Speech-Stimulating Substances in Autism Spectrum Disorders
Abstract
1. Biological Aspects of Speech and Verbal Communication in Autism Spectrum Disorder (ASD)
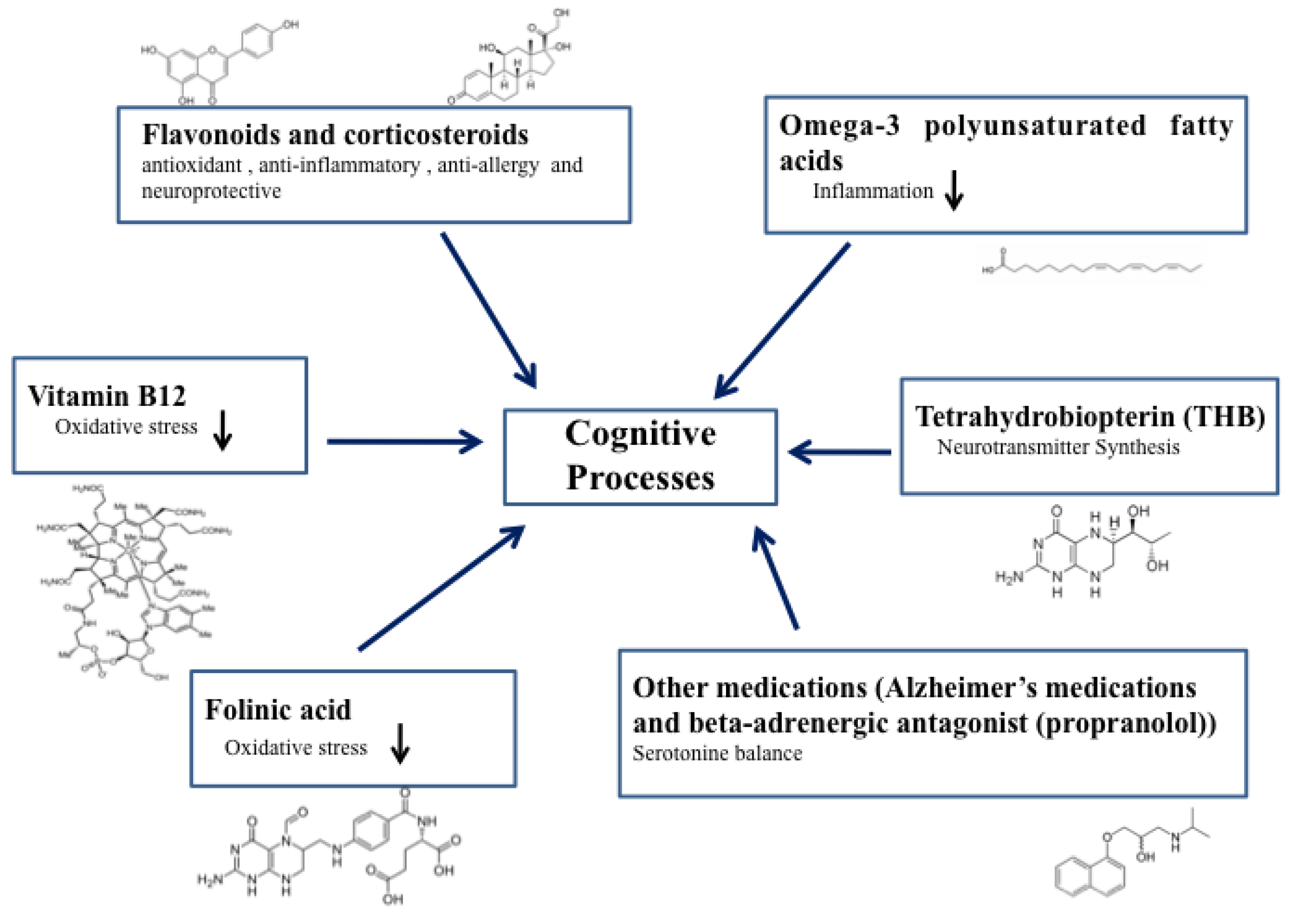
2. Speech-Stimulating Substances in ASD: Overview
2.1. Methylcobalamin (Vitamin B12)
2.2. Tetrahydrobiopterin (THB)
2.3. Folinic Acid
2.4. Omega-3 Polyunsaturated Fatty Acids
2.5. Flavonoids (Luteolin) and Corticosteroids (Prednisolone and Prednisone)
2.6. Other Medications (Alzheimer’s Medications and Beta-Adrenergic Antagonism (Propranolol))
3. Future Perspectives of a Protocol to Stimulate Verbal Communication in ASD
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 4th ed.; (DSM-IV); American Psychiatric Association: Washington, DC, USA, 1994. [Google Scholar]
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 5th ed.; (DSM-V); American Psychiatric Association: Washington, DC, USA, 2013. [Google Scholar]
- Onore, C.; Careaga, M.; Ashwood, P. The role of immune dysfunction in the pathophysiology of autism. Brain Behav. Immun. 2012, 26, 383–392. [Google Scholar] [CrossRef] [PubMed]
- Xu, N.; Li, X.; Zhong, Y. Inflammatory cytokines: Potential biomarkers of immunologic dysfunction in autism spectrum disorders. Med. Inflamm. 2015, 2015, 531518. [Google Scholar] [CrossRef] [PubMed]
- Siniscalco, D.; Schultz, S.; Brigida, A.L.; Antonucci, N. Inflammation and Neuro-Immune Dysregulations in Autism Spectrum Disorders. Pharmaceuticals (Basel) 2018, 11, 56. [Google Scholar] [CrossRef] [PubMed]
- US Centers for Disease Control and Prevention. Prevalence of Autism Spectrum Disorder among Children Aged 8 Years—Autism and Developmental Disabilities Monitoring Network, 11 Sites, United States, 2010, Surveillance Summaries. Morb. Mortal. Wkly. Rep. 2010, 63, 1–21. [Google Scholar]
- Zablotsky, B.; Black, L.I.; Maenner, M.J.; Schieve, L.A.; Blumberg, S.J. Estimated Prevalence of Autism and Other Developmental Disabilities Following Questionnaire Changes in the 2014 National Health Interview Survey. Natl. Health Stat. Rep. Number 2015, 87, 1–21. [Google Scholar]
- Siniscalco, D.; Cirillo, A.; Bradstreet, J.J.; Antonucci, N. Epigenetic findings in autism: New perspectives for therapy. Int. J. Environ. Res. Public Health 2013, 10, 4261–4273. [Google Scholar] [CrossRef] [PubMed]
- Ladd-Acosta, C.; Hansen, K.D.; Briem, E.; Fallin, M.D.; Kaufmann, W.E.; Feinberg, A.P. Common DNA methylation alterations in multiple brain regions in autism. Mol. Psychiatry 2014, 19, 862–871. [Google Scholar] [CrossRef]
- Pu, D.; Shen, Y.; Wu, J. Association between MTHFR gene polymorphisms and the risk of autism spectrum disorders: A meta-analysis. Autism Res. 2013, 6, 384–392. [Google Scholar] [CrossRef]
- Sun, W.; Poschmann, J.; Cruz-Herrera Del Rosario, R.; Parikshak, N.N.; Hajan, H.S.; Kumar, V.; Ramasamy, R.; Belgard, T.G.; Elanggovan, B.; Wong, C.C.Y.; et al. Histone Acetylome-wide Association Study of Autism Spectrum Disorder. Cell 2016, 167, 1385–1397. [Google Scholar] [CrossRef]
- Wu, Y.E.; Parikshak, N.N.; Belgard, T.G.; Geschwind, D.H. Genome-wide, integrative analysis implicates microRNA dysregulation in autism spectrum disorder. Nat. Neurosci. 2016, 19, 1463–1476. [Google Scholar] [CrossRef]
- Lord, C.; Shulman, C.; DiLavore, P. Regression and word loss in autistic spectrum disorders. J. Child Psychol. Psychiatry 2004, 45, 936–955. [Google Scholar] [CrossRef] [PubMed]
- Siperstein, R.; Volkmar, F. Brief report: Parental reporting of regression in children with pervasive developmental disorders. J. Autism Dev. Disord. 2004, 34, 731–734. [Google Scholar] [CrossRef] [PubMed]
- Werner, E.; Dawson, G. Validation of the phenomenon of autistic regression using home videotapes. Arch. Gen. Psychiatry 2005, 62, 889–895. [Google Scholar] [CrossRef]
- Parent Ratings of Behavioral Effects of Biomedical Interventions, Publication 34, Autism Research Institute. Available online: https://www.autism.com/pdf/providers/ParentRatings 2009.pdf (accessed on 28 November 2018).
- Belardo, A.; Gevi, F.; Zolla, L. The concomitant lower concentrations of vitamins B6, B9 and B12 may cause methylation deficiency in autistic children. J. Nutr. Biochem. 2019, 70, 38–46. [Google Scholar] [CrossRef] [PubMed]
- Cannell, J.J. Autism and vitamin D. Med. Hypotheses 2008, 70, 750–759. [Google Scholar] [CrossRef] [PubMed]
- Bittker, S.S.; Bell, K.R. Acetaminophen, antibiotics, ear infection, breastfeeding, vitamin D drops, and autism: An epidemiological study. Neuropsychiatr. Dis. Treat. 2018, 14, 1399–1414. [Google Scholar] [CrossRef]
- Schultz, S.T.; Klonoff-Cohen, H.S.; Wingard, D.L.; Akshoomoff, N.A.; Macera, C.A.; Ji, M.; Bacher, C. Breastfeeding, infant formula supplementation, and Autistic Disorder: The results of a parent survey. Int. Breastfeed. J. 2006, 1, 16. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Schultz, S.T.; Klonoff-Cohen, H.S.; Wingard, D.L.; Akshoomoff, N.A.; Macera, C.A.; Ji, M. Acetaminophen (paracetamol) use, measles-mumps-rubella vaccination, and autistic disorder: The results of a parent survey. Autism 2008, 12, 293–307. [Google Scholar] [CrossRef] [PubMed]
- FAO (Food and Agricultural Organization of the United Nations)/WHO (World Health Organization). Joint Expert Consultation Fats and Oils in Human Nutrition; FAO Food and Nutrition Paper No 57; Food and Agricultural Organization of the United Nations: Rome, Italy, 1994; pp. 49–55. [Google Scholar]
- Berger, A.; Crozier, G.; Bisogno, T.; Cavaliere, P.; Innis, S.; Di Marzo, V. Anandamide and diet: Inclusion of dietary arachidonate and docosahexaenoate leads to increased brain levels of the corresponding N-acylethanolamines in piglets. Proc. Natl. Acad. Sci. USA 2001, 98, 6402–6406. [Google Scholar] [CrossRef] [PubMed]
- Pertwee, R.G. Endocannabinoids and their pharmacological actions. Handb. Exp. Pharmacol. 2015, 231, 1–37. [Google Scholar] [PubMed]
- Keimpema, E.; Barabas, K.; Morozov, Y.M.; Tortoriello, G.; Torii, M.; Cameron, G.; Yanagawa, Y.; Watanabe, M.; Mackie, K.; Harkany, T. Differential subcellular recruitment of monoacylglycerol lipase generates spatial specificity of 2-arachidonoyl glycerol signaling during axonal pathfinding. J. Neurosci. 2010, 30, 13992–14007. [Google Scholar] [CrossRef]
- Carrier, E.J.; Patel, S.; Hillard, C.J. Endocannabinoids in neuroimmunology and stress. Curr. Drug Targets CNS Neurol. Disord. 2005, 4, 657–665. [Google Scholar] [CrossRef]
- Antonucci, N.; Cirillo, A.; Siniscalco, D. Beneficial Effects of Palmitoylethanolamide on Expressive Language, Cognition, and Behaviors in Autism: A Report of Two Cases. Case Rep. Psychiatry 2015, 2015, 325061. [Google Scholar] [CrossRef]
- Siniscalco, D.; Sapone, A.; Giordano, C.; Cirillo, A.; de Magistris, L.; Rossi, F.; Fasano, A.; Bradstreet, J.J.; Maione, S.; Antonucci, N. Cannabinoid receptor type 2, but not type 1, is up-regulated in peripheral blood mononuclear cells of children affected by autistic disorders. J. Autism Dev. Disord. 2013, 43, 2686–2695. [Google Scholar] [CrossRef]
- Högestätt, E.D.; Jönsson, B.A.; Ermund, A.; Andersson, D.A.; Björk, H.; Alexander, J.P.; Cravatt, B.F.; Basbaum, A.I.; Zygmunt, P.M. Conversion of acetaminophen to the bioactive N-acylphenolamine AM404 via fatty acid amide hydrolase-dependent arachidonic acid conjugation in the nervous system. J. Biol. Chem. 2005, 280, 31405–31412. [Google Scholar] [CrossRef]
- Schultz, S.T.; Gould, G.G. Acetaminophen Use for Fever in Children Associated with Autism Spectrum Disorder. Autism Open Access. 2016, 6, 170. [Google Scholar] [CrossRef]
- Karhson, D.S.; Krasinska, K.M.; Dallaire, J.A.; Libove, R.A.; Phillips, J.M.; Chien, A.S.; Garner, J.P.; Hardan, A.Y.; Parker, K.J. Plasma anandamide concentrations are lower in children with autism spectrum disorder. Mol. Autism 2018, 9, 18. [Google Scholar] [CrossRef]
- Zhang, M.; Han, W.; Hu, S.; Xu, H. Methylcobalamin: A potential vitamin of pain killer. Neural Plast. 2013, 2013, 424651. [Google Scholar] [CrossRef]
- Gruber, K.; Puffer, B.; Kräutler, B. Vitamin B12-derivatives-enzyme cofactors and ligands of proteins and nucleic acids. Chem. Soc. Rev. 2011, 40, 4346–4363. [Google Scholar] [CrossRef]
- Hendren, R.L. Autism: Biomedical complementary treatment approaches. Child Adolesc. Psychiatr. Clin. N. Am. 2013, 22, 443–456. [Google Scholar] [CrossRef]
- James, S.J.; Cutler, P.; Melnyk, S.; Jernigan, S.; Janak, L.; Gaylor, D.W.; Neubrander, J.A. Metabolic biomarkers of increased oxidative stress and impaired methylation capacity in children with autism. Am. J. Clin. Nutr. 2004, 80, 1611–1617. [Google Scholar] [CrossRef]
- Bertoglio, K.; Jill James, S.; Deprey, L.; Brule, N.; Hendren, R.L. Pilot study of the effect of methyl B12 treatment on behavioral and biomarker measures in children with autism. J. Altern. Complement. Med. 2010, 16, 555–560. [Google Scholar] [CrossRef]
- Frye, R.E.; Melnyk, S.; Fuchs, G.; Reid, T.; Jernigan, S.; Pavliv, O.; Hubanks, A.; Gaylor, D.W.; Walters, L.; James, S.J. Effectiveness of methylcobalamin and folinic Acid treatment on adaptive behavior in children with autistic disorder is related to glutathione redox status. Autism Res. Treat. 2013, 2013, 609705. [Google Scholar] [CrossRef]
- James, S.J.; Melnyk, S.; Fuchs, G.; Reid, T.; Jernigan, S.; Pavliv, O.; Hubanks, A.; Gaylor, D.W. Efficacy of methylcobalamin and folinic acid treatment on glutathione redox status in children with autism. Am. J. Clin. Nutr. 2009, 89, 425–430. [Google Scholar] [CrossRef]
- Hole, M.; Jorge-Finnigan, A.; Underhaug, J.; Teigen, K.; Martinez, A. Pharmacological Chaperones that Protect Tetrahydrobiopterin Dependent Aromatic Amino Acid Hydroxylases Through Different Mechanisms. Curr. Drug Targets 2016, 17, 1515–1526. [Google Scholar] [CrossRef]
- Kapatos, G. The neurobiology of tetrahydrobiopterin biosynthesis: A model for regulation of GTP cyclohydrolase I gene transcription within nigrostriatal dopamine neurons. IUBMB Life 2013, 65, 323–333. [Google Scholar] [CrossRef]
- Tani, Y.; Fernell, E.; Watanabe, Y.; Kanai, T.; Långström, B. Decrease in 6R-5,6,7,8-tetrahydrobiopterin content in cerebrospinal fluid of autistic patients. Neurosci. Lett. 1994, 181, 169–172. [Google Scholar] [CrossRef]
- Thöny, B.; Auerbach, G.; Blau, N. Tetrahydrobiopterin biosynthesis, regeneration and functions. Biochem. J. 2000, 347 Pt 1, 1–16. [Google Scholar] [CrossRef]
- Komori, H.; Matsuishi, T.; Yamada, S.; Yamashita, Y.; Ohtaki, E.; Kato, H. Cerebrospinal fluid biopterin and biogenic amine metabolites during oral R-THBP therapy for infantile autism. J. Autism Dev. Disord. 1995, 25, 183–193. [Google Scholar] [CrossRef]
- Fernell, E.; Watanabe, Y.; Adolfsson, I.; Tani, Y.; Bergström, M.; Hartvig, P.; Lilja, A.; von Knorring, A.L.; Gillberg, C.; Långström, B. Possible effects of tetrahydrobiopterin treatment in six children with autism—Clinical and positron emission tomography data: A pilot study. Dev. Med. Child Neurol. 1997, 39, 313–318. [Google Scholar] [CrossRef]
- Danfors, T.; von Knorring, A.L.; Hartvig, P.; Langstrom, B.; Moulder, R.; Stromberg, B.; Torstenson, R.; Wester, U.; Watanabe, Y.; Eeg-Olofsson, O. Tetrahydrobiopterin in the treatment of children with autistic disorder: A double-blind placebo-controlled crossover study. J. Clin. Psychopharmacol. 2005, 25, 485–489. [Google Scholar] [CrossRef]
- Frye, R.E.; Huffman, L.C.; Elliott, G.R. Tetrahydrobiopterin as a novel therapeutic intervention for autism. Neurotherapeutics 2010, 7, 241–249. [Google Scholar] [CrossRef]
- Frye, R.E.; DeLatorre, R.; Taylor, H.B.; Slattery, J.; Melnyk, S.; Chowdhury, N.; James, S.J. Metabolic effects of sapropterin treatment in autism spectrum disorder: A preliminary study. Transl. Psychiatry 2013, 3, e237. [Google Scholar] [CrossRef]
- Fluegge, K. A reply to ‘Metabolic effects of sapropterin treatment in autism spectrum disorder: A preliminary study’. Transl. Psychiatry 2016, 6, e793. [Google Scholar] [CrossRef]
- Klaiman, C.; Huffman, L.; Masaki, L.; Elliott, G.R. Tetrahydrobiopterin as a treatment for autism spectrum disorders: A double-blind, placebo-controlled trial. J. Child Adolesc. Psychopharmacol. 2013, 23, 320–328. [Google Scholar] [CrossRef]
- Delhey, L.M.; Tippett, M.; Rose, S.; Bennuri, S.C.; Slattery, J.C.; Melnyk, S.; James, S.J.; Frye, R.E. Comparison of Treatment for Metabolic Disorders Associated with Autism: Reanalysis of Three Clinical Trials. Front. Neurosci. 2018, 12, 19. [Google Scholar] [CrossRef]
- Levine, S.Z.; Kodesh, A.; Viktorin, A.; Smith, L.; Uher, R.; Reichenberg, A.; Sandin, S. Association of Maternal Use of Folic Acid and Multivitamin Supplements in the Periods Before and During Pregnancy with the Risk of Autism Spectrum Disorder in Offspring. JAMA Psychiatry 2018, 75, 176–184. [Google Scholar] [CrossRef]
- Frye, R.E.; Sequeira, J.M.; Quadros, E.V.; James, S.J.; Rossignol, D.A. Cerebral folate receptor autoantibodies in autism spectrum disorder. Mol. Psychiatry 2013, 18, 369–381. [Google Scholar] [CrossRef]
- Sequeira, J.M.; Ramaekers, V.T.; Quadros, E.V. The diagnostic utility of folate receptor autoantibodies in blood. Clin. Chem. Lab. Med. 2013, 51, 545–554. [Google Scholar] [CrossRef]
- Ramaekers, V.; Sequeira, J.M.; Quadros, E.V. Clinical recognition and aspects of the cerebral folate deficiency syndromes. Clin. Chem. Lab. Med. 2013, 51, 497–511. [Google Scholar] [CrossRef]
- Ramaekers, V.T.; Sequeira, J.M.; Quadros, E.V. The basis for folinic acid treatment in neuro-psychiatric disorders. Biochimie 2016, 126, 79–90. [Google Scholar] [CrossRef]
- Desai, A.; Sequeira, J.M.; Quadros, E.V. Prevention of behavioral deficits in rats exposed to folate receptor antibodies: Implication in autism. Mol. Psychiatry 2017, 22, 1291–1297. [Google Scholar] [CrossRef]
- Frye, R.E.; Slattery, J.C.; Quadros, E.V. Folate metabolism abnormalities in autism: Potential biomarkers. Biomark. Med. 2017. [Google Scholar] [CrossRef]
- Frye, R.E.; Slattery, J.; Delhey, L.; Furgerson, B.; Strickland, T.; Tippett, M.; Sailey, A.; Wynne, R.; Rose, S.; Melnyk, S.; et al. Folinic acid improves verbal communication in children with autism and language impairment: A randomized double-blind placebo-controlled trial. Mol. Psychiatry 2018, 23, 247–256. [Google Scholar] [CrossRef]
- Daniels, J.L.; Longnecker, M.P.; Rowland, A.S.; Golding, J.; ALSPAC Study Team. University of Bristol Institute of Child Health. Fish intake during pregnancy and early cognitive development of offspring. Epidemiology 2004, 15, 394–402. [Google Scholar] [CrossRef]
- Hibbeln, J.R.; Davis, J.M.; Steer, C.; Emmett, P.; Rogers, I.; Williams, C.; Golding, J. Maternal seafood consumption in pregnancy and neurodevelopmental outcomes in childhood (ALSPAC study): An observational cohort study. Lancet 2007, 369, 578–585. [Google Scholar] [CrossRef]
- Oken, E.; Radesky, J.S.; Wright, R.O.; Bellinger, D.C.; Amarasiriwardena, C.J.; Kleinman, K.P.; Hu, H.; Gillman, M.W. Maternal fish intake during pregnancy, blood mercury levels, and child cognition at age 3 years in a US cohort. Am. J. Epidemiol. 2008, 167, 1171–1181. [Google Scholar] [CrossRef]
- Posar, A.; Visconti, P. Omega-3 supplementation in autism spectrum disorders: A still open question? J. Pediatr. Neurosci. 2016, 11, 225–227. [Google Scholar] [CrossRef]
- Yadav, S.; Tiwari, V.; Singh, M.; Yadav, R.K.; Roy, S.; Devi, U.; Gautam, S.; Rawat, J.K.; Ansari, M.N.; Saeedan, A.S.; et al. Comparative efficacy of alpha-linolenic acid and gamma-linolenic acid to attenuate valproic acid-induced autism-like features. J. Physiol. Biochem. 2017, 73, 187–198. [Google Scholar] [CrossRef]
- Madore, C.; Leyrolle, Q.; Lacabanne, C.; Benmamar-Badel, A.; Joffre, C.; Nadjar, A.; Layé, S. Neuroinflammation in Autism: Plausible Role of Maternal Inflammation, Dietary Omega 3, and Microbiota. Neural Plast. 2016, 2016, 3597209. [Google Scholar] [CrossRef]
- Mazahery, H.; Stonehouse, W.; Delshad, M.; Kruger, M.C.; Conlon, C.A.; Beck, K.L.; von Hurst, P.R. Relationship between Long Chain n-3 Polyunsaturated Fatty Acids and Autism Spectrum Disorder: Systematic Review and Meta-Analysis of Case-Control and Randomised Controlled Trials. Nutrients 2017, 9, 155. [Google Scholar] [CrossRef] [PubMed]
- Parellada, M.; Llorente, C.; Calvo, R.; Gutierrez, S.; Lázaro, L.; Graell, M.; Guisasola, M.; Dorado, M.L.; Boada, L.; Romo, J.; et al. Randomized trial of omega-3 for autism spectrum disorders: Effect on cell membrane composition and behavior. Eur. Neuropsychopharmacol. 2017, 27, 1319–1330. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.S.; Tseng, P.T.; Chen, Y.W.; Stubbs, B.; Yang, W.C.; Chen, T.Y.; Wu, C.K.; Lin, P.Y. Supplementation of omega 3 fatty acids may improve hyperactivity, lethargy, and stereotypy in children with autism spectrum disorders: A meta-analysis of randomized controlled trials. Neuropsychiatr. Dis. Treat. 2017, 13, 2531–2543. [Google Scholar] [CrossRef] [PubMed]
- Sheppard, K.W.; Boone, K.M.; Gracious, B.; Klebanoff, M.A.; Rogers, L.K.; Rausch, J.; Bartlett, C.; Coury, D.L.; Keim, S.A. Effect of Omega-3 and -6 Supplementation on Language in Preterm Toddlers Exhibiting Autism Spectrum Disorder Symptoms. J. Autism Dev. Disord. 2017, 47, 3358–3369. [Google Scholar] [CrossRef]
- Basil, P.; Li, Q.; Gui, H.; Hui, T.C.K.; Ling, V.H.M.; Wong, C.C.Y.; Mill, J.; McAlonan, G.M.; Sham, P.C. Prenatal immune activation alters the adult neural epigenome but can be partly stabilised by a n-3 polyunsaturated fatty acid diet. Transl. Psychiatry. 2018, 8, 125. [Google Scholar] [CrossRef] [PubMed]
- Mazahery, H.; Conlon, C.A.; Beck, K.L.; Mugridge, O.; Kruger, M.C.; Stonehouse, W.; Camargo, C.A., Jr.; Meyer, B.J.; Tsang, B.; Jones, B.; et al. A Randomised-Controlled Trial of Vitamin D and Omega-3 Long Chain Polyunsaturated Fatty Acids in the Treatment of Core Symptoms of Autism Spectrum Disorder in Children. J. Autism Dev. Disord. 2019. [Google Scholar] [CrossRef]
- Amos, D.; Cook, C.; Santanam, N. Omega 3 rich diet modulates energy metabolism via GPR120-Nrf2 crosstalk in a novel antioxidant mouse model. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2019, 1864, 466–488. [Google Scholar] [CrossRef] [PubMed]
- Theoharides, T.C.; Asadi, S.; Panagiotidou, S. A case series of a luteolin formulation (NeuroProtek®) in children with autism spectrum disorders. Int. J. Immunopathol. Pharmacol. 2012, 25, 317–323. [Google Scholar] [CrossRef]
- Yang, Y.; Bai, L.; Li, X.; Xiong, J.; Xu, P.; Guo, C.; Xue, M. Transport of active flavonoids, based on cytotoxicity and lipophilicity: An evaluation using the blood-brain barrier cell and Caco-2 cell models. Toxicol. In Vitro 2014, 28, 388–396. [Google Scholar] [CrossRef] [PubMed]
- Harwood, M.; Nielewska-Nikiel, B.; Borzelleca, J.F.; Flamm, G.W.; Williams, G.M.; Lines, T.C. A critical review of the data related to the safety of quercetin and lack of evidence of in vivo toxicity, including lack of genotoxic/carcinogenic properties. Food Chem. Toxicol. 2007, 45, 2179–2205. [Google Scholar] [CrossRef] [PubMed]
- Ericson-Neilsen, W.; Kaye, A.D. Steroids: Pharmacology, complications, and practice delivery issues. Ochsner J. 2014, 14, 203–207. [Google Scholar] [PubMed]
- Mordekar, S.R.; Prendergast, M.; Chattopadhyay, A.K.; Baxter, P.S. Corticosteroid treatment of behaviour, language and motor regression in childhood disintegrative disorder. Eur. J. Paediatr. Neurol. 2009, 13, 367–369. [Google Scholar] [CrossRef] [PubMed]
- Stefanatos, G.A.; Grover, W.; Geller, E. Case study: Corticosteroid treatment of language regression in pervasive developmental disorder. J. Am. Acad. Child Adolesc. Psychiatry 1995, 34, 1107–1111. [Google Scholar] [CrossRef] [PubMed]
- Chugani, D.C. Role of altered brain serotonin mechanisms in autism. Mol. Psychiatry 2002, 7 (Suppl. 2), S16–S17. [Google Scholar] [CrossRef] [PubMed]
- Hertzman, M. Galantamine in the Treatment of Adult Autism: A Report of Three Clinical Cases. Int. J. Psychiatry Med. 2003, 33, 395–398. [Google Scholar] [CrossRef] [PubMed]
- Prvulovic, D.; Hampel, H.; Pantel, J. Galantamine for Alzheimer’s disease. Expert Opin. Drug Metab. Toxicol. 2010, 6, 345–354. [Google Scholar] [CrossRef] [PubMed]
- Rossignol, D.A.; Frye, R.E. The use of medications approved for Alzheimer’s disease in autism spectrum disorder: A systematic review. Front. Pediatr. 2014, 2, 87. [Google Scholar] [CrossRef] [PubMed]
- Atzori, M.; Cuevas-Olguin, R.; Esquivel-Rendon, E.; Garcia-Oscos, F.; Salgado-Delgado, R.C.; Saderi, N.; Miranda-Morales, M.; Treviño, M.; Pineda, J.C.; Salgado, H. Locus Ceruleus Norepinephrine Release: A Central Regulator of CNS Spatio-Temporal Activation? Front. Synaptic Neurosci. 2016, 8, 25. [Google Scholar] [CrossRef] [PubMed]
- Harley, C.W. A role for norepinephrine in arousal, emotion and learning?: Limbic modulation by norepinephrine and the Kety hypothesis. Prog. Neuropsychopharmacol. Biol. Psychiatry 1987, 11, 419–458. [Google Scholar] [CrossRef]
- Berridge, C.W.; Waterhouse, B.D. The locus coeruleus-noradrenergic system: Modulation of behavioral state and state-dependent cognitive processes. Brain Res. Rev. 2003, 42, 33–84. [Google Scholar] [CrossRef]
- Lake, C.R.; Ziegler, M.G.; Murphy, D.L. Increased norepinephrine levels and decreased dopamine-beta-hydroxylase activity in primary autism. Arch. Gen. Psychiatry 1977, 34, 553–556. [Google Scholar] [CrossRef] [PubMed]
- Launay, J.M.; Bursztejn, C.; Ferrari, P.; Dreux, C.; Braconnier, A.; Zarifian, E.; Lancrenon, S.; Fermanian, J. Catecholamines metabolism in infantile autism: A controlled study of 22 autistic children. J. Autism Dev. Disord. 1987, 17, 333–347. [Google Scholar] [CrossRef] [PubMed]
- Beversdorf, D.Q.; Hughes, J.D.; Steinberg, B.A.; Lewis, L.D.; Heilman, K.M. Noradrenergic modulation of cognitive flexibility in problem solving. Neuroreport 1999, 10, 2763–2767. [Google Scholar] [CrossRef] [PubMed]
- Minshew, N.J.; Goldstein, G. The pattern of intact and impaired memory functions in autism. J. Child Psychol. Psychiatry 2001, 42, 1095–1101. [Google Scholar] [CrossRef] [PubMed]
- Mehler, M.F.; Purpura, D.P. Autism, fever, epigenetics and the locus coeruleus. Brain Res. Rev. 2009, 59, 388–392. [Google Scholar] [CrossRef] [PubMed]
- Steenen, S.A.; van Wijk, A.J.; van der Heijden, G.J.; van Westrhenen, R.; de Lange, J.; Jongh, A. Propranolol for the treatment of anxiety disorders: Systematic review and meta-analysis. J. Psychopharmacol. 2016, 30, 128–139. [Google Scholar] [CrossRef] [PubMed]
- Beversdorf, D.Q.; Carpenter, A.L.; Miller, R.F.; Cios, J.S.; Hillier, A. Effect of propranolol on verbal problem solving in autism spectrum disorder. Neurocase 2008, 14, 378–383. [Google Scholar] [CrossRef]
- Zamzow, R.M.; Ferguson, J.B.; Ragsdale, A.S.; Lewis, M.L.; Beversdorf, D.Q. Effects of acute beta-adrenergic antagonism on verbal problem solving in autism spectrum disorder and exploration of treatment response markers. J. Clin. Exp. Neuropsychol. 2017, 39, 596–606. [Google Scholar] [CrossRef]
- Ratey, J.J.; Bemporad, J.; Sorgi, P.; Bick, P.; Polakoff, S.; O’Driscoll, G.; Mikkelsen, E. Brief Report: Open Trial Effects of Beta Blockers on Speech and Social Behaviors in 8 Autistic Adults. J. Autism Dev. Disord. 1987, 17, 439–446. [Google Scholar] [CrossRef]
- Zamzow, R.M.; Ferguson, B.J.; Stichter, J.P.; Porges, E.C.; Ragsdale, A.S.; Lewis, M.L.; Beversdorf, D.Q. Effects of propranolol on conversational reciprocity in autism spectrum disorder: A pilot, double-blind, single-dose. Psychopharmacology (Berl.) 2016, 233, 1171–1178. [Google Scholar] [CrossRef]
- Schultz, S.T. Can autism be triggered by acetaminophen activation of the endocannabinoid system? Acta Neurobiol. Exp. 2010, 70, 227–231. [Google Scholar]
- Aran, A.; Eylon, M.; Harel, M.; Polianski, L.; Nemirovski, A.; Tepper, A.; Schnapp, A.; Cassuto, H.; Wattad, N.; Tam, J. Lower circulating endocannabinoid levels in children with autism spectrum disorder. Mol. Autism 2019, 10, 2. [Google Scholar] [CrossRef] [PubMed]
- Leweke, F.M.; Piomelli, D.; Pahlisch, F.; Muhl, D.; Gerth, C.W.; Hoyer, C.; Klosterkötter, J.; Hellmich, M.; Koethe, D. Cannabidiol enhances anandamide signaling and alleviates psychotic symptoms of schizophrenia. Transl. Psychiatry 2012, 2, e94. [Google Scholar] [CrossRef] [PubMed]
- Bar-Lev Schleider, L.; Mechoulam, R.; Saban, N.; Meiri, G.; Novack, V. Real life Experience of Medical Cannabis Treatment in Autism: Analysis of Safety and Efficacy. Sci. Rep. 2019, 9, 200. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Castillo, M.A.; Urdaneta, K.E.; Semprún-Hernández, N.; Brigida, A.L.; Antonucci, N.; Schultz, S.; Siniscalco, D. Speech-Stimulating Substances in Autism Spectrum Disorders. Behav. Sci. 2019, 9, 60. https://doi.org/10.3390/bs9060060
Castillo MA, Urdaneta KE, Semprún-Hernández N, Brigida AL, Antonucci N, Schultz S, Siniscalco D. Speech-Stimulating Substances in Autism Spectrum Disorders. Behavioral Sciences. 2019; 9(6):60. https://doi.org/10.3390/bs9060060
Chicago/Turabian StyleCastillo, María Andrea, Kendy Eduardo Urdaneta, Neomar Semprún-Hernández, Anna Lisa Brigida, Nicola Antonucci, Stephen Schultz, and Dario Siniscalco. 2019. "Speech-Stimulating Substances in Autism Spectrum Disorders" Behavioral Sciences 9, no. 6: 60. https://doi.org/10.3390/bs9060060
APA StyleCastillo, M. A., Urdaneta, K. E., Semprún-Hernández, N., Brigida, A. L., Antonucci, N., Schultz, S., & Siniscalco, D. (2019). Speech-Stimulating Substances in Autism Spectrum Disorders. Behavioral Sciences, 9(6), 60. https://doi.org/10.3390/bs9060060






