1. Introduction
In an increasingly globalised world, in which over half the population is considered bilingual [
1], the ability to communicate in more than one language offers a range of personal and professional advantages. Whether and how the processing of two or more languages in one mind may alter cognition has been the focus of a considerable body of research, and the argument that bilingualism offers genuine cognitive advantages has been increasingly challenged in recent years (see [
2] for a comprehensive overview of the debate). Some empirical evidence suggests that, in comparison to monolinguals, bilingual individuals across the lifespan and from a range of linguistic backgrounds are faster and less affected by conflicting response demands when performing tasks measuring executive function (e.g., [
3,
4]). In particular, a bilingual advantage has been reported on measures of inhibition (e.g., [
4,
5]), attention shifting (e.g., [
6,
7]) and updating in working memory [
4,
8], although such claims are countered by evidence that observed advantages are typically small and statistically unreliable, particularly when considered in the context of publication bias towards reporting positive effects [
9,
10,
11,
12].
One interpretation of these effects derives from psycholinguistic evidence that bilinguals’ two languages are simultaneously activated at all times, even in unilingual contexts [
13]. One of the most influential theoretical frameworks, the Inhibitory Control Model [
14,
15] (ICM), proposes that this unique form of language processing requires the active inhibition of one language in favour of producing the other (see [
16] for an alternative explanation). According to the ICM, in order to resolve the competition between the two languages, cognitive control mechanisms are required. It is the additional cognitive effort associated with processing of two (or more) languages that is, therefore, thought to lead to enhancement in executive function [
17]. A recent development of the ICM, the adaptive control hypothesis [
18], further postulates that the kinds of control mechanisms used in bilingual speech production adapt according to the demands of an individual’s everyday interactional context, with an increased need to switch between two languages leading to a broader range of cognitive control advantages.
However, in the context of recent challenges to theory, concerns about methods and the appropriateness of statistical analyses employed in bilingual cognition research [
2], the question of whether or not bilingualism is associated with enhanced cognitive abilities remains fiercely debated. One theme that has emerged is that if a bilingual advantage does exist, it may be task-specific or otherwise operate only across particular groups of participants [
19,
20]. Crucially, it may also be premature to speak of a universal bilingual
advantage in non-verbal cognitive functioning in light of a recent report of a bilingual disadvantage in metacognition [
21]. Nevertheless, given the prevalence of bilingualism and the implication this may have for professional practice of, for example, educators and clinicians, it remains an important endeavour for scientists to chart and understand the broad implications of multilanguage acquisition on cognition.
An important gap in the literature to date is that, to our knowledge, no studies have addressed the question of whether individual emotional states or traits may differentially affect bilinguals’ and monolinguals’ performance on tasks measuring executive functions. To our knowledge, there is only one study that has investigated emotion processing, specifically emotion regulation, in bilingual individuals [
22]. Janus and Bialystok administered the Emotional Face
n-Back task to 9-year-old monolingual and bilingual children. In this task, participants must indicate whether a letter has been shown on the previous screen (1-back) or on the screen before that (2-back), while faces displaying an angry, happy, or neutral expression are shown on both sides of the letter. The authors found that, whilst bilingual children performed the task more accurately overall and more slowly in the 2-back condition, the effects of emotional valence on reaction time did not differ across groups. They interpreted these findings as evidence that bilingual children may be at an advantage in terms of adjusting their behaviour to task demands but not in terms of emotion regulation. Whilst this study may add to our understanding of emotion processing in bilingual individuals, the focus was on cognitive performance of a task involving emotionally valanced stimuli rather than the effect of the emotional states or traits of the participants themselves. Anxiety and other mood disorders are among the most commonly occurring mental health problems, representing a substantial burden to the economy (e.g., [
23]). The present study explores the effects of trait anxiety and rumination on inhibition, as conceptualised by Attentional Control Theory [
24] (ACT), in young, healthy monolingual and bilingual adults.
The ACT relies on the assumption that anxiety (in both clinical and non-clinical populations) adversely affects processing efficiency (typically inferred via reaction times) to a greater extent than performance effectiveness (i.e., accuracy) [
25]. Specifically, in order to prevent anxiety from adversely affecting their performance, highly anxious individuals are thought to modulate the amount of effort they exert on difficult cognitive tasks, thus operating at a decreased level of efficiency in comparison to individuals with low levels of anxiety. The theory further assumes that there are two attentional systems [
26,
27]: one goal-directed (top-down) and the other stimulus-driven (bottom-up). Anxiety is thought to alter how these two attentional systems are balanced, with the presence of threatening stimuli decreasing goal-directed and increasing stimulus-driven attention. Eysenck and colleagues also argued that the challenges of maintaining goal-directed attention through inhibition and shifting should be most affected by anxiety, whereas storing information (updating) is not directly linked to attentional control and, thus, should not be associated with these effects as strongly and only be observable under particularly stressful conditions (although note that trait worry, a component of trait anxiety, has been related to updating [
28,
29]).
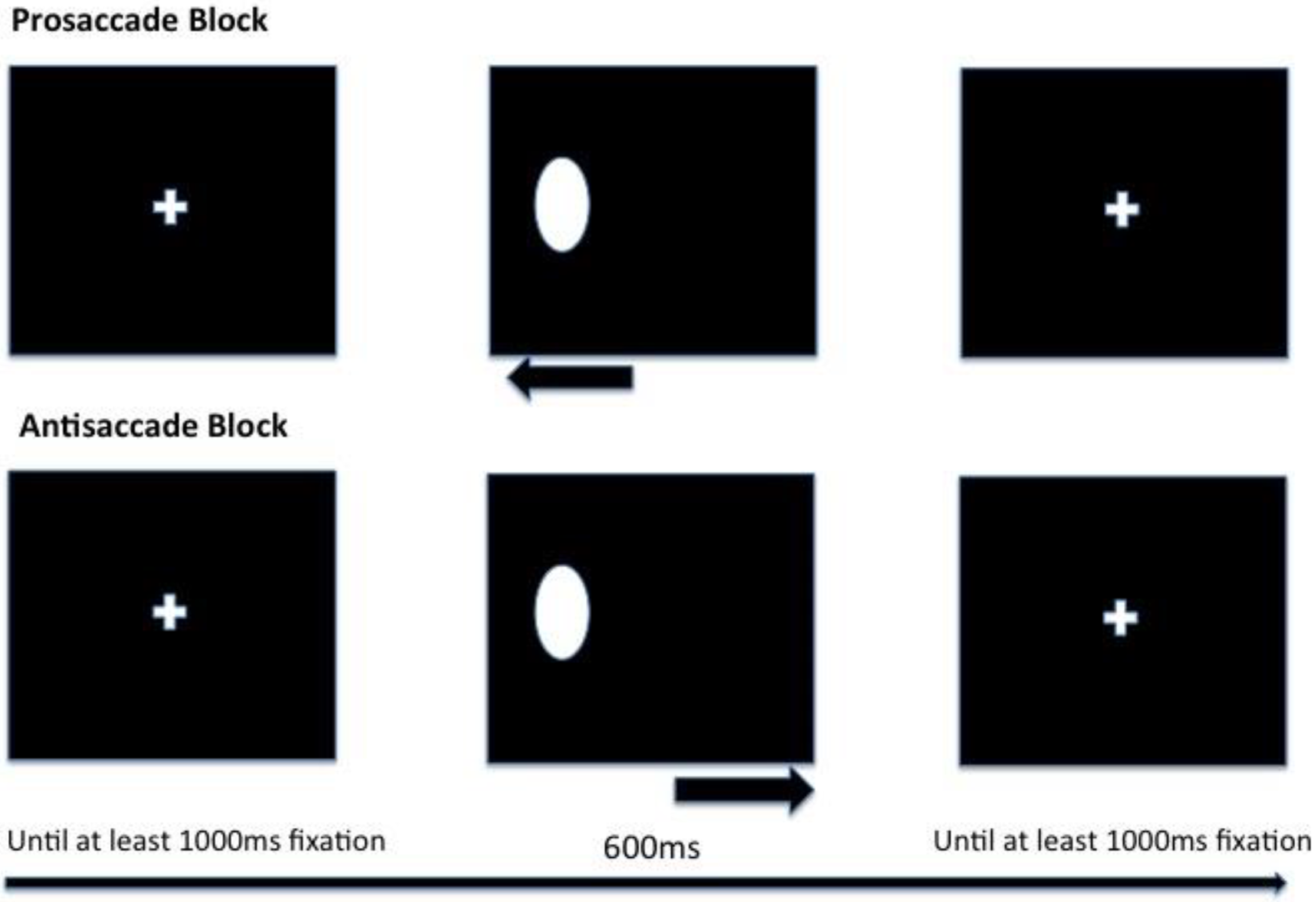
A substantial body of work has provided empirical support for the individual assumptions and hypotheses of the ACT. One method for testing the effects of anxiety on attentional control is the assessment of continuous overt visual attention via analysis of eye movements (saccades) [
30,
31] on the antisaccade task (note that the antisaccade task incorporates both pro- and antisaccade conditions) [
32]. This task provides a measure of visual inhibition [
33] because it incorporates an antisaccade condition in which the participant is required to produce a saccade to the opposite side of space from a visually presented stimulus [
34,
35,
36]. Derakhshan, Ansari, Hansard, Shoker, and Eysenck [
37] tested sixty-one healthy adults on two versions of the task, one featuring an oval stimulus (classic version) and the other neutral, happy, and angry faces. The study mainly focused on the latency of the first saccade made on each antisaccade trial, which is argued to be an indicator of processing efficiency (i.e., it is typically prolonged due to the requirement to inhibit a reflexive saccade to the stimulus). Furthermore, Derakhshan and colleagues assessed saccadic, as well as behavioural, accuracy (performance effectiveness) and corrective behaviours (correcting an erroneous saccade within the same trial). The latter, they argued, could be an indicator of compensatory strategies used in difficult (antisaccade) trials by high-anxiety participants.
All participants completed the Trait Anxiety Scale of the State-Trait Anxiety Inventory [
38], which is a well-established self-report measure assessing how individuals feel about themselves in general (Cronbach’s α = 90 [
39]). The authors conducted a tertile split and only included those with the highest and the lowest trait anxiety scores in the analysis. In line with the assumptions of the ACT, the authors found that high-anxiety individuals showed reduced processing efficiency when the task was difficult (i.e., they produced longer antisaccade latencies), but did not differ from the low-anxiety group on any of the performance effectiveness measures (saccadic and behavioural accuracy, corrective behaviours), or on prosaccade performance. Furthermore, they found that the presence of threatening stimuli (angry faces) disproportionately affected processing efficiency in high-anxiety individuals, thereby supporting the ACT hypothesis that anxiety decreases goal-directed attention in favour of increased stimulus-driven attention.
In a later study, De Lissnyder, Derakhshan, De Raedt, and Koster [
40] assessed these effects in a healthy population differentiated in terms of general depressive symptoms, as well as rumination. Rumination is a cognitive symptom of depression, which manifests itself in recurrent thoughts, contemplating the symptoms, causes, as well as implications of one’s depressive state [
41]. This disposition to self-focus has previously been argued to be a key element of cognitive vulnerability associated with depression [
42,
43] De Lissnyder and colleagues [
40] administered the self-report Ruminative Response Scale [
44] in order to assess participants’ overall ruminative tendencies, as well as the two distinct subtypes of rumination,
reflective pondering (the focus on problem solving; adaptive rumination) and
depressive brooding (the focus on one’s negative mood; maladaptive rumination) [
45]. They administered the classic antisaccade task, in order to assess inhibition, as well as a mixed (shifting) version of the task, in which the direction of the gaze is determined on a trial-by-trial basis by a cue displayed in the fixation period [
46]. The authors found that two groups with high and low general depression did not differ in their performance of the antisaccade task. In contrast, the high-rumination group was found to display slower antisaccade latencies when compared to the low-rumination group, with depressive brooding being a predictor of antisaccade latencies in particular. Thus, the study of De Lissnyder and colleagues [
40] replicated the findings reported by Derakhshan and colleagues [
37], demonstrating that attentional control deficits are not only associated with high trait anxiety, but also with high levels of depressive brooding/maladaptive rumination. In line with previous research [
47], rumination was not associated with deficits in shifting.
Research employing both behavioural methods, as well as neuroimaging, has provided further support for the ACT (see [
48,
49,
50] for reviews). For example, state and trait anxiety, as well as chronic stress, have been found to predict reduced shifting abilities on a variety of tasks [
46,
51,
52]. Furthermore, prefrontal response differences have been identified in neuroimaging and electrophysiological studies between low- and high-anxiety individuals in the absence of behavioural inhibition differences [
53,
54,
55]. Therefore, there is substantial evidence in support of the ACT, deriving from studies using a variety of paradigms.
The Current Study
The current investigation did not seek to address the bilingual executive function advantage per se, but rather sought to evaluate what this commonly postulated advantage may mean within the context of the hypotheses posed by the ACT. Informed by previous literature, we administered a classic version of the antisaccade task, as well as a behavioural measure of inhibition, the Simon task [
56,
57], to young, healthy monolingual and bilingual adults. Whether or not bilingualism is associated with differential oculomotor control abilities is unresolved given the sparse and conflicting evidence currently available [
58]. With regards to the Simon task, Bialystok and colleagues [
59] found evidence that, in the absence of consistent evidence for a behavioural advantage, activation in the dorsolateral prefrontal cortex was associated with faster reaction times in the Simon task only in monolinguals, whereas bilinguals were found to recruit resources in language processing areas of the brain alongside other regions in the left frontal hemisphere. The authors interpreted this to be evidence for the notion that the management of two language systems impacts non-verbal cognitive processing such that bilingual individuals recruit a more diverse network of cortical areas in the service of more efficient processing [
60]. To date, there is a scarcity of studies testing the ACT in light of individual differences. There is some evidence suggesting that increased working memory capacity may serve as a protective mechanism against the adverse effects of anxiety on performance (e.g., [
61,
62]). Given that the ACT predicts that increased levels of anxiety are associated with a more dispersed allocation of attentional resources [
24], it is reasonable to assume that individual differences in cognitive functioning, such as those reported by some studies comparing monolingual and bilingual individuals, will lead to differences in the effect of anxiety on inhibition [
61]. We predicted that trait anxiety and rumination would not impact performance in either group on easy/congruent trials in either task, but that between-group performance would diverge on the more demanding incongruent conditions. The key question here was whether the commonly postulated inhibitory control differences between monolingual and bilingual individuals, behaviourally and/or on a neural level, would lead to differential effects of trait anxiety and rumination on cognitive performance.
4. Discussion
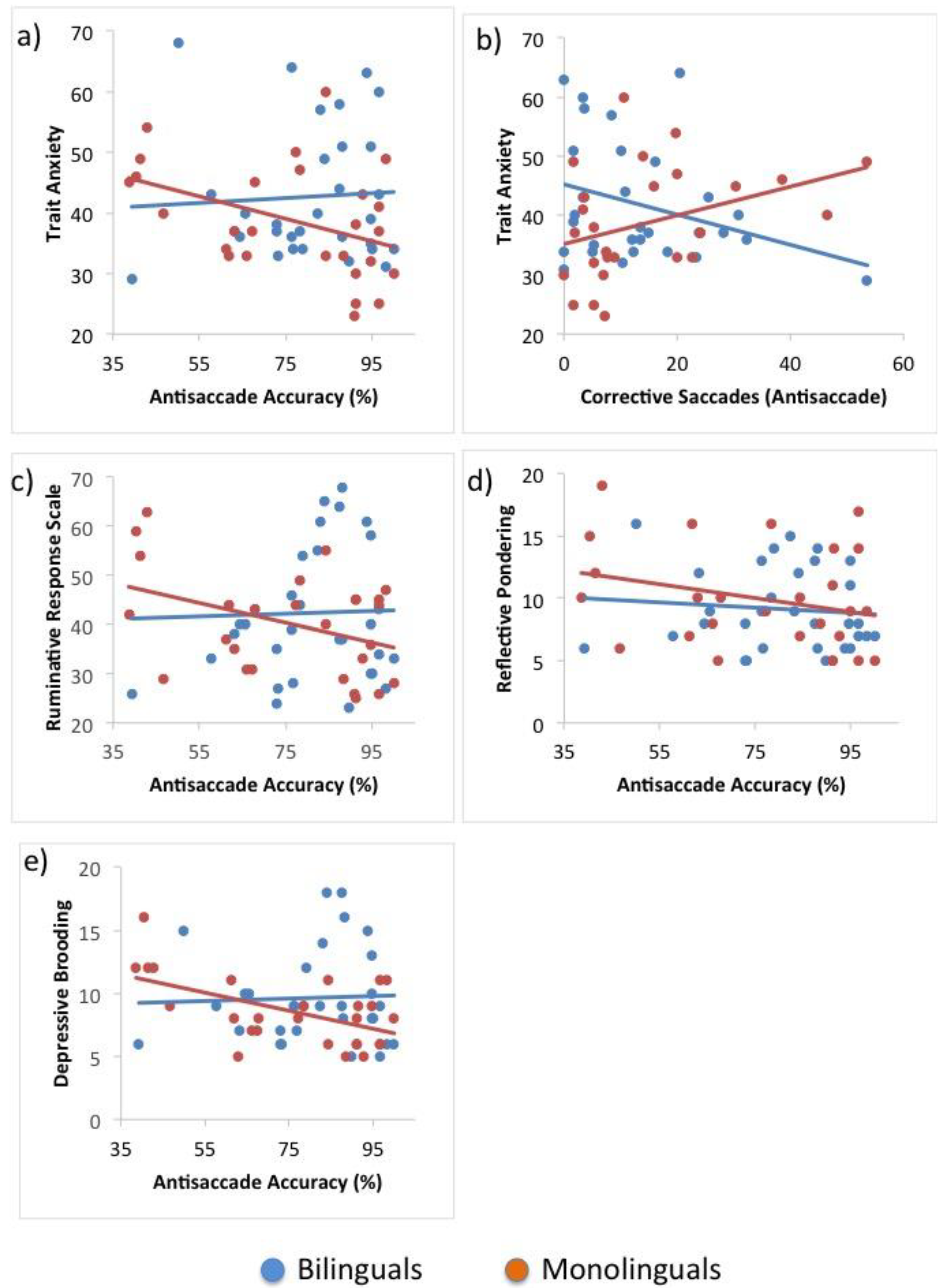
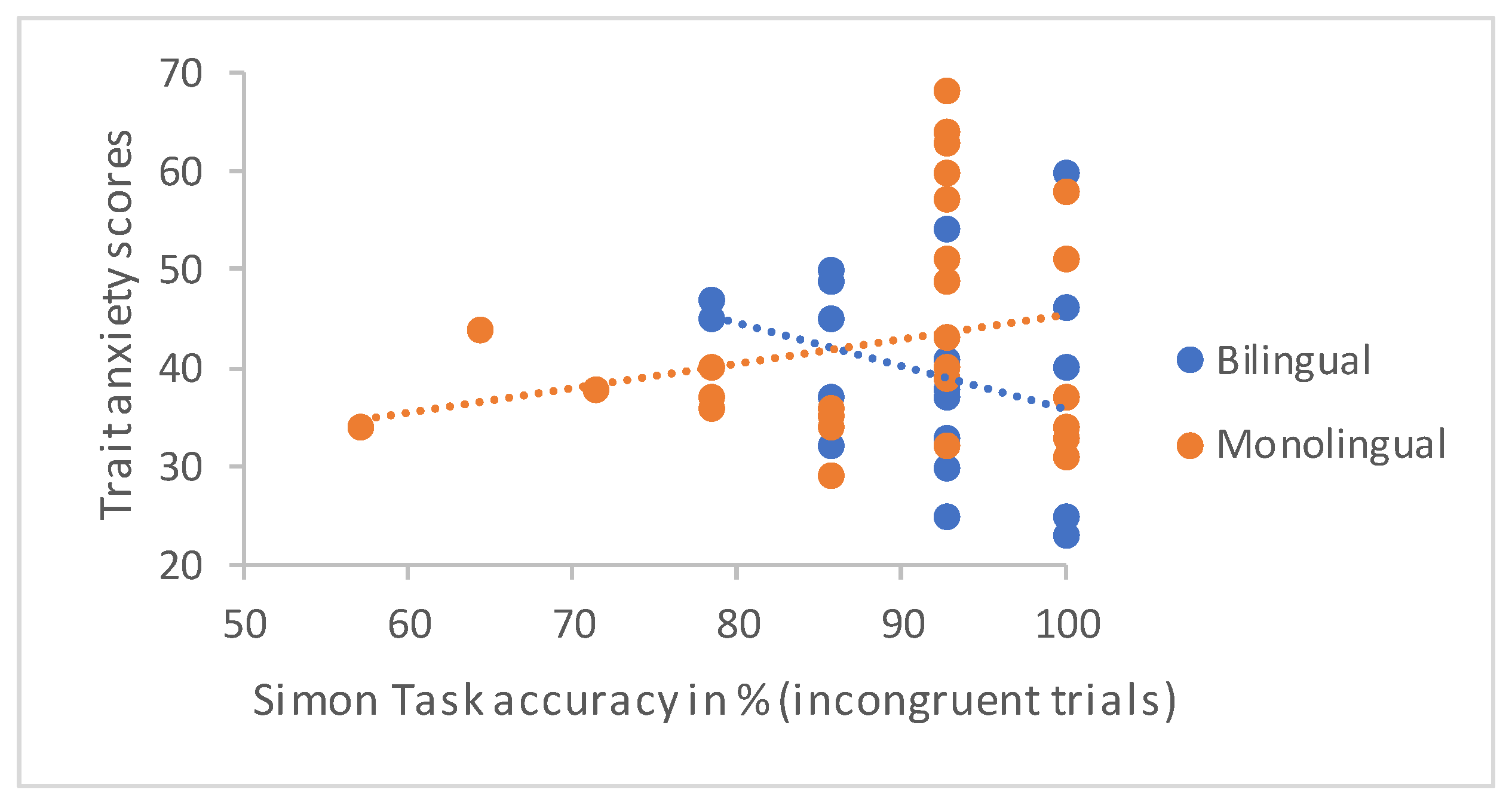
The main objective of this investigation was to evaluate whether monolingual and bilingual individuals differed with regard to the effects of adverse emotional traits on the performance of two measures thought to tap into inhibition. Informed by previous literature, a combined version of the classic pro- and antisaccade tasks, as well as the Simon Task, were employed. The effects of trait anxiety and rumination, as well as the rumination types, reflective pondering and depressive brooding on performance effectiveness and processing efficiency were assessed within both groups and compared across groups.
The main hypothesis of this study, speculating that bilinguals may be differentially affected by adverse emotional states and traits, was confirmed. The analyses revealed that trait anxiety predicted lower levels of performance effectiveness on the most difficult trials of both tasks only in bilingual participants. Remarkably, this effect was found on all variables of performance effectiveness, including saccadic and behavioural accuracy, as well as corrective behaviours. Significant effects of rumination were only found with regard to saccadic accuracy and, in line with findings reported by De Lissnyder and colleagues [
40], depressive brooding but not reflective pondering was found to significantly predict performance. It has previously been noted that the focus on one’s negative mood, which is a characteristic specific to depressive brooding, may lead to attentional inflexibility and, thus, implicate inhibitory resources more so than reflective pondering [
47].
No effects of the emotional variables on processing efficiency were detected in either group. It is important to note here, however, that our study took a different approach to others by focusing specifically on the effects of adverse emotional traits on cognitive performance. Published ACT studies have typically divided participants into low- and high-anxiety individuals (e.g., [
37]) or high- and low ruminators [
40], whilst in this investigation, the effects of adverse emotional traits were assessed on a continuum. Therefore, it is possible that the main assumption of the ACT relevant here, that adverse emotions effect processing efficiency as measured by first antisaccade latencies, can only be observed in a group that scores on the higher end of the spectrum on the emotional measures administered. This interpretation is in line with recent findings suggesting that correlations between performance and anxiety as a continuous variable are not always found, even when high- and low-anxiety individuals differ [
75]. Testing this hypothesis was not possible given the sample size constraints of this study and we thus encourage a replication involving a larger pool of participants.
If bilingual individuals rely on inhibitory mechanisms when controlling their two languages, as hypothesised by the ICM [
14,
15], it is possible that these mechanisms become increasingly efficient over time. In turn, bilingual speakers may have less experience at exerting effort over these mechanisms compared to monolinguals. According to the ACT, anxiety should not have an impact on performance effectiveness (as measured by accuracy) but it should have it on processing efficiency (as indicated by response time). Whereas effectiveness is affected by adverse emotions, efficiency is modulated in order to compensate for these effects. Therefore, performance may still reach the same level, but under a condition of increased effort. If bilinguals do not use effort to modulate inhibitory mechanisms in everyday life, it is plausible that they will not make use of the protective functions of effort when their cognitive functioning is subject to the influence of adverse emotions. However, this interpretation is speculative and at odds with the literature challenging the bilingual advantage, as well as a recent argument that bilingual language control may not rely on executive function beyond the initial stages of second language learning [
76]. Therefore, the evaluation of the differences reported here demands further investigation, for example, in studies employing a broader range of tests of executive function.
With regard to overall task performance, the two groups did not differ on the antisaccade task, which is in line with findings reported by Bialystok, Craik, and Ryan [
77]. Previous research suggests that bilinguals’ level of second language proficiency is a predictor of their cognitive performance (e.g., [
8,
78,
79], although note [
80,
81,
82,
83], who reported contradictory findings). Thus, the finding that bilinguals’ proficiency did not significantly predict performance in the antisaccade task, whilst it did on incongruent trials of the Simon Task, supports the notion proposed by Bialystok and colleagues [
77] that the eye tracking version of the task detects very early processing effects that may not be subject to the bilingual advantage in inhibitory control. In other words, language ability does not appear to predict performance in these early attentional markers. With regard to the Simon Task, similarly to earlier research [
84], no group differences in accuracy or the Simon Effect were detected in this sample of young healthy adults.
Future Directions
Following on from the methodological considerations made above, it will be important for future research to further consider the effects reported in this study in groups of high- and low- anxiety monolinguals and bilinguals. Based on the findings reported here, as well as previous research addressing the assumptions and hypotheses of the ACT, it is possible that these effects are confined to highly anxious bilinguals and will become more pronounced as a result. Furthermore, it will be important to evaluate whether or not processing efficiency is affected differently by anxiety in bilingual individuals. If the current interpretation of these results is correct, i.e., bilinguals do not modulate effort in order to compensate for adverse emotional effects on performance, a high-anxiety bilingual group should not differ from a low-anxiety group with regard to processing efficiency. Alternatively, results could suggest that the effect of trait anxiety and rumination on processing efficiency is reduced in bilinguals, compared to monolinguals. The same pattern of results should emerge in a high-rumination group of bilinguals. However, considering that regression coefficients regarding effects of adverse emotions on processing efficiency did not significantly differ between groups in this study, it is possible that bilinguals experience a more widespread disadvantage in dealing with adverse emotions.
Recent research conducted by Berggen and Derakshan [
75] suggests that anxiety may implicate stimulus–response competition in particular, as opposed to stimulus–stimulus competition. Notably, to our knowledge, this was the first study evaluating the impact of anxiety on distractor cost (i.e., the difference between congruent and incongruent trials), as opposed to treating congruency as an independent variable with two levels, as is common in the ACT literature, e.g., [
37,
40,
85]. Given current challenges to how inhibition has been conceptualised by the literature to date, e.g., [
81,
86], it will be important for future research to systematically evaluate the relationship between adverse emotional traits and different types of inhibition in both monolingual and bilingual individuals.
The question of whether bilingual individuals process threat-related stimuli similarly to monolinguals should also be addressed in future research. Based on the findings reported here, we are hesitant to offer any firm predictions, considering that past research has extensively evaluated a bilingual advantage in inhibiting the presence of task-irrelevant visual stimuli, e.g., [
87,
88]. Research from our lab (see Ouzia and Filippi [
89] for further details) suggests that the relationship between trait anxiety/depressive brooding and sentence comprehension accuracy in the presence of auditory distractors featuring adverse emotions (crying) differs among monolinguals and bilinguals. Specifically, it appears to suggest threat-avoidance in monolinguals, with the presence of a distractor being associated with more accurate performance, whilst bilinguals exhibit attentional bias (i.e., the distractor is associated with a decrease in accuracy). This indicates that, depending on the presentation of the threat-related stimulus, bilinguals may be faced with either an advantage or a disadvantage.
We suggest that evaluating the mechanisms with which adverse emotions affect cognitive functioning in bilingualism will be of great importance for theory, research, and applied work with bilingual individuals. Whilst it may seem counterintuitive at first, bilinguals’ reliance on inhibitory control mechanisms in everyday-life language processing may not always lead to observable advantages, but disadvantages as well. The notion that bilingualism may affect the ways in which individuals are able to exert additional cognitive effort when demanded by internal processes not directly linked to bilingual cognition, such as anxiety and rumination, will require further enquiry. In light of research suggesting that adverse effects of anxiety and how they are dealt with cognitively can impact motivational levels in learning [
90] and also, the inherent importance of this research for understanding cognition in clinical populations [
91], this line of inquiry delivers a promising direction for research on bilingualism.