 15710 Chemical Products; The Chemical Daily Co., Ltd.: Tokyo, Japan, 2010. [Google Scholar]
- Organization for Economic Co-Operation and Development Publications (OECD). Guidelines for the Testing of Chemicals; Organization for Economic Co-Operation and Development Publications: Paris, France, 2000. [Google Scholar]
- ASTM F726-99; Standard Test Method for Sorbent Performance of Adsorbents. ASTM Committee on Standards: West Conshohocken, PA, USA, 1998.
- Langmuir, I. The adsorption of gases on plane surfaces of glass, mica and platinum. J. Am. Chem. Soc. 1918, 40, 1361–1403. [Google Scholar] [CrossRef]
- Freundlich, H. Kapillarchemie: Eine Darstellung der Chemie der Kolloide und Verwandter Gebiete; Akademische Verlagsgesellschaft: New York, NY, USA, 1922. [Google Scholar]
- Huang, J.; Yan, Z. Adsorption Mechanism of Oil by Resilient Graphene Aerogels from Oil-Water Emulsion. Langmuir 2018, 34, 1890–1898. [Google Scholar] [CrossRef]
- Jing, H.; Lu, S.; Ji, L.; Li, S.; Zhu, B.; Guo, J.; Sun, J.; Cai, L.; Wang, Y. Magnetic Wakame-Based Biochar/Ni Composites with Enhanced Adsorption Performance for Diesel. J. Renew. Mater. 2022, 10, 3147–3165. [Google Scholar] [CrossRef]
- Ministry of Natural Resources Environment Vietnam (MoNRE); World Bank; DANIDA. Vietnam Environment Monitor 2006: Water Quality in Vietnam with a Focus on the Cau, Nhue-Day and Dong Nai River Basins; World Bank: Hanoi, Vietnam, 2006.
- Institute for Global Environmental Strategies (IGES). Water Resources Management in Ho Chi Minh City. In Sustainable Groundwater Management in Asian Cities: A Final Report of Research on Sustainable Water Management Policy; Institute for Global Environmental Strategies: Hayama, Japan, 2007; p. 69. [Google Scholar]
- Binh Dinh Provincial People’s Committee (PPC). Environmental and Social Impact Assessment Report on Coastal Cities Sustainable Environment Project (CCSEP): Quy Nhon City Sub-Project; World Bank Group: Washington, DC, USA, 2016.
- Nguyen, H.A. Wastewater management and treatment in urban areas in Viet Nam. In Proceedings of the 1st International Workshop on Water Environment Partnership in Asia (WEPA), Hanoi, Vietnam, 8–9 March 2010; pp. 211–215. [Google Scholar]
- Vinh, L.T.; Tam, P.H. Water quality at Rach Gia monitoring station (1997–2015). Vietnam J. Mar. Sci. Technol. 2017, 17, 222–231. [Google Scholar] [CrossRef]
- Pham, H.T. Coastal Seawater Quality from Data at South Vietnam Monitoring Stations during 2013–2017. VNU J. Sci. Earth Environ. Sci. 2018, 34, 2. [Google Scholar] [CrossRef]
- Pham, H.T. The Variation Trend of Environmental Quality at Corals in Coastal Areas of Khanh Hoa during 2010–2018. VNU J. Sci. Earth Environ. Sci. 2019, 35, 4. [Google Scholar] [CrossRef]
- Ministry of Natural Resources Environment Vietnam (MoNRE). Báo Cáo Hiện Trạng Môi Trường Quốc gia Giai Đoạn 2011–2015 (Vietnamese); Ministry of Natural Resources Environment Vietnam: Hanoi, Vietnam, 2015.
- Japan International Cooperation Agency (JICA). Final Report on JICA Survey on Examination of Monitoring Methodology for Indicator of SDG 6.3.1 in Vietnam; Japan International Cooperation Agency: Tokyo, Japan, 2019.
- Asian Development Bank (ADB); Global Environment Facility (GEF); United Nation Environment Programme (UNEP). Vietnam National Environmental Performance Assessment (EPA) Report, National Performance Assessment and Subregional Strategic Environment Framework in the Greater Mekong Subregion; Asian Development Bank: Mandaluyong, Philippines, 2006. [Google Scholar]
- Japan International Cooperation Agency (JICA). Project Completion Report on the Project for Strengthening Capacity of Water Environmental Management in Vietnam; Japan International Cooperation Agency: Tokyo, Japan, 2013.
- Asian Development Bank (ADB). Environments in Transition: Cambodia, Lao, Thailand, Vietnam; Asian Development Bank: Manila, Philippines, 2000; p. 41. [Google Scholar]
- WWF. WWF REPORT 2018 Textile and Garment Sector in Vietnam: Water Risks and Solutions; WWF: Gland, Switzerland, 2018. [Google Scholar]
- Do Nam Thang, N.T.C. Recent development of water environmental management in Vietnam. Water Environment Partnership in Asia (WEPA). In Proceedings of the Seminar on Current Situation and Future Prospect of Water Environment Governance in Asia, Tokyo, Japan, 23 January 2014. [Google Scholar]
- Vietnam Environment Administration (VEA). Báo Cáo Môi Trường Quốc gia 2012—Môi Trường Nước Mặt; Vietnam Environment Administration: Hanoi, Vietnam, 2012.




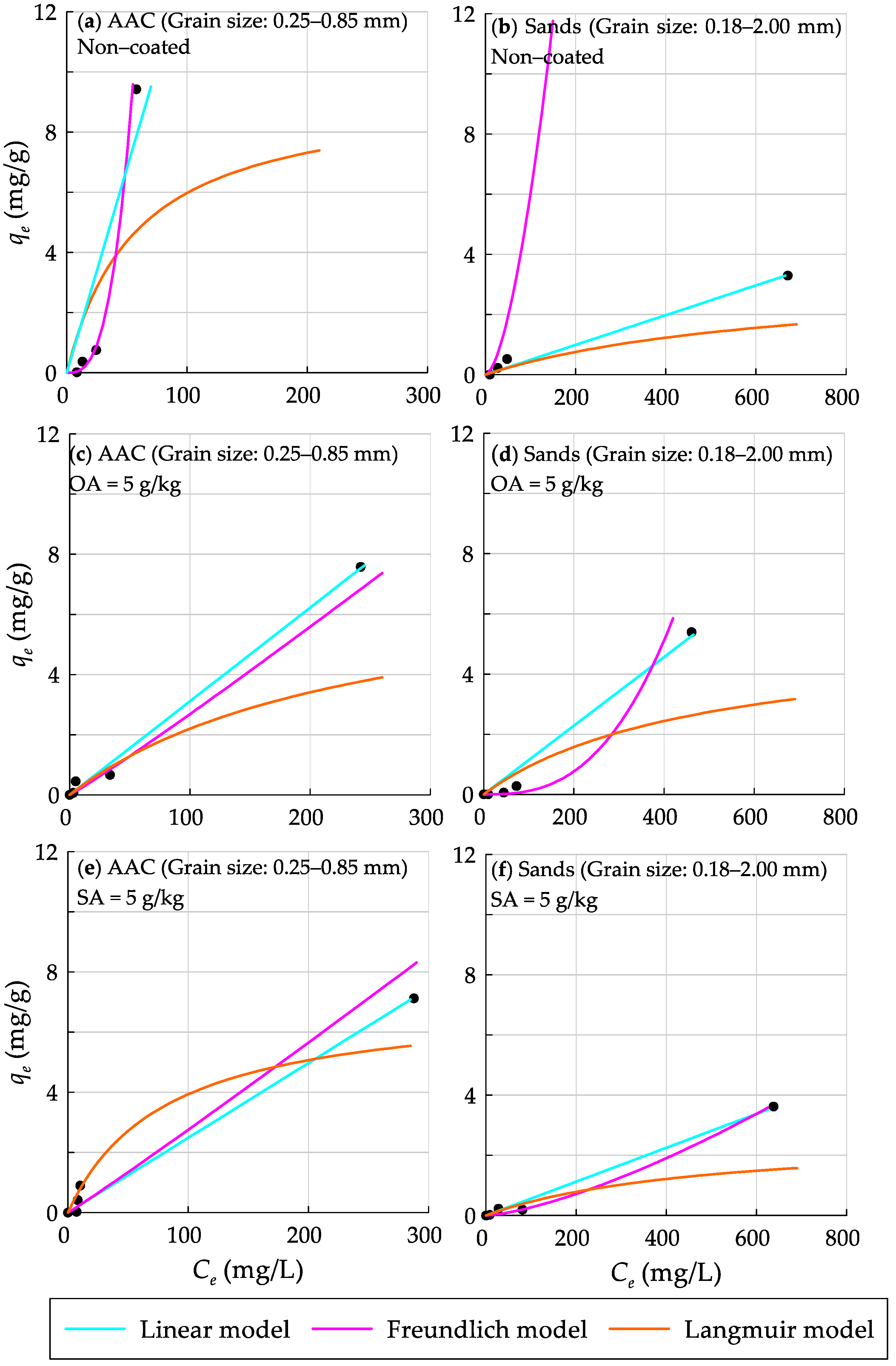
| Langmuir | Freundlich | Linear | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sample | Particle Size (mm) | Coating (g/kg) | qmax (mg/g) | b (L/mg) | R2 | 1/n | KF (mg/g) | R2 | Kd (L/mg) | R2 | Target Oil | Ref. |
| AAC | 0.106–0.25 | 0 | 8.6 | 7.3 × 10−3 | 0.97 | 1.7 | 3.0 × 10−3 | 0.99 | 6.1 × 10−2 | 0.99 | Soybean oil * | This Study |
| 0.25–0.85 | 9.4 | 1.7 × 10−2 | 0.86 | 3.0 | 6.3 × 10−5 | 0.99 | 1.4 × 10−1 | 0.94 | ||||
| 0.85–2.00 | 8.9 | 8.9 × 10−3 | 0.99 | 1.5 | 8.6 × 10−3 | 0.99 | 7.8 × 10−2 | 0.99 | ||||
| 0.106–0.25 | OA = 1 | 9.0 | 1.0 × 10−2 | 0.98 | 0.92 | 1.2 × 10−1 | 0.99 | 9.1 × 10−2 | 0.99 | |||
| 0.25–0.85 | 6.6 | 2.9 × 10−3 | 0.96 | 0.70 | 8.9 × 10−2 | 0.98 | 1.9 × 10−2 | 0.99 | ||||
| 0.85–2.00 | 8.5 | 6.7 × 10−3 | 0.85 | 1.0 | 4.0 × 10−2 | 0.99 | 5.6 × 10−2 | 0.99 | ||||
| 0.106–0.25 | OA = 5 | 8.9 | 8.7 × 10−3 | 0.81 | 1.3 | 2.6 × 10−2 | 0.99 | 7.7 × 10−2 | 0.99 | |||
| 0.25–0.85 | 7.6 | 4.1 × 10−3 | 0.93 | 1.1 | 2.1 × 10−2 | 0.99 | 3.1 × 10−2 | 0.99 | ||||
| 0.85–2.00 | 7.3 | 3.7 × 10−3 | 0.94 | 1.4 | 3.6 × 10−3 | 0.99 | 2.7 × 10−2 | 0.99 | ||||
| 0.106–0.25 | OA = 10 | 9.1 | 8.5 × 10−3 | 0.82 | 3.0 | 8.3 × 10−6 | 0.99 | 1.0 × 10−1 | 0.99 | |||
| 0.25–0.85 | 6.9 | 3.0 × 10−3 | 0.96 | 2.1 | 4.2 × 10−5 | 0.99 | 2.3 × 10−2 | 0.99 | ||||
| 0.85–2.00 | 6.7 | 2.8 × 10−3 | 0.96 | 1.6 | 7.8 × 10−4 | 0.99 | 2.0 × 10−2 | 0.99 | ||||
| 0.106–0.25 | SA = 1 | 7.8 | 4.5 × 10−3 | 0.92 | 0.89 | 6.9 × 10−2 | 0.99 | 3.5 × 10−2 | 0.99 | |||
| 0.25–0.85 | 7.7 | 4.4 × 10−3 | 0.93 | 0.73 | 1.4 × 10−1 | 0.99 | 3.4 × 10−2 | 0.99 | ||||
| 0.85–2.00 | 6.3 | 6.2 × 10−2 | 0.39 | 0.60 | 1.8 × 10−1 | 0.99 | 1.7 × 10−2 | 0.99 | ||||
| 0.106–0.25 | SA = 5 | 8.6 | 6.9 × 10−3 | 0.86 | 1.6 | 4.5 × 10−3 | 0.99 | 6.0 × 10−2 | 0.99 | |||
| 0.25–0.85 | 7.1 | 1.2 × 10−2 | 0.75 | 1.0 | 2.3 × 10−2 | 0.99 | 2.5 × 10−2 | 0.99 | ||||
| 0.85–2.00 | 7.8 | 4.6 × 10−3 | 0.74 | 1.2 | 2.0 × 10−2 | 0.99 | 3.6 × 10−2 | 0.99 | ||||
| 0.106–0.25 | SA = 10 | 8.4 | 6.3 × 10−3 | 0.87 | 1.2 | 2.8 × 10−2 | 0.99 | 5.3 × 10−2 | 0.99 | |||
| 0.25–0.85 | 8.3 | 1.3 × 10−2 | 0.73 | 0.95 | 7.6 × 10−2 | 0.99 | 4.8 × 10−2 | 0.99 | ||||
| 0.85–2.00 | 9.0 | 1.0 × 10−2 | 0.78 | 1.3 | 2.3 × 10−2 | 0.99 | 9.0 × 10−2 | 0.99 | ||||
| Sands | 0.18–2.00 | 0 | 3.3 | 1.5 × 10−3 | 0.99 | 1.5 | 2.0 × 10−2 | 0.99 | 4.9 × 10−3 | 0.99 | Soybean oil * | This Study |
| 0.30–2.00 | 5.2 | 2.1 × 10−3 | 0.97 | 1.7 | 5.6 × 10−3 | 0.99 | 1.1 × 10−2 | 0.99 | ||||
| 0.18–2.00 | OA = 2 | 4.1 | 1.7 × 10−3 | 0.96 | 1.8 | 1.8 × 10−5 | 0.99 | 6.8 × 10−3 | 0.98 | |||
| 0.30–2.00 | OA = 5 | 4.7 | 1.8 × 10−3 | 0.98 | 2.0 | 3.3 × 10−5 | 0.99 | 8.7 × 10−3 | 0.99 | |||
| 0.18–2.00 | OA = 2 | 5.4 | 2.1 × 10−3 | 0.96 | 2.8 | 3.7 × 10−7 | 0.99 | 1.1 × 10−2 | 0.99 | |||
| 0.30–2.00 | OA = 5 | 3.9 | 1.6 × 10−3 | 0.98 | 2.2 | 5.9 × 10−6 | 0.99 | 6.3 × 10−3 | 0.99 | |||
| 0.18–2.00 | OA = 2 | 5.3 | 2.1 × 10−3 | 0.96 | 2.1 | 1.2 × 10−5 | 0.99 | 1.1 × 10−2 | 0.99 | |||
| 0.30–2.00 | OA = 5 | 5.4 | 2.2 × 10−3 | 0.96 | 1.4 | 6.7 × 10−4 | 0.99 | 1.1 × 10−2 | 0.99 | |||
| 0.18–2.00 | SA = 2 | 3.6 | 1.5 × 10−3 | 0.98 | 1.4 | 4.0 × 10−4 | 0.99 | 5.6 × 10−3 | 0.99 | |||
| 0.30–2.00 | SA = 5 | 4.0 | 1.7 × 10−3 | 0.99 | 1.3 | 1.4 × 10−3 | 0.98 | 6.7 × 10−3 | 0.99 | |||
| Graphite powders | <0.03 | Activated carbons | 25 | 3.9 × 10−3 | 0.9 | 0.82 | 0.16 | 0.77 | – | – | Diesel oil * | Huang et al. (2018) [62] |
| Activated carbons | 0.5–2.0 | 0 | 16–86 | 7.0 × 10−3 −1.0 × 10−2 | 0.94–0.99 | 0.18–0.5 | 0.4–5.0 | 0.93–0.99 | – | – | Vegetable oil | Gong et al. (2007) [34] |
| Wakame | – | Biochar /Ni | 8.65–116.5 | 0.113–0.481 | 0.964–0.999 | 0.03–0.1 | 3.45–7.21 | 0.806–0.965 | – | – | Diesel oil | Jing et al. (2022) [63] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Matsuno, A.; Kawamoto, K. Assessment of Dispersed Oil Sorption in Oily Wastewater onto Hydrophobized/Oleophilized Autoclaved Aerated Concrete (AAC) Grains. Environments 2023, 10, 92. https://doi.org/10.3390/environments10060092
Matsuno A, Kawamoto K. Assessment of Dispersed Oil Sorption in Oily Wastewater onto Hydrophobized/Oleophilized Autoclaved Aerated Concrete (AAC) Grains. Environments. 2023; 10(6):92. https://doi.org/10.3390/environments10060092
Chicago/Turabian StyleMatsuno, Akihiro, and Ken Kawamoto. 2023. "Assessment of Dispersed Oil Sorption in Oily Wastewater onto Hydrophobized/Oleophilized Autoclaved Aerated Concrete (AAC) Grains" Environments 10, no. 6: 92. https://doi.org/10.3390/environments10060092
APA StyleMatsuno, A., & Kawamoto, K. (2023). Assessment of Dispersed Oil Sorption in Oily Wastewater onto Hydrophobized/Oleophilized Autoclaved Aerated Concrete (AAC) Grains. Environments, 10(6), 92. https://doi.org/10.3390/environments10060092







