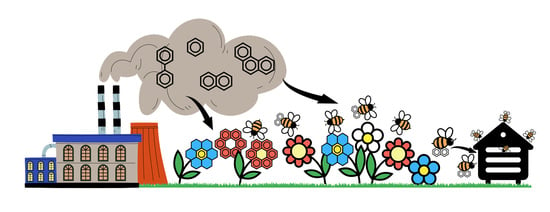
Foraging Honeybees (Apis mellifera ligustica) as Biocenosis Monitors of Pollution in Areas Affected by Cement Industry Emissions
Abstract
1. Introduction
2. Materials and Methods
2.1. Sampling Sites
2.2. Honeybee Samples
2.3. Analytical Procedures (Sample Extraction, Cleanup, and Analysis)
3. Results and Discussion
3.1. Water Content of Foraging Honeybee Samples
3.2. Non-Polar Aliphatic Fraction
3.3. Polycyclic Aromatic Compound Fraction
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Svoboda, J. Industrial poisoning of bees by arsenic (Prumyslove otravy vcel arsenem). Ved. Pract. Vyzk. Ustavu Vcelarskeho CSAZV 1961, 2, 55–60. [Google Scholar]
- Svoboda, J. Teneur en strontium 90 dans les abeilles et dans leurs produits. Bull. Apic. 1962, 5, 101–103. [Google Scholar]
- Bromenschenk, J.J.; Carlson, S.R.; Simpson, J.C.; Thomas, J.M. Pollution Monitoring of Puget Sound with Honey Bees. Science 1985, 227, 532–634. [Google Scholar] [CrossRef] [PubMed]
- Crane, E. Bees, honey and pollen as indicators of metals in the environment. Bee World 1984, 55, 47–49. [Google Scholar] [CrossRef]
- Cunningham, M.M.; Tran, L.; McKee, C.G.; Polo, R.O.; Newman, T.; Lansing, L.; Griffiths, J.S.; Bilodeau, G.J.; Rott, M.; Guarna, M.M. Honeybees as biomonitors of environmental contaminants, pathogens, and climate change. Ecol. Indic. 2022, 134, 108457. [Google Scholar] [CrossRef]
- Di Fiore, C.; Nuzzo, A.; Torino, V.; De Cristofaro, A.; Notardonato, I.; Passarella, S.; Di Giorgi, S.; Avino, P. Honeybees as bioindicators of heavy metal pollution in urban and rural areas in the South of Italy. Atmosphere 2022, 13, 624. [Google Scholar] [CrossRef]
- Perugini, M.; Manera, M.; Grotta, L.; Abete, M.C.; Tarasco, R.; Amorena, M. Heavy metal (Hg, Cr, Cd and Pb) contamination in urban areas and wildlife reserves: Honeybees as bioindicators. Biol. Trace Elem. Res. 2011, 140, 170–176. [Google Scholar] [CrossRef]
- Zugravu, C.A.; Parvu, M.; Patrascu, D.; Stoian, A. Correlations between lead and cadmium pollution of honey and environmental heavy metal presence in two Romanian counties. Bull. UASVM Agric. 2009, 66, 230–233. [Google Scholar] [CrossRef]
- Satta, A.; Verdinelli, M.; Ruiu, L.; Buffa, F.; Salis, S.; Sassu, A.; Floris, I. Combination of beehive matrices analysis and ant biodiversity to study heavy metal pollution impact in a postmining area (Sardinia, Italy). Environ. Sci. Pollut. Res. 2012, 19, 3977–3988. [Google Scholar] [CrossRef]
- Gutierrez, M.; Molero, R.; Gaju, M.; Van der Steen, J.; Porrini, C.; Ruiz, J.A. Assessment of heavy metal pollution in Cordoba (Spain) by biomonitoring foraging honeybee. Environ. Monit. Assess. 2015, 187, 651–665. [Google Scholar] [CrossRef]
- Van der Steen, J.J.M.; Cornelissen, B.; Blacquiere, T.; Pijnenburg, J.E.M.L.; Severijnen, M. Think regionally, act locally: Metals in honeybee workers in the Netherlands surveillance study 2008. Environ. Monit. Assess. 2016, 188, 463. [Google Scholar] [CrossRef] [PubMed]
- Tautz, J. The Buzz about Bees: Biology of a Superorganism, 1st ed.; Springer Science & Business Media: Berlin, Germany, 2008. [Google Scholar]
- Zaric, N.M.; Deljanin, I.; Ilijević, K.; Stanisavljević, L.; Ristić, M.; Gržetić, I. Assessment of spatial and temporal variations in trace element concentrations using honeybees (Apis mellifera) as bioindicators. Peer J. 2018, 6, e5197. [Google Scholar] [CrossRef] [PubMed]
- Hladun, K.R.; Parker, D.R.; Trumble, J.T. Cadmium, Copper, and lead accumulation and bioconcentration in the vegetative and reproductive organs of raphanus sativus: Implications for plant performance and pollination. J. Chem. Ecol. 2015, 41, 386–395. [Google Scholar] [CrossRef] [PubMed]
- Gilbert, M.D.; Lisk, D.J. Honey as an environmental indicator of radionuclide contamination. Bull. Environ. Contam. Toxicol. 1978, 19, 32–34. [Google Scholar] [CrossRef] [PubMed]
- Fresquez, P.R.; Armstrong, D.R.; Pratt, L.H. Radionuclides in bees and honey within and around Los Alamos National Laboratory. J. Environ. Sci. Health Part A 1997, 32, 1309–1323. [Google Scholar] [CrossRef]
- Porrini, C.; Sabatini, A.G.; Girotti, S.; Ghini, S.; Medrzycki, P.; Grillenzoni, F.; Bortolotti, L.; Gattavecchia, E.; Celli, G. Honey bees and bee products as monitors of the environmental contamination. Apiacta 2003, 38, 63–70. [Google Scholar]
- Borowska, M.H.; Kapala, J.; Puścion-Jakubik, A.; Horembala, J.; Markiewicz-Źukowska, R. Radioactivity of honeys from Poland after the Fukushima accident. Bull. Environ. Contam. Toxicol. 2013, 91, 489–492. [Google Scholar] [CrossRef]
- Meli, M.A.; Desideri, D.; Roselli, C.; Feduzi, L.; Benedetti, C. Radioactivity in honey of the central Italy. Food Chem. 2016, 201, 349–355. [Google Scholar] [CrossRef]
- Pareja, L.; Colazzo, M.; Pérez-Parada, A.; Niell, S.; Carrasco-Letelier, L.; Besil, N.; Cesio, M.V.; Heinzen, H. Detection of pesticides in active and depopulated beehives in Uruguay. Int. J. Environ. Res. Public Health 2011, 8, 3844–3858. [Google Scholar] [CrossRef]
- Hungerford, N.L.; Fletcher, M.T.; Tsai, H.H.; Hnatko, D.; Swann, L.J.; Kelly, C.L.; Anuj, S.R.; Tinggi, U.; Webber, D.C.; Were, S.T.; et al. Occurrence of environmental contaminants (pesticides, herbicides, PAHs) in Australian/Queensland Apis mellifera honey. Food Addit. Contam. Part B Surveill. 2021, 14, 193–205. [Google Scholar] [CrossRef]
- Balayiannis, G.; Balayiannis, P. Bee honey as an environmental bioindicator of pesticides occurrence in six agricultural areas of Greece. Arch. Environ. Contam. Toxicol. 2008, 55, 462–470. [Google Scholar] [CrossRef] [PubMed]
- Chauzat, M.P.; Martel, A.C.; Cougoule, N.; Porta, P.; Lachaize, J.; Zeggane, S.; Aubert, M.; Carpentier, P.; Faucon, J.P. An assessment of honeybee colonymatrices, Apis mellifera (Hymenoptera: Apidae) to monitor pesticide presence incontinental France. Environ. Toxicol. Chem. 2011, 30, 103–111. [Google Scholar] [CrossRef] [PubMed]
- Balsebre, A.; Báez, M.E.; Martínez, J.; Fuentes, E. Matrix solid-phase dispersion associated to gas chromatography for the assessment in honey bee of a group of pesticides of concern in the apicultural field. J. Chromatogr. A 2018, 1567, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Perugini, M.; Di Serafino, G.; Giacomelli, A.; Medrzyck, P.; Sabatini, A.G.; Persano Oddo, L.; Marinelli, E.; Amorena, M. Monitoring of polycyclic aromatic hydrocarbons in bees (Apis mellifera) and honey in urban areas and wildlife reserves. J. Agric. Food Chem. 2009, 57, 7440–7444. [Google Scholar] [CrossRef]
- Grenier, E.; Giovenazzo, P.; Julien, C.; Goupil-Sormany, I. Honeybeesas a biomonitoring species to assess environmental airborne pollution in different socioeconomic city districts. Environ. Monit. Assess. 2021, 193, 740. [Google Scholar] [CrossRef] [PubMed]
- Lambert, O.; Veyrand, B.; Durand, S.; Marchand, P.; Le Bizec, B.; Piroux, M.; Puyo, S.; Thorin, C.; Delbac, F.; Pouliquen, H. Polycyclic aromatic hydrocarbons: Bees, honey and pollen as sentinels for environmental chemical contaminants. Chemosphere 2012, 86, 98–104. [Google Scholar] [CrossRef]
- Kargar, N.; Matin, G.; Matin, A.A.; Buyukisik, H.B. Biomonitoring, status and source risk assessment of polycyclic aromatic hydrocarbons (PAHs) using honeybees, pine tree leaves, and propolis. Chemosphere 2017, 186, 140–150. [Google Scholar] [CrossRef] [PubMed]
- Bargańska, Z.; Ślebioda, M.; Namieśnik, J. Honey bees and their products-bioindicators of environmental contamination. Crit. Rev. Environ. Sci. Technol. 2016, 46, 235–248. [Google Scholar] [CrossRef]
- Simon-Delso, N.; San Martin, G.; Bruneau, E.; Delcourt, C.; Hautier, L. The challenges of predicting pesticide exposure of honey bees at landscape level. Sci. Rep. 2017, 7, 3801. [Google Scholar] [CrossRef]
- Stogiannidis, E.; Laane, R. Source characterization of Polycyclic Aromatic Hydrocarbons by using their molecular indices: An overview of Possibilities. In Reviews of Environmental Contamination and Toxicology; Whitacre, D., Ed.; Springer: Cham, Switzerland, 2015; Volume 234, pp. 49–133. [Google Scholar] [CrossRef]
- Mishra, U.C.; Sarsaiya, S.; Gupta, A. A systematic review on the impact of cement industries on the natural environment. Environ. Sci. Pollut. Res. 2022, 29, 18440–18451. [Google Scholar] [CrossRef]
- Palmieri, N.; Panfili, M.; Fioretto, V. Available online: https://www.ilpianetanaturale.org/wp-content/uploads/2018/08/Unico-Api-Gubbio-2004-Protect.pdf (accessed on 6 August 2018).
- De Felip, E.; Miniero, R. AnalyticaI procedures adopted to determine microcontaminants in lagoon sediments. In Rapporti ISTISAN 99/28; Istituto Superiore di Sanità: Roma, Italy, 1999; ISSN 1123-3117. [Google Scholar]
- Abas, M.R.B.; Simoneit, B.R.T. Composition of extractable organic matter of air particles from Malaysia: Initial study. Atmos. Environ. 1996, 30, 2779–2793. [Google Scholar] [CrossRef]
- Ladji, R.; Yassaa, N.; Balducci, C.; Cecinato, A. Organic components of Algerian desert dusts. Chemosphere 2010, 81, 925–931. [Google Scholar] [CrossRef]
- Simoneit, B.R.T. Organic matter of the troposphere V. Application of molecular marker analysis to biogenic emissions into the troposphere for source reconciliations. J. Atmos. Chem. 1989, 8, 251–275. [Google Scholar] [CrossRef]
- Moneti, G.; Dani, F.R.; Pieraccini, G.; Turillazzi, S. Solid-phase Microextraction of Insect Epicuticular Hydrocarbons for Gas Chromatographic/Mass Spectrometric Analysis. Rapid Commun. Mass Spectrom. 1997, 11, 857. [Google Scholar] [CrossRef]
- Sledge, M.F.; Moneti, G.; Pieraccini, G.; Turillazzi, S. Use of solid-phase microextraction in the investigation of chemical communication in social wasps. J. Chromatogr. A 2000, 873, 73–77. [Google Scholar] [CrossRef] [PubMed]
- Buckner, J.S.; Pitts-Singer, T.L.; Guédot, C.; Hagen, M.M.; Fatland, C.L.; Kemp, W.P. Cuticular lipids of female solitary bees, Osmia lignaria Say and Megachile rotundata (F.) (Hymenoptera: Megachilidae). Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2009, 153, 200–205. [Google Scholar] [CrossRef] [PubMed]
- Castellani, F. Fast GC-MS Technique for the Determination of PAHs on Environmental Matrices; 1st Level; University of Perugia: Perugia, Italy, 2016. [Google Scholar]
- INERIS. Available online: www.ineris.fr/sites/ineris.fr/files/contribution/Documents/HAP_4.pdf (accessed on 10 December 2022).
- Colombo, J.C.; Pelletier, C.H.; Brochu, A.; Khalil, M.; Catoggio, J.A. Determination of hydrocarbons sources using n-alkanes and polyaromatic hydrocarbons distribution indexes. Case study: Rio de La Plata Estuary, Argentina. Environ. Sci. Technol. 1989, 23, 888–894. [Google Scholar] [CrossRef]
- Fernández, P.; Grimalt, J.O. On the Global Distribution of Persistent Organic Pollutants. Chimia 2003, 57, 514–521. [Google Scholar] [CrossRef]
- Daly, G.L.; Wania, F. Organic Contaminants in Mountains. Environ. Sci. Technol. 2005, 39, 385–398. [Google Scholar] [CrossRef]
- Sportsøpl, S.; Gjøps, N.; Lichtenthaler, R.G.; Gustavsen, K.O.; Urdal, K.; Oreld, F.; Skei, J. Source Identification of Aromatic Hydrocarbons in Sediments Using GC/MS. Environ. Sci. Technol. 1983, 17, 282–286. [Google Scholar] [CrossRef]
- Tolosa, I.; Bayona, J.M.; Albaigés, A.J. Aliphatic and Polycyclic Aromatic Hydrocarbons and Sulfur/Oxygen Derivatives in Northwestern Mediterranean Sediments: Spatial and Temporal Variability, Fluxes, and Budgets. Environ. Sci. Technol. 1996, 30, 2495–2503. [Google Scholar] [CrossRef]
- Andersson, J.T.; Hegazi, A.H.; Roberz, B. Polycyclic aromatic sulfur heterocycles as information carriers in environmental studies. Anal. Bioanal. Chem. 2006, 386, 891–905. [Google Scholar] [CrossRef] [PubMed]
- Guillen, M.D.; Iglesias, M.J.; Dominguez, A.; Blanco, C.G. Semi quantitative FTIR Analysis of a Coal Tar Pitch and its Extracts and Residues in Several Organic Solvents. Energy Fuels 1992, 6, 518–525. [Google Scholar] [CrossRef]
- Mössner, S.G.; Wise, S.A. Determination of Polycyclic Aromatic Sulfur Heterocycles in Fossil Fuel-Related Samples. Anal. Chem. 1999, 17, 58–69. [Google Scholar] [CrossRef] [PubMed]
- Karcher, W.; Nelen, A.; Depaus, R.; van Eijk, J.; Glaude, P.; Jacob, J. New results in the detection, identification, and mutagenic testing of heterocyclic polycyclic aromatic hydrocarbons. In Chemical Analysis and Biological Fate: Polynuclear Aromatic Hydrocarbons; Batelle Press: Columbus, OH, USA, 1981; pp. 317–327. [Google Scholar]
- Jacob, J. Sulfur Analogues of Polycyclic Aromatic Hydrocarbons (Thiaarenes), 1st ed.; Cambridge University Press: Cambridge, UK, 1990; 281p. [Google Scholar]
- McFall, T.; Booth, G.M.; Lee, M.L.; Tominaga, Y.; Pratap, R.; Tedjamulia, M.; Castle, R.N. Mutagenic activity of methyl-substituted tri-and tetracyclic aromatic sulfur heterocycles. Mutat. Res./Genet. Toxicol. 1984, 135, 97–103. [Google Scholar] [CrossRef] [PubMed]
- Palmieri, N. Available online: https://www.ilpianetanaturale.org/wp-content/uploads/2018/08/Biomonitoraggio_Ambientale_nel_Comune_di_Gualdo_Cattaneo-anno-2008-HiQu_protect.pdf (accessed on 6 August 2018).
- Neff, J.M.; Stout, S.A.; Gunster, D.G. Ecological risk assessment of polycyclic aromatic hydrocarbons in sediments: Identifying sources and ecological hazard. Integr. Environ. Assess. Manag. 2005, 1, 22–33. [Google Scholar] [CrossRef] [PubMed]
- Gogou, A.; Apostolaki, M.; Stephanou, E.G. Determination of organic molrcular markers in marine aerosols and sediments: One-step flash chromatography compound class fractionaction and capillary gas chromathographic analysis. J. Chromathogr. A 1998, 799, 215–231. [Google Scholar] [CrossRef]
- Baumard, P.; Budzinski, H.; Garrigues, P. Polycyclic Aromatic Hydrocarbons in sediments and mussels of the western Mediterranean Sea. Environ. Toxicol. Chem. 1998, 17, 765–776. [Google Scholar] [CrossRef]
- Oliveira, C.; Martins, N.; Tavares, J.; Pio, C.; Cerqueira, M.; Matos, M.; Silva, H.; Oliveira, C.; Camões, F. Size distribution of polycyclic aromatic hydrocarbons in a roadway tunnel in Lisbon, Portugal. Chemosphere 2011, 83, 1588–1596. [Google Scholar] [CrossRef]
- Dallarosa, J.B.; Teixeira, E.C.; Pires, M.; Fachel, J. Study of the profile of polycyclic aromatic hydrocarbons in atmospheric particles (PM10) using multivariate methods. Atmos. Environ. 2005, 39, 6587–6596. [Google Scholar] [CrossRef]
- Tobiszewski, M.; Namieśnik, J. PAH diagnostic ratios for the identification of pollution emission sources. Environ. Pollut. 2012, 162, 110–119. [Google Scholar] [CrossRef] [PubMed]
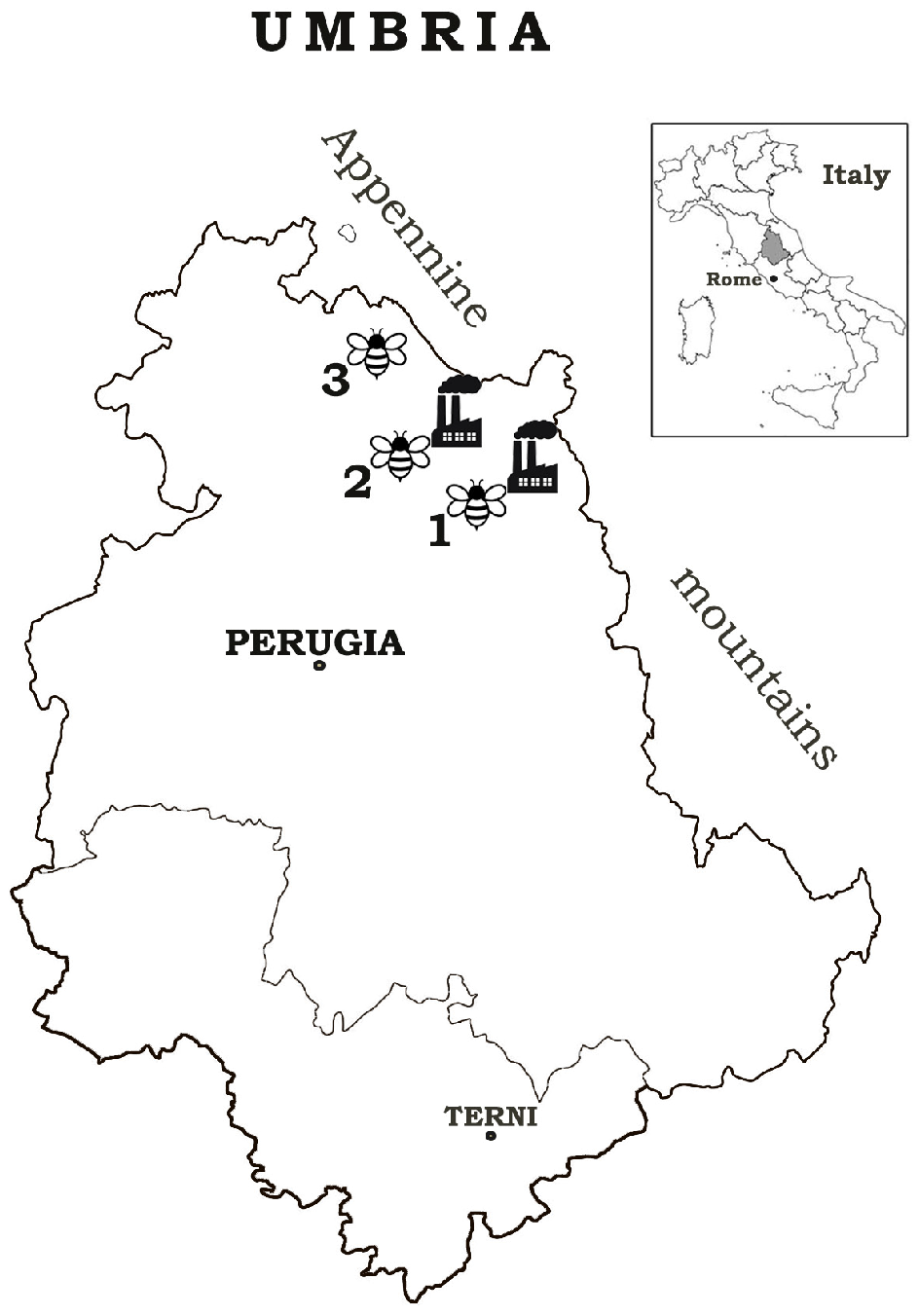
 , affected sites (1, 2), and pristine area (3) position and beehive locations
, affected sites (1, 2), and pristine area (3) position and beehive locations  .
.
 , affected sites (1, 2), and pristine area (3) position and beehive locations
, affected sites (1, 2), and pristine area (3) position and beehive locations  .
.

| Homolog | Monitoring Site | ||||||
|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | |||||
| ng/g d.w. a | % | ng/g d.w. a | % | ng/g d.w. a | % | ||
| nC14 | Tetradecane | 0.00 b | 0.00 b | 2.9 | 0.00 b | 3.8 | 0.00 b |
| nC15 | Pentadecane | 3.8 | 0.00 b | 5.0 | 0.00 b | 5.5 | 0.00 b |
| nC16 | Hexadecane | 5.9 | 0.01 | 6.3 | 0.01 | 12.4 | 0.01 |
| nC17 | Heptadecane | 29.5 | 0.03 | 39.5 | 0.03 | 43.8 | 0.03 |
| nC18 | Octadecane | 19.6 | 0.02 | 44.5 | 0.04 | 15.9 | 0.01 |
| nC19 | Nonadecane | 52.4 | 0.06 | 113.7 | 0.10 | 205.8 | 0.14 |
| nC20 | Eicosane | 23.9 | 0.03 | 28.2 | 0.02 | 26.3 | 0.02 |
| nC21 | Heneicosane | 548.5 | 0.59 | 662.2 | 0.53 | 573.2 | 0.40 |
| nC22 | Docosane | 369.9 | 0.43 | 402.2 | 0.36 | 399.9 | 0.29 |
| nC23 | Tricosane | 24,937.1 | 27.01 | 37,161.9 | 32.95 | 38,767.9 | 27.26 |
| nC24 | Tetracosane | 8611.0 | 0.93 | 1166.3 | 1.03 | 1215.5 | 0.85 |
| nC25 | Pentacosane | 29,576.5 | 32.07 | 45,302.3 | 40.17 | 4105.2 | 38.04 |
| nC26 | Hexacosane | 780.4 | 0.85 | 885.0 | 0.78 | 1039.9 | 0.73 |
| nC27 | Heptacosane | 14,193.2 | 15.39 | 12,732.3 | 11.29 | 23,306.5 | 16.39 |
| nC28 | Octacosane | 489.0 | 0.53 | 371.3 | 0.34 | 618.4 | 0.43 |
| nC29 | Nonacosane | 5277.4 | 10.62 | 7113.6 | 6.31 | 10,959.3 | 7.70 |
| nC30 | Triacontane | 353.1 | 0.38 | 385.6 | 0.30 | 446.7 | 0.31 |
| nC31 | Hentriacontane | 9729.5 | 10.55 | 5353.0 | 4.75 | 8977.0 | 6.30 |
| nC32 | Dotriacontane | 175.6 | 0.19 | 124.2 | 0.11 | 153.2 | 0.11 |
| nC33 | Tritriacontane | 256.1 | 0.28 | 916.5 | 0.81 | 1362.4 | 0.96 |
| Total ng/g | 92,209 | 112,777 | 142,234 | ||||
| CPI25 | 29.6 | 33.0 | 36.1 | ||||
| Peak nr | Compound | Rt | Peak nr | Compound | Rt |
|---|---|---|---|---|---|
| 1 | Naphthalene | 5.080 | 76 | x,y-di-methyl Phen/Anthr | 15.386 |
| 2 | 2-methyl-Naphthalene | 6.316 | 77–78 | x,y-di-methyl Dibenzotiophene | 15.539–15.554 |
| 3 | 1-methyl-Naphthalene | 6.518 | 79 | x,y-di-methyl Phen/Anthr | 15.658 |
| 4 | 1,1′-Biphenyl | 7.318 | 80–81 | x,y-di-methyl Dibenzotiophene | 15.673–15.690 |
| 5 | Biphenyl-3-ol | 7.535 | 82–83 | x,y-di-methyl Phen/Anthr | 15.758–15.842 |
| 6–9 | x,y di-methyl-Naphthalene | 7.651–8.064 | 84 | tetra-methyl-s-Indacene-1,7-dione, tetra-hydro- | 15.975 |
| 10 | Acenaphthene | 8.212 | 85 | x,y-di-methyl Phen/Anthr | 16.009 |
| 11 | 2-acetossi-2-metossi-Biphenyl | 8.462 | 86 | Biphenyl-2-ol-5, 1,1-dimethyletil- | 16.093 |
| 12 | Acenaphthylene | 8.829 | 87–89 | x,y-dimethyl Phen/Anthr | 16.126–16.310 |
| 13 | x,y,z tri-methyl Naphthalene | 8.829 | 90 | Fluoranthene | 16.545 |
| 14 | x,y,z tri-methyl Naphthalene | 8.927 | 91 | x,y-di-methyl 9,10-Anthracenedione | 16.562 |
| 15 | x,y,z tri-methyl Benzo(b) thiophene | 8.993 | 92 | 2-Phenylmethyl Naphthalene | 16.819 |
| 16 | Dibenzofuran | 9.061 | 93–95 | x,y,z-tri-methyl Phen/Anthr | 16.869–17.022 |
| 17–21 | x,y,z tri-methyl Naphthalene | 9.061–9.542 | 96 | x,y-di-methyl Phenanthrene, diidro- | 17.022 |
| 22 | x-y di-methyl Biphenyl | 9.774 | 97 | x,y,z-tri-methyl Phen/Anthr | 17.105 |
| 23 | Fluorene | 9.873 | 98 | x,y-di-methyl 9,10-Anthracenedione | 17.139 |
| 25 | x methyl Biphenyl | 10.041 | 99 | x,y,z-tri-methyl Phen/Anthr | 17.190 |
| 26 | x,y di-methyl diphenyl Methane | 10.124 | 100 | x,y,z-tri-methyl Phen/Anthr | 17.256 |
| 27 | x,y,z,t tetra-methyl Naphthalene | 10.190 | 101 | Pyrene | 17.305 |
| 28 | x,y di-methyl Biphenyl | 10.307 | 102–106 | x,y,z-tri-methyl Phen/Anthr | 17.322–17.657 |
| 29 | Benzophenone | 10.374 | 107–110 | x,y,z,t-tetra-methyl Phen/Anthr | 17.740–18.090 |
| 30–31 | x,y di-ethyl Biphenyl, | 10.524–10.557 | 111 | Methylethyl Benzo(a)acridine | 18.225 |
| 32 | x,y,z,t tetra-methyl Naphthalene | 10.640 | 112–113 | x-methyl Fluoranthene/Pyrene | 18.192–18.275 |
| 33 | diisopropyl Naphthalene | 10.756 | 114 | 3-methyl Phenanthro(4,5-bcd) tiophene | 18.357 |
| 34 | Nitrophenol, ditert-buthyl- | 10.822 | 115–116 | x-methyl Fluoranthene/Pyrene | 18.492–18.610 |
| 35 | Biphenyl, di-ethyl- | 10.873 | 117 | Triphenyl methane | 18.696 |
| 36 | Nitrophenol, diter-buthyl- | 10.956 | 118 | 3-methyl Phenantro(4,5-bcd) tiophene | 18.749 |
| 37–38 | Biphenyl, di-ethyl- | 11.005–11.256 | 119–131 | x-methyl Fluoranthene/Pyrene | 18.850–20.861 |
| 39–41 | diisopropyl Naphthalene | 11.407–11.541 | 132 | Benzo(a)nafto(2,1-d)tiophene | 20.911 |
| 42 | Ethane-1,1-bis(p-ethylphenyl) | 11.608 | 133 | x,y-dimethyl Fluoranthene/Pyrene | 20.945 |
| 43 | 2-vinyl-2,3-di-hydro Nahthofurane | 11.675 | 134 | Benzo(ghi)fluoranthene | 20.978 |
| 44 | Benzene-1,1-methylene bis(4-methyl) | 11.692 | 135 | Benzo(a)naphtho(1,2-d)tiophene | 21.213 |
| 45 | 1,3 Pentadiene, 1,1-di-phenyl | 11.726 | 136 | Benzo(a)anthracene | 21.831 |
| 46 | 1,2-dimethyl Naphtho(2,1-b) furane | 11.825 | 137 | Crisene+Trifenilene | 21.984 |
| 47 | Biphenyl, di-ethyl- | 11.859 | 138 | 6-methyl Benzo(b)nafto(2,3-d) tiophene | 22.198 |
| 48 | x,y,z,t-tetramethyl Biphenyl | 11.875 | 139 | 8-methyl Benzo(b)nafto(2,3-d)tiophene | 22.533 |
| 49 | Benzaldeide-3,5-di-tert-buthyl -4-hydrossi | 11.926 | 140–141 | 1-H-Indene-2,3-diidro-trimethylphenil | 22.600–22.746 |
| 50 | 4-methyl Acridone | 11.976 | 142–147 | x-methyl Benzo(a)anthr/Crisene | 23.297–23.863 |
| 51 | Anthracene, tetrahydro-9-propyl | 11.976 | 148–152 | x,y-di-methyl Benzo(b)naphtho (2,3-d)tiophene | 23.647–24.346 |
| 52 | 10-methyl Acridone | 12.075 | 153–155 | Benzo(b,j,k)fluoranthene | 25.740–26.012 |
| 53 | Biphenyl, di-ethyl- | 12.125 | 156 | Benzo(a)fluoranthene | 26.162 |
| 54 | Dibenzotiophene | 12.175 | 157 | Benzo(e)pyrene | 26.162 |
| 55 | x,y,z-trimethyl-3-phenyl di-hydro indene | 12.209 | 158 | Benzo(a)pyrene | 26.803 |
| 56 | Phenanthrene | 12.557 | 159 | Perilene | 27.063 |
| 57 | Diphenoxyethane | 12.641 | 160 | 5,8-di-methyl Benzo(c)phenanthrene | 27.342 |
| 58 | Anthracene | 12.723 | 161 | methyl Benzo[2,3]phenanthro (4,5-bcd)tiophene | 27.392 |
| 59 | Fluorenone | 12.756 | 162 | 3-methyl Benzo(j)aceantrilene | 27.880 |
| 60 | 1-Indene-2,3-di-hydro-1,1,3- trimethyl-3-phenyl | 12.906 | 163–166 | x-methyl Perilene/Benzo [1,2-b:4,3-b] ditiophene-1-Phenyl | 27.998–28.387 |
| 61 | 9,10-Anthracenedione | 13.635 | 167 | Indenofluoranthene | 29.763 |
| 62 | x-methyl Phen/Anthr | 13.788 | 168 | Indeno(1,2,3-cd)pyrene | 31.314 |
| 63 | 9,10-Anthracenedione, 2-ethyl | 13.804 | 169–170 | Benzo(ac+ah)anthracene | 31.506 |
| 64 | 9-Amminofluorenone | 13.957 | 171 | Benzo(b)crisene | 31.906 |
| 66 | 2-methyl Phenanthrene | 14.207 | 173 | Dibenzo(a,l)pyrene | 34.497 |
| 67 | 1-methyl Phenanthrene | 14.360 | 174 | Dibenzo(a,e)pyrene | 35.353 |
| 68 | x-methyl Phen/Anthr | 14.462 | 175 | Coronene | 39.390 |
| 69 | Dimethyl carbazole | 14.513 | 176 | Dibenzo(a,h)pyrene | 39.670 |
| 70 | 9-methyl Anthracene | 14.548 | 177 | Dibenzo(a,i)pyrene | 43.040 |
| 71–75 | x,y-dimethyl dibenzotiophene | 14.785–15.335 |
| Monitoring Site | ||||
|---|---|---|---|---|
| 1 | 2 | 3 | ||
| Isomer nr | ng/g d.w. a | |||
| trimethyl Benzothiophene | 1 | 0.05 | <0.03 | <0.03 |
| Dibenzothiophene | 1 | 4.36 | 1.15 | 1.55 |
| dimethyl Dibenzothiphene | 9 | 8.24 | 6.01 | <0.03 |
| Benzonaphtho(2,1-d)thiophene | 1 | 3.55 | <0.04 | <0.04 |
| Benzonaphtho(1,2-d)thiophene | 1 | 0.73 | <0.04 | <0.04 |
| methyl Benzo(b)naphthothiophene | 2 | 8.98 | <0.05 | <0.05 |
| dimethyl Benzo(b)naphthothiophene | 5 | 5.15 | <0.05 | <0.05 |
| methyl Phenanthrothiophene | 1 | 2.46 | <0.05 | <0.05 |
| methyl Benzo(b)phenanthrothiophene | 2 | 2.11 | <0.05 | <0.05 |
| Total PAHs ng/g d.w. a | 35.63 | 7.16 | 1.55 | |
| ng/g w.w. b | 13.50 | 2.72 | 0.41 | |
| Monitoring Site | |||
|---|---|---|---|
| 1 | 2 | 3 | |
| Compound | ng/g d.w. a | ||
| Naphthalene * | 33.63 | 19.67 | 31.97 |
| Acenaphthylene * | 0.81 | 0.74 | 0.98 |
| Acenaphthene * | 1.69 | 2.41 | 2.26 |
| Fluorene * | 40.89 | 107.35 | 92.88 |
| Phenanthrene * | 29.84 | 25.16 | 21.49 |
| Anthracene * | 10.31 | 2.87 | 3.10 |
| Fluoranthene * | 7.03 | 9.58 | 9.10 |
| Pyrene * | 16.65 | 7.75 | 6.13 |
| Benzo(ghi)fluoranthene | 0.61 | <0.04 | 0.70 |
| Benzo(a)anthracene * | NQ | NQ | NQ |
| Chrisene+Triphenylene * | 24.92 | 11.62 | 9.15 |
| Benzo(b+k+j)fluoranthene * | 9.36 | <0.05 | <0.05 |
| Benzo(a) fluoranthene | 1.25 | <0.08 | <0.08 |
| Benzo(e)pyrene | 17.79 | <0.08 | <0.08 |
| Benzo(a)pyrene * | 24.91 | <0.08 | <0.08 |
| Indenofluoranthene | 4.40 | <0.08 | <0.08 |
| Indeno(123-cd)pyrene * | 26.12 | <0.10 | <0.10 |
| Dibenzo(ac+ah)anthracene * | 6.68 | <0.10 | <0.10 |
| Benzo(b)chrysene | 19.93 | <0.10 | <0.10 |
| Benzo(ghi)perylene * | 28.51 | <0.10 | <0.10 |
| Dibenzo(al+ae+ai+ah)pyrene | 8.79 | <0.40 | <0.40 |
| Coronene | 27.67 | <0.10 | <0.10 |
| Total PAHs ng/g d.w. a | 341.77 | 187.14 | 177.67 |
| ng/g w.w. b | 129.95 | 71.15 | 67.50 |
| Monitoring Site | Literature | ||||
|---|---|---|---|---|---|
| Diagnostic Ratio | 1 | 2 | 3 | Pyrolitic | Petrogenic |
| Low/High | 0.5 | 3.9 | 4.4 | <1 | >1 |
| Ph/An | 2.9 | 8.8 | 6.9 | <10 | >15 |
| Fl/Pyr | 0.4 | 1.2 | 1.5 | >1 | <1 |
| Fl/Fl+Pyr | 0.3 | 0.6 | 0.6 | >0.5 | <0.5 |
| An/Ph+An | 0.26 | 0.10 | 0.13 | ||
| IP/IP+BghiP | 0.48 | - | - | ||
| fresh | aged | ||||
| BeP/BaP | 0.71 | - | - | <1 | >1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sebastiani, B.; Mariucci, S.; Palmieri, N. Foraging Honeybees (Apis mellifera ligustica) as Biocenosis Monitors of Pollution in Areas Affected by Cement Industry Emissions. Environments 2023, 10, 34. https://doi.org/10.3390/environments10020034
Sebastiani B, Mariucci S, Palmieri N. Foraging Honeybees (Apis mellifera ligustica) as Biocenosis Monitors of Pollution in Areas Affected by Cement Industry Emissions. Environments. 2023; 10(2):34. https://doi.org/10.3390/environments10020034
Chicago/Turabian StyleSebastiani, Bartolomeo, Sara Mariucci, and Nicola Palmieri. 2023. "Foraging Honeybees (Apis mellifera ligustica) as Biocenosis Monitors of Pollution in Areas Affected by Cement Industry Emissions" Environments 10, no. 2: 34. https://doi.org/10.3390/environments10020034
APA StyleSebastiani, B., Mariucci, S., & Palmieri, N. (2023). Foraging Honeybees (Apis mellifera ligustica) as Biocenosis Monitors of Pollution in Areas Affected by Cement Industry Emissions. Environments, 10(2), 34. https://doi.org/10.3390/environments10020034