Slow-Oscillation Neurofeedback: A Narrative Review on Clinical Efficacy in Pediatric Settings
Abstract
1. Introduction
1.1. Slow Oscillations
1.1.1. Slow Cortical Potentials (SCP)
- Negative shifts, associated with depolarization of the neuronal membrane, increasing neuronal excitability and thereby enabling activation of neural pathways (e.g., to perform behavioral or cognitive tasks).
- Positive shifts, linked to hyperpolarization of the neuronal membrane, that reduce cortical excitability and facilitate inhibitory states or rest (e.g., in Go/No-Go paradigm).
1.1.2. Infra-Low Frequency (ILF)
1.1.3. Infra-Slow Fluctuations (ISF)
1.1.4. Summary and Differences Between Protocols
1.2. Aim of the Review
2. Method
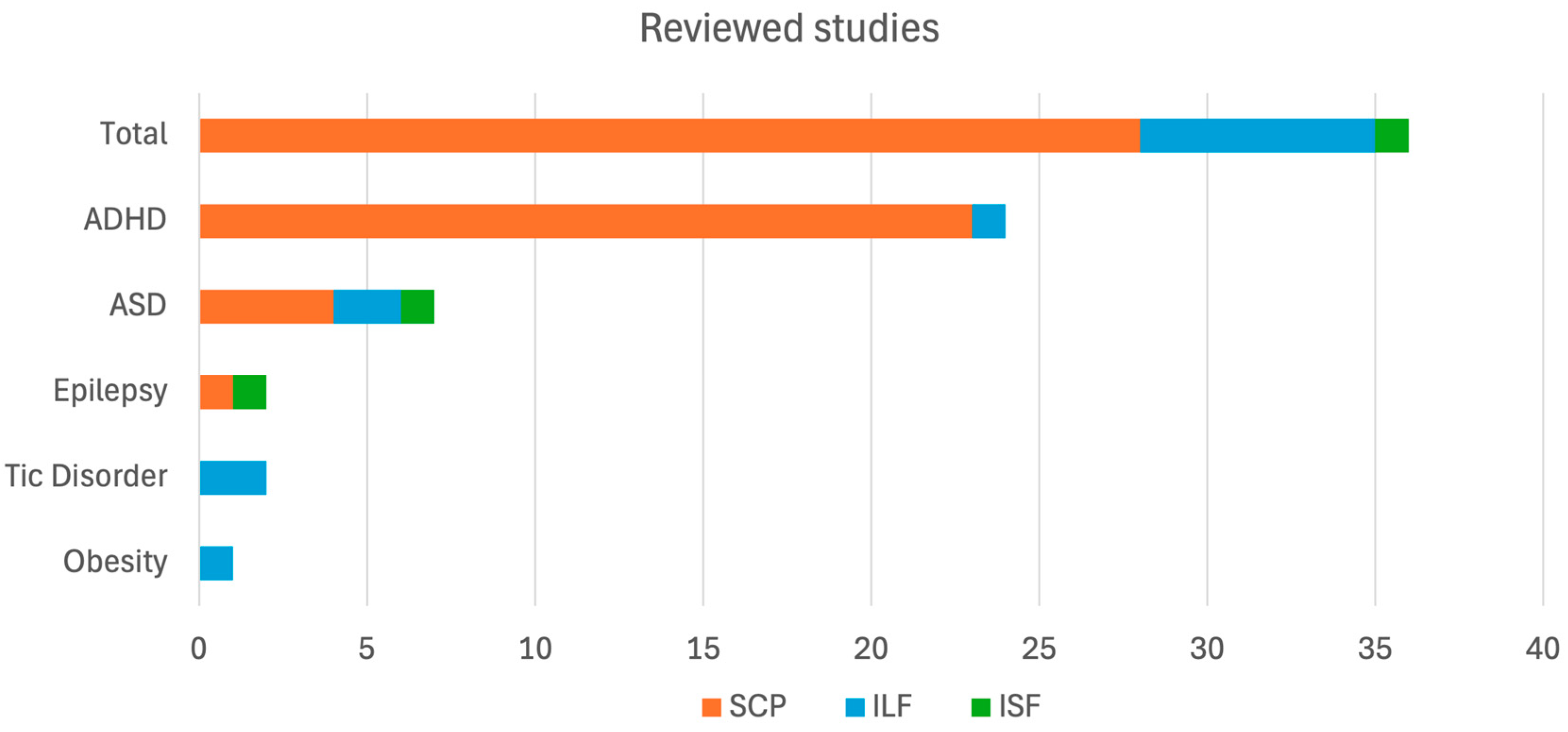
3. Results
3.1. Studies on Attention-Deficit/Hyperactivity Disorder
3.1.1. Efficacy of SCP Neurofeedback and Comparison with Theta/Beta NF Training
3.1.2. Comparison of SCP Neurofeedback with Other Forms of Therapy
3.1.3. Predictors of Neurofeedback Learning
3.2. Studies on Autism Spectrum Disorder
3.2.1. SCP Neurofeedback in Autism Spectrum Disorder
3.2.2. ILF Neurofeedback in Autism Spectrum Disorder
3.3. Studies on Other Disorders
3.3.1. Epilepsy
3.3.2. Studies on Tic Disorders
3.3.3. Studies on Eating Disorders
4. Discussion
4.1. Clinical Insights into Slow-Oscillation Neurofeedback Across Pediatric Disorders
4.1.1. Attention-Deficit/Hyperactivity Disorder
4.1.2. Autism Spectrum Disorder
4.1.3. Other Disorders
4.2. Methodological Limitations
4.3. Future Research Directions
4.4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Aggensteiner, P.-M., Albrecht, B., Strehl, U., Wörz, S., Ruckes, C., Freitag, C. M., Rothenberger, A., Gevensleben, H., Millenet, S., Hohmann, S., Banaschewski, T., Legenbauer, T., Holtmann, M., & Brandeis, D. (2021). Can neurophysiological markers of anticipation and attention predict ADHD severity and neurofeedback outcomes? Biological Psychology, 165, 108169. [Google Scholar] [CrossRef] [PubMed]
- Aggensteiner, P.-M., Brandeis, D., Millenet, S., Hohmann, S., Ruckes, C., Beuth, S., Albrecht, B., Schmitt, G., Schermuly, S., Wörz, S., Gevensleben, H., Freitag, C. M., Banaschewski, T., Rothenberger, A., Strehl, U., & Holtmann, M. (2019). Slow cortical potentials neurofeedback in children with ADHD: Comorbidity, self-regulation and clinical outcomes 6 months after treatment in a multicenter randomized controlled trial. European Child & Adolescent Psychiatry, 28(8), 1087–1095. [Google Scholar] [CrossRef]
- Aladjalova, N. A. (1957). Infra-slow rhythmic oscillations of the steady potential of the cerebral cortex. Nature, 179(4567), 957–959. [Google Scholar] [CrossRef] [PubMed]
- Albrecht, J. S., Bubenzer-Busch, S., Gallien, A., Knospe, E. L., Gaber, T., & Zepf, F. D. (2017). Effects of a structured 20-session slow-cortical-potential-based neurofeedback program on attentional performance in children and adolescents with attention-deficit hyperactivity disorder: Retrospective analysis of an open-label pilot-approach and 6-month-follow-up. Neuropsychiatric Disease and Treatment, 13, 667–683. [Google Scholar] [CrossRef] [PubMed]
- Azza, Y., Grueschow, M., Karlen, W., Seifritz, E., & Kleim, B. (2020). How stress affects sleep and mental health: Nocturnal heart rate increases during prolonged stress and interacts with childhood trauma exposure to predict anxiety. Sleep, 43(6), zsz310. [Google Scholar] [CrossRef]
- Azza, Y., Wilhelm, F. H., Seifritz, E., Junghanns, K., Kleim, B., & Wilhelm, I. (2022). Sleep’s role in updating aversive autobiographical memories. Translational Psychiatry, 12(1), 117. [Google Scholar] [CrossRef] [PubMed]
- Barradas, I., Tschiesner, R., & Peer, A. (2022). Towards a dynamic model for prediction of emotion intensity from peripheral physiological signals. In International conference on human-computer interaction (pp. 18–35). Lecture Notes in Computer Science. Springer Nature Switzerland. [Google Scholar] [CrossRef]
- Barradas, I., Tschiesner, R., & Peer, A. (2024). Emotion-undifferentiated intensity estimation: Dynamical models and physiological insights. In 2024 IEEE 4th international conference on human-machine systems ICHMS. IEEE. [Google Scholar] [CrossRef]
- Barradas, I., Tschiesner, R., & Peer, A. (2025). Dynamic Emotion intensity estimation from physiological signal facilitating interpretation via appraisal theory. PLoS ONE, 20(1), e0315929. [Google Scholar] [CrossRef]
- Baumeister, S., Wolf, I., Hohmann, S., Holz, N., Boecker-Schlier, R., Banaschewski, T., & Brandeis, D. (2019). The impact of successful learning of self-regulation on reward processing in children with ADHD using fMRI. ADHD Attention Deficit and Hyperactivity Disorders, 11, 31–45. [Google Scholar] [CrossRef]
- Baumeister, S., Wolf, I., Holz, N., Boecker-Schlier, R., Adamo, N., Holtmann, M., Ruf, M., Banaschewski, T., Hohmann, S., & Brandeis, D. (2018). Neurofeedback training effects on inhibitory brain activation in ADHD: A matter of learning? Neuroscience, 378, 89–99. [Google Scholar] [CrossRef]
- Bazzana, F., Finzi, S., Di Fini, G., & Veglia, F. (2022). Infra-low frequency neurofeedback: A systematic mixed studies review. Frontiers in Human Neuroscience, 16, 920659. [Google Scholar] [CrossRef] [PubMed]
- Beber, S., Bontempi, G., Miceli, G., & Tettamanti, M. (2024). The neurofunctional correlates of morphosyntactic and thematic impairments in aphasia: A systematic review and meta-analysis. Neuropsychology Review, 35(3), 483–516. [Google Scholar] [CrossRef] [PubMed]
- Beber, S., Capasso, R., Maffei, C., Tettamanti, M., & Miceli, G. (2025). Distinct neural correlates of morphosyntactic and thematic comprehension processes in aphasia. Brain Communications, 7(2), fcaf093. [Google Scholar] [CrossRef]
- Berger, I., Slobodin, O., Aboud, M., Melamed, J., & Cassuto, H. (2013). Maturational delay in ADHD: Evidence from CPT. Frontiers in Human Neuroscience, 7, 691. [Google Scholar] [CrossRef] [PubMed]
- Brunia, C. H. M., & van Boxtel, G. J. M. (2001). Wait and see. International Journal of Psychophysiology, 43(1), 56–75. [Google Scholar] [CrossRef] [PubMed]
- Chirita-Emandi, A., & Puiu, M. (2014). Outcomes of neurofeedback training in childhood obesity management: A pilot study. The Journal of Alternative and Complementary Medicine, 20(11), 831–837. [Google Scholar] [CrossRef]
- Christiansen, H., Reh, V., Schmidt, M. H., & Rief, W. (2014). Slow cortical potential neurofeedback and self-management training in outpatient care for children with ADHD: Study protocol and first preliminary results of a randomized controlled trial. Frontiers in Human Neuroscience, 8, 943. [Google Scholar] [CrossRef]
- Coben, R., Linden, M., & Myers, T. E. (2010). Neurofeedback for autistic spectrum disorder: A review of the literature. Applied Psychophysiology and Biofeedback, 35(1), 83–105. [Google Scholar] [CrossRef] [PubMed]
- Coleman, D. M., Adams, J. B., Anderson, A. L., & Frye, R. E. (2019). Rating of the effectiveness of 26 psychiatric and seizure medications for autism spectrum disorder: Results of a national survey. Journal of Child and Adolescent Psychopharmacology, 29(2), 107–123. [Google Scholar] [CrossRef] [PubMed]
- Davico, C., Secci, I., Vendrametto, V., & Vitiello, B. (2023). Pharmacological treatments in autism spectrum disorder: A narrative review. Journal of Psychopathology, 29, 38–52. [Google Scholar] [CrossRef]
- Doehnert, M., Brandeis, D., Straub, M., Steinhausen, H.-C., & Drechsel, R. (2008). Slow cortical potential neurofeedback in attention deficit hyperactivity disorder: Is there neurophysical evidence for specific effects? Journal of Neural Transmission, 115(10), 1445–1456. [Google Scholar] [CrossRef]
- Drechsler, R., Straub, M., Doehnert, M., Heinrich, H., Steinhausen, H.-C., & Brandeis, D. (2007). Controlled evaluation of a neurofeedback training of slow cortical potentials in children with attention deficit/hyperactivity disorder (ADHD). Behavioral and Brain Functions, 3, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Elbert, T. (1993). Slow cortical potentials reflect the regulation of cortical excitability. In W. C. McCallum, & S. H. Curry (Eds.), Slow potential changes in the human brain (Vol. 254). NATO ASI Series. Springer. [Google Scholar] [CrossRef]
- Elbert, T., Ulrich, R., Rockstroh, B., & Lutzenberger, W. (1991). The processing of temporal intervals reflected by CNV-like brain potentials. Psychophysiology, 28(6), 648–655. [Google Scholar] [CrossRef]
- Esmaeilzadeh Kanafgourabi, S. N., Shabani, M., Mirchi, Z., Aliyari, H., & Mahdavi, P. (2023). The impact of ILF neurofeedback on inhibitory control in high-functioning adolescents with autism spectrum disorder: Preliminary evidence of a randomized controlled trial. Applied Neuropsychology: Child, 14(1), 55–73. [Google Scholar] [CrossRef]
- Gevensleben, H., Albrecht, B., Lütcke, H., Auer, T., Dewiputri, W. I., Schweizer, R., Moll, G., Heinrich, H., & Rothenberger, A. (2014). Neurofeedback of slow cortical potentials: Neural mechanisms and feasibility of a placebo-controlled design in healthy adults. Frontiers in Human Neuroscience, 8, 990. [Google Scholar] [CrossRef] [PubMed]
- Gevensleben, H., Heinrich, H., Moll, G., & Rothenberger, A. (2011). Neurofeedback bei kindern mit ADHS-methodische grundlagen und wissenschaftliche evaluation. Praxis der Kinderpsychologie und Kinderpsychiatrie, 60(8), 666–676. [Google Scholar] [CrossRef] [PubMed]
- Gevensleben, H., Holl, B., Albrecht, B., Schlamp, D., Kratz, O., Studer, P., Wangler, S., Rothenberger, A., Moll, G. H., & Heinrich, H. (2009a). Distinct EEG effects related to neurofeedback training in children with ADHD: A randomized controlled trial. International Journal of Psychophysiology, 74(2), 149–157. [Google Scholar] [CrossRef] [PubMed]
- Gevensleben, H., Holl, B., Albrecht, B., Vogel, C., Schlamp, D., Kratz, O., Studer, P., Rothenberger, A., Moll, G. H., & Heinrich, H. (2009b). Is neurofeedback an efficacious treatment for ADHD? A randomised controlled clinical trial. Journal of Child Psychology and Psychiatry, 50(7), 780–789. [Google Scholar] [CrossRef] [PubMed]
- Gevensleben, H., Moll, G. H., & Heinrich, H. (2010). Neurofeedback-training bei kindern mit aufmerksamkeitsdefizit-/hyperaktivitätsstörung (ADHS): Effekte auf verhaltens- und neurophysiologischer ebene. Zeitschrift für Kinder- und Jugendpsychiatrie und Psychotherapie, 38(6), 409–420. [Google Scholar] [CrossRef]
- Greco, A., Valenza, G., & Scilingo, E. P. (2016). Advances in electrodermal activity processing with applications for mental health. Springer. [Google Scholar] [CrossRef]
- Grin-Yatsenko, V. A., Kara, O., Evdokimov, S. A., Gregory, M., Othmer, S., & Kropotov, J. D. (2020). Infra-low frequency neuro feedback modulates infra-slow oscillations of brain potentials: A controlled study. Journal of Biomedical Engineering and Research, 4, 1–11. [Google Scholar] [CrossRef]
- Grin-Yatsenko, V. A., Ponomarev, V. A., Kara, O., Wandernoth, B., Gregory, M., Ilyukhina, V. A., & Kropotov, J. D. (2018). Effect of infralow frequency neurofeedback on infra-slow EEG fluctuations. In M. Schwartz (Ed.), Biofeedback (pp. 75–87). IntechOpen. [Google Scholar] [CrossRef]
- Hamano, T., Lüders, H. O., Ikeda, A., Collura, T. F., Comair, Y. G., & Shibasaki, H. (1997). The cortical generators of the contingent negative variaton in humans: A study with subdural electrodes. Electroencephalography and Clinical Neurophysiology, 104(3), 257–268. [Google Scholar] [CrossRef]
- Hasslinger, J., Bölte, S., & Jonsson, U. (2022a). Slow cortical potential versus live z-score neurofeedback in children and adolescents with ADHD: A multi-arm pragmatic randomized controlled trial with active and passive comparators. Research on Child and Adolescent Psychopathology, 50(4), 447–462. [Google Scholar] [CrossRef] [PubMed]
- Hasslinger, J., D’Agostini Souto, M., Folkesson Hellstadius, L., & Bölte, S. (2020). Neurofeedback in ADHD: A qualitative study of strategy use in slow cortical potentail training. PLoS ONE, 15(6), e0233343. [Google Scholar] [CrossRef]
- Hasslinger, J., Jonsson, U., & Bölte, S. (2022b). Immediate and sustained effects of neurofeedback and working memory training on cognitive functions in children and adolescents with ADHD: A multi-arm pragmatic randomized controlled trial. Journal of Attention Disorders, 26(11), 1492–1506. [Google Scholar] [CrossRef] [PubMed]
- Heinrich, H., Gevensleben, H., Becker, A., & Rothenberger, A. (2020). Effects of neurofeedback on the dysregulation profile in children with ADHD: SCP NF meets SDQ-DP—A retrospective analysis. Psychological Medicine, 50(2), 258–263. [Google Scholar] [CrossRef] [PubMed]
- Heinrich, H., Gevensleben, H., Freisleder, F. J., Moll, G. H., & Rothenberger, A. (2004). Training of slow cortical potentials in attention-deficit/hyperactivity disorder: Evidence for positive behavioral and neurophysiological effects. Biological Psychiatry, 55(7), 772–775. [Google Scholar] [CrossRef] [PubMed]
- Hiltunen, T., Kantola, J., Abou Elseoud, A., Lepola, P., Suominen, K., Starck, T., Nikkinen, J., Remes, J., Tervonen, O., Palva, S., Kiviniemi, V., & Palva, J. M. (2014). Infra-slow EEG fluctuations are correlated with resting-state network dynamics in fMRI. Journal of Neuroscience, 34(2), 356–362. [Google Scholar] [CrossRef] [PubMed]
- Hinterberger, T., Veit, R., Strehl, U., Trevorrow, T., Erb, M., Kotchoubey, B., Flor, H., & Birbaumer, N. (2003). Brain areas activated in fMRI during self-regulation of slow cortical potentials (SCPs). Experimental Brain Research, 152, 113–122. [Google Scholar] [CrossRef] [PubMed]
- Hinterberger, T., Veit, R., Wilhelm, B., Weiskopf, N., Vatine, J. J., & Birbaumer, N. (2005). Neuronal mechanisms underlying control of a brain-computer interface. European Journal of Neuroscience, 21(11), 3169–3181. [Google Scholar] [CrossRef]
- Infraslow Associates. (2024). Infraslow associates | infraslow neurofeedback training & resources. Infraslow Associates. Available online: https://isfassociates.com/ (accessed on 23 April 2025).
- Kajimura, S., Margulies, D., & Smallwood, J. (2023). Frequency-specific brain network architecture in resting-state fMRI. Scientific Reports, 13(1), 2964. [Google Scholar] [CrossRef]
- Klöbl, M., Prillinger, K., Diehm, R., Doganay, K., Lanzenberger, R., Poustka, L., Plener, P., & Konicar, L. (2023). Individual brain regulation as learned via neurofeedback is related to affective changes in adolescents with autism spectrum disorder. Child and Adolescent Psychiatry and Mental Health, 17(1), 6. [Google Scholar] [CrossRef]
- Konicar, L., Radev, S., Prillinger, K., Klöbl, M., Diehm, R., Birbaumer, N., Lanzenberger, R., Plener, P., & Poustka, L. (2021). Volitional modification of brain activity in adolescents with autism spectrum disorder: A Bayesian analysis of Slow Cortical Potential neurofeedback. NeuroImage: Clinical, 29, 102557. [Google Scholar] [CrossRef]
- Kropotov, J. D. (2022). The enigma of infra-slow fluctuations in the human EEG. Frontiers in Human Neuroscience, 16, 928410. [Google Scholar] [CrossRef]
- Larsen, S., & Sherlin, L. (2013). Neurofeedback: An emerging technology for treating central nervous system dysregulation. Psychiatric Clinics of North America, 36(1), 163–169. [Google Scholar] [CrossRef]
- Legarda, S. B., McMahon, D., Othmer, S., & Othmer, S. (2011). Clinical neurofeedback: Case studies, proposed mechanism, and implications for pediatric neurology practice. Journal of Child Neurology, 26(8), 1045–1051. [Google Scholar] [CrossRef]
- Leins, U., Goth, G., Hinterberger, T., Klinger, C., Rumpf, N., & Strehl, U. (2007). Neurofeedback for children with ADHD: A comparison of SCP and theta/beta protocols. Applied Psychophysiology and Biofeedback, 32(2), 73–88. [Google Scholar] [CrossRef]
- Leins, U., Hinterberger, T., Kaller, S., Schober, F., Weber, C., & Strehl, U. (2006). Neurofeedback der langsamen kortikalen potenziale und der theta/beta-aktivität für Kinder mit einer ADHS: Ein kontrollierter vergleich [Neurofeedback for children with ADHD: A comparison of SCP- and theta/beta-protocols]. Praxis der Kinderpsychologie und Kinderpsychiatrie, 55(5), 384–407. [Google Scholar] [PubMed]
- Linssen, A. M., Vuurman, E. F., Sambeth, A., Nave, S., Spooren, W., Vargas, G., Santarelli, L., & Riedel, W. J. (2011). Contingent negative variation as a dopaminergic biomarker: Evidence from dose-related effects of methylphenidate. Psychopharmacology, 218, 533–542. [Google Scholar] [CrossRef] [PubMed]
- Mazefsky, C. A., Herrington, J., Siegel, M., Scarpa, A., Maddox, B. B., Scahill, L., & White, S. W. (2013). The role of emotion regulation in autism spectrum disorder. Journal of the American Academy of Child & Adolescent Psychiatry, 52(7), 679–688. [Google Scholar] [CrossRef] [PubMed]
- Micoulaud-Franchi, J.-A., Geoffroy, P. A., Fond, G., Lopez, R., Bioulac, S., & Philip, P. (2014). EEG neurofeedback treatments in children with ADHD: An updated meta-analysis of randomized controlled trials. Frontiers in Human Neuroscience, 8, 906. [Google Scholar] [CrossRef] [PubMed]
- Minder, F., Zuberer, A., Brandeis, D., & Drechsler, R. (2018). Informant-related effects of neurofeedback and cognitive training in children with ADHD including a waiting control phase: A randomized-controlled trial. European Child & Adolescent Psychiatri, 27(8), 1055–1066. [Google Scholar] [CrossRef]
- Morales-Quezada, L., Martinez, D., El-Hagrassy, M. M., Kaptchuk, T. J., Sterman, M. B., & Yeh, G. Y. (2019). Neurofeedback impacts cognition and quality of life in pediatric focal epilepsy: An exploratory randomized double-blinded sham-controlled trial. Epilepsy & Behavior, 101, 106570. [Google Scholar] [CrossRef]
- Moreno-García, I., Cano-Crespo, A., & Rivera, F. (2022). Results of neurofeedback in treatment of children with ADHD: A systematic review of randomized controlled trails. Applied Psychophysiology and Biofeedback, 47, 145–181. [Google Scholar] [CrossRef]
- Ning, J., Zhang, Y., Wang, Y., Liu, C., Cheng, Y., Zhu, M., Dong, M., Yang, X., & Lv, Y. (2023). Exploring the cortical habituation in migraine patients based on contingent negative variation. Frontiers in Neurology, 14, 1226554. [Google Scholar] [CrossRef]
- Okumura, Y., Kita, Y., Omori, M., Suzuki, K., Yasumura, A., Fukuda, A., & Inagaki, M. (2019). Predictive factors of success in neurofeedback training for children with ADHD. Developmental Neurorehabilitation, 22(1), 3–12. [Google Scholar] [CrossRef] [PubMed]
- Othmer, S. (2020). History of neurofeedback. In H. W. Kirk (Ed.), Restoring the brain (2nd ed., pp. 23–55). Routledge. [Google Scholar]
- Othmer, S., & Othmer, S. F. (2024). Endogenous neuromodulation at infra-low frequency: Method and theory (preprint). Preprints.org. Available online: https://www.researchgate.net/publication/374784708_Endogenous_Neuromodulation_at_Infra-Low_Frequency_Method_and_Theory (accessed on 23 April 2025). [CrossRef]
- Othmer, S., Othmer, S. F., & Legarda, S. B. (2011). Clinical neurofeedback: Training brain behavior. Treatment Strategies–Pediatric Neurology and Psychiatry, 2(1), 67–73. [Google Scholar]
- Othmer, S. F. (2017). Protocol guide for neurofeedback clinicians (6th ed.). EEG Info. [Google Scholar]
- Pascual-Marqui, R. D. (2002). Standardized low-resolution brain electromagnetic tomography (sLORETA): Technical details. Methods & Findings in Experimental & Clinical Pharmacology, 24(Suppl. D), 5–12. [Google Scholar]
- Pimenta, M. G., Brown, T., Arns, M., & Enriquez-Geppert, S. (2021). Treatment efficacy and clinical effectiveness of EEG neurofeedback as a personalized and multimodal treatment in ADHD: A critical review. Neuropsychiatric Disease and Treatment, 17, 637–648. [Google Scholar] [CrossRef]
- Pineda, J. A., Friedrich, E. V. C., & LaMarca, K. (2014). Neurorehabilitation of social dysfunctions: A model-based neurofeedback approach for low and high-functioning autism. Frontiers in Neuroengineering, 7, 29. [Google Scholar] [CrossRef]
- Prillinger, K., Radev, S. T., Doganay, K., Poustka, L., & Konicar, L. (2022). Impulsivity moderates the effect of neurofeedback training on the contingent negative variation in autism spectrum disorder. Frontiers in Human Neuroscience, 16, 838080. [Google Scholar] [CrossRef] [PubMed]
- Prinz, W. (2015). Neurofeedbacktherapie als spezialtherapieangebot. Fallbericht aus einem kinder-und jugendpsychiatrischen dienst in der Schweiz [Neurofeedback therapy as a special therapeutic option: Case report from a Swiss child and adolescent psychiatric practice]. Psychopraxis. Neuropraxis, 18, 180–183. [Google Scholar] [CrossRef]
- Raut, R. V., Snyder, A. Z., Mitra, A., Yellin, D., Fujii, N., Malach, R., & Raichle, M. E. (2021). Global waves synchronize the brain’s functional systems with fluctuating arousal. Science Advances, 7(30), eabf2709. [Google Scholar] [CrossRef] [PubMed]
- Rauter, A., Schneider, H., & Prinz, W. (2022). Effectivity of ILF neurofeedback on autism spectrum disorder—A case study. Frontiers in Human Neuroscience, 16, 892296. [Google Scholar] [CrossRef] [PubMed]
- Rockstroh, B., Elbert, T., Lutzenberger, W., & Birbaumer, N. (1990). Biofeedback: Evaluation and therapy in children with attentional dysfunctions. In A. Rothenberger (Ed.), Brain and behavior in child psychiatry (pp. 345–357). Springer. [Google Scholar] [CrossRef]
- Schneider, E., & Krombholz, A. (2016). Frequenzbandtraining. In K. M. Haus, C. Held, A. Kowalski, A. Krombholz, M. Novak, M. Schneider, G. Strauß, & M. Wiedemann (Eds.), Praxisbuch biofeedback und neurofeedback (2nd ed., pp. 45–62). Springer. [Google Scholar]
- Schneider, E., & Strauß, G. (2016). Training der selbstkontrolle der langsamen kortiallen potentiale. In K. M. Haus, C. Held, A. Kowalski, A. Krombholz, M. Novak, M. Schneider, G. Strauß, & M. Wiedemann (Eds.), Praxisbuch biofeedback und neurofeedback (2nd ed., pp. 63–90). Springer. [Google Scholar]
- Schneider, H., Riederle, J., & Seuss, S. (2022). Therapeutic effect of infra-low-frequency neurofeedback training on children and adolescents with ADHD. In Brain-computer interface. IntechOpen. [Google Scholar] [CrossRef]
- Schweren, L., Hoekstra, P., van Lieshout, M., Oosterlaan, J., Lambregts-Rommelse, N., Buitelaar, J., Franke, B., & Hartman, C. (2019). Long-term effects of stimulant treatment on ADHD symptoms, social–emotional functioning, and cognition. Psychological Medicine, 49(2), 217–223. [Google Scholar] [CrossRef] [PubMed]
- Smith, M. L. (2013). Infra-slow fluctuation training: On the down-low in neuromodulation. NeuroConnections, Fall 2013, 38–46. [Google Scholar]
- Smith, M. L., Leiderman, L. M., & de Vries, J. (2016). Infra-slow fluctuation (ISF) training for autism spectrum disorders. In T. F. Collura, & J. A. Frederick (Eds.), Handbook of clinical QEEG and neurotherapy (pp. 488–499). Routledge. [Google Scholar]
- Solberg, B., & Solberg, E. (2022). Infra-low frequency neurofeedback in application to Tourette syndrome and other tic disorders: A clinical case series. Frontiers in Human Neuroscience, 16, 891924. [Google Scholar] [CrossRef]
- Spencer, T. J., Biederman, J., Wilens, T. E., & Faraone, S. V. (2002). Novel treatments for attention-deficit/hyperactivity disorder in children. Journal of Clinical Psychiatry, 63, 16–22. [Google Scholar] [PubMed]
- Strehl, U., Aggensteiner, P., Wachtlin, D., Brandeis, D., Albrecht, B., Arana, M., Bach, C., Banaschewski, T., Bogen, T., & Flaig-Röhr, A. (2017). Neurofeedback of slow cortical potentials in children with attention-deficit/hyperactivity disorder: A multicenter randomized trial controlling for unspecific effects. Frontiers in Human Neuroscience, 11, 135. [Google Scholar] [CrossRef]
- Strehl, U., Leins, U., Goth, G., Klinger, C., Hinterberger, T., & Birbaumer, N. (2006). Self-regulation of slow cortical potentials: A new treatment for children with attention-deficit/hyperactivity disorder. Pediatrics, 118(5), e1530–e1540. [Google Scholar] [CrossRef]
- Studer, P., Kratz, O., Gevensleben, H., Rothenberger, A., Moll, G. H., Hautzinger, M., & Heinrich, H. (2014). Slow cortical potential and theta/beta neurofeedback training in adults: Effects on attentional processes and motor system excitability. Frontiers in Human Neuroscience, 8, 555. [Google Scholar] [CrossRef] [PubMed]
- Stuss, D. T., & Alexander, M. P. (2007). Is there a dysexecutive syndrome? Philosophical Transactions of the Royal Society B: Biological Sciences, 362(1481), 901–915. [Google Scholar] [CrossRef]
- Thatcher, R. W., Lubar, J. F., & Koberda, J. L. (2019). Z-score EEG biofeedback: Past, present, and future. Biofeedback, 47(4), 89–103. [Google Scholar] [CrossRef]
- Tschiesner, R. (2023). Infra-low-frequency neurofeedback treatment in Dysthymia: A case study. Behavioral Sciences, 13(9), 711. [Google Scholar] [CrossRef] [PubMed]
- Walter, W. G., Cooper, R., Aldridge, V., McCallum, W., & Winter, A. (1964). Contingent negative variation: An electric sign of sensori-motor association and expectancy in the human brain. Nature, 203(4943), 380–384. [Google Scholar] [CrossRef] [PubMed]
- Wangler, S., Gevensleben, H., Albrecht, B., Studer, P., Rothenberger, A., Moll, G. H., & Heinrich, H. (2011). Neurofeedback in children with ADHD: Specific event-related potential findings of a randomized controlled trial. Clinical Neurophysiology, 122(5), 942–950. [Google Scholar] [CrossRef] [PubMed]
- Watson, B. O. (2018). Cognitive and physiologic impacts of the infraslow oscillation. Frontiers in Systems Neuroscience, 12, 44. [Google Scholar] [CrossRef] [PubMed]
- Werneck-Rohrer, S. G., Lindorfer, T. M., Waleew, C., Philipp, J., Prillinger, K., & Konicar, L. (2022). Effects of an intensive slow cortical potentials neurofeedback training in female and male adolescents with autism spectrum disorder: Are there sex differences? Wiener Klinische Wochenschrift, 134, 60–68. [Google Scholar] [CrossRef] [PubMed]
- Wigal, S. B. (2009). Efficacy and safety limitations of attention-deficit hyperactivity disorder pharmacotherapy in children and adults. CNS Drugs, 23(Suppl. 1), 21–31. [Google Scholar] [CrossRef]
- Winkeler, A., Winkeler, M., & Imgart, H. (2022). Infra-low frequency neurofeedback in the treatment of patients with chronic eating disorder and comorbid post-traumatic stress disorder. Frontiers in Human Neuroscience, 16, 890682. [Google Scholar] [CrossRef]
- Zuberer, A., Minder, F., Brandeis, D., & Drechsler, R. (2018). Mixed-effects modeling of neurofeedback self-regulation performance: Moderators for learning in children with ADHD. Neural Plasticity, 2018(1), 2464310. [Google Scholar] [CrossRef]

| Slow Cortical Potentials (SCPs) | Infra-Low Frequency (ILF) | Infra-Slow Fluctuations (ISFs) | |
|---|---|---|---|
| Frequency Range | Below 0.5 Hz | Below 0.1 Hz | Between 0.1–0.01 Hz |
| Reflection of activity | Neuronal | Neuronal and glial | Neuronal and glial |
| Role | Excitability shifts | State stabilization | Brain state transitions |
| Applications | Task-specific preparation | Arousal stabilization | State transitions |
| Conscious reward | Yes | Primarily non-conscious | Yes |
| Electrode placements | Unipolar (e.g., Cz with mastoid as reference) | Bipolar (e.g., T3–T4, with Cz as reference) | Montage varies by protocol |
| Main equipment | Sintered Ag/AgCl electrodes; Direct Current-coupled amplifier, PC Interface | ||
| Authors | Age | Diagnosis | n(SO-NF) (Sex) | Design | Control | Protocol | Total NF Sessions | Clinical Change |
|---|---|---|---|---|---|---|---|---|
| Strehl et al. (2006) | 8–13 | ADHD | 23 (4 F, 16 M) | Pre–Post-Assessment with 6-month follow-up | X | SCP | 30 | ↑ |
| Leins et al. (2006) | 8–13 | ADHD | 19 out of 38 (3 F, 16 M) | RCT | ✓ | SCP | 30 | ↑ |
| Leins et al. (2007) | 11–13 | ADHD | 19 out of 38 (3 F, 16 M) | Comparative study | ✓ | SCP | 60 | ↑ |
| Drechsler et al. (2007) | 9–13 | ADHD | 17 out of 30 (4 F, 13 M) | RCT | ✓ | SCP | 30 | ⟷ ↑ |
| Doehnert et al. (2008) | 9–12 | ADHD | 14 out of 26 (2 F, 12 M) | RCT | ✓ | SCP | 30 | ↑ |
| Gevensleben et al. (2009a) | 8–12 | ADHD | 46 out of 72 (5 F, 41 M) | RCT | ✓ | SCP and θ/β | 18 and 18 | ↑ |
| Gevensleben et al. (2009b) | 8–12 | ADHD | 59 out of 94 (8 F, 51 M) | RCT | ✓ | SCP and θ/β | 18 and 18 | ↑ |
| Gevensleben et al. (2010) | 8–12 | ADHD | 59 out of 94 (8 F, 51 M) | RCT | ✓ | SCP and θ/β | 18 and 18 | ↑ |
| Wangler et al. (2011) | 8–12 | ADHD | 59 out of 94 (8 F, 51 M) | RCT | ✓ | SCP and θ/β | 18 and 18 | ↑ |
| Christiansen et al. (2014) | 7–11 | ADHD | 29 out of 58 (5 F, 24 M) | RCT | ✓ | SCP | 30 | ⟷ |
| Zuberer et al. (2018) | 8–17 | ADHD | 48 (21 F, 27 M) | RCT | X | SCP | 15 double sessions | ↑ ⟷ |
| Albrecht et al. (2017) | 7–17 | ADHD | 24 (16 F, 8 M) | Pre–Post-Assessment | X | SCP | 20 | ↑ |
| Strehl et al. (2017) | 7–9 | ADHD | 75 out of 144 (14 F, 61 M) | RCT | ✓ | SCP | 25 | ⟷ |
| Minder et al. (2018) | 8–15 | ADHD | 38 out of 77 (15 F, 23 M) | RCT | ✓ | SCP | 20–28 | ⟷ |
| Baumeister et al. (2018) | 10–13 | ADHD | 8 out of 16 (2 F, 6 M) | RCT | ✓ | SCP | 20 | ↑ |
| Baumeister et al. (2019) | 11–13 | ADHD | 8 out of 15 (2 F, 6 M) | RCT | ✓ | SCP | 20 | ↑ |
| Aggensteiner et al. (2019) | 7–9 | ADHD | 75 out of 150 (14 F, 61 M) | RCT | ✓ | SCP | 25 | ↑ |
| Okumura et al. (2019) | 7–17 | ADHD | 22 (5 F, 17 M) | Pre-/Post Assessment | X | SCP | 6 | ⟷ |
| Heinrich et al. (2020) | 8–12 | ADHD | 30 out of 48 (5 F, 25 M) | RCT | ✓ | SCP and θ/β | 18 and 18 | ↑ |
| Hasslinger et al. (2020) | 9–13 | ADHD | 30 (9 F, 21 M) | qualitative study | X | SCP | 25 | ↑ |
| Aggensteiner et al. (2021) | 7–9 | ADHD | 50 out of 103 (10 F, 40 M) | RCT | ✓ | SCP | 25 | ⟷ |
| H. Schneider et al. (2022) | 7–21 | ADHD | 196 (41 F, 155 M) | Pre–/Post Assessment | X | ILF | ~39 | ↑ |
| Hasslinger et al. (2022a) | 9–17 | ADHD | 51 out of 202 (13 F, 38 M) | RCT | ✓ | SCP | 25 | ⟷ |
| Hasslinger et al. (2022b) | 9–17 | ADHD | 51 out of 202 (13 F, 38 M) | RCT | ✓ | SCP | 25 | ⟷ |
| Smith et al. (2016) | 6, 29 | ASD | 2 (2 M) | case report | X | ISF | ~ 80 | ↑ |
| Rauter et al. (2022) | 5 | ASD | 1 (1 M) | case report | X | ILF | 26 | ↑ |
| Konicar et al. (2021) | 12–17 | ASD | 21 out of 41 (21 M) | RCT | ✓ | SCP | 24 | ↑ |
| Prillinger et al. (2022) | 12–17 | ASD | 21 out of 41 (21 M) | RCT | ✓ | SCP | 24 | ↑ |
| Werneck-Rohrer et al. (2022) | 12–17 | ASD | 12 (6 F, 6 M) | clinical trial | X | SCP | 24 | ⟷ |
| Klöbl et al. (2023) | 12–17 | ASD | 21 out of 41 (21 M) | clinical trial | ✓ | SCP | 24 | ⟷ |
| Esmaeilzadeh Kanafgourabi et al. (2023) | 12–16 | ASD | 12 out of 24 (12 M) | RCT | ✓ | ILF | 15 | ↑ |
| Legarda et al. (2011) | 6–19 | Epilepsy | 3 (1 F, 2 M) | case report | X | ISF (ILF) | 13–21 | ↑ |
| Morales-Quezada et al. (2019) | 10–19 | Focal epilepsy | 16 out of 44 (7 F, 9 M) | RCT | ✓ | SCP | 25 | ↑ |
| Prinz (2015) | 14, 17 | Tic Disorder | 2 (1 F, 1 M) | case report | X | ILF | 9–10 | ↑ |
| Solberg and Solberg (2022) | 8–18 | Tic Disorder | 100 (35 F, 65 M) | case studies | X | ILF | 30–40 | ↑ |
| Chirita-Emandi and Puiu (2014) | 6–18 | Obesity | 12 out of 34 (5 F, 7 M) | pilot study | ✓ | ILF | 20 | ↓ ↑ |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Glaubig, L.; Azza, Y.; Beber, S.; Silbernagl, P.; Barradas, I.; Peer, A.; Tschiesner, R. Slow-Oscillation Neurofeedback: A Narrative Review on Clinical Efficacy in Pediatric Settings. Behav. Sci. 2026, 16, 337. https://doi.org/10.3390/bs16030337
Glaubig L, Azza Y, Beber S, Silbernagl P, Barradas I, Peer A, Tschiesner R. Slow-Oscillation Neurofeedback: A Narrative Review on Clinical Efficacy in Pediatric Settings. Behavioral Sciences. 2026; 16(3):337. https://doi.org/10.3390/bs16030337
Chicago/Turabian StyleGlaubig, Lea, Yasmine Azza, Sabrina Beber, Philipp Silbernagl, Isabel Barradas, Angelika Peer, and Reinhard Tschiesner. 2026. "Slow-Oscillation Neurofeedback: A Narrative Review on Clinical Efficacy in Pediatric Settings" Behavioral Sciences 16, no. 3: 337. https://doi.org/10.3390/bs16030337
APA StyleGlaubig, L., Azza, Y., Beber, S., Silbernagl, P., Barradas, I., Peer, A., & Tschiesner, R. (2026). Slow-Oscillation Neurofeedback: A Narrative Review on Clinical Efficacy in Pediatric Settings. Behavioral Sciences, 16(3), 337. https://doi.org/10.3390/bs16030337







